Abstract
In Ayurveda, pure ghee is used as one of the components for the treatment of pain caused by the bone fracture. In this study, novel nanocomposites based on polyurethane (PU), ghee and propolis were developed using the electrospinning technique. Morphology analysis revealed that the novel developed PU/ghee (817 ± 138.39 nm) and PU/ghee/propolis (576 ± 144.96 nm) nanocomposites possessed smaller fiber diameter than the pure PU membrane (890 ± 116.911 nm). The presence of ghee and propolis in PU was confirmed by hydrogen bond formation as revealed in Fourier infrared analysis. The contact angle was found to be increased in PU/ghee (122° ± 1°) indicating the hydrophobic nature while the angle was decreased in PU/ghee/propolis (55° ± 1°) suggesting the hydrophilic nature. The surface roughness of electrospun nanocomposites was found to decrease while thermal stability was observed to be enhanced for the nanocomposite as indicated in atomic force microscopy and thermogravimetric analysis. Further, the blood compatibility assessments revealed the delayed activation of blood clots in the electrospun scaffolds suggesting better blood compatibility. Moreover, the hemolytic assay suggested the developed nanocomposites (PU/ghee – 1.61% and PU/ghee/propolis – 1.52%) as non-hemolytic material and also enhanced safety to RBCs. The cytocompatibility studied revealed the better proliferation rates of human fibroblast cells in the developed nanocomposites (PU/ghee – 148.3% ± 6.028% and PU/ghee/propolis – 156.7% ± 5.686%) indicating its non-toxicity. So, the newly developed PU/ghee and PU/ghee/propolis nanocomposites were found to possess desirable characteristics such as better physicochemical, improved blood compatibility and non-toxic behavior which might be utilized as an appropriate candidate for bone tissue engineering.
Introduction
The growth in the technology of biomedical engineering promoted improvement in tissue engineering. The tissue engineering gained the attention of many and more researchers recently [1]. Now-a-days, the researchers focused on bone tissue engineering to remodel the bone defects due to trauma, infection or tumor [2]. In biomedical applications, bone transplantation is the only method to remodel bone defects. The methods used in the bone transplantation were autogenic, allogenic and xenogenic [3]. The use of autogenic was limited in bone transplantation owing to limited in donor supply, donor site injury and complications in harvesting into a required shape. Compared to the autogenic, the allogenic and xenogenic possess many advantages such as rich sources of supply and easy harvesting. But they possess certain disadvantages such as causing immunogenicity and infection [4–7]. Because of these limitations, researchers are forced to find alternate methods.
In a recent study, an electrospun scaffold based on polycaprolactone (PCL) incorporated with gelatin nanofibers was fabricated for bone tissue engineering application, and the prepared scaffold showed enhanced osteogenic differentiation of human adipose-derived stem cells [8]. Moreover, some researchers reported that the scaffold containing growth factors promoted enhanced cell adhesion and proliferation. For example, in a recent study, an electrospun scaffold based on silk containing bone morphogenetic protein 2 (BMP-2) was fabricated. It was observed that the scaffold with BMP-2 showed enhanced deposition of calcium deposition and suggested a suitable candidate for bone tissue engineering [9]. From the literature, it was observed that the scaffold plays an important role in bone tissue engineering which facilitates the osteogenic differentiation for effective remodeling of damaged bone tissue. There are different methods to fabricate a scaffold, but it was observed that the scaffolds fabricated via electrospinning technique has gained wide attention in biomedical applications because of the nanofibrous nature which resembles the native extra cellular matrix (ECM) matrix [10]. Further, the scaffold with hydrophilic behavior plays an important role in bone tissue engineering which supports the improved adhesion and proliferation of osteoblast cells.
Electrospinning is a versatile technique capable of converting polymer solution into fine nanofibers [11]. The nanofibers obtained through the electrospinning process were observed to be widely used in clinical applications owing to its high surface area with interconnected pore network [12]. Moreover, nanofibers help in increasing the response of cell response in terms of adhesion and proliferation [13]. In bone tissue engineering, the biodegradable polymers were often used to develop porous scaffold. Recently, polyurethane is one of the biodegradable polymers which has been widely used in tissue engineering applications. In this study, the medical grade Tecoflex EG-80A was used for developing a bone scaffold. PU was widely used in tissue engineering applications and it was reported that the spinning of PU to obtain nanofibers was very easy owing to its flexibility [14,15].
Joint is a place in which two bones meet and connected together. When a bone fracture occurs, these joints become inflamed and pain is created at the fracture site. To reduce the pain, different substances were applied to the joints. Substances such as ghee, banana, turmeric, dried ginger and castor oil (http://www.findhomeremedy.com/natural-cure-for-joint-pain/) were the widely chosen candidates in Ayurvedic medicine. In this research, ghee was selected to combine with the PU for fabricating the nanocomposites. Ghee is a class of butter that arose from the Indian subcontinent which contains a high amount of fats and vitamins like E and A. It was used in the Ayurveda medicine along with sesame oil, muscle fat and bone marrow. Further, it was used for curing constipation and ulcers in Ayurvedic medicine [16]. Further, to improve the properties of the PU, the propolis was utilized in this work. Propolis looks like a resinous substance that was extracted by the bees from their salivary extracts [17]. The composition of propolis has numerous biological constituents such as flavonoid, phenolic components, amino acids and some inorganic compounds [18]. Moreover, it was reported to have many medicinal properties such as highly adhesive, antibacterial, antifungal, antiviral, antioxidant and anti-inflammatory activities [17]. Kim et al. fabricated PU scaffold incorporated with propolis for biomedical applications and it was reported that the addition of propolis improved the hydrophilicity nature of the polyurethane [19]. Hence, in this study, the propolis was added with ghee to tailor the surface wettability features. In biomedical applications, the coagulation assessments are one of the main factors which chooses the application of the fabricated materials. When the blood touches the materials, there might be rapid absorption of plasma proteins which enables the platelet surface interaction. The platelet surface interaction might result in thrombus formation which leads to the failure of the fabricated material [20]. In this article, blood compatibility assessments of electrospun membranes were investigated. In this study, PU scaffold incorporated with ghee and propolis were fabricated using the electrospinning technique. Further, the combined effect of ghee and propolis on improving the properties of PU was demonstrated. For the developed PU, PU/ghee and PU/ghee/propolis nanocomposites, the physiochemical properties, blood compatibility assessments and cytocompatibility studies were performed to determine its feasibility for bone tissue engineering.
Materials and methodology
Materials
Tecoflex EG-80A pellets were purchased from Lubrizol. Ghee and propolis were procured from AEON, Johor, Malaysia. Dimethylformamide (DMF) was purchased from Sigma Aldrich, USA. Phosphate-buffered saline (PBS, Biotech Grade) and sodium chloride physiological saline (0.9% w/v) were obtained from Sigma-Aldrich, Malaysia. All reagents used for activated partial thromboplastin time (APTT) and prothrombin time (PT) assay were obtained from the Diagnostic Enterprises, India.
