Abstract
Autologous lung-derived mesenchymal stem cells (LMSCs) were transplanted endoscopically into sheep with experimental emphysema to assess their capacity to regenerate functional tissue. LMSC lines were derived from transbronchial biopsies, cloned at passage 2, expanded in culture, and labeled. A delivery scaffold containing 1% fibrinogen, 20 μg/ml of fibronectin, and 20 μg/ml of poly-L-lysine was used to promote cell attachment and spreading. Treatment animals received scaffold containing 5–10 × 106 cells/site; control animals received scaffold alone. Phenotypic markers, differentiation capacity, extracellular matrix protein expression, and paracrine function of LMSCs were characterized in vitro. Responses to LMSC transplantation in vivo were assessed in terms of clinical toxicity, lung physiology, change in tissue mass (measured by CT scanning) and perfusion (measured by scintigraphy scanning), and tissue histology. At 4-week follow-up, transplants were well tolerated and associated with increased tissue mass and lung perfusion compared to control treatment. Histology confirmed cell retention, increased cellularity, and increased extracellular matrix content following LMSC treatment. Labeled cells were distributed in the alveolar septum and peribronchiolar interstitium. Some label was also present within phagocytes, indicating that a fraction of autologous LMSCs do not survive transplantation. These results suggest that endobronchial delivery of autologous LMSCs has potential therapeutic utility for regenerating functional lung in emphysema.
Introduction
Mesenchymal stem cells (MSC) have recently been considered for use in cell-based therapies because they are readily available, highly proliferative, and display multilineage potential (19,35). Although MSC lines have been isolated from various adult tissues including adipose, liver, lung, and heart, most studies evaluating their regenerative capacity have utilized bone marrow-derived cells (17,25,29,33,36,40). Several lines of evidence suggest that bone marrow-derived mesenchymal stem cells (BM-MSC) could have therapeutic utility for treatment of advanced lung diseases. BM-MSCs have been identified in healing allografts of lung transplant patients, confirming their ability to migrate to, and remodel, lung tissue (3,7,16,34). They have also been shown to express lung epithelial markers in culture, suggesting mesenchymal–epithelial transdifferentiation and the potential to contribute to epithelial regeneration (24,38).
Transplantation studies of BM-MSCs in animal models of lung disease have thus far been only marginally successful, however. Intravenous delivery of syngeneic BM-MSCs in rodents indicates that engraftment requires a toxic “priming” step such as radiation or bleomycin treatment. Even with priming, only low-level BM-MSC retention has been observed in vivo (18,20). Furthermore, therapeutic responses to BM-MSC transplantation have been limited to animal models of inflammatory lung diseases, such as asthma and acute lung injury (21,30,42). Beneficial effects have been attributed to immunomodulation either from cytokine release or activation of the endogenous immune system (22,31). To date there is no evidence that transplanted BM-MSCs contribute directly to regeneration of functional lung tissue, or are capable of differentiating into lung cells in vivo. Thus, the utility of BM-MSCs for treating advanced emphysema, a disease characterized by tissue destruction and loss of structural integrity, is questionable.
Our lab has recently isolated a highly proliferative, clonogenic mesenchymal cell from explants of adult lung parenchyma (LMSC). This cell demonstrates efficient retention in the lung when delivered endobronchially, and expresses basement membrane proteins (e.g., collagen IV, laminin, and fibrillin-1) and growth factors that could promote regeneration of lung tissue through trophic effects on endogenous cells (11).
The present study summarizes responses to autologous LMSC transplantation in a large animal model of experimental emphysema. Results show that LMSCs have multilineage potential similar to BM-MSCs, exert paracrine effects on epithelial cells, and engraft the alveolar septum without causing pathology. Autologous LMSC transplantation improved tissue mass and lung perfusion at sites of preexisting elastase-induced damage. Together these results indicate that autologous LMSC transplantation may be effective for promoting tissue regeneration in emphysema.
Materials and Methods
Experimental Protocol
Large-animal studies were performed under a protocol approved by the Tufts Cummings Veterinary School Institutional Animal Care and Utilization Committee (IACUC). Following acclimation, 13 experimentally naive mixed breed adult female sheep received serial endobronchial elastase treatments monthly for 5 months. Treatment sites were allowed to mature (8 weeks) and experimental emphysema was confirmed by computed tomography (CT) imaging. Ten animals were selected to receive either control (n = 5) treatment with scaffold alone or autologous LMSC transplantation (n = 5). Study animals underwent pretransplantation scintigraphy perfusion scans and assessment of lung mechanics. Animals receiving autologous cell transplants underwent transbronchial biopsies at 8–10 sites in the healthy lung (i.e., non-elastase treated) and explants were established to generate autologous LMSC lines. Control animals did not undergo sham procedures. LMSC clones were derived at passage 2 and expanded to generate 5–10 million cells. Transplants were performed at passage 5. Animals underwent prenecropsy assessments 1 month following transplantation immediately prior to sacrifice. Perfusion and CT scans and lung physiology measurements were repeated, and tissues were harvested for frozen section analysis and paraffin embedding.
