Abstract
N,N′-Dicyclohexyl-2,6-naphthalenedicarboxamide (NU100), a commercial β-nucleating agent (NA) for isotactic polypropylene (iPP), is found dissolvable in iPP melt. Various heating temperatures (T hs) of iPP/NU100 cause different solubilities of NA, resulting in various aggregation morphologies of NU100 in the melt. Moreover, nucleation efficiency of NA NU100 on iPP changes with the T h of iPP/NU100. In this article, the effects of T h on nucleation behaviors of iPP nucleated with different concentrations of NU100 were systematically studied. The results indicated nucleation efficiency of NU100 depended not only on concentration of NA but also on T h of nucleated iPP. With the increase of T h, crystallization peak temperature (T cp) of nucleated iPP decreased remarkably. The whole process that T cp shifted from 126.8°C to 120.8°C was obtained by investigating the nucleation behavior of iPP/0.1 wt% NU100. Polarized optical microscopy observation confirmed lower T cp, representing the crystallization of iPP nucleated by dissolved–recrystallized NA NU100. Further, diverse aggregation morphologies of NA in iPP melt during crystallization were observed. A schematic diagram was proposed illustrating the morphologies formed during recrystallization process of NU100 melted at different T hs.
Keywords
Introduction
Owing to numerous advantages, such as low density and excellent thermal and mechanical properties, isotactic polypropylene (iPP) is widely used in various applications. As one of the semicrystalline polymers, iPP has been intensively researched in the last few years because of its polymorphism characteristics, for example, the monoclinic α-phase, the trigonal β-phase, the orthorhombic γ-phase, and the smectic phase. 1 –4 Recently, β-phase iPP (β-iPP) has attracted great attention due to its remarkable physical properties like higher impact strength and better drawability than α-phase iPP. However, because of the instability of β-iPP, it’s difficult to obtain it in a regular way. It has been reported that β-iPP could be formed under the crystallization conditions of shear field, 5,6 temperature gradients, 7,8 and specific β-phase nucleating agent (NA). 9 –13 Among these, the addition of β-NA is the most effective approach to produce β-iPP. N,N ′-Dicyclohexyl-2,6-naphthalenedicarboxamide (NU100; NJStar) is found to be an efficient β-NA, which has already been commercialized in the past few years. The effect of N,N ′-dicyclohexylo-2,6-naphthalenodicarboxyamide on β-formation in iPP was clearly demonstrated by Romankiewicz and coworkers. 14 –16 It was reported that the maximum of the β-form content (k = 99%) was found for the iPP nucleated with NU100. The highest value of crystallization peak temperature (T cp) was 128°C for iPP/NU100 while that of pure iPP was 116°C. Moreover, the outstanding toughness of iPP nucleated with NU100 was also elucidated. 17 –20
NU100 has been proven capable of dissolving in iPP melt. Varga et al. 26 found that depending on the final heating temperature, NJStar NU100 may be partially or completely dissolved in iPP melt. Because of the different solubility, a wide variety of supramolecular structures may form. Moreover, it was interesting that the peak temperature of nucleated iPP shifted toward lower values with increasing final temperature of heating. To the best of our knowledge, however, the relationship between nucleating efficiency and the solubility of NA NU100 had been investigated primarily in the low-additive concentration range (i.e. 100–300 ppm). Examining this part of concentration range may not provide sufficient instructive information for polymer process since the optimum concentration was generally around 0.2 wt% for NU100.
In this article, a detailed study on the nucleation efficiency of a wide range of concentration of NU100 (from 0.01 wt% to 10 wt%) affected by different heating temperatures (T h: 180°C to 280°C, and 300°C, 320°C for the sample with higher concentrations in addition) was presented. Further, diverse aggregation morphologies of NU100 with different nucleation efficiencies were observed with the aid of in situ polarized optical microscopy (POM) during crystallization. A schematic diagram showing the forming process of these morphologies was proposed.
