Abstract
Neuro-inflammation plays a key role in the pathophysiology of brain infarction. Cell therapy offers a novel therapeutic option due to its effect on immunomodulatory effects. Amniotic stem cells, in particular, show promise owing to their low immunogenicity, tumorigenicity, and easy availability from amniotic membranes discarded following birth. We have successfully isolated and expanded human amniotic mesenchymal stem cells (hAMSCs). Herein, we evaluated the therapeutic effect of hAMSCs on neurological deficits after brain infarction as well as their immunomodulatory effects in a mouse model in order to understand their mechanisms of action. One day after permanent occlusion of the middle cerebral artery (MCAO), hAMSCs were intravenously administered. RT-qPCR for TNFα, iNOS, MMP2, and MMP9, immunofluorescence staining for iNOS and CD11b/c, and a TUNEL assay were performed 8 days following MCAO. An Evans Blue assay and behavioral tests were performed 2 days and several months following MCAO, respectively. The results suggest that the neurological deficits caused by cerebral infarction are improved in dose-dependent manner by the administration of hAMSCs. The mechanism appears to be through a reduction in disruption of the blood brain barrier and apoptosis in the peri-infarct region through the suppression of pro-inflammatory cytokines and the M2-to-M1 phenotype shift.
Background
Ischemic stroke is a major cause of permanent disability and death in people worldwide 1 . The neuro-inflammation that occurs following occlusion and deprivation of the blood supply plays an important role in the pathophysiology of ischemic stroke 2 .
Lack of blood, due to a disruption of the blood supply, initiates ischemic damage through hypoxia, the production of reactive oxygen species, complement activation, alteration of blood brain barrier (BBB) permeability, and the increased expression of adhesion molecules, such as selectin and intracellular adhesion molecule-1 (ICAM-1), on capillary vessels 2 –5 . This is followed by the adhesion and infiltration of neutrophils and circulating monocytes. These blood-borne immune cells and perivascular macrophages release neurotoxic mediators such as matrix metalloproteinase9 (MMP9), tumor necrotic factor α (TNFα), nitric oxide (NO), and interleukin 1β (IL1β), leading to further BBB breakdown and enhanced leukocyte infiltration 2,6 –9 . Neuronal cell death causes the next phase of the inflammatory response, resulting in the release of damage-associated molecular pattern proteins, which activate parenchymal macrophages and microglia resulting in the production of pro-inflammatory cytokines such as interleukin23 (IL23), IL1β, and TNFα 2,7 . This is followed by the induction of IL23 induced interleukin-17-producing γδ T lymphocytes, which then act on macrophages to promote neurotoxic effects 2,7 . Although this process promotes clearance of dead tissue and neuro-regeneration, excessive inflammation often interferes with tissue repair and exacerbates the damage caused by the primary injury 2,7,10 .
Currently, intravenous administration of the recombinant tissue plasminogen activator and endovascular thrombectomy are performed worldwide as standard reperfusion therapies in the hyperacute phase for normalizing blood supply to ischemic penumbra, and for minimizing the ischemic core. However, these therapies are characterized by narrow time windows, and good outcomes are achieved only in a limited number of patients, even when these therapies are successfully performed. Furthermore, anti-inflammatory agents that block a single molecular pathway, such as ICAM1, the IL1 receptor, or neutrophils, have been studied as potential novel therapeutics in the management of cerebral infarction, although most of these studies failed 11,12 . Therefore, there is a need to focus on novel innovative therapeutic options.
Cell therapy has recently been recognized as an innovative therapeutic option for various neurological disorders 13 . It can target both the subacute and chronic phases of cerebral infarction, thereby providing an opportunity to manage chronic symptoms 14 . Stem cells used for treating ischemic infarction may be obtained from several sources, such as human embryonic stem cells (ES cells), induced pluripotent cells, and mesenchymal stem cells (MSCs) 13,14 . Among these, MSCs exhibit a unique feature, wherein these cells can exert their biological effects on neurogenesis, angiogenesis, cytokine production, and immunomodulation 14 –18 .
Amniotic stem cells, which are multipotent stem cells possessing phenotypic features similar to those of bone marrow-derived mesenchymal stem cells 10,19 , modulate immune responses by suppressing the production of excessive pro-inflammatory cytokines and enhancing the production of anti-inflammatory cytokines. Thus, full advantage of these immunomodulatory effects may be taken by administering amniotic stem cells during the early stages of stroke, thereby promoting the beneficial effects of inflammation during the early stages, while regulating its harmful effects later on 10,20 .
Furthermore, amniotic stem cells are unique in their immune tolerance, which is consistent with the functions of the amnion to protect the fetus from the mother’s immune system 10,20,21 . Host responses have not been reported following xenotransplantation into immunocompetent animals, and this feature confers a unique advantage to amniotic stem cells, with particular reference to allogeneic therapeutic applications without immunosuppressants 19 .
We have successfully isolated, expanded, and formulated human amnion derived mesenchymal stem cells (hAMSCs), derived from human fetal membranes 22 , and demonstrated their immunomodulatory effects including the suppression of macrophage 23 and Th1/Th17 immunity 24 . For use as a potential drug, validation of the safety and efficacy of administering hAMSCs intravenously to patients with treatment resistant Crohn’s disease and acute graft-versus-host disease is now under investigation with respect to their immunomodulatory effects via phase I/II clinical trials 25,26 .
The objective of this study was to evaluate the therapeutic effects of hAMSCs on neurological deficits in a mouse model of cerebral infarction. Furthermore, we investigated the mechanisms underlying the immunomodulatory effects of these cells.
Materials and Methods
Animals
The study protocol was approved by the Animal Care Committee of Hyogo College of Medicine (Approval number: 17-034) and performed in accordance with the ‘Guide for the Care and Use of Laboratory Animals’ published by the National Academy of Science of the USA. Seven to nine-week-old male CB-17/Icr-+/+Jcl mice (Clea Japan Inc., Tokyo, Japan) were housed in a temperature (22-24°C) and humidity (55%) controlled room under a 12/12 light–dark schedule. The animals were allowed access to water and standard pellet chow ad libitum.
