Abstract
Hypothyroidism, a prevalent endocrine disorder characterized by insidious onset and prolonged progression, leads to metabolic slowdown and multiorgan dysfunction, significantly compromising patients’ quality of life. While hormone replacement therapy improves most patients’ serum thyroid function, it faces limitations, including individual response variations, lifelong medication requirements, and potential risks, with no reversal of existing tissue damage. In recent years, the mesenchymal stem cells (MSCs) and their derivatives have gained prominence in immunomodulation and regenerative medicine because of their abundant sources, low immunogenicity, potent paracrine effects, and multidirectional differentiation capabilities for tissue repair. Their unique advantages in modulating the immune microenvironment, promoting angiogenesis, reducing fibrosis, and stimulating endogenous cell regeneration offer novel strategies to overcome treatment bottlenecks in hypothyroidism and its complications, effectively repairing thyroid and multiorgan damage in animal models. This review synthesizes literature from the PubMed and Web of Science databases, incorporating relevant basic research and clinical trials. This study examined the systemic impacts of hypothyroidism, evaluated the experimental and clinical roles of the MSCs in thyroid tissue reconstruction and organ protection, and analyzed molecular mechanisms, including immune regulation, the antioxidant stress response, anti-apoptosis, and cellular function restoration. Clinical application potential is assessed alongside critical challenges such as standardized preparation, long-term safety, in vivo homing efficiency, and large-scale production.

Keywords
Introduction
Epidemiological characteristics, clinical diagnosis, and management strategies for hypothyroidism
The thyroid gland, the largest endocrine organ in the human body, secretes thyroid hormones (THs) that regulate cellular metabolism throughout the body, influencing growth and development, metabolism, and multiple systems, including the cardiovascular, skeletal muscle, nervous, digestive, and respiratory systems1,2. Thyroid disorders are the second most common endocrine disease after diabetes, accounting for approximately 40% of all endocrine diseases 3 , with hypothyroidism accounting for 30%–50% of these cases 4 , and the incidence rate ranges from 0.2% to 10% 5 .
Hypothyroidism is caused primarily by insufficient synthesis or secretion of THs or a weakened physiological effect, leading to a systemic reduction in metabolism. The typical clinical manifestations include fatigue, chills, weight gain, memory impairment, constipation, and dry skin. The symptoms of hypothyroidism are subtle and nonspecific, pose significant challenges for clinical diagnosis5,6, and can vary on the basis of factors such as age, sex, and severity of the condition. A definitive diagnosis relies on thyroid function tests combined with imaging assessments, and high-risk factors include iodine deficiency, female sex, pregnancy, autoimmune diseases, and genetic susceptibility. Clinical management involves thyroid hormone therapy (THT) and regular follow-up, with oral levothyroxine (LT4) monotherapy being the cornerstone of hypothyroidism treatment. Its absorption is highly dependent on the gastric acid environment, so it must be taken on an empty stomach to ensure optimal bioavailability 7 . Dietary factors, such as caffeine and calcium and iron supplements, can reduce the blood concentration of LT4. Patients with hypothyroidism who have gastrointestinal diseases or comorbidities affecting the heart, liver, and kidneys may take multiple medications that interfere with LT4 absorption, increasing the likelihood of inadequate treatment 8 . Therefore, it is essential to systematically review drug interactions before starting treatment 9 . Elevated estrogen levels during pregnancy can increase the risk of errors in thyroid function tests. In early pregnancy, it is crucial to increase the appropriate dose of LT4 and shorten the follow-up period, which is essential for fetal neural development and preventing miscarriage or premature birth10,11. By dynamically adjusting the dosage, thyroid function levels can be targeted. If not properly treated, severe cases can progress to myxedema coma, a life-threatening condition 5 .
Long-term follow-up has shown that even with well-controlled biochemical indicators, patients may still experience “residual symptoms” characterized by fatigue, cognitive impairment, and metabolic abnormalities 12 . The mechanism behind these symptoms may be related to differences in the efficiency of local THs conversion or receptor sensitivity 13 . To address the limitations of traditional LT4 monotherapy, research into combination therapies and new THs analogues (such as sobetirom) is becoming increasingly important 14 .
However, whether optimizing alternative therapies or developing novel hormone analogues, their mechanisms of action remain confined to the scope of “hormone replacement”—regulating endocrine indicators through exogenous supplementation. This mechanism lacks the direct repair capabilities for organic or functional damage to multiple systems (e.g. cardiovascular and nervous tissues) caused by hypothyroidism itself. The aforementioned pharmacological advancements still focus on fine-tuning within the TH axis. None of the existing strategies can directly reverse the target organ damage already caused by hypothyroidism. When the disease has led to myocardial remodeling, neuronal dysfunction, or muscle atrophy, simple hormone level correction often proves “too late” 12 .
Therefore, developing novel therapies that achieve “tissue repair” rather than merely hormone replacement has become a critical challenge in this field. While current conventional treatments for hypothyroidism can supplement hormones, they fail to fundamentally repair damaged thyroid tissue and its target organs, presenting inherent limitations (Table 1). Against this backdrop, the mesenchymal stem cells (MSCs) and their derived therapies, which possess immune-regulating, angiogenic, and tissue-repairing functions, emerge as a promising regenerative medicine strategy, offering new possibilities to overcome this therapeutic bottleneck.
Common treatments for hypothyroidism and their advantages and limitations.

Triiodothyronine (T3); thyroxine (T4); thyroid-stimulating hormone (TSH); LT4 (levothyroxine); LT3 (liothyronine).
Definition, origin, and mechanism of the MSCs, extracellular vesicles, and exosomes
The MSCs, which possess self-renewal and multilineage differentiation potential, have become crucial therapeutic tools in regenerative medicine because of their proliferative capacity, differentiation characteristics, and immune regulatory functions. These cells can be isolated from various tissues, including bone marrow, the umbilical cord, adipose tissue, endometrial polyps, the placenta, and menstrual blood28–30. Both in vitro and in vivo studies have demonstrated that, under specific conditions, the MSCs can differentiate into adipocytes, chondrocytes, and osteoblasts 29 . Notably, the MSCs from different tissue sources exhibit significant differences in their differentiation capacity, transcriptomic profiles, and biological functions 31 . The currently recognized phenotype of human MSCs is characterized by nonexpression of hematopoietic markers (CD45, CD34, and CD14) and costimulatory molecules (CD80, CD86, and CD40) but varying levels of expression of CD105, CD73, CD44, CD90, CD71, GD2, and CD271. In addition, they display positive STRO-1 antibody binding, with the expression of these markers potentially influenced by species, tissue origin, and culture conditions 29 . The low immunogenicity and immune-regulating properties of the MSCs make them suitable for both autologous and allogeneic transplantation. Furthermore, their antiapoptotic, paracrine, and multidirectional differentiation capabilities have driven their application in translational research and clinical trials for treating various diseases 32 .
Mesenchymal stem cell–derived extracellular vesicles (MSC-EVs) are lipid-enriched membrane vesicles containing heterologous bioactive molecules from donor cells (including proteins, nucleic acids, lipids, and metabolites) that serve as crucial mediators of both local and long-range intercellular communication33,34. These vesicles inherit the biologically active components and functions of their parent cells while exhibiting lower toxicity, superior biocompatibility, enhanced stability, and reduced immunogenicity. Leveraging their efficient cellular communication capabilities and significant safety advantages, the MSC-EVs have emerged as a promising cell-free strategy to replace traditional MSC therapies 35 . Exos, which are EVs measuring 40–100 nm in diameter, carry bioactive substances such as proteins, lipids, and nucleic acids. Through specific packaging mechanisms, these contents are delivered to target cells to regulate disease progression.
The MSCs and their derivatives have potential applications in soft tissue repair and regeneration, particularly in promoting bone, skeletal muscle, and tendon healing 36 , as well as peripheral nerve regeneration. In cardiovascular regeneration, they facilitate cardiac repair by protecting myocardial cells from ischemic damage, promoting neovascularization, and inhibiting vascular remodeling 37 . Although the MSCs and their derivatives have been widely applied in the treatment of various chronic and refractory diseases, relatively few studies have reported their basic and clinical applications in thyroid-related disorders.
Materials and methods
This review synthesizes current evidence on the therapeutic mechanisms of MSCs and their derivatives for hypothyroidism-induced multi-system injuries. To ensure comprehensive and thematically focused coverage, targeted literature searches were conducted in PubMed and Web of Science up to July 2025.
Customized search strategies were designed for four core pathological domains:
Thyroid dysfunction: Combining MSC-related terms with keywords for hypothyroidism and autoimmune thyroiditis (AIT) models.
Cardiovascular complications: Integrating terminology for specific MSC sources, relevant disease models (e.g. myocardial infarction), and key mechanisms such as angiogenesis and anti-fibrosis.
Skeletal muscle injury: Focusing on MSC types, muscle disease models (e.g. atrophy, dystrophy), and repair pathways like myogenic differentiation.
Neurological impairment: Employing terms for MSCs and their derivatives, neurological disorders (e.g. Alzheimer’s disease (AD), stroke, cerebral ischemia), and neuroprotective mechanisms (e.g. neurogenesis, modulation of neuroinflammation).
Retrieved records were collated and screened using standard literature management and selection processes. Inclusion was limited to original preclinical and early-phase clinical studies published in English, while reviews, editorials, and case reports were excluded.
As a narrative synthesis, this review does not employ the exhaustive search or formal quality appraisal characteristic of a systematic review. However, the structured, domain-specific search approach ensures a representative and logically coherent integration of the current evidence base, while transparently acknowledging the selective nature of the methodology.
Hypothyroidism: Multisystem influence mechanism and treatment difficulties
Molecular mechanism of THs bioactivity regulation and autoimmune hypothyroidism
THs primarily consist of T4 and T3. The thyroid gland synthesizes approximately 85% of T4, whereas the bioactive T3 constitutes only 15%. Peripheral tissues activate T4 into more biologically active T3 through deiodinase enzymes. Type 1 deiodinase (Dio1), which is expressed mainly in the liver and kidneys, contributes to the production of 15%–20% of circulating T3. Type 3 deiodinase (Dio3), predominantly found in the pituitary gland, heart, brain, and adipose tissue, generates approximately two-thirds of circulating T3 and converts THs into inactive forms to terminate their effects 3 . Secreted T4 must undergo 5′-deiodination catalyzed by peripheral deiodinases (primarily in the liver and kidneys) to convert into active T3. Notably, over 80% of circulating T3 originates from this peripheral conversion process rather than direct thyroid secretion 38 . Impaired 5′-deiodinase activity leads to reduced T3 levels in circulation and tissues, resulting in hypothyroidism. This hormonal system is precisely regulated by the hypothalamic–pituitary–thyroid (HPT) axis: thyrotropin-releasing hormone (TRH), which is secreted by the hypothalamus, activates the anterior pituitary to produce thyroid-stimulating hormone (TSH), which stimulates thyroid follicular cells to synthesize and release T4/T3. Simultaneously, T3 and T4 maintain dynamic equilibrium by inhibiting TRH and TSH secretion through negative feedback mechanisms 38 . Any disruption in this integrated regulatory network may cause THs metabolic disorders.
