Abstract
Polyurethanes (PURs) synthesized using hexamethylene diisocyanate (HDI) and polyethylene glycol with molecular weights of 400 g mol−1 and 600 g mol−1 (PUR 400 HDI and PUR 600 HDI) or polyoxypropylene diol (POPD) of molecular weights 1002 g mol−1 and 2002 g mol−1 (PUR 1002 HDI and PUR 2002 HDI) were used as modifiers for diglycidyl ether of bisphenol A. It was found that maximum improvement in impact strength and critical stress intensity factor (
Introduction
Epoxy resins (EPs) are widely used as matrices for high-performance composite materials, electric insulators, printing circuit boards, surface coatings and adhesive for metals. However, cured EPs exhibit low impact strength (IS), poor resistance to crack propagation and small elongation at break. 1 –3
In the last few decades, a lot of studies were carried out to reduce the brittleness and improve the mechanical properties and at the same time maintain the thermal stability of EPs. One of the methods used to improve the EPs mechanical properties included the formation of interpenetrating polymer networks (IPNs).
Due to its excellent elasticity, polyurethane (PUR) was used to improve the resistance to brittle fracture of EP and also to improve other mechanical properties. Most of the conducted studies concerned the modification of EP with PUR based on toluene diisocyanate (TDI). 4 –9
Hsieh and Han 4 evaluated the mechanical properties of graft IPN structure based on diglycidyl ether of bisphenol A and PURs based on polyols with different chain length. They showed that the significant improvement in the tensile strength was not only due to the grafted structure but also due to the shortness of PUR chains. In a separate work, Raymond and Bui 5 modified EP using urethane prepolymers based on TDI and castor oil with a molar ratio NCO/OH equal to 1.5. They confirmed that hydroxyl groups of EP and that of rycyn oil reacted with the excess of isocyanate groups, therefore enhancing the properties of the polymer matrix. Moreover, the thermomechanical tests demonstrated a good compatibility between the polymer matrix and the incorporated modifier.
However, Stefani et al. 6 investigated the properties of EP modified with urethane prepolymer containing blocked isocyanate groups. PURs with different NCO/OH ratios were synthesized and used as epoxy modifiers. Composition containing PUR with an excess of isocyanate groups showed increased IS without a decrease in the elastic modulus or the compressive yield stress. However, within the stoichiometric system, it was observed that it had more enhanced IS but with a pronounced decrease in the stiffness and resistance to compression.
Li and Mao 7 prepared semi-IPN EP/PUR, and evaluated their thermal and mechanical properties demonstrating that the two polymers are compatible at weight ratio of EP/PUR equating 70/30. Moreover, the scanning electron microscopic (SEM) analysis indicated that the obtained semi-IPN had a two-phase continuous structure that changed with different weight compositions. Also, the occurrence of the glass transition temperature between those of EP and PUR was related to the EP/PUR interface.
In order to further increase the level of reaction between EP with modifier, it is appropriate and recommended to use a PUR containing other reactive groups. For example, Wang and Chen 8 modified diglycidyl ether of bisphenol A with PUR prepolymer terminated with aromatic amine groups as well as phenol hydroxyl groups. They determined the effect of the diol type and content in PUR on the final properties and morphology of the obtained compositions. They concluded that EP modified with PUR with phenol hydroxyl groups exhibits better resistance properties and higher glass transition temperature than EP containing PUR with amine groups. PUR can be linked to EP through physical entanglements and chemical bonding to form grafted IPN.
The effect of TDI-based PUR content on the mechanical and thermal properties of diglycidyl ether of bisphenol A cured with triethylene tetramine was also investigated by Pokropski and Balas. 9 They confirmed a significant improvement in the mechanical properties of the polymer matrix with PUR containing excess of isocyanate groups.
In another work, Harani et al. 10 confirmed that isocyanate-terminated PUR reacts with EP leading to a significant fracture toughness improvement. Moreover, the use of chain extender with PUR prepolymer caused a sevenfold increase in IS and almost twofold increase in critical stress intensity factor in comparison with the unmodified EP. The improvement in the EP fracture toughness was attributed to the grafting reactions between the polymer matrix and the modifier.
PUR with an excess of isocyanate groups was synthesized and used as modifier for EP. 11 It was confirmed that maximum improvement in the fracture toughness was reached with PUR with highest content of isocyanate. Furthermore, the elastic modulus of the polymer matrix decreased due to PUR incorporation, implying the softening of the former. Moreover, infrared spectra indicated that an excess of isocyanate groups led to a grafting process between the modifier and the matrix, explaining the toughening of the latter.
