Abstract
One of the greatest challenges in the bone remodeling is to fabricate the structure resembling the extracellular matrix. This research aims to fabricate a novel bone scaffold comprising polyurethane (PU) added with almond nanofibers via electrospinning technique. The PU/almond oil nanocomposites showed smaller fiber diameter (629 ± 148.92 nm) compared to the pristine PU (890 ± 116.91 nm). The interaction of PU with almond oil was confirmed in the infrared spectrum by the strong formation of a hydrogen bond. The wettability analysis showed that the prepared PU/almond oil nanocomposites were hydrophobic in nature (107° ± 1) compared with the pure PU (100° ± 0.5774). Thermal analysis revealed enhancement of the thermal stability with the addition of the almond oil. The addition of almond oil into the PU matrix increased the surface roughness and blood compatibility properties. Further, the fabricated PU/almond oil nanocomposites showed less toxicity to red blood cells (RBCs), as indicated in the hemolytic assay. Hence, the novel fabricated scaffold possesses better physicochemical properties and is nontoxic to the RBCs, which may be utilized for bone tissue regeneration.
Introduction
The greatest challenge of bone tissue engineering is the repair of the damaged bone tissue affected by trauma, disease, or congenital malformations. The small bone defects would heal naturally, but the critical size defects would take a long time to heal. 1 In bone tissue engineering, the repair of the damaged bone tissue is done by improving the health of musculoskeletal tissue by substituting for the living bone graft. 2 The substitute used for the bone graft was made of materials that aid the bone formation from the surrounding tissue and also as a carrier for embedded bone cells. 3 The use of these methods was limited in the biomedical applications owing to certain disadvantages like donor scarcity, supply limitation, infections, and immune rejection. 4 With advancements in technology, the tissue-engineered scaffold emerged as an alternate for these methods to overcome these limitations. An ideal bone scaffold used for the bone regeneration must resemble the native function of the extracellular matrix (ECM). Moreover, it should have other necessary characteristics like nontoxic, biodegradable, biocompatible, porosity, and facilitating the osteoblast cell adhesion and growth. 5,6
In tissue engineering applications, one of the increasing interests is developing a fibrous scaffold resembling the native structure of the ECM. Recently, the electrospinning technique is emerging as a prominent technique to develop a fibrous structure. Electrospinning technique involves different adjustable process parameter that facilitates the formation of a structure with the required morphological and topographical features. 7 The ultimate aim in the biomedical research areas is to fabricate a nanofibrous-based 3-D scaffold that resembles the ECM. Electrospinning is a versatile method, which produces nanofibers from the polymeric materials in the nanoscale range resembling the ECM matrix. Nowadays, the nanofibers were widespread in tissue engineering applications because of its porous structure with large surface area to volume ratio. 8 Further, the nanofibers fabricated through electrospinning technique were reported to facilitate gaseous exchange, nutrients transport, cell adhesion, and proliferation to support the tissue regrowth. The present study employs Tecoflex EG-80A (Lubrizol, Wickliffe, Ohio, USA), a medical-grade polyurethane (PU), to fabricate the bone scaffold. PU possesses desirable characteristics such as biodegradable behavior, biocompatibility, and good mechanical properties, which makes them the most commonly used polymer in biomedical applications. 9,10
In a human body, bones are connected together by joints. When there is a bone fracture, inflammation occurs at the wound site, creating pain immediately. In Ayurvedic medicine, plant oils are applied to the joints to reduce pain. In a recent study, Jaganathan et al. fabricated a scaffold based on polymer loaded with various plant oils, such as corn, neem oil, olive oil, and grape seed oil for bone tissue repair. 11 -13 From this literature, it was evident that the oils play a key role in bone tissue repair. In this study, almond oil was selected. The almond oil is rich in fatty acids, carbohydrates, and proteins. It possesses a highly nutritional source of vitamins and minerals, which make them widespread in medical applications for health benefits. 14 Further, it also possesses more vitamin B complex and zinc content, which support and maintain the skin in healthy condition. The almond oil was reported to possess many multifaceted properties, such as anti-inflammatory, immunity-boosting, and anti-hepatotoxicity. 15 -17 For the first time, we had analyzed the role of almond oil with PU for bone tissue repair applications. The scaffolds used in tissue engineering applications should be nontoxic and also should not cause infection. Further, it should also support cell adhesion and proliferation. Almond oil is reported to possess nontoxic and anti-inflammatory behavior. 14,18 Hence, the developed scaffold would be nontoxic and also render infection-free environment at the site. Since the almond oil also contains a rich source of antioxidants, it would help the cells damaged from free radicals suitable for cell survival and growth. 19 The coagulation assessments play the main role in determining the application of the fabricated material. When the materials contact with the blood, immediate absorption of plasma proteins will occur on the surface. In extension, it might lead to platelet surface interaction, which may cause thrombus formation resulting in material failure. 20 In this study, the PU and PU/almond oil membrane was fabricated using electrospinning technique, and their physicochemical characteristics and blood compatibility assessments were studied.
