Abstract
This study aimed to identify the beneficial effects of adipose tissue-derived mesenchymal stem cells (ASCs) and ASCs that overexpress the CTLA4Ig gene (CTLA4Ig-ASCs) on established autoimmune thyroiditis and to examine changes in clinical chemistry parameters and the presence of humoral responses upon repeated long-term administration of autologous ASCs. This study also aimed to acquire desirable results in a preclinical study by using large-sized lab animals and applying ASCs that overexpress therapeutic genes. Experimental autoimmune thyroiditis was induced by immunization with thyroglobulin. Experimental dogs were divided into five groups: (i) ASC IT + IV, (ii) ASC IV, (iii) CTLA4Ig-ASC IT + IV, (iv) CTLA4Ig-ASC IV, and (v) control IT + IV (saline only), and they received intrathyroidal (IT; 10 million cells/250 μl saline per thyroid) administration one time or intravenous (IV; 20 million cells/5 ml) administration seven times within a 101-day period. Blood samples were collected every week, and thyroids were harvested on days 104-106. After serial ASC or CTLA4Ig transplantation, the levels of canine thyroglobulin autoantibodies (TgAA) in serum and the infiltration of T-lymphocytes between the follicles of the thyroid glands were decreased. The expression of FoxP3 in submandibular lymph nodes was significantly increased. Repeated long-term administration of autologous ASCs or CTLA4Ig-ASCs did not generate changes in clinical chemistry parameters or humoral responses. The TgAA test can detect autoimmune thyroiditis years before clinical signs of hypothyroidism occur. Thus, ASC and CTLA4Ig-ASC transplantation in that period can be attractive candidates to ameliorate autoimmune thyroiditis and prevent the development of hypothyroidism.
Keywords
Introduction
CTLA4 (cytotoxic T-lymphocyte antigen 4), also known as cluster of differentiation 152 (CD152), is similar to the T-cell costimulatory protein CD28 and acts as a negative regulator of T-cell function. Many studies have shown that CTLA4 has a critical role in maintaining peripheral tolerance, immunosuppression, and autoimmunity (38). In 1995, for example, a different research group demonstrated that mice lacking the Ctla4 gene developed lethal autoimmune lymphoproliferative disease with massive T-lymphocyte infiltration and multiorgan tissue destruction (40,43). It has also been reported that CTLA4 polymorphisms are associated with autoimmune disorders in humans, including autoimmune thyroiditis (9,18).
Owing to the comparatively higher binding affinity of CTLA4, the CTLA4Ig fusion protein can block the B7:CD28 costimulatory pathway, which results in the prevention of T-cell proliferation, induction of T-cell clonal anergy, and amelioration of autoimmune disease (32). Hence, there has been great interest in the therapeutic potential of CTLA4Ig for autoimmune disorders.
Previously, we induced an experimental autoimmune thyroiditis (EAT) in mice (12). In this study, CTLA4Ig gene-transduced human adipose tissue-derived mesenchymal stem cells (hASCs) were injected intravenously prior to the onset of disease, and a preventive effect of ASCs on EAT was found.
Although murine EAT mimics some immunologic features of Hashimoto's thyroiditis (40), the symptoms of EAT spontaneously regress over several weeks without further thyroid follicular disruption. Thus, long-term therapeutic study is difficult to achieve using the murine EAT model. The small size of the thyroid is another restriction in the murine EAT model. Even under a surgical microscope, some leakage of injected substance has been observed. Therefore, we have attempted to address these drawbacks by using a large animal EAT model.
In terms of genetic or environmental factors, dogs have advantages in replicating some aspects of human disease (10,34). As in humans, hypothyroidism is the most common endocrine disorder in dogs, and up to 80% of cases result from autoimmune (lymphocytic) thyroiditis (6,19). Thus, we investigated this by inducing experimental autoimmune thyroiditis in dogs.
Mesenchymal stem cells (MSCs) are well known to have multipotency, immune privilege, and robust trophic, anti-inflammatory, and regenerative properties (4,8,31,37). Thus, many trials using MSCs have been applied to various autoimmune and immune-mediated diseases. MSCs can be isolated from various tissues (24), and adipose tissue has many advantages as it is easily acquired, easily expanded, and causes minimal patient discomfort (33,47). Moreover, the ethical issues associated with ASCs are limited, making them an ideal source for use in practical regenerative medicine.
In this study, we followed up the long-term therapeutic effect of canine CTLA4 gene-transduced autologous ASCs by monitoring the thyroglobulin autoantibody (TgAA) concentration and thyroid histopathology. Furthermore, we investigated whether repeated injections of ASCs or CTLA4Ig-ASCs (every 2 weeks, a total of seven times) affected organ function by serum chemistry examination over 3 months. In addition, we investigated the change in the proportion of regulatory T cells (Tregs) in peripheral blood mononuclear cells (PBMCs) and FoxP3 expression in the submandibular lymph nodes.
Materials and Methods
Experimental Animals
Fifteen 1-year-old male beagle dogs (Gaoyao Kangda Laboratory Animals and Technology Co. Ltd., Gaoyao City, Guangdong, China) determined to be healthy after routine physical examination, complete blood counts, serum biochemistry analysis, and urinalysis were used. This study was reviewed and approved by the Institutional Animal Care and Use Committee of the Samsung Biomedical Research Institute (SBRI). SBRI is an Association for Assessment and Accreditation of Laboratory Animal Care International-accredited facility and abides by the Institute of Laboratory Animal Resources guide.
All efforts were made to minimize animal suffering. The dogs were housed separately in metal cages and maintained on commercial dry dog food and water ad libitum. The cages were kept at a constant temperature (22-25°C) and humidity (50-55%). The animals were acclimatized at least 2 weeks prior to use.
Preparation of Canine ASCs
Adipose tissue was aseptically obtained from the inguinal subcutaneous fat of each dog under isoflurane (Hana Pharm Co. Ltd., Seoul, Korea) anesthesia at least 3 weeks before immunization with thyroglobulin. Dog ASCs were prepared as described in our previous studies (12) except for the extension media (RCME-P; K-Stemcell, Seoul, Korea). Autologous ASCs at passage 4 were used for transplantation.