Induction of Permanent Focal Cerebral Infarction by Direct Occlusion of the Middle Cerebral Artery (MCAO) in Mice
The procedure was performed according to a previously described method 27 . Prior to surgery, the mice were anesthetized using 2% isoflurane. Briefly, an incision was made in the skin between the left eyeball and the left ear hole. The left zygoma was dissected and detached under an operating microscope. A bone window (diameter: 1.5 mm) was made using a dental drill, and the main trunk of middle cerebral artery (MCA) was electrocauterized and cut down just distal to the olfactory tract to induce a permanent focal cerebral infarction (Mylan, Canonsburg, Pennsylvania, USA). This method has been reported to confer a number of advantages when investigating the therapeutic effects of experimental treatments on ischemic infarction. These advantages are as follows: (a) the model produces highly reproducible lesions of cortical infarction; (b) mice show long-term survival; and (c) it is a model of permanent focal ischemia which mimics the pathophysiology of actual human cerebral infarctions. A sham surgery was also performed in control mice. For sham surgery, all the arteries were exposed for the surgical period, but the MCA was not occluded.
Cell Preparation and Administration of hAMSCs
The hAMSCs were prepared as previously described 22 . Briefly, informed consent was obtained from all donors who provided fetal membranes according to the protocol approved by our institutional ethic committee (approval number 325). The fetal membranes were obtained from healthy donor mothers following a cesarean section. The amnion was mechanically separated from the chorion and processed with collagenase/dispase solution. Following this, the cells were suspended in minimal essential medium with bovine-derived platelet lysate “NeoSERA®” (https://www.japan-biomedical.jp) and incubated at 37°C for cell expansion. Mice were randomly assigned to four groups as follows: high-dose hAMSC group (MCAO followed by administration with a high dose of hAMSCs); low-dose hAMSC group (MCAO followed by administration with a low dose of hAMSCs); the vehicle control group (MCAO followed by administration of lactated Ringer’s solution alone); and sham group (administration of lactated Ringer’s solution after sham surgery). Mice in the first and second groups received an intravenous injection of either 100 µL or 50 µL of hAMSCs (4 × 106 and 2 × 106 cells/kg) via the carotid vein under direct view 24 h after MCAO the induction of MCAO, respectively. Mice in the third and fourth groups received an intravenous injection of lactated Ringer’s solution alone.
Behavioral Tasks
During the period between 1 and 2 months post-MCAO, a series of behavioral tasks were conducted on mice who had undergone MCAO, including the open field test, the wire hang test, the Y-maze task, the water maze learning task, the passive avoidance learning task, and the open space swimming test. These tasks were conducted to assess the functional deficits and recovery of the animals. Assessments were conducted by independent experimenters who were blinded to the experimental groups. Increase in general activity and anxiety after ischemic infarction 28,29 and the degree of impairment in habituation after repeated exposure, which reflects memory disturbance 30 , were measured as locomotion counts by the open field test. Motor coordination and muscular strength were tested with the help of a wire hang test. Cognitive functions were assessed by the Y-maze, water maze learning, and passive avoidance learning tasks. Finally depression-like symptoms were also assessed using the open space swimming test 31 . Details of the methods used are described in the supplemental material.
Quantitative Real-Time RT–PCR (q-PCR)
Seven days post-MCAO, mice in three groups (i.e., the high-dose hAMSC group, vehicle control group, and sham surgery group) were euthanized using isoflurane and decapitated. Ipsilateral hemispheres of the brain samples were quickly isolated and incubated in RNAlater RNA Stabilization Reagent (Qiagen, Hilden, Germany). Total RNA was extracted using a RNeasy mini kit (Qiagen) according to the manufacturer’s instructions. The purified total RNA was converted into cDNA using ReverTra Ace qPCR RT Master Mix (Toyobo, Osaka, Japan), following which real-time PCR was performed using THUNDERBIRD SYBR qPCR Mix (Toyobo) according to the manufacturer’s instructions. The relative expression levels of the housekeeping genes glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as well as TNFα, MMP2, MMP9, and inducible nitric oxide synthase (iNOS) were determined. The primer sequences used for real-time PCR are listed [Additional file 1]. The levels of target gene expression were calculated via the ΔΔCT method and normalized against those of GAPDH.
Immunohistochemistry
Seven days post-MCAO, mice in two groups (i.e., the high-dose hAMSC group, and vehicle control group) were euthanized using isoflurane and decapitated. Brain tissues were fixed overnight in 4% paraformaldehyde, dehydrated in 30% sucrose, frozen at –80°C, and sliced into 10 µm coronal sections using a cryostat. Frozen sections were placed in PBS and a blocking solution (0.3% H2O2 in methanol) consecutively for 10 min each. They were then washed with PBS thrice for 3 min each time and placed in a permeabilization solution for 10 min (1% TritonX-100 in PBS). They were washed again with PBS twice for 5 min each time and placed in the blocking solution for 30 min at 25°C (0.1 M TBS containing 20% bovine serum and 3% BSA). The sections were then incubated overnight at 4°C with an anti stem101 antibody (1:100; Takara, Shiga, Japan, Y-40400), Diaminobenzidine (DAB) was used as a chromogen, and the nuclei were counterstained with hematoxylin. In fluorescent immunohistochemistry, non-specific binding was blocked with Blocking One Histo (Nacalai Tesque, Kyoto, Japan) according to the manufacturer’s instructions, and permeabilized with 1% Triton X-100 in PBS for 10 min. The sections were incubated overnight at 4°C with an anti-iNOS antibody (1:500; Funakoshi, Tokyo, Japan, MAB9502), or an anti-CD11b/c antibody (1:100; Bioss, Woburn, USA, bs-1014R), and then incubated with corresponding Alexa Fluor 488- or 555-conjugated secondary antibodies (Cell Signaling Technologies, Danvers, Massachusetts, USA) at room temperature for 2 h. Nuclei were counterstained with 4′,6-diamino-2-phenylindole (DAPI; Sera Care, Milford, Iowa, USA). Histological images were captured using a fluorescence microscope BZ-X (Keyence Corporation, Osaka, Japan) at a magnification of 200 x in order to count iNOS or CD11b/c positive cells. Eighteen fields in each of the peri-infarct areas (three non-overlapping random visual fields per coronal section and two sections per brain;
The ischemic core was observed as a clearly demarcated necrotic area with low cell density and rich in debris and the peri-infarct area was observed as an area with a high degree of cell accumulation surrounding an ischemic core (Fig. 2a) 32,33 .
