Abstract
The main objective of soft tissue engineering (STE) is to improve the quality of life by replacing damaged tissues with synthetic scaffolds. Collagen is the key structural protein of the native extracellular matrix with nano-scale diameter. Accordingly, this study focused on the fabrication of the blended and co-electrospinning hybrid synthetic polycaprolactone/polyurethane (PCL/PU) nanofibers scaffolds and investigates their
Introduction
Soft tissue engineering (STE) has been introduced as a new strategy to replace damaged or diseased soft tissues to overcome the limitations of current therapies. 1 The collagen fibers are a major component in the extracellular matrix (ECM) of soft tissue and are found in the form of nano-fibrous structure.2–4 Therefore, nanofibers are great importance to mimic the structural, mechanical, and biological environment of native ECM.5,6
Generally, designing biocompatible synthetic nanofibers scaffolds by bio-materials that they can properly interact with cells and tissue is a functional approach in tissue engineering and regenerative medicine.4,7
In this regard, using the histological analysis as the gold standard for biocompatibility tissue examination to assess the inflammation or healing stage and to monitor the presence and distribution of degradation products that dissolved into the surrounding tissue is necessary, because the synthetic nanofibers scaffolds are recognized as foreign bodies and evoke an array of inflammatory responses and unwanted reactions. 8 In this regard, finding suitable bio-materials and methods for the fabrication of biocompatible synthetic nanofibers scaffolds for STE applications with improvement in immune response and acceptable histological examination is an interesting field of research.
According to the author’s knowledge, there are many studies about applying different polymers such as polycaprolactone (PCL), polyethylene terephthalate, polyurethane (PU), Poly (lactic acid) , and poly (glycolic acid) as neat and hybrid synthetic nanofibers scaffold in STE.
Polycaprolactone is well known as a strong, non-toxic, low degradable, and semi-crystalline polymer.9,10 Despite the advantages of PCL, but due to the lack of elasticity, it has limitations in STE applications in the form of a neat structure. Therefore, hybrid PCL-based structures of a blend of two or more polymers are suggested to overcome the limitations of PCL for successful clinical applications. 11 Polyurethane is a flexible, biocompatible, non-toxic polymer with a low degradation rate. Also, PU consists of soft and hard segments which allow the PU to behave in elastomer and thermoplastic state. 12 Collagen and elastin are responsible for the stiffness and elasticity properties in the ECM. Accordingly, next to the PCL (with excellent mechanical properties), the flexible polymer such as PU should be used for simulation of the elasticity of elastin in the ECM. 13
Different methods were developed for the fabrication of three-dimensional synthetic nanofibers scaffolds for STE.14–17 Electrospinning is an attractive, ideal, and effective method because it has enabled the production of a large variety of materials with an extremely high specific surface-to-volume ratio or mass ratio (due to their small diameters), low density, high pore volume, and high flexibility.18,19
It is confirmed that PCL and PU as two biopolymers have been investigated in the form of neat structures to be used for STE due to high biocompatibility and controllable degradation rate.
But the tissue response and immune histological biocompatible evaluation of hybrid synthetic polycaprolactone/polyurethane (PCL/PU) nanofibers in the form of blended (blend-electrospinning) and co-electrospinning (co-electrospinning) nanofibers
In the following characterizing, the morphology and 1. Inflammation: that it consisted of acute and chronic response. Breakdown products of complement activation, chemokine released from activated platelets, and other mediators that produced at the site of implantation section as chemotactic agents to recruit neutrophils and then monocytes over the next 6 to 48 h. These inflammatory cells eliminate the offending agents and clear the debris. In the following, the inflammation resolves as a result of clearing the injurious agents and necrotic cells. 2. Cell proliferation: this stage, which takes up to 10 Days. Epithelial cells, endothelial cells, and fibroblasts are proliferated in this stage. In this step, endothelial cells proliferate to form new blood vessels (angiogenesis), and in following, fibroblasts proliferate and migrate into the site of injury.
20
Migration and proliferation of fibroblasts and deposition of loose connective tissue, together with the vessels and interspersed mononuclear leukocytes, form granulation tissue. The term granulation tissue is characterized by proliferation of fibroblasts and new thin-walled, delicate capillaries in a loose ECM, often with admixed inflammatory cells, mainly macrophages. Granulation tissue progressively fills the site of injury.21,22 The amount of granulation tissue that is formed depends on the size of the tissue defect created by the wound and the intensity of inflammation.
20
3. Remodeling: After fibroblasts form and deposit connective tissue, the connective tissue in the scar continues to be modified and remodeled. This process begins 2–3 weeks after implantation and may last for months or years.
23
Accordingly, a significant emphasis and object of the present article are the development and evaluation of the biocompatible hybrid synthetic PCL/PU nanofibers scaffolds (PCL/PU scaffolds) and examination
In this regard, the present study has focused on the fabrication of the hybrid synthetic PCL/PU as nanofibers scaffolds with different composite ratios by blend-electrospinning and co-electrospinning methods.