Large-Animal Model Development
The ovine emphysema model used in this study has been described in detail elsewhere (37). In brief, animals received 20 ml doses (200 U/ml of High Purity Porcine Elastase, Elastin Products, #EC134) of purified elastase dissolved in phosphate-buffered saline, pH 7.4, at two subsegmental airways delivered bronchoscopically while mechanically ventilated under general anesthesia. Treatment was repeated every 3–4 weeks at the same anatomic location for a total of five doses. Following the last dose, animals were observed for 8 weeks and then underwent CT imaging to confirm the presence of destructive changes. At the time of the confirmatory CT scan, transbronchial biopsies were performed at 8–10 sites in the lung that did not receive elastase treatments. Tissues were transferred immediately into iced transport media (Invitrogen Gibco, Product #18045) and transported to a sterile cell culture hood for processing.
Large-Animal Imaging and Physiology Studies
CT images were obtained with a Picker 5000 scanner using a spiral acquisition protocol, 10 mm collimation, pitch of 10, and 5 mm overlap. Scans were performed on intubated, anesthetized, mechanically ventilated animals following lung inflation to 25 cm H2O of transpulmonary pressure.
Radionucleotide perfusion scans were performed in awake animals while standing in an animal transport cart with the camera positioned over the dorsal aspect of the thorax. Single-view images were obtained immediately following jugular vein injection of radioisotope (Tm 99m microaggregate albumin).
Lung physiology assessments included measurements of lung resistance (RL), dynamic compliance (CL), and quasistatic deflation pressure–volume curves summarized using the exponential equation V(P) = Vmax – (Vmax – Vmin)e–kP, where V is volume, P is pressure, Vmax is lung volume at infinite inflation pressure, Vmin is lung volume at 0 inflation pressure, and k is the shape factor describing the curvature of the exponential. Measurements were performed in anesthetized intubated animals as previously described (9,12).
LMSC Isolation and Characterization
Cell Isolation and Proliferation
LMSCs for transplantation were grown from tissue explants of transbronchial biopsy samples using an established protocol (11). In brief, the tissue fragments were rinsed in antibiotic/antifungal solution diluted in Dulbecco's phosphate-buffered saline [DPBS; 100 U/ml penicillin (PCN), 100 μg/ml streptomycin, 2.5 μg/ml amphotericin], and trypsinized for 15 min at 37°C (5% CO2, 100% humidity). Tissues were washed in DMEM-F12 media with 10% serum, and placed into individual wells of fibronectin-coated six-well culture plates containing 0.5 ml of the same media. (The growth conditions were determined as optimal for mesenchymal progenitor cell expansion based on preliminary studies confirming maximal doubling rates with DMEM-F12 + 10% serum compared to DMEM, MEM, RPMI, LHC-9, and LHC-basal media with equivalent additives and similar growth conditions.) Additional media was added to the explants once the cells were approximately 25% confluent (day 7–10), and cells were passaged at 90% confluence. All subsequent passages were performed on standard tissue culture plastic (polystyrene). Under these conditions, LMSCs demonstrated a population doubling time of 74.3 ± 3.9 h and a maximum cell density of 6.3 times; 104 cells/cm2 at confluence. Cells were cloned at passage 2 by dilutional plating, and five clones with similar morphology were selected for further passage. Cells were split 1:4 through passage 5 to generate sufficient cells for transplantation (approximately 5 times; 107 cells/site) and phenotypic characterization.
Cell Characterization: ECM Expression by Immunostaining
LMSC expression was assessed by growing cells on plastic cover slips (Thermonox, TPX Inc.), fixing them in ice-cold methanol, and performing immunocytochemistry for a panel of proteins. Expression of integrins α2 (Abcam, ab30486, clone AK7), α3 (Abcam, ab30488, clone 17C6), α5 (Abcam, ab25076, clone 5H10-27), αv (Abcam, ab78438, clone P156), β1 (Abcam, ab21845, clone MEM-101A), β5 (Abcam, ab55359, polyclonal), intermediate filaments (vimentin, Santa Cruz Biotech, clone RV203, sc-58901), CD45 (Serotec Ab 220F, clone 1.11.32 mouse IgG anti-sheep), and extracellular matrix components (collagen IV, Santa Cruz Biotech, clone CIV22, sc-59813; laminin-5, Santa Cruz Biotech, clone P3E4) was evaluated. Immunocytochemistry was performed using standard methods employing a 1-h primary antibody incubation without antigen retrieval. Signal detection was performed with species-specific peroxidase-conjugated secondary antibodies (Vector Laboratories, Burlingame, CA). Controls were performed using 1-h species-specific serum incubation in place of primary antibody.
Cell Characterization: Paracrine Effects
LMSC effects on epithelial cell growth and attachment were assessed in vitro. Growth curves of primary ovine lung epithelial cells were measured in 12-well transwell assays (3-μm pores) with confluent LMSCs in the upper chamber. Control experiments were performed using transwell inserts with either no cells or confluent epithelial cells. Each experiment was repeated five times to evaluate consistency of response. Growth was measured by directly counting trypsinized cells in the lower well at specific times following seeding.
To further characterize paracrine effects, RNA was isolated from confluent LMSCs in culture, and gene expression analyzed using the Affymetrix Unigene Build 57 bovine chip with confirmed ovine cross-reactivity (1). RNA was isolated using the SA Biosciences qPCR-grade RNA isolation system (SA Biosciences, Fredrick, MD, #PA-001). RNA quality and quantity were confirmed spectrophotometrically. Expression analysis and corresponding bioinformatics were performed by Tufts University Molecular Biology Core Facility.