Western Blotting
Seven days post-MCAO, mice in three groups (i.e., the high-dose hAMSC group, vehicle control group, and sham surgery group) were euthanized using isoflurane and decapitated. Ipsilateral hemispheres of the brain samples taken from all the groups were quickly frozen at −80°C, homogenized, and lysed using an EzRIPA Lysis kit according to the manufacturer’s instructions (Atto Corporation, Tokyo, Japan). Protein levels in the whole lysates were measured using a BCA assay (Thermo Scientific, Waltham, Massachusetts, USA), and 20 µg of each protein sample was separated by 10% SDS-PAGE and then electrophoretically transferred to a PVDF membrane (Atto Corporation). Membranes were blocked in EzBlock Chemi (Atto Corporation) and probed with each antibody overnight. The membranes were then incubated with appropriate horseradish peroxidase-conjugated secondary antibodies after washing with 1% Triton X-100 in Tris buffered saline (TBS). Immunoreactive proteins were detected using an enhanced chemiluminescence kit (EzWestLumi plus, Atto Corporation) and visualized using LuminoGraph 1 (Atto Corporation). Quantification of immunoreactive proteins was performed using Image J software (NIH, Bethesda, MD, USA). The following antibodies were used in these experiments: anti-TNFα (1:200, Santa Cruz, Dallas, TX, USA, sc-52746), and anti-caspase3 (1:200, Santa Cruz, CA, USA, sc-7272).
Gelatine Zymography
The protein extracts were prepared and normalized using the process described for western blotting. Gelatine zymography was performed to measure MMP gelatinase activities according to the manufacturer’s instructions (Cosmo Bio Corporation, Tokyo, Japan). The samples were subjected to electrophoresis in the precast gels. Following this, the gels were incubated in renaturing buffer to remove SDS for 1 h, incubated in developing buffer at 37°C for 48 h, and then stained with Coomassie Blue for 30 min. Finally, the gels were decolorized and scanned using the white light image scanner ES-8500 (Epson, Suwa, Japan).
Evans Blue Assay
BBB permeability was assessed in mice from two groups (i.e., the high-dose hAMSC group, and vehicle control group) via the extravasation of Evans blue (EB), an index of albumin leakage. As previously described 34 , Evans blue dye (4% in PBS, 4 mL/kg) was administered via the carotid vein 48 h following MCAO, which was 24 h after the administration of hAMSCs. Mice were euthanized and transcardially perfused with 100 mL of saline 2 h after EB dye administration at room temperature to wash out intravascular EB. Next, whole brains were quickly obtained. The ipsilateral hemisphere, contralateral hemisphere, and cerebellum, from each whole brain were separated from each other, and weighed. The samples were incubated in formamide (0.5 mL) at 55°C for 2 days. Supernatants were separated via centrifugation (15,000 × g, 10 min) following which the EB levels in supernatants were estimated, by measuring corresponding fluorescence at a wavelength of 620 nm, using a spectrophotometer (SPECTRA max PLUS384, Molecular Devices, San Jose, CA, USA). Tissues were then oven dried at 95°C for 5 days to obtain their dry weight. The EB levels in the brain samples was quantified using a standard linear regression curve using 11 concentrations (0–104 ng/mL) of EB dye and levels expressed as µg/brain. Relative EB levels were expressed as fold change of the contralateral side, corrected with brain dry weight.
Terminal Deoxynucleotidyl Transferase (TdT)-Mediated dUTP Nick End Labelling (TUNEL) Assay
In order to detect apoptotic cells, a TUNEL assay was performed in the mice from two groups (i.e., the high-dose hAMSC group, and the vehicle control group) according to the manufacturer’s instructions (In situ cell death detection kit, POD; Sigma-Aldrich, St. Louis, Missouri, USA). Briefly, brain tissues were fixed overnight with 4% paraformaldehyde and cryoprotected in 30% sucrose. Frozen sections were placed in PBS for 30 min, blocking solution for 10 min (3% H2O2 in methanol), PBS for 10 min, permeabilization solution for 2 min on ice (0.1% TritonX-100 in 0.1% sodium citrate), washed three times with PBS for 3 min each time, placed in blocking solution for 30 min at room temperature (0.1 M TBS containing 20% bovine serum and 3% BSA), and then washed three times with PBS for 3 min each time. DNA strand breaks were labeled using fluorescein-12-dUTP by incubating with TdT buffer for 60 min at 37°C. As a negative control, the incubation was performed without the TdT enzyme. DAB was used as a chromogen, and the nuclei were counterstained with hematoxylin. Neurons showing chromatin condensation, nuclear budding and fragmentation, or apoptotic bodies were identified as being apoptotic, whereas cells showing diffuse cytoplasmic labeling were considered as necrotic cells. Cells labelled as positive were manually counted using a light microscope (Keyence Corporation) at 400× magnification. The sample size and method for image capture were in accordance with immunofluorescence staining requirements.