Solution preparation
First, PU (9 wt %) was prepared by dissolving 0.405 g of PU pellets in 4.5 ml of DMF and stirred overnight for complete dissolution. Similarly, ghee (4 wt %) and propolis (9 wt %) were prepared by dissolving the calculated amount of ghee and propolis in 3 ml of DMF and stirred for a maximum of 1 h. Finally, PU, ghee and propolis solutions were mixed at volume ratios of 7:2 (v/v%) (PU and ghee) and 7:1:1 (v/v%) (PU, ghee and propolis) and stirred for a maximum of 2 h for even dissolution.
Preparation of the membranes
The electrospinning technique was used to extract the nanofibers from the prepared homogeneous solution. Briefly, the prepared solution was loaded in the 10 ml of the syringe and placed in the syringe pump. The voltage was applied to attain fine nanofibers, and for all experiments, the applied voltage was 10.5 kV. Further, the flow rate and the collector distance were set as 0.5 ml/h and 20 cm, respectively. Nanofibers deposited on the aluminum foil were detached and dried under vacuum at room temperature.
Characterization
Scanning electron microscopy analysis
The obtained electrospun nanofibers were cut into a small pieces of sample and sputtered with gold. Then the gold plated samples were imaged through a scanning electron microscope unit to identify the morphology of the prepared samples. From the scanning electron microscopy (SEM) photographs, the distribution of fiber diameter was calculated using Image J software.
Fourier infrared analysis
The spectra of the electrospun fibers were recorded in Nicolet FT-IR Spectrometer. A small piece of electrospun fiber mat was placed on the measuring surface and the spectra were recorded from 600 to 4000 cm−1 wavelength at a resolution of 4 cm−1 resolutions with 32 scans per minute.
Contact angle measurements
The water drop method was utilized to measure the contact angle of the developed membranes and was evaluated in the video contact angle equipment. The distilled water droplet with a size of 0.5 µL was placed on the electrospun membrane, and within a few seconds, the static image was captured using a video camera. The manual contact angles were measured from the captured image of the water droplet through computer integrated software.
Thermal analysis
Thermal analysis for the electrospun membranes was carried out by the thermogravimetric (TGA) unit. The thermal analysis was done under a nitrogen atmosphere, and each sample was run from 30 to 1000°C at a heating rate of 10°C/min.
Atomic force microscopy analysis
The surface morphology of electrospun nanofibers was characterized using atomic force microscopy (AFM) equipment. The scanning of the of the electrospun nanofibers was conducted in a normal atmosphere, and the data were collected by scanning in a 20 µm × 20 µm size and images were obtained through JPKSPM data processing software with 256 × 256 pixels.
Mechanical testing
Mechanical properties were carried out in uniaxial testing machine according to ASTM D882-10 standards to determine the deformation rate of the electrospun membranes. The samples were cut into a size of 40 mm × 10 mm and clamped in the grip ends of the tensile machine. Then, the deformation rate was measured under a crosshead speed of 5 mm/min with a load cell of 500 N. Finally, the tensile strength and the Young’s modulus were calculated through machine generated stress–strain curve.
Coagulation assay
APTT and PT assay
The anticoagulant properties of the electrospun membranes were evaluated through PT and APTT. To start APTT, the samples were cut into square samples with a size of 0.5 × 0.5 cm2 and were pre-incubated with 100 µl of PPP at 37°C. Then, the mixture was added with 100 µl of rabbit brain cephalin for 3 min, followed by the addition of calcium chloride (0.025 M). Finally, using steel hook, the clot was initiated and the clotting times were measured by chronometer. Similarly, to begin the PT, the samples were cut into square samples with a size of 0.5 × 0.5 cm2 and were pre-incubated with 100 µl of PPP at 37°C followed by the addition of 0.9% sodium chloride (NaCl)–thromboplastin containing calcium (Ca2+) ions. Finally, using steel hook, the clot was initiated and the clotting times were measured by chronometer [21].
Hemolysis assay
To evaluate the amounts of hemoglobin released from the electrospun membranes, the hemolysis assay was performed. To begin, the electrospun samples were cut into 1 cm × 1 cm size and soaked in 37°C of saline (0.9% w/v) for 30 min followed by adding with 4:5 v/v% ratio of citrated blood and diluted saline for 1 hour at 37°C. Then, the samples were retrieved and centrifuged for 15 min at 3000 r/min. Finally, the clear supernatant was pipetted and the amount of hemoglobin released was recorded at a wavelength of 542 nm. The hemolytic percentage was measured as reported earlier [21].
Cell proliferation study
Human fibroblast (HDF) cells were cultured in the medium of Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and maintained at 37°C with 5% carbon dioxide (CO2). To start the assay, the cultured HDF cells with a density of 10 × 103 cells/cm2 were seeded on the sterilized electrospun membranes placed in the 24 well plates and placed in the CO2 incubator. The viability of HDF cells was determined after three days culturing using MTS (3-(4,5-dimethylthiazol-2-yl)-5-(3- carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H tetrazolium, inner salt) assay. After three days of culture, the membranes with cell grown were added with 20% of MTS and incubated for 4 h. After 4 h, the culture medium was taken out carefully and the cell counts were measured by recording the absorbance at 490 nm using spectrophotometric plate reader.
Statistical analysis
All experiments were performed three times independently. One-way analysis of variance was carried out followed by Dunnett post hoc test to determine the statistical significance (p < 0.05). The obtained results are expressed as mean ± SD and for qualitative experiments; a representative of three images is indicated.
Result and discussion
One of the goals in bone tissue engineering is to develop and design materials which should resemble the native bone structure effectively. Now-a-days, in bone tissue engineering, the widely used material is the electrospun synthetic polymers which were reported to mimic the ECM. But the synthetic materials used in the bone tissue engineering possess a lack of bioactivity and blood compatibility. In order to overcome these issues, ghee and propolis were incorporated into the PU substrate. The ghee and propolis were used owing to its non-toxicity and also its medicinal constituents present in their composition.
Figures 1 and 2 depict the morphology and fiber diameters distributions of PU, PU/ghee and PU/ghee/propolis nanocomposites analyzed through SEM analysis.

SEM images of (a) polyurethane, (b) polyurethane/ghee nanocomposites and (c) olyurethane/ghee/propolis nanocomposites.