Ismail and colleagues 12 confirmed that conventional EP modified with an aliphatic PUR synthesized from poly(ethylene glycol) (PEG) and hexamethylene diisocyanate (HDI) without a solvent exhibited enhanced thermal stability, flexural strength, storage modulus and adhesion strength.
More recently, we have modified diglycidyl ether of bisphenol A with PURs obtained from different polyols and TDI or diphenylmethane diisocyanate (MDI). 13,14 It was shown that the addition of 10% or 15% of PUR prepared from TDI gave compositions with enhanced mechanical properties. Moreover, it was shown that the incorporation of 5% and 10% PUR based on MDI resulted in the formation of a grafted IPN structure with improved mechanical properties. 13 Composites containing PUR based on MDI and higher molecular weight PUR (obtained from polyoxypropylene diols (POPD)) with long flexible segments exhibited higher IS, while PUR prepared from polyethylene glycol (PEG) had a higher flexural energy to break and a higher flexural modulus.
The purpose of the present work was to investigate the effect of PUR based on HDI and polyols with different molecular weights on the mechanical properties and morphology of EP. The polyols were based on PEG and POPD.
Experimental
Materials
The following ingredients were used in the present work.
The clear liquid EP with an epoxide equivalent weight of 185–190 g equiv− 1 and number average molecular weight of 380 was diglycidyl ether of bisphenol A (Epidian 5). The curing agent was triethylene tetramine (trade name Z1). Both the EP and curing agent were obtained from Organika Szarzyna Co. (Nowa Sarzyna, Poland). PEG with the molecular weights of 400 g mol−1 (PEG 400 having hydroxyl number = 277 mg KOH g−1) and 600 g mol−1 (PEG 600, hydroxyl number = 169 mg KOH g−1) were purchased from Merck (Germany).
POPD with the molecular weights of 1002 g mol−1 (POPD 1002 with hydroxyl number of 180 mg KOH g−1) and 2002 g mol−1 (POPD 2002) with hydroxyl number = 46 mg KOH g−1) were purchased from Rokita Co. (Poland). The diisocyanate was HDI from Merck and the catalyst was dibutyltin dilaurate (Merck).
Preparation of PUR prepolymer
First, the dehydratation of polyols was carried out by means of azeotropic distillation using toluene. The hydroxyl number was then estimated after removal of the solvent under lower pressure using Polish norm PN-93/C-8905203.
The synthesis of PUR was carried out in a reactor under a nitrogen atmosphere. First, 50 g of polyol and 0.2 g of dibutyltin dilaurate were introduced in the reactor then the diisocyanate was added. The reaction was conducted for 10–15 min maintaining the temperature below 20°C and ended with the increase in PUR viscosity.
Samples preparation
Different epoxy compositions containing 5%, 10% and 15% PUR were prepared. The compositions were obtained from HDI and PEG with the molecular weights of 400 g mol−1 and 600 g mol−1 (PUR 400 HDI and PUR 600 HDI). However, the compositions designated PUR 1002 HDI and PUR 2002 HDI were prepared using HDI and POPD with 1002 g mol−1 and 2002 g mol−1 of molecular weights.
First, PUR and EP were mixed for 5 min in a homogenizer at 2400 r/min. The compositions were then placed in a vacuum oven to remove air bubbles. A stoichiometric amount of hardener (Z1) was added and mixing continued for 5 min. The mixture was then poured to coated metallic forms with adequate geometries for 48 h curing. Postcuring was carried out for 3 h at 80°C.
Properties evaluation
The critical stress intensity factor
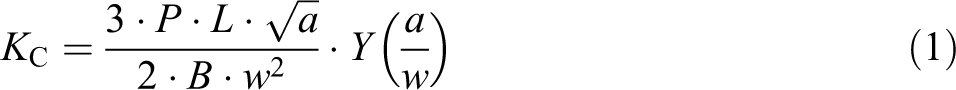
The geometric factor was calculated according to the following equation
15
Impact strength was measured according to Charpy method using a Zwick 5012 apparatus (ISO 179) on the samples of 80 × 10 × 4 mm3 and 1 mm notch. Three-point bending tests (ISO 178) were carried out at room temperature on specimens of the same dimensions as for impact tests using an Instron 5566 at deformation rate of 5 mm min−
1. The distance between the spans was 60 mm. Five samples were used for each data point of impact and flexural tests.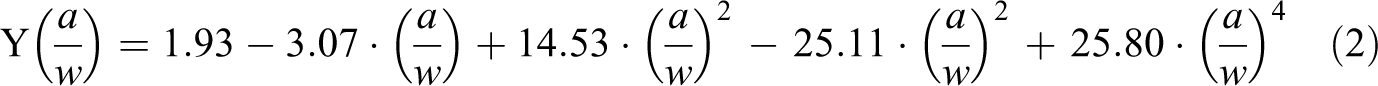
Structure and morphology analyses
Fourier transform infrared (FTIR) spectroscopy was performed on a IRT C JASCO spectrophotometer recording the infrared spectra from 450 to 4000 cm−1.
