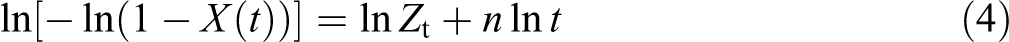Abstract
Isotactic polypropylene (iPP) composites with carbon nanotube (CNT) networks at relatively high loadings could have various applications such as electromagnetic interference shielding and thermal conductivity. The crystallization behavior of iPP inside CNT networks could be very much related to the above properties, which was found to be quite different from that of neat iPP in this work. In CNT networks, CNTs not only act as effective heterogeneous nucleating agents to noticeably increase the onset temperature (more than 18°C) of iPP crystallization but also bring strong confinement on the mobility of iPP chains and then reduce the overall crystallization rate of iPP matrix. It is interesting to find that CNT networks, especially in the case of ultrahigh loading (90 wt%), have remarkable confinement effect on crystallization of iPP, overcoming the heterogeneous nucleation of CNTs, resulting in a decline of crystallization rate of iPP. The nonisothermal crystallization kinetics of iPP in the dense CNT network quite fits to the modified Avrami mode by Jeziorny, even more satisfactorily than the case of neat iPP. When confinement effect is dominated during crystallization, we found that the perfection and size of crystallites are extremely decreased at ultrahigh loading, leading to a very low melting point of iPP (approximately 140°C).
Introduction
Despite the excellent mechanical and electrical properties of carbon nanotube (CNT), it has been proved predominantly that CNTs are superior nucleating agents for various semicrystalline polymers,
1–3
such as isotactic polypropylene (iPP), polyethylene, poly(ethylene terephthalate), poly(butylene terephthalate), poly(
Various parameters could affect the nucleating and crystallization kinetics of semicrystalline polymers induced by CNTs, such as aspect ratio, surface group, and concentration. With regard to the loadings of CNTs in polymer matrix, it is usually found that the efficiency of nucleating effect reaches a maximum with the increase of CNTs loading and then keeps a saturation value. 12,13 For instance, during nonisothermal crystallization of iPP in the presence of CNTs, a gradual elevation of onset temperature appeared when CNT loading increased up to approximately 1 wt%, and subsequently it remained unchanged during the CNTs loading ranging from 1 to 2 wt%. 13 It should be noted that this conclusion is usually drawn in the case of CNT loading less than 5 wt%, probably because a few percentage of loading are normally added into polymer matrix as a nucleating agent. However, when high loadings of CNTs to polymer matrices are added, a remarkable improvement in mechanical properties 14,15 or novel functional properties such as electrical properties, 16 thermal properties, 17 and electromagnetic interference shielding 18,19 can be achieved. For instance, a high connectivity at a high loading of 20 vol% single-walled carbon nanotube (SWNT) is required to improve the thermal conductivity of SWNT/high-density polyethylene nanocomposites, 17 which is usually very hard to tune at a low CNT content. Therefore, it is believed that the crystallization behavior as well as morphology in the case of high CNT loadings will be helpful for a better understanding of the relationship between structure and properties of polymeric composites containing CNTs.
A formation of CNT networks has been revealed at above a critical loading, which can be reflected by a gelation behavior from rheological viewpoint. 9,20 Consequently, the crystallization will be expected to occur in a confined environment of CNT networks since the diffusion of polymer chains into crystallites could be restrained by the CNT networks. 21,22 It is found that the crystallization of semicrystalline polymer in other cases of confined environment results in distinctive and amazing phenomena, which could be much different from the corresponding bulk crystallization. 23 For example, a rare crystallization behavior (e.g. homogeneous nucleated crystallization) occurs during three-dimensional nanoconfined crystallization; 24,25 a unique crystal orientation appears by confining crystalline polymers in nanoscale lamellae and cylinders; 26 a phase transition (like melting behavior) can be altered dramatically by nanoconfinement in confined electrospun nanofibers. 27 However, very few studies have addressed the question regarding the interplay of heterogeneous nucleation and confinement in CNT networks on the crystallization behavior.
Due to the attractiveness of novel crystallization behaviors in the aforementioned confined environment as well as distinct importance of polymeric composites with high CNT loadings, we previously investigated the crystallization of ethylene–vinyl acetate copolymer (EVA) at a high MWNT loading, distinctly indicating the existence of both heterogeneous nucleation and growth confinement. 28 That is, although the peak crystallization temperature (T p) of EVA-containing 10 wt% of MWCNTs increased by more than 5°C, the overall crystallization rate was reduced compared to neat EVA. It was speculated that the strong interactions between the ester groups in the EVA chains and the MWNTs significantly confined and restricted the movement of EVA chains toward the crystal lattice. Thereby, another system of iPP and MWCNTs, probably existing relatively weak interactions between the two substances, was chosen to give more insight into the crystallization in confined environment, which revealed preliminarily that geometrical confinement of CNTs networks still can affect the nucleation and crystallization rate of iPP. 29 In this work, the nonisothermal crystallization of iPP in dense CNT networks at ultrahigh loading (90 wt%) was carried out and analyzed by the model of crystallization kinetics. The results present more details on the nucleation and crystal growth of iPP in the dense CNT networks. The crystalline structure, crystallinity, and melting behavior are also altered dramatically by this highly dense CNT networks at ultrahigh loading, which is not observed in the case of moderate loadings.
Experimental
Materials
iPP (trade marked as T30s) used in this study was provided by Dushanzi Petroleum Chemical Co., China, with the melt flow index of 3 g/10 min (230°C, 2.16 kg), weight-average molecular weight (M w) = 39.9 × 104 g mol−1, and M w/number-average molecular weight (M n) = 4.6. CNTs were provided by Chengdu Organic Chemicals Co. Ltd, the Chinese Academy of Sciences R&D Center for Carbon Nanotubes (China). The CNTs have a length of approximately 10–20 μm and an outer diameter of approximately 30–50 nm and used as received.
Preparation of iPP/CNT composites
A solution dispersion process was utilized to prepare the CNT/iPP composites. The ultrahigh CNT content of 90 wt% was selected to form very dense CNT networks. The experimental procedure was as follows: 9 g of CNTs was added to 900 ml of dimethyl formamide (DMF), and the suspension was subjected to ultrasound and mechanical stirring for 90 min to obtain a uniform dispersion. At the same time, iPP (1 g) was completely dissolved in xylene (100 ml) at 140°C. By dropping the stable CNT/DMF suspension into the iPP/xylene solution, coagulated material precipitated continuously. Thereafter, ethanol was poured into the mixture until no more coagulated material precipitated. The coagulates made up of the CNT/iPP composites were transferred to petri dishes, left at room temperature for 24 h, and then dried in a vacuum at 80°C for 48 h to evaporate any residual solvent. The samples were then compression molded into disks with a thickness of 1 mm and a diameter of 25 mm at 190°C and 10 MPa. For comparison, CNT/iPP sample with 20 wt% of CNT loading and neat iPP were also prepared. The CNT/iPP composites with 20 and 90 wt% of CNTs were noted as PPCN20 and PPCN90, respectively, and the neat iPP as PP0.