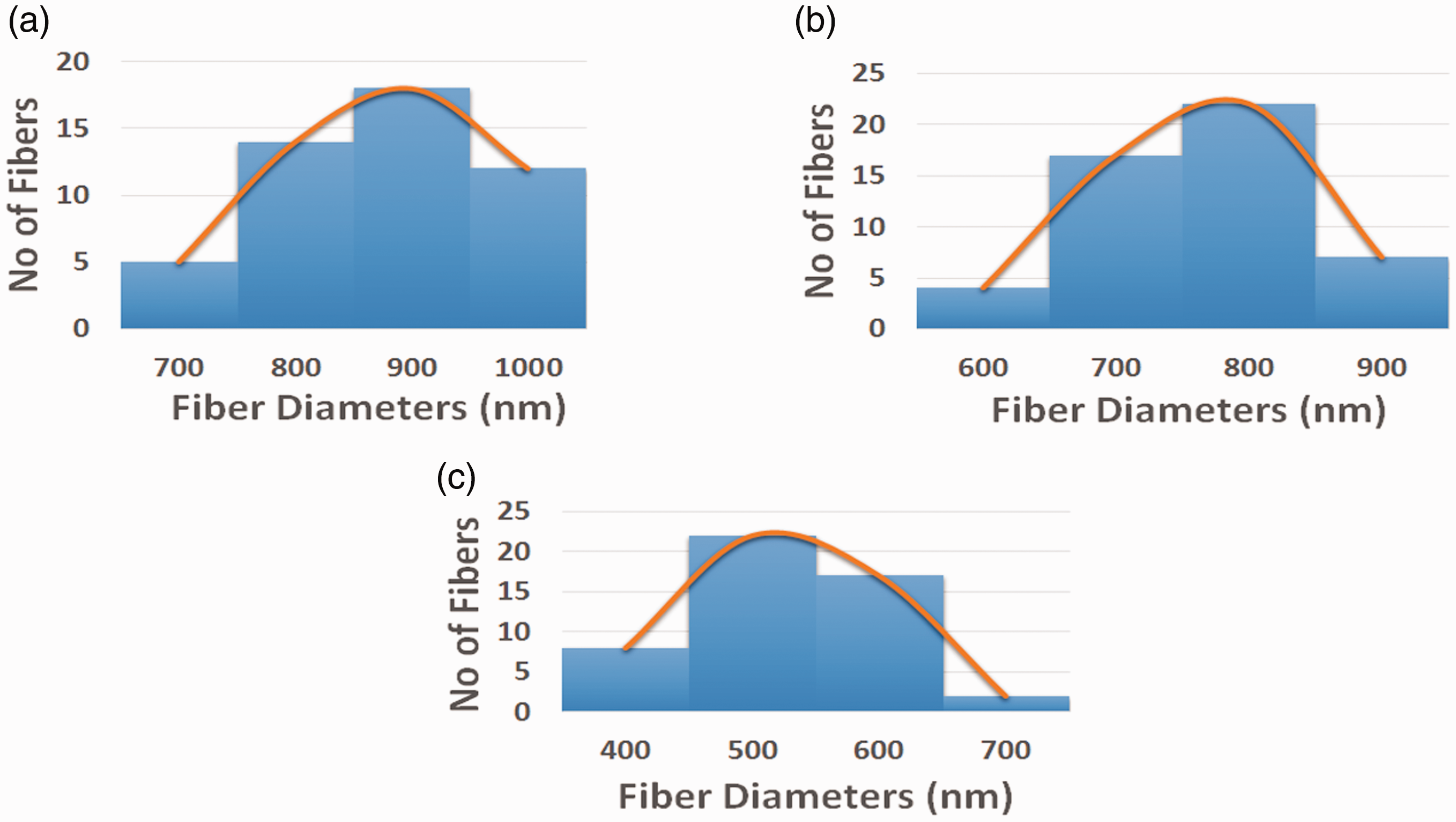
Fiber diameter distribution of (a) polyurethane, (b) polyurethane/ghee nanocomposites and (c) polyurethane/ghee/propolis nanocomposites.
From SEM images, it was observed that the PU and blended composite membranes exhibited continuous randomly oriented fibers without any sign of bead defects. The measured fiber diameters of PU and PU nanocomposites fabricated with ghee and ghee/propolis using Image J were observed to be 890 ± 116.911 nm, 817 ± 138.39 nm and 576 ± 144.96 nm, respectively. From the measurement of the fiber diameter, it was observed reduction in the diameter of fibers in PU membrane when mixed with ghee and propolis. Further, with the addition of propolis in ghee membrane, there was a synergistic reduction in fiber diameter compared to the PU. The decrease in the smaller fiber diameter was because of the decrease in the polymer concentration while incorporating ghee and propolis into the polyurethane membrane [22,23]. Further, this behavior was also due to the plasticizing effect of the ghee and propolis with the PU molecules resulting in the reduction of the glass transition temperature. Liu et al, prepared polylactic acid (PLA)/nanohydroxyapatite (nHA) scaffold for bone tissue engineering. It was observed that the prepared PLA/nHA nanocomposites showed reduced fiber diameter and exhibited increased growth of osteoblast cells compared to the control [24]. The developed nanocomposites with reduced fiber diameter compared to polyurethane scaffold might favor the osteoblast cell growth.
Figure 3 depicts the Fourier infrared (FTIR) results of developed PU, PU/ghee and PU/ghee/propolis nanocomposites.