Scanning electron microscope (Hitachi S-2460 N) was employed to examine the fracture surfaces of specimens obtained from the impact tests.
Results and discussion
Mechanical properties
Figure 1 shows the effect of PUR based on PEG (PUR 400 HDI and PUR 600 HDI) and POPD (PUR 1002 HDI and PUR 2002 HDI) content on the IS of EP. It can be noted that the maximum IS value, representing approximately 150% increase in relation to unmodified epoxy sample, was obtained by the composites containing 15% PUR 400 (abbreviation of PUR 400 HDI) and 5% PUR 1002 (abbreviation of PUR 1002 HDI). Such IS improvement due to PUR incorporation might be related to the presence of flexible segments of PEG and POPD and most probably also attributed to the formation of an IPN structure between EP and the polymeric modifier. It has to be mentioned that the addition of more than 15% PUR was not possible because of viscosity problems of the compositions.
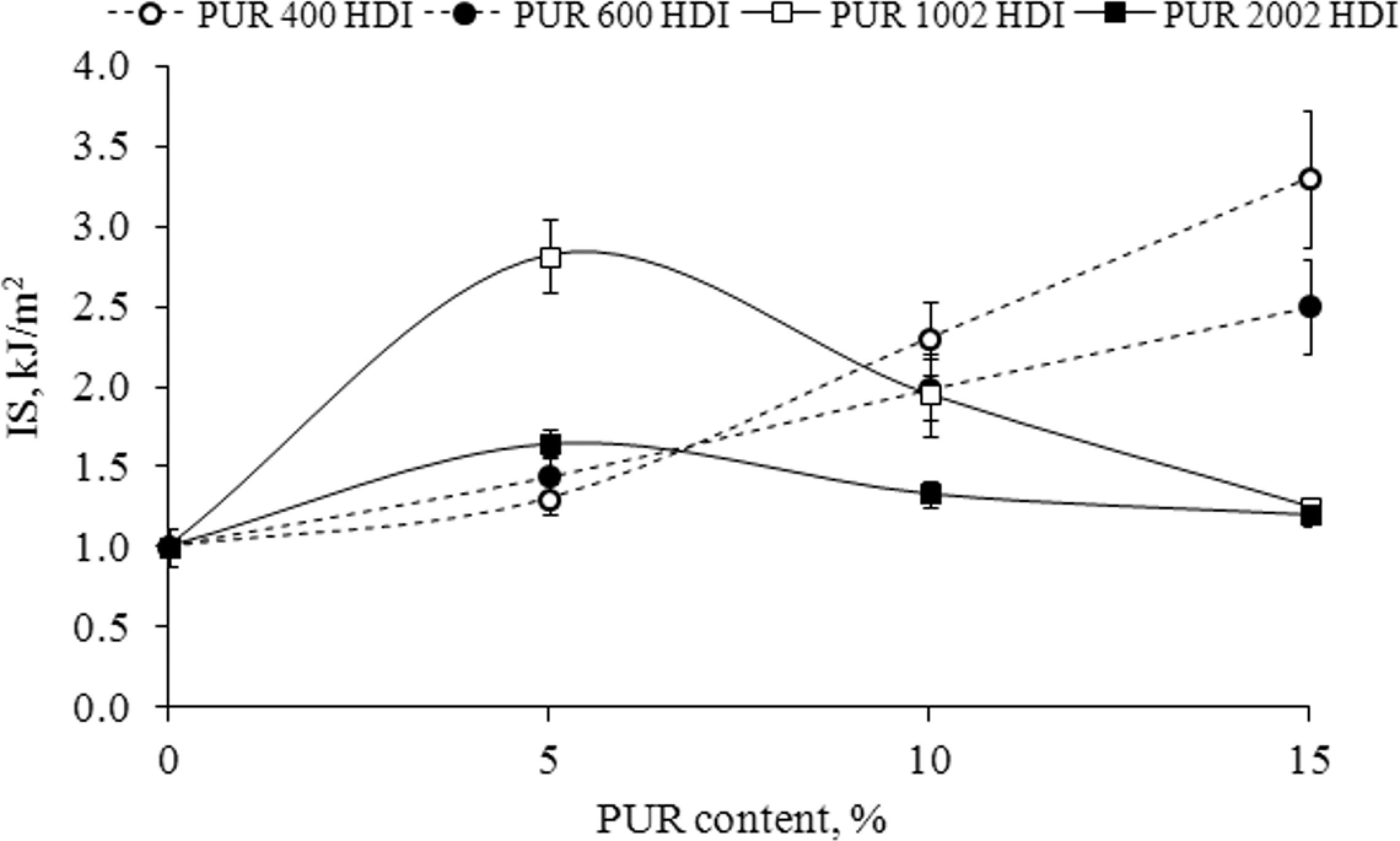
IS of epoxy resin as a function of added PUR: (a) PUR 400 and PUR 600, (b) PUR 1002 and PUR 2002. IS: impact strength; PUR: polyurethane.
