Abstract
The fatty acid ternary eutectics such as capric-lauric-myristic acid, capric-lauric-palmitic acid, capric-lauric-stearic acid, capric-myristic-palmitic acid, capric-myristic-stearic acid, and capric-palmitic-stearic acid and quaternary eutectics such as capric-lauric-myristic-palmitic acid, capric-lauric-myristic-stearic acid, capric-lauric-palmitic-stearic acid, capric-myristic-palmitic-stearic acid, and lauric-myristic-palmitic-stearic acid were selected as solid-liquid phase-change materials (PCMs). And then the innovative ternary eutectic/polystyrene (PS)/ternary eutectic and ternary eutectic/PS/quaternary eutectic form-stable phase-change composite fibrous membranes (PCCFMs) were directly prepared by the co-electrospinning method under the same spinning parameters. The as-prepared form-stable PCCFMs exhibited three-dimensional fibrous network structure with the average fiber diameter of about 410–780 nm according to the scanning electron microscopy images. The adhesion among nanofiber intersections was observed occasionally. The differential scanning calorimetry results showed that fatty acid eutectics have been successfully combined into the form-stable PCCFMs, and their melting peak temperatures mostly ranged from 15°C to 21°C with a temperature interval of 1°C. Moreover, the phase-change enthalpies reached about 40–61 kJ kg−1. It is concluded that these electrospun form-stable PCCFMs with appropriate phase-change temperatures could be considered as potential PCMs to be applied in thermo-regulating fibers and textiles.
Keywords
Introduction
Thermal energy storage is a technology of storing thermal energy in thermal reservoirs and then releasing the stored thermal energy to alleviate the imbalance between intermittent supply and demand. Currently, thermal energy storage materials with different heat storage forms have been extensively studied by researchers in order to realize the effective use of energy and improve the utilization of renewable resources. Thermal energy storage in the form of latent heat occurs through the phase transition from one phase to another phase of phase-change materials (PCMs) at a constant temperature. Additionally, the latent heat PCMs show higher heat storage density than that of the sensible heat PCMs. Hence, latent heat storage using PCMs can satisfy the high-performance thermal energy storage requirements for different application areas such as advanced cooling packaging, 1 solar domestic hot water systems, 2 solar-powered air-cooling systems, 3 household refrigerating, 4 energy conservation buildings, 5 thermal management of electronics, 6 phase-change fibers, 7 and so on.
Fatty acids and their derivative substances including fatty acid esters as well as eutectic mixtures have been widely recommended as common organic solid–liquid PCMs for storage and retrieval of thermal energy due to their excellent characteristics such as easy incorporation into polymers or inorganic materials, abundance in natural resources, good thermal and chemical stabilities, high thermal energy storage capacity, reversible phase-change behavior, nonflammability, nontoxicity, noncorrosive, no phase segregation, no supercooling, and so on. 8 -13 However, these solid–liquid PCMs have a notable drawback of liquid leakage problem, which seriously limits their applications. One of the most effective solutions to address this problem is to prepare form-stable phase-change composite systems in which polymers (e.g. poly(vinyl alcohol), 8 poly(styrene-co-acrylonitrile), 9 polyacrylonitrile, 10 polyurethane, 11 and polyethylene terephthalate 12 ) or inorganic materials (e.g. silica, 13 single-walled carbon nanotube, 14 hydrophobic expanded perlite, 15 expanded graphite, 16 and expanded vermiculite 17 ) provide good mechanical property and play the role of supporting materials. There are several methods for the preparation of form-stable PCMs such as electrospinning, 7,12 a Pickering emulsion route in an aqueous medium, 8 chemical synthesis, 9 physical adsorption method, 10,11 sol-gel method, 13 impregnation method, 14,15 -17 and so on. Electrospinning is an efficient technique for the fabrication of polymer nanofibers, which has also been recognized as an innovative and simple processing technique for the production of form-stable phase-change composite fibrous membranes (PCCFMs) with fiber diameter ranging from hundreds of nanometers to microns. Electrospun form-stable PCCFMs not only have good thermal energy storage performance but also have excellent flexibility and high morphological controllability.