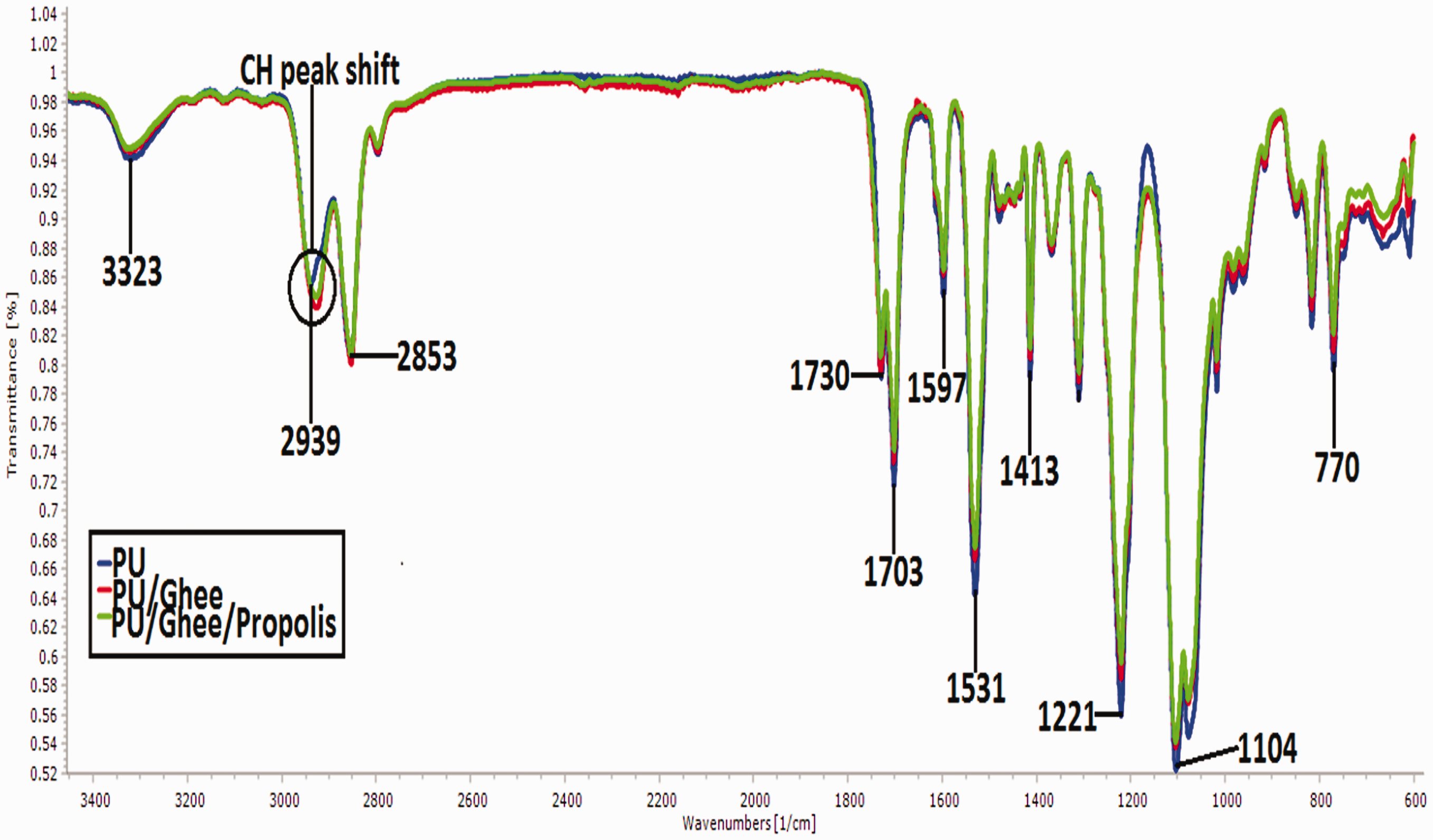
FTIR spectra of (a) polyurethane, (b) polyurethane/ghee nanocomposites and (c) polyurethane/ghee/propolis nanocomposites. PU: polyurethane.
In the spectra of PU membrane, a broad peak at 3323 cm−1 corresponds to NH stretch and the peaks at 1597 cm−1 and 1531 cm−1 were attributed to the vibration of NH stretching. The CH2 stretching was observed at sharp bands 2939 cm−1 and 2853 cm−1, while its vibrations were seen at 1413 cm−1. The twin peak at 1730 cm−1 and 1703 cm−1 corresponds to CO stretching and its vibrations of alcohol group were observed at 1221 cm−1, 1104 cm−1 and 770 cm−1, respectively [20]. From the spectra of PU/ghee and PU/ghee/propolis nanocomposites, no new peak formation was observed, but the peak intensity was decreased in PU/ghee and PU/ghee/propolis nanocomposites indicating the formation of hydrogen bond [25]. The formation of hydrogen bond was due to the linking of OH and CH molecules in the ghee and propolis with molecules of the polyurethane. The existence of ghee and propolis in PU was indicated by shifting of CH band from 2939 cm−1 in PU to 2929 cm−1 and 2929 cm−1 in PU/ghee and PU/ghee/propolis which further confirms the interaction of PU with ghee and propolis [26].
The wetting behavior of electrospun PU, PU/ghee and PU/ghee/propolis nanocomposites was presented. It was observed that the wettability of PU/ghee was decreased than PU and in PU/ghee/propolis nanocomposites and was improved than PU membrane. The measured contact angle of PU membrane was found to around 100° ± 0.5774°, while the contact angles of electrospun PU/ghee and PU/ghee/propolis nanocomposites were reported to be 122° ± 1° and 55° ± 1°, respectively. Scaffold used in tissue engineering requires an optimal range of wettability to support cell adhesion and proliferation. The contact angle of the PU/ghee membrane was found to be 122° ± 1°. The observed contact angle was beyond the optimum range of osteoblast cell adhesion and proliferation (<106°) which necessitates the need for tailoring the contact angle [27]. Fortunately, the addition of propolis to the PU/ghee nanocomposite resulted in the marked decrease in the contact angle making it hydrophilic. Fu et al. developed an electrospun scaffold based on poly(lactic-co-glycolic acid) (PLGA)/nHA incorporated with graphene oxide for bone tissue engineering. It was reported that the prepared PLGA/HA/graphene oxide membrane showed hydrophilic nature and favors the enhanced growth of osteoblast cells [28]. Hence, our tailor-made nanocomposite with optimal wettability can serve as a putative candidate for bone tissue engineering.
Figure 4 shows the tensile results of PU, PU/ghee and PU/ghee/propolis nanocomposites investigated through the uniaxial tensile machine. The tensile result values were listed in Table 1.