Experimental
Materials
iPP (trade name F401, weight-average molecular weight = 307,000; number-average molecular weight = 81,000; molecular weight distribution (MWD) = 3.8) used was bought from Yangzi Petrochemical Corporation (China), which has a melt flow index of 3.4 g/10 min. NAs used in this experimental were NJStar NU100 from New Japan Chemical Corporation (Japan). All the other chemicals were of reagent grade, purchased from commercial sources, and used without further purification.
Sample preparation
Various concentrations of NA were uniformly mixed with solution of iPP in boiling p-xylene (138°C). The concentration of iPP was 10 mg ml−1. Then the solution was dropped onto glass substrates. After evaporation of solvent, thin iPP/NU100 film was obtained for further investigation. The pure iPP was prepared by the same method for comparison.
Differential scanning calorimeter
Differential scanning calorimetry (DSC; Diamond, Perkin-Elmer, Waltham, Massachusetts, USA) was carried out to study the crystallization behavior of pure iPP and iPP/NU100. Temperature was calibrated before the measurements using indium as a standard medium. The samples of about 3 mg were heated from 50°C to the predetermined melting temperature (T m) at a heating rate of 10°C min−1 and maintained for 5 min to erase their thermal history. Then the samples were cooled to 50°C at a cooling rate of 10°C min−1.
Polarized optical microscopy
The studies of crystal morphologies of pure iPP and iPP/NU100 were performed with the aid of a polarized optical microscope (BX51, Olympus, Japan) attached with a DP70 digital camera and an LNP94 hot stage (Linkam, UK). The samples were placed between two microscopic slides, heated to the predetermined T m for 5 min to erase any trace of polymer, and then cooled to room temperature at the rate of 10°C min−1. Photographs were taken during the crystallization process.
Results and discussion
Effects of heating temperature on the nucleation behaviors of iPP nucleated with different concentrations of NU100
