Abstract
In the recent decade, the growth of medical textiles is enormous and it paves the sustained development in the quality of life. In this research, an electrospun textile scaffold comprising polyurethane impregnated with
Introduction
The standard methods utilized for bone repair are autologous and allogeneic transplantations. However, it is limited due to certain drawbacks like limited donor site supply, immune rejection, donor site morbidity, and transfer of pathogens [1]. With the development of technology, the growth of the bone tissue engineering was found to be enormous and demanding for over the years mainly due to their some advantages like limitless supply and no disease transmission. The objective of the bone tissue engineering is to provide functional bone regeneration through a combination of scaffolds, cells and growth factors [1]. Among these, the scaffold plays a vital role in mimicking the native extracellular matrix (ECM) of human tissue. Further, they also help in supporting the cell adhesion and proliferation for new tissue growth. Recently, the fabric based on textile technologies were widely utilized in different application areas such as liquid fuel purification, textile based sensors, protective clothing, and tissue engineering applications [2–7]. It is possible to fabricate complex structures and three-dimensional designs with the help of textile technologies. Further, the studies have shown that functionalized fabrics were reported to possess outstanding biological properties with less toxicity [8–13] Although several techniques like weaving, knitting, braiding were utilized in the fabrication of fabrics, usage of electrospinning to produce nonwoven scaffolds is indispensable [14].
Electrospinning is a versatile and cost-effective technique, which could be able to fabricate fibers ranging from micrometers to nanometers. The nanofibers fabricated through electrospinning technique was widely utilized in biomedical applications owing to its high surface area to volume ratio, microporous structure, and also the ability to mimic the ECM structure of the human tissue [15]. Further, the nanofibers enhance cell adhesion, proliferation, and growth for remodeling of the damaged tissue. Further, the important properties considered in the design of scaffolds for the bone tissue engineering were biodegradability, biocompatibility, porosity, nontoxicity, and good mechanical properties [16,17]. A wide range of synthetic and natural polymers have been employed in obtaining nanofibers for the past few years either in pure or blended form. Polyurethane (PU) is one among these, which possesses good mechanical property and water insolubility that finds applications in biosensors, protective cloths, and enhancing epithelial growth [18]. It was reported to be biocompatible, biodegradable, good oxidation, and thermal stability [19,20]. Further, it can be inferred that PU-based scaffolds hold great potential in tissue engineering applications like skin, bone, and cardiac [21–24]. In this study, Tecoflex EG-80A PU was used to fabricate the bone scaffold.
Hydroxyapatite nanoparticles (nano HA) is a prospective biomaterial that is utilized for making bone substitutes owing to their biocompatible, biodegradable nature, osteo-conductive, osteo-inductive, and osteo-integrative properties [25]. Apart from these outstanding properties, it was found to be risky when they were applied in the clinical applications due to their nanoscale particle size [26]. Despite their promising potential in the clinical applications, the safety and toxicity of HA are still considered as a challenging one. Hence, it has forced the researchers to find alternatives to HA particles. In Asian countries, the essential oil plays an important role in alleviating the pains created at the bone joints. The essential oils derived from the plants were reported to be less toxic and cost effective. Recent research suggested the use of oils in the bone tissue engineering applications [6,7]. This motivated us to use
AS is a member of Amaranthaceae family, which grows both in wetlands and uplands. It is rich in vitamin A, dietary fiber and also contains various bioactive constituents like flavonols, steroids, tannins, triterpenoids, β′-sitosterol, campesterol, stigmasterol, and lupeol. The chemical structure of AS is indicated in Figure 1. Further, it also possesses minerals like sodium (Na), potassium (K), magnesium (Mg), calcium (Ca) and a small amount of zinc, copper, and iron (Zn, Cu, and Fe) [27]. AS is reported as a remedy for various ailments like night blindness, dysentery, intestinal cramps, anaemia, hepatitis, diarrhoea, bronchitis, tight chest, lung troubles, asthma, treating fever, and wounds [28–30]. Further, it has been reported that this plant possesses antibacterial, antiulcer, antipyretic, diuretic, and hematinic properties [28]. Several researches indicated its versatile nature in biomedical applications. However, there is no single report on the use of AS oil in the tissue engineering application. The aim of the study is to fabricate an electrospun novel bone scaffold based on PU incorporated with the AS oil. In addition, the fabricated membranes were subjected to the physico-chemical characterization, blood compatibility assessments, and cytocompatibility studies.