Methods and materials
Materials
The PCL with an average 80,000 molecular weight (Mn = 80,000) and PU (biodegradable and biomedical grade) were supplied by Sigma Aldrich, USA. Dichloromethane, ethanol, chloroform, Tetrahydrofuran (THF), N, N-dimethylformamide (DMF), and 1,1,1,3,3-hexafluoro-2-propanol (HFIP) were purchased from Merck, Germany. The solvents were used without further purification. The animals (rats) were purchased from the Institute of Medical Sciences, Mashhad, Iran.
Electrospinning section
Fabrication of the PCL/PU scaffolds by blend-electrospinning method
For blend-electrospinning, the PCL/PU solution was prepared by dissolving 8 w/v% of the PCL and PU polymers (2 g) in 25 mL HFIP at 50°C through magnetic stirring for 3 h. In following, the mixed polymer solution was loaded into a plastic syringe connected to a 20 gauge needle. The electrospun PCL/PU scaffolds with different composite ratios (25:75, 50:50, and 75:25) were fabricated by a custom-designed electrospinning setup. A cylinder collector covered with aluminum foil was applied for the collection of the blended PCL/PU scaffolds.
Fabrication of the PCL/PU scaffolds by Co-electrospinning method
Co-electrospinning and Blend-electrospinning conditions.

PCL: polycaprolactone; PU: polyurethane.
The custom-designed electrospinning setup (Figure 1(a) and (b) Schematic diagrams of electrospinning design, (a) blended electrospinning method, and (b) Co-electrospinning method.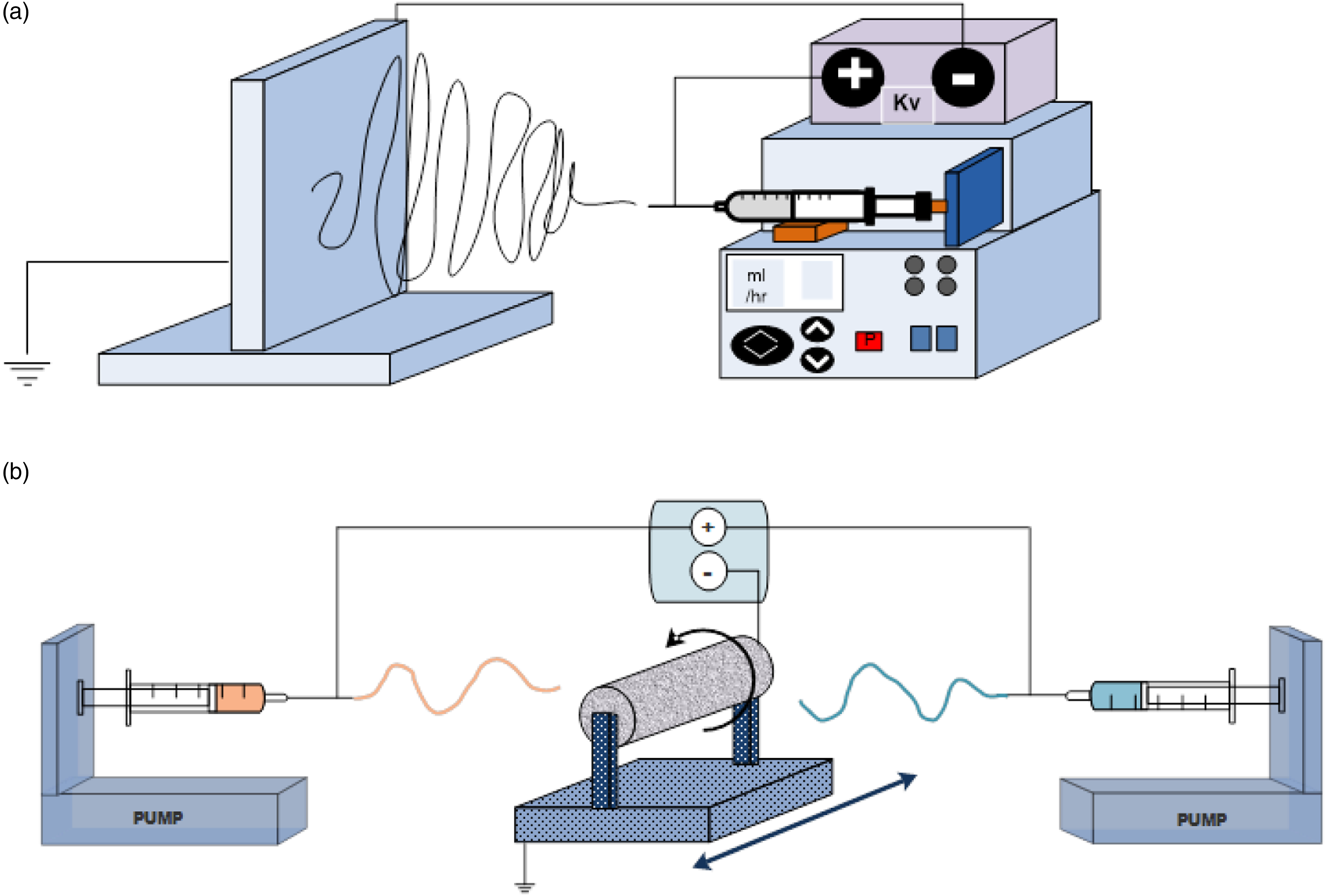
In blend and co-electrospinning methods, the temperature and relative humidity as environment parameters were kept at 28 C and 30%, respectively. The details of the optimum blend and co-electrospinning conditions were reported in Table 1.