Concentrations of NU100 studied in this article ranged from 0.01 wt% to 10 wt%. T h was chosen from 180°C to 320°C. The exothermic curves under non-isothermal crystallization conditions after melting at various temperatures were obtained by DSC. Crystallization curves of pure iPP and the samples with different concentrations of NU100 (0.02, 0.1, 3, 5, and 10 wt%) are shown in Figure 1.
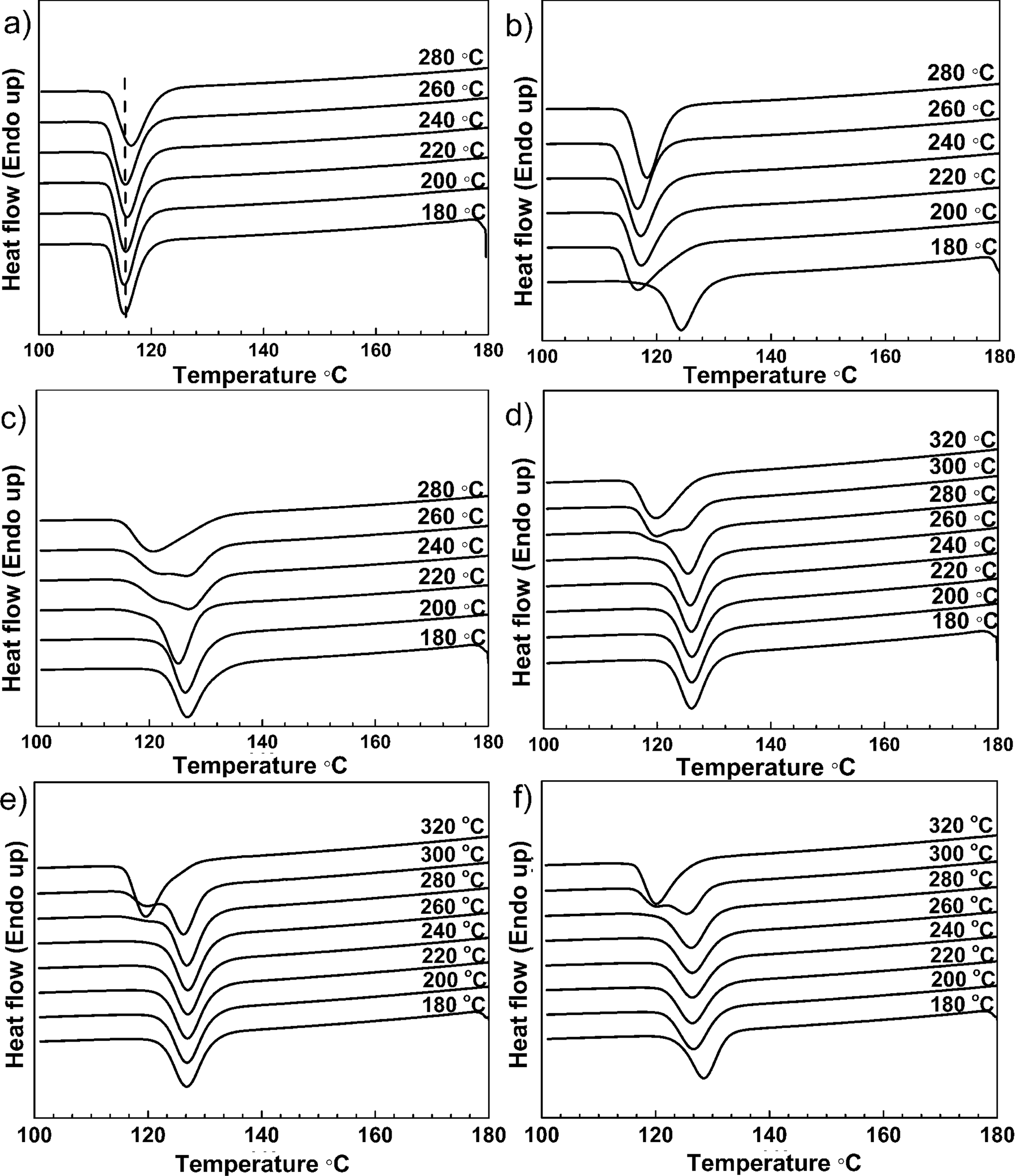
Crystallization curves of iPP and iPP nucleated with NU100 suffered different T hs. (a) iPP, (b) 0.02 wt%, (c). 0.1 wt%, (d) 3 wt%, (e) 5 wt%, and (f) 10 wt%. NU100: N,N′-dicyclohexyl-2,6-naphthalenedicarboxamide; iPP: isotactic polypropylene; T h: heating temperature.
From Figure 1(a) it can be observed that the T cp of pure iPP changed little with the increase of T h. With the addition of 0.02 wt% NU100, T cp of iPP increased to 124.2°C from 115.3°C when T h was 180°C (Figure 1(b)), which indicated that NU100 was an efficient NA for iPP. Meanwhile, it can be noticed that the nucleating efficiency of 0.02 wt% NU100 significantly changed with the increase of T h (Figure 1(b)). When T h was increased to 200°C, T cp of iPP/0.02 wt% NU100 decreased to 116.7°C from 124.2°C. Then it remained nearly the same with the further increase of T h. Normally, the nucleating efficiency of an NA depends on the species and its addition amounts. 21,22 But, here crystallization behaviors of iPP nucleated with NU100 were determined not only by the concentration of NA but by the T h of nucleated iPP.
The whole process of T cp shifting from higher to lower values after being heated at different temperature was detected in the sample of iPP/0.1 wt% NU100 (Figure 1(c)). A single peak crystallization curve with T cp of 126.8°C was obtained when the T h was 180°C. Both the shape of crystallization curve and T cp of nucleated iPP slightly changed when the sample was heated to 200 and 220°C. However, after melting at 240°C, there appeared a shoulder peak around 120°C on the crystallization curve of iPP/0.1 wt% NU100. Then the shoulder peak at lower temperature became more and more obvious with the increase of T h. Finally, the curve turned into a single peak once again, resulting in a great decrease in T cp. It dropped to 120.8°C after being heated at 280°C.
