Abstract
Red mud (RM) is one kind of basic solid waste produced from aluminum industry. RM is usually stored by the method of land stockpiling, which has caused serious environmental and safety issues. Bittern, a by-product of salt manufacture, is abundant in China. But the utilization rate is low, less than 20%. The disposal of waste bittern has become a challenge that attracts much attention in water industry. In this article, Mg/Al/Fe layered double hydroxides (Mg/Al/Fe-LDHs) were synthesized using the above two wastes by a coprecipitation method and characterized by X-ray diffraction (XRD) and scanning electron microscopy. The XRD results showed that LDHs were successfully synthesized. Then, the flame-retardant properties and thermal properties of Mg/Al/Fe-LDHs in ethylene vinyl acetate (EVA)/LDHs composites had been tested by cone calorimeter test (CCT), limiting oxygen index (LOI), smoke density test (SDT), and thermogravimetry–Fourier transform infrared spectrometry (TG-FTIR). The CCT results showed that the heat release rate (HRR) of the EVA/LDHs composites significantly decreased in comparison with that of pure EVA, and EVA2 sample showed the lowest peak value of HRR value of 204.59 kW m−2. The LOI results showed that EVA3 had a highest LOI value of 28.3% in all the samples. The SDT test indicated that Mg/Al/Fe-LDHs were beneficial to the smoke suppression performance of EVA composites. TG-IR results showed that EVA/LDHs composites had better thermal stability than EVA.
Introduction
Seawater bittern or called brine is a byproduct of salt manufacture, which is generally regarded as harmful waste. 1 As the world’s largest producer of sea salt, China produces bittern close to 2 × 107 m3 every year. Although the bittern resources are very rich in China, the utilization rate is low, even less than 20%. The disposal of waste bittern has become a challenge that attracts much attention in water industry. 2 -4 This by-product is rich in magnesium, potassium, bromine, and so on. Its Mg2+ content is about 32 g L−1, which is 27 times as that of seawater, 5 so bittern is considered to be one of the ideal resources to extract magnesium salt. Red mud (RM) is a strongly basic solid residue in aluminum industry. It is estimated that the aluminum industry produces RM more than 6 × 107 tons every year all over the world. 6,7 Most of them are disposed by wet storage technology, causing a lot of environmental and safety problems. The composition of RM is complicated owing to various bauxite ingredients or different production processes. Its general compositions are Fe2O3, Al2O3, SiO2, TiO2, CaO, Na2O, and so on. 8 -10
Layered double hydroxides (LDHs) are called anionic layered clays, and its general formula can be described by [M1−x2+M x 3+(OH)2]A x /n n −·mH2O. The M2+ for divalent metal ions, M3+ for trivalent metal ions, the A n − for interlayer anions. 11 One of the most typical LDHs is Mg6Al2(OH)16CO3·4H2O, whose structure is similar to Mg(OH)2. 12 Owing to their special layered structure, it has been widely used as flame retardant, catalyst, 13,14 medicine, 15,16 absorbent, 17 and so on. When used as flame-retardant materials, it showed many advantages, such as nonhalogenated, nontoxic, and no corrosive gas generation, which is very promising. 12
RM and bittern are two typical wastes, which are abundant in China. Although many methods have been proposed to reutilize them, they have been rarely used as materials to synthesize LDHs, which is a kind of promising flame-retardant fillers. Moreover, the Fe and Al elements contained in RM and the abundant Mg in bittern are suitable to prepare Mg/Al/Fe-LDHs. 18 In this article, Mg/Al/Fe-LDHs were synthesized using the above two wastes by a coprecipitation method and characterized by X-ray diffraction (XRD) and scanning electron microscopy (SEM). Then, the flame-retardant properties and thermal properties of Mg/Al/Fe-LDHs in ethylene vinyl acetate (EVA)/LDHs composites had been tested by cone calorimeter test (CCT), limiting oxygen index (LOI), smoke density test (SDT), and thermogravimetry–Fourier transform infrared spectrometry (TG-FTIR).
Experiment
Materials
EVA18 (containing 18% vinyl acetate) was purchased from Beijing Eastern Petrochemical Co., Ltd (China). RM was supplied by Aluminum Corporation of China Shandong Branch. It contains O (37.12%), Na (7.11%), Al (11.33%), Si (5.16%), S (0.4%), Ca (0.62%), Ti (2.15%), Fe (35.61%), and Co (0.5%). (The result is examined by an INCA Energy X-ray energy spectrometer (EDS, Oxford) after roasted at a temperature of 650°C for 4 h.) Bittern was supplied by Weidong Chemical Saltwork. It contains Mg (3.2%), K(0.43%), Na(3.2%), Fe(0.0012%), Al(0.00051%), Cr(0.00066%), and Zn(0.00020%). The result is examined by inductively coupled plasma atomic emission spectrometry. Other reagents were standard laboratory reagents and used as received without further purification.
Synthesis of LDHs
RM was grounded and filtered using a sieve and was dried at a temperature of 100°C for 4 h, then the above RM was soaked in hydrochloric acid. Bittern was prepared using similar dry and filtration processes. Then, RM and bittern were mixed with different Mg/(Al + Fe) molar ratios of 1.0/1.0–5.0/1.0 (solution A). An aqueous solution with 0.4 mol L−1 Na2CO3 and 1.5 mol L−1 NaOH was also necessary (solution B). Solutions A and B were added at the same speed to a three-necked, round-bottomed flask provided with a mechanical stirrer and a water bath. The stirring speed kept unchanged and the temperature was 80°C. After the reaction was finished, the resulting slurry was filtered, washed thoroughly, and dried at 80°C to obtain Mg/Al/Fe-LDHs. Then, the Mg/Al/Fe-LDHs were ground into small particles (with a particle size of about 48 μm).