In clinical practice, thyroidectomy, AIT, and radioactive iodine-131 therapy are the most common causes of hypothyroidism 39 . AIT is the primary etiology of primary hypothyroidism, characterized by lymphocytic infiltration of the thyroid gland and positive serum anti-thyroid peroxidase and anti-thyroglobulin antibodies 32 . Although the exact pathogenesis remains incompletely understood, immune abnormalities, genetic factors, and environmental influences are considered to play significant roles 40 . As AIT progresses, destruction of the thyroid follicular epithelium leads to reduced hormone synthesis, accelerating the development of subclinical and clinical hypothyroidism 41 (Figure 1). In addition to having impaired thyroid function, patients with hypothyroidism also face increased risks of malignancies and cardiovascular diseases42,43. Currently, oral LT4 is the standard replacement therapy for hypothyroidism of various etiologies 44 , which typically requires lifelong medication.

Molecular mechanisms of thyroid hormone synthesis, peripheral activation and regulation, and pathological states of imbalance. The hypothalamic–pituitary–thyroid (HPT) axis regulates T4/T3 synthesis, with hormone release inhibiting further production via negative feedback. Most circulating T3 derives from peripheral conversion of T4 by deiodinases. Destruction of follicular cells by autoimmune disease, surgery, or radioiodine therapy impairs hormones synthesis, leading to hypothyroidism and necessitating lifelong THs replacement. Created with BioRender.com.
Hypothyroidism: Mechanisms of cardiovascular injury and limitations of treatment
Cardiovascular injury: Multiple pathomechanisms and clinical risks mediated by THs deficiency
Bioactive T3 has significant effects on cardiomyocytes. Hypothyroidism increases the risk of ischemic heart disease, myocardial infarction, and cardiovascular mortality through multiple mechanisms, including metabolic disorders, endothelial damage, atherosclerosis, and myocardial remodeling 38 (Figure 2). This condition directly regulates the cardiovascular system via both genomic and nongenomic pathways while mediating systemic metabolic‒cardiovascular dysregulation.

Pathophysiological mechanisms of cardiovascular manifestations in hypothyroidism. The diagram links thyroid hormone deficiency to cardiovascular disease through interconnected pathways. This includes impaired vasodilation and reduced vascular compliance, promoting hypertension and atherosclerosis via inflammation and arterial stiffening. Concurrent metabolic dysregulation elevates cholesterol and triglycerides. Clinically, this manifests as electrocardiographic abnormalities (e.g. bradycardia, prolonged QT interval). Ultimately, these changes impair cardiac relaxation and filling, leading to diastolic dysfunction, reduced output, and increased vascular resistance. Created with BioRender.com.
At the metabolic level, hypothyroidism activates HMG-CoA reductase to promote cholesterol biosynthesis. Simultaneously, it downregulates hepatic low-density lipoprotein (LDL) receptor expression and inhibits the CYP7A1-mediated clearance of low-density lipoprotein cholesterol (LDL-C), leading to elevated total cholesterol (TC), LDL-C, and triglyceride (TG) levels while compensating for the reduction in high-density lipoprotein cholesterol (HDL-C) 3 . Reduced lipoprotein lipase (LPL) activity further hinders the hydrolysis of TG-rich chylomicrons, resulting in a characteristic lipid metabolism imbalance. Hypothyroidism also causes glucose transporter 4 (GLUT4) translocation disorders in peripheral tissues and increases hepatic phosphoenolpyruvate carboxykinase and glucose-6-phosphatase expression, promoting gluconeogenesis and increasing type 2 diabetes risk 38 . Through mechanisms such as disrupting lipid/glycogen metabolism homeostasis, inducing insulin resistance, and enhancing oxidative stress, hypothyroidism exacerbates the pathological process of metabolic dysfunction. This amplifies metabolic-related cardiovascular risks—including accelerated atherosclerosis, deteriorated endothelial function, and imbalance in myocardial energy metabolism—ultimately significantly increasing the incidence of adverse cardiovascular events.
Cardiac changes associated with hypothyroidism manifest through electrocardiogram abnormalities and echocardiographic findings. These alterations result primarily from the direct regulation of myocardial cell electrophysiological properties by THs (particularly T3) and vascular dysfunction. During disease progression, early-stage myocardial injury predominantly involves left ventricular diastolic dysfunction, whereas advanced stages progress to dual impairment of both systolic and diastolic functions, accompanied by significant electrophysiological abnormalities that substantially increase the risk of malignant arrhythmias 45 . Even in subclinical hypothyroidism (SCH), clinical observations reveal increased carotid intima‒media thickness, diminished flow-mediated dilation (FMD), and elevated arterial stiffness—pathological changes collectively promoting inflammatory responses, oxidative stress, and endothelial dysfunction46,47. Within the pathological context of hypothyroidism, these vascular abnormalities accelerate atherosclerosis through mechanisms including impaired synthesis of vascular endothelial nitric oxide (NO), leading to peripheral resistance elevation, coagulation‒fibrinolysis system imbalance, and abnormal levels of inflammatory markers such as lipoprotein A and homocysteine. Clinical studies have demonstrated an independent correlation between elevated serum TSH levels and increased cardiovascular risk in young SCH patients 48 ; hypothyroidism significantly increases hospitalization risk due to heart failure exacerbation 47 . In addition, hypothyroidism-associated thyroxine deficiency syndrome occurs in 20%–30% of chronic heart failure patients and is strongly associated with all-cause mortality 47 . Severe hypothyroidism may cause hyperproteinous pericardial effusion due to increased capillary permeability and impaired lymphatic drainage, with rare cases progressing to cardiac tamponade. Therefore, early identification and intervention are critical to effectively reduce the risk of cardiovascular events.
Cardiovascular protection by LT4: Controversy over improved surrogate markers and the absence of hard endpoints
Vascular endothelial function serves as a critical biomarker for cardiovascular risk. Small-scale studies indicate that THT may improve endothelial function, with preliminary evidence supporting its potential cardiocerebrovascular protective effects (e.g. enhanced cardiac function and vascular endothelial function) 49 . Experimental data show that LT4 treatment not only corrects thyroid dysfunction but also significantly reduces TC, LDL-C, C-reactive protein (CRP), and bradykinin B1 (BNP) levels, suggesting improvements in metabolic and inflammatory states 49 . More importantly, LT4 treatment has been proven to markedly increase blood flow–mediated FMD and decrease pulse wave velocity (PWV), indicating its ability to improve endothelial function and arterial stiffness 49 . These synergistic hemodynamic improvements support the hypothesis that LT4 exerts cardiovascular protective effects through multiple pathways, including metabolic regulation, anti-inflammatory effects, and vascular repair. Although limited in sample size, these studies provide new perspectives on the pathological link between thyroid function and cardiovascular risk, highlighting the potential value of LT4 in the secondary prevention of cardiovascular diseases.
However, large-scale research findings remain controversial. Studies by Feller et al. 50 indicated that LT4 therapy failed to significantly reduce all-cause mortality, cardiovascular events, atrial fibrillation, or heart failure risk in elderly patients with SCH regardless of their cardiovascular history or age. Consistently, a meta-analysis revealed that LT4 treatment did not substantially lower systolic blood pressure in SCH patients 3 . McAninch et al. noted that among multiple lipid profile indicators (TC, LDL-C, HDL-C, and TG), only TC significantly improved after LT4 therapy. They further emphasized that while such therapy corrects hypothyroidism, it cannot resolve tissue-specific T3/T4 conversion disorders or reverse the progression of myocardial fibrosis 51 (Figure 3).
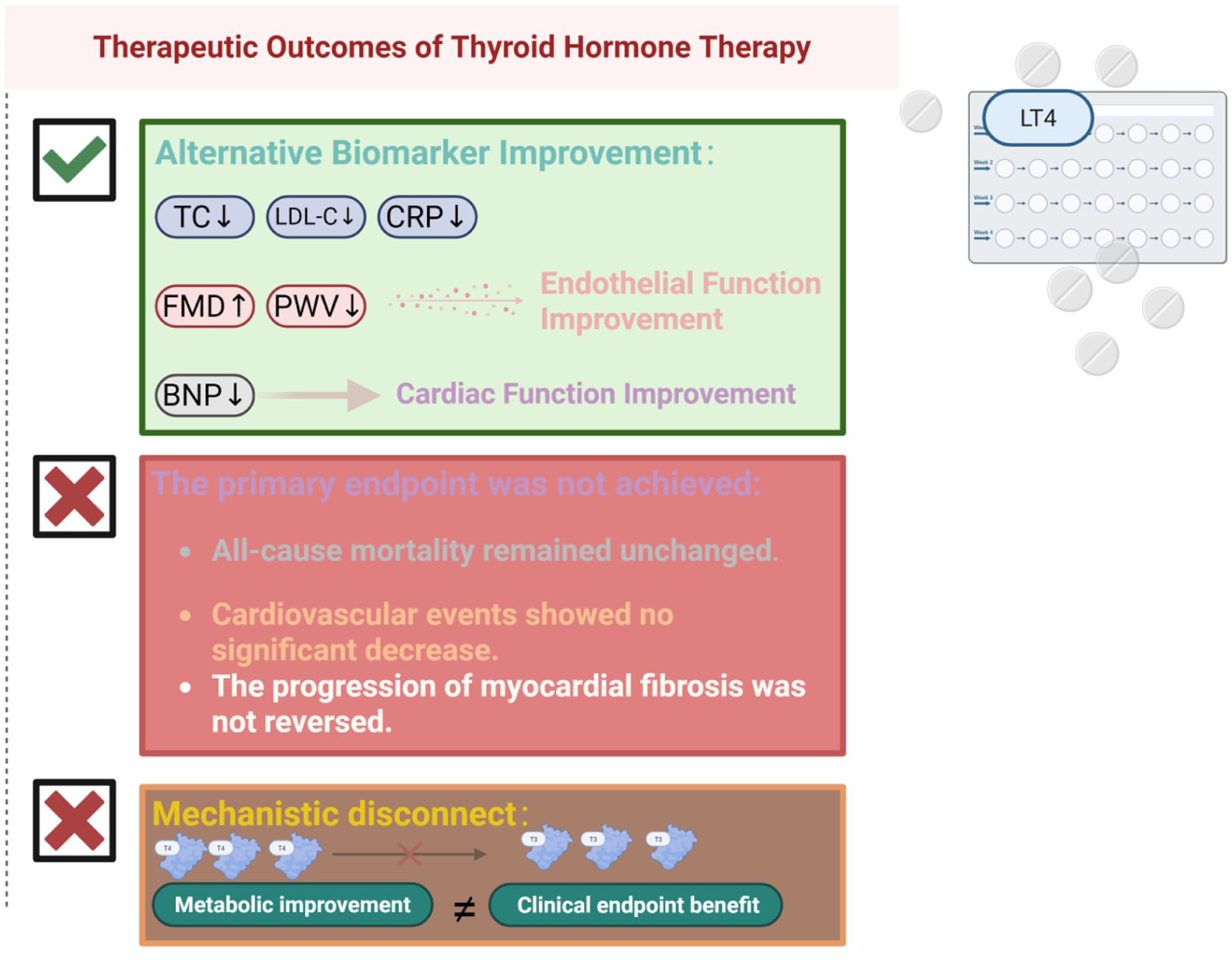
Observed therapeutic outcomes and clinical endpoint paradox in thyroid hormone therapy (THT) for hypothyroidism. The diagram illustrates the disconnect between biomarker improvement and clinical outcomes. While THT effectively improves cardiovascular biomarkers—such as lowering cholesterol, CRP, and NTproBNP, and enhancing endothelial function—these changes do not translate into reduced mortality, fewer cardiovascular events, or reversal of myocardial fibrosis. This highlights a mechanistic gap between intermediate functional benefits and meaningful clinical endpoints. Created with BioRender.com.