Experiment
Materials
The following materials were used for the experimental study: Tecoflex EG-80A pellets from Lubrizol (Wickliffe, Ohio, USA), almond oil from AEON (Johor, Malaysia), dimethylformamide (DMF) from Merck (Burlington and New Jersey, USA), phosphate-buffered saline, sodium chloride (NaCl) physiological saline (0.9% w/v) from Sigma-Aldrich (Malaysia), and coagulation assay reagents from Diagnostic Enterprises (Thiruvananthapuram and Kerala, India).
Methodology
In this research, PU homogeneous solution was prepared at a concentration of 9 wt%. For almond oil homogeneous solution, the weight fraction used was 2 wt%. The prepared almond oil solution was added to PU solution at a ratio of 8:1 (v/v) and stirred for 2 h maximum for even dissolution. Firstly, PU was electrospun for different weight concentration and optimized based on the morphology before choosing 9 wt% for further studies. For PU/almond oil, we had spun three volume ratios before arriving 8:1 v/v. The other ratios either yielded beaded fibers or spitting of the polymer/oil solution. The chosen 8:1 v/v displayed the uniform formation of nanofibers without beads and spitting of the composite solution. Further, they displayed significant differences in the fiber diameter and contact angle values compared to the pure PU.
Fabrication of scaffolds
Electrospinning technique was used to convert the prepared homogeneous solution into scaffolds. The electrospinning parameters were set constant for all solution as follows: (1) flow rate: 0.5 ml/h, (2) applied voltage: 10.5 kV, and (3) distance: 20 cm. The collected fibers on the aluminum foil were dried in vacuum to remove any residual content of DMF.
Physicochemical characterization
Scanning electron microscopy analysis
Scanning electron microscopy (SEM) unit (Hitachi Tabletop TM3000, Tokyo, Japan) was utilized to detect the surface morphology and to calculate the diameters of the electrospun membranes. Before scanning was performed, the samples from the electrospun membranes were gold plated at 20 mA for 2 min. The average diameters were measured using ImageJ by choosing 30 individual fibers randomly in the captured images.
Fourier transform infrared spectroscopy analysis
The fibrous membranes were inspected in Fourier transform infrared spectroscopy spectrometer (Nicolet iS 5, Thermo Fischer Scientific, Waltham, MA, USA) to analyze the characteristics chemical bands present. The membranes were examined over the wavelength of 600–4000 cm−1 with a resolution of 4 cm−1. The attenuated total reflectance (ATR) crystal used was the zinc selenium.
Contact angle measurements
Wettability of the electrospun membranes was determined through video contact angle optimum contact angle unit (AST Products, Inc., Billerica, MA, USA). Initially, a water droplet with a size of 0.5 μl was deposited on the surface of the electrospun membranes. After a few seconds, the static image of the droplet deposition was recorded using a video camera. The experiment was repeated for three trials, and the manual contact angle was measured using computer-integrated software.
Atomic force microscopy analysis
Surface analysis was performed in an atomic force microscopy (AFM) unit (NanoWizard®, JPK Instruments, Berlin, Germany) under a normal atmosphere to measure the roughness of the developed fibrous membrane. The membranes were scanned in 10 × 10 μm2 size, and using JPKSPM software, a digital 3-D image with 256 × 256 pixels was captured.
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was conducted in a Perkin Elmer (Waltham, MA, USA) instrument to evaluate the degradation behavior of the fibrous membranes. The heating was given to the samples (3 mg) placed in an aluminum pan under nitrogen atmosphere in the temperature range of 30–1000°C at a rate of 10°C min−1.
