Abstract
Cesarean section (CS) scar tissue fibrosis and decreased muscle density increase the risk of uterine rupture and placental implantation in subsequent pregnancies. Given the potent anti-fibrotic and regenerative properties of human amniotic mesenchymal stem cells (hAMSCs), this study investigated their therapeutic potential in repairing uterine scars in a rat model. A full-thickness uterine wall excision model was established to mimic CS scarring. hAMSCs were transplanted at the edge of the incision. Myometrial thickness and collagen deposition within the scar were assessed histologically using hematoxylin and eosin (H&E) and Masson’s staining. Immunohistochemistry evaluated the expression of MMP8, TGF-β1, VEGFA, and α-SMA within the scar region. mRNA transcriptome sequencing and quantitative real-time polymerase chain reaction (qRT-PCR) were employed to explore possible mechanisms. A total of 110 rats were used in the study, 30 in the sham group, 40 in the phosphate-buffered saline (PBS) control group and 40 in the hAMSCs treatment group. Compared with the PBS group, the hAMSCs group exhibited a 35% reduction in collagen fiber area and a 28% increase in smooth muscle cell density (

Introduction
Cesarean section plays an important role in the management of high-risk pregnancy deliveries, and this technique is mature and widely used. However, the uterine smooth muscle has a limited regenerative capacity, making it unable to fully repair myometrial damage caused by cesarean section. As a result, the injured myometrial tissue is replaced by scar tissue. Scar tissue is less elastic and contractile, leading to increased risk of adverse outcomes in repeat pregnancies such as uterine rupture, placental implantation, and cesarean section keloid pregnancies 1 . With the increase in cesarean section rates, the repair of uterine scars has also attracted the attention of obstetricians. However, no effective method has been established to improve healing at the uterine incision site2,3. Wound healing is a complex, continuous, and dynamic evolutionary process, in which cell proliferation, neovascularization and other complex biological events are intertwined and regulated by a variety of cytokines and signal transduction pathways 4 . Among them, the TGF-β/Smad and Wnt/β-catenin pathways are particularly important and play a key role in the balance between fibrotic and regenerative repair at the site of injury5–8. During fibrotic repair, on the one hand, TGF-β signaling is continuously activated and regeneration at the site of injury is inhibited while fibrosis is enhanced5,9, while on the other hand, insufficient neovascularization of regenerating tissues leads to nutrient deficiencies, which leads to the regenerating tissues to re-denature and ultimately still be replaced by fibrous tissues10–12. The regenerative repair of smooth muscle at the site of uterine injury also faces these challenges, coupled with the lack of a strong cell proliferation capacity of the uterine smooth muscle tissue itself, making regenerative repair of uterine smooth muscle particularly difficult.
Stem cell therapy is a new medical technology that uses the regenerative ability of stem cells to repair or replace damaged cells and tissues to restore the form and function of the original tissues. Mesenchymal stem cells (MSCs) are important members of the stem cell family and have received increasing attention because of their multidirectional differentiation potential, immune regulation, and self-replication. MSCs can differentiate into bone, cartilage, muscle, nerves, liver, myocardium, endothelium, and other tissues
In this study, hAMSCs were applied to rat uterine incisions to study effects on tissue repair and the possible underlying mechanism, providing an experimental basis for the treatment of poor healing of uterine incision scars using stem cells.
Materials and methods
hAMSCs source
Discarded amniotic membranes were obtained from five healthy pregnant women without infectious diseases or other chronic diseases after full-term delivery at the Department of Obstetrics and Gynecology, General Hospital of Northern Theater Command (all of whom provided informed consent for the abandonment of amniotic membranes). To obtain hAMSCs, amniotic membranes were repeatedly rinsed with phosphate-buffered saline (PBS) under aseptic conditions, isolated, and cultured using enzyme digestion. The study protocol was reviewed and approved by the Medical Ethics Committee of the General Hospital of the Northern Theater (Approval No. Y2021 (123), approved on November 26, 2021).
Experimental animals
A total of 110 SPF-grade Sprague-Dawley (SD) female rats, aged 7–8 weeks and weighing 220 ± 20 g, were purchased from Liaoning Changsheng Biotechnology Co., Ltd. (Benxi City, Liaoning Province, China). (Animal Quality Certificate No. 210726210101097576; Animal Use License No. SCXK [Liao] 2020-0001). The animals were kept in the SPF-class animal feeding room of the Department of Obstetrics and Gynecology, General Hospital of Northern Theater Command, and were fed special feed and clean drinking water in a well-ventilated environment with moderate temperature and humidity, and intermittent day and night lighting (12 h/12 h). The experimental animal feeding environment and operation procedures were in accordance with Regulations on the Management of Experimental Animals. This study followed the triple-blind method, and the protocol was reviewed and approved by the Animal Medical Research Ethics Committee Branch of the General Hospital of Northern Theater Command (Approval No. 2021-008, Approved on October 26, 2021). All efforts were made to minimize the number of animals used and their suffering. Animal experiments were conducted in strict accordance with the standards of the ARRIVE guidelines. This study was in line with the ARRIVE guidelines 2.0.
Isolation, culture, and characterization of hAMSCs
Extraction and culture of hAMSCs
hAMSCs were isolated and cultured by trypsin and collagenase digestion of fresh amniotic membrane 22 . Amniotic tissues were obtained from the Department of Obstetrics and Gynecology of the General Hospital of the Northern Theater Command. The donor placenta was delivered by cesarean section in accordance with medical standards, and the amniotic membrane was bluntly separated from the placenta within 10 min of delivery. Cell isolation was completed within 2 h of specimen collection.