According to the literatures, thermal energy storage performances including phase-change temperatures and enthalpies are extremely significant parameters that directly define whether or not these form-stable PCMs are suitable for a certain engineering field. The phase-change temperatures and enthalpies of fatty acids such as capric acid (CA), lauric acid (LA), myristic acid (MA), palmitic acid (PA), and stearic acid (SA), as well as their eutectics, have been systematically investigated in our previous work. 10,11,18 The research results indicated that fatty acid eutectics exhibited the lower phase-change temperatures in comparison with those of the single fatty acids. Obviously, the selectivity and diversity of phase transition temperatures of these solid–liquid PCMs were greatly expanded through developing fatty acid eutectics. Nevertheless, there is still a lack of some solid–liquid PCMs with the special melting peak temperatures such as 15°C, 16°C, 17°C, and so on. For example, the application of PCMs for smart textiles has also been reported by Mondal. 19 Mondal reported that one of the required properties for the PCMs applied in the field of the temperature-regulating textiles is that their melting temperatures should be between 15°C and 35°C. Moreover, Sarı and Karaipekli 20 also reported that the indoor comfortable temperature is usually in 15–25°C for energy-saving building application. In addition, Akeiber et al. 21 reported a review on PCMs for sustainable passive cooling in building envelopes. They described that from a physical point of view, the melting temperature of the PCMs should be in the range of 10–30°C to provide thermal comfort for occupants.
Therefore, this article focuses on the design and development of novel electrospun polystyrene (PS)-based form-stable PCCFMs with melting peak temperatures mainly ranging from about 15°C to 21°C with a temperature interval of 1°C via co-electrospinning method. The fatty acid ternary eutectics such as capric-lauric-myristic acid (CLM), capric-lauric-palmitic acid (CLP), capric-lauric-stearic acid (CLS), capric-myristic-palmitic acid (CMP), capric-myristic-stearic acid (CMS), and capric-palmitic-stearic acid (CPS) and quaternary eutectics such as capric-lauric-myristic-palmitic acid (CLMP), capric-lauric-myristic-stearic acid (CLMS), capric-lauric-palmitic-stearic acid (CLPS), capric-myristic-palmitic-stearic acid (CMPS), and lauric-myristic-palmitic-stearic acid (LMPS) were selected as solid–liquid PCMs. The morphological structure and thermal properties of the developed electrospun ternary eutectic/PS/ternary eutectic and ternary eutectic/PS/quaternary eutectic form-stable PCCFMs were characterized by scanning electron microscopy (SEM) and differential scanning calorimetry (DSC), respectively.
Experimental setup
Materials
The PS (Mw = 260,000) particles were purchased from Scientific Polymer Products, Inc. The raw materials such as CA, LA, MA, PA, SA, and N,N-dimethyl formamide (DMF) solvent were supplied by the Sinopharm Group Chemical Reagent Co., Ltd (Shanghai, China). The fatty acid ternary eutectics (i.e. CLM, CLP, CLS, CMP, CMS, and CPS) and quaternary eutectics (i.e. CLMP, CLMS, CLPS, CMPS, and LMPS) were prepared and selected as solid–liquid PCMs base on the data published in my previous articles. 18 All of the chemicals were used as received without further purifications.
Fabrication of electrospun PS-based form-stable PCCFMs
The PS particles were chosen in this study as the polymer materials of co-electrospinning because of their excellent properties such as good spinnability, low cost, and so on. The 10 wt% PS particles were first added into DMF solvent and then followed by magnetic stirring to achieve the homogeneous PS solutions. Subsequently, ternary and quaternary eutectics were respectively dissolved into the above PS solutions to prepare ternary eutectic/PS and quaternary eutectic/PS composite solutions with the 50% mass fraction of fatty acid eutectic. Figure 1 reveals the schematic diagram of the co-electrospinning experimental setup. The obtained composite solutions were held in the plastic syringes fitted with a metal needle with an inner diameter of 0.6 mm. The electrode of the high voltage power supply was clamped to the metal needle tip. A rotating roller with the rotation rate of 200 r min−1 for the collection of phase-change composite nanofibers was grounded. The tip-to-collector distance, working voltage, and flow rate of the syringe pump were set at 18 cm, 18 kV, and 0.5 mL h−1, respectively. The ternary eutectic/PS and quaternary eutectic/PS phase-change composite nanofibers were respectively electrospun and deposited on an aluminum foil covered on the surface of the grounded roller to form ternary eutectic/PS/ternary eutectic and ternary eutectic/PS/quaternary eutectic form-stable PCCFMs under the same spinning parameters. The detailed sample codes are summarized in Tables 1 and 2.