Chemical structure of
Materials and methodology
Materials
Medical grade Tecoflex EG-80A PU (molecular weight = 1000 g/mol) was obtained from LubriZol, USA and N, N-dimethylformamide (DMF) was supplied by Merck Millipore, Germany. AS oil was obtained from Thurgas Trading, Pulau Pinang, Malaysia. Phosphate buffered saline (PBS, biotech Grade) and sodium chloride physiological saline (0.9% w/v) used in the blood compatibility assays were supplied by Sigma-Aldrich, Malaysia. The activated partial thromboplastin time (APTT) and prothrombin time (PT) reagents such as rabbit brain activated cephaloplastin, calcium chloride (0.025 M), and thromboplastin (Factor III) were purchased from Diagnostic Enterprises, India.
Preparation of PU and biocomponent PU/AS electrospun solutions
To prepare 9 wt% of homogeneous PU solution, 480 mg of PU was dissolved in 4.5 mL of DMF and stirred overnight at room temperature. Similarly, 4 v/v% of homogeneous AS solution was made by adding 120 µL of AS in 3 mL of DMF respectively and stirred for 1 h maximum at room temperature. Finally, PU/AS solution with the ratio of 8:1 v/v % was obtained by slowly adding the prepared homogeneous PU and AS solution and stirred for 2 h at room temperature to obtain a uniform dissolution.
Fabrication of electrospun PU and biocomponent PU/AS oil membranes for bone scaffolds
The electrospun membranes were fabricated through electrospinning technique (Progene Link Sdn Bhd, Malaysia) operated at room temperature with a humidity of 55%. To begin, the prepared homogeneous solution was loaded in 10 mL syringe attached with 18 G stainless steel needle (outer diameter of 1.28 mm and an inner diameter of 0.92 mm, Unicontrols Singapore Pte. Ltd) and fitted to the syringe pump. In our study, both PU and biocomponent PU/AS oil membranes were electrospun at a flow rate of 0.5 mL/h with an applied voltage of 10 kV. The deposited fibers membranes were deposited on the static collector drum for nearly 5 h, which was placed 20 cm from the needle. Finally, the obtained fibers were dried under vacuum at room temperature for 24 h to remove any residual DMF. The images of electrospun PU and biocomponent PU/AS oil membranes are shown in Figure 2.

Electrospun membranes of PU and biocomponent PU/AS oil membrane.
Scanning electron microscopy (SEM)
SEM (Hitachi Tabletop TM3000, Tokyo, Japan) was utilized to analyze the fibrous morphology and their fiber diameters. Before attaining photographs, the samples were gold coated with gold at 20 mA for 2 min and the scanning was performed. The photographs were captured at different magnifications at an accelerated voltage of 10 kV. Using Image J software, the average fiber diameter and fiber distribution were calculated by measuring at least individual 50 fibers randomly from the captured image with a magnification of × 2.5 k.
Contact angle measurements
The contact angles for the electrospun membranes were measured through a video contact angle (VCA) measurement unit (AST Products, Inc., Billerica, MA, USA). The contact angle of the electrospun membranes was measured in static mode. A droplet of deionized water with a size of 0.5 µL was placed on the fabricated membranes and the image of the water droplet was captured using video cam. Using computer integrated software, the contact angle was calculated and the experiment was repeated for three different trials to measure the average value.
Atomic force microscopy (AFM)
Surface analysis was carried out in an AFM equipment (NanoWizard®, JPK Instruments, Berlin, Germany) under a normal atmosphere to measure the average surface roughness of the electrospun membranes. The surface roughness of the electrospun membranes was determined by scanning in 20 × 20 µm area with 256 × 256 pixels.