Morphological and chemical characterization of neat and hybrid PCL/PU nanofibers
Fiber diameter and porosity measurement
Characterization of the fiber diameter distribution of the all blended and co-electrospinning PCL/PU scaffolds was evaluated by Scanning Electron Microscopy ((VEGA II TESCAN and MIRA II TESCAN with 15 kV accelerating voltage in both cases)) with 15 kV accelerating voltage after gold–platinum coating (
The apparent density and porosity of the fabricated synthetic PCL/PU nanofibers (neat and hybrid nanostructures) were measured by the gravimetric method using the Equation (1) and (2), respectively.24,25

The bulk density of the PCL and PU is 1.58 g/cm3 and 1.699 g/cm3, respectively.
Fourier Transform Infrared Spectrometer
Fourier transform infrared spectra (FTIR) of nanofibers were evaluated using a FTIR spectrophotometer (FTIR, Nicolet Thermo Scientific Co.). Spectra were recorded and used to determine the presence of specific functional groups using KBR pellets containing 3 mg of each sample between 400 to 4000 cm−1 ranges.
In-vivo study
The nanofibers structures due to their unique properties can be used as synthetic nanofibers scaffolds for clinical application in STE. In this regard, the present study tries to examine the healing effect of electrospun PCL/PU nanofibers in the form of co-electrospinning and blended PCL/PU scaffolds by
Animals study
The evaluation of the histological analysis of fabricated electrospun nanofibers (neat, blended, and co-electrospinning nanofibers), Wistar male rats (age 6 weeks), weighing approximately 0.17 kg, were used for the
For investigating the immune response and histological examination of the blended and co-electrospinning PCL/PU scaffolds in
Histopathological evaluation of implanted samples
The histopathological analysis was evaluated by determining seven healing parameters including edema, foreign body granulomatous reaction, granulation tissue formation, inflammation, calcification, fibroblastic change, and Necrosis. After 45 days, the samples (the implanted sample with the surrounding of the healthy tissue) were fixed in 10% neutral buffered formalin. After 24 h, the specimens were cut from the middle and passed in an auto technical apparatus, and were exposed to liquid paraffin. After 8 h, the biopsy specimens were extracted from the liquid paraffin and cut into 5–6 microns thick bands, and fixed on the slide to study the mentioned parameters. The Hematoxylin and Eosin (H&E) staining were performed following the standard techniques for microscopically histopathological evaluation. The score of absent (−), mild (+), moderate (++), severe (+++), and very severe (++++) was allocated to the healing parameters in the wound healing process.26-28
Result
Fiber diameter and porosity measurement of blended and co-electrospinning PCL/PU scaffolds
The blend-electrospinning and co-electrospinning methods of the PCL/PU were successfully carried out in the nano-scale range without any beads and with random distribution (Figure 2(a)–(c) and Table 2). The average fiber diameters of PCL and PU were reported at 433 ± 80 nm and 470 ± 95 nm, respectively. According to the obtained results, each method leads to fabricate the nanostructure by close average fiber diameter together. By adding the PU in PCL/PU scaffolds (blended and co-electrospinning), the increase of fiber diameter was observed. The obtained results indicated that the co-electrospinning PCL/PU scaffolds have shown a smaller fiber diameter than blended PCL/PU scaffolds. The mean porosity of the neat synthetic scaffolds (neat PCL and PU) were reported 89.1±0.69% and 63±0.46%, respectively. In the blended and co-electrospinning PCL/PU scaffolds, with increasing the 25 to 75% PCL, the porosity increases from (75.33 ± 1.15 to 83.75 ± 1.70) and (74.30 ± 0.15 to 81.00 ± 1.30), respectively. The results related to the porosity of the blended and co-electrospinning PCL/PU scaffolds are presented in Table 2. Morphological characterization of electrospun nanofibers (a) FESEM images of neat and hybrid nanofibers, (b) Average fiber diameter of blended polycaprolactone/polyurethane scaffolds, (b) Average fiber diameter of co-electrospinning polycaprolactone/polyurethane scaffolds, and (c) Average fiber diameter of blended polycaprolactone/polyurethane scaffolds. Average fiber diameter and porosity of neat, blended, and co-electrospinning PCL/PU scaffolds. PCL: polycaprolactone; PU: polyurethane.