Immunophenotype of Dog ASCs
Dog ASCs were immunophenotyped by flow cytometric analysis at culture passage 4. Fluorescein isothiocyanate (FITC)-conjugated hamster anti-mouse CD29 [0.5 μg/test (5 × 105 to 106 cells, a final volume 20 μl), clone: Ha2/5, 555005; BD Biosciences, San Jose, CA, USA], phycoerythrin (PE)-conjugated mouse anti-canine CD34 (0.05 μg/test, clone: 1H6, 12-0340; eBioscience, San Diego, CA, USA), FITC-conjugated rat anti-canine CD44 (0.025 μg/test, clone: YKIX337.8, 11-5440; eBioscience), FITC-conjugated rat anti-canine CD45 (0.05 μg/test, clone: YKIX716.13, 11-5450; eBioscience), PE-conjugated rat anti-canine CD90 (0.1 μg/test, clone: YKIX337.217, 12-5900; eBioscience), and FITC-conjugated rat anti-canine MHC class II (0.1 μg/test, clone: YKIX334.2, 11-5909; eBioscience) were used for cell surface antigen detection. ASC immunophenotypes were analyzed using a FACSAria flow cytometer (BD Bioscience) using BD FACSDiva software. Isotype control antibodies [1 μl/each test, 553960 (isotype control 1: CD29), 555749 (isotype control 2: CD34), 554688 (isotype control 3: CD44 and MHC class II), 556923 (isotype control 4: CD45) (all BD Biosciences), and 12-4031 (isotype control 5: CD90) (eBioscience)] were used to estimate the nonspecific binding of target primary antibodies to cell surface antigens. The procedure was performed following the manufacturer's instructions.
Construction of the Therapeutic Gene
The construction of the therapeutic canine CTLA4Ig gene was as described in our previous study (14). Briefly, we constructed a therapeutic gene composed of the functional extracellular domain of canine CTLA4 to block the B7:CD28 costimulatory pathway and the hinge-CH2-CH3 domains of the canine immunoglobulin α1 constant (IGHAC) region to extend the half-life of the therapeutic protein in vivo. For secretion into the body fluid, the human oncostatin M signal sequence was ligated to the therapeutic gene. The fusion sequence consisting of oncostatin M, the CTLA4 extracellular domain, and the hinge-CH2-CH3 domains of the IGHAC region was ligated to the HindIII and XbaI sites in pcDNA3.1+ (Invitrogen, Carlsbad, CA, USA). Primer pairs were prepared: C1-F 5′-CACCATGGGTGTACTGCTCA-3′ and C2-R 5′-TTAGTAGCAGATGCCGTCCA-3′. With these primers, approximately 1,195 bp fragments were amplified using pcDNA 3.1+/CTLA4Ig as the template by Pfu polymerase (TaKaRa, Otsu, Japan). This product was cloned into the pLenti6/V5-D-TOPO® vector (Invitrogen) according to the manufacturer's instructions.
Canine CTLA4Ig Gene Transduction Into Canine ASCs
The canine CTLA4Ig gene was transduced into canine ASCs using ViraPower™ Lentiviral Expression Systems (Invitrogen) according to the manufacturer's instructions.
Briefly, the cloning of the canine CTLA4Ig gene into the pLenti6/V5-D-TOPO® vector (Invitrogen) was confirmed by restriction enzyme digestion and DNA sequencing. The ViraPower packaging mix and canine CTLA4Ig-pLenti6/V5 TOPO expression plasmid were cotransfected into the 293FT cell line (Invitrogen) using Lipofectamine 2000 (Invitrogen). Virus particles were collected and transfected into canine ASCs. Five days after selection with blasticidin (5 μg/ml; Invitrogen), total RNA was isolated using Easyblue (iNtron Biotechnology, Sungnam, Korea) from nontransduced ASCs (negative control) and canine CTLA4Ig gene-transduced ASCs (CTLA4Ig-ASCs) to determine the transcription of the canine CTLA4Ig gene by reverse transcriptase polymerase chain reaction (RT-PCR).
Detection of the Transcription of the Canine CTLA4 Gene by RT-PCR
Detection of the transcription of the canine CTLA4 gene by RT-PCR was conducted as in the previous study (11,14). In vivo, to detect transcription of the therapeutic gene from CTLA4Ig-ASCs in the thyroid, a pair of primers amplifying the fusion portion of CTLA4Ig, which does not exist in normal dogs, was used for RT-PCR analysis (5′-ACAAAGTGAACCTCACCATC-3′ and 5′-ACACTGGACACACTGTAGCA-3′). Canine glyceraldehyde-3-phosphate dehydrogenase was used as housekeeping gene (5′-GGTCACCAGGGCTGCTTT-3′ and 5′-ATTTGATGTTGGCGGGAT-3′).
ELISA to Determine the Concentration of Dog IgA in Stem Cell Culture Supernatant
Dog IgA concentrations in stem cell culture supernatants were assessed by enzyme-linked immunosorbent assay (ELISA) using a Dog IgA ELISA kit (assay range: 31.25-1,000 ng/ml, KT 513; Kamiya Biomedical Company, Osaka, Japan) according to the manufacturer's instructions.
