Abstract
The present work investigates the properties and structure of epoxy resin (EP) modified with polyurethane (PUR). This latter was prepared using polymeric diphenylmethane diisocyanate and polyethylene glycol (PEG 400) or polyoxypropylene diol with different molecular weights (POPD 1002 and POPD 2002). It was confirmed that 5% of either PUR improved significantly the mechanical properties of EP. Impact strength and critical stress intensity factor values of modified epoxy compositions were increased by 200 and 100%, respectively, relative to pristine samples. Fourier-transform infrared spectra confirmed the occurrence of chemical reactions between reactive groups of EP and PUR. The fracture surface of EP modified with PUR based on PEG exhibited less plastic yielding, while river-like lines with more pronounced plastic deformations were shown by EP modified with 5% PUR with longest chains.
Introduction
Due to its excellent elasticity, high impact strength (IS), and very good chemical and thermal resistance, 1,2 polyurethane (PUR) is employed to produce interpenetrating polymer networks (IPNs) with epoxy resin (EP) for improving the toughness and elasticity of EPs. 3 -18 Most of the conducted works concerned the effects of different type and modifier content of PUR based on toluene diisocyanate (TDI) on the structure and mechanical properties of EP. However, only a few studies have investigated the properties and morphologies of composites based on EP and PUR based on other diisocyanates such as 4,4′-diphenylmethane diisocyanate (MDI) or 1,6-hexamethylene diisocyanate (HMDI).
A series of semi-IPNs based on EP and PUR were also prepared and characterised by Cristea et al. 13 They synthesised PUR from poly(ethylene adipate) diol, MDI and butylene glycol. They studied the damping properties of the obtained semi-IPNs and found that the heat treatment conditions affected significantly the viscoelastic properties of the blends. Moreover, weak softening and completion of the cross-linking were observed upon temperature increase. Wang and Jia 14 modified the diglycidyl ether of bisphenol A with polyol cross-linked PUR. They showed that the thermal stability of prepared IPNs was enhanced with increasing content of grafted PUR and that the tensile and IS reached maximum values at 10% of the polymeric modifier. Shi et al. 19 investigated the morphology and damping properties by dynamic mechanical analysis (DMA) of synthesised PUR/EP blends. DMA analysis indicated that the addition of PUR to EP lowered the glass transition temperature of the polymer matrix. In another work, Wang and Chen 20 modified an EP based on bisphenol A using a PUR prepolymer synthesised from polyether diol and MDI with the use of a coupling agent. They found that the addition of PUR to EP improved the fracture energy upon an increase of modifier content. The study revealed also that the use of hydroxyl-terminated PUR gave a composite with the best mechanical properties in comparison with amine- or anhydride-terminated PUR. Analysis of the scanning electron microscopy (SEM) and transmission electron microscopy pictures showed that phase separation occurred for the obtained composites, which might take part in the toughening mechanism.
Natarajan and Anu prepared PUR/EP blend to be used as matrix for polymer nanocomposites. 21 Obtained results showed that the addition of MDI-based PUR led to a significant improvement of EP mechanical properties than in case of TDI. Moreover, Fourier-transform infrared (FTIR) and differential scanning calorimetry analyses confirmed the formation of chemical bonding between the hydroxyl groups of EP and the PUR isocyanate groups.
Pan et al. 22 studied the effect of PUR based on MDI and HMDI on the EP thermal stability. It was shown that the glass transition temperature and thermal stability decreased with increasing PUR content. However, PUR-modified EP exhibited improved tensile strength and shear strength. Patel et al. 23 characterised the structure and properties of blends based on EP and PUR synthesised from different diisocyanates (TDI, isophorone diisocyanate and HMDI). The IS and elongation at break of the blends containing HMDI were the highest in comparison with virgin EP matrix. However, Ong et al. 24 modified an EP with an aliphatic PUR synthesised from polyethylene glycol (PEG) and HMDI. An amine-cured epoxy composition containing 5 phr PUR exhibited enhanced thermal stability, flexural strength, storage modulus and adhesion strength.
In our previous studies, we modified EP with HMDI- and MDI-based PUR. IS and critical stress intensity factor (
The present work investigates the effect of PURs based on polymeric MDI and different polyols having various chain lengths on the structure, morphology and the mechanical properties of EP.