With increasing T h from 180°C to 280°C, T cp of 3 wt% NU100 nucleated iPP remained around 126°C (Figure 1(d)). Similar to iPP/0.1 wt% NU100, there appeared a shoulder peak at lower temperature in the crystallization curve of iPP/3 wt% NU100 after being heated at 280°C. When T h rose to 300°C, the shoulder peak grew larger. Finally, the crystallization curve of nucleated iPP turned into a single peak when the sample melted at 320°C. A decrease in T cp of nucleated iPP took place. T cp of iPP/3 wt% NU100 dropped to 120.1 and 119.8°C when T h was 300 and 320°C. Similar phenomena appeared in the crystallization curves of iPP/5 wt% NU100 and iPP iPP/10 wt% NU100 (Figure 1(e) and (f)). Table 1 summarizes the values of T cp calculated from different samples after melting at various temperatures.
Crystallization peak temperature of iPP and iPP nucleated with NU100 melting at different temperature.
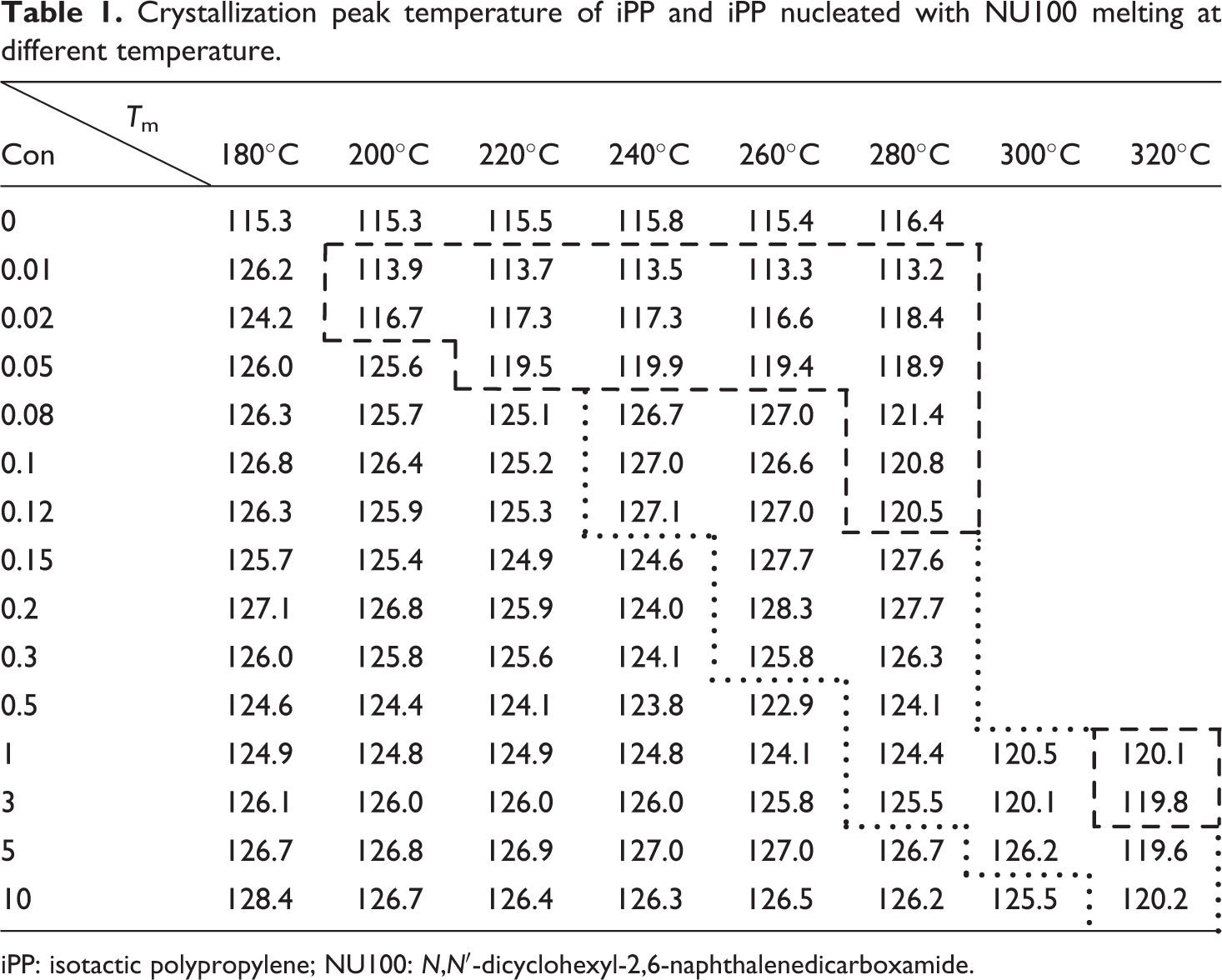
iPP: isotactic polypropylene; NU100: N,N′-dicyclohexyl-2,6-naphthalenedicarboxamide.