Chemical and structural characteristics
Infrared (IR) spectrum of the electrospun membranes was inspected in Fourier transform infrared spectroscopy (FTIR) unit (Nicolet IS 5, Thermo Fischer Scientific, Waltham, MA, USA) equipped with zinc selenium (ZnSe) as an attenuated total reflectance (ATR) crystal. The absorption bands of the electrospun membranes were measured once at a wavelength of 600 and 4000 cm−1 with a resolution of 4 cm−1. The recorded spectra were normalized using standard Speckwin 32 software to identify the peaks present in it.
TGA analysis
TGA equipment (PerkinElmer, Waltham, MA, USA) was utilized to determine the thermal behavior of the electrospun membranes. A small piece weighing 3 mg is placed in an aluminum pan and the heating was performed once under a nitrogen atmosphere at a rate of 10°C min−1 in the temperature range of 30–1000°C.
Coagulation assay
Prothrombin test
Measurement of prothrombin time was used to predict the extrinsic pathway. To start the assay, 50 µL of platelet poor plasma (PPP) was applied on the surface of the electrospun membranes and incubated at 37°C. In the next step, it was added with 50 µL of NaCl-thromboplastin (Factor III) containing Ca2+ ions. Finally, the time taken for the blood clot was measured using the stopwatch and a steel hook [6,7].
Activated partial thromboplastin test (APTT)
On the other hand, the measurement of APTT was used to predict the intrinsic pathway. To start the assay, the samples were incubated with 50 µL of PPP at 37°C. Later, it was further incubated with 50 µL rabbit brain encephalin for 5 min followed by adding calcium chloride (0.025 M). Finally, the time taken for the blood clot was measured using the stopwatch and a steel hook [6,7].
Hemolysis test
To begin the assay, the samples were soaked with physiologic saline at 37°C for 30 min. Initially, the citrated blood was diluted in saline at a ratio of 4 : 5 v/v and then the samples were incubated for 60 min at 37°C. Then, these mixtures were centrifuged at 3000 r/min for 5 min. Finally, the clear supernatant was pipetted and the absorbance was measured at 542 nm, which expresses the release of hemoglobin. The percentage of hemolysis was calculated as per the formula as discussed earlier [6,7].
Statistical analysis
All experiments were performed three times individually unless stated otherwise. The statistical significance was determined through unpaired
Result and discussion
Figure 3(a) and (b) illustrates the morphology of PU and biocomponent PU/AS oil membrane determined through SEM analysis. It was observed from the SEM images that the electrospun PU and biocomponent PU/AS oil membranes showed continuous fibers without any beads. The calculated fiber diameters of PU and biocomponent PU/AS oil membrane using Image J were found to be 0.890 ± 0.119 µm and 0.821 ± 0.141 µm, respectively. The corresponding fiber diameter distribution curve was indicated in Figure 3(c) and (d). The pristine showed 6 fibers in the range of 700–800, 15 fibers in the range of 800–900, 25 fibers in the range of 900–1000, and 4 fibers in the range of 1000–1100, respectively. The PU/AS oil showed 20 fibers in the range of 700–800, 19 fibers in the range of 800–900, 7 fibers in the range of 900–1000, and 4 fibers in the range of 1000–1100, respectively. The fiber diameter measurements revealed the reduction in the fiber diameter of PU membrane when incorporating of AS oil and this might be due to the decrease in the polymer concentration while adding AS oil into the PU membrane. Further, this may be due to the plasticizing effect of the AS oil addition with the PU. The AS oil components may have reduced the viscosity of the melt solution of PU/AS oil resulting in the lower fiber diameter [31,32]. This may be corroborated using our SEM results as we observed smooth morphology for biocomponent PU/AS membrane compared to the more rigid pristine PU. Jaganathan et al. developed PU scaffold for the bone tissue engineering added with corn and neem oil. It was reported that the addition of corn and neem oil resulted in the reduction of the fiber diameter of the pristine PU which resembles our findings. They concluded that fabricated composite as a suitable candidate for the bone tissue engineering [6]. Our electrospun biocomponent PU/AS oil membrane exhibited reduced fiber diameter, which might be conducive to the bone tissue engineering.