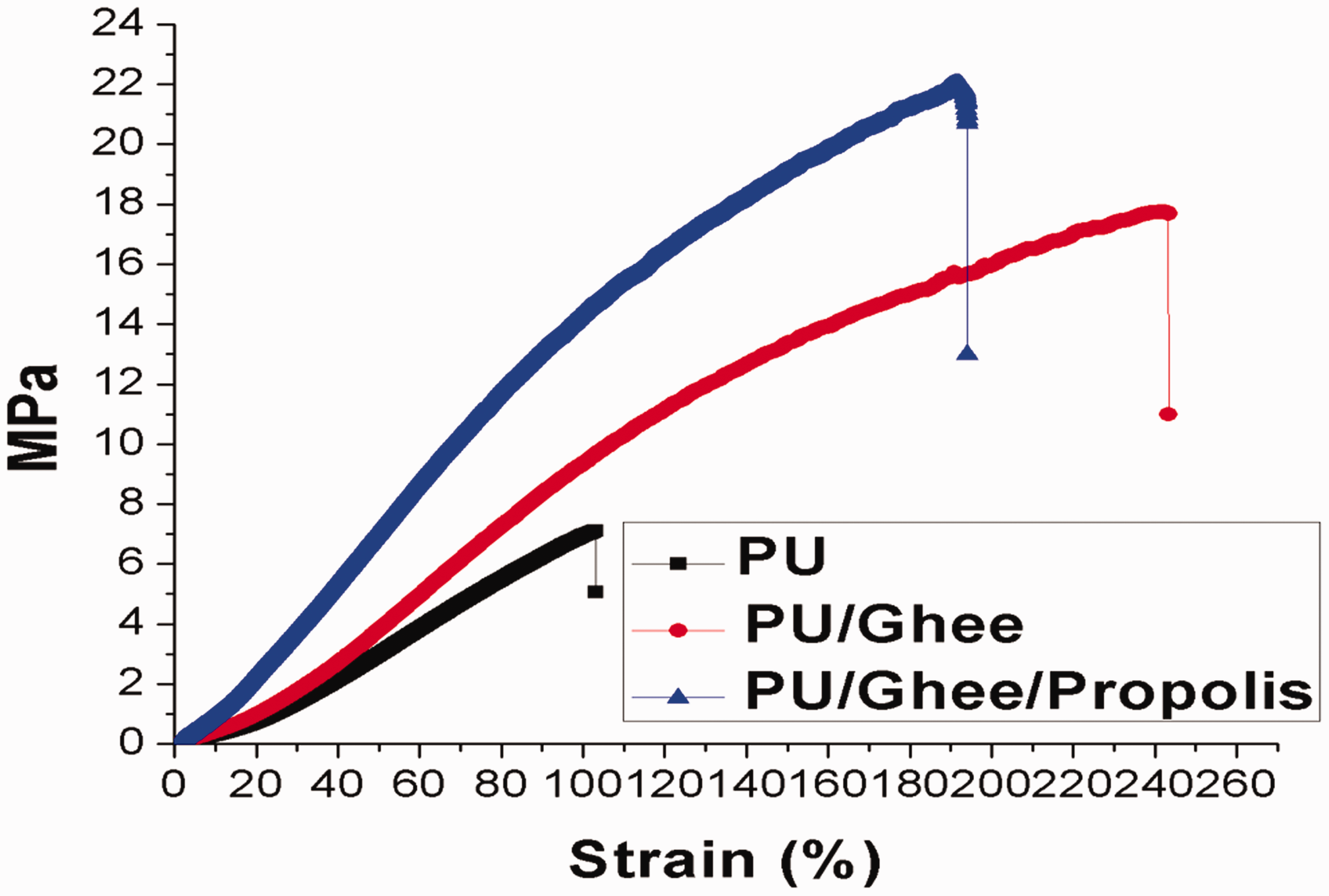
Mechanical testing of (a) polyurethane, (b) polyurethane/ghee oil nanocomposites and (c) polyurethane/ghee/propolis nanocomposites. PU: polyurethane.
Mechanical testing values of electrospun membranes.

It was observed from the stress–strain curve that the PU/ghee and PU/ghee/propolis nanocomposites exhibited a significant increase in the tensile strength compared to the pure PU membrane. The reported tensile strength of electrospun PU/ghee and PU/ghee/propolis nanocomposites was found to 17.75 MPa and 22.02 M Pa, while the electrospun PU membrane showed the strength of 7.12 MPa only. Hence, the addition of ghee and propolis improved the mechanical strength of the PU significantly. Qian et al. developed PCL scaffold blended with the gelatin and chitosan nanofibers. It was observed that the developed PCL/gelatin/chitosan nanocomposites showed higher tensile strength and suggested that the increase in tensile strength was because of thin fibers of the developed membranes [29]. In this study, the smaller fiber diameters of the electrospun PU/ghee and PU/ghee/propolis nanocomposites could favor the enhanced tensile strength. In another study, Unnithan et al. fabricated a wound dressing scaffold based on polyurethane added with emu oil nanofibers. It was observed that the addition of emu oil favored the enhancement of the tensile strength which correlates with our findings. They concluded that the addition of emu oil could provide the attachment of fibers because of hydrogen bond formation between PU and emu oil molecules [25]. FTIR analysis of the fabricated composites revealed the hydrogen bond formation as reported earlier. The molecules of ghee and propolis might have favored the bonding with the polyurethane matrix resulting in enhancement of the tensile strength.
Figure 5 depicts the measured Ra of PU, PU/ghee and PU/ghee/propolis nanocomposites. It was observed that the addition of ghee and propolis into the PU matrix decreased surface roughness. The measured Ra electrospun PU membrane was found to 576 nm, while the electrospun PU/ghee and PU/ghee/propolis showed Ra of 521 nm and 491 nm, respectively. The decrease in the surface roughness was due to the presence of ghee and propolis in the pristine PU and this might be because of bioactive constituents present in the ghee and propolis. A similar finding was observed in a recent study by Jaganathan et al. [30]. It was reported that the lower surface roughness exhibited a high response of bone marrow cell attachment and proliferation compared to the rougher surfaces [31]. Hence, our developed nanocomposites exhibiting smooth surfaces may warrant higher osteoblast cell adhesion and proliferation.
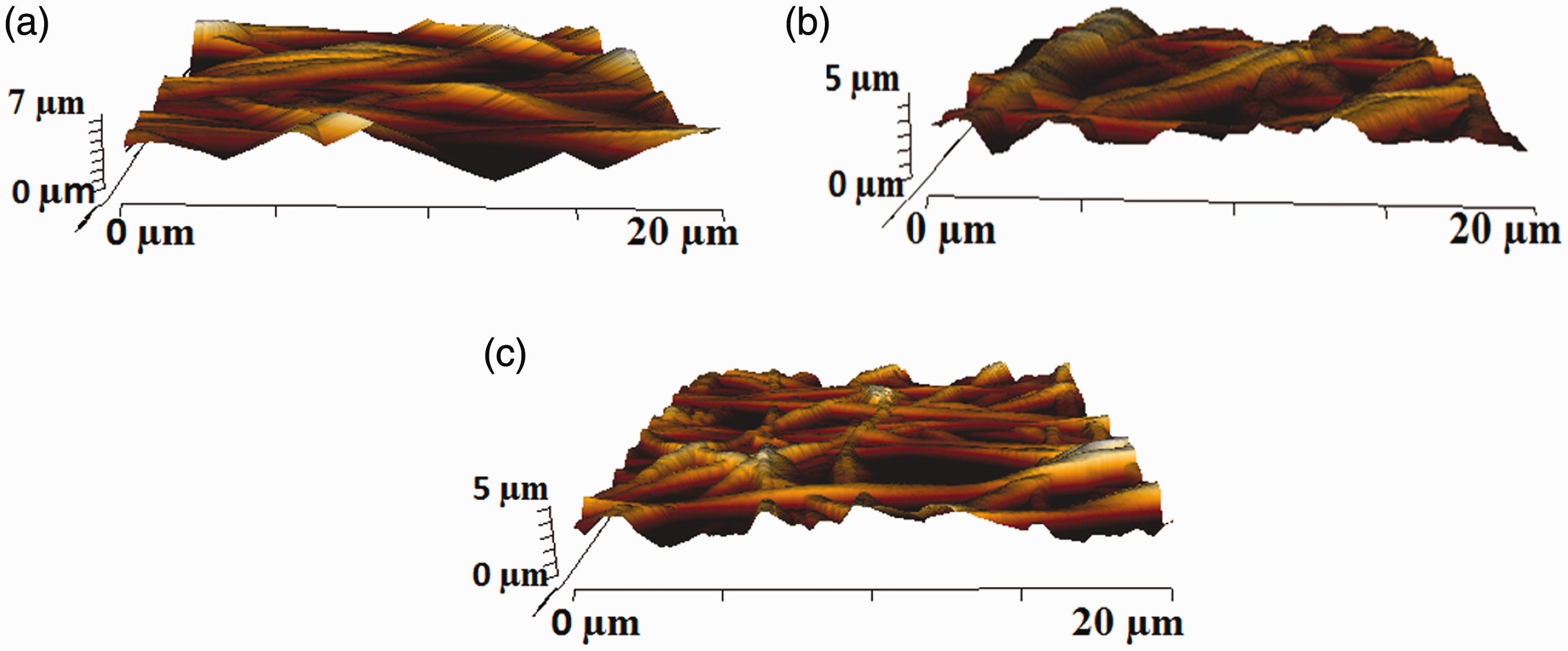
AFM images of (a) polyurethane, (b) polyurethane/ghee oil nanocomposites and (c) polyurethane/ghee/propolis nanocomposites.