According to the results, the process associating with the nucleation of NU100 at varied T hs comprised mainly of three different states. First, T cp of nucleated iPP remained constant with the increase of T h. Then a shoulder peak at lower temperature appeared in the crystallization curve at a specific T h, and it became larger with the increase of T h. T cp of nucleated iPP at this time was determined by the crystallization peak with higher intensity in the shoulder peak, which was noted in the block surrounded by dot line in Table 1. Finally, the crystallization curve of nucleated iPP turned into a single peak again. T cp of nucleated iPP significantly decreased, noted in the block surrounded by dash line in Table 1. The results also showed that the specific T h at which a shoulder peak initially appeared in the crystallization curve of iPP/NU100 was gradually promoted with the increase of the NU100 content.
Different concentration of NA leads to the difference in the density of nuclei during nucleation stage of crystallization, hence resulting in distinct nucleation efficiency on iPP. 23,24 To further understand how T h affected the nucleation efficiency of NU100 and what caused the double peak in the crystallization curves, the crystallization process of nucleated iPP melted at different T h was studied.
Crystallization process of iPP/NU100 melting at different temperature
POM with a hot stage was applied to study the crystallization process of iPP nucleated with different concentrations of NU100. Crystallization morphologies of iPP/0.02 wt% NU100 were investigated first. Figure 2 shows the non-isothermal crystallization of iPP/0.02 wt% NU100 after being heated at 180, 220, and 260°, respectively. Each figure shows three stages during crystallization process of iPP/NU100, molten state at certain temperature, crystallization of NA NU100, and crystallization of iPP induced by NU100. As seen in Figure 2(a), particles of dispersed NAs were detected in the molten iPP at 180°C. During crystallization, the crystal of NA NU100 emerged prior to the crystal of iPP. However, the density of NA was so high that individual crystal shape could not be clearly recognized at this condition. When T h increased to 220°C, it was interesting to find that the crystal of NA NU100 appeared to be a cluster-like crystal (Figure 2(b)), which was a totally different form from the former one. Then iPP crystallized on NU100 aggregation, forming a hexagon-like crystal. Different aggregation morphologies of NU100 led to different crystallization morphologies of nucleated iPP. Like di-(p-methylbenzylidene) sorbitol, NU100 has been proved to be a dissolvable NA in iPP melt. 25,26 As is well known, the solubility of a compound is affected by its concentration and solvent temperature. Change of temperature at which iPP/0.02 wt% NU100 melted generated the variation in the solubility of NU100. According to Figure 2(a), 0.02 wt% NU100 was not fully dissolved in iPP melted at 180°C, while NU100 completely dissolved in the molten iPP when the sample was heated to 220°C. Comparing Figure 2(a) with Figure 2(b), the crystal aggregation form of dissolved NU100 was different from the non-dissolved one when it recrystallized from the melt crystal during the cooling process. When the T h increased to 260°C, the same morphology could be obtained as the sample melted at 220°C (Figure 2(c)), which was in good accordance with their changed T cp. From Table 1, the T cp of iPP/0.02 wt% NU100 dropped from 124.2°C to 117.3°C when T h increased from 180°C to 220°C, and then maintained nearly the same with further increase of T h. Thus, it indicated that dissolved–recrystallized NA NU100, displaying different morphology from the non-dissolved one, had different nucleating efficiencies for iPP. Peak at lower temperature in the crystallization curve represented the crystallization of iPP nucleated with the dissolved–recrystallized NU100, which demonstrated dissolved–recrystallized NU100 had relatively lower nucleation efficiency.