SEM images of (a) PU membrane; (b) biocomponent PU/AS membrane and Fiber diameter distribution of (c) PU membrane; (d) biocomponent PU/AS membrane.
The obtained results using density bottle method showed the average porosity of the electrospun PU and PU/AS oil membranes was found to be 75% and 72%, respectively. Further, the pore size was calculated through Image J, which showed the average pore size of electrospun PU and PU/AS oil membranes was found to be 2834 ± 74 and 2162 ± 74 nm respectively. Prabhakaran et al. reported in their work that the prepared poly(DL-lactide-co-glycolide) blended with gelatin fibers showed reduced pore size and attributed this behavior was because of smaller fiber diameter [33]. In our study, the prepared PU/AS oil membrane showed smaller fiber which might have resulted in the reduced pore size.
Figure 4(a) and (b) presents the IR spectrum of developed PU and biocomponent PU/AS oil membrane. The spectra of PU showing a wide band at 3323 cm−1 indicates the N–H stretching vibrations and the peak at 1597 cm−1 and 1531 cm−1 indicates the N–H bending vibrations. The symmetric and asymmetric CH2 stretching vibrations were observed at the peak 2939 cm−1 and 2853 cm−1, while the peak at 1413 cm−1 indicates the CH2 bending vibrations, respectively. The sharp peaks at 1730 cm−1 and 1703 cm−1 represents the C=O stretching vibration and further peaks seen between 1200 and 1000 cm−1 represents the stretching vibration of C–O–C and C–OH, respectively [34]. From the spectra of biocomponent PU/AS oil membrane, it was noted no new band was formed but the intensity of PU peak was increased with the addition of AS oil due to strong hydrogen bond formation as denoted in Figure 4(b) [21]. OH and CH molecules present in the AS oil might have interacted with the PU. C=O present in PU have interacted with the AS oil-based hydrogen coming from OH and CH resulting in hydrogen bonding and their schematic diagram was represented in Figure 5. Further, there was a peak shift found, which also depicts the interaction of PU with AS oil. The CH band in PU from 2939 cm−1 was shifted to 2924 cm−1 in the biocomponent PU/AS oil membrane, which confirms the presence of AS oil in the PU [35]. Similar findings of peak shift and change in peak intensity were reported in oils incorporated PU scaffold [6,21]. The formation of hydrogen bonds and peak shift was due to the interaction of OH and CH molecules in the AS oil with molecules of the PU.