Hypothyroidism myopathy: Damage to skeletal muscle structure and function mediated by multiple mechanisms and networks
Hypothyroidism can induce myopathy characterized by skeletal muscle dysfunction, with clinical manifestations including muscle weakness, fatigue (occurring in up to 80% of cases), muscle spasms, and stiffness. Severe cases may develop pseudohypertrophy and pseudo-rigidity 52 (Figure 4). The pathological mechanism involves abnormal TH signaling: inactive T4 in skeletal muscle cells must be converted into active T3, which regulates muscle development, contraction function, and thermogenesis through nuclear receptors. In hypothyroidism, the balance between muscle protein synthesis and degradation is disrupted, leading to a net catabolic state that causes muscle fiber atrophy and reduced muscle mass 53 . Clinical evidence shows that patients with hypothyroidism following unilateral thyroid lobectomy exhibit significantly lower skeletal muscle mass than those with normal thyroid function does, suggesting a correlation with postoperative complication risk 54 .

Pathological mechanisms and clinical manifestations of hypothyroidism-induced skeletal muscle injury. The diagram depicts how thyroid hormone signal defects cause metabolic dysregulation and alter drug sensitivity, disrupting the balance between protein synthesis and degradation. This leads to a net catabolic state, resulting in muscle fiber atrophy and mass loss, while concurrently increasing susceptibility to statin-induced myopathy. These cellular injuries clinically manifest as muscle weakness, fatigue, cramps, and pseudohypertrophy. Created with BioRender.com.
Metabolically, hypothyroidism enhances the adaptive response of skeletal muscle to chronic exercise stimuli, as manifested by increased β-oxidation capacity, an elevated proportion of major histocompatibility complex class I (MHC I) slow-twitch muscle fibers, and a compensatory increase in the protein synthesis rate 55 . However, such compensatory mechanisms may be accompanied by mitochondrial dysfunction and impaired regeneration capacity, leading to decreased muscular endurance and significantly elevated serum creatine kinase levels. Notably, hypothyroid patients often require statin therapy due to concurrent endogenous hypercholesterolemia. However, their skeletal muscles demonstrate heightened sensitivity to drug toxicity, with approximately 10% of patients developing myopathy symptoms ranging from mild spasms to rhabdomyolysis, which undoubtedly exacerbates skeletal muscle damage 52 .
Age-related factors further complicate the progression of hypothyroidism-induced myopathy. Although Schwarzler’s hypertrophic myopathy is prevalent among elderly people, treatment with THT does not significantly improve muscle strength, function, or quality in this population 52 . Moreover, dysregulation of skeletal muscle-system-wide metabolic interactions may exacerbate age-related disease development. In summary, hypothyroidism impairs skeletal muscle structure and function through multiple mechanisms, including hormonal signaling defects, metabolic pathway abnormalities, and altered drug sensitivity. Interventions targeting these therapeutic targets may provide novel approaches for clinical management 56 .
Mechanisms of nerve damage: From developmental brain region–specific damage to glial immune damage
Multidimensional damage to the nervous system during development and selective brain region mechanisms
THs regulate brain development and function throughout the lifespan, with homeostatic imbalance causing multidimensional neurological damage (Figure 5). Studies on rodent models of hypothyroidism revealed structural brain damage characterized by significant atrophy, including whole-brain volume reduction, cortical thinning, decreased vascular density, and reduced myelin sheathing in white matter regions such as the corpus callosum57,58. These effects exhibit notable sex-specific differences: male mice, owing to high thyroid hormone receptor alpha (TRα) expression in the hippocampus, show greater sensitivity of the neural stem cells (NSCs) to THs fluctuations, whereas females demonstrate greater functional compensatory capacity 59 . The combined effects of these structural changes and sex-specific mechanisms ultimately lead to cognitive deficits, enhanced anxiety-like behaviors, and white matter developmental abnormalities in model animals60,61.

The characteristics and clinical manifestations of the effects of hypothyroidism on the nervous system. The diagram illustrates the progressive damage to the central nervous system resulting from impaired thyroid hormone signaling. Dysfunction of nuclear receptors (TRα/TRβ), along with reduced hormone transport (MCT8) and local activation (DIO2↓), suppresses expression of key genes such as BDNF and MBP. This suppression triggers glial cell damage and neuronal injury, characterized by mitochondrial dysfunction and impaired synaptic plasticity. These combined cellular deficits drive extensive neurostructural remodeling, including gray and white matter atrophy, neuronal migration disorders, blood–brain barrier dysfunction, and vascular network rarefaction. Created with BioRender.com.
Although rodent models provide valuable insights into the neuropathological mechanisms of hypothyroidism, significant differences in brain cell types, neural network topology, and spatiotemporal myelination patterns between rodents and humans limit the clinical translation of findings 62 . Human neuroimaging evidence indicates that hypothyroidism causes cortical volume reduction, decreased white matter myelination, and vascular network thinning 63 , with a critical developmental window for the brain’s sensitivity to THs. Hypothyroidism occurring during pregnancy or early life can significantly impair neuronal density in the cerebral cortex, hippocampal synaptic plasticity, and the signal integration function of cerebellar Purkinje cells. Children with SCH exhibit atrophy of the frontal/parietal cortex, abnormal elevation of cholinergic compounds and N-acetyl-aspartate levels in the hippocampus and cerebellum, accompanied by disordered white matter fiber structures such as the corpus callosum64,65. Owing to impaired myelin integrity in major fiber tracts such as the corpus callosum and corticospinal tracts in hypothyroid patients, axonal conduction delays occur66,67, leading to language, cognitive, memory, and motor coordination disorders59,64,68. In addition, in hereditary THs signaling defects, TRα mutations cause resistance to TH alpha, which is characterized by reduced nonverbal IQ and sensory‒motor integration disorders; thyroid hormone receptor beta (TRβ) dominant‒negative mutations result in resistance to TH beta, which is characterized by severe intellectual disability and attention deficit symptoms69,70; and SLC16A2 mutations cause monocarboxylate transporter 8 (MCT8) dysfunction, triggering Allan–Herndon–Dudley syndrome (AHDS), which manifests as progressive myelin degeneration, abnormal neuronal migration, and oligodendrocyte maturation arrest71,72.
In adults and elderly individuals, thyroid dysfunction is closely associated with neuropsychiatric disorders: reduced gray matter volume in the frontal/occipital regions and decreased GABA concentration in the medial prefrontal cortex lead to an imbalance in the inhibitory–excitatory network, causing depression and executive dysfunction 71 . Notably, elderly hypothyroid patients have a significantly increased risk of cognitive decline, involving mechanisms related to impaired β-amyloid clearance and excessive Tau protein phosphorylation 73 . Intervention studies have shown that early initiation of LT4 therapy can reverse GABA deficiency and improve memory function 72 .
In conclusion, hypothyroidism exhibits distinct developmental stage specificity and regional selectivity in neurodegenerative processes. THs regulate myelin formation, synaptic plasticity, and neurotransmitter homeostasis to maintain brain function. Deficiencies in the spatiotemporal specificity of THs signaling pathways—manifested as developmental transport disorders, metabolic dysregulation in adulthood, and clearance impairment in old age—constitute a potential common mechanism underlying neuropsychiatric disorders.
Neuroregulation of THs: Imbalance of two pathways and glial-immune injury
At the molecular level, THs regulate neural function through dual genomic and nongenomic pathways. In the genomic pathway, THs bind to the nuclear receptors TRα/TRβ to form complexes that directly activate the expression of key genes such as brain-derived neurotrophic factor (BDNF) and myelin basic protein (MBP). Hypothyroidism disrupts this signaling pathway, significantly inhibiting neurogenesis and myelination, thereby affecting memory formation and anxiety-like behaviors74–76. Research has revealed that T3 specifically suppresses Sox2 expression via the nuclear receptor TRα1, driving the differentiation of the NSCs into neurons. THs deficiency during development impairs Sox2 suppression while simultaneously downregulating the EGFR pathway (which promotes glial differentiation), ultimately hindering neurogenesis and disrupting the neuron/glia output balance, impacting nervous system function 77 . In the nongenomic pathway, THs primarily mediates the rapid regulation of cellular energy homeostasis, including the modulation of mitochondrial respiratory chain complex activity and reactive oxygen species (ROS) metabolic balance. In the central nervous system, approximately 80% of active T3 rely on local conversion from T4 by type 2 deiodinase (Dio2) within glial cells, such as astrocytes, followed by transport into neurons via MCT8. The hypothyroid state inhibits Dio2 activity, reducing intracellular active T3 generation and exacerbating energy metabolism dysregulation78–80.