From Figure 2, showing the effect of incorporated PUR on the critical stress intensity factor (
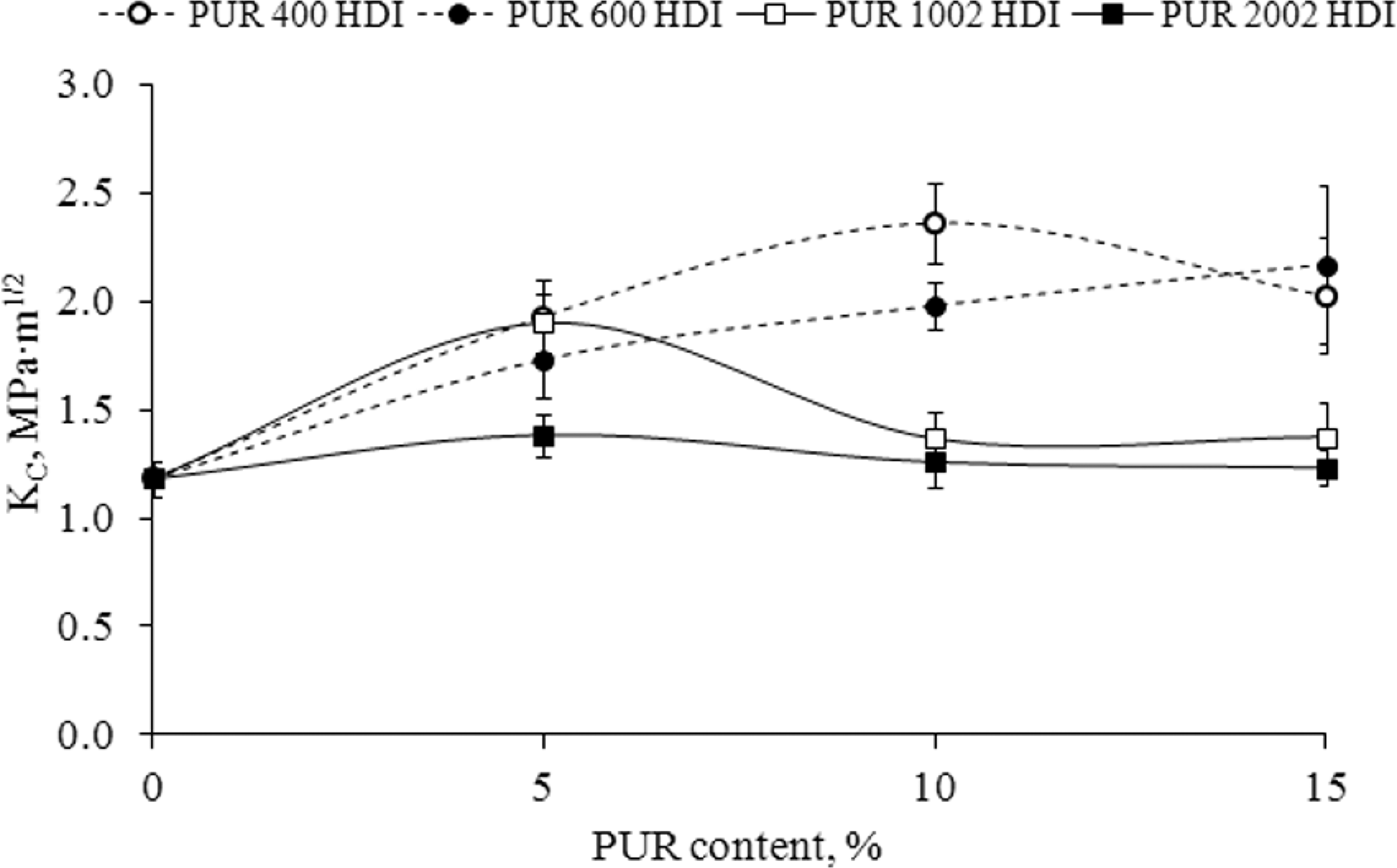
Critical stress intensity factor (
The decrease in IS and
The flexural properties as estimated from three-point bending mode are shown in Table 1 for epoxy compositions containing different amounts of PURs. From Table 1, it can be noted that, similarly to IS and
Flexural properties of epoxy composition containing different polyurethanes.