After obtaining the amniotic membrane, it was repeatedly washed with PBS to remove blood and mucus. The tissue was digested 3–4 times with trypsin-ethylenediaminetetraacetic acid (EDTA; Thermo Fisher Scientific, Waltham, MA, USA). The remaining tissues were digested with 1 mg/ml collagenase IV (Thermo Fisher, USA) for 60 min, and the resulting digestion mixture was filtered through a 200-mesh cell sieve, and the filtered liquid was centrifuged at 800 rpm for 10 min. The supernatant was discarded, and the cells were resuspended in serum-free MSC medium (YOCON, N0.7 fengxian Street, Haidian District, Beijing, China). The cells were inoculated in a 25-cm2 culture bottle with a density of 2.5 × 109 cells/l, cultured at 5% CO2 and 37°C saturated humidity, and the complete medium was replaced 24 h later. Cells were continuously expanded up to the third passage (P3).
Identification of multiple differentiation potential of hAMSCs
The P3 generation of adherent hAMSCs was obtained and cultured until 90% confluence was reached. OriCell human adipose MSCs adipogenic induction differentiation medium, OriCell human adipose MSCs osteogenic induction differentiation medium, and OriCell human adipose MSCs chondrogenic induction differentiation medium (Cyagen, N0.162 Renmin Street, Suzhou City, Jiangsu Province, China) were used to induce adipogenic, osteogenic and chondrogenic differentiation of hAMSCs, respectively. After 4 weeks, induced differentiation of the tissue cells was detected.
Identification of hAMSCs
P3 hAMSCs were extracted, digested with trypsin, and neutralized in serum. The collected cell sediments were washed three times with PBS. 1 × 106 hAMSCs were suspended in 100 μl PBS. Then, 5 μl of each PE-labeled antibody (CD31, CD44, CD45, CD73, CD90, CD105, and HLA-DR; BioLegend, San Diego, CA, USA) was added, and incubated at room temperature for 30 min away from light. After cleaning with PBS, hAMSCs were suspended with 500 μl PBS and analyzed by flow cytometry.
Rat uterine scar models and treatment groups
The rats were divided into sham operation group (n = 30), PBS control group (n = 40), and hAMSCs group (n = 40) using the randomized numerical table method. The rats were anesthetized by the inhalation of a mixture of 1.5% isoflurane and oxygen at a flow rate mask of 3.0 l/min. All experimental rats underwent laparotomy. After entering the abdominal cavity, no treatment was administered to the sham-operated group. In the other two groups, the entire layer of the uterus was incised upward and longitudinally at the bifurcation of the cervix at the contralateral margin of the mesometrium, with a length of approximately 1.5 cm. In the hAMSCs group, 0.1 ml of hAMSCs (2 × 10⁶/ml) was injected into the myometrium at multiple points along both sides of the incision margin. The PBS control group received an equal volume of PBS following the same surgical procedure. In both groups, the uterine incision was closed using 7-0 absorbable sutures, and a No. 1 silk thread was placed at the upper edge of the incision to mark its location. Ten rats in each group were randomly selected for execution on days 7 and 14 after surgery, and the remaining rats were sacrificed on day 30 after surgery by an overdose of anesthesia (5.0 l/min, 5% isoflurane; RWD Life Science Co., Shenzhen, China) followed by euthanasia. The sample size selection and rat uterine scar model were developed according to the experimental protocol described by Fan et al. 27
Histological evaluation of myometrium thickness and collagen fibers in rat uterine scars
Three rats were randomly selected from each group at each observation time point and the uterus was removed. Hematoxylin and eosin (H&E) staining was performed to evaluate the morphological structure of the uterus. Paraformaldehyde (4%; Solarbio, Beijing, China) was used to fix mouse uterus specimens for 24 h. Mouse uterus was embedded in the paraffin and then cut transversely into sections with a thickness of 5 μm. Sections were stained according to the instructions of the Hematoxylin and Eosin (HE) Staining Kit (Solarbio), and then sealed with neutral gum. Local capillaries, collagenous connective tissue, and smooth muscles in the scar tissue were observed under a light microscope. Five fields of view (×100) were randomly selected from the blue-stained area of the scar in each section and analyzed using ImageJ computer image analysis software. Masson’s trichrome staining was performed to observe collagen deposition around the uterine scar. Paraffin sections were stained using the Masson Trichrome Staining Kit (Solarbio), and after staining was completed, the sections were also sealed with neutral resin. After Masson’s staining, collagen fibers were blue, muscle fibers were red, and nuclei were blue-black under a light microscope. Each section was randomly selected from five fields of view (×100) in the blue-stained area of the scar and analyzed using ImageJ image analysis software. The ratio of collagen deposition in the scar area to that in the smooth muscle area was measured, and the mean value was calculated.