Characterization
Differential scanning calorimetric (DSC) measurements were performed on a TA DSC Q200 instrument (New Castle, Delaware, USA). About 5 mg of dried samples were heated to 180°C at a constant heating rate and held at this temperature for 5 min to eliminate any thermal history. Then, the samples were cooled to 50°C at various cooling rates (2, 5, 10, 20, and 40°C min−1) for the investigation of nonisothermal crystallization, followed by heating to 180°C at a rate of 10°C min−1 to study melting behavior. The viscoelasticity of PP0 and CNT/iPP composites was carried out with a stress-controlled dynamic rheometer AR2000ex (TA Instruments) using a parallel plate geometry. The dynamic rheological tests of these samples were then conducted at 210°C under nitrogen atmosphere. Frequency sweeps between 0.1 rad s−1 and 100 rad s−1 were carried out at a low strain (2%), which was within the linear elastic range for these materials. The morphology of CNT/iPP composites was examined with a JEOL JSM 820 scanning electron microscopy (SEM; Japan). The specimens were fractured in liquid nitrogen and coated with a thin gold layer before SEM observation. X-Ray diffraction (XRD) characterizations of the samples were taken on a Rigaku diffractometer (Japan) with copper K α radiation with a wavelength of 0.1542 nm. The scanning rate is 3.6° min−1. FTIR spectroscopy measurements were collected over the wave number range of 600–4000 cm−1 using a Bruker TENSOR 37 FTIR spectrometer (Billerica, Massachusetts, USA) with a deuterated triglycine sulfate detector. The spectra were obtained by averaging 16 scans at a resolution of 2 cm−1 with 1 min intervals. Background spectra used for reduction were collected at the same temperatures as those of the samples. Before the XRD and FTIR tests, the samples were nonisothermally crystallized at a cooling rate of 10°C min−1 with a Linkam CSS-450 high-temperature stage.
Results and discussion
The formation of dense CNT networks in iPP/CNT composites at ultrahigh loading
First, we characterize the geometrical figuration of CNTs in iPP matrix. Figure 1 shows SEM image of PPCN20 and PPCN90. It can be seen from Figure 1 that CNTs are very crowded and contact with each other in iPP matrix due to high loading, which implies that CNTs may form interconnected network in iPP matrix. Second, the presence of CNT networks in a polymer matrix makes the composite like a “jammed solid” or “dispersed gel,” 20 contributing polymer matrix to additional rigidity or stiffness. Thereby, we measured the rheological behaviors of iPP/CNT nanocomposites as shown in Figure 2. Here, the test of PPCN90 is difficult to set out, because it is hard to determine the accurate values of the composites with such a high CNT content. Fortunately, PPCN20 displays elevated storage modulus in comparison with PP0, especially in the region of low frequency. Specifically, with the decrease of frequency, PP0 has a drastic sensitivity of storage modulus to frequency, while the storage modulus of PCN20 already reaches the plateaus at the region of low frequency, which refers to as the sight of CNT networks, 9 suggesting that the addition of 20 wt% CNTs is enough to form CNT networks. 22 Therefore, a dense CNT network can be expected to be achieved in PPCN90.

SEM images of iPP/CNT composites with different loadings: (a) 20 wt% and (b) 90 wt%. SEM: scanning electron microscopy; iPP: isotactic polypropylene; CNT: carbon nanotube.
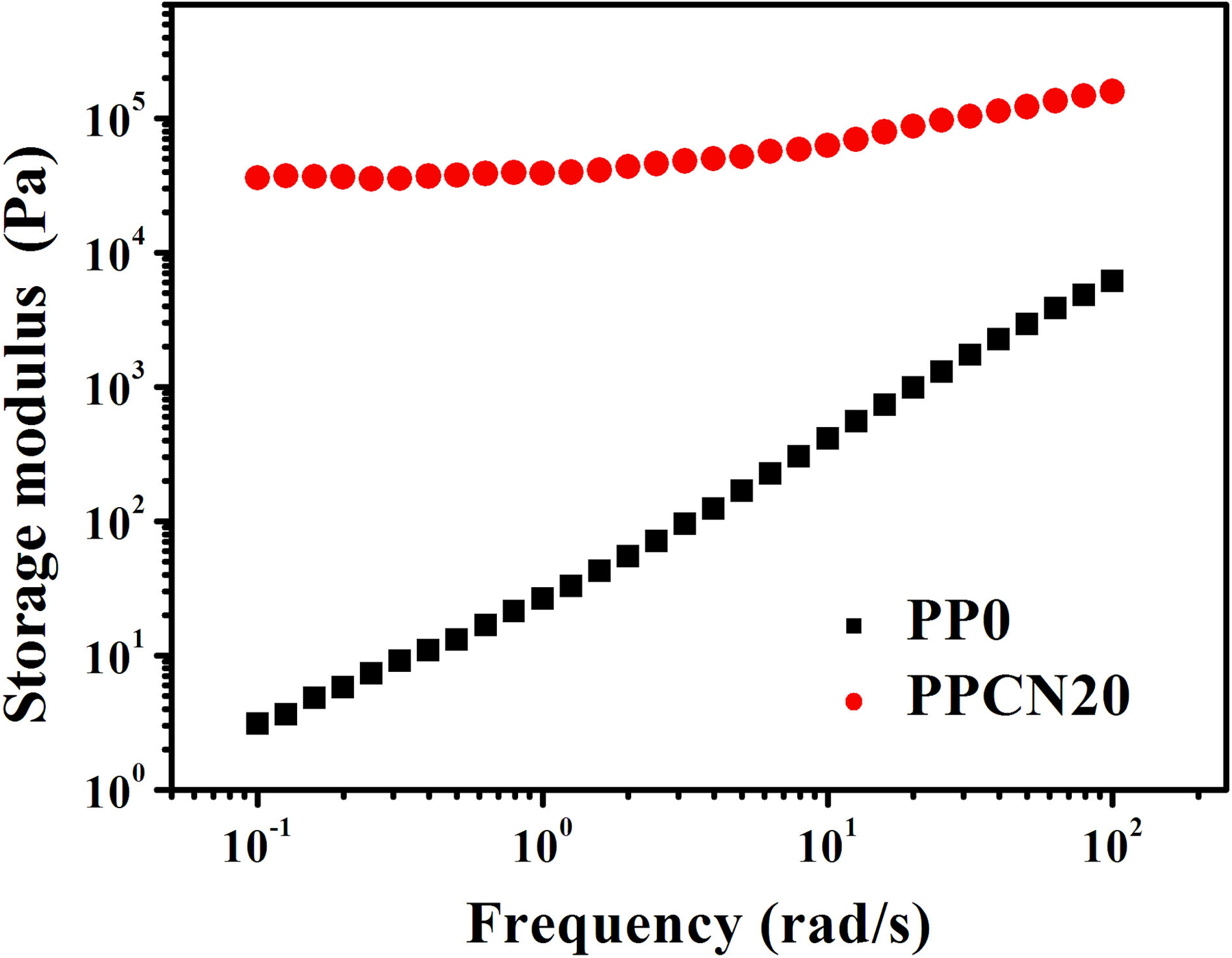
Storage modulus as a function of frequency for PP0 and PPCN20. iPP: isotactic polypropylene; PPCN: carbon nanotube/isotactic polypropylene composites.