Induction of Autoimmune Thyroiditis
Experimental groups were divided into control (n=3), ASC IT + IV (n = 3), ASC IV (n = 3), CTLA4Ig-ASC IT + IV (n = 3), and CTLA4Ig-ASC IV (n = 3) groups. We induced experimental autoimmune thyroiditis by porcine thyroglobulin (Tg; Sigma-Aldrich, St. Louis, MO, USA). For induction, 30 mg of porcine Tg in deionized water (0.5 ml) was emulsified in Freund's complete adjuvant (1:1, v/v; Sigma-Aldrich), and 1 ml was given subcutaneously (SC) to each dog. The boosting injection on day 13 was given SC using porcine Tg in incomplete Freund's adjuvant (Sigma-Aldrich). On day 13 postimmunization, all the experimental dogs showed positive results in the canine TgAA assay and confirmed the development of autoimmune thyroiditis. On day 16 or 17, the control, ASC IT + IV, and CTLA4Ig-ASC IT + IV groups were treated with 250 μl of saline, 107 ASC/250 μl of saline, and 107 CTLA4Ig-ASC/250 μl of saline per thyroid, respectively. Intrathyroidal injections were conducted under isoflurane anesthesia (Fig. 1). On days 16 (or 17), 32, 46, 60, 73, 87, and 101, 2 × 107 autologous ASCs/5 ml of saline (the ASC IT+IV group and ASC IV group; n=6) or the same amount of canine CTLA4Ig gene-transduced autologous ASCs/5 ml of saline (the CTLA4Ig-ASC IT + IV group and the CTLA4Ig-ASC IV group; n = 6) were administered intravenously. Control dogs were treated identically, but were infused with 5 ml of saline instead of cells (n = 3).

Immunophenotyping of canine CTLA4Ig-ASCs. Dog ASCs were immunophenotyped by flow cytometric analysis at culture passage 4. FITC-conjugated hamster anti-mouse CD29, PE-conjugated mouse anti-canine CD34, FITC-conjugated rat anti-canine CD44, FITC-conjugated rat anti-canine CD45, PE-conjugated rat anti-canine CD90, and FITC-conjugated rat anti-canine MHC class II were used for cell surface antigen detection. Isotype control antibodies were used to estimate the nonspecific binding of target primary antibodies to cell surface antigens. (A) Nontransduced canine ASCs and (B) CTLA4Ig-ASCs.
Detection of Canine TgAA
During the experiment, serum samples were collected once per week and kept frozen at −70°C until analysis of canine TgAA using a commercially available ELISA kit (VT12; Oxford Biomedical Research, Rochester Hills, MI, USA) according to the manufacturer's instructions. The sample was considered to be positive at values >25% for the TgAA assay. Values <10% were counted as negative.
Serum Biochemistry
Serum concentrations of alanine aminotransferase (ALT), aspartate aminotransferase (AST), blood urea nitrogen (BUN), creatinine, total protein, albumin, and total bilirubin were measured by a Selectra IV autoanalyzer (Merck, Dieren, the Netherlands).
Canine TSH and Canine-Free T4
Serum samples were collected from the experimental groups on days 27 (peak TgAA period) and 104 (last blood collection). Serum-free T4 by equilibrium dialysis and TSH were determined by referring to the ANTEC laboratory (Metairie, LA, USA).
Suppression Test of the Supernatant From Canine ASCs or Canine CTLA4Ig-ASCs on the Proliferation of PBMCs From Experimental Autoimmune Thyroiditis (EAT) Dogs
On day 63, PBMCs were harvested from EAT control dogs and supplemented with sodium heparin (BD, Franklin Lakes, NY, USA) as an anticoagulant. Ficoll-Paque (Pharmacia Biotech, Uppsala, Sweden) gradient centrifugation was performed at 400 x g for 30 min. The PBMCs were washed twice with phosphate-buffered saline without calcium chloride and magnesium chloride (DPBS; Wel Gene, Seoul, Korea) and adjusted to 5 × 106 viable cells per milliliter of complete endotoxin-free medium RPMI-1640 (Invitrogen) supplemented with 10% fetal calf serum (Boehringer Mannheim, Australia) and 1x antibiotic–antimycotic (Gibco, Grand Island, NY, USA). PBMCs (4 × 105 cells/80 μl per well) were seeded in 96-well plates (Nunc, Roskilde, Denmark) in the presence of 100 μl uncultured medium or supernatants obtained from the culture of ASCs or CTLA4Ig-ASCs in a T175 flask (Corning, Corning, NY, USA) at 70-80% confluence. Cells were added with or without pTg (40 μg/ml), ConA (2.5 μg/ml; Sigma-Aldrich), and LPS (2.5 μg/ml; Sigma-Aldrich), and cultured for 72 h (total volume: 200 μl/well). After 3 days in culture, the BrdU assay was carried out (BrdU labeling time: 6 h) according to the manufacturer's instructions (Cell Proliferation ELISA BrdU, colorimetric; Roche Diagnostics, Mannheim, Germany). The reaction was quantified by measuring the optical density at a wavelength of 450 nm and a reference range wavelength of 690 nm. The stimulation index value (mean optical density values of Tg or mitogen-stimulated cultures/mean optical density values of medium-only cultures) was calculated for each culture treatment.
Detection of Multiple Cytokine Levels in Sera and Thyroid Tissue Extracts
Serum samples were collected and stored at −70°C before assay. Thyroids were weighed on a microgram scale and then transferred to 25 μl homogenizing solution [500 mM NaH2PO4/Na2HPO4 (Sigma-Aldrich) buffer and 0.2% Triton X-100 (Sigma-Aldrich) in H2O with anti-protease complete TM cocktail (Boehringer) per milligram of wet weight tissue]. The tissue was homogenized using a freeze-crushing cell homogenizer, Automill TK-AM5 (W280 mm x D 390 mm x H 290 mm, Tokken Inc., Chiba, Japan), at 1,400 rpm for 90 s twice and centrifuged at 16,000 x g for 15 min at 4°C. The supernatant was collected and stored at −70°C. Then the serum samples and the supernatant samples of the thyroid tissue extract were assayed using a multiplex cytokine/chemokine kit for granulocyte macrophage colony-stimulation factor (GM-CSF), tumor necrosis factor (TNF)-α, interferon (IFN)-γ, interleukin (IL)-2, IL-4, IL-6, IL-8, IL-10, IL-15, IL-18, and monocyte chemoattractant protein (MCP)-1 (Millipore, Bedford, MA, USA) according to the manufacturer's instructions.