Schematic diagram of the experimental setup for co-electrospinning.
The sample codes of electrospun fatty acid ternary eutectic/PS/fatty acid ternary eutectic form-stable PCCFMs.

PS: polystyrene; PCCFM: phase-change composite fibrous membrane; CLM: capric-lauric-myristic acid; CLP: capric-lauric-palmitic acid; CLS: capric-lauric-stearic acid; CMP: capric-myristic-palmitic acid; CMS: capric-myristic-stearic acid; CPS: capric-palmitic-stearic acid.
The sample codes of electrospun CMS/PS/quaternary eutectic and ternary eutectic/PS/LMPS form-stable PCCFMs.

CMS: capric-myristic-stearic acid; PS: polystyrene; LMPS: lauric-myristic-palmitic-stearic acid; PCCFM: phase-change composite fibrous membrane; CLMP: capric-lauric-myristic-palmitic acid; CLMS: capric-lauric-myristic-stearic acid; CLPS: capric-lauric-palmitic-stearic acid; CMPS: capric-myristic-palmitic-stearic acid; CLM: capric-lauric-myristic acid; CLP: capric-lauric-palmitic acid; CLS: capric-lauric-stearic acid; CMP: capric-myristic-palmitic acid; CPS: capric-palmitic-stearic acid.
Characterizations
The surface morphology of electrospun ternary eutectic/PS/ternary eutectic and ternary eutectic/PS/quaternary eutectic form-stable PCCFMs were observed by scanning electronic microscopy (SEM, S3400, Hitachi, Tokyo, Japan). The DSC (214Polyma, Netzsch, Selb, Germany) was used to study the thermal behaviors of electrospun ternary eutectic/PS/ternary eutectic and ternary eutectic/PS/quaternary eutectic form-stable PCCFMs. The phase-change enthalpies were determined by numerical integration of the area under the peaks. The membranes were heated and then cooled in the temperature range of −20°C to 60°C at the scanning rate of 8°C min−1 under a 50 mL min−1 nitrogen atmosphere.
Results and discussion
Morphological structure
Figures 2 to 4 respectively show the representative SEM images of electrospun ternary eutectic/PS/ternary eutectic (i.e. CLM/PS/CLP, CLM/PS/CLS, CLM/PS/CMP, CLM/PS/CMS, CLM/PS/CPS, CLP/PS/CLS, CLS/PS/CMP, CLS/PS/CMS, and CLS/PS/CPS), CMS/PS/quaternary eutectic (i.e. CMS/PS/CLMP, CMS/PS/CLMS, CMS/PS/CLPS, and CMS/PS/CMPS), and ternary eutectic/PS/LMPS (i.e. CLM/PS/LMPS, CLP/PS/LMPS, CLS/PS/LMPS, CMP/PS/LMPS, CMS/PS/LMPS, and CPS/PS/LMPS) form-stable PCCFMs obtained by varying the species of fatty acid eutectics in the solutions, under the same co-electrospinning parameters. As can be seen in Figure 2, the ternary eutectic/PS/ternary eutectic form-stable PCCFMs consisting of two kinds of electrospun ternary eutectic/PS composite nanofibers presented the three-dimensional porous network structure. The SEM images revealed that the obtained ternary eutectic/PS composite nanofibers exhibited nano-scale fiber diameter and their average fiber diameter was in the range of about 410–780 nm measured from SEM micrographs. Meanwhile, it is interesting to find that the ternary eutectic/PS composite nanofibers slightly adhered together at some bonding sites. Some nanofibers presented a smooth and cylindrical morphological structure, whereas some nanofibers had a rough surface. It can be clearly seen from Figures 3 and 4 that electrospun CMS/PS/quaternary eutectic and ternary eutectic/PS/LMPS form-stable PCCFMs presented similar morphology and structure characteristics with those of ternary eutectic/PS/ternary eutectic form-stable PCCFMs. The type of the loaded fatty acid eutectics had no remarkable effect on the morphological structure of the form-stable PCCFMs. The SEM observation indicated that fatty acid eutectics encapsulated into the PS nanofibers formed one component of composite nanofibers and presented the form-stable state even when the working temperature of the SEM characterization (about 25°C) was slightly higher than the melting temperatures of fatty acid eutectics, except for LMPS (see Table 3). Obviously, electrospun PS nanofibers provided the supporting skeleton and mechanical strength for the whole membranes, as well as prevented the seepage of the molten fatty acid eutectics due to the effect of capillary and surface tension forces.

