Abstract
This study deals with the temperature adjusting performance of thermoregulated woven fabric based on phase-change microcapsules in low-temperature environment. Phase-change microcapsules containing n-octadecane (MicroC18) with melamine–urea–formaldehyde as shell were synthesized by an in situ polymerization using styrene maleic anhydride copolymer as emulsifying agent. Surface morphology, chemical structure, and thermal properties of MicroC18 were, respectively, characterized using field emission scanning electron microscope, Fourier transform infrared spectroscopy, X-ray diffraction, differential scanning calorimetry, and thermal gravimetric analysis. The results indicate that a series of microcapsules with spherical shapes were fabricated with about 20.6-μm weight-average particle size. Latent heat is about 188.2 J/g and encapsulation efficiency of n-octadecane (C18) is 85.2%. Phase-change microcapsule composite fabric was prepared through foaming method with plain weave, twill weave, and satin weave as substrates. Thermal insulation property, low-temperature resistance, air permeability, and mechanical property of the finished fabric were investigated. The results show that the cooling rate of finished fabric is significantly slower, and low-temperature resistance time increases. Finished satin fabric has the best thermal resistance performance. The air permeability of finished fabrics is lightly reduced, and final elongation in warp and weft are increased by 16.5% and 15.2%, respectively.
Introduction
Ultra-low-temperature protection technology has important applications in the fields of aerospace, biology, and medicine. It is a generally accepted traditional method to improve the low-temperature protection performance by increasing the density and thickness of materials, whereas by which the using effects are significantly affected. However, the development of phase-change materials (PCMs) 1 and microcapsule 2 technology makes an innovative solution to this dilemma.
PCMs absorb, store, and release large amounts of latent heat over a defined temperature range during phase changes 3 and thus the application of which in thermal energy storage has been well known in many fields.4,5 In common PCMs, n-octadecane (C18) is recognized as the most frequently used PCM due to its advantages in low costs, low corrosivity, and better environmental protection.6,7 Microencapsulation is the technology of packaging micronized materials in capsules with the size ranged from less than 1 μm to more than 1000 μm. Microencapsulated PCMs (microPCMs) have been rapidly developed in recent 30 years and widely used in aerospace, textile, and other fields.8,9 Due to the smaller particle size and larger specific surface area, microPCMs exhibit relatively higher phase-change enthalpy and more remarkable effect on heat storage and temperature adjustment.10,11 The most widely accepted microcapsule shell materials include polyurethane 12 (PU), melamine–formaldehyde resin 13 (MF), and urea–formaldehyde resin 14 (UF), which possess excellent high-temperature performance, and so on.
Among them, MF is toxic due to residual formaldehyde. Melamine possesses more cross-links and higher brittleness, which limit its further development. Studies have shown that melamine–urea–formaldehyde (MUF) resin effectively solves the problem of high brittleness in MF. 15
There are many processing methods of finishing microPCM composite fabric, which mainly include dipping treatment, fabric coating, spinning process, and hollow fiber filling. 16 Foam coating possesses the advantages of thin coating film and uniform coating. At present, various aspects in finishing process of microPCM composite material and the microcapsule material17,18 are under study around the world. It has been shown that phase-change microcapsule coating significantly improves the heat storage and temperature regulation performance of the fabric. 19 However, most studies are focused on microPCM composite materials under normal temperature, whereas studies on which under low and ultra-low temperature are still in the preliminary stage. This study revealed the preparation process of MUF microcapsules containing C18 (MicroC18) as phase-change core material for thermal energy storage. MicroC18 was prepared by in situ polymerization using styrene maleic anhydride copolymer (SMA) as the emulsifying agent and analyzed by optical microscope (OM), field emission scanning electron microscope (FE-SEM), Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), differential scanning calorimetry (DSC), and thermal gravimetric analysis (TGA). The microcapsules were coated to the surface of plain, twill, and satin fabrics by foam coating. The thermal resistance performance, low-temperature resistance, air permeability, and mechanical property of finished fabrics were investigated, respectively.