Determination of the Proportion of Tregs in PBMCs
Before EAT induction, EAT induction/before therapy, and after therapy (on days 0, 15, and 34), blood collection was performed in all experimental animals. PBMCs were harvested from approximately 10 ml of venous blood, supplemented with sodium heparin as an anticoagulant. PBMCs were analyzed for Treg markers using CD4, CD25, and FoxP3 by flow cytometry (FACSCalibur flow cytometer; BD Bioscience) as in previous canine studies (5,36).
Histopathology
On days 104-106, the thyroid glands were surgically removed from dogs and bisected along the midsagittal line. One hemithyroid gland was fixed by immersion in 10% neutral-buffered formalin (Sigma-Aldrich), embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin (Dako, Carpinteria, CA, USA) and immunostained using antimacrophage mouse IgG1 (Abcam, Cambridge, UK) for histopathology. The other hemithyroid gland was snap frozen and stored at −70°C. Frozen sections were subjected to immunostaining using anti-canine CD4, anti-canine CD8 mouse IgGs, and anti-canine CD11c mouse IgG1 (Serotec Ltd., Oxford, UK). The immunostaining method was the same as in our previous study (10). The severity of the infiltration index of CD4+ or CD8+ cells was scored on a graded scale of 0-4. For immunohistochemical localization of anti-macrophage antibody, paraffin-embedded sections were deparaffinized in xylene, rehydrated in graded alcohol, and transferred to 0.01 M phosphate-buffered saline (PBS, pH 7.4). Subsequently, sections underwent heat-induced epitope retrieval with citrate buffer (pH 6.0; Dako) for 5 min at 121°C to reveal hidden antigen epitopes followed by cooling down at room temperature. For immunohistochemical localization of anti-CD11c antibody, frozen sectioned slides were washed in PBS. Then endogenous peroxidase was blocked by incubating slides with 3% hydrogen peroxide in PBS for 10 min at room temperature. After washing in PBS, sections were treated with serum-free blocking solution (Dako) for 20 min at room temperature to block nonspecific binding. Subsequently, sections were incubated with anti-macrophage mouse monoclonal antibody (1 μg/ml; Abcam) or anti-CD11c mouse monoclonal antibody (1/200; AbD Serotec, Kidlington, UK) for 60 min at room temperature. After washing in PBS, the sections were incubated for 30 min at room temperature with HRP-labeled polymer-conjugated secondary antibodies against mouse IgG (Dako). The color reaction was developed using the ready-to-use DAB (3,3′-diaminobenzidine) substrate-chromogen solution (Dako) for 5 min and then washed with distilled water. Finally, sections were lightly counterstained with Mayer's hematoxylin for 30 s before dehydration and mounting.
Confocal Staining for FoxP3
Frozen sections of the submandibular lymph node were fixed in acetone (Daejung Chemicals & Metals Co. Ltd., Seoul, Korea) for 10 min and washed in PBS three times for 5 min each. Nonspecific binding was blocked using 1% BSA/PBST (Koma Biotech, Seoul, Korea) for 30 min at room temperature. The slides were stained using FITC-labeled anti-FoxP3 Ab (1:100; eBioscience). After a 1-h incubation in the dark at room temperature, the slides were washed in PBS (Sigma-Aldrich) three times for 5 min each and stained sections (by counterstaining with mounting medium containing DAPI) were analyzed using a laser scanning confocal microscope (LSM 700; Carl Zeiss, Jena, Germany). The fluorescence intensity of the FITC-positive cells was scored on a graded scale of 0-5.
Confocal Microscopy for Finding CM-DiI-Labeled ASCs
One dog per group was given ASCs fluorescently labeled using conjugated red fluorochrome Cell Tracker CM-DiI (Life Technologies, Carlsbad, CA, USA) according to the manufacturer's instructions for identification in histopathological sections. The presence of CM-DiI-labeled cells was examined in various tissues such as thyroid, submandibular lymph node, spleen, kidney, liver, lung, and heart (by counterstaining with mounting medium containing DAPI) with a laser scanning confocal microscope (Radiance 2100 confocal microscope; Bio-Rad, Hercules, CA, USA). Excitation wavelengths of 405 and 543 nm were used.
Humoral Response to Infused Cells
For the detection of antibodies against administered ASCs, flow cytometric cross-match for anti-ASC IgG antibodies or anti-CTLA4Ig-ASC IgG antibodies was conducted using sera collected from recipient and control dogs. In detail, autologous recipient sera (1:25 dilution in DPBS) and control sera (non-ASC infused, negative control) were incubated with donor ASCs for 30 min at 37°C. After washing, ASCs were incubated with FITC-anti-canine IgG antibody (1:1,000 per 106 ASCs, AP308F; Millipore) for 1 h at 4°C, then washed in DPBS and assayed in a flow cytometer (FACS Aria, BD Bioscience). Analyses were performed with BD FACSDiva software (BD Biosciences). Fluorescence signals from 10,000 cells were counted, and the percentage of FITC-positive cells recorded.
Statistical Analysis
All results are expressed as the mean with the standard error of the mean (SEM). Data obtained from groups were compared using a one-way analysis of variance (ANOVA) followed by Tukey's post hoc multiple comparison tests. The paired t-test or Wilcoxon signed ranks test was used to compare the values of means from two related samples (before vs. after), as appropriate. Differences with a confidence level of 95% or higher were considered statistically significant (p < 0.05). All statistical analyses were conducted using SPSS version 17.0 (SPSS Inc., Chicago, IL, USA).
Results
Detection of the Transcription of Canine CTLA4 Gene by RT-PCR
RT-PCR to detect transcription of the therapeutic gene was carried out using RNA isolated from ASCs as the template. Agarose gel electrophoresis of the RT-PCR products from canine CTLA4Ig-ASCs revealed a single band of the expected size (394 bp), whereas no band was observed from the nontransduced control ASCs (data not shown). Electrophoresis of thyroid RT-PCR samples from the CTLA4Ig-ASC group showed a band of 394 bp, demonstrating that transcription of the therapeutic gene occurs in vivo. However, no band was detected in the saline-treated control group (data not shown).