Nonisothermal crystallization behaviors of iPP/CNT composites
Figure 3 shows the DSC thermograms of PP0, PPCN20, and PPCN90 during nonisothermal crystallization at various cooling rates. In each sample, the crystallization peak becomes wider and onset temperature (T 0) shifts to lower temperature with increasing cooling rate due to time-dependent nucleation and crystal growth, which is in agreement with many previous studies. 30–32 From these curves, some useful parameters such as the T 0 and T p, and the values of T 0−T p of the samples can be obtained, as shown in Figure 4. It can be seen from Figure 4(a) that the T 0 of PPCN20 increases by more than 18°C compared to PP0 at all the cooling rates, indicating an appreciable heterogeneous nucleation effect of CNT network on iPP crystallization, which could be attributed to the easy attachment of iPP segments in melted state onto the surface of CNTs and subsequently the initiation of iPP nucleation at higher temperature. 31,33 For PPCN90, a substantial addition of CNTs also moves T 0 to higher temperature; while T 0 of PPCN90 shows 5–10°C decline compared with the PPCN20 (see Figure 4(a)), suggesting the nucleation effect decreases as the CNT network becomes denser. The tendency of T p with respect to CNT loading is well consistent with that of T 0 (see Figure 4(b)), showing a more than 10°C of decrease. These results may refer that the dense CNT network restrains the nucleation on the surface of CNTs due to confined diffusion of iPP chains. On the other hand, PPCN20 and PPCN90 exhibit obviously broader peaks, especially at lower cooling rates, which could be confirmed clearly by the value of T 0−T p accounting for crystallization rate to some extent. It can be seen from Figure 4(c) that by incorporating CNT networks, the value of T 0−T p shows an impressive increase (approximately 5°C for PP0, 4–9°C for PPCN20, and 8–14°C for PPCN90), implying preliminarily that the overall crystallization rate at ultrahigh CNT loading is much decelerated by CNT network. The results regarding T 0, T p, and the broadness of crystallization indicate that the heterogeneous nucleation and crystallization rate can be affected distinctively by the geometrical configuration of CNTs.
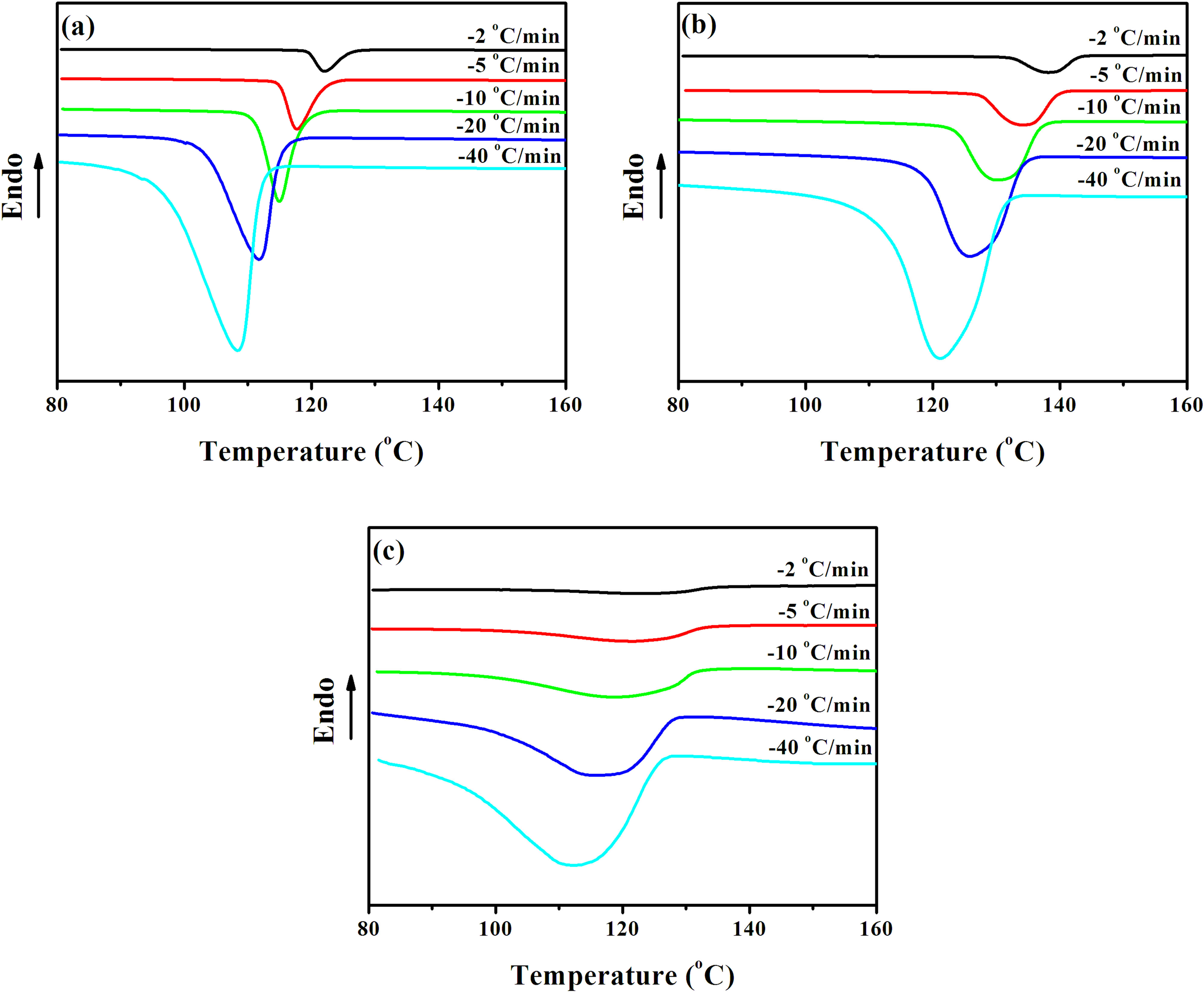
DSC thermograms of the nonisothermal crystallization for (a) PP0, (b) PPCN20, and (c) PPCN90 at various cooling rates. DSC: differential scanning calorimetry; PPCN: carbon nanotube/isotactic polypropylene composites.