Experimental
Materials
The following ingredients were used in the study: EP (Epidian 5; Organika Sarzyna, Poland) with a molecular weight of about 380 g mol−1, viscosity at 25°C max 30 Pa s and epoxy number 0.49–0.52 mol/100 g; PEG (molecular weight of 400 g mol−1 and hydroxyl number of 277 mg g−1) from Merck (Germany); polyoxypropylene diol (molecular weight of 1000 g mol−1 and a hydroxyl number of 180 mg g−1) from Rokita Company, Poland; polyoxypropylene diol (molecular weight of 2000 g mol−1 and a hydroxyl number of 46 mg g−1) from Rokita Company; curing agent: triethylenetetramine (Organika Sarzyna Company, Poland); and polymeric 4,4′-diphenylmethane diisocyanate (PMDI) Organodur viscosity = 150 mPa. s obtained from BorsodChem (Czech Republic).
Synthesis of PUR
Three types of PURs were prepared using polymeric diphenylmethane diisocyanate (PMDI) and polyols (PEG and polyoxypropylene diols 1002 and 2002). Polyols were purified by means of azeotropic and vacuum distillation with toluene to eliminate water. First, 50 g of polyol and 0.25 cm3 of dibutyltin dilaurate were stirred for 5 min under vacuum and cooled to below 10°C to slow down the exothermic reaction between isocyanate and hydroxyl groups, and finally PMDI was rapidly added. The reaction was performed in a nitrogen atmosphere with vigorously mixing for 2 min and the temperature was maintained below 10°C until reaching a homogeneous mixture. All the prepared PURs had an isocyanate index equal to 1.05.
Preparation of modified epoxy compositions
Different amounts of PURs (5, 10, 15 and 20 wt %) were added to EP and mixed for 10 min using an ultrasonic homogeniser with an amplitude of 240 µm. The mixtures were then placed in a vacuum oven to eliminate air bubbles. After cooling to room temperature, the curing agent was added (12 g for 100 g of resin), and the mixture was stirred for 5 min with a mechanical stirrer. Finally, the obtained compositions were poured into moulds with standard geometries prior to mechanical testing. The curing process was carried out at room temperature for 24 h followed by post-curing in an air circulating oven for 3 h at 80°C.
Properties and structure of composites
IS was measured according to the Charpy method (ISO 179) using a Zwick 5012 apparatus (Ulm German) on rectangular-shaped samples with 80 mm in length, 10 mm in width, 4 mm in thickness and 1 mm in notch length. Three-point bending tests (ISO 178) were conducted using a Zwick/Roell 010 universal testing machine (Ulm German) on samples of the same dimensions as for impact. The tests were carried out with a deformation rate of 5 mm min−1 and 60 mm distance between the spans.

where
The geometrical factor
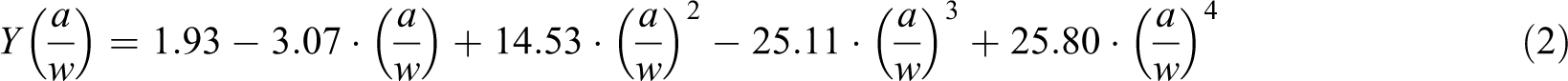
FTIR spectroscopy was performed using a JASCO 6200 spectrophotometer (Japan) recording the IR spectra from 400 cm−1 to 4000 cm−1 with KBr pellets. SEM was used to examine the fracture surfaces of specimens obtained from the impact tests.
Results and discussion
Mechanical properties
Figure 1 shows the effect of added PURs based on PEG 400 (PUR 400), polyoxypropylene diol 1002 (PUR 1002) and polyoxypropylene diol 2002 (PUR 2002) on the IS of EP. It can be observed that all the tested compositions with 5 wt% PUR content exhibited a three-fold increase of IS in relation to unmodified EP. Nevertheless, additional amount of incorporated modifier did not lead to further increase of IS.

IS of PUR-modified EP.
The values of

The improvement of IS and
Moreover, strong interactions may occur between the excess of isocyanate groups and the hydroxyl or amine groups from epoxy system, which leads to the formation of grafted IPNs. 5,12 It should be mentioned that the resistance to slow or fast crack propagation of EP/PUR blends based on polymeric MDI is more important than in case of blends containing low molecular weight MDI, as reported previously. 26 Indeed, the chain length plays an important role in determining the system chains’ flexibility.