IR spectrum of PU membrane and biocomponent PU/AS membrane.
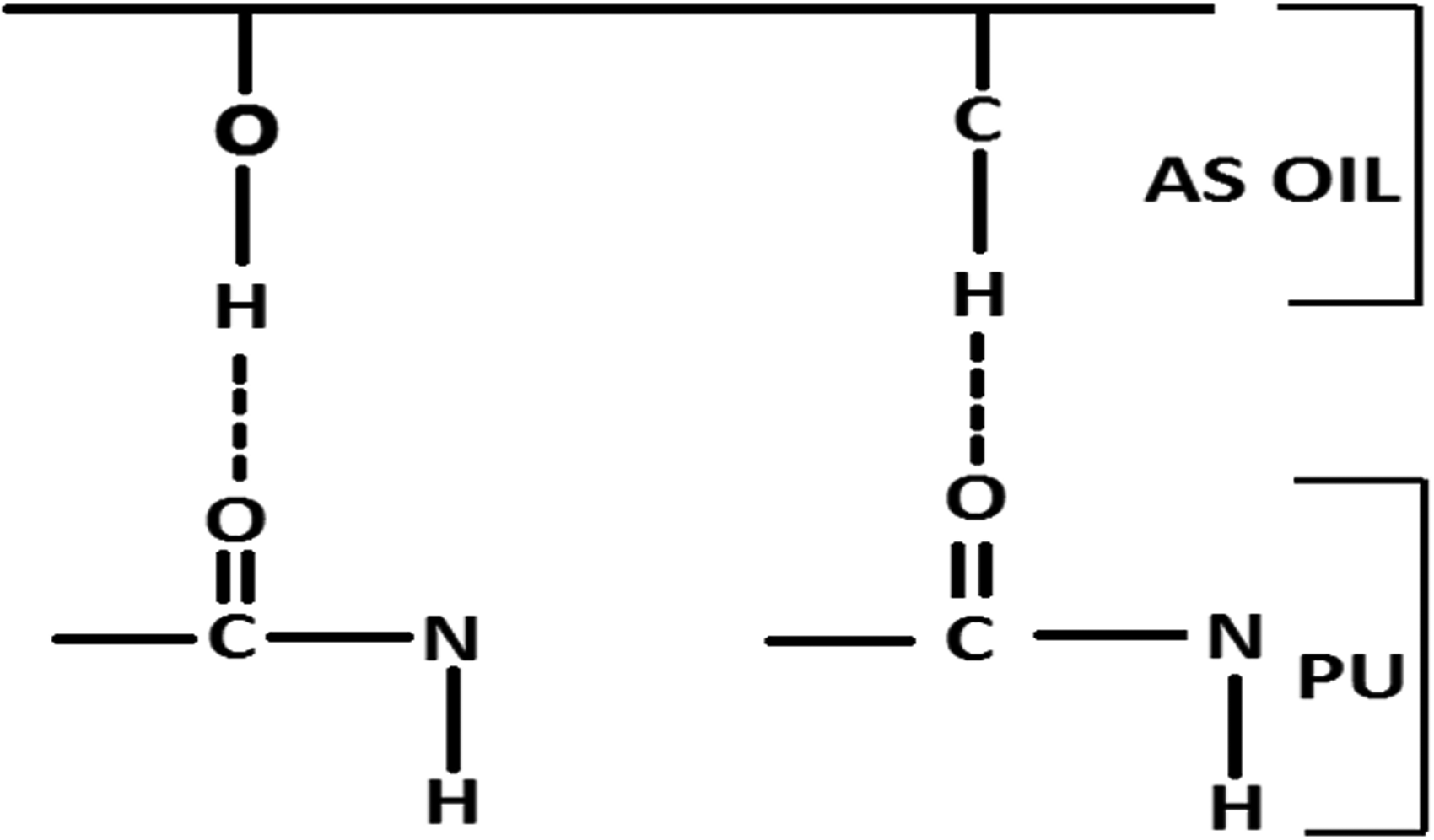
Schematic representation of the hydrogen bond formation in PU/AS membrane.
The contact angle measurements of electrospun PU and biocomponent PU/AS oil membrane were discussed. It was observed that the wettability of biocomponent PU/AS membrane was lower than the PU membrane. The contact angle of the pristine PU membrane was found to be 100° ± 0.5774, while the contact angles of the biocomponent PU/AS oil membrane were found to 113.7° ± 3.215, respectively. Hence, the incorporation of AS oil increased the hydrophobic nature of the PU. Cui et al. developed poly(D,L-lactide) and poly(D,L-lactide)-poly(ethylene glycol) membranes using electrospinning. It was observed that the electrospun scaffold with smaller fiber diameter resulted in the improvement of contact angle [36]. In another research, Ceylan et al. fabricated electrospun scaffold utilizing polystyrene and polyvinyl chloride fibers. It was shown that the water contact angle was enhanced when the diameter of fiber becomes smaller [37]. In our study, the developed biocomponent PU/AS oil membrane showed a smaller fiber diameter than PU membrane, which might have favored the increase in the contact angle. Cui et al. reported that the decrease in fiber diameters and the pore sizes would cause higher air entrapment in the surface, which might favor the larger contact angle [36]. Our smaller fiber diameter of the fabricated biocomponent PU/AS oil membrane might cause higher air entrapment, which favored the larger contact angle. In a recent study, Jaganathan et al. fabricated PU scaffold added with sunflower and neem oil for the bone tissue engineering. The contact angle of the fabricated biocomponent PU/sunflower oil was reported to be 112° ± 1 and exhibited enhanced cellular response than the pristine PU [7]. Hence, our biocomponent PU/AS oil membrane showed contact angle within the reported range and might be suitable for the bone tissue engineering.