Statistical Analysis
All data were expressed as mean ± SEM. JMP ver. 13 (SAS Institute Inc., Cary, NC, USA) was used to analyze data. Differences between two groups (i.e., the high-dose hAMSC group versus the vehicle control group) were analyzed for statistical significance via an unpaired two-tailed
Results
Acute Intravenous Administration of hAMSCs Improves the Neurological Deficits Induced by Cerebral Infarction in a Mouse Model of MCAO
On the day following the MCAO operation hAMSCs were administered intravenously and their therapeutic effects were then evaluated during the chronic phase of the disease (Fig. 1a). Mice who underwent the MCAO operation (
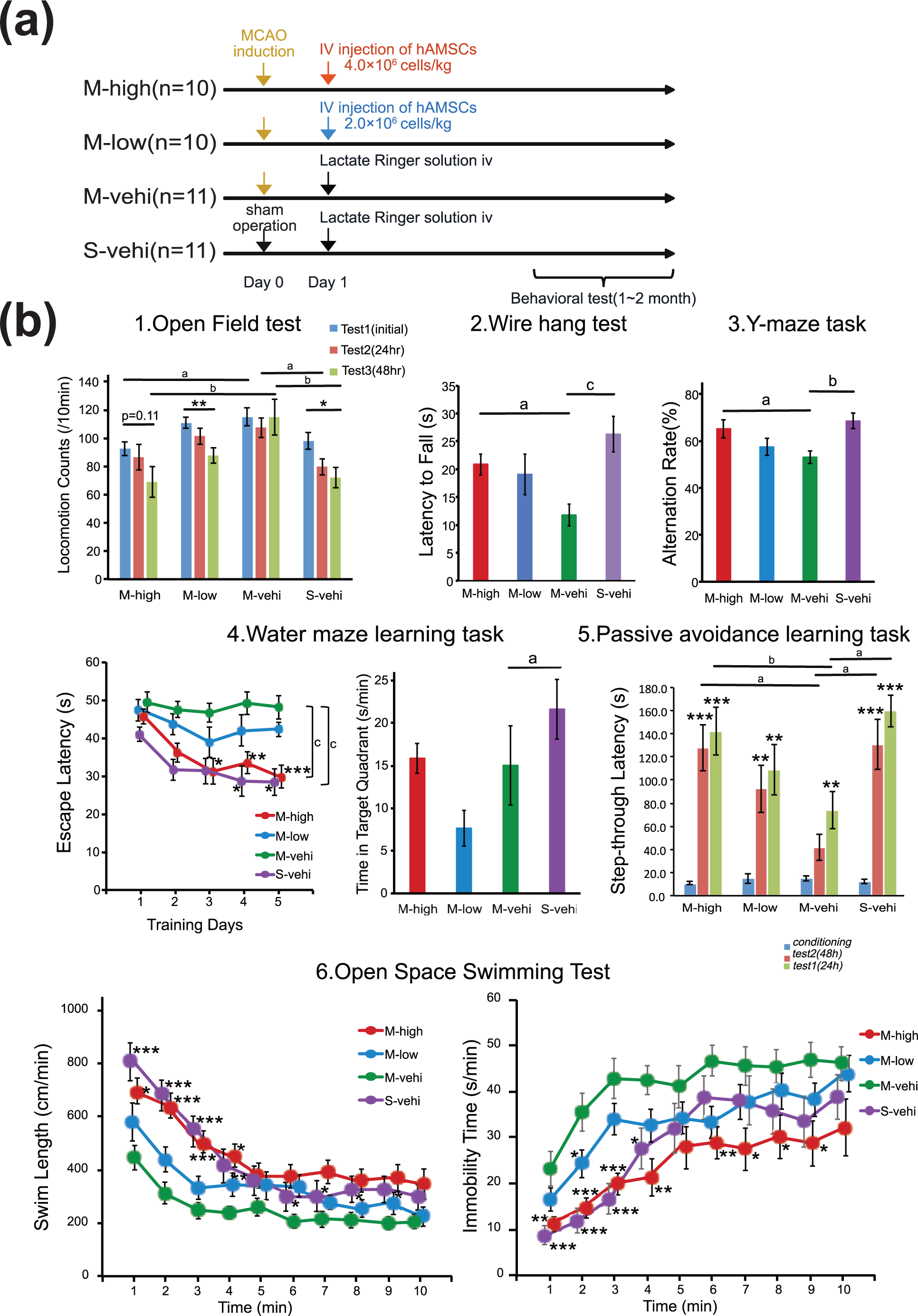
Acute intravenous administration of hAMSCs improves functional recovery following permanent MCAO. (a) Protocol for the evaluation of behavioral tasks. Behavioral testing was conducted using CB17 male mice randomly assigned to one of four arms: M-high (MCAO followed by the intravenous administration of a high dose of hAMSCs at 24 h;
For data arising from repeated measures (i.e., the open field test, escape latency of the water maze learning task, the passive avoidance learning task, and the open space swimming test), there were significant main effects of group and group by time interaction effects, except for an interaction effect for locomotion counts in the open field test, F (5, 62) = 0.817,
Compared with mice in the S-vehi group, mice in the M-vehi group exhibited significant behavioral abnormalities in several behavioral tasks. In the open field test, across-session habituation decreases, which reflect a time-dependent decline in locomotion counts, were significantly more evident in the M-vehi group than in the S-vehi group (*
To evaluate the effect of administration of hAMSCs on general activities, motor function deficits, cognitive impairment, and depression-like symptoms in MCAO mice, we compared performance on the behavioral tests between MCAO mice who had administered hAMSCs (M-high and M-low groups) and the vehicle control (M-vehi). The open field test showed significant between-group differences for the M-high and M-vehi group in the sessions performed on day 1 and 3 (a
Taken together, these results indicate (i) that previously mentioned neurological and behavioral impairments, such as general activities, motor weakness, memory impairment, and depression-like symptoms, in the mouse model of MCAO were indeed induced by cerebral infarction, and (ii) that these deficits were improved by administering hAMSCs in a dose dependent manner 2 to 3 months after MCAO.
The Administration of hAMSCs Suppresses mRNA Expression and Protein Expression Levels of Pro-Inflammatory Cytokines Around the Infarcted Brain During the Early Phase of Ischemic Infarction
The significant differences in the scores in the behavioral tasks between the M-high and M-vehi groups led us to assess the underlying mechanisms of the amelioration of symptoms after the administration of hAMSCs. The mRNA expression levels of pro-inflammatory cytokines in the brain were assessed 7 days after the induction of MCAO (Fig. 2a).