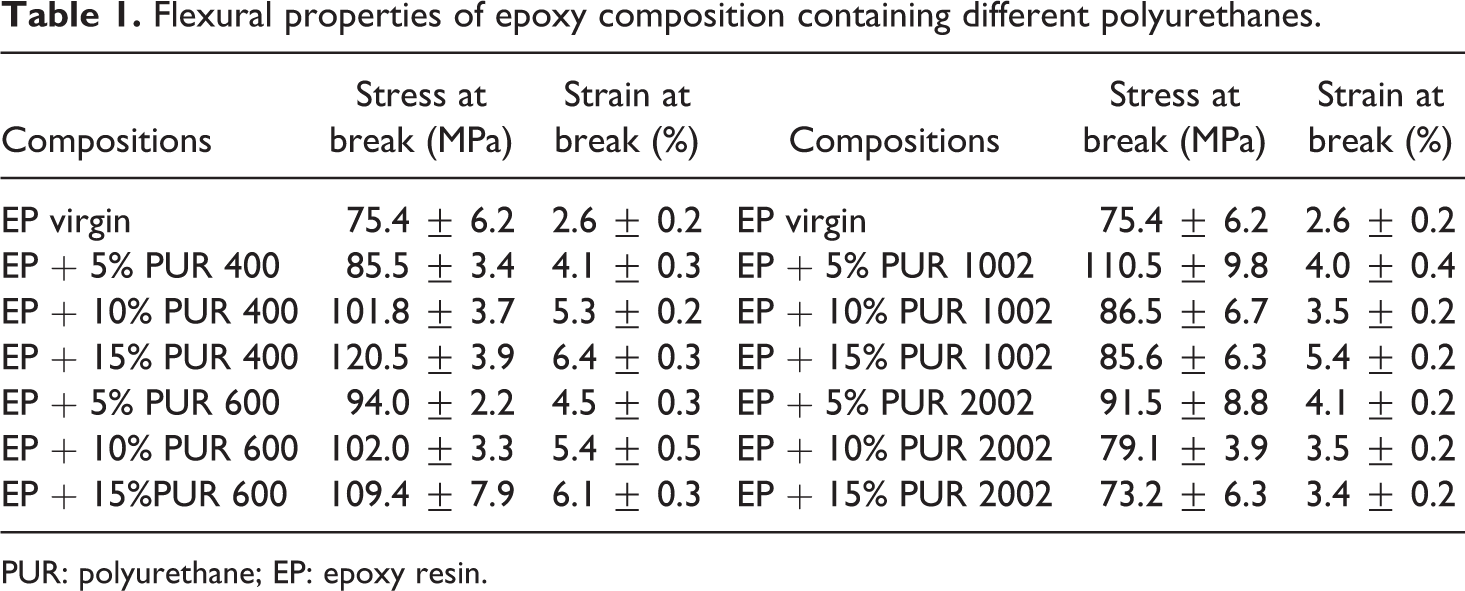
PUR: polyurethane; EP: epoxy resin.