The samples were numbered LDH1(Mg/(Al + Fe) molar ratios of 1.0/1.0), LDH2(Mg/(Al + Fe) molar ratios of 2.0/1.0), LDH3(Mg/(Al + Fe) molar ratios of 3.0/1.0), LDH4(Mg/(Al + Fe) molar ratios of 4.0/1.0), and LDH5(Mg/(Al + Fe) molar ratios of 5.0/1.0).
Preparation of the EVA composites
All compositions were melt-compounded with a mixer at about 120°C for 10 min. After mixing, the mixtures were then compression-molded at about 120°C into sheets under a pressure of 10 MPa for 10 min. The sheets were cut into suitably sized specimens for fire testing. For all the samples, the additive level of all samples was 50%, and they were named EVA0 (pure EVA), EVA1 (containing 50% LDH1), EVA2 (containing 50% LDH2), EVA3 (containing 50% LDH3), EVA4 (containing 50% LDH4), EVA5 (containing 50% LDH5), and EVA6 (containing 50% RM).
Measurements
X-Ray diffraction
XRD data were recorded at room temperature on Philips X’Pert Pro Super apparatus (Nicolet Instrument Co., Madison, Wisconsin, USA) using copper Kα radiation with a nickel filter (wavelength =1.5418 Å) at a scan rate of 0.0167° s−1.
Scanning electron microscopy
The SEM studies were performed using a Hitachi X650 scanning electron microscope (Qingdao University of Science and Technology) at an accelerating voltage of 8 kV.
Limiting oxygen index
LOI was measured according to ASTM D 2863. The apparatus used was an HC-2 oxygen index meter (Jiangning Analysis Instrument Company, China). The specimens used for the test were of dimensions 100 × 6.5 × 3 mm3.
Cone calorimeter test
The cone calorimeter (Stanton Redcroft, London, UK) tests were performed according to ISO 5660 standard procedures. Each specimen of dimensions 100 × 100 × 4 mm3 was wrapped in aluminum foil and exposed horizontally to an external heat flux of 50 kW m−2.
Smoke density test
A SDT machine JQMY-2 (Jianqiao Co, China) was used to measure the smoke characteristics according to ISO 5659-2(2006). Each specimen of dimensions 75 × 75 × 2.5 mm3 was wrapped in aluminum foil and exposed horizontally to an external heat flux of 25 kW m−2 with or without the application of a pilot flame.
Thermogravimetry–Fourier transform infrared spectrometry
The TG-FTIR instrument consists of a thermogravimeter (TG2009 F1, Netzsch Instruments, Germany), a FTIR spectrometer (Vertex 70, Bruker Optics, Germany), and a transfer tube with an inner diameter of 1 mm connected to the TG and the infrared cell. The investigation was carried out from 30°C to 900°C at a linear heating rate of 10°C min−1 under the nitrogen flow rate of 3 × 10−5 m3 min−1.
Results and Discussion
XRD characterization of Mg/Al/Fe-LDHs