Immunohistochemical detection of MMP8, TGF-β1, VEGFA, and α-SMA in rat uterine scars
Six rats were randomly selected from each group at each observation time point, and the sections of each group were stained with IHC to detect the expression and tissue localization of α-smooth muscle actin (α-SAM), transforming growth factor (TGF-β1), vascular endothelial growth factor (VEGFA), and anti-matrix metalloproteinase-8 (MMP8) in the uterine scar region. The dewaxing and rehydration procedures were the same as those used for HE staining. The tissues were blocked with goat serum (Solarbio). A primary antibody (rabbit anti-αSMA polyclonal antibody (Abcam, Discovery Drive, Cambridge, UK), rabbit anti-TGF-β1 polyclonal antibody (Abcam) 1:200, rabbit anti-VEGF polyclonal antibody (Abcam) 1:200, rabbit anti-MMP8 polyclonal antibody (Abcam) 1:200) was added and refrigerated at 4°C overnight. The next day, goat anti-rabbit IgG antibody (HRP-labeled) was added dropwise, and the slides were dried and sealed with neutral gum. The α-SMA was positively expressed in the cytoplasm of tissue cells in the form of slender, long spindle-shaped brown-yellow and brown particles. TGF-β1 was positively expressed in the cytoplasm and cell membrane in brown-yellow color. MMP8 is positively expressed in the cytoplasm of tissue cells (yellow). VEGFA is usually positively stained in the cytoplasm or nucleus of cells. Six slices from each section were randomly selected. Six fields of view were randomly selected from each section, magnified 100×, and the percentages of α-SAM, TGF-β1, VEGFA, and MMP8-positive cells were determined by ImageJ, and the mean values were taken.
Fluorescence tracing of hAMSCs after transplantation
The hAMSCs were labeled with PKH26 to track their trajectory according to the instructions of the CFDA SE Cell Proliferation and Cell Tracking Kit (Yeasen, Shanghai, China). Rats were injected hAMSCs labeled with PKH26. Modeling and injection methods as before. The uterine tissues were removed on day 3 after laparotomy and cut into 5-μm sections. Antifade mounting medium was added after adding DAPI (Beyotime Institute of Biotechnology, Shanghai, China) onto the slide, and fluorescence images were obtained using a fluorescence microscope (ZEISS, Jena, Germany).
RNA-seq transcriptomic profiling: differential expression and pathway analysis
Transcriptome analysis was performed on five rats in the PBS group and five rats in the hAMSCs group. Thirty days after surgery, five mice were randomly selected from each PBS- and hAMSC-treated group. Laparotomy was performed on all experimental rats, and the entire uterus was removed. The scar tissue of 50- to 100-mg rat uterus was quickly cut into small pieces using micro-scissors. Total RNA was extracted using the TRIzol reagent (Thermo Fisher Scientific) according to the manufacturer’s instructions. The extracted nucleic acids were assessed for concentration using a Nanodrop 2000 (Thermo Scientific, USA) and integrity using an Agilent 2100 LabChip GX (Agilent, Santa Clara, CA, USA). For the RNA sequencing protocol, a library was constructed by screening mRNAs that met the criteria from five tissue samples. Screening requirements are as follows: total amount ≥2 (μg), two times of library construction, concentration ≥40 (ng/μl), volume ≥10 (μl), OD260/280 between 1.7 and 2.5, OD260/230 between 0.5 and 2.5, 260 nm absorption peak display normal, RIN value ≥7. The mRNA in these tissue samples were enriched using magnetic beads containing oligonucleotides (dT). cDNA libraries were constructed according to the manufacturer’s Library construction Kit Hieff NGS Ultima Dual-mode mRNA Library Prep Kit for Illumina (Yizheng, China). The constructed library was sequenced using Illumina NovaSeq6000 and a 150-bp double-ended sequencing was generated. Finally, the signaling pathways of the Differentially Expressed Genes (DEGs) were analyzed.
RT-qPCR validations of differentially expressed genes in rat uterine scar tissues
Based on the gene sequencing results of the transcriptomes of PBS and hAMSCs treatment groups, six genes were randomly selected for reverse transcription-polymerase chain reaction (RT-PCR) verification. The expression levels of Lamc2, Lamb3, Wnt4, Fzd5, Wnt5a, and PPARD were determined. Five rats were randomly selected from the PBS and hAMSCs treatment groups, and total RNA was extracted using the TRIzol method. The RNA reverse-transcribed into cDNA using the PrimeScript RT kit (Takara, Kusatsu, Shiga, Japan). Target genes were amplified by quantitative polymerase chain reaction (qPCR) using the SYBR Premix Ex Taq II kit (Takara, Shiga, Japan) with specific primers listed in Table 1. PCR amplification program parameters were set: pre-denaturation at 95°C for 30 s, denaturation at 95°C for 5 s, annealing at 60°C for 30 s, and 45 cycles. The Ct value was calculated using the internal software of ABI 7500 Fast Real-Time PCR System. Target gene expression was determined based on the Ct value, and GAPDH was used as an internal reference. For each group, the experiment was performed in triplicate, with three technical replicate wells prepared for each sample. The target gene amplification level was obtained by calculating the 2−ΔΔCt method.
Primer information for real-time fluorescence quantitative PCR.