ELISA to Determine the Concentration of Dog IgA in Stem Cell Culture Supernatants
We determined the concentration of dog IgA in stem cell culture supernatants to know whether CTLA4Ig-transduced ASCs really expressed CTLA4Ig and secreted CTLA4Ig effectively into the extracellular space. There is no antibody to the canine CTLA4 protein, and antibodies to CTLA4 of other species do not have cross-reactivity with canine CTLA4. Thus, we looked for a constant IgA region and determined dog IgA levels. Five days after selection with blasticidin (5 μg/ml), the cell culture supernatant was collected to determine the concentration of dog IgA by ELISA. The concentration of dog IgA in the culture supernatant of canine CTLA4Ig-ASCs was 79.9 ± 21.1 ng/ml, while that of nontransduced control ASCs was undetectable.
Immunophenotype of Dog ASCs
Nontransduced ASCs and CTLA4Ig-ASCs positively expressed CD29, CD44, and CD90 and did not express CD34, CD45, or MHC class II (Fig. 1).
Detection of Canine TgAA
Following the primer injection with porcine Tg, all 15 dogs were positive for TgAA on day 13 postimmunization. The highest TgAAs were observed on day 27 in all dogs. After the seventh CTLA4Ig-ASC IT+IV treatment, the TgAA titer on day 104 was decreased significantly compared to day 27 (paired t-test, p = 0.034), and these changes were also observed in the ASC IT+IV treatment group and the ASC IV treatment group (paired t-test, p = 0.023 and p = 0.013). By contrast, this change was not observed in the control group (Fig. 2).

(A) Detection of canine TgAA titers after immunization by porcine Tg. (B) Intrathyroidal injection. Experimental autoimmune thyroiditis was induced by injection of porcine Tg on days 0 and 13. On day 16 or 17, control, ASC IT + IV, and CTLA4Ig-ASC IT + IV groups were treated with 250 μl saline, 107 ASCs/250 μl saline, and 107 CTLA4Ig-ASCs/250 μl saline per thyroid, respectively. On days 16 or 17, 32, 46, 60, 73, 87, and 101, 2 × 107 ASCs/5 ml of saline (the ASC IT + IV group and ASC IV group; n = 6) or the same amount of CTLA4Ig gene-transduced ASCs/5 ml of saline (the CTLA4Ig-ASC IT + IV group and the CTLA4Ig-ASC IV group; n = 6) were administered intravenously. Control dogs were treated identically, but infused with 5 ml of saline instead of cells (n = 3). Serum samples were collected, and canine TgAAs were measured. The sample was considered to be positive when the value was >25% for the TgAA assay. Values <10% were counted as negative. The paired t-test was used to compare the values of means from two related samples. *Significant (p < 0.05) differences between the TgAA titer on day 27 and that on day 104.
On day 104, the TgAA levels were not significantly different among the groups (by the ANOVA followed by Tukey's post hoc multiple comparison tests) due to the small sample size and many numbers of the groups.
The suppression index [(1 - mean TgAA levels on day 104/mean TgAA levels on day 27) x 100%] of ASC IT + IV, ASC IV, CTLA4Ig-ASC IT + IV, CTLA4Ig-ASC IV, and control IT+IV groups were 45.3%, 37.8%, 62.8%, 46.2%, and 33.9%, respectively. CTLA4Ig-ASC IT + IV group showed the best results in the suppression index.
Serum Biochemistry
Serum concentrations of ALT, AST, BUN, creatinine, total protein, albumin, and total bilirubin were not significantly different among the groups before and after EAT induction. They were not significantly different before and after therapy. Almost all data were within the reference ranges (data not shown).
Canine TSH and Canine-Free T4
There was no significant effect on free T4 levels. Free T4 levels from sera acquired on day 27 (peak TgAA period) and day 104 (after last transplantation) were within the reference range (8-40 pmol/L). On day 27, one dog from the ASC IT+IV group and one dog of the ASC IV group had a TSH level higher than the reference range (0-0.6 ng/ml); these TSH levels were 1.94 and 1.11 ng/ml, respectively. After ASC transplantation, these TSH levels were within the reference range (0.1 and 0.51 ng/ml, respectively). There was no significant effect on TSH levels in other dogs. These findings can be ascribed to the fact that the dogs did not develop hypothyroidism. According to a veterinary textbook, clinical signs of hypothyroidism develop when over 75% of the thyroid gland has been destroyed (21).
Suppression Test of the Supernatant From Canine ASCs or Canine CTLA4Ig-ASCs on the Proliferation of PBMCs From EAT Dogs
Supernatants from cultures of ASCs or CTLA4Ig-ASCs were added to PBMC cultures that were stimulated with autoantigen or mitogens to determine whether the soluble factor of ASCs was associated with the immunomodulatory effect. PBMC proliferation in response to autoantigenic and mitogenic stimuli was significantly suppressed on adding culture supernatants of ASCs or CTLA4Ig-ASCs by ANOVA (pTg: p = 0.001, ConA: p = 0.001, LPS: p = 0.044) and Tukey's multiple comparisons (Fig. 3). The stimulation index of the supernatant from CTLA4Ig-ASC cultures was lower than that of ASC cultures, but it was not significantly different.

Suppression test of the supernatant from canine ASCs or canine CTLA4Ig-ASCs on the proliferation of PBMCs from dogs with EAT. PBMCs were harvested from EAT control dogs. PBMCs (4 × 105 cells/80 pl per well) were seeded in 96-well plates in the presence of 100 μl of uncultured medium or supernatants obtained from cultures of ASCs or CTLA4Ig-ASCs added with or without pTg (40 μg/ml), ConA (2.5 μg/ml), and LPS (2.5 μg/ml) then cultured for 72 h (total volume: 200 μl/well). After 3 days in culture, the BrdU assay was carried out. The stimulation index value (mean optical density value of Tg or mitogen-stimulated cultures/mean optical density value of medium-only cultures) was calculated for each culture treatment. Data obtained from groups were compared using a oneway ANOVA followed by post hoc Tukey's multiple comparison tests. *Significant (p < 0.05) differences from the control.