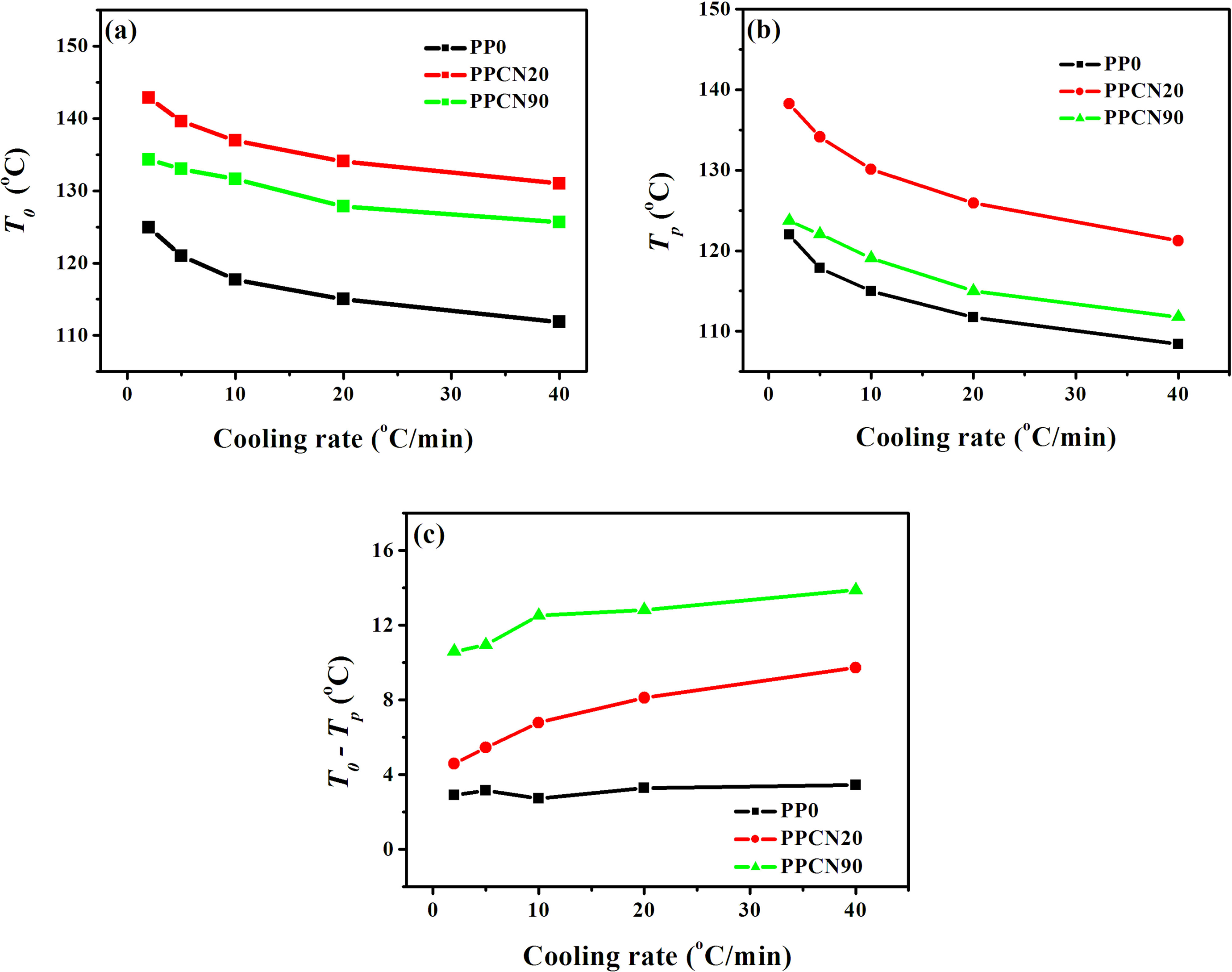
The values of (a) T0 , (b) T p, and (c) T 0−T p of PP0, PPCN20, and PPCN90 at various cooling rates. PPCN: carbon nanotube/isotactic polypropylene composites; T 0: onset temperature; T p: peak crystallization temperature.
According to the DSC curves in Figure 3, the relative degree of crystallinity as a function of temperature, X(T), can be determined by the following equation
34
:
where T 0 and T ∞ represent the onset and the end of crystallization temperatures, respectively, and dH C/dT is the heat-evolution rate. From equation (1), X(T)s for PP0, PPCN20, and PPCN90 at various cooling rates are shown in Figure 5. It can be found that all curves have a sigmoidal shape, indicating the lag effect of the cooling rate upon crystallization. 35,36 The slopes of the curves for PPCN20 and PPCN90 are much lesser than that for PP0 at the same cooling rate, suggesting that the crystallization rates of CNT/iPP composites are much slower than that of PP0. Furthermore, the slope of PPCN90 is obviously less than that of PPCN20, inferring that the crystallization rate decreases significantly with an ultrahigh content of CNTs (i.e. the high density of CNT networks).
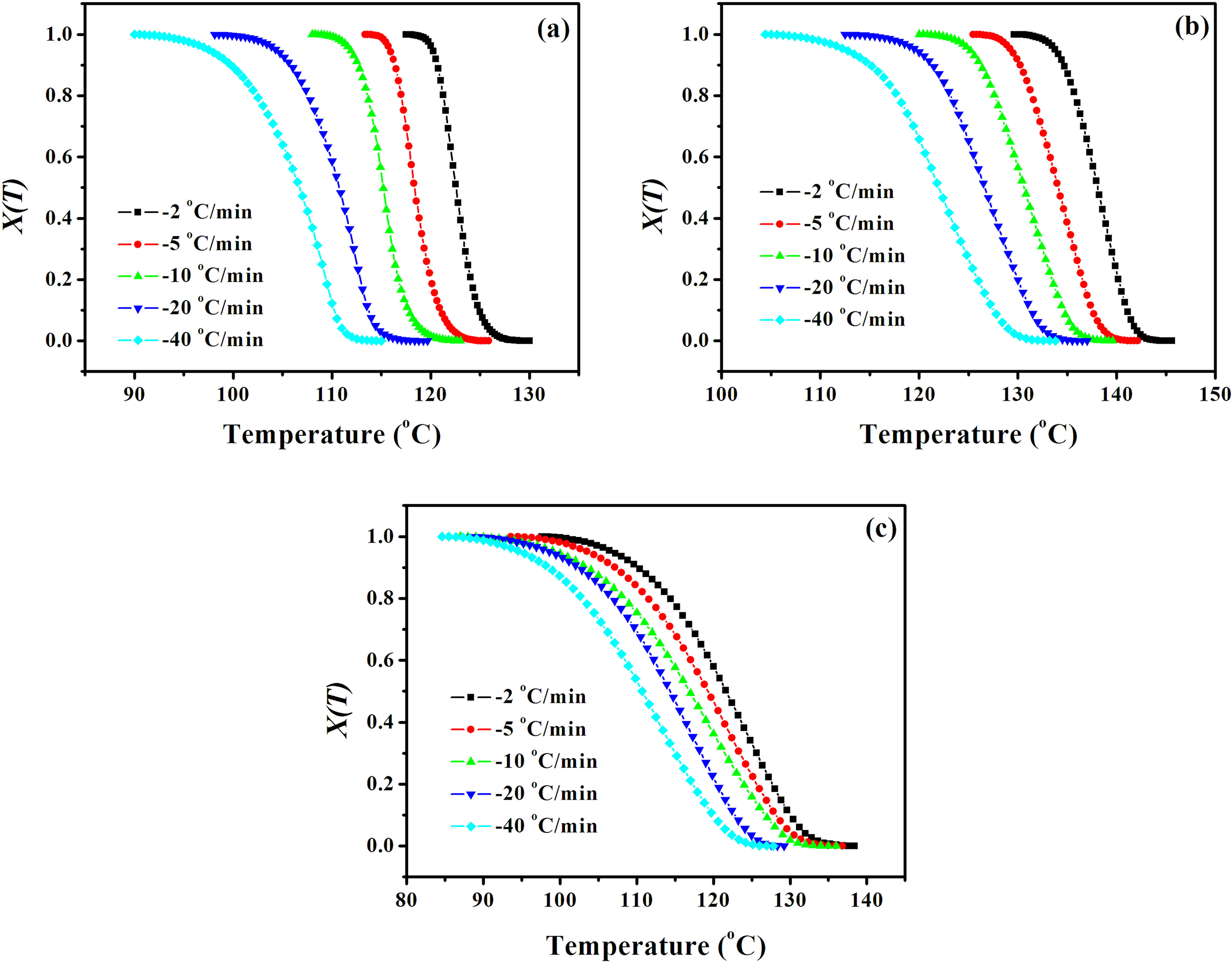
Plots of X(T) versus temperature for (a) PP0, (b) PPCN20, and (c) PPCN90 at various cooling rates. PPCN: carbon nanotube/isotactic polypropylene composites.