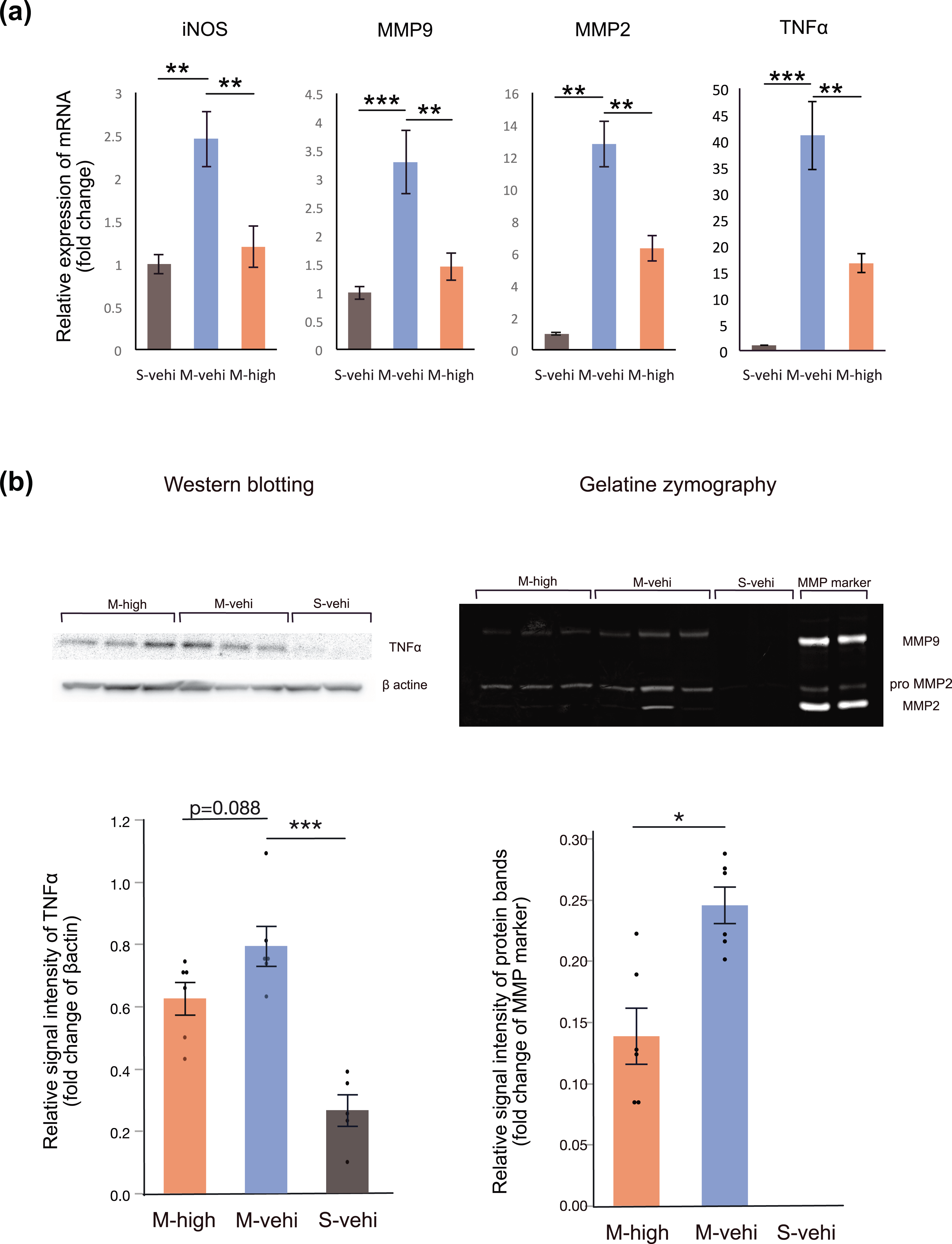
Administration of hAMSCs suppresses pro-inflammatory cytokines produced by immune cells following MCAO. We randomly assigned MCAO mice (
The relative mRNA expression levels of iNOS, MMP2, MMP9, and TNFα were significantly higher in the M-vehi group compared with the S-vehi group (
These data suggest that administering hAMSCs suppresses the mRNA expression levels and the amount of pro-inflammatory cytokines around the infarcted brain in the early phase of ischemic infarction.
After the Intravenous Administration of hAMSCs, Stem101-Positive Cells were Detected and the Infiltration of iNOS-Positive Pro-Inflammatory Immune Cells Was Suppressed in the Peri-Infarct Area
To investigate whether intravenously administered hAMSCs migrated into the brain parenchyma and to analyze the distribution of immune cells showing pro-inflammatory neurotoxic phenotypes in the peri-infarct area, frozen brain sections of mice in the M-high and M-vehi groups were assessed via immunohistochemistry 7 days after MCAO. In the frozen brain sections DAB-stained with the anti-stem101 antibody, which specifically reacted with the human nucleus protein Ku80 and can differentiate implanted human cells from mouse brain tissues, we identified stem101-positive cells around the peri-infarct area in M-high mice (Supplemental Fig. S1). Although it was difficult to verify the absence of stem101-positive cells in the M-vehi group due to the non-specific DAB staining characteristic of the ischemic core, stem101-positive cell counts around the peri-infarct area in the M-high group were significantly higher than those in the M-vehi group, which suggested that grafted stem101-positive hAMSCs migrated and survived in the the peri-infarct area of the mouse model at least 6 days after administration.
Fluorescent immunohistochemistry of CD11b/c showed an accumulation of CD11b/c positive immune cells in the peri-infarct area, suggesting the activation of macrophages and microglia. The distribution of iNOS-positive cells showed the same trend as the distribution of CD11b/c positive cells (Fig. 3a, b).
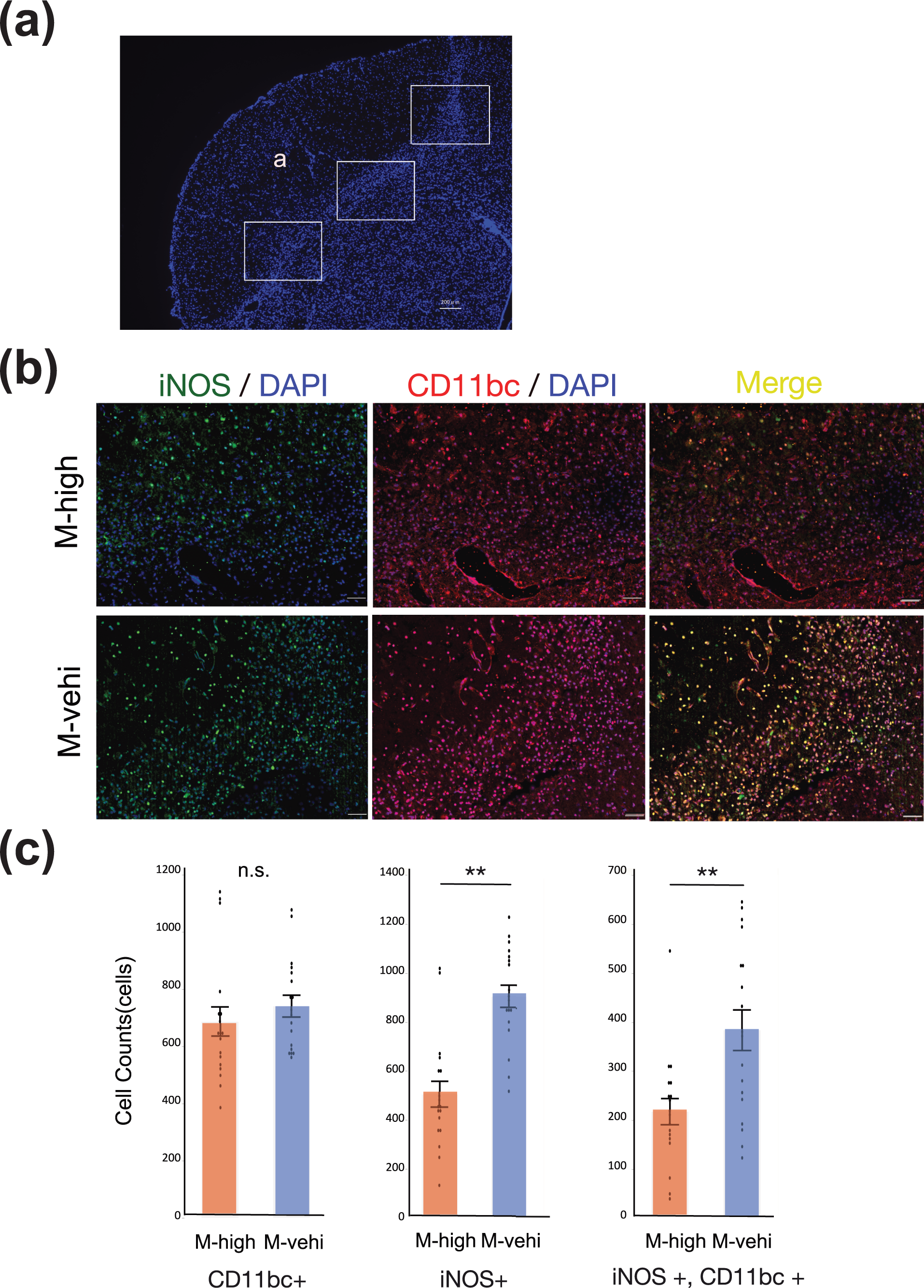