Non-isothermal crystallization process of iPP nucleated by 0.02 wt% NU100 (T m (a) 180°C, (b) 220°C, and (c) 260°C). iPP: isotactic polypropylene; NU100: N,N′-dicyclohexyl-2,6-naphthalenedicarboxamide; T m: melting temperature.
Then we chose a high concentration at which T cp of nucleated iPP remained constant with the change of T h. Figure 3 shows the crystallization process of iPP/3 wt% NU100 melted at different T hs. It can be seen that although the non-dissolved NA became fewer with the increase of T h, there were still abundant needle-like non-dissolved NU100 residuals in melt. Meanwhile, iPP crystals were mostly induced by these non-dissolved ones. Therefore, the nucleating behaviors of iPP/3 wt% NU100 heated at 180, 220, and 260°C appeared constant.

Non-isothermal crystallization process of iPP nucleated by 3 wt% NU100 (T m (a) 180°C, (b) 220°C, and (c) 260°C). iPP: isotactic polypropylene; NU100: N,N′-dicyclohexyl-2,6-naphthalenedicarboxamide; T m: melting temperature.
The crystallization process of iPP/0.15 wt% NU100 heated at 240°C was further investigated. Branch-like NA crystals were observed before iPP crystallized, as shown in Figure 4. It clearly demonstrated the existence of two parts of NA NU100. Dissolved NA first grew at the tip of the non-dissolved needle-like crystal. Then due to the lack of NA “nucleators”, other dissolved NA still recrystallized on this crystal during cooling. Finally, it became a huge branch-like form. During crystallization, two fractions of NU100 crystals respectively induced the crystallization of iPP. Thus the double peak in crystallization curve of nucleated iPP was due to different nucleating efficiencies of different fractions of NU100 crystals. With the increase of T h, there would be more and more dissolved NA. The peak at lower temperature in the crystallization curve would become more remarkable with the increase of T h and finally turned into a single peak with a great decrease in T cp.

Crystallization of NU100 in iPP melt (concentration 0.15 wt%, T m 240°C). iPP: isotactic polypropylene; NU100: N,N′-dicyclohexyl-2,6-naphthalenedicarboxamide; T m: melting temperature.
So far, we have confirmed that non-dissolved and dissolved–recrystallized NU100 differed from each other in nucleation efficiency on iPP. Dissolved–recrystallized NU100 has relatively lower nucleation efficiency. Moreover, several aggregation morphologies of NA in iPP melt during crystallization were observed, such as needle-like, branch-like, and cluster-like crystals. Based on these experimental results, we proposed a schematic diagram showing how these morphologies formed during recrystallization process of NU100 melted at different T hs (Figure 5).
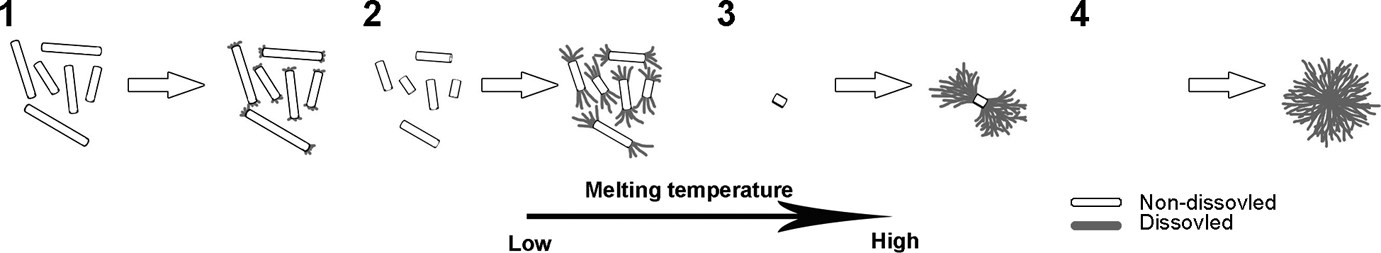
Schematic diagram of NU100 recrystallization from iPP melt at different T ms. iPP: isotactic polypropylene; NU100: N,N′-dicyclohexyl-2,6-naphthalenedicarboxamide; T m: melting temperature.