In the nonisothermal crystallization process, the crystallization time (t) has relation with the corresponding temperatures by means of
34
:
where T is the temperature at time t and φ is the cooling rate. Thereby, half-crystallization time (t 1/2), the time at 50% of relative crystallinity can be obtained. Figure 6 summarizes t 1/2 at various cooling rates for PP0, PPCN20, and PPCN90. An obvious increase in t 1/2 of iPP in CNT/iPP composites, especially in PPCN90, has been found. For instance, compared with the t 1/2 of 1.82 min for PP0, the t 1/2 reaches 2.43 min for PPCN20 and 5.95 min for PPCN90 at the cooling rate of 2°C min−1, representing 34% and 227% of increase compared to PP0, respectively. The results on T 0−T p, slope presented in Figure 5 as well as t 1/2 provide compelling evidence for the confined crystallization of iPP existing in the CNT networks, and the confinement effect was more obvious in the composite with a denser CNT networks of 90 wt%. This obviously observable phenomenon is quite different from that in recent literature, 37,38 where the crystallization rate of iPP containing low-content CNTs was accelerated observably during nonisothermal crystallization owing to the strong nucleating effect of CNTs on the crystallization of iPP. Therefore, it is reasonably suggested that CNT networks, especially in the case of high CNT loading (PPCN90), may have dramatically influenced due remarkable confinement effect on crystallization of iPP, prevailing over the heterogeneous nucleation of CNTs, resulting in a decline of crystallization rate of iPP.
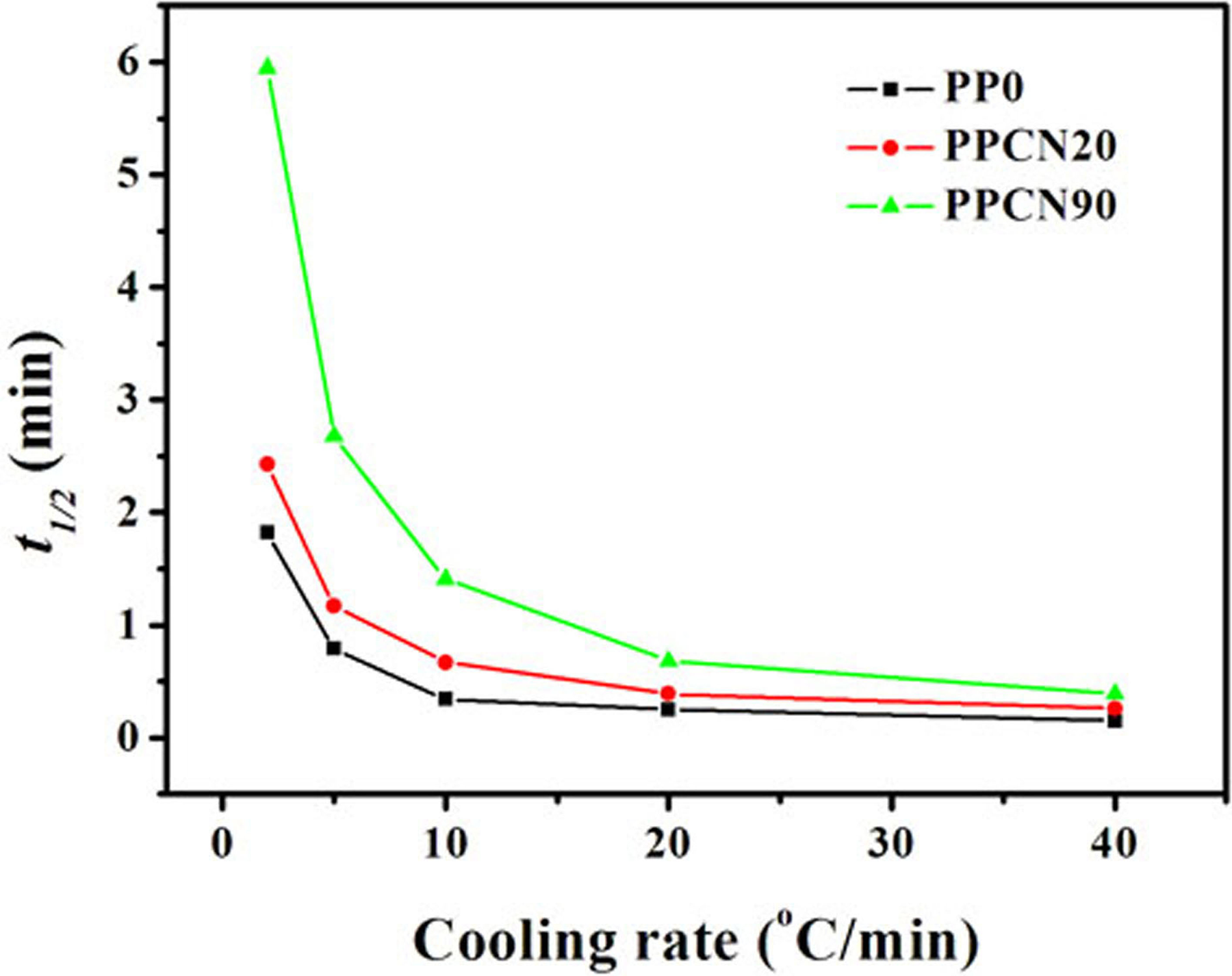
Comparison of crystallization half-time (t 1/2) of PP0, PPCN20, and PPCN90. PPCN: carbon nanotube/isotactic polypropylene composites.
Nonisothermal crystallization of iPP/CNT composites analyzed by kinetics model
To quantitatively describe the crystallization kinetics of iPP crystallization in dense CNT networks, Avrami equation, modified by Jeziorny, is used to analyze nonisothermal crystallization behavior of PP0, PPCN20, and PPCN90
39
:
or
where the exponent n is the Avrami exponent depending on the type of nucleation and the dimensionality of crystal growth, t is the crystallization time, and Z
t is growth rate constant during nonisothermal crystallization. Considering the influence of the cooling rate in the nonisothermal process and assuming that the cooling rate is constant, the parameter, Z
t should be corrected as follows:
where Z c is the modified growth rate constant.