Figure 1 shows the powder XRD patterns of RM and Mg/Al/Fe-LDHs samples. Typical peaks of LDHs, which are correlated with characteristic planes (003), (006), (009), (015), and (110), were clearly seen for the Mg/Al/Fe-LDHs samples, while similar peaks do not appear in the XRD pattern of RM. The crystal planes of (003), (006), and (009) have good multiple relationship, indicating that the synthesized LDHs were well crystallized with layered structures. 19 -21 It should be noticed that LDH3 and LDH4 show much sharper peaks and lower baselines, which means they are of higher crystallinity than the other samples. Owing to the complicated compositions of RM and bittern, small spikes can also be found in the XRD patterns.
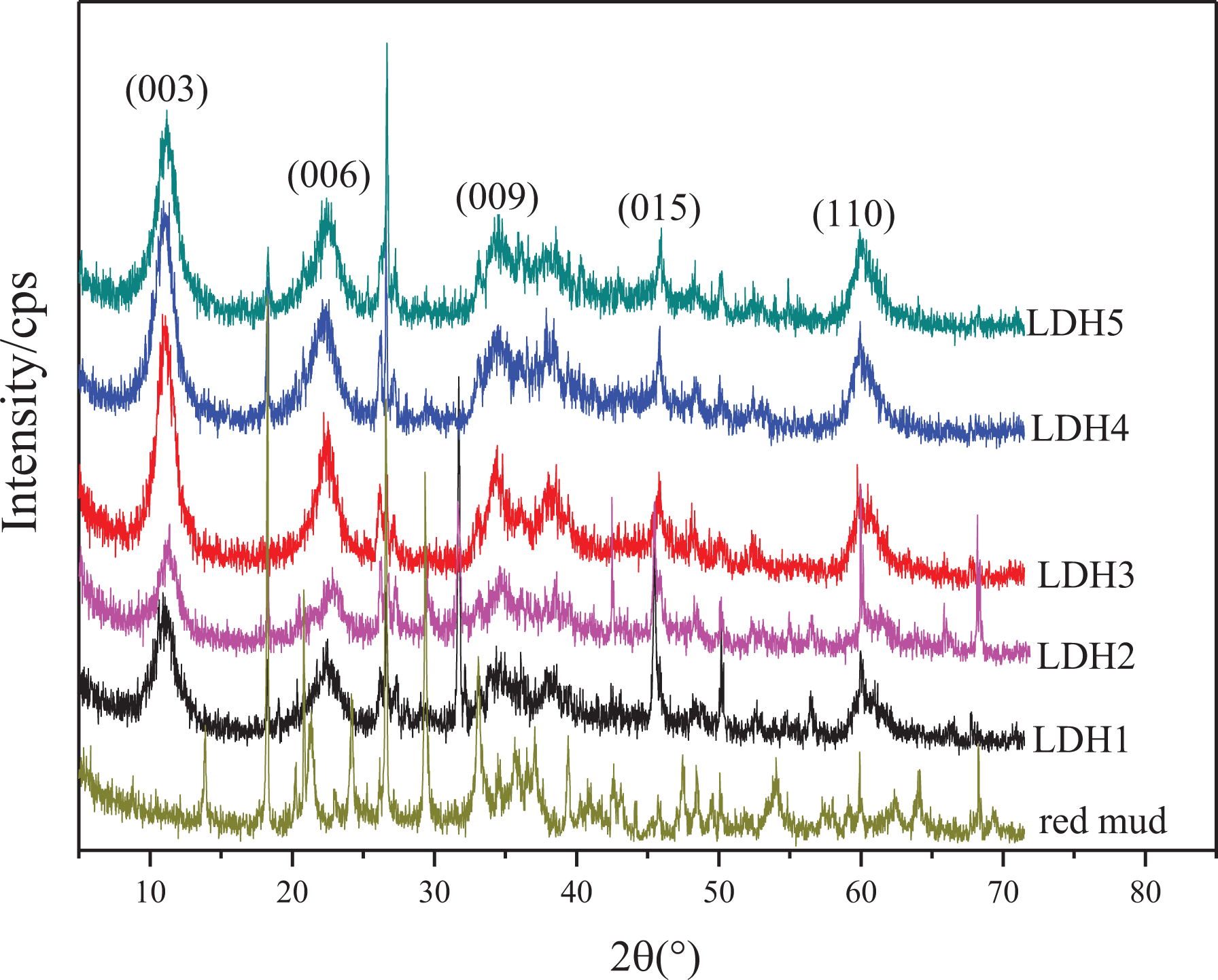
XRD patterns of red mud and Mg/Al/Fe-LDHs samples. XRD: X-ray diffraction; LDH: layered double hydroxide.
SEM of Mg/Al/Fe-LDHs
Figure 2 shows the SEM photographs of RM and Mg/Al/Fe-LDHs powders. RM powder appears serious reunion phenomenon and its particles are in a wide range of sizes. However, the particle size of LDH3 powder is relatively well-distributed, which may be beneficial for its compatible with EVA, improving the flame-retardant and smoke suppression properties of the composites.

SEM photographs of (a) RM and (b) LDH3. SEM: scanning electron microscopy; RM: red mud; LDH: layered double hydroxide.
Flame-retardant properties of EVA/LDHs composites
CCT of EVA/LDHs composites
CCT is widely used for assessing the fire reaction behavior of polymer materials, which is based on the principle of oxygen consumption. It can provide a wealth of information on the combustion behavior, such as heat release rate (HRR), mass loss rate, mass, total heat release (THR), and so on. 22 -24
HRR of EVA/LDHs composites
HRR is an important parameter in CCT and can be used to express the intensity of fire. 25 HRR plots of EVA/LDHs composites are shown in Figure 3 and the correlated data are listed in Table 1. It can be found that the HRR curve of EVA0 rises up quickly and the peak value of HRR (PHRR) reached 1645.88 kW m− 2 . On the other hand, PHRR value of EVA6 (addition of RM) is 396.84 kW m−2, much lower than that of EVA0 and higher than the other EVA/LDHs composites. Among all the EVA/LDH composites, EVA2 exhibited a much smoother HRR curve and showed the lowest PHRR value of 204.59 kW m−2. It should also be noticed that the average HRR (AHRR) and the time to flameout showed the same trend with HRR for EVA0-EVA6. The AHRR value of EVA1–EVA6 is lower than EVA0, and EVA2 has the lowest AHRR value of 106.58 kW m− 2 . Composites containing LDHs need more time to be ignited than the other samples. The combustion process was prolonged to 757 s.