ELISA of Multiple Cytokine Levels in Sera and Thyroid Tissues
Compared with the levels seen before EAT induction (sera on day 0), the levels of GM-CSF, IFN-γ, IL-15, IL-18, MCP-1, and TNF-α were significantly increased after EAT induction/before therapy (sera on day 15) by Wilcoxon signed ranks test (data were not normally distributed, and we therefore used nonparametric tests for analysis) (Table 1). However, the cytokine levels of the sera were not significantly different among the groups due to the small number of samples in the groups along with larger values of standard deviation and differences in the baselines of the cytokines. Average IL-18, IL-2, and TNF-α levels from the thyroids were highest in the control group, but it did not reach statistical significance.
Multiple Cytokine Levels in Sera Before and After Experimental Autoimmune Thyroiditis (EAT)

Wilcoxon signed ranks test was used to compare the values of means from two related samples (before vs. after EAT induction). Min, minimum; Q, quartile; Max, maximum.
Determination of the Proportion of Tregs in PBMCs
Compared with that before EAT induction, the average of the proportion of Tregs was decreased in PBMCs after EAT induction/before therapy. The proportion of Tregs increased in all dogs after ASC or CTLA4Ig-ASC therapy, but decreased in two of the three control dogs treated with saline (Fig. 4).

Proportion of Tregs in peripheral blood by flow cytometry. Blood was collected from all experimental animals before EAT induction, EAT induction/before therapy and after therapy (on days 0, 15, and 34). PBMCs were analyzed for Treg markers, CD4, CD25, and FoxP3. Representative figures and data (mean ± SEM) are presented.
Histopathology
Immunoperoxidase staining of frozen thyroid sections showed severe infiltration of CD4, CD8, CD11c, and macrophage marker-positive cells between the follicles of the thyroid glands in the control group, but only mild infiltration in the ASC- or CTLA4Ig-ASC-treated groups (Fig. 5). The CD4+ cell infiltration index values of the control, ASC IT + IV, ASC IV, CTLA4Ig-ASC IT + IV, and CTLA4Ig-ASC IV groups were 2.3 ± 0.9, 1.0 ± 0.6, 0.7 ± 0.3, 1.0 ± 0.6, and 1.0 ± 0.6, respectively (mean ± SEM, ANOVA: p = 0.116). The CD8+ cell infiltration index of the control, ASC IT + IV, ASC IV, CTLA4Ig-ASC IT + IV, and CTLA4Ig-ASC IV groups were 3.0 ± 0.6, 1.3 ± 0.7, 1.0 ± 0.0, 1.3 ± 0.3, and 1.0 ± 0.0, respectively (mean ± SEM, ANOVA: p = 0.036).
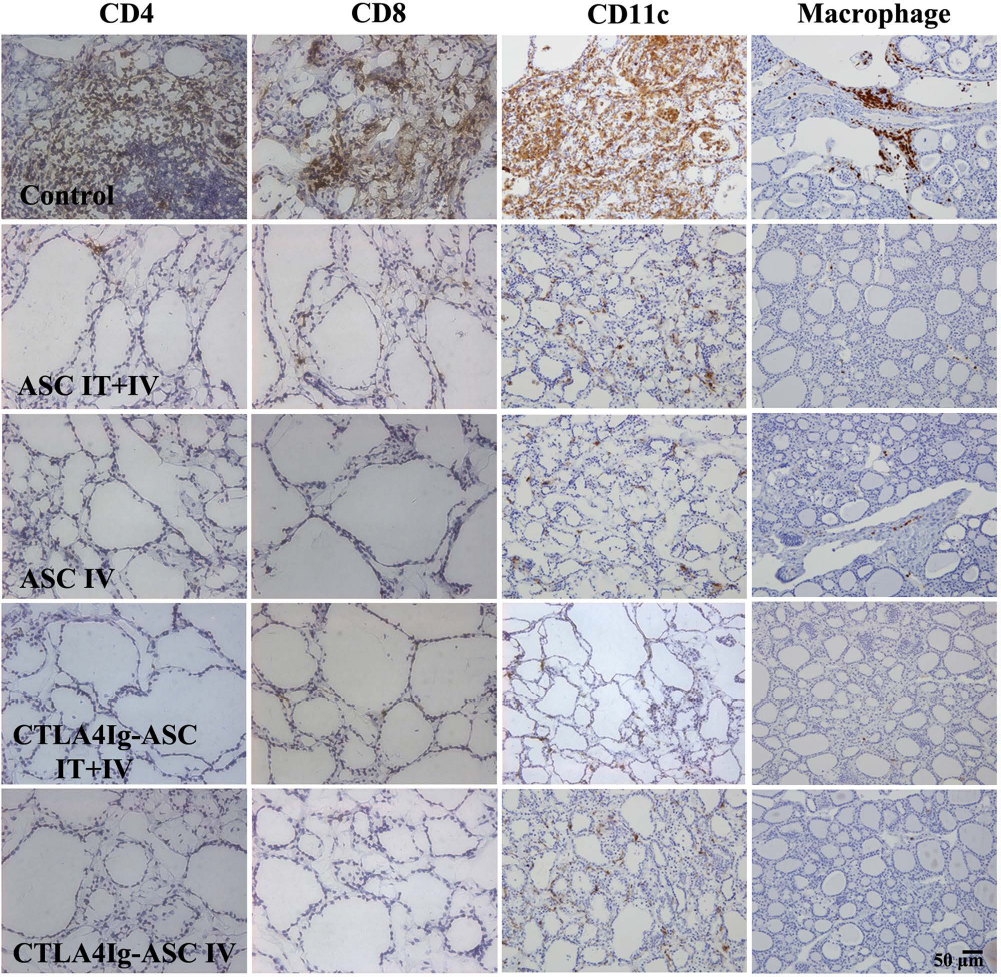
Immunohistochemistry for CD4, CD8, CD11c, and macrophage. Dogs with EAT were divided into five groups: (i) ASC IT + IV, (ii) ASC IV, (iii) CTLA4Ig-ASC IT + IV, (iv) CTLA4Ig-ASC IV, or (v) control IT + IV (saline only), to which treatments were administered intrathyroidally (10 million/250 μl saline per thyroid) one time or intravenously (20 million/5 ml) seven times within a 101-day period. Blood samples were collected every week, and thyroids were harvested on days 104~106. The thyroid gland sections were immunostained using anti-canine CD4, anti-canine CD8 mouse IgGs, anti-canine CD11c mouse IgG1, and anti-macrophage mouse IgG1.