Representative SEM images of electrospun ternary eutectic/PS/ternary eutectic form-stable PCCFMs: (a) CLM/PS/CLP, (b) CLM/PS/CLS, (c) CLM/PS/CMP, (d) CLM/PS/CMS, (e) CLM/PS/CPS, (f) CLP/PS/CLS, (g) CLS/PS/CMP, (h) CLS/PS/CMS, and (i) CLS/PS/CPS.

Representative SEM images of electrospun CMS/PS/quaternary eutectic form-stable PCCFMs with the different kinds of quaternary eutectics: (a) CMS/PS/CLMP, (b) CMS/PS/CLMS, (c) CMS/PS/CLPS, and (d) CMS/PS/CMPS.

Representative SEM images of electrospun ternary eutectic/PS/LMPS form-stable PCCFMs with the different kinds of ternary eutectics: (a) CLM/PS/LMPS, (b) CLP/PS/LMPS, (c) CLS/PS/LMPS, (d) CMP/PS/LMPS, (e) CMS/PS/LMPS, and (f) CPS/PS/LMPS.
The peak onset temperatures (T o), peak end temperatures (T e), melting peak temperatures (T m), freezing peak temperatures (T f), melting enthalpies (ΔH m), and freezing enthalpies (ΔH f) of fatty acid ternary and quaternary eutectics.

CLM: capric-lauric-myristic acid; CLP: capric-lauric-palmitic acid; CLS: capric-lauric-stearic acid; CMP: capric-myristic-palmitic acid; CMS: capric-myristic-stearic acid; CPS: capric-palmitic-stearic acid; CLMP: capric-lauric-myristic-palmitic acid; CLMS: capric-lauric-myristic-stearic acid: CLPS: capric-lauric-palmitic-stearic acid; CMPS: capric-myristic-palmitic-stearic acid; LMPS: lauric-myristic-palmitic-stearic acid.
Thermal energy storage properties
The DSC curves of electrospun ternary eutectic/PS/ternary eutectic, CMS/PS/quaternary eutectic, and ternary eutectic/PS/LMPS form-stable PCCFMs are plotted in Figures 5 to 7. Table 3 lists the peak onset temperatures (To ), peak end temperatures (T e), melting peak temperatures (T m), freezing peak temperatures (T f), melting enthalpies (ΔH m), and freezing enthalpies (ΔH f) of fatty acid ternary and quaternary eutectics. Tables 4 and 5 summarize the corresponding thermal energy storage data of electrospun ternary eutectic/PS/ternary eutectic (i.e. CLM/PS/CLP, CLM/PS/CLS, CLM/PS/CMP, CLM/PS/CMS, CLM/PS/CPS, CLP/PS/CLS, CLS/PS/CMP, CLS/PS/CMS, and CLS/PS/CPS) and ternary eutectic/PS/quaternary eutectic (i.e. CMS/PS/CLMP, CMS/PS/CLMS, CMS/PS/CLPS, CMS/PS/CMPS, CLM/PS/LMPS, CLP/PS/LMPS, CLS/PS/LMPS, CMP/PS/LMPS, CMS/PS/LMPS, and CPS/PS/LMPS) form-stable PCCFMs, respectively. The DSC curves shown in Figures 5 to 7 indicate that the significant endothermic and exothermic peaks were clearly observed due to the introduction of fatty acid ternary and quaternary eutectics acting as solid–liquid PCMs, which also demonstrated that ternary/quaternary eutectics have been well encapsulated into the electrospun PS nanofibers during the co-electrospinning process. In order to better observe the phase-change temperature difference between the fatty acid ternary/quaternary eutectics and the obtained PS-based form-stable PCCFMs, Figure 8 provides the distribution of the melting peak temperatures of fatty acid ternary and quaternary eutectics. Additionally, the distribution of the melting peak temperatures of electrospun form-stable PCCFMs is shown in Figure 9. It was clearly found that the melting peak temperatures of electrospun ternary eutectic/PS/ternary eutectic and ternary eutectic/PS/quaternary eutectic form-stable PCCFMs were slightly lower than those of the loaded fatty acid ternary/quaternary eutectics. In other words, the phase-change temperatures of electrospun form-stable PCCFMs were shifted to the lower temperature range, which could be due to the synergy effect of the two different types of fatty acid ternary/quaternary eutectics. This result also confirmed that electrospun PS-based form-stable PCCFMs with the new phase-change temperatures could be flexibly designed through the co-electrospinning method by changing the types of fatty acid eutectics in the composite solutions.

DSC curves of electrospun ternary eutectic/PS/ternary eutectic form-stable PCCFMs during the melting and freezing processes.