Notably, hypothyroidism causes multifaceted damage to glial cells: Dysfunction of the TH transporter (MCT8/OATP1C1) in astrocytes leads to autometabolic abnormalities and mitochondrial respiratory defects, thereby weakening their neurotrophic support capacity 78 . During developmental hypothyroidism, the Sonic Hedgehog signaling pathway is inhibited, reducing the number of parvalbumin-positive GABAergic interneurons in the cortex and disrupting inhibitory neural microcirculation 81 . Immune interactions further amplify neural injury: thyroid peroxidase antibodies (TPO-Ab) bind to astrocytes, triggering autoimmune inflammation. Pro-inflammatory factors such as interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α) enhance neuronal excitability by downregulating GABA receptor expression 78 . This triple dysfunction involving the glial-metabolic-immune systems creates a cascading damage cascade, ultimately manifesting as neuromotor imbalance, synaptic plasticity impairment, and neural network remodeling disorders. Clinically, this results in cognitive impairment, psychiatric disorders, and increased susceptibility to epilepsy 82 .
Cross-system repair in stem cell engineering: From thyroid immune regulation, cardiac and skeletal muscle-targeted regeneration to neural multimechanism synergy
Stem cell engineering and immune regulation: Cutting-edge advances in modeling, functional repair, and regenerative therapy for thyroid diseases
Improving hypothyroidism and its multisystem complications
The MSCs and their derivatives have demonstrated remarkable efficacy in treating hypothyroidism and related organ damage (Table S2). Transplantation of human amniotic mesenchymal stem cells (hAMSCs) significantly improved thyroid function and multiorgan damage in a mouse model of age-related of subclinical hypothyroidism (AR-SCH). Starting from day 10 of treatment, serum TSH levels decreased markedly, and thyroid autoantibodies (TPO-Ab, TgAb) were reduced, alleviating the destruction of thyroid follicular structures and inflammatory infiltration. In addition, the hAMSCs effectively regulated lipid metabolism, improved hepatic steatosis, and enhanced cardiac function. Mechanistically, the hAMSCs restored the immune balance and inhibited apoptosis in the thyroid, liver, and cardiomyocytes. This therapy reverses core pathological changes in AR-SCH through the dual effects of immunomodulation and anti-apoptosis, effectively improving thyroid function, metabolic disorders, and tissue damage, providing a novel cellular therapeutic strategy for patients unresponsive to conventional treatments 83 . Hypothyroidism significantly impairs bone repair capacity in rats, as manifested by decreased biomechanical properties and degeneration of the bone microstructure. The combined application of low-intensity laser therapy and MSC-conditioned medium (MSC-CM) effectively reversed the hypothyroidism-induced inhibition of bone regeneration. In the treatment group, the bone biomechanical parameters returned to normal levels, with increased trabecular volume and osteoblast count, demonstrating that this synergistic therapy improved bone formation metabolism and mechanical strength, suggesting a new therapeutic approach for hypothyroidism-related bone healing disorders. The use of the MSC-CM alone also effectively improved the biomechanical properties and bone morphology of hypothyroidism rats with bone defects 84 . Furthermore, bone marrow–derived mesenchymal stem cells (BM-MSCs) transplantation can repair hypothyroidism-induced pancreatic injury by improving hyperglycemia and insulin secretion, increasing total serum antioxidant capacity, and reducing the levels of the oxidative stress marker malondialdehyde (MDA). Histological evidence confirmed the recovery of the exocrine acinar structure in the pancreas, reducing inflammatory infiltration, improving the islet structure in the endocrine system, and decreasing apoptosis 85 .
Collectively, the studies above demonstrate the therapeutic potential of MSC-based strategies. It is important to distinguish the maturity of this evidence. The capacity of the hAMSCs or BM-MSCs to improve functional and histological outcomes in specific hypothyroid animal models represents a well-established and reproducible finding across independent studies83,85. Similarly, the anti-apoptotic and general antioxidant effects of the MSCs in these contexts are strongly supported. However, other aspects remain in the early-stage or mechanistic hypothesis phase. For instance, the specific contribution of metabolic reprogramming versus direct differentiation to tissue repair, and the long-term stability of immune modulation beyond the experimental period, are inferences based on compelling but preliminary data. The translation of bone repair synergy using laser therapy 84 to clinical settings is also a promising yet speculative avenue requiring validation in more complex models. Thus, while the therapeutic foundation is solid, the precise mechanistic hierarchy and optimal application protocols constitute the current frontier of investigation.
Molecular mechanism regulating the immune microenvironment in the treatment of AIT
The core mechanism by which the MSCs treat AIT lies in restoring immune homeostasis and suppressing inflammatory damage. Specifically, in both AIT patients and experimental autoimmune thyroiditis (EAT) models, thyroid tissue exhibits significant oxidative stress and abnormal activation of inflammatory pathways, including ROS accumulation, overactivation of the STING pathway, NLRP3 inflammatory body activation, infiltration of M1 macrophages (iNOS+), and endoplasmic reticulum stress 86 . Notably, the MSCs effectively ameliorate thyroid pathological damage in EAT. The key mechanism involves inhibiting the STING pathway, thereby suppressing downstream NLRP3 inflammatory body activation and M1 macrophage polarization while significantly alleviating endoplasmic reticulum stress. This pathway has been identified as a critical target in the EAT model. Although its primacy in human AIT and across different MSC sources awaits further confirmation 87 .
In addition to the aforementioned mechanisms, another study revealed the importance of regulating T-cell signaling pathways. Specifically, the human umbilical cord mesenchymal stem cells (hUC-MSCs) can suppress the phosphorylation of signal transduction and activation factor 3 (STAT3) 86 by increasing the expression of protein tyrosine phosphatase non-receptor type 2 (PTPN2) in T cells. This regulatory effect directly leads to a reduction in the proportion of IL-17α+ Th17 cells and an increase in CD25+FOXP3+ Treg cells in the spleen, effectively improving the Th17/Treg imbalance. Concurrently, it is accompanied by decreased serum levels of the Th1-type cytokine interferon-gamma (IFN-γ) and an increased IFN-γ/IL-4 ratio. Crucially, shRNA-mediated knockout of PTPN2 in T cells partially reversed the immunomodulatory effects of the hUCMSCs, strongly confirming that the PTPN2/STAT3 signaling axis plays a central role in hUCMSCs-mediated immune homeostasis reconstruction 86 . Finally, these findings were robustly validated in large animal models. In the canine EAT model, both the adipose-derived stem cells (ADSCs) and CTLA4Ig-ASCs effectively reduce TgAA titers, alleviate thyroid inflammatory infiltration, and increase the proportion of regulatory T cells in the peripheral blood and lymph nodes 85 . In addition, its secretory group can significantly inhibit the proliferation of peripheral blood mononuclear cells, and the transplanted cells are located mainly in immune-related organs such as the spleen, thyroid, and lymph nodes 88 , which provides direct evidence for its mode of action.
The MSC therapy effectively reverses the pathological progression of AIT by synergistically inhibiting inflammatory pathways and reshaping immune homeostasis (Figure 6). Breakthroughs in key mechanistic studies, combined with the application of genetic engineering strategies and successful validation in large animal models, mark pivotal advancements in breakthrough treatments for refractory AIT, accelerating its clinical translation.

Study on the mechanism and pathway of stem cells and their derivatives in treating hypothyroidism and Hashimoto’s thyroiditis. They target key drivers such as immune dysregulation and endoplasmic reticulum stress, which activate harmful inflammatory pathways like the NLRP3 inflammasome. Through immunomodulation (e.g. promoting Tregs and M2 macrophages), cytoprotection (anti-apoptotic, antioxidant), and specific pathway inhibition (notably suppressing STING overactivation), MSCs alleviate inflammation and cellular stress. This multi-targeted action restores tissue homeostasis. Created with BioRender.com.
Synergistic systems of the MSCs and derivative materials: Mechanism and transformation of targeted repair and cell communication driving heart regeneration
Multisource MSCs and derivatives: Heart regeneration is driven by synergistic, paracrine, and targeted communication networks
The MSCs and their derivatives serve as core therapeutic agents in cardiac regeneration, promoting myocardial repair through multiple synergistic mechanisms (Figure 7). The MSCs derived from adipose tissue, bone marrow, and the umbilical cord can differentiate into cardiomyocyte-like cells both in vitro and in vivo; these cells express cardiac-specific genes and induce angiogenesis 89 . Their paracrine effects enhance cardiac function by regulating cardiomyocyte and vascular smooth muscle functions, reducing inflammatory fibrosis, and inhibiting apoptosis 90 . Exos involved in paracrine signaling show promising potential for cardiovascular disease–related cardiac repair. The therapeutic potential stems from multiple beneficial mechanisms, including antifibrotic, anti-inflammatory, proangiogenic, antioxidant, and antiapoptotic effects, which collectively improve cardiac function 91 . MicroRNAs (miRNAs) regulate angiogenesis through three mechanisms: in situ transcriptional suppression (targeted degradation of endothelial/pericyte-derived angiogenic mRNAs), paracrine-mediated delivery (Exos-transmitted miRNA to distal tissues), and microenvironmental reprogramming, ultimately dynamically shaping the tissue microenvironment and demonstrating significant potential in cardiovascular diseases 92 . The precise regulation mediated by the MSC-Exos content is particularly crucial: miR-223-targeted inhibition of inflammatory genes such as IL-6 and IL-1β reduces myocardial injury93,94. As carriers of bioactive molecules (TIMP4), the MSC-Exos achieve functional reprogramming of distal target cells (T cells) through myocardial Exos, regulating the Th17/Treg immune balance to ultimately improve heart failure progression 95 . The overexpression of HIF2α enhances the angiogenic effects of the hUC-MSC-EVs by upregulating miR-146a to inhibit Spred1, thereby activating the AKT/ERK pathway to promote endothelial cell proliferation, migration, and tubule formation 96 .