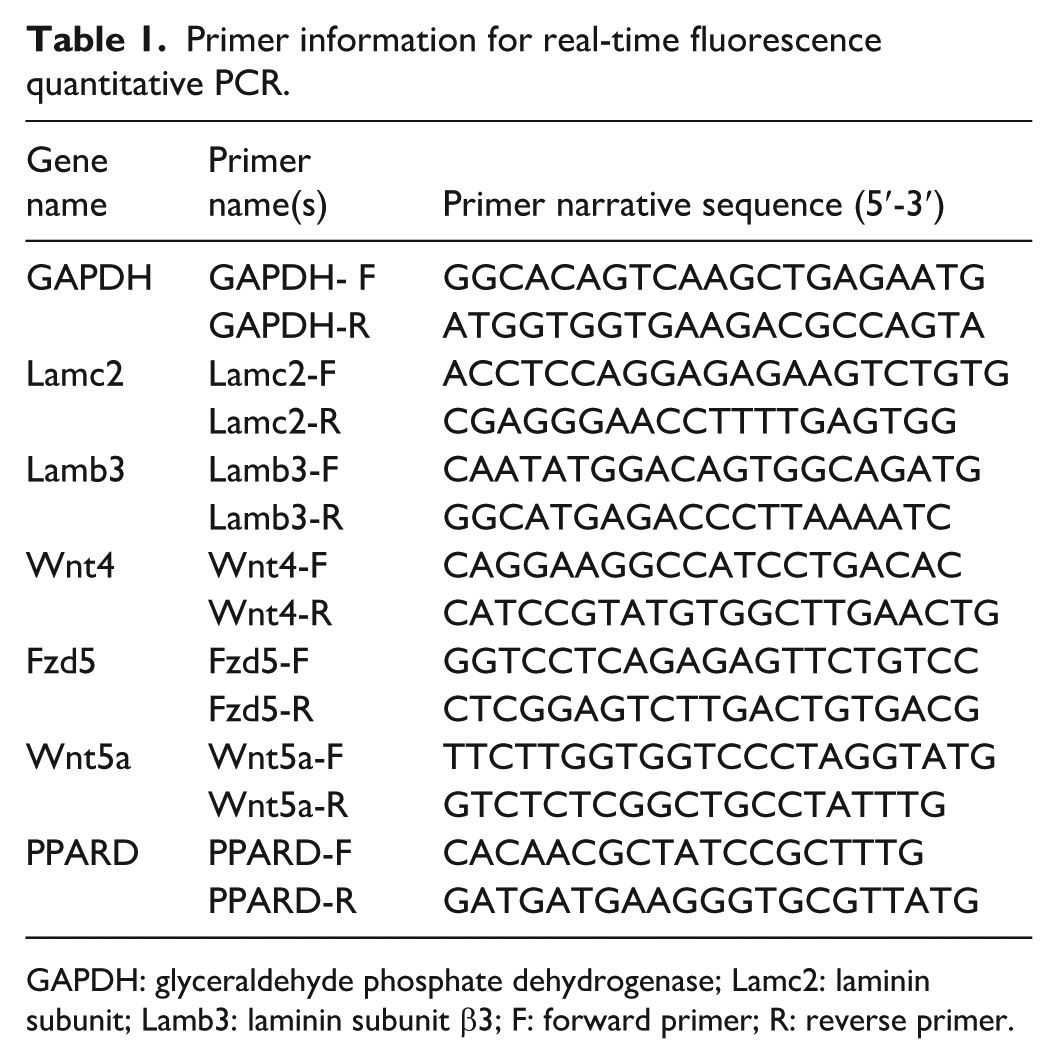
GAPDH: glyceraldehyde phosphate dehydrogenase; Lamc2: laminin subunit; Lamb3: laminin subunit β3; F: forward primer; R: reverse primer.
Statistical analysis
Statistical analyses were performed using GraphPad Prism 9. Data were expressed as mean ± standard deviation (SD) of at least three independent experiments. One-way analysis of variance (ANOVA) and the least significant difference (LSD) test were used for comparisons between groups. GAPDH was used as an endogenous control. Statistical significance was set at
Results
hAMSC has the potential for polydifferentiation into lipids, osteogenesis, and cartilage
hAMSCs extracted from the human amniotic membrane were morphologically homogeneous and pike-shaped. After culturing in lipogenic, osteogenic, and chondrogenic differentiation media, hAMSCs expressed lipid droplets, calcium nodules, and acidic mucopolysaccharides, respectively, indicating that they have lipogenic, osteogenic, and chondrogenic differentiation abilities. Further details are shown in Fig. 1.

Lipogenic, osteogenic, and chondrogenic differentiation of hAMSCs. (a) The hAMSCs extracted from human amniotic membrane showed homogeneous cell morphology and pike shape under the microscope. (b) Calcific nodules stained orange-red by alizarin red. (c) Fat droplets were stained reddish-orange by Oil Red O. (d) Chondrocytes and stroma stained light blue by Alcian blue.
Identification of specific surface markers of hAMSCs
After passage to the third generation, hAMSCs showed positive expression of CD44, CD73, CD90, and CD105, but no expression of CD31, CD45, or HLA-DR. This was consistent with the characteristics of hAMSCs. Further details are shown in Fig. 2.

Specific expression of surface markers on hAMSCs. Flow cytometry revealed that hAMSCs were positive for CD44, CD73, CD90, and CD105, and negative for CD31, CD45, and HLA-DR.
hAMSCs improve uterine morphology and reduce fibrosis in rat uterine scars
According to the results of HE staining, the myometrium of the PBS group was thinned, myofibers were not neatly arranged or were broken, more inflammatory cells infiltrated, and capillaries were sparse. The myometrium of the hAMSCs group was thicker than that of the PBS group, myofibers were more neatly arranged, fewer inflammatory cells were infiltrated, and more neonatal capillaries were observed. There was no statistically significant difference in myometrial thickness between the hAMSCs and sham operation groups. Masson’s trichrome staining revealed a large number of dark blue collagen fibers in the myofibers of the PBS group, and the number of red muscle fibers was significantly reduced. The hAMSCs group showed significantly more collagen degradation and increased number of muscle fibers, and the arrangement was more regular than that in the PBS control group. The difference in the area of collagen fibers between the hAMSCs and sham groups was not statistically significant. Further details are shown in Fig. 3 and in the Supplementary Material (Supplementary Figs. S1, S2, and S5).