Expression of specific growth factors identified by gene array analysis was performed on concentrates of conditioned media harvested from LMSCs that had been grown to confluence, washed, and switched to serum-free media for 24 h. Samples were concentrated under vacuum and resuspended in 10% of their initial volume. ELISA (Antigenix America, Huntington Station) was performed for fibroblast growth factor 2 (FGF2) and FGF10 according to the manufacturer's instructions. Each analysis was performed in triplicate.
The ability of LMSCs to affect epithelial cell attachment and spreading through expression of extracellular matrix was assayed by performing epithelial cell attachment assays on LMSC-conditioned surfaces prepared by growing LMSCs to confluence in 48-well polystyrene plates, washing with 0.5% Triton X-100 and PBS, and then incubating with serum-free media containing antibiotics for 48 h at 37°C and 5% CO2. Attachment assays were repeated four times to assess consistency.
Cell Characterization: LMSC Differentiation Capacity In Vitro
The differentiation capacity of LMSCs was assessed by seeding at low density onto plastic cover slips in osteogenic (Invitrogen-Gibco, A10072-01), chondrogenic (Invitrogen-Gibco A10071-01), and adipogenic (Invitrogen-Gibco, A10070-01) differentiation media. After 14 days (70–80% confluence), cells were fixed with 5% buffered formalin and stained with Alcian blue (to detect chondrocyte differentiation), Alizarin red (to detect osteocyte differentiation), or Lipid Tox® green (Invitrogen-Gibco, H34475, to detect adipocyte differentiation).
Cell Characterization: LMSC Niche In Vivo
To characterize the LMSC niche in vivo, explants were harvested at various stages after culture initiation and frozen sections (10-μm thickness) were prepared for staining. Cells in the explants were labeled with a panel of primary antibodies to characterize their phenotype (S100A4, α-smooth muscle actin (α-SMA), vimentin, and collagen 1) and state of proliferation [proliferating cell nuclear antigen (PCNA)].
Scaffold Design and Testing
In Vitro Attachment, Spreading, and Proliferation Assays
Scaffolds have previously been shown to facilitate engraftment of exogenous attachment-dependent mesenchymal stem cells in non-pulmonary applications (15). For this application, LMSC scaffolds were designed using biologic substrates to promote engraftment, prevent anoikis, and promote cell cycling through integrin, syndecan, and glypican binding. Attachment and spreading assays were performed in standard fashion using substrate coated 96-well plates as previously described (11).
The effect of attachment substrates on the lag time to initiation of cell cycling was assessed using a similar approach with omission of the albumin blocking step. To assess proliferation, cells were trypsinized and counted at specified times following plating.
Assessment of LMSC Engraftment and Postengraftment Expression in Organotypic Culture Using Devitalized Lung Tissue
To further characterize the capacity of LMSCs to attach and spread using biologic scaffolds, attachment assays were performed using sections of devitalized lung prepared from blocks of freshly frozen, healthy ovine tissue harvested at necropsy. Tissue core (12 mm diameter) prepared from frozen tissue blocks were sliced to generate disks 3–4 mm in thickness. These were washed in 0.5% Triton X-100 for 24 h to remove dead cells, rinsed extensively in distilled water, and sterilized in 70% ethanol. Tissue was rehydrated in three exchanges of sterile DPBS, then incubated in tissue culture media with our without attachment substrates for an additional 24 h at 37°C, 5% CO2. Disks were then transferred to 12-well culture plates and seeded with 104 GFP-labeled LMSCs/disk. Samples were harvested 24 h postseeding. Attached cells in control and scaffold-conditioned tissues were quantified by counting GFP-labeled cells/high power field (hpf; 200× magnification) for 10 randomly selected fields.
Postengraftment LMSC expression was assessed 14 days following seeding of ovine LMSCs onto devitalized lung tissue samples that had been pretreated with either media containing 10% lamb serum or fibrinogen/fibronectin/poly-L-lysine (PLL) (FFP) scaffold. Staining was performed for vimentin, α-SMA, fibronectin, laminin, elastin, and collagen IV.
Autologous Transplantation in an Ovine Model of Emphysema
Description of Transplant Procedure
In vivo testing of autologous LMSC transplantation was performed in sheep with experimental emphysema by comparing treatment with cells plus scaffold to scaffold alone. To assess retention and engraftment of transplanted cells, LMSCs were labeled with the fluorescent membrane dye PKH-26 (Sigma Chemical, St. Louis, MO; 018K1741) according to the manufacturer's instructions, and incubated for 60 min at 37°C, 5% CO2 in culture media. Prior in vitro studies confirmed that PKH labeling does not alter cell viability, attachment, or spreading, and that labeled cells can be detected by fluorescent microscopy after four to five passages in culture.
Each animal received treatment at two subsegmental sites. Cell suspensions, consisting of 20 ml of scaffold + ~107 PKH-labeled autologous cells were administered endobronchially to animals that received autologous cell transplants. Twenty milliliters of scaffold alone was administered in similar fashion to control animals. Treatments were performed in anesthetized, mechanically ventilated animals through a fiberoptic endoscope advanced into wedge position in a subsegmental airway. Each site was “primed” immediately prior to treatment with 20 ml of 0.25% trypsin solution injected directly through the instrument channel of the scope (10). A dual lumen delivery catheter was positioned with its tip 2–3 cm beyond the end of the bronchoscope. The syringe containing each 20 ml treatment and the syringe containing the hydrogel scaffold cross-linker (2.5 ml of thrombin) were attached to the appropriate connectors at the proximal end of the dual-lumen delivery catheter and delivered simultaneously (10).