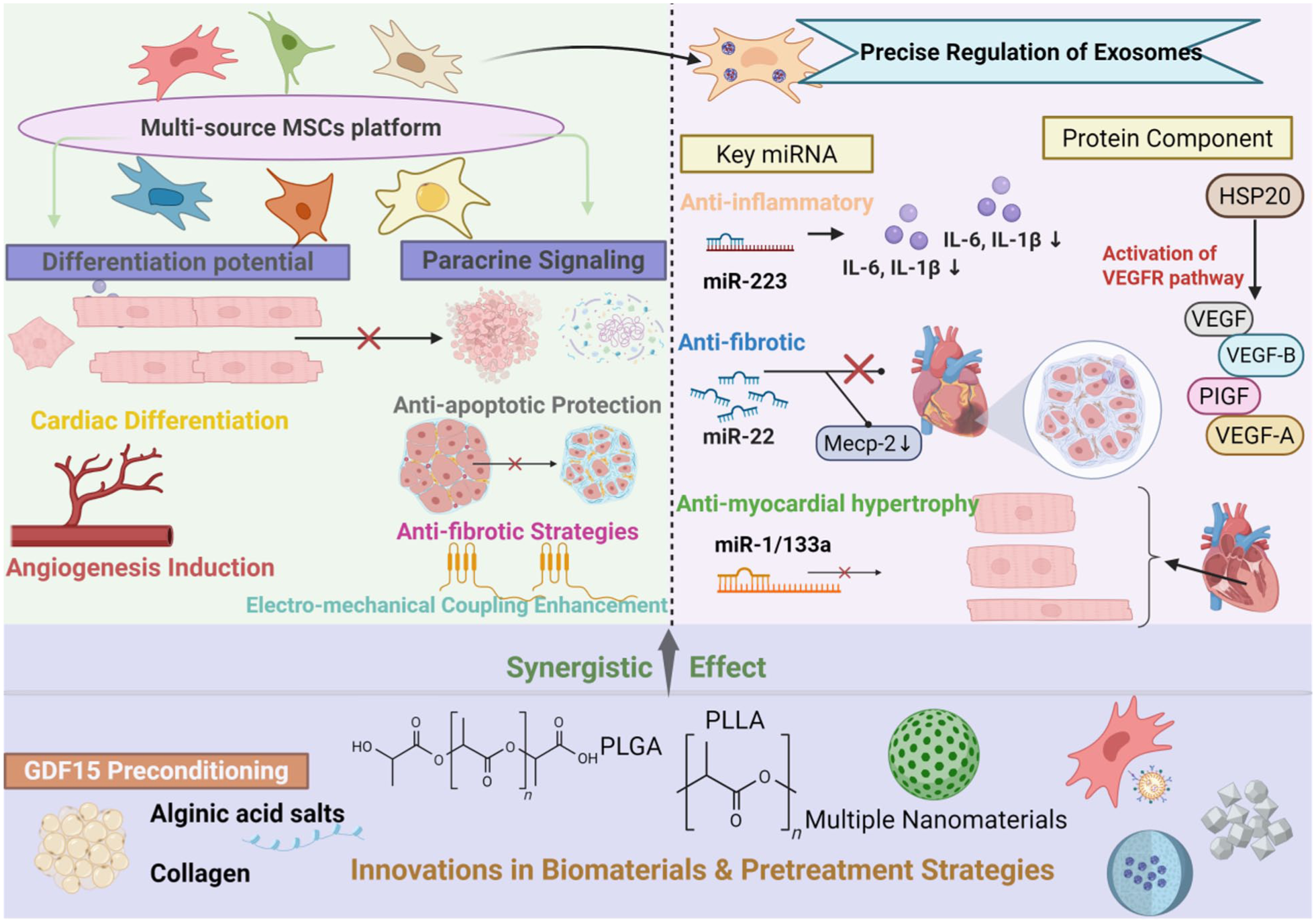
An integrated framework for cardiac regeneration: synergistic MSC networks, animal model validation, and biomaterial-based strategies to overcome therapeutic bottlenecks. This schematic summarizes the integrated therapeutic profile of MSCs derived from various tissues. The platform possesses inherent differentiation potential and operates primarily through a potent paracrine network, which provides anti-apoptotic, anti-fibrotic, and pro-regenerative effects. Its secretome contains key regulatory miRNAs and proteins that modulate inflammation, activate pro-angiogenic pathways (e.g. via VEGF), and deliver protective factors. To amplify these effects, synergistic strategies—including biomaterial encapsulation (e.g. in alginate or PLLA matrices) and preconditioning (e.g. with GDF15)—are employed to enhance the platform’s therapeutic efficacy. Created with BioRender.com.
Mechanistic exploration and translational value of animal models in cardiac regeneration research
Owing to their high similarity to human hearts, large animal models provide crucial platforms for the clinical translation of the MSC and MSC-Exo therapies. Current technological optimizations in this field focus on improving cell sourcing, exploring combination therapy strategies, and applying gene-editing technologies to reduce the risk of immune rejection. For instance, Seibt et al. 97 demonstrated that intramyocardial injection of the ADSCs effectively reduced the incidence of ventricular arrhythmia in a myocardial infarction rat model. The cardioprotective mechanisms include the upregulation of Cx43 expression in infarcted areas, the suppression of the levels of the pro-inflammatory cytokine interleukin-1 beta (IL-1β), and the reduction in the levels of oxidative stress markers such as 4-HNE, indicating that the ADSCs exert therapeutic effects through anti-inflammatory, antioxidant, and electrophysiological coupling improvements. However, the harsh microenvironment following myocardial infarction severely limits the survival and efficacy of the transplanted MSCs. To address this, researchers have developed multiple strategies. Zhuo Wang et al.’s 98 team created an ultrasound-responsive phase-change biomimetic nanoparticle that integrates the MSC membranes with macrophage membranes and carries the key molecule miRNA-125b to regulate cardiomyocyte apoptosis and fibroblast activity. Through ultrasound-targeted microbubble disruption technology, the nanoparticles achieved precise delivery to inflamed myocardial regions, effectively overcoming microenvironmental constraints and significantly promoting cardiac function recovery. In a rat model of myocardial infarction, pretreatment of the MSCs with 2,4-dinitrophenol (DNP) before transplantation significantly increased cell survival in infarcted areas. This treatment also improved cellular homing, adhesion, and differentiation potential, resulting in reduced scar tissue formation, better preservation of left ventricular wall thickness, enhanced angiogenic activity, and improved cardiac function. In addition, the MSC-Exos themselves serve as therapeutic agents through dual mechanisms: upregulating the long noncoding RNA HAND2-AS1 and inhibiting miR-17-5p to activate the Mfn2 pathway, thereby mitigating myocardial injury, apoptosis, oxidative stress, and inflammation induced by hypoxia/reoxygenation 99 . Moreover, it delivers the key molecule miR-21-5p to macrophages, promoting their polarization from the pro-inflammatory M1 phenotype to the anti-inflammatory M2 phenotype. This transformation is characterized by decreased expression of M1 markers (iNOS, IL-1β, IL-6, and TNF-α) and increased M2 marker levels, accompanied by reduced inflammatory factor levels and elevated anti-inflammatory agent levels, effectively reducing inflammatory responses and promoting cardiac repair 100 . Notably, this protective effect is miR-21-5p dependent.
In summary, researchers have effectively overcome microenvironmental constraints through multiple approaches, significantly improving transplant cell survival rates, targeting efficiency, and repairing efficacy, ultimately facilitating the restoration of cardiac structure and function.
Biological materials and pretreatment strategies work together to overcome the efficacy bottleneck of heart regeneration therapy
While stem cells and Exos therapies show significant potential in myocardial repair, their therapeutic efficacy is constrained by the low differentiation efficiency of stem cells into cardiomyocytes. There is an urgent need to develop novel biomaterials and pretreatment strategies to synergistically optimize transplantation outcomes. In biomaterial development, natural polymers such as collagen and alginate, along with synthetic degradable materials such as poly-lactic-co-glycolic acid (PLGA) and polylactic acid, have been widely applied to reshape the extracellular matrix microenvironment, regulate paracrine signaling, and serve as delivery carriers 101 . Moreover, nanomaterials enhance repair efficacy through multiple pathways: gold nanoparticles improve cardiac troponin detection sensitivity; injectable hydrogels loaded with vascular endothelial growth factor (VEGF) and other factors promote angiogenesis; and PLGA nanoparticles target macrophage polarization to restore immune homeostasis. Notably, alginate hydrogels encapsulating miR-126/146a-loaded Exos synergistically promoted angiogenesis, enhanced component retention, and achieved multitarget antifibrotic effects. This significantly reduces the infarcted myocardial area and increases CD31/Connexin43 expression, providing a novel strategy for preventing and treating postmyocardial infarction heart failure102,103. In response to stem cells pretreatment, the GDF15-pretreated MSCs inhibited mitochondrial division and ROS generation by activating the AMPK pathway, significantly improving cells survival under ischemic conditions while enhancing paracrine functions. This approach improved cardiac function and fibrosis 104 in a mouse model of myocardial infarction. In addition, the hAMSCs combined with PGS-co-PCL films synergistically enhanced cardiac function parameters (left ventricular ejection fraction, fractional shortening, and stroke volume) and increased VEGF expression in myocardial ischemia‒reperfusion injury models 105 (Table S3). Furthermore, HSP20-mediated myocardial Exos effectively improved cardiac function in myocardial infarction model mice by activating the AKT signaling pathway 106 .
Research on the mechanism and optimization strategy of the use of the MSCs and their derivatives in skeletal muscle regeneration
The BM-MSCs and the multidimensional mechanism and optimization scheme of Exos regulating skeletal muscle regeneration
Muscle and bone tissue injuries with degenerative changes are common pathological conditions leading to patient disability, where endogenous regeneration is limited by injury severity and the inflammatory microenvironment. The BM-MSCs promote skeletal muscle regeneration through paracrine effects, cellular interactions, and microenvironment regulation. Kinnaird et al. 107 demonstrated significant enrichment of VEGF, basic fibroblast growth factor (bFGF), placental growth factor (PlGF), and monocyte chemotactic protein-1 (MCP-1) in the BM-MSCs-conditioned medium. Sassoli Chiara’s team further confirmed that key secreted factors such as VEGF and sphingosine 1-phosphate (S1P) synergistically activate the Notch-1/AKT signaling pathway in myoblasts, driving myoblast proliferation and myotubule formation 108 . In BALB/c mouse ischemic limb models, the BM-MSCs transplantation significantly increased the mid-thigh conduit vessel density and cross-sectional area, enhanced blood flow velocity and distal limb perfusion, reduced the muscle atrophy index and fibrotic area, and improved limb function scores 109 . Molecular validation revealed upregulated bFGF/VEGF expression at transplantation sites, confirming that paracrine mechanisms achieve functional remodeling through angiogenesis promotion and antifibrosis effects.
The Exos act as core signaling mediators for precise regulation. The miR-210-3p molecules they carry target and inhibit KLF7 gene expression, activating the PI3K/AKT pathway to increase myoblast satellite cell activity 110 . Moreover, miR-215 suppresses oxidative stress-induced myocyte apoptosis through the FABP3 axis 111 . However, the regenerative capacity of the BM-MSCs is limited by donor age. In contrast, the hDE-MSCs exhibit enhanced myogenic differentiation capability and improved in vivo implantation efficiency during early development because of their high serum response factor expression level 112 .