Confocal Staining for FoxP3
Confocal staining of frozen submandibular lymph node sections showed many FoxP3-positive cells in all of the ASC- or CTLA4Ig-ASC-treated groups but only a few positive cells in the control group (Fig. 6). The fluorescence intensity of the FITC-positive cells was significantly higher in all of the ASC- or CTLA4Ig-ASC-treated groups compared to that in the control group by ANOVA (p = 0.003).

Confocal staining for FoxP3. Frozen sections of the submandibular lymph node were obtained from experimental beagle dogs with autoimmune thyroiditis. Slides were stained using FITC-labeled anti-FoxP3 antibody and counterstained with mounting medium containing DAPI. The slides were analyzed using a laser scanning confocal microscope (original magnification 200x, LSM 700). The fluorescence intensity of the FITC-positive cells was scored on a graded scale of 0-5. Data obtained from groups were compared using a one-way ANOVA followed by post hoc Tukey's multiple comparison tests. *Significant (p < 0.05) differences from the control.
Confocal Microscopy to Identify CM-DiI-Labeled ASCs
ASCs labeled with the CM-DiI red fluorescent tracker dye were mostly present in the spleen, thyroid, and submandibular lymph node (Fig. 7).

Confocal microscopy in thyroids, spleens, and submandibular lymph node. One dog per group was given ASCs fluorescently labeled using the conjugated red fluorochrome Cell Tracker CM-DiI for identification in histopathological sections. After counterstaining with mounting medium containing DAPI, the presence of CM-DiI-labeled cells was examined by confocal microscopy (original magnification 400x, Radiance 2100 confocal microscope).
Humoral Response to Infused Cells
The levels of IgG against autologous transplanted cells (percentage of IgG-positive cells) were not significantly different between recipient dogs and saline-treated control dogs (duplicate, Table 2). Furthermore, the level of IgG against autologous transplanted cells and the level of IgG against CTLA4Ig-overexpressing autologous transplanted cells were not significantly different (Table 2).
Humoral Response to Infused Adipose Tissue-Derived Stem Cells; Percentage of IgG-Positive Cells (Against ASCs; Unit: %)

Discussion
Animal models are an invaluable source to elucidate the development and progression of diseases, to study disease-associated pathways, and to evaluate new therapies (25,29). Murine EAT typically occurs on day 21, maximal lymphocytic infiltrations occur on days 28-35, then EAT remission occurs on days 42-49 after immunization for the induction of EAT (2). EAT model mice are typically sacrificed between day 21 and 28 after the first immunization to analyze the outcome of interest (7). Thus, it is difficult to study long-term therapeutic effects using the murine EAT model.
In a previous study, we established a canine model of autoimmune thyroiditis by immunizing normal dogs with bovine Tg (10). Using that method, a total of five injections of bovine Tg (every 2 weeks) and TgAA were monitored for about 10 months (11). Autoimmune thyroiditis lasted for over 10 months judging from the TgAA concentrations and thyroid histopathology of the control dogs (11). In that study, following the primer and the first booster injection with bovine Tg, all six treated dogs were positive for TgAA and the mRNA expression of IFN-γ, IL-15, and IL-18 in PBMCs during the development of autoimmune thyroiditis, but serum levels of cytokines were not determined at that time.
The sale of bovine Tg has been discontinued; thus, a canine model of autoimmune thyroiditis was induced in beagles using porcine Tg in this study. According to a report from Michigan State University, some breeds including beagles have a significantly higher laboratory prevalence of anti-TgAA compared to other dogs. After the second porcine Tg injection, autoimmune thyroiditis lasted for over at least 3 months judging from canine TgAA concentrations and thyroid histopathology of control dogs. Thus, a total of two injections of porcine Tg can induce autoantibodies against canine Tg, which results in the development of autoimmune thyroiditis in experimental beagle dogs. A canine model of experimental autoimmune thyroiditis can serve as a surrogate for studying the immunopathogenesis of the disease and for evaluating the therapeutic effects of new drugs over about 3 months of evaluation. Recent developments in serum cytokine quantification technology include multiplex arrays (30) and luminex multiplexing system. Analysis of cytokine expression patterns has been performed by ELISA for each separate sample. Luminex multiplexing system offers substantial cost and sample savings over traditional ELISA measurements and facilitates the simultaneous evaluation of multiple immune mediators (17). We determined the serum levels of various cytokines using a multiplex cytokine/chemokine kit for GM-CSF, TNF-α, IFN-γ, IL-2, IL-4, IL-6, IL-8, IL-10, IL-15, IL-18, and MCP-1. Serum cytokine levels of GM-CSF, IFN-γ, IL-15, IL-18, MCP-1, and TNF-α were significantly increased after the induction of autoimmune thyroiditis.
Autoimmune thyroiditis is characterized by the development of TgAA and the infiltration of inflammatory cells, especially T-lymphocytes, into the thyroid, causing the gradual destruction of thyroid follicles (3).
In the animal study, intrathyroidal and intravenous transplantation of CTLA4Ig-ASCs yielded the best results in controlling autoantibodies in established autoimmune thyroiditis.