Figure 6(a) and (b) indicates the TGA and DTG curves of the electrospun PU and biocomponent PU/AS oil membrane. The electrospun PU membrane showed no change in the thermal behavior with the addition of AS oil. It was noted from the TGA that both PU and biocomponent PU/AS oil membrane starts to degrade initially at 276°C, respectively. Further, from the figure it was evident that the biocomponent PU/AS membrane showed initial weight gain, which might be due to the bonding of AS oil molecules with PU. This observation is further correlated with the FTIR results, which depicts the formation of hydrogen bonding as confirmed by the shifting of CH band in PU from 2939 cm−1 to 2924 cm−1 in the biocomponent PU/AS oil membrane. Moreover, at 1000°C, the remaining weight residue percentage of pure PU membrane was observed to be 0.47%, while for the electrospun biocomponent PU/AS oil membrane, it was increased to 1.58%, which might due to higher unburnable/ash content. Further, the derivate weight loss curve depicted that the weight loss in the electrospun biocomponent PU/AS oil membrane was lower than the pure PU. From the DTG diagram, it was revealed the electrospun PU membrane showed three weight loss in which the first loss occurs at 223°C to 348°C (degradation of urethane groups), the second loss begins at 348°C to 446°C (degradation of ester bonds), and the final loss was seen at 557°C to 684°C (degradation of aromatic), respectively [38,39]. In the case of electrospun biocomponent PU/AS membrane, it showed two weight loss peaks in which the first weight loss occurs at 225°C to 363°C and the second loss at 363°C to 514°C, respectively. The first loss of biocomponent PU/AS oil may be attributed to the degradation of the urethane group. The second loss seems to be wider compared to the PU and this may be due to the melting of ester and partly aromatic groups. It was observed from the curve that the weight loss peak intensity of the fabricated biocomponent PU/AS membrane was observed to lower than the pristine PU indicating their reduced weight loss.

(a) TGA and (b) DTG analysis of PU membrane and biocomponent PU/AS membrane.
Figure 7 indicates the AFM images of PU and biocomponent PU/AS oil membrane, which depicts the average surface roughness. It was noted that the incorporation of AS into the PU membrane increased the average surface roughness. The calculated surface roughness for the pristine PU membrane was found to be 372 ± 169, while the electrospun biocomponent PU/AS oil membrane showed Ra of 508 ± 249. The increase in the surface roughness was due to the addition of AS oil into the pristine PU. The bioactive constituents present in the AS oil might have favored the increase in the surface roughness value. A similar increase in the surface roughness was observed in a recent study by Jaganathan et al.. In their work, electrospun PU scaffold added mustard oil was developed. They showed an increase in the surface roughness than the pristine PU due to the bioactive constituents present in the mustard oil [40]. It has been reported that the measurement of surface roughness was correlated to the adhesion, proliferation, and growth of osteoblastic cell. Rough surfaces enhance the osteoconductive process by favoring entrapment of fibrin protein and adhesion of an osteogenic cell [41]. Our fabricated biocomponent PU/AS oil membrane showed rougher surfaces, which might be suitable for enhancing the osteoconductive process.

AFM images of (a) PU membrane and (b) biocomponent PU/AS membrane.
The blood compatibility assessments of the electrospun PU and biocomponent PU/AS oil membrane measured through APTT and PT assay. Both APTT and PT assessments revealed marginally enhanced blood clotting time exhibited by the biocomponent PU/AS oil membrane than the PU membrane. In the APTT assay, the blood clotting time for PU membrane was found to be 175.3 ± 4.041 s, while the electrospun biocomponent PU/AS oil membrane showed blood clotting time of 183 ± 2.646 s. Similarly, in PT assay, the blood clotting time for PU membrane was found to be 76.33 ± 2.517 s while the electrospun biocomponent PU/AS oil membrane showed blood clotting time of 80.67 ± 1.528 s. The anticoagulant nature of the biocomponent PU/AS oil is higher than the pure PU but the results are not statistically significant. However, it can be inferred that the introduction of AS oil does not have any negative influence on the anticoagulant behavior of the fabricated membrane.