FTIR
The FTIR spectra of the functional groups of the neat and hybrid nanofibers composed of PCL and PU were presented in Figure 3 in the spectral range of 400–4000 cm−1. Fourier Transform Infrared Spectrometer spectra of electrospun polycaprolactone/polyurethane nanofibers.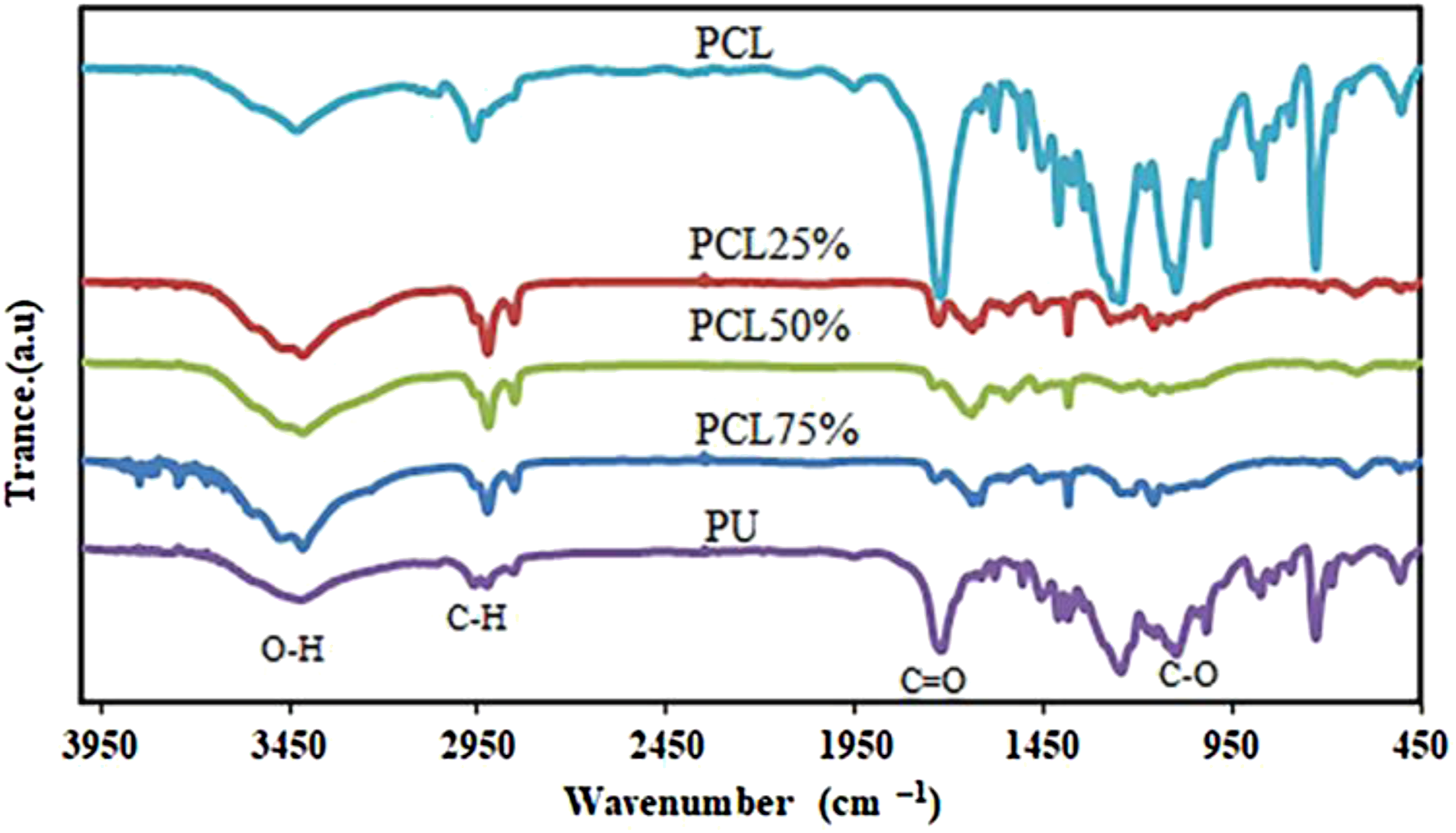
For the PCL nanofibers, the characteristic broad band exhibited at 3400–3500 cm−1 belongs to N–H (stretching) group. The broad absorptions observed at the range of (2850–3000 cm−1), (1705–1725 cm−1), and (1000–1300 cm−1) were attributed to the –CH2 (stretching), carbonyl ketone –C = O (stretching), and C–O groups, respectively. The FTIR spectrum of N–H (stretching), C–H stretching (aromatic group), and –C = O (carbonyl ketone – stretching) was observed at (3000–3500), 2930, and (1730–1750) cm−1, respectively, and were attributed to the PU. The existence of all peaks in the PCL/PU scaffolds with different composite ratios confirmed the presence of both polymers in the hybrid fabricated nanofibers.