The administration of hAMSCs suppresses pro-inflammatory immune cells around the peri-infarct area. (a) Tissue section stained with 4′,6-diamino-2-phenylindole (DAPI) showing regions of analysis in the peri-infarct area ((a) ischemic core). Three random regions, indicated by solid boxes (200× magnification), per coronal section and two coronal sections per brain were analyzed to count iNOS+/CD11bc+ cells [M-high:
The numbers of iNOS-positive cells as well as iNOS and CD11 b/c colocalized cells, in the peri-infarct area of mice in the M-high group were significantly suppressed compared to mice in the M-vehi group (
These results suggest that the number of immune cells, including macrophages and microglia, along with pro-inflammatory and neurotoxic phenotypes, are suppressed by the intravenous administration, of hAMSCs, although the total number of immune cells was not altered.
Blood Brain Barrier Permeability is Suppressed by the Administration of hAMSCs
In order to investigate the association between BBB permeability and neuro-inflammation, we performedan EB assay 24 h after administering the high dose of hAMSCs. BBB disruption was determined by the leakage of the EB stain (dark blue) into ipsilateral brain tissues (Fig. 4a). The EB levels in brain tissue were significantly decreased in the M-high group compared to those in the M-vehi group (
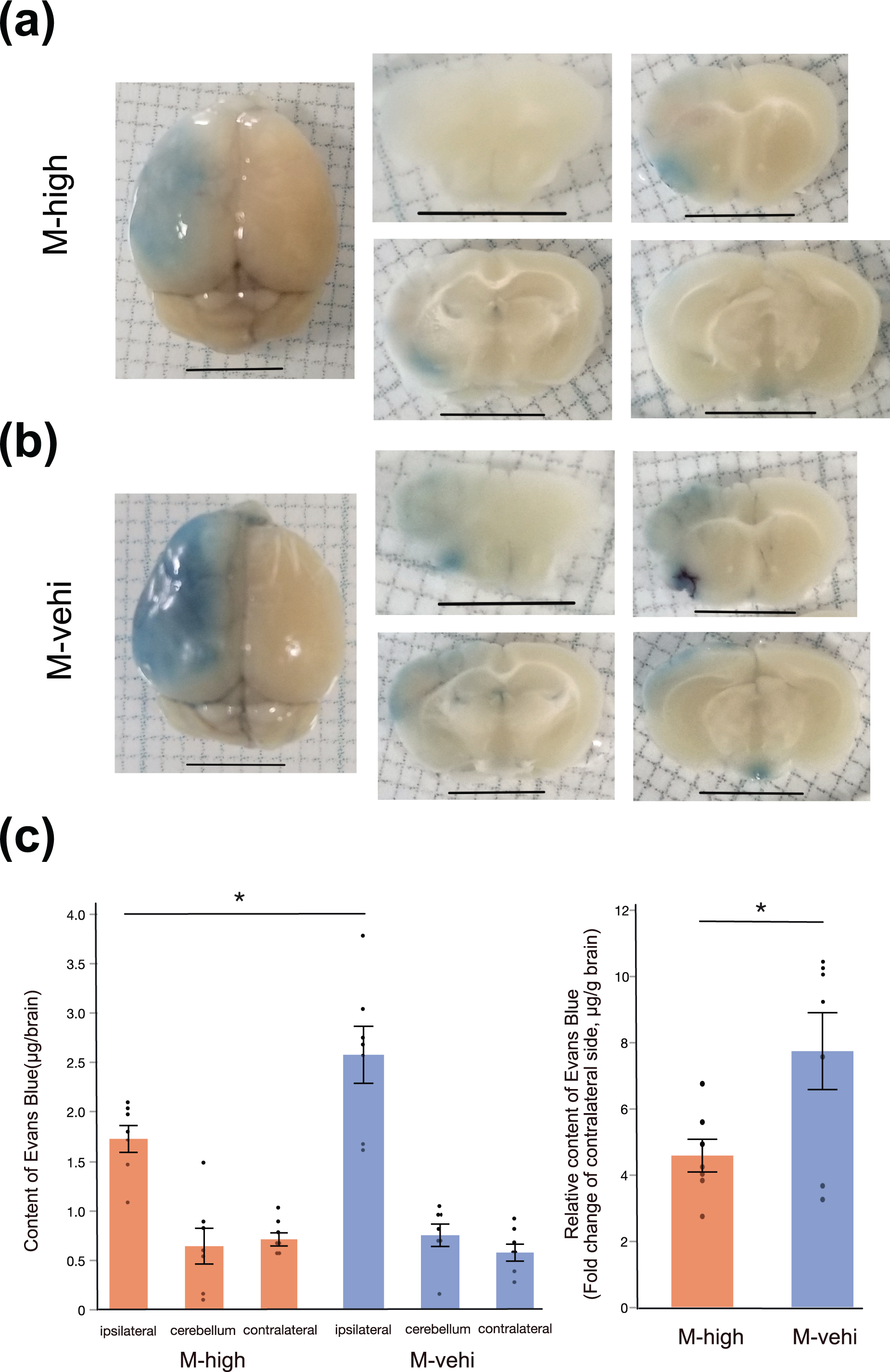
Evans Blue assay demonstrates the suppression of BBB permeability by the administration of hAMSCs. (a) Representative images of whole brain (left column) and coronal sections (middle and right columns). EB deposited in the brain parenchyma of the ischemic core and the peri-infarct area (middle cerebral artery territory of the left hemisphere), where the BBB was disrupted. Scale bar: 5 mm. (b) Quantitative analysis of EB content [M-high:
The Administration of hAMSCs Suppresses Delayed Neuronal Cell Death in the Peri-Infarct Area
Unlike necrosis at the ischemic core, which occurs within minutes following the onset of infarction, many neurons in the peri-infarct area undergo apoptosis after several hours or days following the onset of infarction. In fact, several reports have shown that neuronal death in the peri-infarct area and at the ischemic core are temporally and spatially different phenomena 36 –38 .