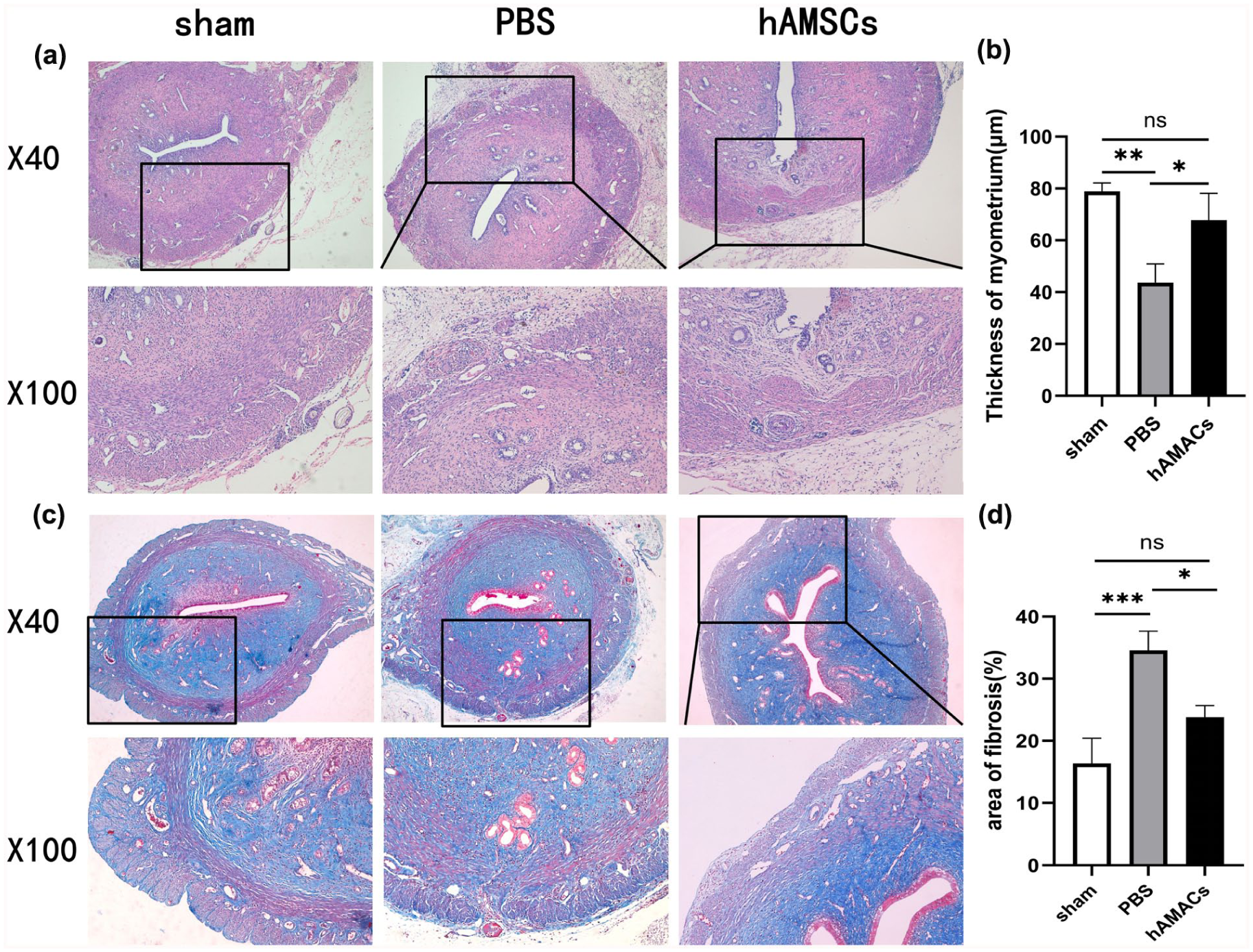
HE staining and Masson’s staining of rat uterine incision tissues. (a, c) Hematoxylin and eosin (HE) and Masson staining of rat uterine scars in each group. (b). Myometrial thickness of rats in each group. (d) Proportion of collagen fibers in the region of uterine scarring in each group of rats (
hAMSCs upregulate MMP8/VEGFA/α-SMA and suppress TGF-β1 in rat uterine scars
MMP8 was expressed in the plasma of uterine scar tissues in all three groups, and the positive expression rate of MMP8 in the hAMSCs group was higher than that in the PBS and sham groups, whereas the positive expression rate of MMP8 was not significantly different between the PBS and sham groups (

Immunohistochemistry of rat uterine incision tissues. (a, c, e, g) Representative images of MMP8, TGF-β1, VEGF, and α-SMA immunohistochemistry of rat uterine incision tissues from each groups. (b, d, f, h) Percentage of MMP8, TGF-β1, VEGF, and α-SMA-positive areas at the uterine incision site in each group of rats (×200,
hAMSCs migrate into the wound after transplantation
Fluorescence microscopy revealed that pKH26 (red fluorescence)-labeled hAMSCs migrated into the wound, suggesting that hAMSCs have the ability to converge toward uterine wounds. After transplantation hAMSCs were able to migrate into the wound and function within the wound (Fig. 5).

Migration of hAMSCs into the wound after transplantation. The hAMSCs were labeled using pKH26 (red fluorescence), and hAMSCs were observed migrating into the wound on day 3 after transplantation.
Transcriptomic profiling identifies DEGs between PBS and hAMSCs-treated rat uterine scars
Under the condition of satisfying the

DEGs at the site of uterine incision in hAMSCs and PBS group rats. (a) The histogram of differentially expressed genes; gray and blue indicate the genes upregulated and downregulated in the hAMSCs group compared to the PBS group. (b) The volcano map of differentially expressed genes (red and green) indicates the genes upregulated and downregulated in the hAMSCs group compared with the PBS group.