Outcome Measures
Response to treatment was assessed in terms of procedural and postprocedural safety and efficacy. Safety outcome measures included assessments of vital signs, oxygen saturation, body weight, activity, food consumption, and clinical pathology (blood chemistry, hematology, and coagulation studies). Efficacy was assessed in terms of lung physiology (lung resistance, and static and dynamic compliance, single breath diffusing capacity), radiographic changes in tissue mass (by CT imaging) and perfusion (by quantitative perfusion scanning), and histological assessments of cell retention and tissue remodeling.
Statistical Analysis
Results are presented either as mean values ± SDs or median values with ranges as appropriate. Comparisons for continuous outcome variables pre- and posttreatment for each group, and between treatment groups, were performed by t-test or nonparametric testing (Mann-Whitney test) as appropriate. Statistical significance was defined as a value of p < 0.05. Where appropriate, correction for multiple comparisons using the Bonferroni method was applied.
Results
Isolation and Characterization of Autologous LMSCs Obtained From Lung Biopsies
LMSC In Vivo Localization, Cloning Efficiency, and Growth Characteristics
Photomicrographs of explant-derived LMSCs at various stages of growth are presented in Figure 1. Spindle-shaped cells first appeared at the edge of explanted tissues on fibronectin-coated plates between day 4 and 7 (Fig. 1a) and formed a monolayer by day 21 (Fig. 1b). Immunofluorescent staining demonstrated a large subset of PCNA+ S100A4+ cells within the explant arising from the lung interstitium and perivascular adventitia (Fig. 1f). S100A4+ cells did not express collagen IV (Fig. 1h), fibrillin-1, CD45, α-SMA, or collagen 1, indicating that: the S100A4+ cells were highly proliferative but not actively synthesizing basement membrane proteins at this stage, and S100A4+ cells displayed phenotypic markers distinct from fibrocytes (collagen 1+, CD13+, CD45+, vimentin+), myofibroblasts (collagen 1+, α-SMA+, vimentin+, CD45–), pericytes (CD31+, CD90+, α-SMA+, Ang-1+), and leukocytes (CD45+). Limiting dilution cloning assays at passage 2 (Fig. 1c) demonstrated LMSCs possess high cloning efficiency (8.5 ± 2.3%).

LMSC characterization during growth from explants. (a) Lung-derived mesenchymal stem cells (LMSCs) growing from explanted tissue at passage 0, day 10 on fibronectin-coated plates in DMEM-F12 media. (b) Near-confluent LMSCs at passage 0, day 22. (c) Cloned cells at passage 2. (d) Confluent LMSCs at passage 5. (e) Immunofluorescence controls of an explant processed at passage 0. (f) Explant at P0 stained for S100A4 (green) and collagen I (red) showing S100A4+ mesenchymal cells (LMSCs) migrating towards the periphery of the explant. (g) S100A4+ (green) LMSCs co-stained for proliferating cell nuclear antigen (PCNA) (red), showing that the cells are actively proliferating. At this stage of proliferation and migration, the cells are vimentin+ (not shown), but do not express (h) collagen IV (red) or fibrillin (not shown) and are not CD45+ (not shown).
Assessment of Multipotency
Figure 2 summarizes results of adipogenic, osteogenic, and chondrogenic differentiation assays performed at passage 5. Cells were assayed after 14 days in culture at 70–80% confluence. LMSC differentiation capacity was similar to that of other mesenchymal stem cells, displaying chondrocyte (Fig. 2a), osteocyte (Fig. 2b), and adipocyte (Fig. 2c) phenotypes under the appropriate culture conditions.

LMSC differentiation in chondrogenic, osteogenic, and adipogenic media. (a) Alcian blue stain performed on LMSCs after 14 days in chondrocyte selection media showing chondrocyte differentiation. (b) Alizarin red stain performed on LMSCs after 14 days in osteocyte selection media showing osteocyte differentiation. (c) Lipid Tox® green stain performed on LMSCs after 14 days in adipocyte selection media showing adipocyte differentiation. (d–f) LMSCs 14 days (passage 2) after transfer back to DMEM-F12 media showing loss of mesenchymal differentiation phenotypes.
To assess the stability of lineage-specific differentiation, LMSCs were transferred back into DMEM-F12 media and reassayed after two passages. Loss of chondrocyte (Fig. 2d), osteocyte (Fig. 2e), and adipocyte (Fig. 2f) phenotypes was observed, showing media-dependent reversion to a lung fibroblast phenotype.
Characterization of LMSC Protein Expression in Culture: Immunocytochemistry
Characterization of lineage markers, growth factor receptors, integrins, and extracellular matrix proteins of ovine LMSCs at passage 5 is summarized in Figure 3. Findings were consistent across all clones tested. Confluent LMSCs expressed the mesenchymal markers vimentin (Fig. 3b), α-SMA (Fig. 3c), and S100A4 (Fig. 3d), and were pancytokeratin (Fig. 3e) and CD45 (Fig. 3p) negative. LMSCs also expressed high levels of platelet-derived growth factor receptor-α (PDGFR-α) (Fig. 3g) and transforming growth factor-β2 receptor (TGF-β2R) (Fig. 3i). epidermal growth factor receptor (EGFR) (Fig. 3j) was undetectable. Integrins α5, αv, and β1 were expressed at high levels, while β3 (Fig. 3l–o) was expressed at low levels. At confluence, LMSCs expressed collagen IV (Fig. 3r), laminin (Fig. 3s), fibrillin-1 (Fig. 3t), fibronectin (not shown), and vitronectin (not shown), but little collagen 1 (Fig. 3q). Cellular expression of ECM components at confluence was greater than at low density (Fig. 3w, x).