This prompted us to investigate the number of dying cells in the peri-infarct area at a relatively late phase of MCAO, namely 7 days following MCAO induction, using mice that were administered a high dose of hAMSCs on the day following MCAO.
While disappearance of neuronal cells and cellular debris without nuclei were mainly observed in the ischemic core, TUNEL staining revealed that the number of TUNEL-positive neurons in the peri-infarcted area was significantly lower in the M-high group compared to the in M-vehi group (
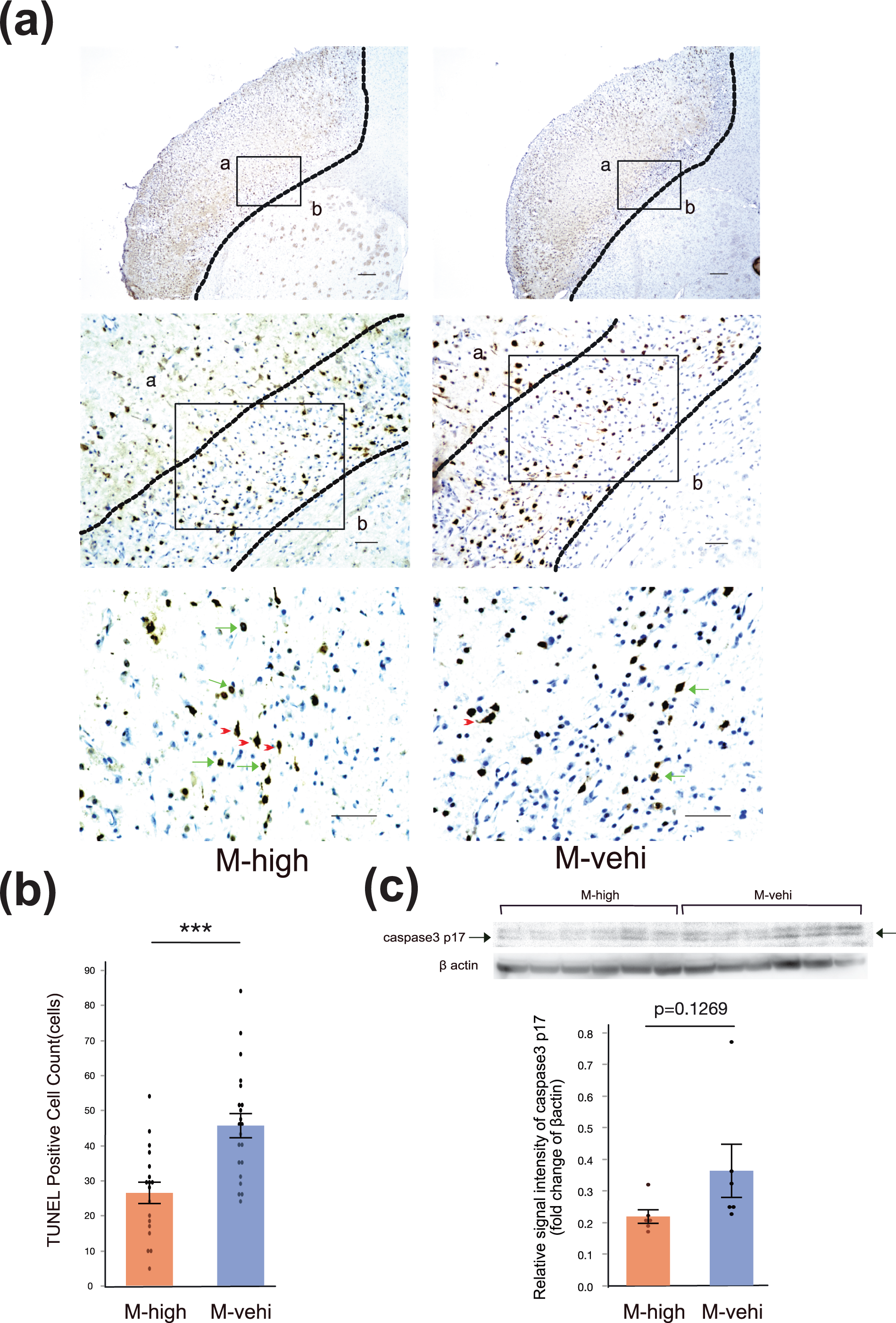
Administration of hAMSCs suppresses delayed neuronal apoptosis in the peri-infarct area. (a) Representative image of terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick end labelling (TUNEL) staining. Regions of analysis in the peri-infarct area are indicated by solid boxes (a: ischemic core, b: normal brain). Three random regions, indicated by solid boxes (400× magnification), per coronal section and two coronal sections per brain were analyzed to estimate the number of TUNEL positive cells [M-high:
These results suggest that the administration of hAMSCs suppresses delayed neuronal death in the peri-infarct area, particularly in relation with apoptosis.
Discussion
Standard reperfusion therapies, such as the use of recombinant tissue plasminogen activator and endovascular thrombectomy, have very narrow therapeutic time windows. Because neuro-inflammation is considered to play an important role in the pathophysiology of ischemic stroke, immunosuppressive treatments that target single molecular pathways that are activated during the acute and subacute phases of ischemic infarction have been attempted, but have failed to demonstrate sufficient effects 11,12 . This may partly be due to post-ischemic inflammation, which acts through multiple pathways, and also plays an important role in tissue repair, and so immunosuppressive treatments that inhibit inflammation could interfere with these repair processes 2 .
Cell therapy also targets the subacute phases of ischemic stroke, thereby preventing early secondary cell death by suppressing apoptosis and inflammation 14 . Cell therapy has the potential to suppress multiple injury mechanisms and facilitate the mechanisms of cerebral repairment and reorganization during the subacute and chronic phases via angiogenesis, neurogenesis, and synaptogenesis 14,21 .