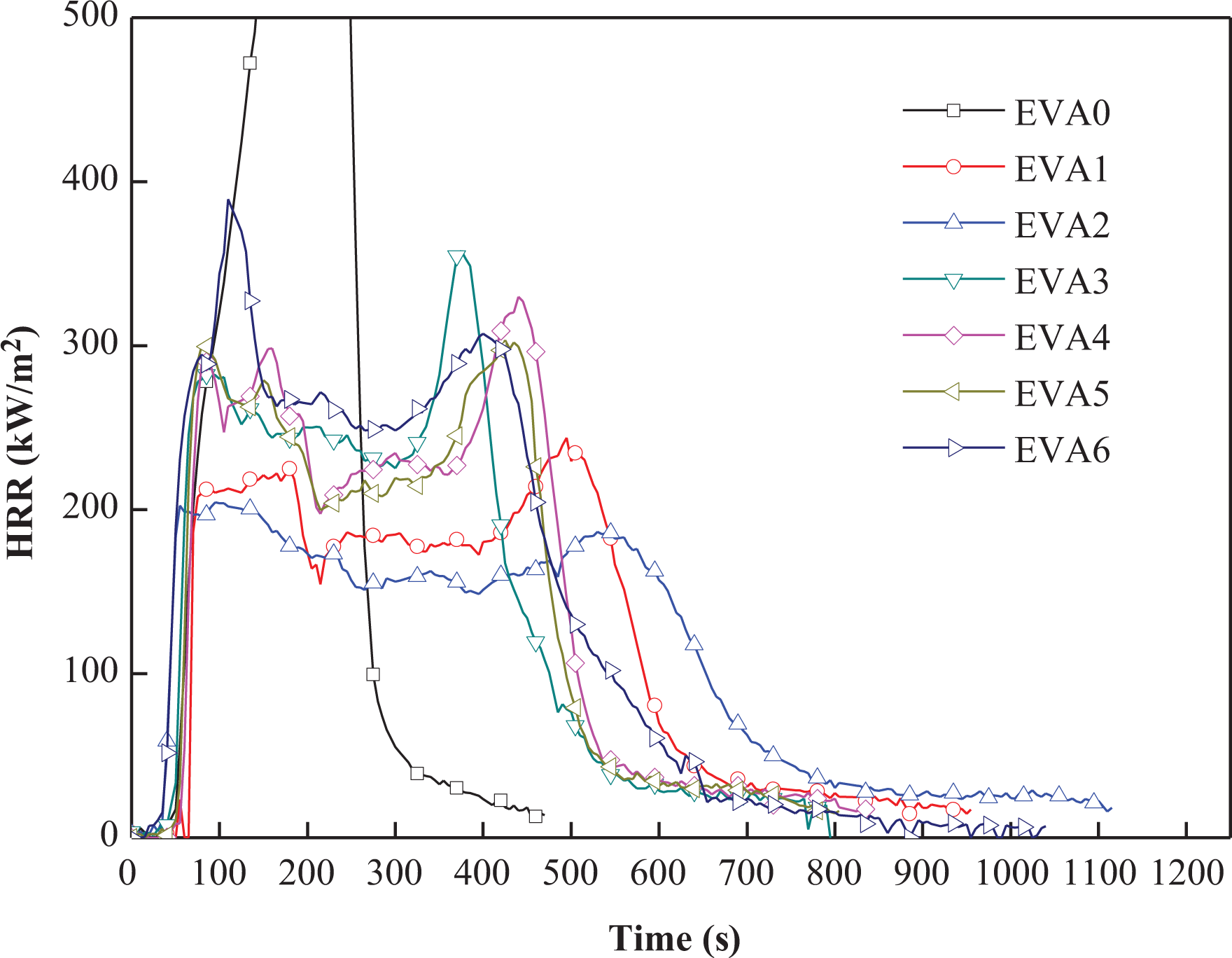
HRR curves of EVA/LDHs composites. HRR: heat release rate; EVA: ethylene vinyl acetate; LDH: layered double hydroxide.
Data from cone calorimeter test.

PHRR: peak value of heat release rate; AHRR: average heat release rate; THR: total heat release; EVA: ethylene vinyl acetate.
HRR results indicated that RM is also helpful for the flame retardant performance of the composites, the reason may be that oxides, such as CaCO3, TiO2, and SiO2, can be used as flame retardants to retard the burning of the composites. 26,27 It is interesting to find that although LDH3 and LDH4 were well crystallized, the flame-retardant properties of related composites were well expected. Unfortunately, the reason for these phenomena is not clear now.
MASS of EVA/LDHs composites
Figure 4 shows the weight variation of the char residues. The curve of EVA0 drops quickly compared with that of EVA1∼EVA6. For EVA1∼EVA5, at the beginning of the combustion, the surfaces of EVA/LDHs composites form a charred layer, which