Experiment
Materials
n-octadecane (C18) used as core material was purchased from Alfa Aesar, USA. Melamine (Sigma-Aldrich, USA), urea (Sigma-Aldrich, USA), and formaldehyde solution, 37 wt% in H2O (Sigma-Aldrich, USA) were used as shell materials. SMA used as emulsifying agent, sodium dodecyl sulfate (SDS) used as foaming agent, and polyvinyl alcohol (PVA) used as thickener, were obtained from Tianjin Guangfu Technology Development Co., Ltd, China. Waterborne polyurethane (WPU), supplied by Huizhou Duoli Co., Ltd, China, was used as binder agent, in order to bond microcapsules on the fabrics.
Preparation of MicroC18
The microencapsulated C18 was fabricated through in situ polymerization. The pre-polymer solution was prepared by mixing 2.1 wt% melamine, 1.3 wt% urea, 5.6 wt% formaldehyde, and 71 wt% distilled water. Then, the mixture was adjusted to the pH of 8.0–8.5 with triethanolamine and stirred at 60°C. The solution was cooled to 35°C using cold water when it was clear. After adding 10.0 wt% SMA into pre-polymer solution, the reaction solution was dispersed by XHF-D type high-speed disperser at the speed of 6000 r/min for 30 min to form stable oil-in-water (O/W) emulsion. In order to enhance the effect of emulsification, C18 was dripped into the reaction solution during the dispersing process. Subsequently, the reaction beaker was transferred to the 80°C water bath. The reaction solution was agitated with a three-bladed low-shear mixing impeller at 600 r/min for 3 h. Then, the resultant microcapsules were filtered and washed with 30 wt% ethanol aqueous solution at approximately 60°C. The wet microcapsule powder was dried in a vacuum oven at 80°C for 24 h to remove water.
Preparation of low-temperature resistance fabric
The finishing solution prepared by mixing 15.0 wt% WPUs, 10.0 wt% MicroC18, 2.5 wt% PVA, 2.5 wt% SDS, and 70.0 wt% deionized water was injected into CPU20F-Y low-pressure PU foaming machine with high foaming to make the foam uniform. Foam finishing solution was evenly coated to the surface of plain, twill, and satin cotton fabric by NM-450 vertical mill. The foaming time was 4.5 min, the foam density was 100 g/L, and the coating thickness was about 1.2 mm. Fabrics after finishing were pre-cured for 10 min at 80°C and cured for 3 min at 120°C. Foam finishing solution formed film attached to the surface of the fabric. The finishing solution compositions are shown in Table 1, and the organization chart of composite fabric is shown in Figure 1.
Test samples.
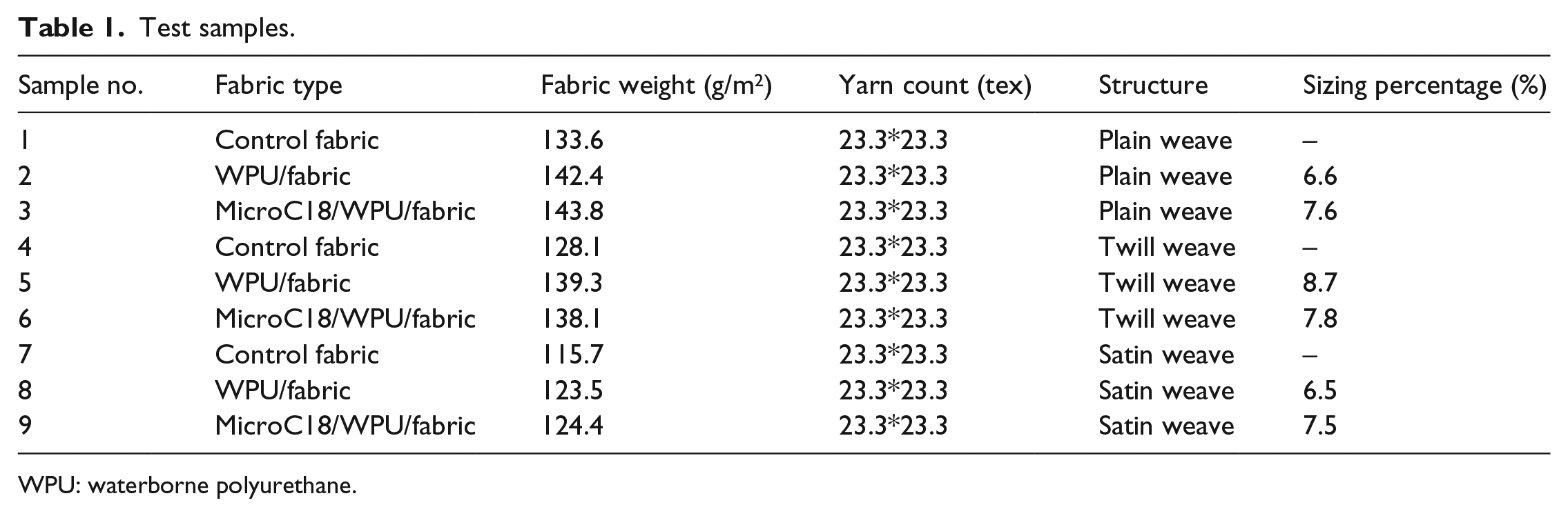
WPU: waterborne polyurethane.