Functional enrichment reveals core DEGs and signaling pathways: PBS vs. hAMSCs-treated uterine scars
Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis results showed that the genes Laminin, Collagen, Wnt, Frizzled, and PPARD were differentially expressed between the hAMSCs and PBS groups. The hAMSCs group showed lower expression of the Laminin, Collagen, Wnt, Frizzled, and PPARD genes than the PBS group. Differential genes encoding laminin and collagen are involved in the production of collagen fibrils and fibronectin in the extracellular matrix (ECM). wnt, Frizzled, and PPARD differential genes were mainly involved in the classical Wnt4/β-catenin and Wnt5/Ca2+ signaling pathways. Further details are shown on Fig. 7.

Differential gene pathway analysis. Green indicates upregulated or active interactions and red indicates downregulated or repressed interactions. (a) ECM: extracellular matrix, Syndecan: transmembrane proteins, Perlecan: string-beaded proteoglycan. (b) Frizzled: for the receptor for the Wnt gene, PPARδ (PPARD): peroxisome proliferator–activated receptor δ.
RT-qPCR validated core DEGs in uterine scar tissues
In this study, the mRNA expression of Lamb3, Lamc2, Wnt4, Wnt5a, Fzd5, and PPARD genes in the uterine incision tissues of rats in the hAMSCs and PBS groups was detected using real-time quantitative PCR. The gene sequencing group showed that the mRNA expression levels of each gene in the hAMSCs group were significantly lower than those in the PBS control group, and the differences were statistically significant (

Expression of Lamc2, Lamb3, Wnt4, Fzd5, Wnt5a, PPARD in PBS group and hAMSCs group. The mRNA relative expression level of (a) Lamb3; (b) Lamc2; (c) Wnt4; (d) Wnt5a; (e) Fzd5; (f) PPARD. Control: control group administered phosphate buffer injection; hAMSCs: experimental group administered human amniotic membrane mesenchymal stem cell injection (n = 3, *
Discussion
Cesarean section is an important obstetric surgery used to treat high-risk pregnancies and abnormal deliveries, and to save the lives of mothers and perinatal infants. With the increase in older and high-risk pregnant women, the cesarean section rate in China has risen sharply in recent years and is much higher than the cesarean section rate (10%-15%) recommended by the World Health Organization (WHO) 1 . In the early 2000s, the cesarean section rate in China was approximately 20%. By 2008-2018, the cesarean section rate had increased to approximately 30%–40% in certain areas 2 . The integrity of the uterus provides the necessary internal environment for embryo attachment and the maintenance of pregnancy. Uterine smooth muscle cells are stable cells with a weak regenerative capacity after injury. The repair connection process for damaged myocytes is long, and the smooth muscle at the break is mainly connected by fiber scarring. Invasive procedures, such as cesarean section and myoma nucleation, can cause severe uterine trauma and damage the myometrium, triggering the overactivation of fibroblasts and continuous secretion of collagen. However, these processes hinder the proliferation, differentiation, and migration of the original uterine cells. As a result, damaged uterine walls experience loss of muscle cells and the formation of collagen scars28–30. The texture of the scar is tougher than that of normal myometrial tissue; therefore, it is less elastic and sparsely vascularized, which significantly affects tissue function31,32. This leads to an increased risk of scar pregnancy, uterine rupture, and placental implantation in subsequent pregnancy, seriously threatening the health and life of the mother and child 3 . Therefore, uterine scar repair is a critical clinical concern. However, studies on the repair of uterine scar myometrium are still lacking. Therefore, there is an urgent need to explore novel and feasible treatments that promote uterine scar repair.
MSCs are a class of stem cells with multidirectional differentiation potential and are widely found in a variety of tissues, such as the bone marrow, adipose tissue, umbilical cord, and placenta. They can multidirectionally differentiate and proliferate, secrete various cytokines and growth factors, and promote tissue repair and anti-inflammatory effects. They can be readily obtained and induced to differentiate into smooth muscle cells under specific conditions. MSCs from the bone marrow and umbilical cord have been widely used to study uterine injury repair33–35. As perinatal stem cells, hAMSCs isolated from discarded amniotic membranes have the advantages of immune tolerance, non-tumorigenicity, low cost, and abundant cell sources, making them promising candidates for clinical applications36,37. Currently, hAMSCs are widely used in the treatment of pulmonary fibrosis, myocardial infarction, skin scarring, and intrauterine adhesions 32 . The application of hAMSCs in cesarean section scar repair is a promising research direction.