Table 1 shows the data from the three-point bending tests. In general, the flexural properties increased and then decreased with increasing amount of PUR. In PUR/EP systems, the ultimate strength can be expressed by the stress value that the material can bear before its breakage and depends significantly on the nature of the interactions as well as the system formed between the matrix and the polymeric modifier chains. 15,17 In fact, maximum increase of the flexural strength was observed for epoxy blends based on 15% PUR 400, 10% PUR 1002 and 5% PUR 2002. The flexural strength increase was 115, 80 and 130%, respectively, in comparison with virgin samples. These results confirmed that the addition of small amount of PUR strongly affects the flexural stress at break of EP independently of the polyol used. However, maximum enhancement of strain at break was noted for blends based on 15% PUR 400, 15% PUR 1002 and 10% PUR 2002. Consequently, the flexural energy to break of EP was also significantly enhanced by PUR incorporation, since it is directly related to flexural strength and strain at break increase. The improvement of mechanical properties might be related to the interpenetration of flexible PUR chains into EP rigid structure as well as chemical reactions between reactive groups between both components. However, Chen et al. 28 attributed the enhancement of mechanical properties of EP modified with PUR based on poly(tetramethylene glycol) to hydrogen bonding within IPN system.
Flexural properties of EP modified with PUR.
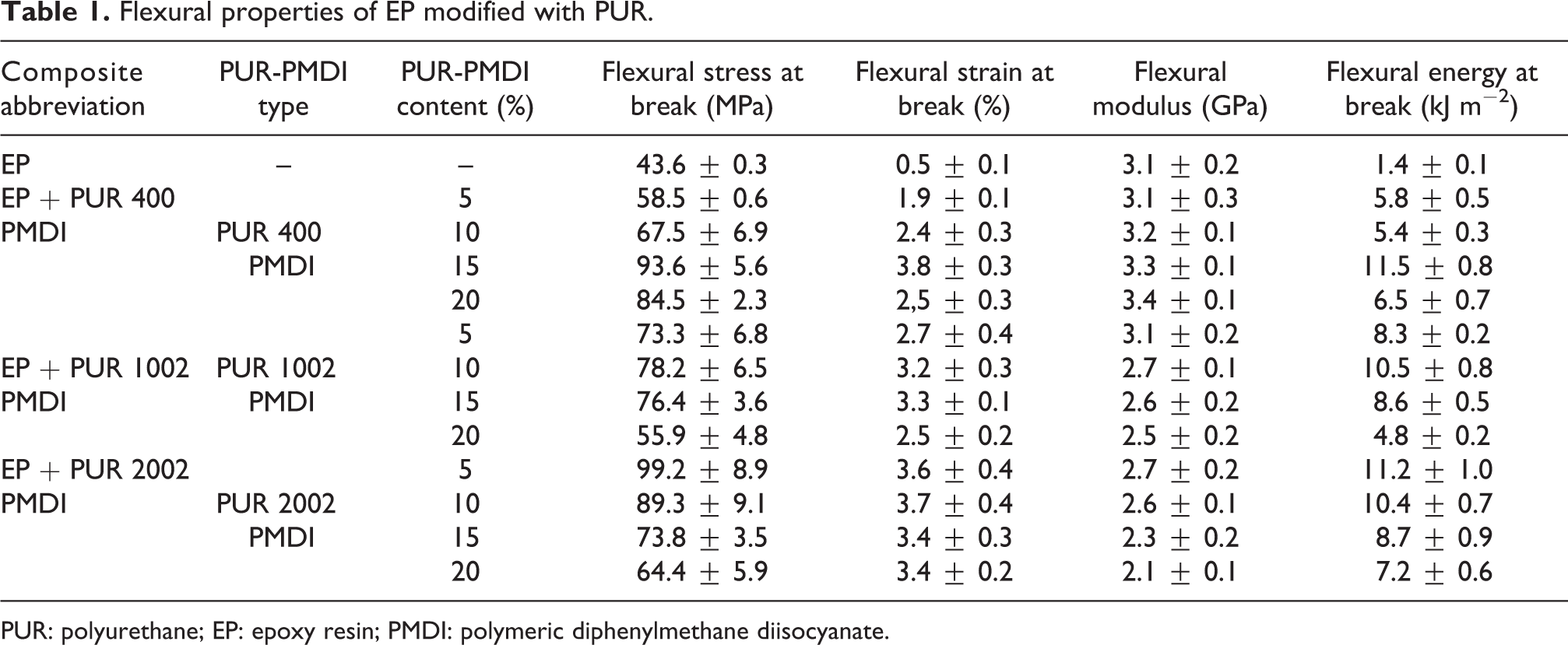
PUR: polyurethane; EP: epoxy resin; PMDI: polymeric diphenylmethane diisocyanate.