Illustration of composite fabric and MicroC18.
FTIR spectra
The FTIR spectroscopic analyses of MicroC18, MUF resin, and C18 were obtained by FTIR spectrophotometer (Tensor 37 FT-IR; Bruker Corporation, Germany).
Crystallography of microcapsules
The diffraction patterns of C18 and MicroC18 were obtained using an XRD (D8; Bruker Corporation) at 20°C. The scanning range was 5°–40°. The wavelength of the X-ray was 1.5416 nm.
Morphology
The morphology of MicroC18 was obtained using Optical Microscope (BA2000; Chongqing Photoelectric Instrument Co., Ltd) and FE-SEM (S-4800; Japan Hitachi Limited, Japan). The weight-average diameters of MicroC18 were measured on the OM. More than 200 MicroC18 were counted, and the formula is shown as follows

where Dm represents the weight-average diameters of MicroC18, Ni represents the number of MicroC18, and Di represents the average diameter.
Phase-change behavior
The phase-change properties of the C18 and MicroC18 were measured by differential scanning calorimeter (DSC 200 F3; NETZSCH, Germany) in the range of −20°C to 80°C at the rate of ±10°C/min in a nitrogen atmosphere. The microencapsulation ratio of C18 in MUF was calculated with the following equation

where η is the microencapsulation ratio of C18; ΔΗµ,MicroC18 and ΔHc,MicroC18 are melting enthalpy and crystallization enthalpy of MicroC18, respectively; ΔHm,C18 and ΔHc,C18 are melting enthalpy and crystallization enthalpy of C18, respectively.
Supercooling ΔT refers to the difference between the theoretical crystallization temperature Tcrystalize and the actual crystallization temperature Tcurrent. The calculation formula is as follows

Thermal stability
Thermal stability of C18 and MicroC18 were investigated using a thermogravimetric analyzer (TGA; STA449F3; NETZSCH) at a scanning rate of 10°C/min in the range of 30°C–600°C in a nitrogen atmosphere.
Thermal resistance performance
The thermal resistance performance of control and finished fabrics was measured using Fabric Heat Preservation Performance Tester (YG606D; Shenzhen Haibin Instrument Co., Ltd, China) in ISO constant temperature and humidity. Clo is defined as follows: the room temperature is 21.1°C, the relative humidity is below 50%, the airflow is 10 cm/s (no wind), subjects were quietly at sitting position and feeling comfortable, the skin temperature is maintained at 33.3°C, and the average basal metabolism is 58.1 W/m2. Under the above conditions, the thermal resistance of the clothing worn by the subject is 1 clo.
Low-temperature resistance
Low-temperature resistance test equipment (LTRTE) was designed, and low-temperature resistance of samples 1–9 was measured by LTRTE. First, the fabric sample was made into a cylindrical shape (bottom diameter: 2.5 cm, height: 8 cm), and the probe of the temperature measuring device was inserted and sealed. The samples were placed in the constant temperature and humidity environment (temperature is 25°C and relative humidity is 50%) for 24 h. After reaching heat balance, the samples were placed in the environment of −15°C, −25°C, −40°C, and −50°C for 30 min, respectively. The low-temperature resistance curve of the sample is obtained by measuring the internal ambient temperature of the sample per minute with the temperature measuring device.