Figure 7 presents the plots of
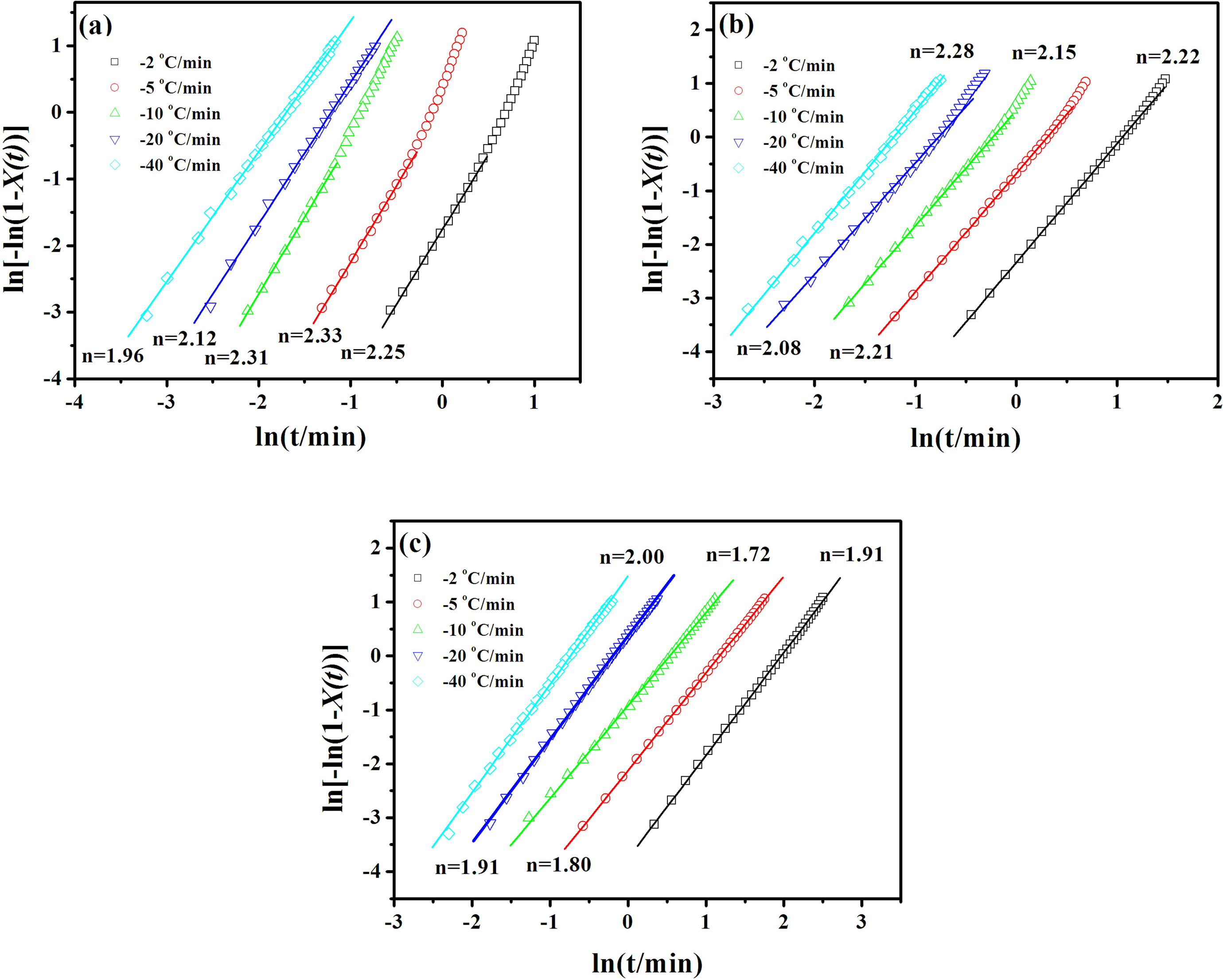
Plots of ln[−ln(1–X(t))] versus ln t for (a) PP0, (b) PPCN20, and (c) PPCN90. PPCN: carbon nanotube/isotactic polypropylene composites.
Values of n, ln Z t, and Z c from modified Avrami equation for PP0, PPCN20, and PPCN90.
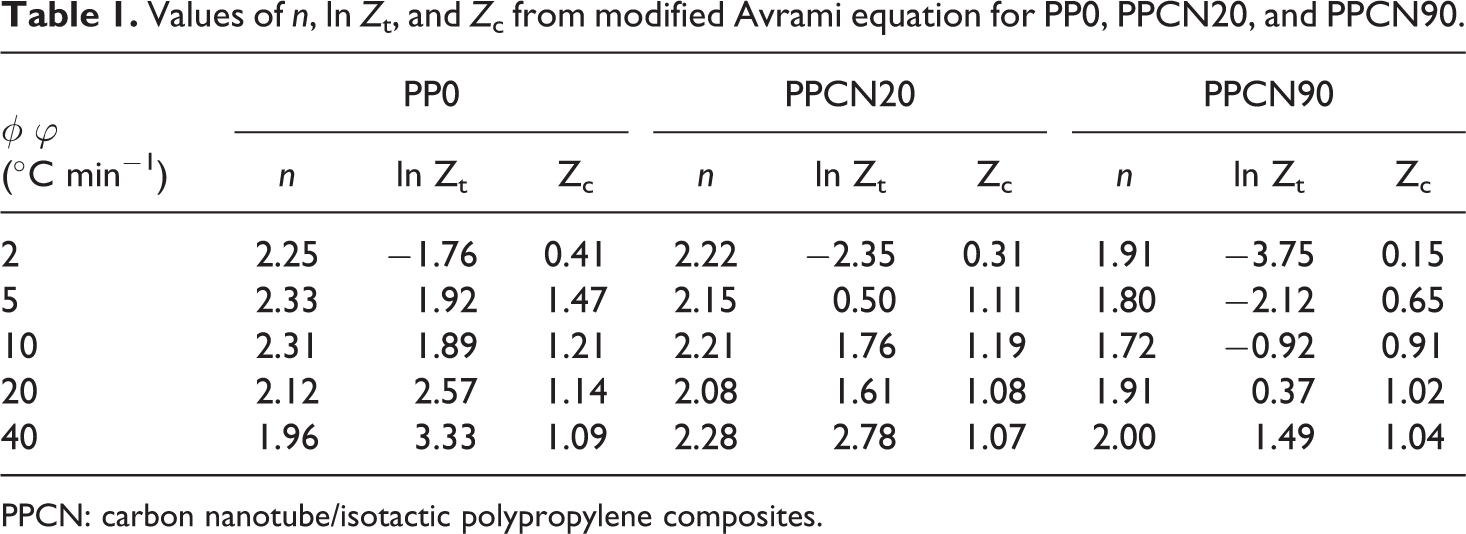
PPCN: carbon nanotube/isotactic polypropylene composites.