The histopathological study of blended PCL/PU scaffolds
Edema
Results of pathology evaluation of the fabricated Polycaprolactone/Polyurethane (PCL/PU) blended nanofibers.
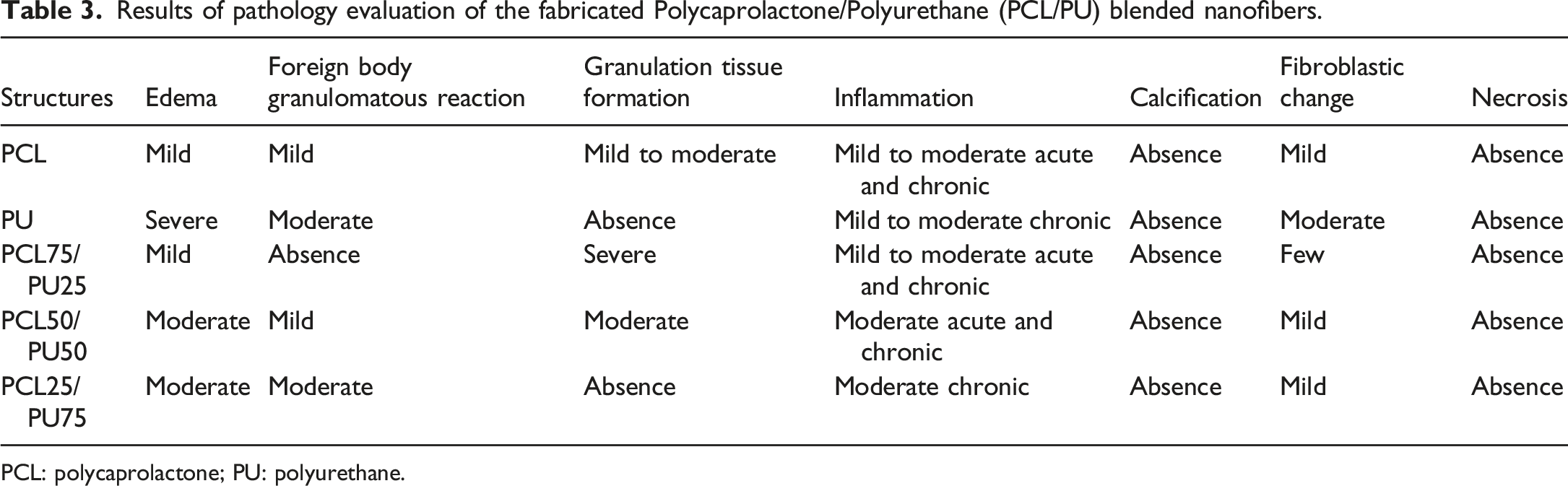
PCL: polycaprolactone; PU: polyurethane.

Histopathological assessment of the neat polycaprolactone and polyurethane nanofibers by hematoxylin and eosin staining. On days 45 after surgery, randomly a group of rats was selected and sacrificed. *: Edema, #: Fibroblastic change, &: Foreign body granulomatous reaction, $: Inflammation, ∼: granulation tissue.
Foreign body granulomatous reaction
According to the histopathological results (Table 3, Figure 5), by increasing the composite ratio of PCL from 25 to 75%, the amount of foreign body giant cell decreased in the blended PCL/PU scaffolds (Table 3, Figure 4). These results are confirmed according to the foreign body giant cell results in neat PCL and PU nanofibers that were reported scant and severe, respectively. These results indicate the effect of the composite ratio of PCL and PU on the body’s adaptation of blended PCL/PU scaffolds. Histopathological assessment of blended polycaprolactone/polyurethane scaffolds by hematoxylin and eosin staining. On days 45 after surgery, randomly a group of rats was selected and sacrificed. *: Edema, #: Fibroblastic change, &: Foreign body granulomatous reaction, $: Inflammation, ∼: granulation tissue.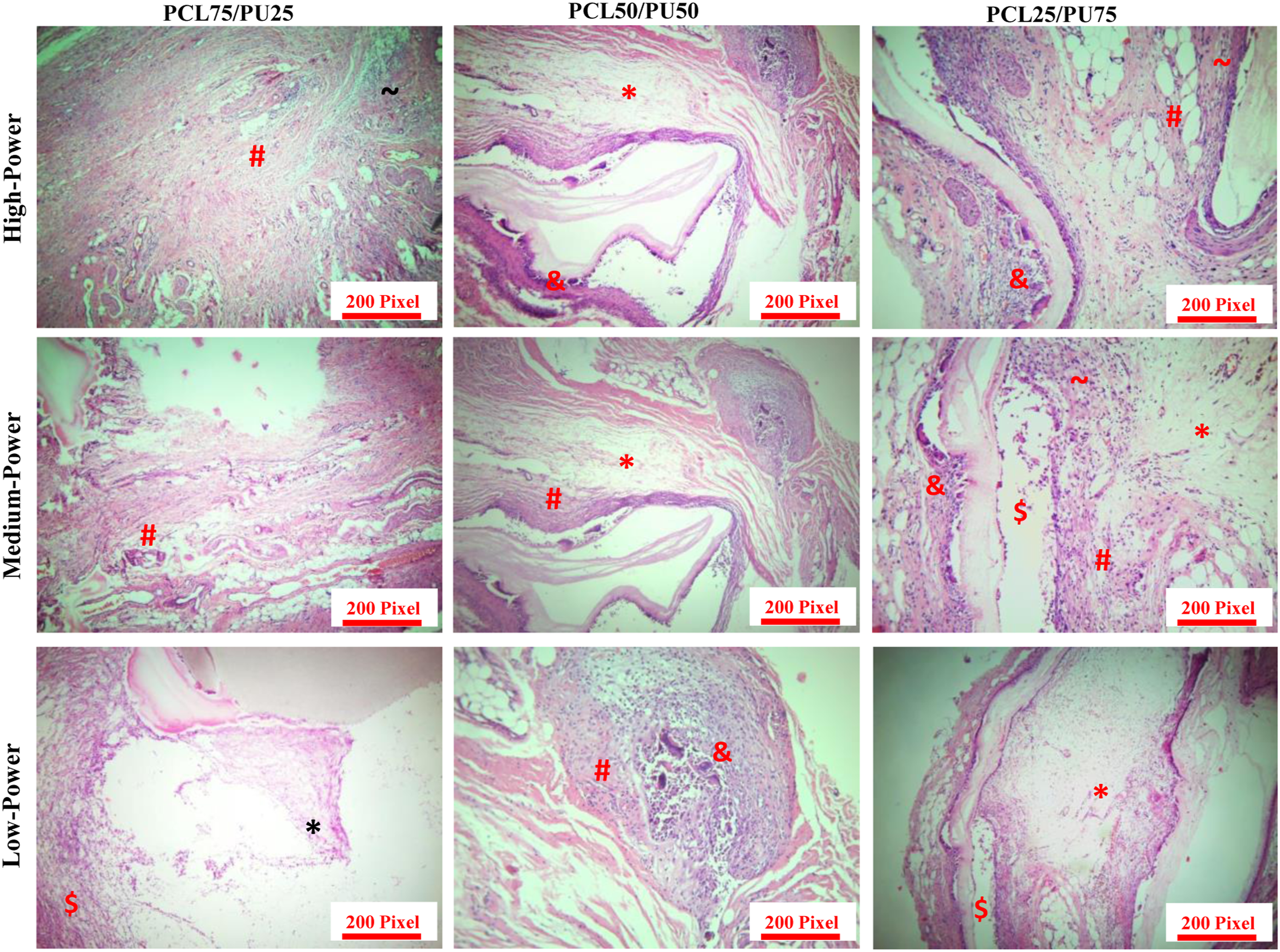
Granulation tissue formation