Coagulation measurements
Activated partial thromboplastin time and prothrombin time measurements
The intrinsic pathway of pristine PU membrane and PU/almond oil nanocomposite was analyzed through activated partial thromboplastin time (APTT). For APTT, the samples were mixed with 50 μl of platelet-poor plasma, followed by incubating 50 µl of rabbit brain cephalin for 3 min and then added with 50 µl of calcium chloride (CaCl2). Finally, the clot formation was investigated by stirring the mixture using the needle and the time taken for the formation of clot was measured using a chronometer. On the other hand, an extrinsic pathway of pristine PU membrane and PU/almond oil nanocomposite was measured through prothrombin time (PT). For PT assay, the samples were added with 50 μl of platelet-poor plasma, followed by incubation with 0.9% NaCl-thromboplastin (factor III, 100 ml)-containing calcium (Ca2+) ions. Finally, the clot formation was analyzed by string the mixture using the needle and the time taken for the formation of clot was measured using chronometer. 12
Hemolysis assay
Initially, the samples were soaked in physiological saline (0.9% w/v; 37°C, 30 min) and exposed to 3 ml of diluted blood. The positive and negative controls were made by mixing human blood into distilled water in the ratio 4:5 (complete hemolysis) and physiological saline, respectively. After that, the samples were removed and centrifuged at 1000 r min−1 for 5 min. Finally, optical density was measured at 545 nm for the resultant supernatant, which indicates the RBC damage. The percentage of hemolysis or hemolytic index was calculated, as reported previously. 12
Statistical analysis
In this study, the experiments performed were carried out thrice independently. Unpaired
Results and discussion
SEM analysis
SEM images of fibrous PR membranes and their blends are displayed in Figure 1(a) and (b). SEM result shows that the fabricated PU nanofibers and their composites showed randomly oriented fibers without any bead formation. Further, reduction in fiber diameter was observed in the PU/almond oil nanocomposites than the pristine PU. The average fiber diameter of electrospun PU and PU/almond oil nanocomposites was found to be 890 ± 116.91 nm and 629 ± 148.92 nm, respectively, and their fiber distribution curves are displayed in Figure 2(a) and (b). Ngiam et al. electrospun poly(lactic-

SEM images of (a) PU membrane and (b) PU/almond oil nanocomposites.

Fiber diameter distribution of (a) PU membrane and (b) PU/almond oil nanocomposites.
FTIR analysis