Figures 6 and 7 represent the TGA and DTG curves of PU, PU/ghee and PU/ghee/propolis nanocomposites. It was observed that the thermal behavior of PU was increased with the addition of ghee and propolis. It was observed that the PU begins to degrade at 276°C, while that temperature was moved to 299°C and 306°C in the developed PU/ghee and PU/ghee/propolis nanocomposites indicating its improved thermal behavior as shown in Figure 6(a) to (c). At 5% degradation, the temperature of PU, PU/ghee and PU/ghee/propolis was found to be 286.4°C, 313.45°C and 319.4°C, respectively. The obtained results depict the degradation temperature of composites was found to be higher than the pristine PU. Jaganathan et al. incorporated castor oil in polyurethane matrix and reported that the incorporation of castor oil improved the thermal behavior of polyurethane membrane which correlates with the observed findings [32]. The increase in thermal stability was due to the presence of ghee and propolis constituents in the polyurethane matrix. Hayashi and Tournilhac reported in their work that the intermolecular hydrogen bonding between the molecules will favor the increase in thermal stability [33]. As reported previously, the addition of ghee and propolis in the polyurethane matrix has formed the hydrogen bonding which might have resulted in the enhancement of the thermal stability. Further, the results of derivate weight loss curve for the PU, PU/ghee and PU/ghee/propolis were depicted in Figure 7(a) to (c). It was observed that the weight loss in the electrospun PU/ghee was decreased than the PU membrane and PU/ghee/propolis nanocomposites. From the DTG diagram, it was revealed three weight loss peaks were exhibited by the electrospun PU membrane in which the first loss starts at 223°C to 348°C, the second loss starts 348°C to 446°C and the final loss happens at 557°C to 684°C, respectively. In the case of electrospun PU/ghee nanocomposites, it showed three weight loss in which the first loss starts at 221°C to 375°C, the second loss was seen at 375°C to 556°C and the final loss occurs at 556°C to 739°C. In the case of PU/ghee/propolis nanocomposites, it showed two weight losses in which the first weight loss happens at 220°C to 376°C and the second loss happens at 376°C to 626°C. In PU/ghee nanocomposites, it was noted that the intensity of the first weight loss peak was decreased than the PU membrane suggesting low weight loss. Although PU/ghee/propolis nanocomposites showed only two step degradation, the peak intensity of first weight loss curve was major than the PU/ghee mat suggesting the increased weight loss in PU/ghee/propolis nanocomposites was owing to the existence of propolis into the PU matrix.
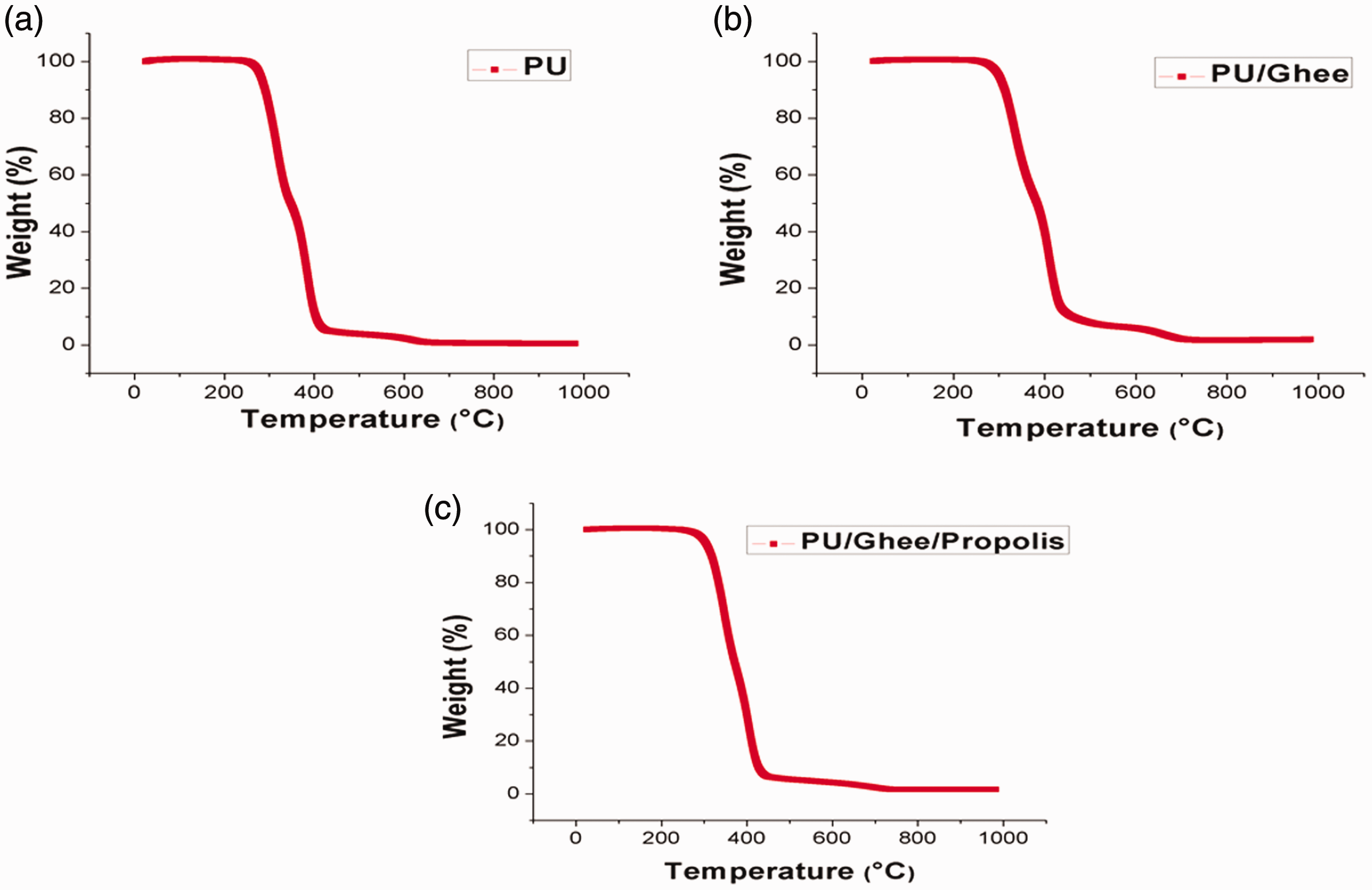
TGA analysis of (a) polyurethane, (b) polyurethane/ghee nanocomposites and (c) polyurethane/ghee/propolis nanocomposites. PU: polyurethane.