According to Wang and Jia, 14 the significant improvement of tensile and IS of EP modified with polyol cross-linked PUR can be attributed to the plasticising effect of dispersed grafted PUR phase. Furthermore, at higher modifier loading, they stipulated that the degree of entanglement between PUR and EP increased, thus restricting the mobility of different molecular chains and leading to the reduction of mechanical properties. A possible reaction between EP- and PMDI-based PUR, leading to grafted structure of the EP/PUR system, is shown in Figure 3.
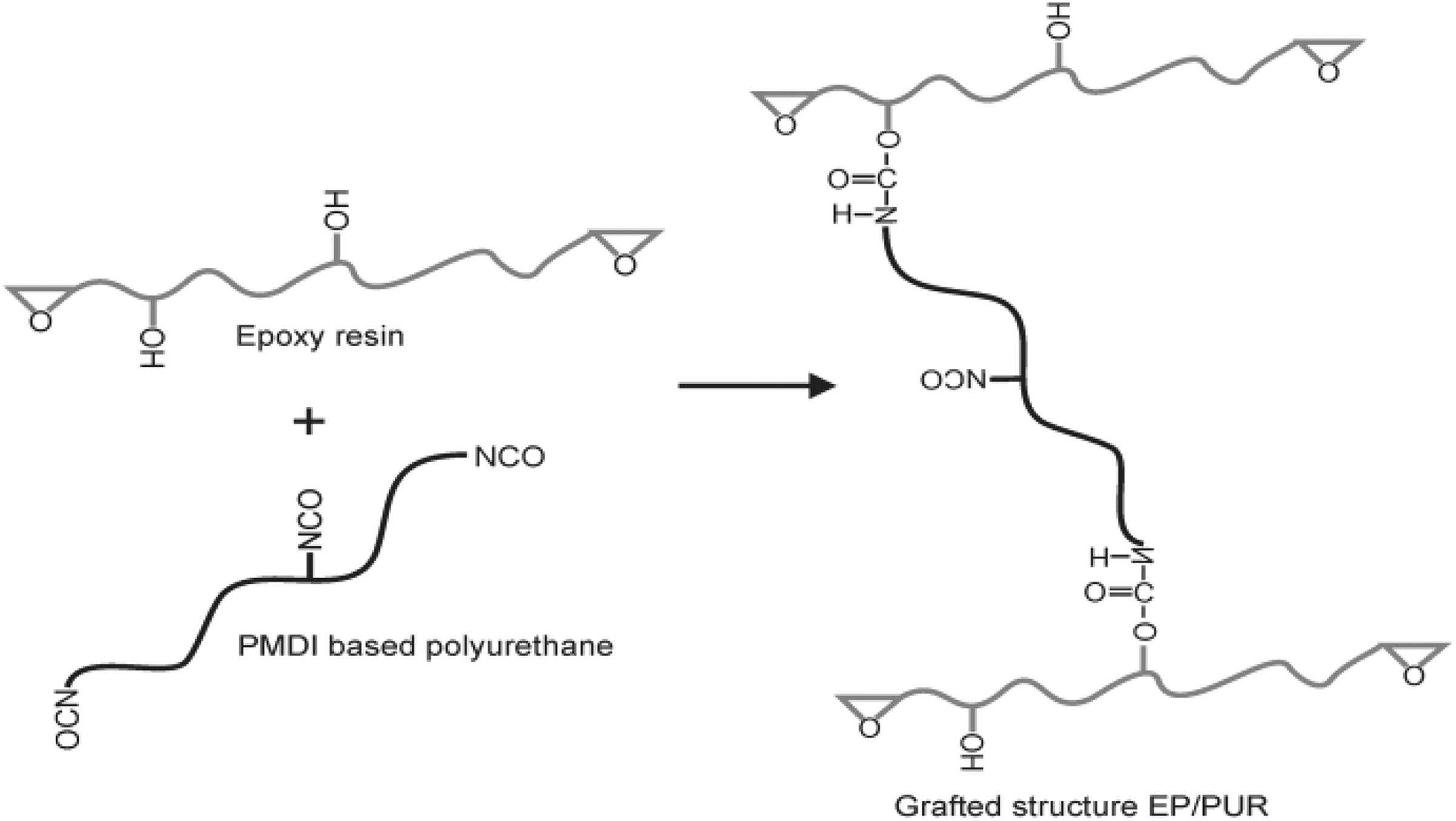
Grafting reaction between hydroxyl (OH) of EP and isocyanate (NCO) groups of PUR.