Urethane is reported to comprise of four common elements like carbon, hydrogen, nitrogen, and oxygen. For the name urethane, it should have the molecular urethane linkage (NHCO2). The real chemistry behind is having a series of block copolymers with segments of hard and soft phases. The hard phase may be aromatic or aliphatic, and the soft phase may be polyether or polyester. 22 -25 Figure 3 depicts the infrared spectra of fibrous PU and PU/almond oil nanocomposite. The major peaks present in the PU membrane were 3323, 2939, 2853, 1730, 1703 cm−1, respectively. The band shown at 3319 cm−1 was attributed to N–H (aliphatic primary amine) peak and its vibrations occurred at 1597 and 1531 cm−1. The presence of C–H (alkane groups) band was seen at 2938 and 2855 cm−1, while the band at 1413 cm−1 represents its vibrations. Further, the sharp peak seen at 1728 and 1703 cm−1 denotes the C=O (carbonyl groups) band and the peaks at 1220, 1104, and 770 cm−1 correspond to the C–O (carbonyl group) with respect to alcohol group. 11 -13 In the spectra of PU/almond oil nanocomposite, no additional peaks were found, but the PU peak intensity was altered (decreased) with a blending of almond oil owing to strong hydrogen bond formation. Always there is a good probability for the formation of interhydrogen bond between two different macromolecular species rather than between the molecules of the same polymer. 26 Hence, the linking of CH and CO molecules present in the PU with the molecules of almond oil might result in the formation of the hydrogen bond. The formation of hydrogen bond insinuated the interaction between PU and almond oil. 27
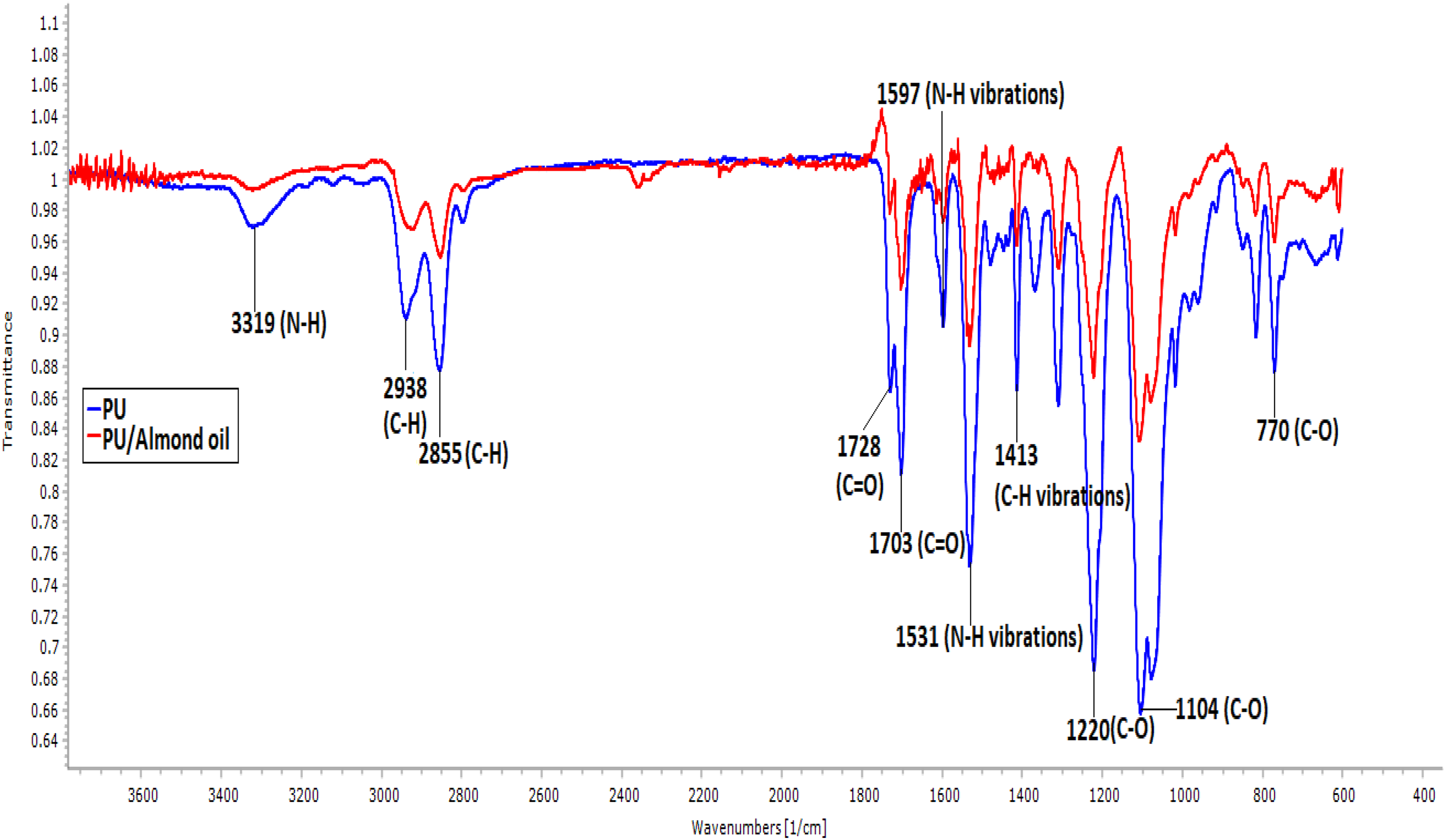
IR spectrum of PU membrane and PU/almond oil nanocomposites.
Wettability behavior
The surface wettability of the fibrous membranes was calculated through water contact angle measurements and their images are indicated in Figure 4. From the obtained results, the pristine PU fibrous membrane showed an average contact angle of 100 ± 0.57°, and it was increased to 107 ± 1° for PU/almond oil nanocomposites. Hence, the blending of almond oil decreases the wettability of PU revealing hydrophobic behavior. Jose and Alagar fabricated bone scaffold comprising polyoxymethylene (POM) added with hydroxyapatite nanoparticle and noted that the developed POM/hydroxyapatite membrane exhibited hydrophobic nature. Further, they exhibited more absorption of proteins compared to neat POM and suggested a suitable candidate for bone tissue engineering. 28 Hence, the developed PU/almond nanocomposites displaying hydrophobic nature may facilitate the enhanced absorption of proteins suitable for bone tissue engineering.