Weight residue percentage of (a) polyurethane, (b) polyurethane/ghee nanocomposites and (c) polyurethane/ghee/propolis nanocomposites.
Figures 8 and 9 depict the blood compatibility parameters for PU, PU/ghee and PU/ghee/propolis nanocomposites determined through APTT and PT assay. It was observed that the blood clotting time of PU was enhanced by adding ghee and propolis. In APTT assay, the measured blood clotting time for PU membrane was 152.7 ± 3.055 s, and for PU/ghee and PU/ghee/propolis nanocomposites, it was observed to be 164.7 ± 4.163 s and 153 ± 2.646 s, respectively. Similarly, in PT assay, the measured blood clotting time for PU membrane was 88.67 ± 2.517 s, and for PU/ghee and PU/ghee/propolis nanocomposites, the measured blood clotting time was found to be 93 ± 2.646 s and 90.33 ± 2.082 s, respectively. It was found that PU/ghee nanocomposites were significant in delaying the coagulation times. This may be due to the hydrophobic nature of the scaffold. It was reported that hydrophobic surface binds the plasma protein irreversibly resulting in the passivation of the surface and promoting the blood compatibility. Further, when there is an addition of propolis, this tendency seems to be shifted primarily due to the balance of hydrophobic/hydrophilic moieties of propolis which resulted in the slight reduction of the coagulation times [34]. Although there is a reduction, still the PU/ghee/propolis nanocomposites blood compatibility is improved compared to the pure polyurethane. Further, the release of hemoglobin was measured for all electrospun membranes to depict their safety on RBC. The induced hemolytic percentage for electrospun PU/ghee and PU/ghee/propolis nanocomposites was 1.61% and 1.52%, while the PU membrane showed a hemolytic percentage of 2.48% as indicated in Figure 10. It was reported when the hemolytic percentage was below 2%, it was non-hemolytic and if the percentage was above 2%, it was hemolytic material [21]. Our fabricated nanocomposite scaffolds showed a hemolytic percentage of less than 2% and so it was considered as a non-hemolytic material. It was reported that the blood compatibility was improved significantly by the smaller fiber diameter of the scaffolds [35]. In this study, the smaller fiber diameter of the developed nanocomposites might have favored the enhanced blood compatibility of the nanocomposites.
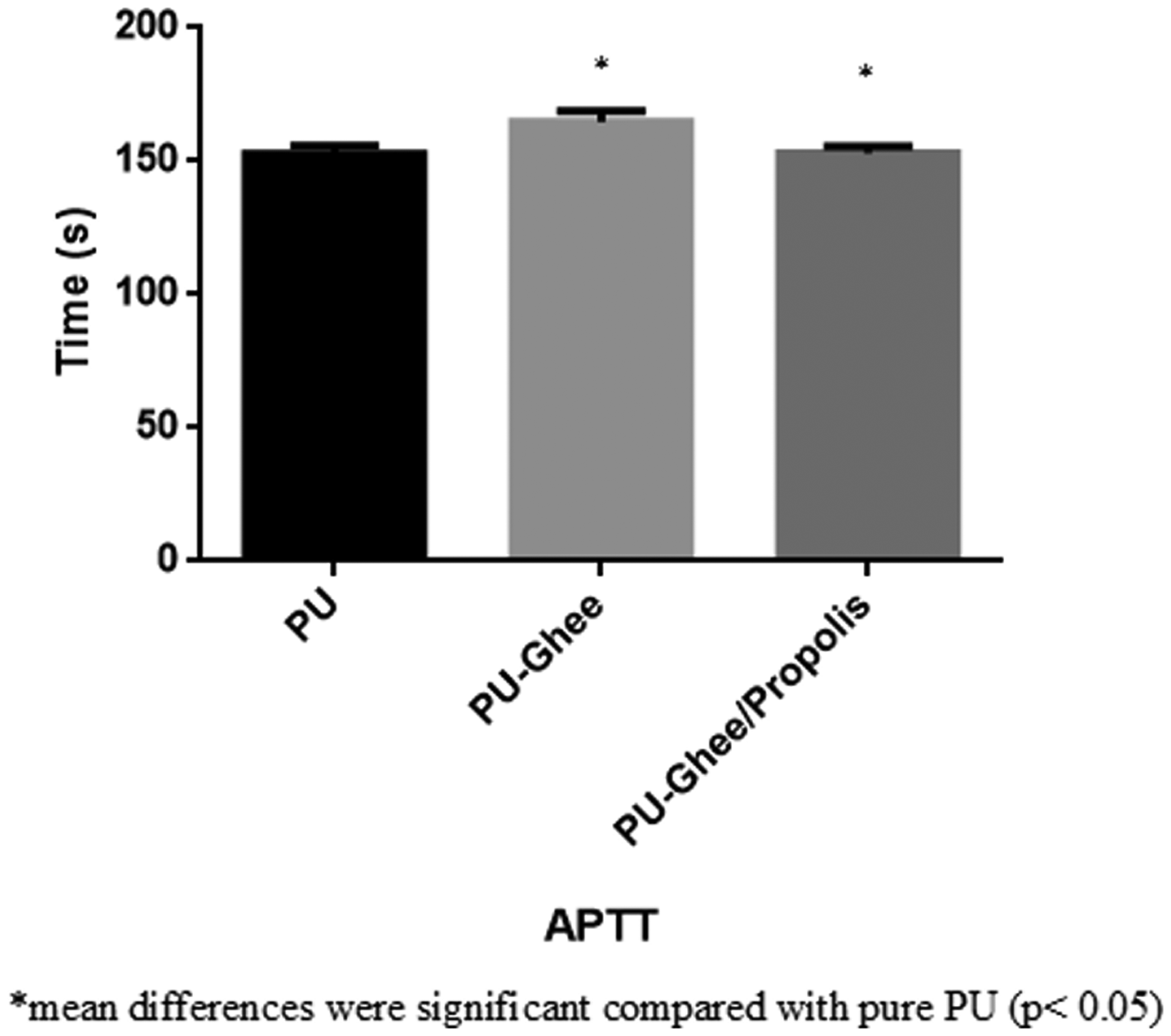
APTT assay of (a) polyurethane, (b) polyurethane/ghee nanocomposites and (c) polyurethane/ghee/propolis nanocomposite. APTT: activated partial thromboplastin time; PU: polyurethane.