Air permeability
Air permeability of samples was measured by automatic air permeability instrument testing (YG461H; Ningbo Textile Instrument Factory, China). Six parts of each sample were taken for measurement, and the average values were obtained by calculation.
Mechanical property
Final elongation and final elongation ratio of plain fabric in warp and weft were measured by multi-function fabric machine (YG026D; Dongguan Fangyuan Instrument Co., Ltd, China) using samples with the size of 25 cm × 5 cm.
Results and discussion
FTIR spectra
The FTIR spectra of the MicroC18, MUF shell, and C18 are presented in Figure 2, in which a characteristic broad band responsible for (N–H) and (O–H) stretching vibration peak superposition was observed around 3380 cm−1. The peaks at 2960, 1460, and 1370 cm−1 presented (–CH3) antisymmetric stretching vibration, asymmetric bending vibration, and symmetrical bending vibration in C18 spectra, respectively. These assignments confirmed that the C18 was successfully encapsulated into the MUF copolymers. The peaks at 2920 and 2850 cm−1 presented (–CH2) stretching vibration. (C–H) stretching vibration and in-plane bending vibration were found at 1340 and 1490 cm−1. The characteristic absorption bands for the aliphatic (C–N) vibration appeared at 1170 cm−1, while the characteristic triazine ring bending was observed at 810 cm−1. Ether bond (C–O–C) stretching vibration peak appeared near the 1010 cm−1. The condensation polymerization took place with methylol melamine part of hydroxy (–OH) in pre-polymers and generated ether bond, which was named as crosslinked pre-polymer. Aldehyde carbonyl (C=O) was not discovered near 1720 cm−1 in the spectra, which indicated that no formaldehyde existed in MicroC18.

FTIR spectra of C18 (a), MUF shell (b) and MicroC18 (c).
Morphology
Figure 3 shows the OM image, FE-SEM images, and particle size distribution of the microcapsules. Obviously, the microcapsules prepared by SMA possessed spherical shape and smooth surface with regular pattern, as is shown in Figure 3(a) to (c). The volume effect of core material during phase change accompanied with collision and extrusion behavior between microcapsules resulted in significant depression of some