DSC curves of electrospun CMS/PS/quaternary eutectic form-stable PCCFMs with the different kinds of quaternary eutectics during the melting and freezing processes.

DSC curves of electrospun ternary eutectic/PS/LMPS form-stable PCCFMs with the different kinds of ternary eutectics during the melting and freezing processes.

Distribution of melting peak temperatures of fatty acid ternary and quaternary eutectics.

Distribution of melting peak temperatures of electrospun ternary eutectic/PS/ternary eutectic, CMS/PS/quaternary eutectic, and ternary eutectic/PS/LMPS form-stable PCCFMs.
The peak onset temperatures (T o), peak end temperatures (T e), melting peak temperatures (T m), freezing peak temperatures (T f), melting enthalpies (ΔH m), and freezing enthalpies (ΔH f) of electrospun ternary eutectic/PS/ternary eutectic form-stable PCCFMs.
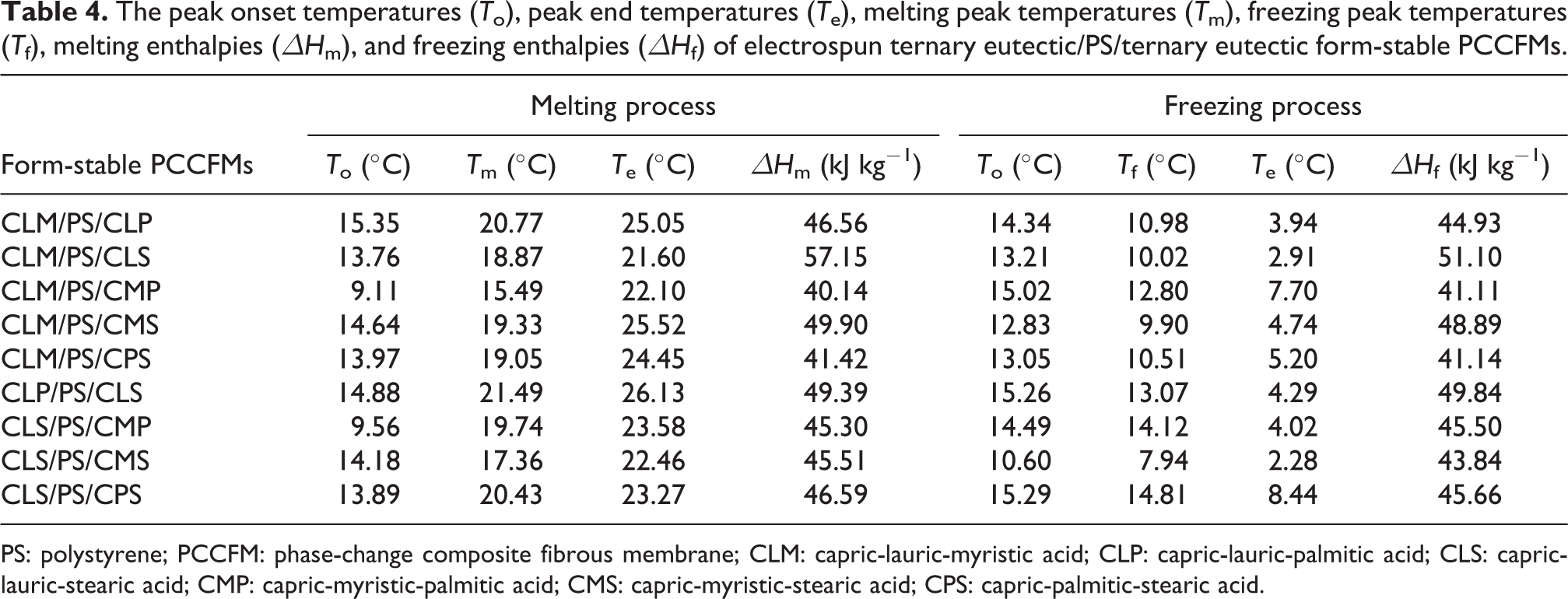
PS: polystyrene; PCCFM: phase-change composite fibrous membrane; CLM: capric-lauric-myristic acid; CLP: capric-lauric-palmitic acid; CLS: capric-lauric-stearic acid; CMP: capric-myristic-palmitic acid; CMS: capric-myristic-stearic acid; CPS: capric-palmitic-stearic acid.
The peak onset temperatures (T o), peak end temperatures (T e), melting peak temperatures (T m), freezing peak temperatures (T f), melting enthalpies (ΔH m), and freezing enthalpies (ΔH f) of electrospun ternary eutectic/PS/quaternary eutectic form-stable PCCFMs.