According to Table 3 and Figure 4 in the neat PU and PCL nanofibers were observed in the absent and mild to moderate of granulation tissue formation, respectively. The results in Table 3 have shown that the blended PCL/PU scaffolds with the more composite ratio of PCL have more granulation tissue formation compared to blended PCL/PU scaffolds with the more composite ratio of PU (Figure 5). On the other hand, in the blended PCL/PU scaffolds with 50 and 75% of PCL, the amount of the granulation tissue formation reported moderate and serve, while in blended PCL/PU scaffolds, this parameter decreased by increasing the composite ratio of PU from 25 to 75%. The obtained results confirmed that the amount of the granulation tissue formation is directly related to composite ratio of PCL and PU in blended PCL/PU scaffolds. The degree of granulation tissue formation indicates the body’s ability to adapt with fabricated structures to a foreign body and its tissue repair.
Inflammation
As shown in Figures 4 and 5, all structures (neat and blended PCL/PU scaffolds) showed mild to moderate acute and chronic results about inflammation after implantation in the rat model, and in all structures, the rate of inflammation is almost the same.
Calcification
The histopathological reports confirmed no calcification in the neat and blended PCL/PU scaffolds (Figures 4 and 5).
Fibroblastic change
The fibroblastic changes in PCL and PU as neat nanofibers were reported mild and moderate, respectively (Figure 4). As you can see in Figure 5, the rate of fibroblast reaction in the blended PCL/PU scaffolds with 50 and 75% of PU was determined moderate that this result is based on the amount of fibroblastic change in neat PU nanofibers.