Engineering and combined therapies can effectively overcome therapeutic bottlenecks: Hypoxia pretreatment enhances Exos’ regenerative efficacy 110 , whereas coadministration with platelet-rich plasma synergistically activates mitochondrial biosynthesis and promotes myogenic marker expression 113 . The BM-MSCs and their Exos inhibit the ubiquitin‒proteasome system by activating the Sirt1/PGC-1α pathway, thereby reducing weight loss-induced skeletal muscle atrophy through the suppression of oxidative stress and inflammation 114 . Cross-tissue comparisons demonstrated that the BM-MSCs, with their strong osteogenic differentiation capacity, are suitable for bone‒muscle interface repair, whereas skeletal muscle‒derived stromal cells have greater advantages in tendon regeneration because of their high expression of tendon regulatory proteins 115 . In summary, the BM-MSCs provide a translational foundation for muscle degenerative diseases and trauma repair through three-tier networks: paracrine factors, Exos-miRNAs, and microenvironment reprogramming.
The ADSCs through the study of multimodal paracrine networks, immune metabolism, and engineering enhancement
The ADSCs demonstrate multimodal regulatory mechanisms and significant therapeutic potential in muscle regeneration. With respect to differentiation regulation, specific growth factors can induce the ADSCs to express smooth muscle/skeletal muscle markers and functional ion channels, enabling them to acquire contractile function 116 . IL-1β-activated macrophages drive ADSCs toward angiogenic smooth muscle cell transformation by secreting prostaglandin F2α 117 . In vitro myogenic differentiation models using human adipose-derived stem cells (hADSCs) confirmed that insulin-like growth factor 1 (IGF1) and fibroblast growth factor 2 (FGF2) synergistically activate myogenic differentiation signaling pathways, characterized by significantly upregulated expression of the key regulator MyoD1 (MyoD1↑) 118 . Their paracrine effects, particularly through secreted matrices and EVs, where miRNAs in EVs and proteins in soluble proteins each play unique roles, collectively regulate cell proliferation, differentiation, anti-inflammatory responses, and angiogenesis, thereby accelerating impaired muscle repair 119 . Mouse hindlimb muscle defect models demonstrate that Exos deliver bioactive molecules (e.g. miRNAs) to significantly promote myocyte proliferation and differentiation, which is marked by elevated expression levels of the key myogenic regulators MyoD1 and myoblastin (MyoD1, myoblastin↑), ultimately accelerating muscle tissue regeneration 120 . In the SD rat hindlimb ischemia model, adipose-derived stromal vascular components (AD-SVFs) and ADSCs were demonstrated to significantly promote angiogenesis and blood flow recovery in ischemic tissues by secreting key angiogenic factors such as VEGF 121 . The ADSCs also exhibit significant immunomodulatory properties, inducing macrophages to polarize into the repair-promoting M2 phenotype through the HIF-1α/IL-10/Stat3/ARG-1 axis while coordinating neutrophil and bone marrow progenitor cell infiltration to reshape the ischemic microenvironment and accelerate vascular maturation 122 . Clinical translational studies revealed that, compared with the BM-MSCs, ADSCs demonstrate superior efficacy in reducing collagen deposition, promoting myelination, and facilitating reinnervation 123 . Combined strategies involving hypoxic environments—such as the use of Se@SiO2 nanomaterials to enhance antioxidant and mitochondrial functions or the integration of exercise interventions to synergistically regulate AMPK pathways—can further optimize the therapeutic outcomes of the ADSCs 124 . Lee and Kemp 125 confirmed that a 2% hypoxic environment inhibits hADSC osteoblast and adipocyte differentiation but promotes their myogenic differentiation potential. By integrating multiple targets, including the inflammatory‒oxidative stress balance, angiogenesis, and autonomous cell differentiation, cross-model studies have demonstrated that the ADSCs effectively improve the structural and functional integrity of damaged muscle tissue.
The umbilical cord–derived mesenchymal stromal cells dominate skeletal muscle repair through a paracrine mechanism
The role of umbilical cord–derived mesenchymal stromal cells (UC-MSCs), including umbilical cord blood CD133⁺ hematopoietic stem cells and the Wharton’s Jelly–derived mesenchymal stem cells (WJ-MSCs) from Wollaston’s gel, in skeletal muscle regeneration is mainly dependent on paracrine effects rather than direct differentiation. Studies have shown that although the UC-MSCs can express some myogenic markers, their direct participation in muscle regeneration is low: Koponen et al. 126 found that transplanted UCB progenitor cells with VEGF-D were unable to achieve long-term colonization, and the gene modification did not significantly improve angiogenesis or muscle regeneration in the hindlimb ischemia model, indicating that the repair effect was mainly derived from paracrine mechanism. Similarly, Grabowska’s team demonstrated that the transplanted WJ-MSCs can be infused into new muscle fibers only at a low frequency in vivo but still significantly increase muscle mass posttransplantation, a mechanism likely related to the secretion of regenerative-promoting factors 127 .
Notably, pretreatment strategies can partially enhance therapeutic efficacy. For example, SDF-1 pretreatment did not alter the myofiber incorporation rate in the WJ-MSCs but significantly increased regenerative muscle mass 127 . Han et al. 128 further demonstrated that a 1% oxygen concentration activated antiapoptotic pathways and enhanced angiogenic factor secretion, thereby markedly improving capillary neovascularization in the UC-MSCs and significantly improving ischemic injury in mouse hindlimbs. These findings collectively indicate that the UC-MSCs primarily promote muscle repair through microenvironment regulation. The limited myogenic differentiation potential of these materials may be overcome by optimizing the combination of paracrine factors or integrating bioengineering strategies.
In summary, the BM-MSCs, ADSCs, and UC-MSCs primarily regulate inflammation, angiogenesis, and cellular activity through paracrine mechanisms (secreted factors and Exos) to promote muscle regeneration. Among these, the BM-MSCs excel in repairing the bone‒muscle interface, whereas the ADSCs demonstrate multimodal regulatory capabilities (immunomodulation and differentiation) and engineered enhancement potential. In contrast, the UC-MSCs rely mainly on paracrine mechanisms with relatively lower myogenic differentiation efficiency (Figure 8) (Table S4).
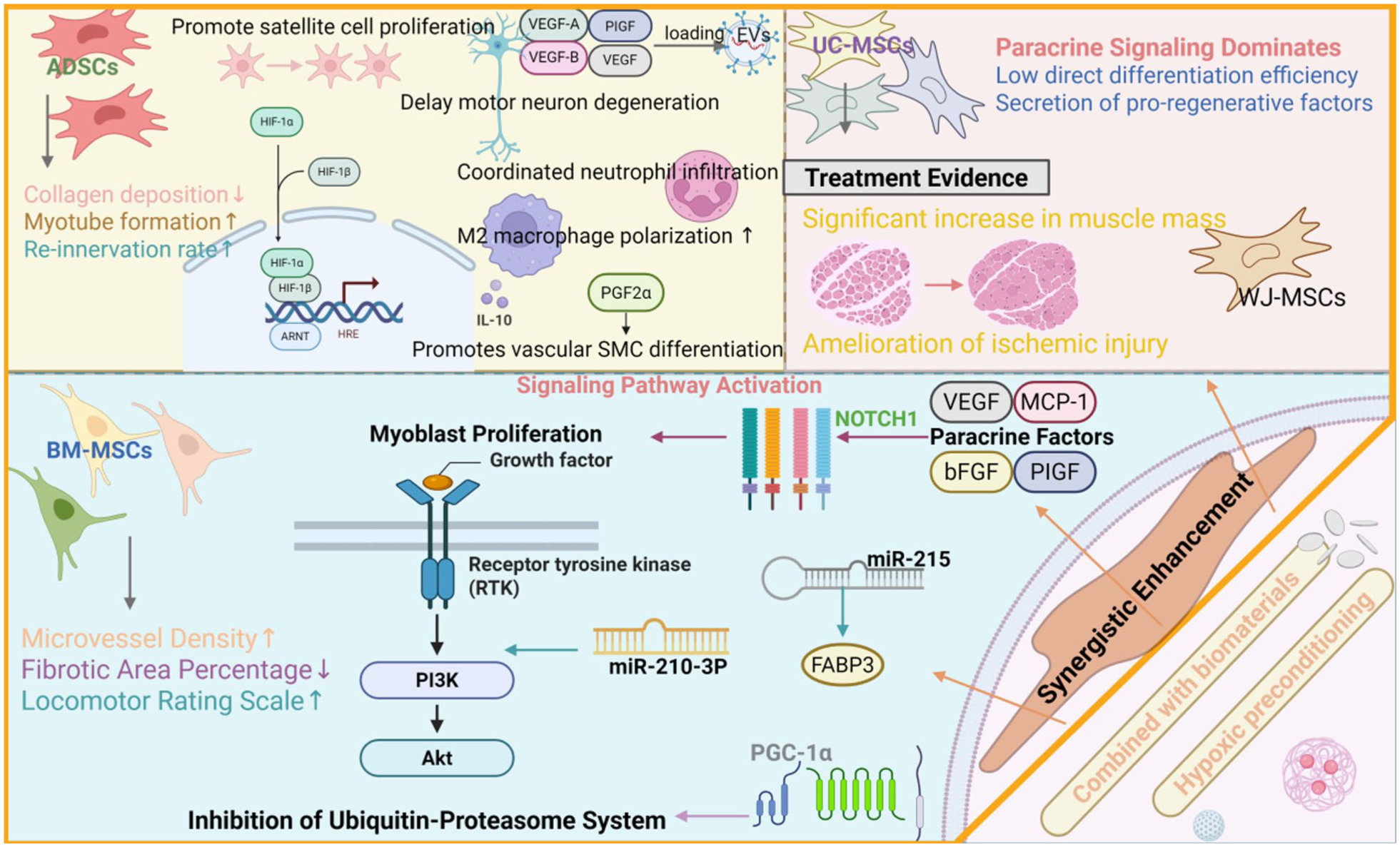
Mechanisms underlying the promotion of skeletal muscle regeneration by MSCs. This schematic outlines how MSCs promote muscle repair through integrated mechanisms. They stimulate satellite cells, modulate the immune response toward regeneration (e.g. promoting M2 macrophages), and secrete key factors (VEGFs, bFGF) that reduce fibrosis and enhance re-innervation. Central to this process is the activation of the PI3K/AKT pathway, which inhibits protein degradation. This potent paracrine action leads to measurable improvements in muscle mass, vascularization, and functional recovery. Created with BioRender.com.