microcapsules, although the microcapsules were still intact, which suggested that the elasticity and toughness of wall were both excellent. Consequently, excess urea reacted with redundant formaldehyde, and by which the crosslinking degree of resin during MicroC18 preparation was increased, thus the toughness of the composite resin was improved. According to Figure 3(d), the particle size of microcapsules was about 20.6 μm with a relatively concentrated distribution which presented normal distribution characteristics. Uniform particle size distribution improved the evenness of surface finishing and temperature adjusting performance of fabric. From Figure 3(e) and (f), the microcapsule wall was thin with the thickness of about 180 nm, which benefited heat release and diffusion.
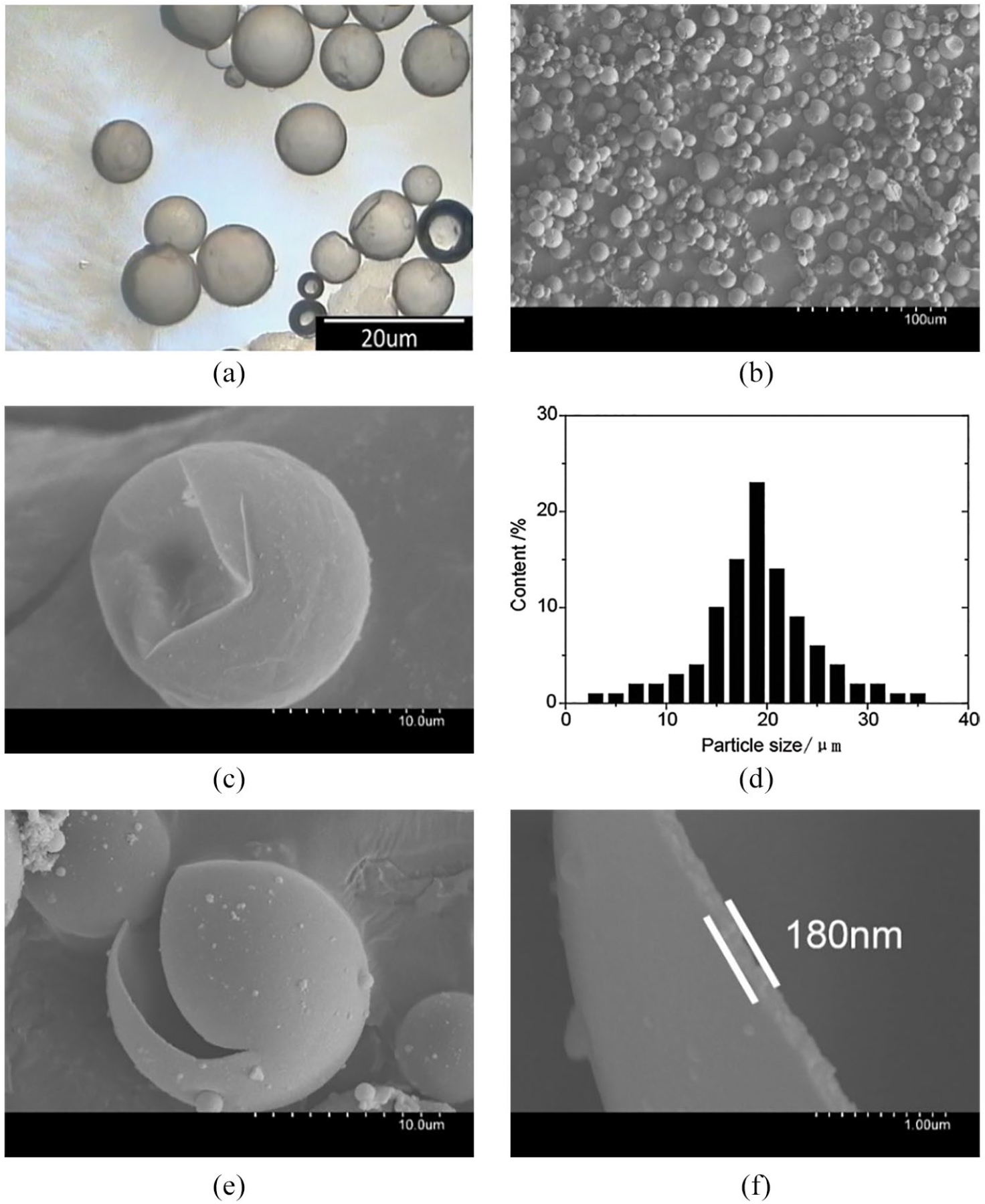
OM and FE-SEM images of MicroC18: (a) OM image (800×), (b) FE-SEM image (500×), (c) FE-SEM image (5000×), (d) particle size distribution of microcapsules, (e) FE-SEM of cracked microcapsules (5000×), and (f) FE-SEM of MUF shell (35,000×).
Crystallography of microcapsules
The crystallinities of C18 and MicroC18 were investigated by means of XRD, and their patterns are displayed in Figure 4. It was revealed that both C18 and MicroC18 were highly crystallized. It was convenient to analyze the crystallographic forms of encapsulated C18 due to the amorphous form of the MUF copolymer shell of MicroC18. The XRD pattern of C18 exhibited four characteristic peaks of triclinic (0 1 0), (0 1 1), (1 0 0), and (1 1 1) observed in the pattern of all samples at 19.2°, 19.6°, 23.2°, and 24.6°, which corresponded to the characteristic peaks of C18, respectively. In addition, the crystal structure of C18 in MicroC18 was the same as that of C18, indicating that they were both triclinic. Nevertheless, the intensities in the diffractions patterns at 6.8°, 10.2°, and 13.7° decreased dramatically after microencapsulation, which indicated that the small space of MicroC18 inhibited the motion of the C18 chains to some extent, resulting in decreased crystallinity. These results confirmed that the crystal structure of the paraffin remained unchanged during the synthesis process.

XRD patterns of C18 and MicroC18.