Initially, abundant needle-like non-dissolved NU100 would be found in the iPP melt because of the low solubility in the case of low T h. These non-dissolved NU100 remained in the iPP melt and served as nucleators for the dissolved NA, which emerged prior to iPP crystals during cooling. Dissolved NU100 was proved to likely recrystallize at the tip of non-dissolved needle-like crystal. 27,28 The increase of T h brought higher solubility of NU100 in molten iPP, leading to the shrinkage of the non-dissolved needle crystal. A mass of dissolved–recrystallized NA made the NU100 crystal into a branch-like form due to the decrease of needle-like nucleus with further increasing of T h. When the T h was high enough, the NU100 could be completely dissolved in the iPP melt. In this case, there would be no such nucleators that existed during crystallization process. Consequently, the crystallization of NA NU100 changed from a heterogeneous nucleation to a homogeneous one forming a cluster-like crystal.
Here, several stages, listed in Figure 5, were observed by investigating iPP nucleated with 0.1 wt% concentration of NU100 melted at different T hs, 180, 240, 260, and 320°C. The whole process of the alteration from a heterogeneous nucleation to a homogeneous one of NU100 recrystallization was obtained. As shown in Figure 6(a) and (b), the non-dissolved NA NU100 was easy to find in iPP melt at both 180 and 220°C. Dissolved NA recrystallized by these nucleators during crystallization like iPP/0.02 wt% NU100 melted at 180°C and iPP/3 wt% NU100 melted at 180, 220, and 260°C. Then, when the sample was heated to a moderate temperature, tremendous branch-like NA crystals like iPP/0.15 wt% NU100 melted at 240°C could be observed. When the sample was heated to 320°C, there was nothing left in the melt. During cooling, the same cluster-like crystal emerged from the melt first as iPP/0.02 wt% NU100 heated to 220°C or above. It is proved that after being completely dissolved in the iPP melt, NU100 would recrystallize in different form and then induce the formation of iPP into a hexagon-like crystal, which showed totally different nucleation efficiency.

Non-isothermal crystallization process of iPP nucleated by 0.1 wt% NU100 (T m (a) 180°C, (b) 220°C, (c) 260°C, and (d) 320°C). iPP: isotactic polypropylene; NU100: N,N′-dicyclohexyl-2,6-naphthalenedicarboxamide; T m: melting temperature.
Conclusions
In this work, variable nucleating efficiency of NU100 was systematically studied by investigating the effect of T h on nucleating behaviors of iPP nucleated with a wide range of concentration of NU100 systematically. The results demonstrated nucleation efficiency of NU100 depended not only on the concentration of NA but also on the T h of nucleated iPP. With the increase of T h, T cp of nucleated iPP decreased remarkably. When the T h reached a specific value, a shoulder peak at lower temperature became visible in the crystallization curve. When T h continued to increase, the shoulder peak became more and more obvious and finally the crystallization curve of nucleated iPP turned into a single peak once again with the lower peak temperature. Results obtained by in situ POM indicated dissolved–recrystallized NA NU100, displaying different morphology from the non-dissolved one, had lower nucleation efficiency for iPP. The changes in the T cp of iPP/NU100 were the results induced by various fractions of NA NU100. Moreover, diverse aggregation morphologies of NA in iPP melt during crystallization were observed. A schematic diagram showing how these morphologies formed was proposed. Once there was existence of abundant NA in the iPP melt, the crystallization pattern of NU100 presented a heterogeneous one. Non-dissolved NU100 in iPP melt served as nucleators for the dissolved NA. Non-dissolved needle-like crystal and dissolved–recrystallized NA made the NU100 crystal a branch-like form, which contained two fractions of NA. With further increasing the T h, the branch-like crystal became more and more similar to cluster-like crystal due to the decrease of needle-like nucleus. When T h was high enough that the NAs could be fully dissolved in the molten iPP, the pattern of NU100 crystallization would change to a homogeneous one, which were confirmed by POM investigations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the National Natural Science Funds of China (U1162110, 21306047), the Program of Shanghai Subject Chief Scientist (10XD1401500), General Financial Grant for China Postdoctoral Science Foundation (2013M541489), and the Fundamental Research Funds for the central universities of China (WA1013012, WA1314051) for financial support.