Moreover, EP modified with PURs containing longer flexible chains, that is, PUR 1002 and PUR 2002 (PUR 2002 HDI), exhibited different resistance to flexure. EP modified with 5% PUR 1002 or 5% PUR 2002 showed maximum flexural strength representing approximately 45% and 20% increase, respectively, in respect with neat epoxy samples. Furthermore, all modified epoxy composites showed enhanced flexural strain at break. The strain at break increased from 2.6% (neat EP) to approximately 4% with the incorporation of 5% of either modifier. This can be partly explained by the free volume increase due to the presence of PUR, which acted as plasticizer. The chains movement is facilitated and hence can undergo large extension before the occurrence of the sample fracture. 17 However, it was reported in literature that the addition of modifiers containing flexible segments contribute to the improvement in mechanical properties of EP through better compatibility between this latter and the modifier. 12,18
Structure and morphology analyses
FTIR spectroscopy was used to analyze the structures of obtained modified EPs and check the possible occurrence of chemical reactions between reactive groups of PURs and the polymer matrix.
Figure 3 shows FTIR spectra of virgin EP as well as compositions containing 15% PUR 400 and that with 15% PUR 600. Characteristic peaks at 1716 cm−1 are associated with the presence of urethanes groups in the epoxy compositions.
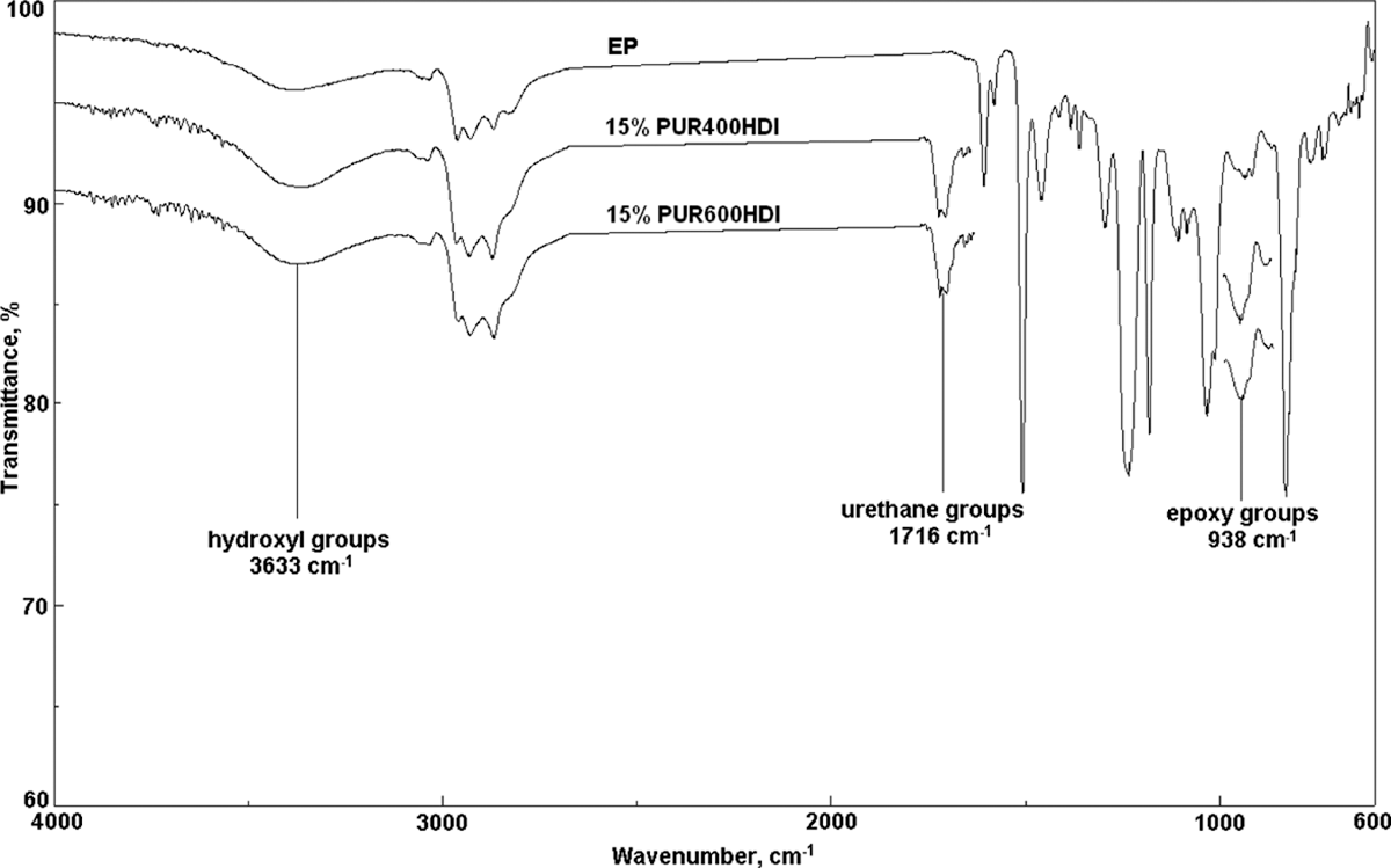
Fourier transform infrared spectra of neat epoxy resin composition containing 15% PUR 400 and composition containing 15% PUR 600. PUR: polyurethane.