Phase-change properties
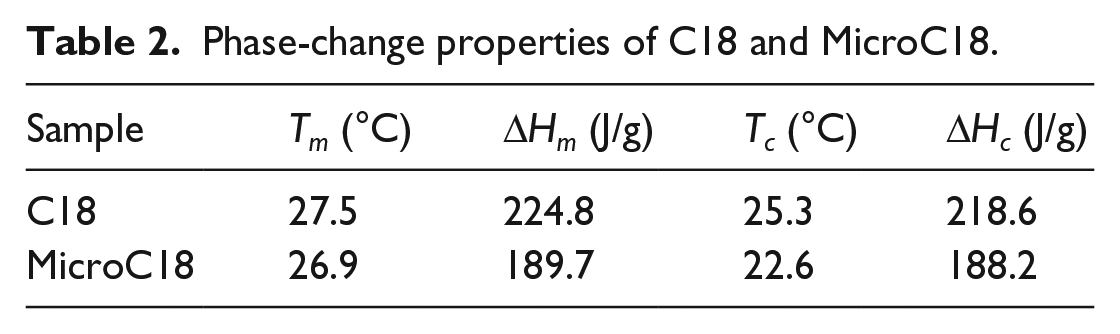
The phase-change behaviors of C18 and MicroC18 were investigated by DSC with the thermograms displayed in Figure 5. The melting and crystallization properties of C18 and MicroC18 were obtained from DSC analysis and summarized in Table 2. The phase-change behavior of MicroC18 was described with phase-change temperature, Tc and Tm, and both of which were determined by DSC analysis. As shown in Table 2, phase-transition temperatures of C18 and MicroC18 were close during raising temperature, indicating that the influence of MicroC18 on the phase-change behavior of C18 was small and the thermal conductive performance of wall was better. The supercooling temperature values of C18 and MicroC18 were 2.2°C and 4.3°C in cooling process, respectively. This was due to the fact that microencapsulated C18 was coated in small space and the crystallization way was converted into homogeneous crystallization which increased the supercooling degree. Meanwhile, phase-transition temperatures of MicroC18 and human body were relatively close, which contributed to the temperature adjusting performance of compound fabric. The microencapsulation ratio of C18 calculated by formula was 85.2%, and the coating effect was ideal.

DSC spectra of C18 and MicroC18.
Phase-change properties of C18 and MicroC18.
Thermal stabilities
The thermal stabilities of the C18 and MicroC18 were evaluated by means of TGA. The degradation data obtained from the thermal gravimetric (TG) thermograms in Figure 6(a) and (b). C18 starts to lose weight at approximately 160°C, and it completely loses its weight at 235°C (Figure 6(a)). The TG curve of C18 is one step and sharp because it is composed of linear alkane molecules with a lower decompose temperature. The TG curve had three steps for MicroC18, as shown in Figure 6(b). In the first step, MicroC18 had an initial mass loss of about 4.8%, which was due to the gasification of water and oligomer. The second step had an initial mass loss of about 38.9%, which was caused by the decomposition of MUF and C18 evolution. As the internal pressure of the core materials built up with increasing temperature, the MicroC18 burst at the particular temperature where the shell wall degraded was beyond a critical level. The third step was from 330°C to 600°C because of further thermal decomposition of MUF. From these results, it is known that the MUF shell can improve the thermal stability of the microencapsulated C18 and prevent the leakage of the melted C18. The life of the MicroC18 is important for thermal energy storage applications in textile (Table 3).

TG spectra of C18 (a) and MicroC18 (b).
TGA data for C18 and MicroC18.

Thermal insulation property
Figure 7 shows the thermal insulation property of samples 1–9. It was revealed that the finished fabrics, compared with the control fabrics, exhibited better thermal insulation property, higher coefficient of heat preservation and clo value, as well as lower coefficient of thermal conductivity. This resulted from the satin weave fabric which possessed relatively less compact structure, larger porosity, and higher still air content that greatly decreased the thermal conductivity. Plain weave exhibited excellent thermal performance due to its advantages in dense structure and warp and weft interlace points. However, the heat preservation performance was not desirable resulting from the lowest still air content. In comparison with the control fabrics, the finished samples exhibited increased thermal insulation ratio, clo value, and thermal insulation property and decreased thermal conductivity coefficient. The reason was that the WPU molecules form polymer films with low thermal conductivity on the surface of the fabric, which blocked the tiny pores on the surface of the fabric, making still air content increased inside the fabric. In addition, the thermal conductivity of WPU-treated fabric was substantially the same as that of the fabric subjected to mixed treatment, since the PCM in MicroC18 had poor thermal conductivity.