PS: polystyrene; PCCFM: phase-change composite fibrous membrane; CMS: capric-myristic-stearic acid; CLM: capric-lauric-myristic acid; CLP: capric-lauric-palmitic acid; CLS: capric-lauric-stearic acid; CMP: capric-myristic-palmitic acid; CPS: capric-palmitic-stearic acid; CLMP: capric-lauric-myristic-palmitic acid; CLMS: capric-lauric-myristic-stearic acid: CLPS: capric-lauric-palmitic-stearic acid; CMPS: capric-myristic-palmitic-stearic acid; LMPS: lauric-myristic-palmitic-stearic acid.
Furthermore, the phase-change enthalpies of electrospun ternary eutectic/PS/ternary eutectic and ternary eutectic/PS/quaternary eutectic form-stable PCCFMs were in the range of about 40–61 kJ kg−1 (see Tables 4 and 5), which obviously decreased compared to those of the pure fatty acid ternary and quaternary eutectics owing to the introduction of the PS supporting matrix. The efficiency of melting enthalpies (the ratio between the experimental value and the theoretical value) for electrospun CLM/PS/CLP, CLM/PS/CLS, CLM/PS/CMP, CLM/PS/CMS, CLM/PS/CPS, CLP/PS/CLS, CLS/PS/CMP, CLS/PS/CMS, and CLS/PS/CPS form-stable PCCFMs were about 70%, 87%, 60%, 72%, 60%, 74%, 67%, 65%, and 67%, respectively. Moreover, the efficiency of melting enthalpies for electrospun CMS/PS/CLMP, CMS/PS/CLMS, CMS/PS/CLPS, CMS/PS/CMPS, CLM/PS/LMPS, CLP/PS/LMPS, CLS/PS/LMPS, CMP/PS/LMPS, CMS/PS/LMPS, and CPS/PS/LMPS form-stable PCCFMs were also determined as approximately 65%, 85%, 66%, 69%, 58%, 71%, 63%, 61%, 83%, and 60%, respectively. Table 6 gives a comparison of thermal properties of some composite form-stable PCMs in the literature. It is noteworthy that the phase-change temperatures of the reported composite form-stable PCMs with the high phase-change enthalpies are usually higher than 30°C. The electrospun PS-based form-stable PCCFMs in this work exhibited relatively appropriate phase-change temperatures for temperature-regulated fibers and textiles applications.
Comparison of melting and freezing peak temperatures and enthalpies (T m, T f, ΔH m, and ΔH f) of some form-stable composite PCMs in the published literature.

PCM: phase-change material; PA: palmitic acid; SAN: poly(styrene-co-acrylonitrile); GMS: glycerol monostearate; PET: polyethylene terephthalate; SA: stearic acid; PEG: polyethylene glycol; SWCNs: single-walled carbon nanotube; EPO: hydrophobic expanded perlite; EG: expanded graphite; LA-MA-SA: lauric-myristic-stearic acid; EV: expanded vermiculite; LA-PA-SA: lauric-palmitic-stearic acid; VMT: vermiculite.
Conclusions
A series of electrospun form-stable PCCFMs of ternary eutectic/PS/ternary eutectic, CMS/PS/quaternary eutectic, and ternary eutectic/PS/LMPS were prepared by co-electrospinning method for thermal energy storage. The SEM images revealed that the obtained fatty acid eutectic/PS composite nanofibers exhibited nano-scale fiber diameter. Some nanofibers presented a smooth and cylindrical morphological structure, whereas some nanofibers had a rough surface. The DSC results indicated that the fatty acid eutectics were well confined into the PS-based fibrous membranes. The melting peak temperatures mainly ranged from 15°C to 21°C with a temperature interval of 1°C and the enthalpies were about 40–61 kJ kg−1. And the efficiency of melting enthalpies of fatty acid eutectics retained in form-stable PCCFMs was determined as about 58–87%. These form-stable PCCFMs could be considered as preferential potential thermal energy storage materials for improving the thermal comfort of fibers and textiles.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by the National Natural Science Foundation of China (grant number 51706092), Natural Science Foundation of Fujian Province of China (grant number 2018J05091), Cultivation Program for Outstanding Young Scholars in Colleges and Universities of Fujian Province of China (grant number 2018), and Open Project Program of Fujian Key Laboratory of Novel Functional Textile Fibers and Materials, Minjiang University (grant number FKLTFM2006).