can slow down the combustion process. With the development of combustion process, the charred layer begins to break, resulting in the mass loss of the composites.
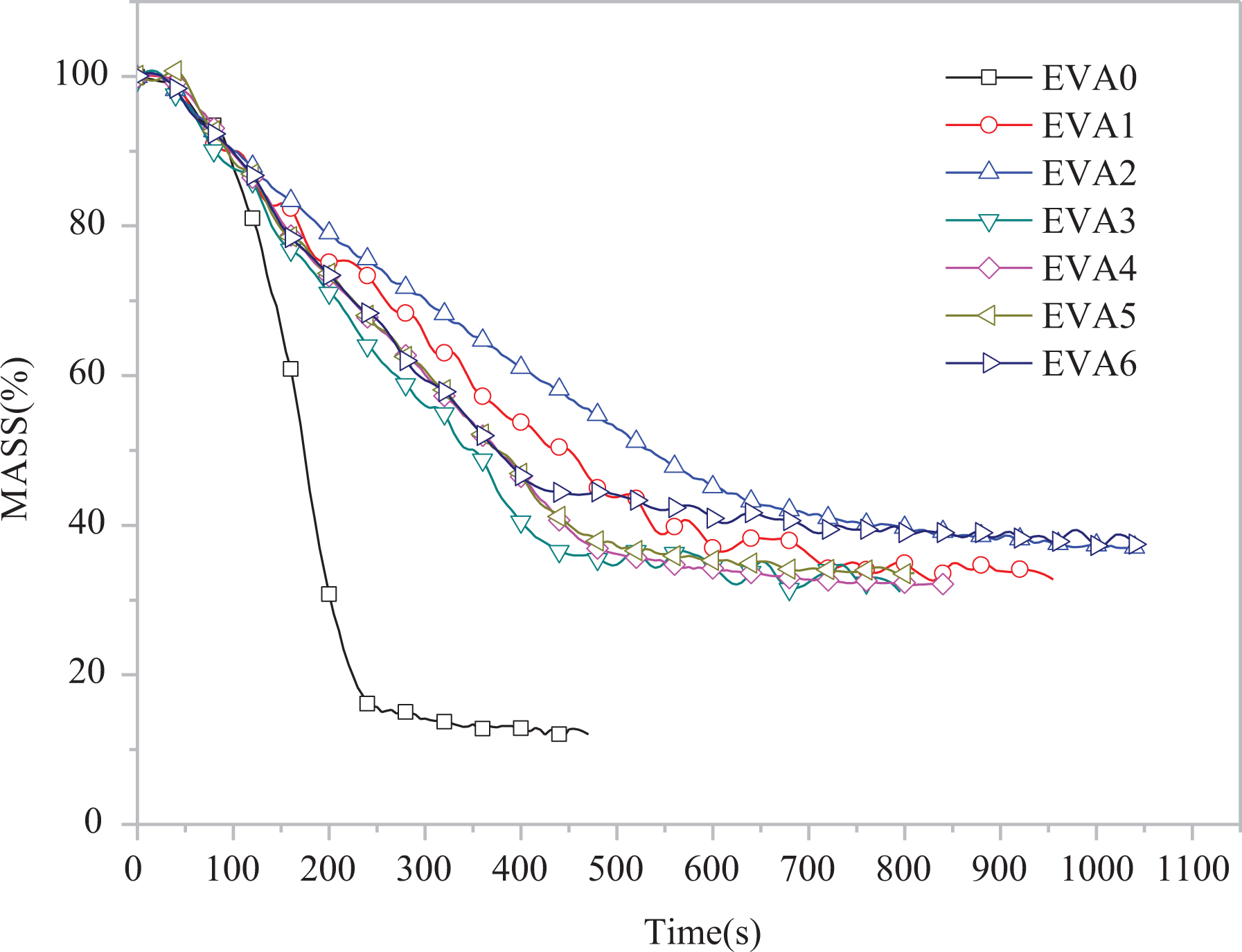
Mass curves of EVA/LDHs composites. EVA: ethylene vinyl acetate; LDH: layered double hydroxide.
THR of EVA/LDHs composites
Figure 5 presents the THR of all the samples. The slope of the THR curve can be assumed as a representative of fire spread. 28 The THR plots are in accordance with the HRR curves. It can be seen that THR is decreased by the addition of LDHs. The THR values are all below 120.26 mJ m−2, in comparison with 145.41 mJ m−2 of EVA0, and EVA1 has the lowest value of 111.72 mJ m−2. During the combustion, the released CO2 can cover the flame and dilute flammable gas. Then, the protective layer may be formed to prevent the fire from spreading, decreasing the THR.
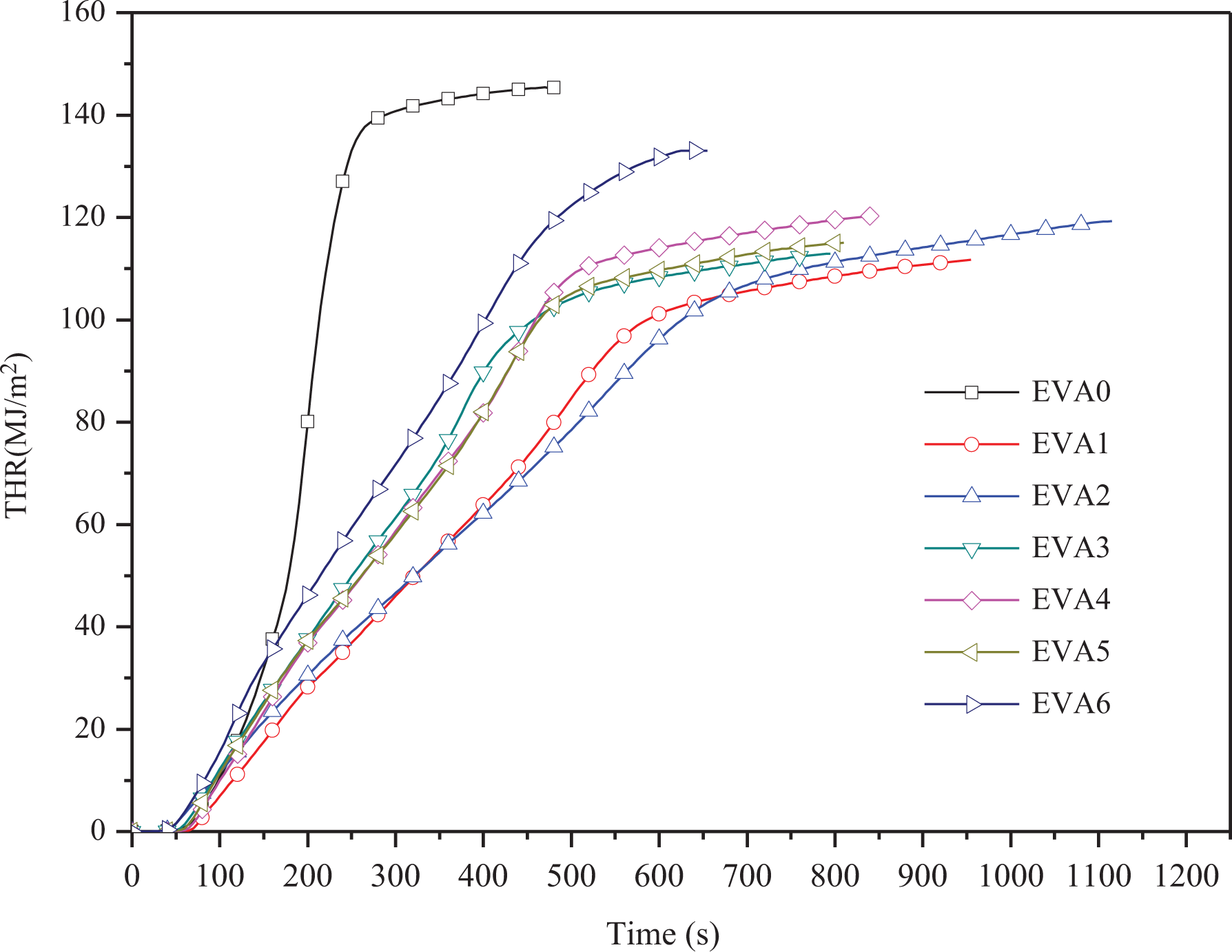
THR curves of EVA/LDHs composites. THR: total heat release; EVA: ethylene vinyl acetate; LDH: layered double hydroxide.