Integrin, growth factor receptor, and extracellular matrix protein expression of LMSCs in culture. (Top row) Ovine LMSCs express multiple mesenchymal markers including vimentin, α-SMA, and S100A4 but are cytokeratin and CD45 (p) negative. (Second row) They strongly express platelet-derived growth factor receptor-α (PDGFR-α) and transforming growth factor-β2 receptor (TGF-β2R), and (third row) integrins α5, αv, and β1. At confluence (fourth row) LMSCs express basement membrane proteins including collagen IV, laminin, and fibrillin but not collagen I. Expression of basement membrane proteins by LMSCs at low density (bottom row) was substantially less.
Paracrine Effects of LMSCs on Epithelial Cells
Paracrine effects of LMSCs on the growth of primary ovine epithelial cells are summarized in Figure 4. LMSCs grown to confluence on transwell filters increased proliferation of cocultured epithelial cells (Fig. 4a). Population doubling time (PDT) for control epithelial cells was 305 ± 22 h in DMEM-F12 media and 127 ± 17 h in LHC9 media. In transwell assays performed with LMSCs, PDT was 106 ± 23 h in DMEM-F12 media (i.e., a 65% reduction) and 93 ± 14 h in LHC9 media (i.e., a 27% reduction). Controls performed with confluent epithelial cells in the upper transwell showed no effect on PDT (DMEM-F12 = 289 ± 44 h; LHC9 = 88 ± 32 h).

Paracrine effects of LMSCs on primary ovine epithelial cells. (a) Paracrine effects of LMSCs on epithelial cell growth is shown. In transwell coculture experiments, confluent LMSCs on three microfilters in the upper chamber reduced epithelial cell population doubling time by tw0-to threefold relative to experiments in which confluent epithelial cells were used in the upper chamber. (b) Matrix secreted by LMSCs onto tissue culture plastic promoted epithelial cell attachment that was nearly equivalent to plastic coated with purified collagen (Col I), fibronectin (FBN), and matrigel (ECM). Albumin partially blocked epithelial binding to LMSC matrix. Addition of purified matrix components produced minimal additional epithelial cell attachment to LMSC substrate.
Gene array analysis indicated that LMSCs express transcripts for FGF2 (probe set Bt 4834.1.21.at; FC vs. chip set control 14.96) and FGF10 (probe set Bt 12991.1.s1.at; FC vs. chip set control 9.07). Protein expression was confirmed by ELISA, which demonstrated measurable quantities of both FGFs (62 ± 34 ng/ml for FGF2, 30 ± 27 ng/ml for FGF10; average of three measurements for each) in conditioned media compared to control samples (<20 ng/ml) prepared using nonviable (lysed) cells.
LMSCs expressed ECM components that promote attachment and spreading of lung epithelial cells (Fig. 4b). LMSC-conditioned plates promoted epithelial cell attachment similar to purified ECM components. Addition of selected ECM components to LMSC-conditioned substrate produced minimal increases in epithelial cell attachment, suggesting optimal responses to LMSC substrate alone.
Design of Hydrogel Scaffold to Promote LMSC Attachment for In Vivo Use
Attachment and Spreading Assays Using Purified ECM Components
Delivery scaffolds tested in this study were designed to include a fibrin hydrogel component because fibrin is an important constituent of the biologic matrix that forms on the epithelial surface of the lung to promote cell spreading following injury (32). Attachment and spreading assays performed using purified ECM components alone, and fibrinogen plus selected purified ECM components, are summarized in Figure 5a. Results show that optimal attachment and spreading is achieved with multicomponent hydrogels. A hydrogel comprised of 1% fibrinogen + 20 μg/ml of fibronectin + 20 μg/ml poly-L-lysine (Fig. 5e) was selected for in vivo use because it promoted better spreading than other multicomponent systems.