Thermal insulation property test histogram of fabric: (a) warmth retention, (b) conductivity, and (c) clo value.
Low-temperature resistance
Low-temperature resistance curves of samples 1–9 under −15°C, −25°C, −40°C, and −50°C were shown in Figure 8(a) to (i). In order to facilitate discussion, in this study, it was assumed that low-temperature resistance time of fabric was the time taken for the internal environment of fabric to be cooled down from −25°C to 0°C. From Figure 8(a), (d), and (g) and Table 4, it is shown that, low-temperature resistance performance of sample 7 reached the best in the control fabric, which was consistent with the results of thermal performance test. However, the low-temperature resistance time of sample 7 was only 2.1 min at 15°C, and the performance of the other two control fabrics was worse. Therefore, low-temperature resistance of control fabric performance was poor under the condition of low temperature.

Low-temperature resistance curves: (a) sample 1, (b) sample 2, (c) sample 3, (d) sample 4, (e) sample 5, (f) sample 6, (g) sample 7, (h) sample 8, and (i) sample 9.
Low-temperature resistance time of the fabric.

The low-temperature resistance of samples 2, 5, and 8, compared with samples 1, 4, and 7, was extended by 1.6, 1.0, and 1.5 min, respectively, in −15°C environment due to the fact that WPU formed a polymer film on the surface of the fabric, which consequently encapsulated a certain amount of still air inside the fabric, reduced the thermal conductivity, and improved the low-temperature resistance. The low-temperature resistance of samples 3, 6, and 9, compared with samples 2, 5, and 8, was extended by 2.7, 1.9, and 2.2 min, respectively, in −15°C environment, which indicated that the addition of MicroC18 improved the low-temperature resistance of composite fabrics. The reason lied in the fact that the PCMs in MicroC18 released heat at specific temperatures, which slowed down the rate of temperature drop. Compared with the samples of other two structures, the satin fabric has the longest resistance time at low temperature. This is because the satin fabric has relatively loose structure, large porosity, and high air content. After finishing the coating, it encapsulates a lot of still air and increases the thermal resistance performance of the composite fabric.
The low-temperature resistance properties of the fabric finished by WPU/MicroC18 were significantly improved, which consequently provided better protection under low-temperature during a short period of time. As the environmental temperature decreased, the low-temperature resistant time ranges of samples 3, 6, and 9 were more obviously shortened. Low-temperature resistance time of samples 3, 6, and 9 were extended by 0.9, 1.1, and 0.9 min, respectively, in −50°C environment. Obviously, the heat released by C18 phase change was relatively limited for extreme temperature. Overall, the improvement of MicroC18 on low-temperature resistance performance of fabric was more remarkable.
Air permeability
Air permeability of samples is shown in Figure 9. It is clear that sample 7 exhibited the most outstanding air permeability among control fabrics, since the structure of satin weave was loose with less interlacing points in warp and weft and more porosity. The quantity of interlacing points in warp and weft of plain weave was highest and the porosity of which was low, resulting in a relatively inferior air permeability. Among finished fabrics, air permeability of samples 3, 6, and 9 was reduced by 17.7%, 16.4%, and 17.1%, respectively. The ideal air permeability performance of finished fabrics was due to the WPU coating with thin thickness, more holes and certain permeability.

Air permeability of samples 1, 3, 4, 6, 7, and 9.