The flexural modulus of composites based on PUR with shorter chain length (PUR 400) did not vary significantly with modifier addition, most probably due to the combined occurrence of grafting reactions and the plasticisation process. However, we observed about 20 and 30% decrease in the flexural modulus of epoxy composites containing 20% PUR 1002 and PUR 2002, respectively, due most probably to the plasticising effect of a dispersed PUR phase.
Structural analysis
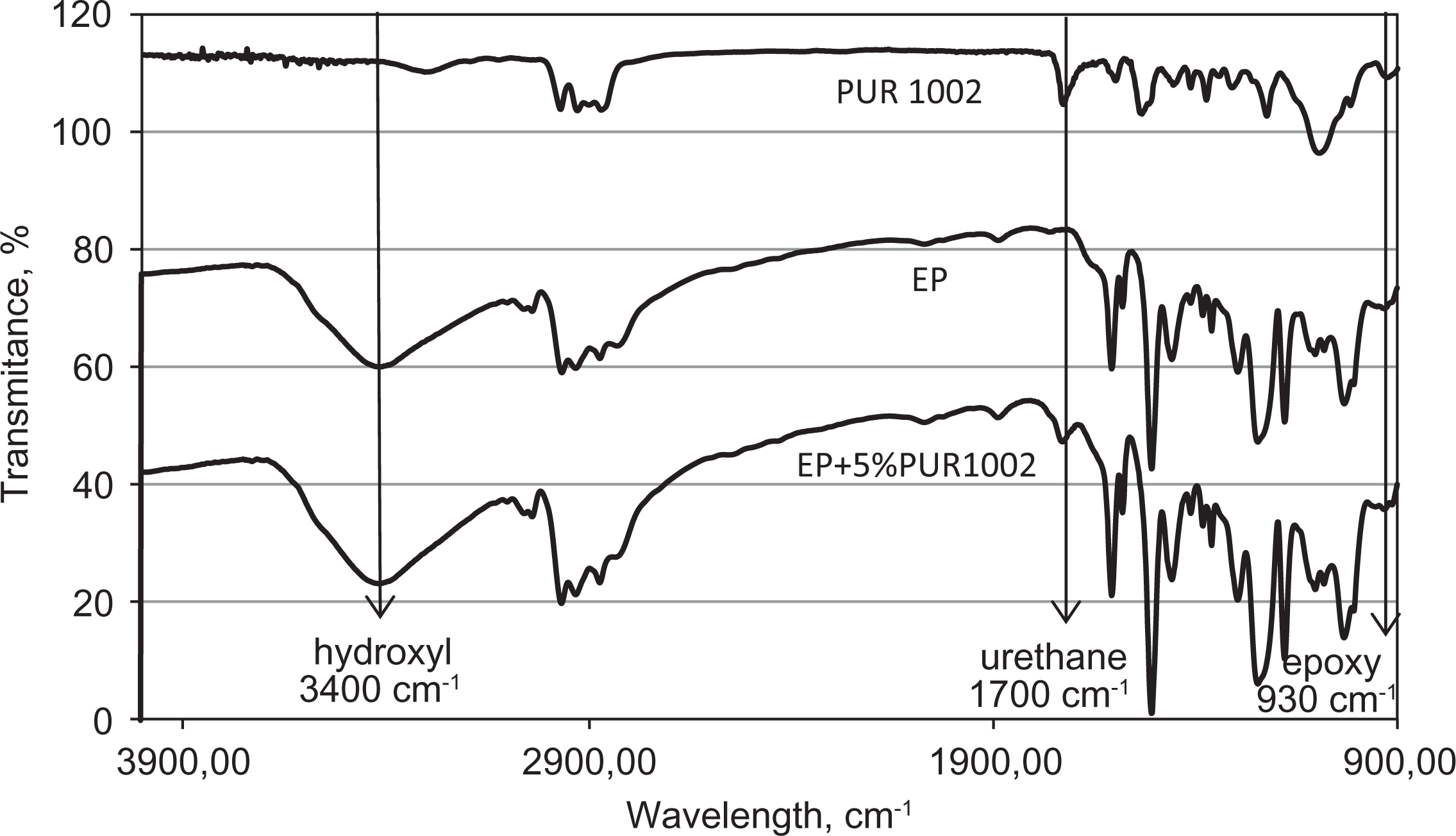
The FTIR spectra shown in Figures 4 to 6 were obtained to indicate the possible chemical reactions that might occur between the polymer matrix and the polymeric modifier. We can observe the characteristic peaks attributed to functional groups of EP appearing at approximately 3420 cm−1 for hydroxyl and amine groups and approximately 930 cm−1 for epoxy groups. However, those appearing at about 1610 cm−1 indicate the presence of urethane and allophanate groups.