Necrosis
According to the histopathological results (Table 3, Figures 4 and 5), no necrosis was observed in PCL, PU, and blended PCL/PU scaffolds.
The histopathological study of co-electrospinning PCL/PU nanofibers
Edema
Results of pathology evaluation of the fabricated PCL/PU co-electrospinning nanofibers.

mild (+), moderate (++), sever (+++), very severe (++++), and absence (−).
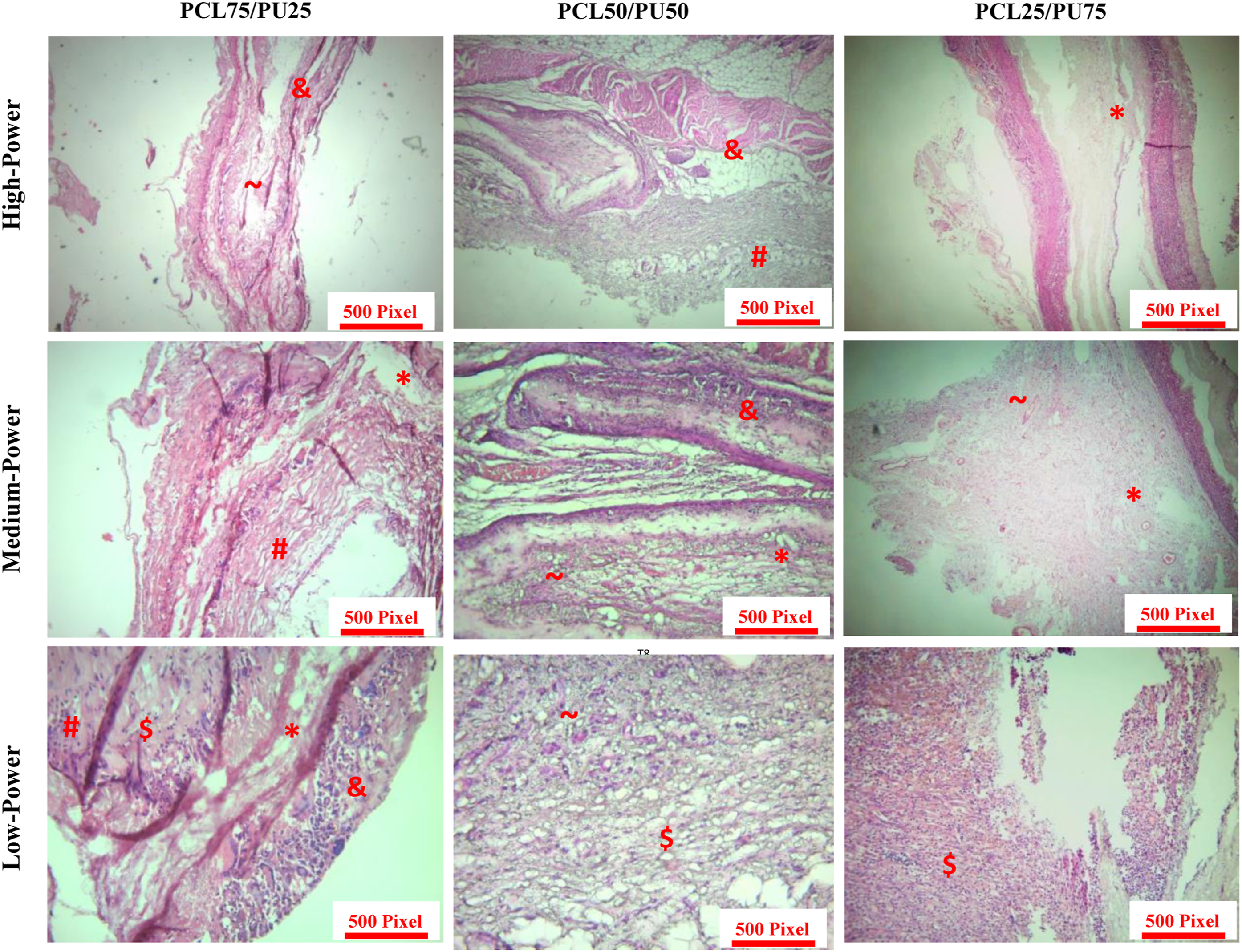
Histopathological assessment of co-electrospinning polycaprolactone/polyurethane scaffolds by hematoxylin and eosin staining. On days 45 after surgery, randomly a group of rats was selected and sacrificed. *: Edema, #: Fibroblastic change, &: Foreign body granulomatous reaction, $: Inflammation, ∼: granulation tissue.
Foreign body granulomatous reaction
As you can see in Table 4, the rate of foreign body giant cells is mild for neat PCL nanofibers and moderate for other groups. In other words, the presence of PU in the co-electrospinning PCL/PU scaffolds has played an important role in determining foreign body granulomatous reaction parameters in PCL/PU scaffolds (Figure 6).