Histopathological findings showed the possibility that either ASCs or CTLA4Ig-ASCs can reduce the infiltration of lymphocytes into the thyroid. According to a study by Roura-Mir et al., T cells isolated from intrathyroidal lymphocytic infiltrates of patients with Hashimoto's thyroiditis release high levels of IFN-γ and TNF-α (39). The combination of IFN-γ and TNF-α accelerated thyrocyte apoptosis through the Fas pathway and developed follicular disruption in EAT (42). Thus, the reduction of serum TgAA levels and the intrathyroidal infiltration of T-lymphocytes by ASC or CTLA4Ig-ASC transplantation are expected to prevent the development of autoimmune thyroiditis and the destruction of thyroid follicles.
The proliferation of PBMCs (from the dogs with established autoimmune thyroiditis) in response to autoantigenic and mitogenic stimuli was significantly suppressed by the addition of the culture supernatant from ASCs or CTLA4Ig-ASCs. It goes well with other previous studies; many previous reports showed that the culture supernatant of MSCs inhibited leukocyte proliferation, indicating that suppression by MSCs was partially mediated by a soluble factor (1,27).
Because MSCs secrete beneficial soluble factors such as immunomodulatory factors and home to injured targets, MSCs have great therapeutic potential for various diseases, including autoimmune diseases (16,22,23,44,45).
Our previous study reported that the transplantation of murine CTLA4Ig gene-transduced human ASCs reduces the inflammatory immune response and improves the Th1/Th2 balance in murine experimental autoimmune thyroiditis. However, a previous murine study showed the preventive effects of ASC transplantation before disease onset; thus, ASCs labeled with the CM-DiI red fluorescent tracker dye were mostly present in the spleen but not in the thyroid. In thyroids, a few very small fluorescent dots were seen, but they were not considered CM-DiI-labeled ASCs due to their weak signals. This migration might be because the stem cells primarily function in systemic immune modulation, due to the cells being given prior to disease induction (12).
In this canine study, many ASCs labeled with the CM-DiI red fluorescent tracker dye were present in the spleen and also in the thyroid and submandibular lymph node. This migration might be due to the cells being given after disease development (homing effect).
Flow cytometry detected high expression levels of CD29, CD44, and CD90, and the absence of CD34 and CD45 surface markers on the canine ASCs. The immunophenotype of CTLA4Ig-ASC was found to retain similar expression patterns with nontransduced ASCs (Fig. 1). Thus, we confirmed again that ASCs can be a useful vehicle for transferring therapeutic genes without interfering with an innate immunophenotype characteristic of ASCs (12).
The identification of FoxP3 as the master regulator of Tregs was a critical step in defining Tregs as a distinct T-cell lineage. Tregs expressing FoxP3 play a critical role in the maintenance of peripheral homeostasis and the prevention of autoimmune diseases (35,41). Furthermore, ectopic FoxP3 expression in CD4+ non-Tregs is sufficient to provide suppressor function in vitro and even in vivo (20,26,28,46). A recent study reported that cell-permeable FoxP3 protein regulates inflammatory immune responses and attenuates autoimmune diseases associated with inflammatory bowel disease and allergic airway inflammation (15). Thus, FoxP3 can be a very important therapeutic target for autoimmune diseases. In our previous study using (NZB x NZW) F1 mice, ASC transplantation increased the numbers of CD4+FoxP3+ cells in the spleen in a murine systemic lupus erythematosus model. In this study, the average of the proportion of Tregs from PBMCs was increased in all dogs treated with ASCs or CTLA4Ig-ASCs, but decreased in two of the three control dogs treated with saline. ASC or CTLA4Ig-ASC transplantation significantly increased the proportion of FoxP3-positive cells in the submandibular lymph nodes judging from the results of confocal staining. FoxP3 expression in lymph nodes near the target organ might be involved in the therapeutic mechanism of ASCs in autoimmune thyroiditis (as an organ-specific autoimmune disease).
Regarding humoral response to infused cells, our previous study on the therapeutic comparison between syngeneic and allogeneic ASC transplantation in murine EAT showed that both ASCs had a similar preventive effect on development of EAT, but the strongest humoral immune response was induced by xenogeneic transplantation, followed by allogeneic and syngeneic administration (13). As expected, transplantation of autologous ASCs did not induce any humoral response in this study. Thus, if there is no functional defect in one's stem cells, autologous transplantation of stem cells is ideal (38).
We intended to determine whether serial injections of ASCs or CTLA4Ig-ASCs (every 2 weeks, total seven times) affect the functions of various organs in this disease model, based on serum chemistry examination for about 3 months. ALT, AST, ALP, total bilirubin, BUN, creatinine, total protein, albumin, inorganic phosphate, and calcium were not significantly changed after serial injections of ASCs or CTLA4Ig-ASCs.
Although small numbers of animals were used in this study and a large number of experimental dogs should be tested before application in animal practice or in the clinic, this study aimed to describe the use of ASCs as a vehicle, the subsequent functional gene expression for correction of the abnormal status over long periods of time, and the effects of multiple injections of CTLA4Ig-ASCs in a large animal model.
In terms of applying the desirable results of the study to large-sized animals, ASCs that overexpress therapeutic genes may be effective in treating diseases and are profoundly significant from the aspect of translational study.
Ultrasound-guided stem cell injection without surgery is a possible clinical application for humans. The TgAA test can detect autoimmune thyroiditis years before clinical signs of hypothyroidism occur. Thus, intravenous and/or intrathyroidal transplantation of ASCs or CTLA4Ig-ASCs in that period can be attractive candidates to ameliorate autoimmune thyroiditis and prevent the development of hypothyroidism.
Footnotes
Acknowledgments
The authors thank the staff of the Laboratory Animal Research Center, Samsung Biomedical Research Institute for their technical support and assistance, especially So Young Park and Ji Hyun Park. This study was supported by the Samsung Biomedical Research Institute (grant SBRIP-A9-005/SMX1131211/SMX1140141) to Eun Wha Choi. Additional support was provided by the Basic Science Research Program through the National Research Foundation of Korea, which is funded by the Ministry of Education, Science, and Technology (NRF-2012R1A1A2006701) to Eun Wha Choi. The authors declare no conflict of interest.