In this study, it was observed at both the HE and Masson tissue levels that the hAMSCs group could effectively reduce collagen deposition in wound-healing tissues. The hAMSCs group was similar to the sham group in its ability to restore the integrity of the myometrium and promote uterine incision healing in rats. This is consistent with the results of Sun et al. 38 in a study of transplanted MSCs on cesarean section scar healing in rats. Further studies at the molecular level have found that hAMSCs group showed high expression of α-SMA, VEGFR, and MMP8 in the uterine scar region compared with the PBS group, while the positive expression of TGF-β1 was reduced. TGF-β1 is a key factor involved in regulating ECM metabolism and initiating the fibrosis process. It induces fibrosis primarily by driving fibroblast differentiation, promoting fibronectin synthesis, promoting interstitial proliferation, and inhibiting the breakdown of the ECM. In particular, the TGF-β/Smad pathway is widely recognized as the core pathway of fibrosis, and TGF-β1 plays a key role in generating the fibroblast phenotype, which is the main cause of the deposition of large amounts of collagen fibers of type I and type III, and is highly correlated with scarring29,34,39. α-SMA is a type of actin specifically expressed in smooth muscle cells. α-SMA expression usually correlates with smooth muscle cells. SMA expression is usually associated with contractile function and tissue remodeling in smooth muscle cells. It is a key marker of smooth muscle cells and is mainly involved in cell contraction and the maintenance of vascular tone40,41. VEGF is a potent proangiogenic factor that stimulates the proliferation, migration, and neovascularization of vascular endothelial cells, thus promoting tissue repair42,43. Li et al. 22 showed that human amniotic epithelial cells promoted VEGF expression in a mouse model of intrauterine adhesions and improved angiogenesis and stromal cell proliferation. MMP-8 is an important matrix metalloproteinase that helps myoblasts and myogenic stem cells migrate to the wound site by degrading damaged ECM during muscle injury, thereby promoting muscle regeneration and repair of the injury site 44 . Duerr et al. 45 demonstrated in their study that MMP/TIMP expression results in reduced cardiomyocyte apoptosis, smaller scar size, and preservation of cardiac function. Fan et al. 27 demonstrated that MMP-8 can cleave mesenchymal collagen I, II, and III and is involved in the tissue remodeling process of the uterus in a rat uterine scar model in a study of human amniotic epithelial cell transplantation to promote morphologic and functional regeneration. Therefore, our results suggest that hAMSCs may promote the remodeling of muscle tissue structure and the maintenance of function in uterine scar sites through various pathways, such as promoting myoblast regeneration and decreasing fibroblast proliferation, upregulating the expression of vascular endothelial growth factor (VEGF), which promotes angiogenesis, improving the microenvironment of the damaged site, and promoting the degradation of extracellular ECM in the scarred area.
At the gene level, hAMSCs downregulated the expression of Laminin, Collagen, Wnt, Frizzled, and PPARD by gene sequencing. The differential expression of Lamc2, Lamb3, Wnt4, Fzd5, Wnt5a, and PPARD was verified using fluorescence quantitative PCR. Laminin and Collagen genes promote the production of collagen fibers and fibronectin to promote scar formation 46 . Laminin subunit β3 (Laminin β3, Lamb3) and Laminin subunit γ2 (Laminin γ2, Lamc2) are different isoforms of Laminin, and Lamb3 is involved in the coding of laminin. Silencing of Lamb3 reduced the protein expression levels of collagen I in myofibroblast models and reversed TGF-β-induced phenotypic transformation of embryonic stem cell myofibroblasts 47 . Lamc2, as a part of laminin, promotes cell (e.g. fibroblasts, endothelial cells) adhesion and migration, activates cell growth-related signaling pathways, and thus promotes the proliferation of fibroblasts and other cells, and upregulation of Lamc2 promotes fibrosis of scarring and the persistence of inflammation 48 . The results of this study showed that Lamc2 and Lamb3 were less expressed in the hAMSCs group than in the PBS group in the rat uterine scar region. This suggests that hAMSCs reduce laminin synthesis during scar repair by affecting the expression of Lamc2 and Lamb3, which facilitate scar repair. Wnt4 (Wnt Family Member 4) mediates myofibroblast differentiation in the skin, kidneys, lungs, and liver, especially during scarring or fibrosis. VEGF is also a downstream target gene of the Wnt/β-catenin signaling pathway. hAMSCs can play a role in promoting wound healing through activation of the classical Wnt4/β-catenin signaling pathway, which regulates the endothelial cell proliferation, migration, and angiogenesis 49 . Wnt5a (Wnt Family Member 5A) usually acts through the non-classical Wnt5a/Ca2+ signaling pathway, and Fzd5 acts as a receptor for wnt5 co-participating in the expression of the pathway. La et al. 50 found that both Wnt4 and Wnt5a levels were increased in the lungs of elderly individuals, and myofibroblast-like differentiation was increased. In a study by Kovacs et al., 51 it was found that in cultured activated HSCs, the expression of the Wnt gene was increased by approximately 3–12 times compared with resting HSCs, and there was excessive ECM deposition. The peroxisome proliferator-activated receptor delta (PPARD) is a nuclear receptor that regulates lipid metabolism, inflammation, and fibrosis. Although its role in uterine repair is poorly understood, PPARD activation inhibits myofibroblast differentiation in other tissues. In a study on the role of PPARD in liver fibrosis 52 , the expression of PPARD was found to be increased in fibrotic liver tissue, promoting liver fibrosis. In another study 53 , PPARD agonist could reduce TGF-β1-mediated activation of HSCs and fibrosis. Our transcriptomic data showed a significant downregulation of PPARD in hAMSC-treated scars. This may reflect a shift from profibrotic to regenerative signaling. In summary, the expression of Wnt4, Fzd5, Wnt5a, and PPARD genes in the uterine scar area of rats in the hAMSCs group was lower than that in the PBS group. This suggests that hAMSCs may affect the activity of Wnt4/β-catenin and Wnt5/Ca2+ signaling pathways by influencing the expression of related genes, thereby reducing the deposition of collagen fibrin and facilitating the repair of normal muscle tissue.