FTIR spectra of selected epoxy compositions and PUR 400.
FTIR spectra of selected epoxy compositions and PUR 1002.

FTIR spectra of selected epoxy compositions and PUR 2002.
From Figure 4, it can be observed that the epoxy group peak appearing at 930 cm−1 for the unmodified EP had almost the same intensity as the epoxy composition modified with 5% PUR 400. Moreover, it was shown that the height of the peak associated with hydroxyl groups (3420 cm−1) increased by approximately 10% due to the addition of 5% PUR 400, indicating the possible occurrence of chemical reactions between the reactive groups. Moreover, the absence of any characteristic peak for free isocyanate groups within the wavelength range from 2270 cm−1 to 2100 cm−1 confirms the formation of interchain bonds due to the reaction between isocyanate groups from PUR and hydroxyl groups from EP within an IPN blend, explaining the improvement of mechanical properties of epoxy matrix. In the case of pure PUR 400, the lack of an absorbance peak in this range confirms the complete reaction between isocyanate groups and hydroxy groups to form PUR. However, from Figure 5 representing the spectra of selected epoxy compositions and PUR 1002, it is evident that the addition of PUR resulted in about 20% increase of the hydroxyl groups peak height, thus confirming again the occurrence of chemical reactions between the polymer matrix and the incorporated polymeric modifier. It is demonstrated that the addition of 5% PUR 1002 forms an IPN structure with a high density of interchain bonds, as shown by the disappearance of the urethane peak from PUR at 1700 cm−1. This kind of grafted IPN structure was also reported by other researchers. 12,17
FTIR spectra of PUR 2002, unmodified EP and epoxy composite based on 5% PUR are exhibited in Figure 6. Similar to the previous spectra, the absence of the urethane peak from PUR 2002 at 1700 cm−1 stipulates that chemical reactions occurred between reactive groups of the different IPN components. This might explain the higher values of IS and
Morphology characterisation
SEM micrographs were obtained from impact test samples near the crack tip for neat EP and selected compositions containing 10% PUR. Figure 7 shows the micrographs of cured unmodified EP and of the one modified with 5% PUR. As expected, the fracture surface of unmodified EP (Figure 7(a)) is rather smooth and homogeneous with quite a large glassy area with several microcracks formed during impact tests. This is a typical fracture surface of brittle glassy polymers having low IS and a rather small elongation at break. It should be noted that the micrographs of composites modified with PUR show completely different morphologies in comparison with neat EP. The fracture surfaces of EP compositions modified with PUR 400 and PUR 1002 exhibited a most heterogeneous microstructure with plastic yielding and shear bands, together with flat glassy surface because of the cross-linked network structure (Figure 7(b)). However, river-like lines with more pronounced plastic deformations were shown by the epoxy composition modified with 5% PUR with longer chains (PUR 2002 and Figure 7(d)). A relatively important plastic yielding region was also observed in the case of epoxy composite containing PUR based on polyoxypropylene diol 1002 having a medium chain length (Figure 7(c)). The microstructural changes induced by PUR might be attributed to the flexible modifier chains.

Fracture surfaces of selected epoxy compositions: neat EP (a), 5% PUR 400 (b), 5% PUR 1002 (c) and 5% PUR 2002 (d).
The formation of plastic deformations and shear bands due to PUR incorporation is used to explain the increase of IS, fracture toughness parameter
Conclusions
The following conclusions can be drawn from the obtained results.
The addition of polymeric modifier resulted in the improvement of the mechanical properties of the EP. The composition containing 5 wt% PUR exhibited maximum enhancement of IS and
The fracture surface morphology analysis suggested that the improvement of mechanical properties of brittle epoxy matrix was due to the heterogeneous microstructure together with plastic yielding and shear bands induced by the incorporated polymeric modifier. The FTIR spectra analysis confirmed the occurrence of chemical reactions between reactive groups of polymer matrix and modifier, thus explaining further the improvement of the fracture toughness of EP.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