Taking into account the great influence of cooling rate on nonisothermal crystallization process, Kissinger proposed a method to determine the crystallization activation energy by calculating the variation of crystallization peak temperature as a function of cooling rate:
42

where φ is the cooling rate, R is the gas constant, and T
p is the crystallization peak temperature. The crystallization activation energy could be determined from the slope of the plot of
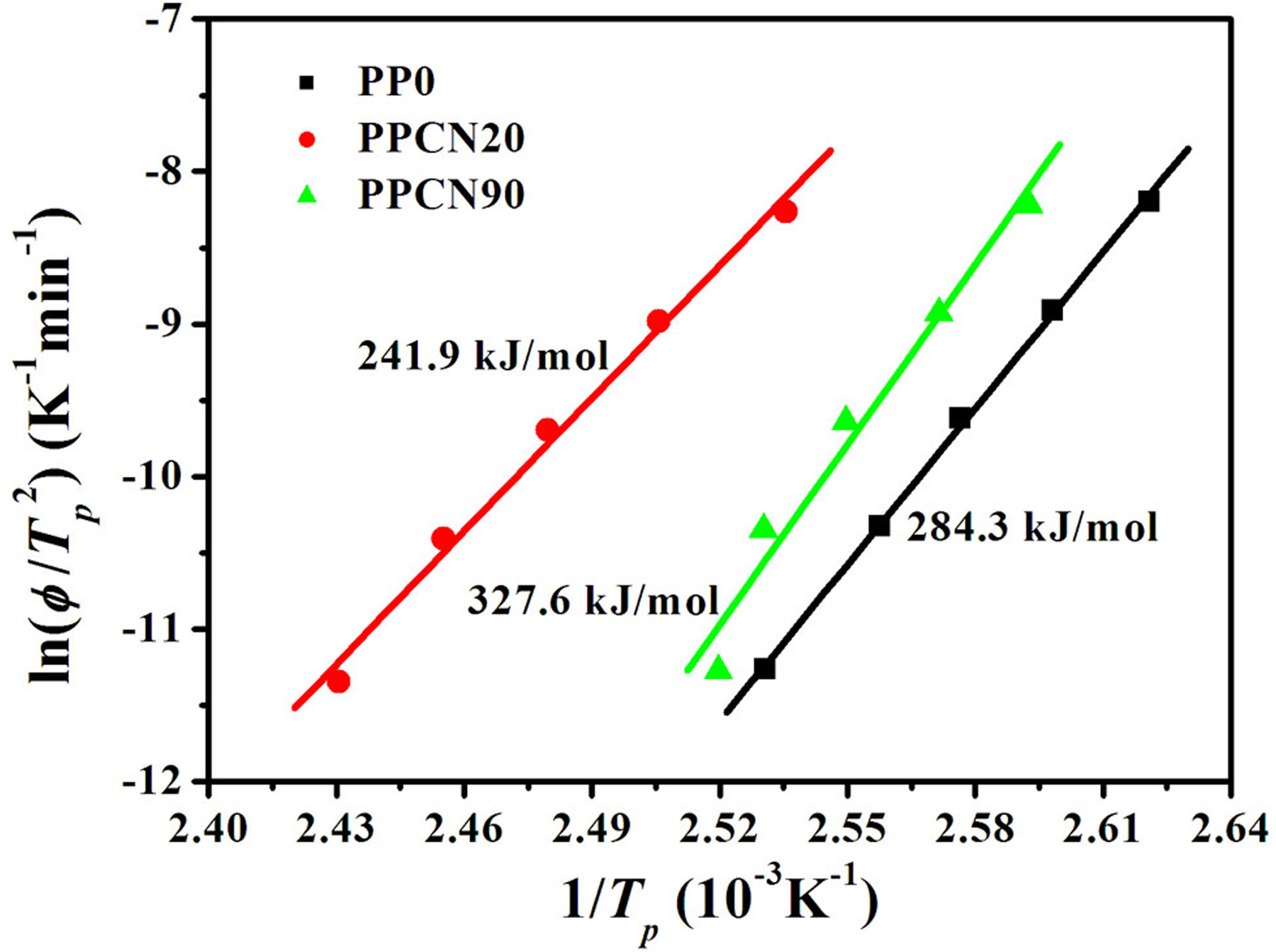
Plots of
Crystalline structure and melting behavior of iPP in highly dense CNT networks
To understand how the confinement affect the crystalline structure and melting behavior of iPP, XRD and FTIR analyses were performed as shown in Figure 9. XRD result shows obvious feature of α monoclinic crystal in the samples of PP0 and PPCN20, which has the characteristic reflections of (110) at 2θ of 14.1°, (040) at 2θ of 16.9°, (130) at 2θ of 18.5°, and (−131) at 2θ of 21.8°. 43 More interestingly, PPCN90 is quite different from the other two, showing two broad reflections at 2θ of 16.9° and 22.5°, respectively, which could be attributed to low perfection of crystallites with preferential growth in highly dense CNT networks. Note that the reflection at 2θ of 26.5° is a characteristic reflection of CNTs due to graphite structure of (002) reflection. 44 The FTIR bands at 940, 1220, 1303, 1167, 841, 998, 900, and 973 cm−1 correspond to 31 helical structures with a degree of order from high to low, 45 and the 1303 cm−1 bond corresponding to the helical length with 13 monomers is usually taken as the crystalline signal. 46 Unlike the distinct peak of 1303 cm−1 for PP0 and PPCN20 (see Figure 9(b) and (c)), the undetectable band for PPCN90 further proves the formation of crystallites with low perfection or very tiny size.
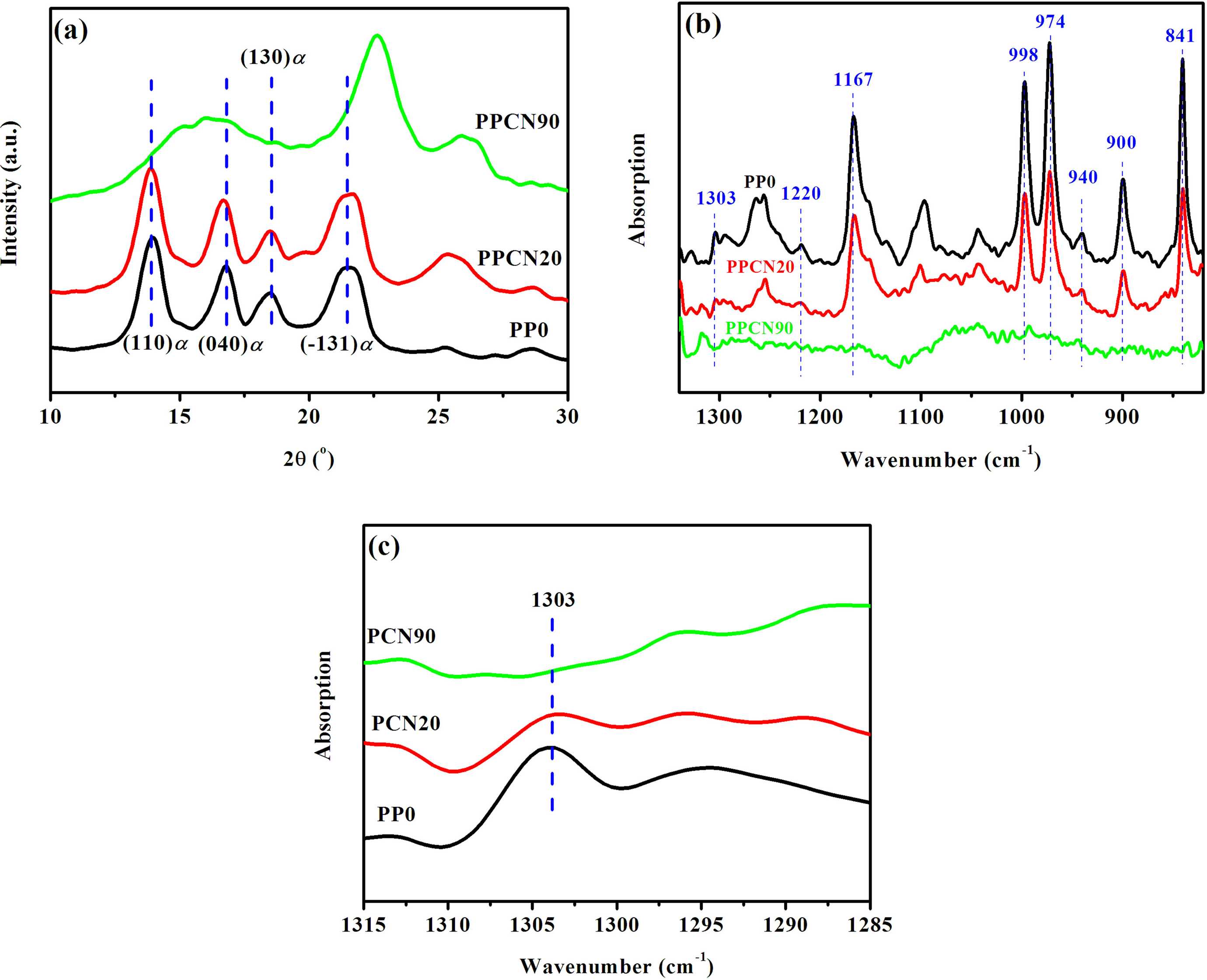
The crystalline structure of iPP and iPP/CNT composites: (a) XRD and (b) and (c) FTIR. Before those tests, the samples were nonisothermally crystallized at a cooling rate of 10oC min−1. XRD: X-ray diffraction; FTIR: Fourier transform infrared; iPP: isotactic polypropylene; CNT: carbon nanotube.