This study demonstrates for the first time that hAMSCs can promote smooth muscle tissue regeneration and inhibit scar formation in a rat uterine incision. The results of this study provide experimental evidence for the use of hAMSCs to repair cesarean incision scarring. Based on the results of this study, it may be possible to develop cesarean incision repair strategies utilizing hAMSCs in the future, so that the chances of placenta implantation and keloidal pregnancies can be effectively reduced, and in addition, with the adjustment of the fertility policy in some large populated countries (e.g. China), it can be predicted that the proportion of pregnant women who undergo ≥3 cesarean deliveries will rise in the world. It is expected that the proportion of pregnant women undergoing ≥3 cesarean deliveries will increase, and the results of this study may be able to contribute to the reduction of the incidence of uterine rupture in this group of pregnant women. In terms of study design and implementation, this study has some advantages. First, the study design included a PBS group and a sham-operated group, which provided a bidirectional control for the observation of the efficacy of hAMSCs and enabled the efficacy of hAMSCs to be reflected more objectively. Second, this study explored the mechanism by which hAMSCs promoted the regeneration of uterine incisional smooth muscle tissues using transcriptome sequencing, which provided a direction for subsequent mechanistic exploration.
However, owing to the preliminary nature of this study, there were still many limitations. First, the observation period of this study was 30 days, which lacked the observation of the long-term efficacy and safety of hAMSCs after transplantation, and ignored the investigation of the survival and functional maintenance duration of hAMSCs in immunocompetent rats’ models, weakening the exploration of the potential clinical applicability of the results of this study. Second, this study did not assess functional outcomes such as uterine contractility and rat fertility after hAMSCs transplantation, which further limited the exploration of the clinical translational potential of the results of this study. Third, the present study was conducted in a rat model and failed to evaluate immune indicators such as inflammatory cytokines and immune cell infiltration, which weakened the exploration of the safety of the clinical use of the present findings. Fourth, in terms of mechanistic studies, although this study used transcriptome sequencing and real-time fluorescence quantitative PCR to explore the pathway perturbations occurring at the uterine incision site after transplantation of hAMSCs, the lack of experimental validation of the relevant pathways reduces the corroboration and depth of the mechanistic studies. In addition, this study did not explore the specific mode of action of hAMSCs transplantation; that is, whether it involves direct differentiation and proliferation after transplantation or crosstalk with other cells in the local microenvironment through paracrine effects. More importantly, the findings of this study are derived from a xenogeneic transplantation model. Beyond the aforementioned concerns regarding immune responses and the precise mechanisms of hAMSCs post-transplantation, their application in humans faces additional challenges. These include the cell survival rates, long-term efficacy, risks of immune rejection, tumorigenic potential, and aberrant differentiation in allogeneic transplantation. These critical issues necessitate further in-depth, systematic, and longitudinal investigations.
Conclusion
In conclusion, this study demonstrated that hAMSCs transplantation can promote the regeneration of smooth muscle and inhibit scar tissue generation in rat uterine incisions and explored its possible mechanisms. At the tissue level, hAMSCs may promote scar repair by promoting collagen fiber degradation and myofibril production in the uterus. At the molecular level, hAMSCs may affect the formation of fibroblasts in the rat uterine scar region by decreasing TGF-β expression. hAMSCs may promote smooth muscle cell proliferation, angiogenesis, and degradation of the ECM at the uterine scar by affecting the expression of α-SAM, VEGF, and MMP8, thereby promoting the reconstruction of normal uterine muscle tissues to promote the repair of uterine incision scar. Through mRNA transcriptome sequencing and qPCR validation, we found that hAMSCs inhibited the activity of multiple fibrotic pathways at the gene level, possibly by downregulating the expression of Lamc2, Lamb3, Wnt4, Fzd5, Wnt5a, and PPARD genes. The results of this study provide new research avenues for uterine incision repair and potential medical options for the prevention and treatment of cesarean section scarring. However, our study has limitations. In future, we should further design experiments to systematically explore the precise mechanism of hAMSCs transplantation in promoting uterine smooth muscle regeneration. We should also assess the long-term effects, functional utility, and immune response of hAMSCs transplantation to provide more experimental evidence for the clinical translation of hAMSCs.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251387398 – Supplemental material for The effect and mechanism of human amniotic mesenchymal stem cells on scar formation in rat uterine incision
Supplemental material, sj-docx-1-cll-10.1177_09636897251387398 for The effect and mechanism of human amniotic mesenchymal stem cells on scar formation in rat uterine incision by Caixi Li, Feng Zhao, Jia Song, Ting Zhang, Rui Yang, Yi Xiao, Yujia Zheng, Jingli Sun and Zhenyu Chen in Cell Transplantation
Footnotes
Acknowledgements
We would like to thank Chao Chen for the technical help in this study.
Ethical Considerations and Consent to Participate
For experiments on human amniotic stem cells, the study entitled “Effect of Human Amniotic Stem Cells on Uterine Scar Healing in Rats” was approved by the Medical Ethics Committee of the General Hospital of Northern Theater Command (Approval No. Y2021 (123), approved on November 26, 2021). The study titled “Effect of Human Amniotic Stem Cells on Uterine Scar Healing in Rats” was approved by the Animal Medical Research Ethics Committee Branch of the General Hospital of the Northern Theater Command (Approval No. 2021-008, Approved on October 26, 2021). Animal experiments were conducted in strict accordance with the standards of the ARRIVE guidelines.
Consent for Publication
All authors confirmed their consent for publication.
Author Contributions
CL and FZ: Conception and design; acquisition, analysis, and interpretation of the data. Manuscript preparation and revision. ZC: Conception and design. Manuscript revision, project administration, and funding. CL, JS, TZ, RY, YX, and YZ performed the experiments and drafted the manuscript. JS contributed to scientific design. CL, JS, TZ, RY, and ZC designed the experiments and wrote the manuscript. Both the authors have read and agreed to the submitted version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Program of Liaoning Province (2022JH2, 101500029).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statements
Data supporting the findings of this study are available from the corresponding author upon request.
Artificial Intelligence (AI)
The authors declare that they did not use AI-generated work in this manuscript.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