Mechanical property
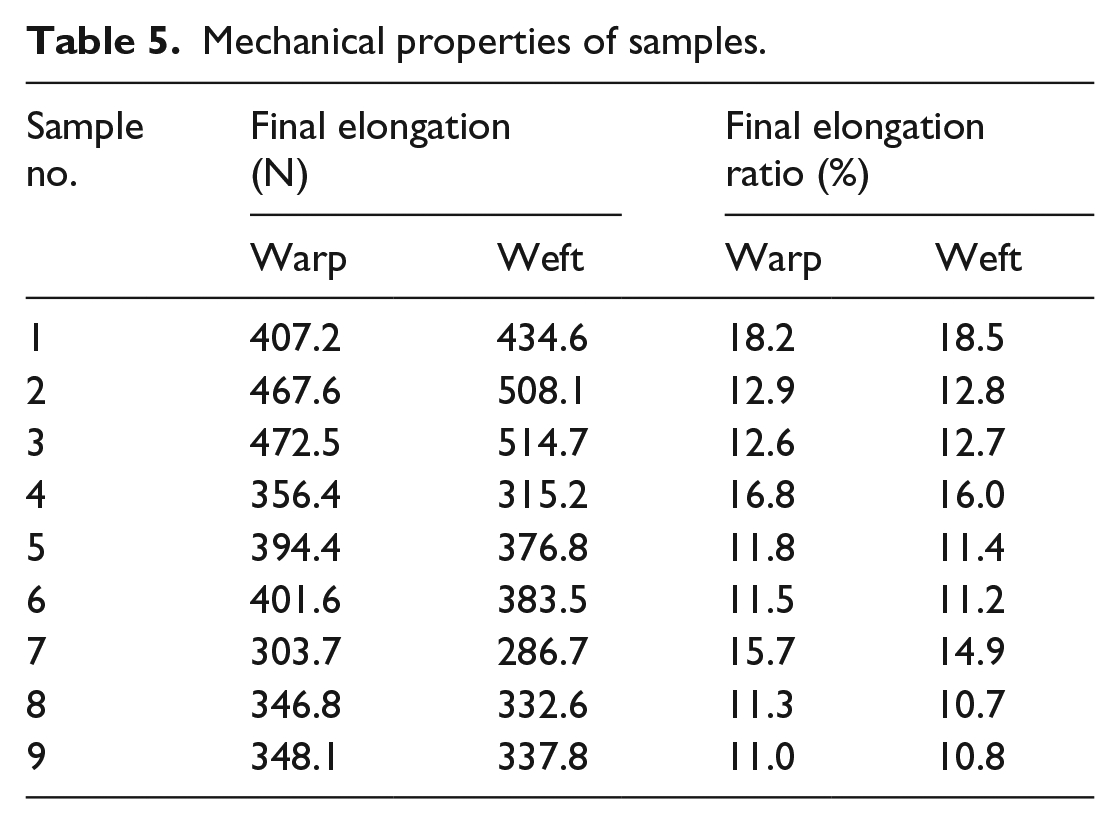
Table 5 shows the final elongation and final elongation ratio of fabric. Compared with control fabric, the final elongation of finished fabric in warp and weft increased significantly while the final elongation ratio dropped slightly. The reason was that WPU molecules in finishing fluid contained a large number of polar groups, and strong intermolecular force between them resulted in excellent film-forming property, by which a polymer membrane was established on the fabric surface. Meanwhile, part of the finishing liquid permeated into the inside of fabric, which significantly increased the final elongation of finished fabric. In addition, compared with the WPU-treated fabrics, the mechanical properties of fabrics subjected to mixed treatment were slightly reduced since the phase-change microcapsules destroyed the integrity of the polymer film to some extent. However, the reduction was not statistically significant due to the relatively small quantity. In addition, tabular fabrics exhibited the best tensile properties, followed by twill and satin.
Mechanical properties of samples.
Conclusion
Phase-change microcapsules containing C18 with MUF shell was prepared by in situ polymerization in this study. The phase-change microcapsules were spherical with relatively smooth surface and particle size of about 20.6 μm. The phase-transition enthalpy of phase-change microcapsules was 188.2 J/g in the cooling process, and the microencapsulation ratio of C18 reached 85.2%. Phase-change microcapsules can provide the fabric with regenerative thermal control performance. The heat preservation performance of finished fabric was obviously improved. The cooling rate of finished fabrics was significantly slower in low-temperature environment, and the low-temperature resistance time increased remarkably. This phenomenon was formed by combining WPU polymer film and MicroC18. Low-temperature resistance of satin fabric reached the best, and low-temperature resistance of twill and plain fabric were slightly undesirable. The air permeability of treated fabrics and elongation at break declined slightly, while the final elongation in warp and weft increased slightly. The polymer coating containing MicroC18 significantly improved the temperature regulation of fabrics in low-temperature environment and can be widely used in outdoor work and activities.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded, in part, through a grant by Tianjin Research Program of Application Foundation and Advanced Technology (No. 15JCZDJC38400).