Digital photos of residues
Figure 6 shows the digital photos of the residues of EVA/LDHs composites. It is clear that EVA0 sample leaves no charred residues at the end of combustion. Moreover, the charred residue of EVA2 is compact and integrated, while the charred residues of other samples are loose and incomplete. A better charred layer can protect composites from further burning and retard the pyrolysis of the polymer.

Digital photos after cone calorimeter test.
LOI values of EVA/LDHs composites
The LOI is widely used to evaluate flame retardant properties of materials and to screen flame-retardant formulations. 29 The results are shown in Figure 7, the LOI value of pure EVA is only 19.8%, and this value increased to 21.0% by the addition of RM. It is obvious that the values of EVA1∼EVA5 are higher than that of EVA6. An interesting phenomenon is that the LOI result of EVA2 is not as good as its CCT performance, so the combustion behavior of the composited may rather be different under different combustion conditions.

LOI values of EVA/LDHs composites. LOI: limiting oxygen index; EVA: ethylene vinyl acetate; LDH: layered double hydroxide.
SDT of EVA/LDHs composites
SDT can be used to evaluate the amount of smoke production, which gives detailed information about the smoke production. As is shown in Figure 8, EVA0 and EVA6 produced more smoke than EVA1∼EVA5 before 900 s. The amount of smoke production for EVA1 and EVA2 is less than the other samples. These results are in good agreement with the CCT results. The reason may be that the oxides formed during the combustion process are helpful for improving the polymer melting viscosity and thus formed a