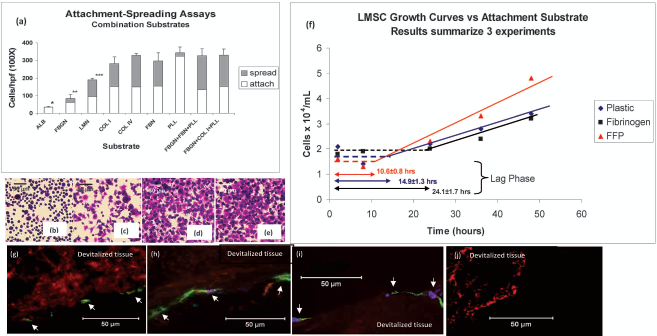
Scaffold design and cell attachment studies. (a) Results of attachment assays for single substrate and combination substrate hydrogel scaffolds (n = 4 per condition). Albumin (ALB), fibrinogen (FBGN), and laminin (LMN) produced less attachment and spreading in this assay system than combination substrates (*, **, *** denotes that each of these scaffolds produced significantly less cell attachment + spreading than either of the multicomponent combination substrates tested in this assay assessed by paired analysis with Bonferroni correction, p < 0.005). (b–f) Cell spreading postattachment is shown for poly-L-lysine (PLL) (b), fibrinogen (c), fibrinogen + collagen I + PLL (d), and fibrinogen + fibronectin + PLL (FFP) (e). (f) Scaffold effects on cell proliferation over the first 48 h. Testing was performed in triplicate. Results show that FFP scaffolds influence cell proliferation primarily by altering lag phase, consistent with facilitation of engraftment. (g–i) Effects of FFP scaffold on attachment of cells to devitalized lung tissue [9.4 ± 5.7 cells/high power field (hpf); n = 3 samples, 5 hpf/sample]. No cells (0 ± 0 cells/hpf; n = 3, 5 hpf/sample) samples were observed attached to media-treated control tissue (j).
Assessment of Cell Cycle Lag Times for Hydrogel Scaffolds
The effect of selected hydrogels on lag times to the initiation of LMSC proliferation is summarized in Figure 5f. Results demonstrated that hydrogels containing fibrinogen, fibronectin, and PLL shortened lag times and promoted cell proliferation compared to plastic or fibrinogen alone.
Testing of Scaffolds Using Devitalized Lung Tissue
Attachment, spreading, and proliferation of LMSCs on devitalized ovine lung are presented in Figure 5g–j. Twenty-four hours following seeding, GFP-labeled LMSCs (9.4 ± 4.7 cells/200× hpf) in lung tissue pretreated with FFP scaffold solution were attached and spread on the surface of the tissue. No attached cells were observed on media-treated tissue samples.
At day 14 following seeding, LMSCs cultured on media-treated and FFP-treated devitalized lung showed similar expression profiles (Fig. 6, FFP samples shown). LMSCs were vimentin+, α-SMA+, and expressed fibronectin and laminin. Day 14 samples that had been treated with FFP scaffold had substantially more cells/hpf than samples treated with media alone (not shown).

LMSC expression after being cultured of devitalized sheep lung tissue for 14 days in DMEM-F12 media. Immunofluorescent (IF) images of 10 μm frozen sections of ovine devitalized lung tissue seeded with LMSCs after soaking in FFP scaffold for 1 h and harvested after 14 days in culture. Expression by IF staining was similar for cells seeded onto media-treated and FFP-treated tissue samples, although the number of cells on FFP samples was substantially greater at 14 days. LMSC expression in this organotypic assay system was similar to that observed in standard cultures for LMSCs; cells expressed α-smooth muscle actin (α-SMA), vimentin, fibronectin, and laminin. Collagen IV expression was not readily detected in this assay system due to high background from tissues.
In Vivo Response to Autologous LMSC Transplantation in Experimental Emphysema
Safety Results: Clinical Observations and Clinical Pathology
In vivo, autologous LMSC transplantation was uniformly well tolerated. One LMSC-treated animal experienced acute fever within 24 h of treatment, which resolved spontaneously. Two animals from each group had transient cough. None of the animals in either group displayed signs of respiratory distress or hypoxemia (i.e., Spo2 <90%). Activity levels, alertness, food and water consumption, and urination and defecation patterns were normal in both groups throughout follow-up.
Clinical pathology at baseline (immediately pretreatment), acutely posttreatment (~1 h posttreatment), and 1 month posttreatment (prior to necropsy) showed no evidence of systemic toxicity in either group. Immediate posttreatment decreases in hematocrit and serum potassium levels were observed in all animals, and were attributed to acute hemodilution due to fluid administration during anesthesia. Complete hematologic, coagulation, and serum chemistry profiles were otherwise normal at all time points.
Physiologic Responses to Autologous LMSC Transplantation After 28 Days
Physiological responses to treatment with autologous LMSCs + scaffold and scaffold alone immediately following treatment and after 28 days were similar. In LMSC-treated animals, a small but statistically significant reduction from baseline in Vmin (Δ Vmin = −293 ± 220 ml, p = 0.04) was observed at 28 days. There were no changes in RL (ΔRL = −0.15 ± 0.77 cm H20/L/s, p = 0.68), CL (ΔCL = −0.02 ± 0.07 L/cm H20), Vmax (ΔVmax = −21 ± 256 ml, p = 0.86), or k (Δk = −0.039 ± 0.081, p = 0.33). In control animals, there were no posttreatment changes in either static (ΔVmax = 257 ± 229 ml, p = 0.06; ΔVmin = 246 ± 300 ml, p = 0.14; Δk = 0.018 ± 0.041, p = 0.37) or dynamic (ΔRL = −0.22 ± 0.33 cm H2O/L/s, p = 0.22; ΔCL = −0.01 ± 0.03 L/cm H2O, p = 0.57) physiology measures.
Radiologic Responses to Autologous LMSC Transplantation After 28 Days
LMSC treatment was associated with a statistically significant increase in tissue mass (ΔHU = +10.7pl;ux3.9%, p = 0.006) from baseline, while control treatment was associated with no change (ΔHU = +2.9 ± 3.4%, p = 0.07). Perfusion to the treated lung 28 days following LMSC treatment increased significantly from baseline (?Q = +7.1 ± 4.4%, p = 0.022) while control treatment was not associated with a significant change (–4.5 ± 3.6%, p = 0.053).