Figure 10 shows the melting curves of PP0, PPCN20, and PPCN90 at a heating rate of 10°C min−1 after the nonisothermal crystallization at the cooling rate of 10°C min−1. The melting point (T m) of PPCN20 is 165.6°C, which is very close to the one of PP0 (165.3°C). We consider that unchanged melting point may be attributed to similar crystallite size (or perfection), and the confinement effect has an influence only on crystallization kinetics. Intriguingly, PPCN90 shows a T m of 142.9°C, which is much lower than that of PP0, suggesting that both crystallization kinetics and crystalline structure have been dramatically influenced due to the confinement effect stem from highly dense CNT networks. To the best of our knowledge, this is the first time to observe the restrained melting of iPP due to confinement, though the melting point of poly(vinylidene fluoride-co-trifluoroethylene) random copolymer shows declined tendency when the pore diameter in long (>10 μm) ordered anodic aluminum oxide (AAO) reduces to below 60 nm. 47 The crystallinity (X c) is calculated according to equation (7), which takes into account the filler fraction of the compound.

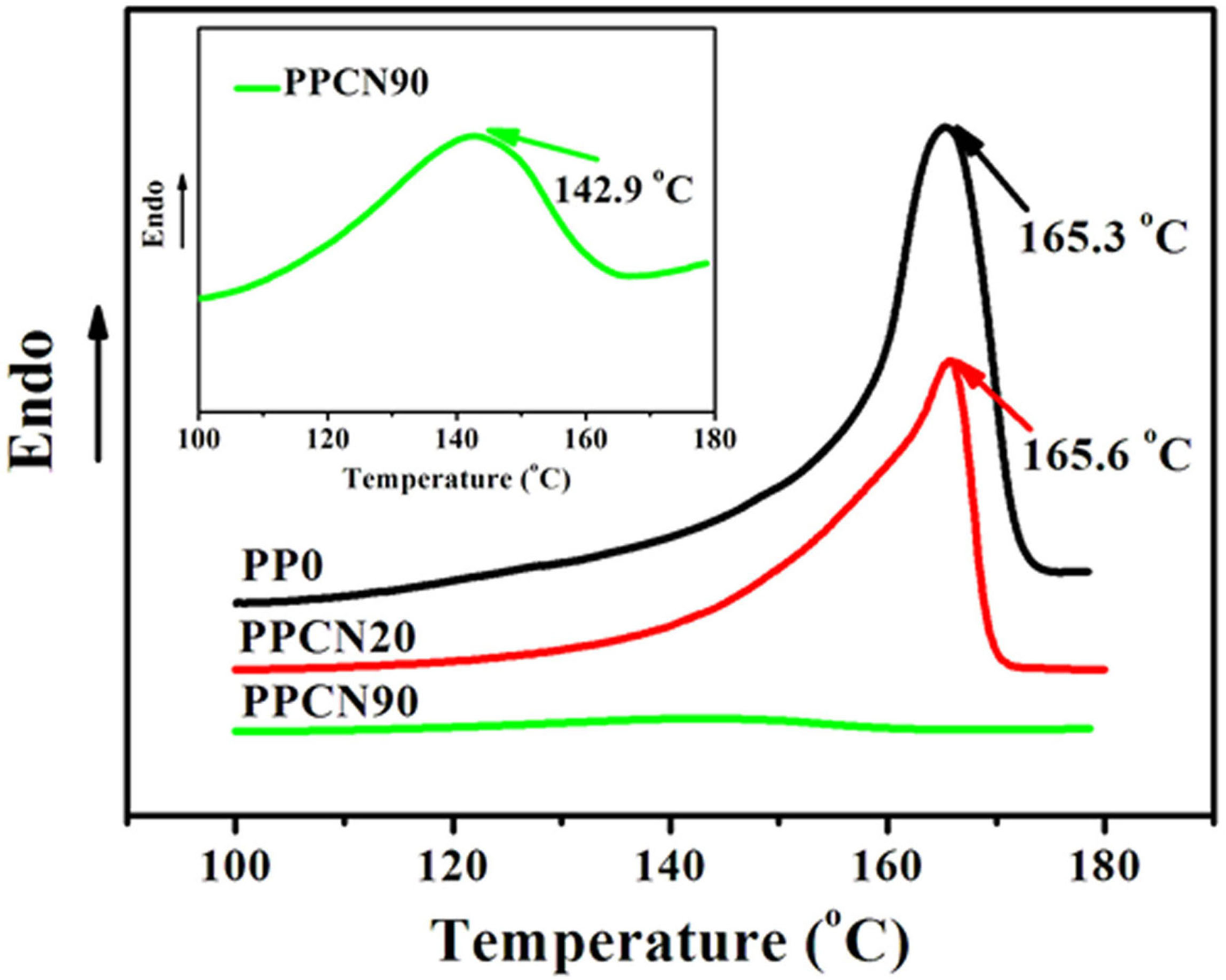
DSC thermograms of the melting behavior of PP0, PPCN20, and PPCN90 at a heating rate of 10°C min−1 after nonisothermal crystallization at a cooling rate of 10°C min−1. DSC: differential scanning calorimeter; PPCN: carbon nanotube/isotactic polypropylene composites.
where ΔHm
is the enthalpy at melting point,
Conclusions
We investigated the nonisothermal crystallization behavior of CNT/iPP composites with ultrahigh CNT loading, in which a high-dense degree CNT network forms. The results reveal that crystallization behavior of iPP inside the highly dense CNT networks is noticeably different from that of neat iPP. On one hand, the CNT networks obviously improve crystallization onset temperatures of iPP, demonstrating the inherent nucleation effect of CNTs on iPP crystallization. On the other hand, the molecular mobility of iPP chains is significantly confined by the highly dense CNT networks; this overcomes the nucleation effect of CNTs, resulting in decreased crystallization rates of iPP. Nonisothermal crystallization kinetics study shows that modified Avrami model can be used to describe satisfactorily the nonisothermal crystallization of iPP in highly dense CNT networks. Additionally, in the iPP composite containing ultrahigh CNT loading (90 wt%), the crystalline structure has been dramatically influenced due to the confinement effect stemming from highly dense CNT networks as well as the crystallization kinetics.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the National Natural Science of China (grant nos: 51273131, 21276168, and 51273114), China Postdoctoral Science Foundation (grant no: 2012M521691), and the Specialized Research Fund for the Doctoral Program of Higher Education (grant no: 20120181120101). Our work was also supported by the Fundamental Research Funds for the Central Universities.