However, we did not observe any shift in the absorption peaks appearing at 938 cm−1 and 3366 cm−1 and related with epoxy and hydroxyl groups of the polymer matrix, indicating most probably the formation of an IPN structure. The formed structure may explain the improvement in IS and
One can observe that the addition of PUR based on PEG has resulted in an increase in hydroxyl and epoxy groups peaks heights. The increase in epoxy groups attained 75% and 45% with the addition of PUR 400 HDI and PUR 600 HDI, respectively. However, the increase in OH groups height was about 70% and 50%, respectively. It seems that grafting reactions took place between PUR and EP, thus, explaining the mechanical properties enhancement.
Moreover, it can be noted that the height of hydroxyl and epoxy groups peaks was not influenced by the addition of PUR based on POPD (Figure 4). In this case, the suggested structure of obtained PUR/EP is that of IPN with no grafting reaction.
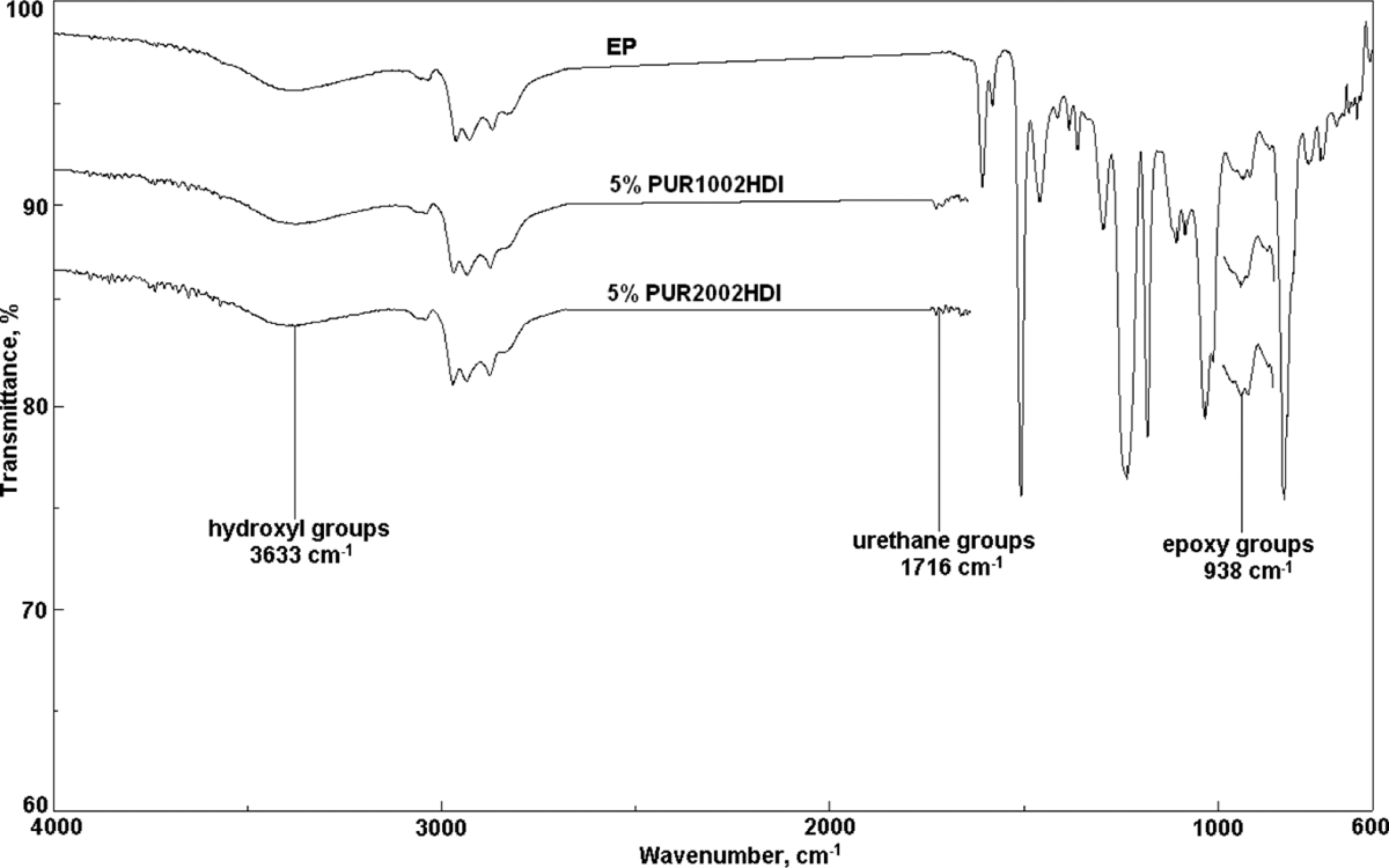
Fourier transform infrared spectra of neat epoxy resin and compositions containing 15% PUR 1002 and 15% PUR 2002. PUR: polyurethane.