Neural repair of the MSCs and derivatives: From multitarget mechanisms to clinical transformation
Neural repair potential: Multilineage characteristics, Exo-mediated mechanisms, and advantages of clinical transformation
The developmental origins of the MSCs involve the mesoderm and cranial neural crest 29 . These cells express multiple lineage markers, including α-smooth muscle actin (α-SMA) and smooth muscle myosin heavy chain from the mesenchymal lineage; neuro-associated markers, such as Nestin and Tuj-1; and endothelial molecules, such as CD146 and CD105, along with key signaling receptors, including transforming growth factor-beta receptor (TGF-β receptor) and integrins. This marker profile suggests their potential to participate in tissue repair processes through integrin-mediated extracellular matrix adhesion and paracrine signals (e.g. the TGF-β pathway). The MSCs possess multidirectional differentiation capabilities, not only differentiating into mesodermal cells (such as bone, adipose, and cartilage cells) but also demonstrating potential for endodermal and neuroectodermal differentiation 129 . In nervous system repair, the MSCs and their derivatives function primarily through paracrine mechanisms, particularly by secreting EVs 130 containing bioactive molecules such as proteins, miRNAs, and mRNAs. These vesicles enable cellular replacement to repair damaged tissues, reshape regenerative microenvironments, and protect existing neurons/glioblasts. These vesicles, with their specific membrane structures, can be precisely targeted and fused with cells. By delivering critical cargo molecules such as amyloid-β (Aβ), tau proteins (Tau), and miRNAs, they exert bidirectional regulatory effects on brain diseases 131 , modulating neuronal functions and behaviors while suppressing pro-inflammatory cytokine release, reducing neuroinflammation, promoting the NSCs proliferation and differentiation, and enhancing the nervous system’s self-repair capacity—providing new avenues for neurological disease diagnosis and treatment 130 . Although the NSCs are considered ideal candidates for central nervous system repair because of their neural differentiation potential, their clinical applications remain limited. In contrast, the MSCs, the more commonly used clinical counterparts, primarily exert therapeutic effects through secretome-mediated immune regulation and nutritional support. Strategies such as promoting bidirectional interactions between the MSCs and NSCs, inducing the MSCs (particularly neural crest-derived subpopulations) to differentiate into neurons, and coculturing with biological scaffolds or reconstructing neural ecological niches can further increase the potential for MSCs-mediated neurorepair 132 .
Mediated neurotrophic multitarget repair: Myelin, neurons, glial cells, and neurons
Myelination repair mechanism
The MSCs and their derivatives demonstrate remarkable repair potential in various demyelinating disease models. In a neonatal rat white matter injury (WMI) model, the hUC-MSCs effectively repaired white matter structure by inhibiting the microglia-mediated NF-κB signaling pathway, promoting oligodendrocyte maturation and neuronal survival 133 . Studies in copper-induced demyelination models indicate that the MSCs transplantation reduces A1 astrocyte marker expression, increases oligodendrocyte numbers, and enhances the expression of neurotrophic factors such as BDNF and transforming growth factor-beta (TGF-β) while decreasing the expression of pro-inflammatory cytokines such as IL-1 and TNF-α, thereby synergistically enhancing myelin regeneration 134 . For neuromyelitis optica spectrum disorders (NMOSDs), the hUC-MSCs transplantation inhibits spinal cord inflammation, protects myelin integrity, and maintains blood‒brain barrier function by downregulating matrix metalloproteinase-9 activity, improving motor dysfunction 135 . In multiple sclerosis treatment, a combined strategy of ablating pathological astrocytes and administering MSCs-secreted factors can reshape the microenvironment, targeting oligodendrocyte-mediated myelin regeneration 136 .
Hippocampal function and cognitive behavior improvement
The MSCs play multidimensional roles in improving hippocampal-related cognitive impairments. In a streptozotocin-induced sporadic Alzheimer’s disease (sAD) rat model, long-term intravenous infusion of the hUC-MSCs effectively reversed multiple cognitive and behavioral deficits through neuroprotective effects, reduced neuroinflammation, and partially restored synaptic/structural integrity 137 . Intraventricular injection of the bone marrow mesenchymal stem cells exosomes (BMSC-Exos) significantly improved behavioral disorders and neuropathological damage in STZ-AD mice. The mechanism involves inhibiting the activation of hippocampal microglia and astrocytes, reducing pro-inflammatory factor levels, decreasing Aβ deposition and BACE/phosphorylated Tau expression, and upregulating the expression of synaptic-associated proteins (synaptograftin-1 and synaptophysin-1) and BDNF. This synergistic regulation activates the “neuroinflammation suppression‒AD pathologic remission‒synaptic repair” axis 138 . Engineering strategies such as EVs delivering complement inhibitors (EVs-T-ALZ01) or coloaded with berberine/palmine (EVs-Ber/Pal) have also improved cognition in AD models. In depression treatment, the MSCs drive microglia to adopt the Arg1⁺ anti-inflammatory phenotype by secreting TGF-β1, demonstrating superior antidepressant efficacy to that of fluoxetine in chronic stress models 139 . Moreover, the BMSC-Exos enhance hippocampal superoxide dismutase expression and reduce MDA/lactate dehydrogenase (LDH) levels through miR-26a delivery while suppressing inflammatory factors and inhibiting neuronal apoptosis 140 .
Glial cell phenotype regulation
The MSCs and their derivatives exert neuroprotective effects by precisely regulating glial cell phenotypes. In microglia, the MSC-EVs can be internalized to deliver miR-467f/miR-466q, which targets the Map3k8/Mk2 gene to block the p38 MAPK pathway. This significantly reduces the production of pro-inflammatory factors such as TNF-α and IL-1β while inhibiting hyperactivation 141 . Light-bioregulated pretreated hUC-MSCs promote anti-inflammatory M2 polarization by downregulating Notch signaling pathways, thereby improving cognitive function in models 142 . Oligoglycoxin (OGD)-pretreated microglia-derived EVs deliver TGF-β1 to activate the Smad2/3 pathway, synergistically promoting angiogenesis and resistance to neuronal apoptosis. They also induce resident microglia to polarize into M2 cells to regulate inflammatory responses, significantly enhancing neurological recovery in stroke models 143 . With respect to astrocyte regulation, hypoxia-preconditioned vesicles (H-EVs) deliver miR-21 to inhibit the JAK2/STAT3 pathway, driving the A1 inflammatory phenotype to the A2 protective phenotype, thus facilitating motor function recovery after spinal cord injury 144 . In addition, the MSC-EVs overexpressing miR-138-5p impart neuroprotective effects on astrocytes via the inhibition of lipocalcin 2 (LCN2) 145 . Notably, the hUC-MSCs can specifically inhibit the upregulation of interferon-γ (IFNγ)-induced NSCs and astrocyte major histocompatibility complex class Ⅱ molecules, providing a novel strategy for reducing allograft immune rejection 146 . Engineered extracellular vesicile-lipid nanoparticle hybrids achieve dual-pathway inhibition of Aβ generation and induce anti-inflammatory transformation of microglia by co-delivering siRNA targeting BACE1 with angiopep-2-modified TREM2 plasmids 147 .
Neuronal protection and regeneration
The MSC technology exerts therapeutic effects by directly promoting neuronal protection and regeneration. In terms of neuroprotective mechanisms, the hUC-MSCs can reduce central nervous system inflammation, preserve blood‒brain barrier integrity, and counteract complement-mediated astrocytic apoptosis 148 . The human amniotic fluid stem cell (hAFSC-Exos) inhibits lipopolysaccharide-activated microglial inflammatory responses while alleviating neuronal oxidative stress and apoptosis in Aβ-induced coculture models with neurons and microglia 136 . Targeted delivery strategies such as Fe65 protein-modified EVs encapsulating the autophagy-inducing agent Crey-B (EV-Crey-B) specifically bind to amyloid precursor protein (APP) and induce autophagy clearance, thereby improving cognitive function in AD 149 . With respect to cell differentiation, human endometrial polyp–derived mesenchymal stem cells (EP-MSCs) can differentiate into glial-like cells and form neural networks under specific conditions 150 . The WJ-MSCs cross the blood‒brain barrier, differentiate into β-III tubulin-positive neurons and astrocytes during focal cerebral ischemic injury, and are accompanied by neurological recovery 28 .
Clinical transformation path: Treatment challenges and precise control strategies for neurological diseases
In addition to animal studies, research has further revealed the therapeutic potential of the MSCs and EVs in neurological disorders. The MSCs deliver neurotrophic compounds via secreted EVs, while the MSC-EVs can penetrate biological barriers to target central nervous system injury sites, demonstrating promise for treating neurodegenerative diseases 151 . In addition, both the MSCs and their released Exos promote neural repair and alleviate neuropathic pain through mechanisms including immune regulation, anti-inflammatory effects, and axonal/myelination regeneration130,152. Preliminary clinical trials have evaluated the safety and feasibility of EVs in neurological conditions. In an acute ischemic stroke trial, five male patients with malignant middle cerebral artery occlusion treated with craniectomy received intracerebral injections of allogeneic placental MSC-derived EVs within 48 h of symptom onset, with no reported adverse events. In a mild-to-moderate AD trial (NCT04388982), phase I/Ⅱ clinical data revealed good tolerability and safety of intranasal administration of the allogeneic human adipose-derived MSC-Exos (hADMSC-Exos) in nine patients. The study established a minimum effective dose of 4 × 108 particles for subsequent trials and suggested comparable therapeutic potential to existing AD medications such as GV-971 or donepezil. Together, these findings demonstrate that the MSC-EVs-based cell-free therapies have promising clinical applications in the field of neurological diseases, and their preliminary safety and feasibility data lay the foundation for deeper clinical translation.
In summary, the MSCs and their Exos, among other EVs, as “cell-free” therapies, deliver bioactive substances to regulate neuroinflammation at multiple targets, promote myelin regeneration, protect neurons, and improve cognitive function. These therapies have unique advantages in treating various neurological diseases, such as stroke and Alzheimer’s disease (Figure 9, Table S5). They avoid the risks associated with cell transplantation, have shown safety and efficacy in preliminary clinical trials, and represent a breakthrough in the field of neural repair. Future efforts should focus on engineering optimization and large-scale clinical validation to accelerate their application.

Mechanisms of remyelination and neuroprotection mediated by MSCs. This diagram illustrates the multi-target strategy of MSCs for promoting neural repair. By inhibiting key inflammatory pathways (e.g. NF-κB, JAK2/STAT3), MSCs reduce neuroinflammation, promote an anti-inflammatory glial phenotype, and help preserve blood–brain barrier integrity, thereby fostering remyelination and synaptic repair. In addition, certain MSCs (e.g. WJ-MSCs) can directly differentiate into neural cells to support structural regeneration. Through these combined actions, MSCs enhance neuroprotection and facilitate functional recovery. Created with BioRender.com.