Contact angle images (a) PU membrane and (b) PU/almond oil nanocomposites.
Thermal analysis
Thermal stability of the fibrous membranes was presented. TGA results clearly revealed that the thermal behavior of PU/almond oil nanocomposites was higher compared to the pristine PU membrane. The initial onset degradation temperature for PU membrane was seen at 276°C, and it was increased to 284°C in PU/almond oil nanocomposites denoting the enhanced thermal stability because of the presence of almond oil in the pristine PU. Ayyar et al. fabricated PU/indhulekha nanocomposites for biomedical applications and reported that the blending of indhulekha into the PU matrix resulted in improved thermal stability, which resembles our findings. 29
Surface analysis
Figure 5(a) and (b) presents the AFM images of the developed fibrous PU and their blends. The calculated surface roughness of the electrospun neat PU and PU/almond oil nanocomposites was observed to be 313 and 321 nm, respectively. Hence, the blending of almond oil into the PU matrix resulted in the increase in the surface roughness, which might be due to constituents of almond oil in the PU matrix. Tijing et al. fabricated PU scaffold added with multiwalled carbon nanotube (MWCN) for tissue engineering applications. It was reported that the addition of MWCN into PU resulted in the enhanced surface roughness, which correlates with our observations. 30

AFM images of (a) PU membrane and (b) PU/almond oil nanocomposites.
Blood compatibility investigation
Figures 6 and 7 depict the blood compatibility assessments of electrospun membranes through APTT and PT assay. The coagulation assessments revealed that the blending of almond oil delayed the blood clotting time of the pristine PU. The APTT registers the blood clotting time of PU/almond oil nanocomposites as 185 ± 2.646 s, while for pristine PU, it was observed as 175.3 ± 4.041 s. Similarly, in PT assay, blood clotting time of PU/almond oil nanocomposites was observed as 82 ± 2 s, while for pristine PU, it was observed to be 76 ± 2.517 s. Further, to analyze the osmotic stress of the fabricated membranes, the hemolysis assay was carried. From the results of hemolysis, it was noted that the fabricated PU/almond oil nanocomposites showed low hemolytic index indicating less toxic to RBCs, as shown in Figure 8. The hemolytic percentage of PU membrane was observed to be 2.48%, and for developed PU/almond oil nanocomposites, it showed only 1.54%, which is considered as a nonhemolytic material. 11 -13 The blood compatibility of the developed material is influenced by multiple surface properties, such as fiber diameter, wettability, surface roughness, surface energy, and surface tension rather than a single factor. 31 Jaganathan et al. studied the blood compatibility properties of electrospun PU added with carotino oil. It was reported that the PU/carotino oil membranes showed improved anticoagulant nature than the pure PU because of an increase in surface roughness. 32 In another study, scaffold based on PU was added with sesame oil, honey, and propolis. The addition of sesame oil improved the anticoagulant nature of the pure PU. They concluded that the reason for improved blood compatibility behavior was due to smaller fiber diameter and hydrophobic behavior of the electrospun composites. 33 In our study, the delayed blood clotting time might be due to the reduced fiber diameters, hydrophobic nature, and increased surface roughness of PU/almond oil nanocomposites.