Granulation tissue formation
According to Figure 4, in the neat nanofibers, the granulation tissue formation parameter was reported mild to moderate (for PCL) and absence (for PU), respectively. In the co-electrospinning PCL/PU scaffolds, the mentioned parameter was evaluated mild for PCL75/PU25, and moderate for PCL50/PU50 and PCL25/PU75 nanofibers. (Figures 4 and 6)
Inflammation
According to the obtained results in Table 4 and Figure 6, by changing the composite ratio of PU from 25 to 50 and 75% in the co-electrospinning PCL/PU scaffolds, the inflammation increased and reported mild to moderate chronic, moderate chronic, and severe acute and chronic, respectively.
Calcification
In PCL/PU scaffolds in the form of co-electrospinning nanofibers by increasing the composite ratio of PU from 25 to 50%, the calcification has changed from scant to few. In the other groups did not report any calcification (Table 4 and Figure 6
Fibroblastic change
The histopathological results (Table 4 and Figures 4 and 6) indicated that the neat PU nanofibers and co-electrospinning PCL/PU scaffolds with the more composite ratio of PU (50 and 75%) have shown similar fibroblastic change after
Necrosis
Similar to blended PCL/PU scaffolds, no necrosis was observed in any group of co-electrospinning PCL/PU scaffolds. (Figure 6)
Discussion
The main objective of this study was to investigate the
Electrospinning by employing the strong electric field can convert a wide range of suitable biopolymer fluids into fibers with diameters in the scale from nanometers to microns. 32
Blend-electrospinning is based on mixing polymeric solution in the similar solvent prior to the electrospinning process and spinning by one syringe. The blend-electrospinning as a simple and effective method leads to favorably blended different composite ratios with attractive properties to overcome limitations and drawbacks associated with the PCL and PU in form of single polymers. In this method, it is possible to load drugs in a dissolved polymeric matrix upon favorable conditions. 33 In contrast, the co-electrospinning method is based on dissolving the polymers in the special solvents, and in following, they spun by separated syringe. In this method, the unique properties of each polymer are protected during the co-electrospinning process. In the present study, the blend-electrospinning and co-electrospinning methods of PCL/PU scaffolds were done successfully in nanometer scale, without any beads, and with random distribution and optimum porosity. In the co-electrospinning PCL/PU scaffolds, a smaller average fiber diameter was observed than blended PCL/PU scaffolds. This phenomenon could be related to the stronger electric field in the co-electrospinning method, because in the abovementioned method, each polymer solution is affected by the electric field individually. The stronger electric field can help to fabricate the smaller fiber diameters which are in agreement with the reported results by Yang et al. 34
The successful electrospinning of PCL and PU was confirmed by FTIR spectroscopy. Similar to obtained results in another research, the PCL and PU characteristic peaks in hybrid PCL/PU nanofibers peaks were confirmed. 35
Edema introduces fluid retention or swelling caused by the leak of fluid of the small blood vessels into nearby tissues.
36
According to histopathological results, in co-electrospinning PCL/PU scaffolds with 25 and 75% of PCL, higher exudate absorption capacity (higher edema) was seen. According to the long time duration of the
Inflammation is introduced as a part of the complex biological response of body tissues against harmful stimuli, such as pathogens, damaged cells, or irritants. The inflammation process is a protective response which consists of immune cells, blood vessels, and molecular mediators. 45 The function of inflammation is to eliminate the initial cause of cell injury, clear out necrotic cells and tissues damaged from the original insult and the inflammatory process, and initiate tissue repair. It is reported that structures with nano-scale features possess surface nano-topography and influence inflammatory cell response. 46 In the rate of inflammation, a similar condition was observed among blended and co-electrospinning PCL/PU scaffolds.
Calcification is the accumulation of calcium salts in body tissue and normally occurs in the formation of bone, but calcium can be deposited abnormally in soft tissue, causing it to harden. 47 It is reported that calcification is one of the most common issues that arise concerning biocompatibility, and the naturally occurring state of the body, as well as when it exists as a result of biomaterial implants. Our results confirmed more calcification in the co-electrospinning hybrid PCL/PU nanofibers compared to blended PCL/PU scaffolds. Necrosis is a form of cell injury which results in the premature death of cells in living tissue by autolysis. Necrosis is caused by factors external to the cell or tissue, such as infection, or trauma which result in the unregulated digestion of cell components. 48 No necrosis was observed in any of the neat and hybrid structures (blended and co-electrospinning PCL/PU scaffolds).
Conclusion
The native ECM has a composite structure. Hence, fabrication of the biocompatible synthetic nanofibers scaffolds (similar to native ECM) for improvement in the
Footnotes
Acknowledgment
The authors also gratefully acknowledge of the University of Sistan and Baluchestan, Mashhad University of Medical Sciences
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute for Medical Research Development (NIMAD), for supporting this research (Grant No. 932/2/1006 and 931488).