compact char which can suppress the production of smoke. Moreover, when a pilot flame is used in the test, the amount of smoke is less than that without a pilot flame. The possible reason may be that the solid particles formed during the depolymerization of the polymers are mainly burned out to form gases, but do not migrate directly into the gas phase. 30

luminous flux curves of EVA/LDHs composites: (a) with the application of a pilot flame and (b) without the application of a pilot flame.
The SDT results indicate that LDHs can suppress smoke production significantly. The good performance of the composites in the SDT test is due to the decomposition of LDHs and the formation of a char layer, which have been mentioned previously.
TG-IR of EVA/LDHs composites
TG analysis
Most polymers will degrade at high temperature via a radical reaction in spite of the inert or air environment. 31 Figures 9 and 10 show the TG and derivative thermogravimetric (DTG) curves of EVA0, EVA1, and EVA2 under the flow of nitrogen atmosphere. The pyrolytic decomposition of EVA is a two-step process, the first decomposition step occurs in the temperature range of 290–390°C (Tmax of 356°C) with a mass loss of 13.5 wt%. It is mainly correlated with the decarboxylation of EVA, generating H2O and CO2. In the temperature range of 390–500°C (Tmax of 471°C), the second decomposition step takes place with only 0.05% residue at 500°C. During this step, the main chain of EVA begins to rupture, producing olefins like butene, ethylene, and so on.
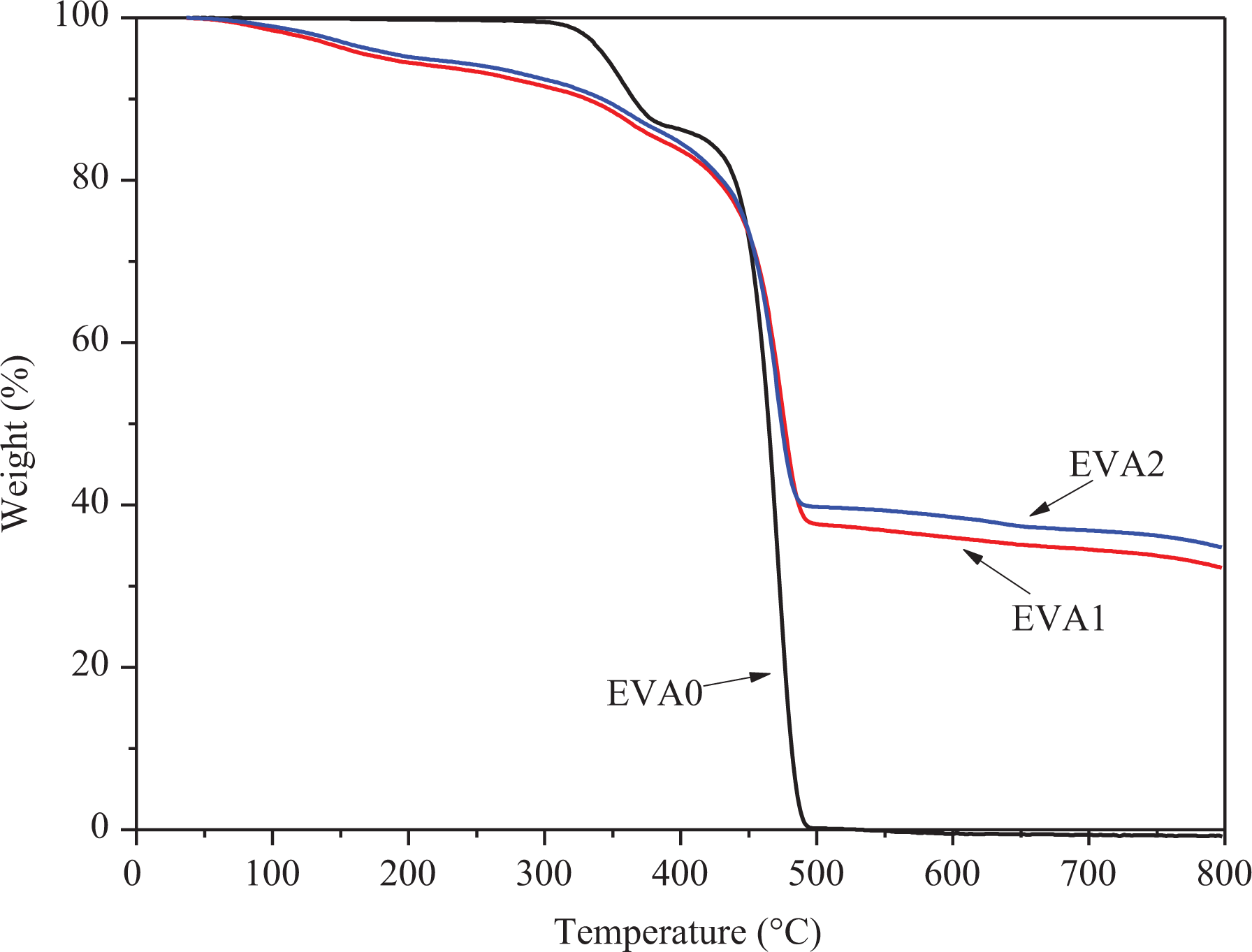
TG curves of EVA/LDHs composites.
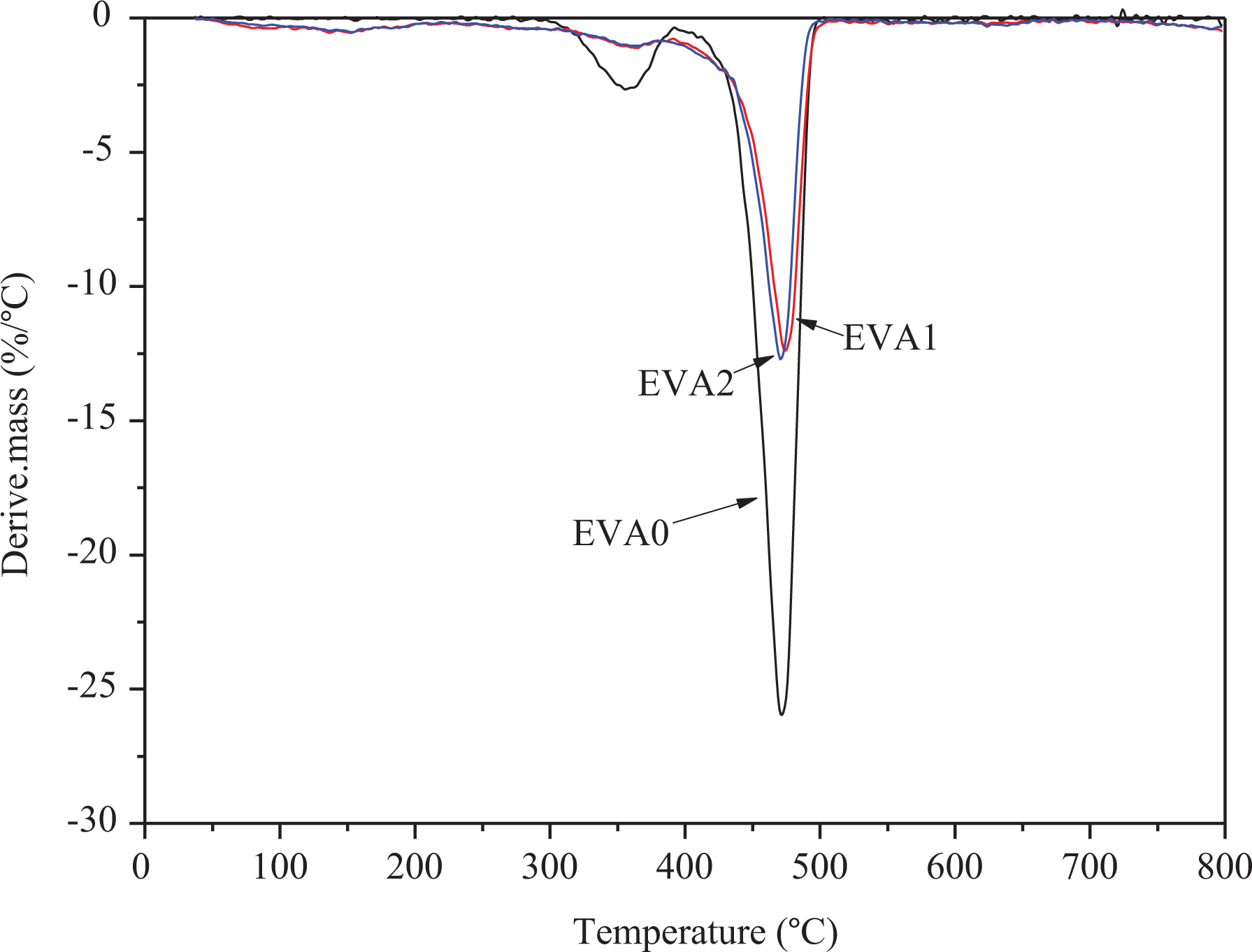
DTG curves of EVA/LDHs composites.