Example CT and perfusion scans from an LMSC-treated and control animal are shown in Figure 7. Increases in tissue mass and perfusion following LMSC treatments were observed specifically in areas where treatment was administered, confirming site-specific effects.

CT and lung perfusion scans from a control and LMSC-treated animal. Examples of a control (a) and LMSC-treated (c) animal at baseline following generation of bullous emphysema, and 1 month following endoscopic site-directed treatment [(b) control scaffold treatment, (d) LMSC treatment]. Insets show the corresponding lung perfusion scans at each time point. Treatment with scaffold alone was associated with no radiographic evidence of tissue regeneration, while LMSC treatment was associated with a reduction in size of the damaged region and increased perfusion.
Assessment of Cell Engraftment and Tissue Remodeling Following Autologous LMSC Transplantation
Representative 10 μm frozen sections of lung tissue from LMSC-treated and untreated sites in transplanted animals, and from treated sites in control animals after 28 ± 4 days are presented in Figure 8. Following autologous LMSC transplantation, cells labeled with PKH-26 membrane dye (red fluorescent signal) were observed in several tissue compartments. In areas of lung with intact alveolar structure, transplanted LMSCs were present within the alveolar septum (Fig. 8a), occupying a niche similar to that from which they appeared to originate in explants. In areas where tissue architecture was distorted and fibrotic, PKH label was observed in alveolar and peribronchiolar spaces. Much of the label colocalized with CD45, suggesting that cells in these areas had been taken up by phagocytes (macrophages and/or dendritic cells).

Histology, immunofluorescence, and immunohistochemistry of LMSC-treated and control lung tissue. (a–c) Cytofluorescence of 10 μm frozen sections with collagen autofluorescence on the FITC (green) channel at 400×. PKH-labeled LMSCs (a) appear engrafted to the alveolar septum at day 28. Control (scaffold alone) treatment (b) and untreated sites (c) show no fluorescence on the rhodamine (red) channel, confirming the absence of PKH-labeled cells. Immunocytochemistry (d–f) of 5 μm formalin-fixed, paraffin-embedded sections with primary antibody against elastin stained with DAB at 100×. LSMC treatment (d) is associated with increased elastin signal compared to Control treatment (e) and untreated healthy tissue outside of the treatment area (f). H&E-stained tissues show increased cellularity in LMSC-treated tissues relative to control treatment and untreated sites. Brightfield photomicrographs of Sirius red stained sections (j–l) show collagen within the alveolar walls for LMSC-treated areas, control treated areas, and untreated areas, respectively. Photomicrographs of the corresponding samples under polarized light (m–o) show the extent of organized collagen fibril formation. Compared to control treatment, LMSC treatment appears to produce partial restoration of the collagen and elastin network.
Histology and immunohistochemistry performed on formalin-fixed tissues from treatment (Fig. 8d, g) and control (Fig. 8e, h) animal sites showed LMSC treatment-specific increases in cellularity, collagen (Fig. 8j, m), and elastin (Fig. 8g), suggestive of tissue healing and regeneration. Animals that received scaffold alone had no detectable PKH label (Fig. 8b), substantially fewer cells (Fig. 8h), and ECM, and less detectable collagen (Fig. 8k, n) and elastin (Fig. 8e) at sites of treatment.
Discussion
Regenerative therapies that utilize stem cell technology could revolutionize transplantation medicine for end stage lung disease. Unfortunately, identification of multipotent stem cells appropriate for pulmonary applications, development of methods for promoting site-specific engraftment, and nontoxic therapies for achieving durable immune tolerance are not presently available (41). This study describes pilot testing of an alternative to conventional stem cell-based therapy that could address some of these limitations and make regenerative treatment for advanced lung disease more achievable.
In a large-animal model of experimental emphysema, evidence of tissue regeneration with increased blood perfusion and extracellular matrix (ECM) content was observed using autologous lung-derived mesenchymal cells grown from transbronchial biopsies. Cell transplants were performed endobronchially employing a delivery scaffold composed of natural ECM components that promote attachment and spreading via integrin, syndecan, and glypican interactions. The apparent benefits of autologous cell therapy reported here were not present following treatment with scaffold alone, suggesting that cells were indeed required. Together these findings indicate that regeneration of functional lung tissue in areas of emphysema may be possible using autologous lung-derived reparative cells of mesenchymal lineage.
The fibroblast-like LMSCs employed in this study express surface markers commonly associated with MSCs, and displayed adipocyte, chondrocyte, and osteocyte differentiating capacity similar to BM-MSCs and the lung-derived mesenchymal cell recently described by Karoubi et al. and others (2,16,27,36). These cells display regenerative capacity through expression of basement membrane proteins and growth factors. While these properties are not unique to LMPCs, this cell, which normally resides in the lung, engrafts following endobronchial delivery, making it well suited for designing a cell-based therapy to reverse parenchymal destruction in emphysema. Following transplantation, immunohistochemistry analysis of tissue sections at sites treated with LMSCs showed increases in extracellular matrix in vivo compared to controls, suggesting de novo synthesis by resident cells and/or implanted LMSCs. Despite expressing α-SMA in conventional and organotypic cultures, a marker associated with myofibroblasts and a profibrogenic phenotype, LMSCs did not express collagen I in vitro, or cause fibrosis at sites of transplantation in vivo. By contrast, α-SMA+ CD45+ cells of bone marrow origin have been shown to express collagen I at sites of experimental tissue injury or neoplasia, which results in fibrosis (13,14,23). Whether this difference in collagen I expression reflects inherent phenotypic differences between α-SMA+ CD45– LMSCs and α-SMA+ CD45+ bone marrow-derived mesenchymal cells, differences in the state of tissue inflammation and cytokine expression at engraftment sites, or effects of the scaffold used to promote LMSC engraftment in this study is unclear. Independent of mechanism, our findings do suggest that LMSCs are capable of contributing to lung remodeling in a manner that leads to functional improvement rather than scarring at 28 days. However, it is possible that LMSCs assume a more fibrogenic phenotype at later time points that could ultimately lead to scarring at the site of engraftment. Longer term studies are needed to address this question.