APTT assay of PU membrane and PU/almond oil nanocomposites. Asterisks indicate mean differences were significant compared with pure PU (

PT assay of PU membrane and PU/almond oil nanocomposites. Asterisks indicate mean differences were significant compared with pure PU (
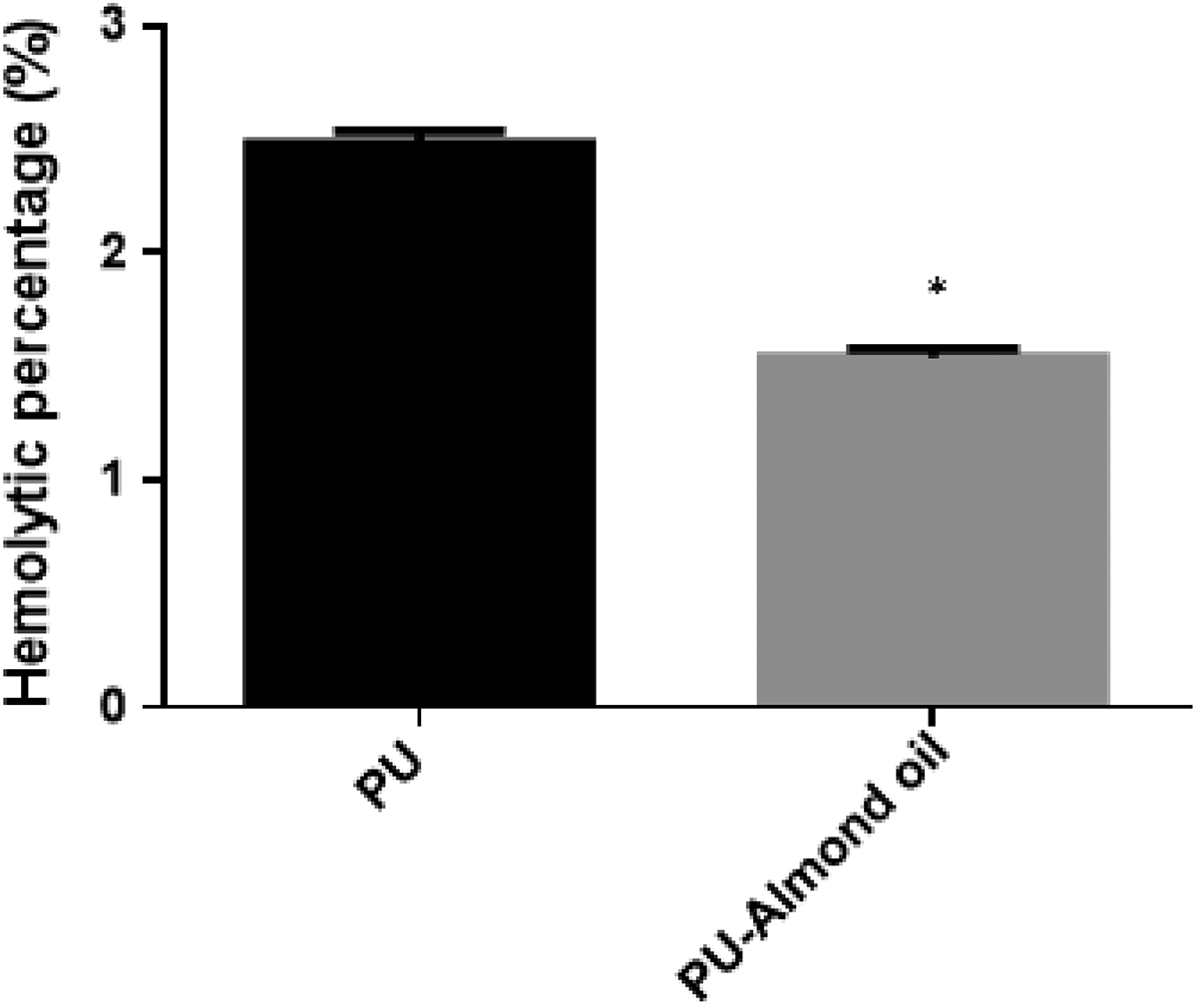
Hemolytic assay of PU membrane and PU/almond oil nanocomposites. Asterisks indicate mean differences were significant compared with pure PU (
Conclusion
The present study summarized the physiocochemical characterization and blood compatibility assessments of the fabricated PU and PU/almond nanocomposites. The PU/almond oil nanocomposites showed smaller fiber diameter compared to the pristine PU. The interaction of PU with almond oil was confirmed in the infrared spectrum by the strong formation of a hydrogen bond. The wettability analysis indicated the hydrophobic nature of the prepared PU/almond oil nanocomposites than the pure PU. Thermal analysis revealed the enhancement of the thermal stability with the addition of the almond oil. The addition of almond oil into the PU matrix increased the surface roughness and blood compatibility properties. Further, the fabricated PU/almond oil nanocomposites showed less toxicity to RBCs, as indicated in the hemolytic assay. Hence, the novel fabricated scaffold possesses better physicochemical properties and is nontoxic to the RBCs, which may be utilized for bone tissue regeneration. The addition of almond oil resulted in the improvement of the physicochemical and biocompatibility properties. However, the almond oil contains various biological components, and it was difficult to predict which component facilitated the improvement of the physicochemical and biocompatibility properties. In the future, it would be interesting to investigate the mechanical properties of the fabricated composites for its suitability in bone tissue regeneration.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