For EVA1 and EVA2, three steps were observed for the pyrolytic decomposition process of the composites. For EVA1, the first step occurs in the range of 100–220°C with a mass change of 6.1% (Tmax of 152°C), which is due to the loss of loosely bound water in the interlayer space of LDHs in the composites. The second step occurs from 220°C to 380°C with a mass change of 9.4% (Tmax of 365°C), which corresponds to the dehydroxylation and decarbonization of LDHs. And the third step occurs after 380°C with a mass change of 52.3% (Tmax of 474°C), which is attributed to the decarboxylation and main chain rupture of EVA. For EVA2, the Tmax of three steps is 152, 362, and 471°C with the mass change of 5.4%, 8.7%, and 51.7%. It is obvious to see that the temperature of pyrolytic decomposition is close to that of EVA1, indicating that two samples have complete structure, which can accommodate more water and anions to keep properties stable.
FTIR characterization of the EVA/LDHs composites
Figure 11 shows the 3D TG-FTIR spectra of pyrolysis products of the pure EVA and EVA/LDHs composites during the thermal degradation. As is shown, the gas products of four samples exhibit characteristic bands at 3400−4000 cm−1, 2800−3150 cm−1, 2250−2400 cm−1, 1700−1850 cm−1, 1250−1500 cm−1, and 950−1150 cm−1, which correspond to H2O (3400−4000 cm−1), CO2 (2300−2400 cm−1), CO (2250−2300 cm−1), carboxylic acid (1700−1850 cm−1), and aliphatic hydrocarbon (2800−3150 cm−1, 1250−1500 cm−1, and 950−1150 cm−1). 27,29,32 It is clear that pure EVA produces more carboxylic acid and aliphatic hydrocarbon, while EVA/LDHs composites produce more CO2 during thermal degradation. This phenomenon fits well with the TG results.

3D TG-FTIR spectra of pyrolysis products of the composites during the thermal degradation: (a) EVA0, (b) EVA1, and (c) EVA2.
More detailed information about FTIR spectra of pyrolysis products of the composites at a temperature from 30°C to 900°C during the thermal degradation is shown in Figure 12. Pure EVA started to degrade at about 300°C, which is higher than that of EVA1 and EVA2. The results may be mainly caused by the CO32− in LDH, which can be transformed into CO2 when heated.

FTIR spectra of pyrolysis products of the composites at different temperatures: (a) EVA0, (b) EVA1, and (c) EVA2.
With the temperature growing up, the release of CO, CO2, and H2O could be detected. It can be seen that a lot of carboxylic acid is evolved from pure EVA, while little can be found for EVA1 and EVA2. The reason may be that LDH can react with a carboxylic acid to form H2O. While the evolved carboxylic acid reflects the decarboxylation of EVA, the release of aliphatic hydrocarbons can be used to evaluate the break of the main chain. More detailed information about the pyrolysis products of the composites at different temperatures is shown in Figure 13.

Variations in the evolved (a) H2O, (b) CO2, (c) CO, (d) carboxylic acid, and (e) aliphatic hydrocarbons gases from EVA0, EVA1, and EVA2 samples.
From Figure 13(a), it can be seen that the release of H2O for pure EVA shows two steps, the first step begins at about 330°C and reaches its first peak at about 370°C. And the second step at above 500°C, the release of H2O reaches a high level. For EVA1 and EVA2, the process of water release has three steps, which fits well with the DTG results. It is obvious that EVA1 and EVA2 release more water than EVA0 samples. One possible reason is that LDHs can load more water. Another reason is that –OH groups in LDHs could contribute to the dehydrogenation reaction of EVA.
From Figure 13(b) and (c), it can be found that no CO2 peak can be found for pure EVA until about 500°C, while a signal can be seen at about 350°C for EVA/LDHs samples. The peak for EVA1 and EVA2 may be mainly caused by the CO32− present in LDHs, which can be transformed into CO2 when heated. However, the release of CO is reduced significantly while adding LDHs to EVA. The decrease of EVA may be one reason for this, but the main reason may be the increase of CO2.
It can be seen in Figure 13(d) and (e) that the curve of EVA0 shows a sharp peak at 470°C. And it produced more carboxylic acid and aliphatic hydrocarbons than EVA/LDHs samples. It may be inferred that LDHs can react with carboxylic acid and aliphatic hydrocarbons to form H2O, thus, the release of carboxylic acid and aliphatic hydrocarbons is maintained at a low level. So it could be concluded that MgAlFe-LDH is an efficient flame retardant to EVA.
Conclusions
This article has proposed a new way to reuse seawater bittern and RM. It has been proved that Mg/Al/Fe-LDHs were synthesized successfully by a coprecipitation method. Then, the flame retardant properties of the EVA/LDHs composites were carefully investigated by CCT, LOI, SDT, and TG-FTIR. The results showed that Mg/Al/Fe-LDHs could improve the flame retardant and thermal stability properties of the composites significantly. Besides, when Mg/(Al + Fe) molar ratio is 2.0/1.0, the composites show best flame retardant properties and thermal stability. This method is promising for improving the utilization rate of solid waste and the development of low-cost flame-retardant materials.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant no. 51572138), the Shandong Provincial Natural Science Foundation, China (grant no. ZR2018BB072), the Foundation of State Key Laboratory of High-efficiency Utilization of Coal and Green Chemical Engineering (grant nos 2018-K09 and 2018-K43) and Key Laboratory of Coastal Environmental Processes and Ecological Remediation, YICCAS (grant no. 2018KFJJ02).