PT assay of (a) polyurethane, (b) polyurethane/ghee nanocomposites and(c) polyurethane/ghee/propolis nanocomposites. PT: prothrombin time; PU: polyurethane.

Hemolytic assay of (a) polyurethane, (b) polyurethane/ghee nanocomposites and (c) polyurethane/ghee/propolis nanocomposites. PU: polyurethane.
Figure 11 depicts the MTS assay results of the PU, PU/ghee and PU/ghee/propolis nanocomposites cultured with HDF cells. The cell viability results after three days of culture showed the enhanced proliferation rate of fibroblast cells on the surfaces of nanocomposites. Further, it was observed that the incorporation of ghee and propolis significantly improved the cell adhesion and proliferation rates than the pristine PU membrane indicating its non-toxic nature to the HDF cells. The cell viability of the electrospun PU/ghee and PU/ghee/propolis nanocomposites was observed to be 148.3% ± 6.028% and 156.7% ± 5.686%, while the PU membrane showed a cell viability rate of 133.33% ± 8.963%. The results of the cell viability of the fabricated nanocomposite scaffolds were higher than the pure polyurethane. Further, the electrospun PU/ghee/propolis scaffold improved fibroblast attachment than the PU/ghee membrane. A closer analysis of this behavior suggested that hydrophilic scaffolds (PU/ghee/propolis nanocomposites) wettability were in the range of 40° to 70°. This is the region of maximum fibroblast cell adhesion and proliferation which was reported in a recent study [36].
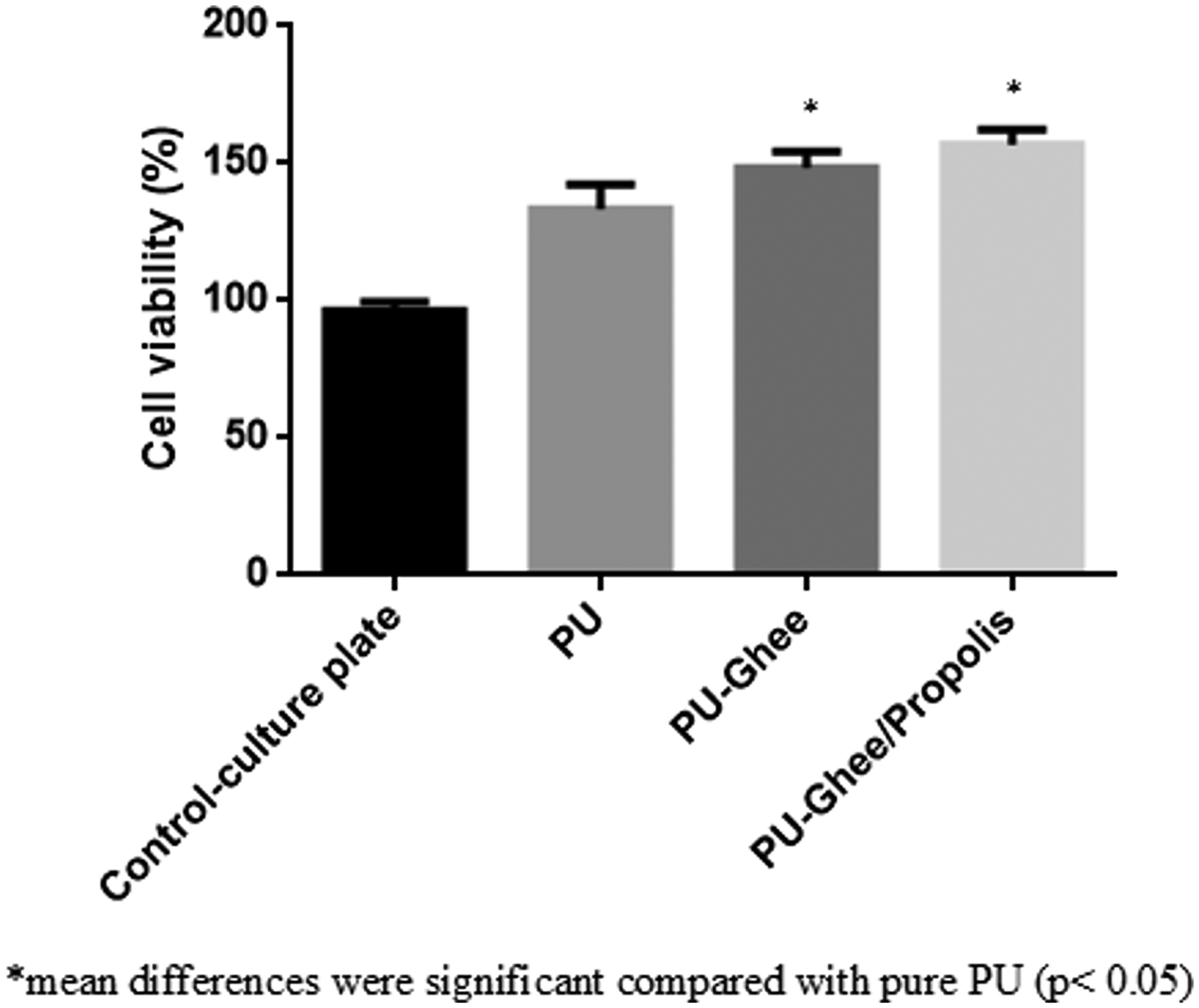
MTS assay of (a) polyurethane, (b) polyurethane/ghee nanocomposites and (c) polyurethane/ghee/propolis nanocomposites. PU: polyurethane.
Conclusion
In this study, a novel bone scaffold comprising PU incorporated with ghee and propolis was successfully fabricated using the electrospinning technique. From overall results, it was observed that the novel developed nanocomposites possessed smaller fiber diameter than the pure PU. The presence of ghee and propolis in PU was confirmed by CH peak shift band and also hydrogen bond formation. The contact angle was found to be increased in PU/ghee nanocomposites indicating the hydrophobic nature while the angle was decreased in PU/ghee/propolis nanocomposites suggesting the hydrophilic nature. The surface roughness of electrospun nanocomposites was found to decrease while heat stability was observed to be enhanced as noted by TGA analysis. Further, the blood compatibility assessments revealed the delayed activation of blood clots in the electrospun nanocomposites suggesting better blood compatibility. Moreover, the hemolytic assay and cytocompatibility studies showed that the developed nanocomposites possess enhanced safety to RBC and better proliferation of HDF cells indicating non-toxic nature. Hence, this study concluded that the newly developed PU/ghee and PU/ghee/propolis nanocomposites were found to possess desirable characteristics such as better physicochemical, improved blood compatibility and non-toxic behavior which might be utilized as an appropriate candidate for bone tissue engineering.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