The SEM micrographs obtained from fractured surfaces of samples after impact tests near the crack tip were used to explain the toughening mechanism due to the addition of the polymeric modifier.
Figure 5 shows the micrograph of the unmodified epoxy composition fracture surface. The flat glassy surface suggests the occurrence of regular crack propagation path and low fracture energies of the tested samples. The lack of specific features or significant plastic deformation associated with the smooth surface indicates that the specimen fractured in a brittle manner.

Micrograph of unmodified epoxy resin.
Figure 6 shows the micrographs of EP containing 5% PUR 1002 HDI (Figure 6(a)) and 5% PUR 2002 HDI (Figure 6(b)). It can be noted that the addition of PUR to the polymer matrix had resulted in an obvious change in the morphology of the neat polymer matrix. The fracture surface of composition containing 5% PUR 1002 (Figure 6(a)) exhibited a deformed leaf-like morphology with larger plastic deformations zones with the presence of microcracks, which may explain the significant improvement in the IS of the polymer matrix (Figure 1).

Micrographs of epoxy compositions containing 5% PUR 1002 HDI (a) and 5% PUR 2002 HDI (b). PUR: polyurethane; HDI: hexamethylene diisocyanate.
However, the fracture surface of composition containing 5% PUR 2002 HDI (Figure 6(b)) is more stratified with more elongated, sea wave-like and rougher structure with significant plastic deformations and microcracks as well as the presence of delamination zones. The differences in the mentioned fracture surface morphologies stands behind the differences in the measured mechanical properties. The observed significant structure deformation might be responsible for IS improvement due to considerable absorbed energy during the crack propagation process.
Micrograph of composition containing 15% of PUR 400 HDI (Figure 7(a)) is very similar to that of neat EP with a smooth surface without any plastic deformation. However, the deformed fracture surface presented in Figure 7(b) shows an elongated structure with some stratification. This might be related to the length of PURs chains, which will not allow large deformations before the fracture of the tested samples.

Micrographs of epoxy compositions containing 5% PUR 400 (a) and 5% PUR 600 (b).
Conclusions
The following conclusions can be drawn based on the obtained results: Maximum IS value, representing approximately 150% increase in relation to unmodified epoxy sample, was exhibited by the composites containing 15% PUR 400 and 5% PUR 1002. Such IS improvement due to PUR incorporation might be related to the soft segments of PEG and POPD. Moreover, the critical stress intensity factor ( FTIR spectra showed that the peaks appearing at 938 cm−1 and 3366 cm−1 and related to epoxy and hydroxyl groups did not shift upon PUR addition. However, the height of OH groups was not influenced by the addition of PUR based on POPD and increased with PEG-based PUR. This finding might be related to the formation of an IPN with no grafting reaction for the former and occurrence of grafting reaction for the latter modifier. Moreover, SEM micrographs of epoxy composites PUR 2002 revealed the presence of a rough, stratified, more elongated and sea wave-like structure with significant plastic deformations and microcracks. However, epoxy composites with PUR 400 showed either typical regular elongated stratified structure (PEG 400) or flat and glassy surface (PEG 600) as for unmodified EP.
Footnotes
Authors’ Note
The present work was carried out within the Strategic Program ‘Innovative Systems of Technical Support for Sustainable Development of Economy’ in Innovative Economy Operational Program.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