In culture, LMSC expression of growth factors was associated with biologically relevant paracrine effects. LMSCs grown on transwell filters promoted epithelial cell proliferation. While FGF2 and FGF10 were the only growth factors for which protein expression was confirmed, LMSCs expressed transcripts for other growth factors that could also contribute to regenerative responses, including vascular endothelial growth factor-A (VEGF-A), VEGF-C, and angiopoetin-1. LMSCs may promote epithelial and endothelial cell haptotaxis through secretion of ECM components, another potentially important paracrine pathway. Attachment assays showed that ECM deposited by LMSCs promoted epithelial cell attachment and spreading equivalent to that produced by type 1 collagen and Matrigel.
The FFP scaffold used to facilitate cell attachment and spreading in these experiments functioned better in vitro than the fibrinogen–fibronectin–vitronectin (FFV) scaffold system described previously (10). FFP also promoted cell cycling by minimizing LMSC lag phase following passage. This may simply reflect more efficient cell attachment, as LMSCs are attachment dependent cells. Alternatively, it may reflect specific cell–substrate interactions. Syndecan-2, which is abundantly expressed on the surface of LMSCs, interacts with PLL through its heparan sulfate side chains. Simultaneous syndecan-2 (through PLL binding) and α5β1 integrin activation (through fibronectin binding) have been shown to activate mitogen activated protein (MAP) kinase, which could lead to initiation of cell cycling (4,5).
Despite the use of autologous cells, only a fraction of LMSCs delivered to the alveolar compartment appears to engraft the alveolar septum. This may be due to several factors. Pretreatment processing of LMSCs for endobronchial administration is associated with cellular injury. FACS analysis indicates that trypsinization, mixing of cells with delivery scaffold, and administration through the catheter reduces cell viability 10–15%, presumably due to trauma that leads to necrosis and apoptosis. Additional cell death in situ likely results from failure of LMSCs to home to and bind their niche. This is most likely to occur in regions where the niche has been modified by inflammation or fibrosis. Because these cells are attachment dependent, failure to attach likely results in cell death through anoikis, leading to phagocytosis by macrophages (6). Alternatively, low-grade chronic inflammation in areas of damaged lung could lead to clearance due to downregulation of CD47+ on the surface of LMSCs. CD47+, a transmembrane regulatory molecule that promotes cell attachment and spreading through interactions with ECM components, also suppresses macrophage-mediated phagocytosis through SIRPα binding (8,26). Present on virtually all cells, downregulation of CD47+ on LMSCs following processing and bronchoscopic delivery could blunt attachment and spreading, resulting in phagocytosis (28,39).
Although many LMPC-treated areas displaying increased cellularity did not have the histological appearance of normal lung, they did have more normal architecture and greater surface area than control regions, and thus could represent regions of improved gas exchange. Indeed, scintigraphy scanning confirmed increases in overall perfusion, showing these sites were not avascular scars. Single lung measurements of diffusing capacity (DLco) were completed in only a subset of test animals (three treated animals, two control animals) for technical reasons (incomplete DLco assessments resulted from equipment failure at the time of our final measurements), but results showed increases in gas exchange following LMSC treatment compared to controls. One month following LMSC treatment, single breath DLco increased 10.4 ± 4.4% from baseline, while no change from baseline (–0.4 mean change; range −2.2% to +1.4%) was observed in control animals. However, definitive conclusions cannot be made from this small sample, and confirmation of treatment-related changes in diffusing capacity awaits future studies.
In conclusion, our findings suggest it may be possible to regenerate functional lung tissue in emphysema using autologous lung-derived mesenchymal cells that secrete basement membrane proteins and growth factors. Although data are presented for only a small number of animals and results are largely descriptive, results show that autologous LMSC therapy using a biocompatible scaffold to promote engraftment is associated with tissue remodeling and increased perfusion without scarring or inflammation. Questions concerning mechanism of action and pattern of physiological response following autologous LMSC therapy remain topics for future investigation. Specifically, the relative importance of direct versus paracrine LMSC contributions to regeneration, effects of transplantation on gas exchange, determinants of LMSC survival following transplantation, and long-term responses to treatment all remain to be characterized. Despite these limitations, this approach could represent a practical alternative to conventional stem cell-based therapy for treatment of emphysema.
Footnotes
Acknowledgments
Supported by NIH R01-HL090145, FAMRI grant #072161-CIA, and the Alpha-1 Foundation & American Thoracic Society research award #103342. The authors declare no conflicts of interest.