Comparative analysis of therapeutic paradigms: MSCs versus conventional TH replacement therapy
Traditional therapies, through exogenous hormone supplementation, can effectively correct the systemic metabolic status and alleviate symptoms directly caused by hormone deficiency 7 . Their core advantages lie in clear mechanisms, simple application, and low cost, enabling effective correction of serum TH levels, relief of most classic clinical symptoms, and prevention of severe hypothyroidism complications 5 . However, their mechanism is essentially “substitution” rather than “repair,” which can be summarized as “metabolic targets achieved without tissue recovery.” This leads to two inherent limitations: First, the improvement of residual symptoms in some patients (especially those with transport or conversion disorders) is limited. Second—and more crucially—it is unable to reverse organic damage to multiple systems such as cardiovascular, neurological, and muscular tissues that has already occurred during the progression of hypothyroidism. This means that even with “metabolic targets achieved,” patients’ long-term prognosis and quality of life may still be significantly compromised due to irreversible tissue damage 12 .
In contrast, the core theoretical advantages and potential clinical value of MSCs and their derivatives therapy lie in its potential to break through these bottlenecks of traditional therapies. Rather than directly supplementing hormones, it functions as an “active repair biotherapy” through homing, immunomodulation, and potent paracrine effects 99 . On one hand, it may improve the local microenvironment of the thyroid. On the other hand, it can directly target distal organs affected by hypothyroidism, such as promoting myocardial cell survival and angiogenesis to reduce fibrosis 90 , supporting neuronal function and myelin repair 130 , and improving muscle metabolism and function 119 . Thus, beyond “correcting targets,” it achieves active intervention in “multi-system damage.”
Therefore, the MSCs and their derivatives therapy should not be viewed as a simple replacement for existing treatments, but rather as a strategic complement in the future. Its precise positioning may lie in providing a new pathway for patients with refractory multisystem complications who remain unresponsive to adequate hormone replacement therapy, transitioning from “biochemical remission” to “tissue repair” and “functional recovery”35,36. This contrast not only clarifies the fundamental differences and complementary relationship between the two therapies, but also defines the key direction for future translational research—how to translate the systemic repair potential of MSCs and their derivatives into verifiable clinical benefits.
In conclusion, the MSCs and their derivatives therapy present a novel paradigm in the treatment of hypothyroidism and its complications. It differs from traditional hormone replacement therapy by focusing on tissue repair.
Challenges and prospects of the use of the MSCs and their derivatives in the treatment of hypothyroidism and related pathological changes
The high heterogeneity of the MSCs and their derivatives poses a core challenge to their standardized application. Factors such as cell source (bone marrow, adipose tissue, umbilical cord, etc.), donor variability, culture passage, and expansion conditions significantly influence their phenotypic characteristics, functional properties, and in vivo behavior. These variations make it difficult to directly compare and replicate results across studies, thereby undermining the generalizability of conclusions and the standardization of therapeutic protocols 153 . Furthermore, the lack of a unified protocol for exosome isolation, purification, and characterization exacerbates inter-batch quality inconsistencies 154 .
Despite the abundance of preclinical data, the mechanisms underlying MSCs and their derivatives remain largely uncharted territory. Most studies have only identified efficacy correlations, lacking spatiotemporal analyses of their survival rates, terminal differentiation, and the specific signaling networks and target points of their paracrine components within the complex pathological microenvironment of hypothyroidism 155 . The maintenance of their functional pluripotency in vivo remains controversial, partly due to the absence of reliable cell tracking technologies. Furthermore, their biological distribution and long-term safety as delivery vectors are unclear. Intravenous administration is rapidly cleared by the mononuclear phagocyte system, resulting in low bioavailability 156 , while local injection may prolong retention but could induce localized inflammatory responses 154 .
The transition from animal models to clinical applications faces significant challenges. Currently, preclinical studies predominantly utilize acute or mild hypothyroidism models, which differ substantially from the chronic, multisystem involvement observed in the progression of human diseases. Early clinical trials frequently encounter issues such as limited sample sizes, short follow-up periods, the absence of control groups, and single assessment metrics. In particular, they lack long-term data with multi-organ function recovery as a hard endpoint. Under specific hypothyroidism pathologies, several critical questions remain unanswered: Can MSCs and their derivatives maintain functionality in hypometabolic microenvironments with potential autoimmune attacks? Do they exhibit differential efficacy or prioritization in repairing the thyroid and distal organs? And can their therapeutic benefits and safety exceed or complement existing simplified hormone replacement therapies?
In conclusion, advancing MSCs and their derivatives therapy to clinical application requires systematic solutions to three critical challenges: controlling cellular and product heterogeneity, elucidating the mechanistic depth, and establishing clinical relevance in translational research.
Therefore, to overcome the aforementioned bottlenecks, such as insufficient bioavailability and targeting, optimizing delivery strategies remains a key research direction for improving therapeutic efficacy. Experimental evidence shows that encapsulating MSC exosomes with sodium alginate hydrogel can significantly delay release kinetics, thereby increasing central myocardial targeting efficiency by 100% in rat models 156 .
Engineering improvements have further expanded the application prospects of these methods. The Exos engineered to display ligands for the TH transporter MCT8 enhance blood–brain barrier permeability, successfully restoring cerebellar myelin protein expression in AHDS model mice 157 . High-throughput proteomic analysis revealed that compared to untreated controls, IFN-γ pretreated MSC exosomes accumulated PD-L1 and TGF-β, resulting in a 100% increase in immunomodulatory activity 156 . In addition, the design and synthesis of functional bio-nanomaterials hold great potential in stem cell-based therapies. Their unique properties provide a new technical platform for the precise regulation of stem cell behavior, including proliferation, differentiation, and delivery 154 .
Conclusion and limitations
Although LT4 replacement therapy can restore the serum indicators of most hypothyroid patients to normal levels, irreversible systemic damage, including neuronal apoptosis, mitochondrial energy depletion, and vascular endothelial degeneration, and notably, skeletal muscle damage characterized by myopathy, fiber atrophy, and impaired regenerative capacity, still results in persistent symptoms such as cognitive impairment, metabolic disorders, fatigue, and depression. This not only significantly increases the risk of cardiovascular disease, neurodegenerative diseases, and autoimmune comorbidities but also leads to a prolonged decline in quality of life and treatment resistance. To address this challenge, the MSCs and their derivatives have shown transformative potential. Preclinical evidence supports their ability to promote neuroprotection and synaptic regeneration, enhance vascular repair and cardiac mitochondrial homeostasis, and stimulate skeletal muscle regeneration and satellite cell activation. However, to transition from animal studies to clinical applications, three major obstacles must be overcome: controlling cell product heterogeneity, addressing species-specific pathophysiological differences, and managing long-term biosafety risks. Therefore, global researchers need to collaborate on innovations in targeted delivery systems, functionalized EVs, and multicenter clinical trials. Ultimately, this will provide new treatment models for refractory diseases, offering patients new treatment strategies and options, thereby improving their quality of life and clinical outcomes.
This review has certain limitations. First, as a narrative rather than a systematic review, it does not employ formally predefined search strategies or explicit eligibility criteria, which may introduce selection bias and limit the comprehensive retrieval of all relevant literature. Second, the significant heterogeneity among the included studies, such as variations in MSCs and their derivatives sources (e.g. bone marrow, adipose tissue), preparation protocols, animal models of hypothyroidism, and outcome measurement methods, makes direct comparison and quantitative synthesis challenging, and may affect the generalizability of the summarized conclusions. Third, the mechanistic discussions and therapeutic prospects are primarily grounded in preclinical (animal) evidence; while clinical data are emerging, they remain limited in scale and duration, necessitating cautious interpretation regarding direct human application. Finally, potential publication bias toward positive results may overestimate the therapeutic efficacy of MSC-based interventions. Despite these limitations, this review systematically examines the pathological impacts of hypothyroidism across multiple organ systems, with particular emphasis on the comprehensive and critical evaluation of the mechanisms and therapeutic potential of MSCs and their derivatives in this condition. By elucidating key signaling pathways, the study identifies promising translational research directions, providing a structured theoretical framework for subsequent pre-clinical optimization and clinical trial design. Our research team has already initiated relevant studies in this field.

Supplemental Material
sj-docx-1-cll-10.1177_09636897261418754 – Supplemental material for Mechanisms and clinical prospects of mesenchymal stem cells and their derivatives in treating hypothyroidism-induced injuries: A focus on cardiovascular, skeletal muscle, and nervous systems
Supplemental material, sj-docx-1-cll-10.1177_09636897261418754 for Mechanisms and clinical prospects of mesenchymal stem cells and their derivatives in treating hypothyroidism-induced injuries: A focus on cardiovascular, skeletal muscle, and nervous systems by Yue Wang, Jiayu Zhu, Zhonghui Li, Fang Yu, Yuanyuan Fu, Han Gao, Meng Wang, Yuan Liu, Luming Zheng and Qingqing He in Cell Transplantation
Footnotes
Acknowledgements
Not applicable.
Ethical Statement
Not applicable.
Consent Statement
Not applicable.
Author Contributions
Yue Wang: Writing—original draft, Visualization, Formal analysis, Conceptualization; Jiayu Zhu: Writing—original draft, Methodology, Data curation; Zhonghui Li: Writing—original draft, Investigation, Validation; Fang Yu: Writing—original draft, Resources, Data curation; Yuanyuan Fu: Writing—review & editing, Supervision, Project administration; Han Gao: Writing—review & editing, Methodology, Formal analysis; Meng Wang: Writing—review & editing, Visualization, Software; Yuan Liu: Writing—review & editing, Supervision, Funding acquisition; Luming Zheng: Writing—review & editing, Resources, Validation; Qingqing He: Writing—review & editing, Supervision, Conceptualization, Funding acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Key Medical Discipline Project of Joint Logistics Support Force of the People’s Liberation Army of China; Key Discipline Project of Shandong Province Medical and Health; Shangdong Provincial Natural Science Foundation General Project (ZR2021MH328); Shandong Province Medical and Health Technology Development Plan (202204011069); 2023 Ji’nan Science and Technology Innovation Development Program (202317016); 2024 Ji’nan Clinical Medicine Science and Technology Innovation Program(202430064).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
AI Use
We confirm that no AI tools were used for language refinement or enhancement in the preparation of this manuscript. Furthermore, no scientific data in this manuscript has been generated or modified using AI. Our work fully complies with Sage’s policy on the use of AI tools.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