Further, the release of hemoglobin was measured for all electrospun membranes through hemolysis assay and the obtained results were presented. The assay results showed that the hemolytic percentage for the electrospun biocomponent PU/AS oil membrane was lowered than the pristine PU. The measured hemolytic index for electrospun biocomponent PU/AS oil membrane was found to be 1.35%, while the pristine PU membrane induced hemolytic index of 2.48%. It was reported when the measured hemolytic index was below 2%, then the material is called as non-hemolytic material. Hence, the developed biocomponent PU/AS oil membrane showed hemolytic index below than 2% and it was a nonhemolytic material [6,7]. In this experiment, the percentage of hemolysis was significantly lower than the PU. This shows our fabricated biocomponent PU/AS oil membrane have relatively edge over the pure PU. Reduced hemolysis with the anticoagulant nature may collectively promote the fabricated biocomponent PU/AS oil membrane as a candidate with enhanced blood compatibility for the bone tissue engineering applications. The overall anticoagulant results were denoted in Table 1.
Anticoagulant results of electrospun membranes.

PU: polyurethane; AS:
Jaganathan et al. fabricated PU scaffold added with corn and neem oil. The reported blood clotting time of the PU/corn/neem oil was found to increase compared to the pristine PU. There was found to be a 3.47% and 3.75% enhancement in APTT and PT of PU/corn/neem oil than the control suggesting a suitable candidate for the bone tissue engineering [6]. In our study, there was found to be a 4.39% and 5.68% enhancement in APTT and PT time of PU/AS oil than the pristine PU. Hence, the developed biocomponent PU/AS oil membrane showed better compatibility than the reported values suggesting its suitability for the bone tissue engineering. It was reported that the blood compatibility was influenced by several surface parameters such as fiber diameter, wettability, roughness, surface tension, etc. [42]. Jaganathan et al. fabricated PU scaffold added with sunflower and neem oil and it have been reported that the scaffold with smaller fiber diameter exhibited enhanced blood clotting time [7]. In another study, Jaganathan et al. prepared a scaffold based on PU added with carotino oil. It was observed that the prepared composite showed hydrophobic nature resulting in increased blood clotting time [43]. Further, they also fabricated PU scaffold added with mustard oil and reported the reason for the increase in blood clotting time was because of the increase in the surface roughness [40]. Moreover, Chen et al reported in their study that the films with small surface tension also influenced the blood compatibility behaviour [44]. In our study, biocomponent PU/AS oil membrane showed smaller fiber diameter, hydrophobic and enhanced surface roughness, which might have influenced the blood compatibility of the PU membrane.
Conclusion
In this study, the PU incorporated with AS oil was successfully electrospun using the electrospinning technique. The morphology analysis showed that the addition of AS oil in the PU membrane resulted in reduced fiber diameter compared to the control sample. The presence of AS oil in PU membrane was confirmed through hydrogen bond formation. Further, the PU blended with AS oil showed hydrophobic behavior and increased surface roughness than the pristine PU. Moreover, the blood compatibility assessments revealed that the developed biocomponent PU/AS oil membrane possess enhanced anticoagulant nature compared to control. Finally, this study suggests that the newly developed biocomponent PU/AS oil membrane with better physico-chemical characteristics and biological properties enabled them as a potential candidate for the bone tissue engineering applications. It would be interesting to investigate the combined effect of HA in the biocomponent PU/AS membrane and this may throw further light on the feasibility of the biocomponent PU/AS oil membrane in the bone tissue engineering. Moreover, in vitro and in vivo testing of the fabricated biocomponent PU/AS oil membrane would make them suitable in the bone tissue engineering.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