Among the various transplantable cells, amniotic stem cells exhibit several features that make them an ideal donor source for cell therapy purposes 19 . Firstly, these cells are easily and less invasively obtained from amniotic membranes, which are usually discarded after birth, compared to bone-marrow derived and adipose derived MSCs. Secondly, unlike the uses of ES cells, there are no ethical barriers to the use of amniotic stem cells. Thirdly, they have low immunogenicity resulting in a lack of host response following xenotransplantation, thereby enhancing the suitability of these cells for allogeneic therapeutic application. Finally, there is no evidence of tumorigenicity following the transplantation of isolated human amniotic cells into animal models 19,21 .
The current study demonstrated that the expression levels of major pro-inflammatory cytokines, such as TNFα, iNOS, MMP2, and MMP9, were significantly decreased by the administration of hAMSCs to a mouse model of MCAO. These findings are substantiated by previous studies which have shown the expression levels of pro-inflammatory cytokines are suppressed by cell therapies 39,40 .
We analyzed MMP gelatinase activities in the MCAO mice and showed that the MMP9 activity was decreased by hAMSC administration whereas MMP2 gelatinase activity was unchanged. This may be partly due to MMP2 being associated with the reversible disruption of the BBB during the early phases of infarction, in contrast to MMP9, which is elevated in the late phase resulting in the loss of tight junctions and is therefore considered to be principally responsible for disruption of the BBB 41,42 . These findings are compatible with the results of our evaluation of BBB integrity via EB extravasation 43 .
We also demonstrated that the number of CD11b/c and iNOS double positive immune cells infiltrating the peri-infarct area, 7 days following MCAO, was significantly decreased by the hAMSC administration although there was no obvious difference in the number of CD11 b/c positive immune cells between the group administered hAMSCs and the vehicle control group.
CD11b is a phenotypic marker of macrophages, such as monocyte derived infiltrating macrophages and microglias (yolk-sac derived resident brain parenchymal macrophages), as well as meningeal and perivascular macrophages 44 . CD11c is expressed on granulocytes and dendritic cells as well as on macrophages 45 . However, iNOS is expressed in macrophages, neutrophils, and microglia in response to pro-inflammatory cytokines, and generates excessive NO, leading to the production of peroxynitrite, which exhibits neurotoxic action 46 . Its expression level is generally used to distinguish M1 pro-inflammatory macrophages/microglia from M2 anti-inflammatory macrophages/microglia 47 –49 .
Macrophages and microglia are known to have a spectrum of different but overlapping functional phenotypes, namely the M1 and M2 phenotypes 6 . Similar phenotypic differentiation is also observed in dendritic cells 50 . M1 macrophages typically release tissue-damaging pro-inflammatory mediators whereas M2 macrophages work as scavengers of cellular debris and produces various protective/trophic factors. A unique chronological phenotype shift from M2-dominant to M1-dominant is caused by the M2-to-M1 conversion within activated microglia/macrophages and the continuing recruitment of pro-inflammatory M1 microglia/macrophages following ischemic infarction 49 . Amnion derived stem cells reportedly limit M1 polarization and promote M2 polarization 51 . These results substantiate our findings showing that although the number of M1 phenotypic iNOS-positive immune cells was reduced by hAMSC administration, the total amount of immune cells including M2 phenotypic immune cells was not altered, suggesting that hAMSC administration suppresses the M2-to-M1 phenotypic shift during the subacute phase. This finding was also compatible with the results of a previous study of ours which investigated the effect of adipose-derived mesenchymal stem cells (ADSCs) on ischemic stroke, where the total CD11b+ cell counts in the subacute phase were not very different, but the M1 phenotype immune cells were found to be significantly higher in the vehicle control than in the group administered ADSCs 52 .
Finally, we evaluated the effect of hAMSCs on apoptosis in the peri-infarct areas. Despite differences in the routes used for administration (i.e., intracerebral, intravenous) and the time points of assessment (from 24 h to 21 d after transient MCAO), several studies have indicated that neuronal apoptosis around the ischemic infarction is suppressed by the administration of amniotic stem cells 39,43,53 –55 . Although TUNEL staining is not always a specific indicator of apoptosis 32 , when combined with typical changes in nuclear morphology, it can be utilized to distinguish the apoptotic cells from the necrotic ones. Our data on the peri-infarct areas, 7 days after MCAO, showed suppression of delayed cell death, with particular reference to apoptotic neuronal death, by hAMSCs, which substantiates previous findings. Western blotting of the caspase 3 p17 subunit also suggested that the administration of hAMSCs has a suppressive effect on activated apoptotic pathways, although this effect was not statistically significant. This may be because apoptotic neuronal death only accounts for a portion of total cell death, since necrotic and apoptotic cell deaths occur concomitantly in ischemic lesions, where the dominant cell death process depends on the relative speed of each process 33 . Apoptotic cell death in the peri-infarct areas appears to occur during the later phases, compared to necrosis in the ischemic core, which may enable the hAMSCs to extend the therapeutic window and be effective, even after the failure of reperfusion therapy for ischemic stroke.
Taken together, our findings indicate that suppression of pro-inflammatory cytokines and the M2-to-M1 phenotype shift, by administering hAMSCs intravenously, appears to inhibit BBB breakdown and apoptosis in the peri-infarct areas, resulting, in turn, in an improvement of neurological outcomes.
Compared to intravenous administration, the clinical use of intracranial injections has some limitations. Intracranial injection, which requires suitable imaging facilities and surgical expertise, may cause adverse effects such as periprocedural hemorrhage, surgical site infection, and brain damage leading to further BBB breakdown and inflammatory responses 21 . Moreover, most importantly, this approach does not target the systemic immune response that contributes to impaired outcomes following ischemic infarction 51 .
The relatively small diameter of amniotic stem cells (8–15 µm) probably reduces the likelihood of these cells clumping inside lung capillaries, and indeed, it has been suggested that amnion derived stem cells acutely administered intravenously to mice following the onset of cerebral ischemia underwent a significant migration to the spleen as well as the injured brain via stromal cell derived factor 1(SDF1)-CXCR4 signaling 54,56,57 . Additionally, in several reports, splenectomy before the induction of stroke has shown to reduce infarct size, as well as the number of activated mononuclear immune cells in the ischemic hemisphere, and to diminish the efficacy of intravenously administered amnion derived stem cells 54,58 .
Although there are challenges in differentiating monocyte-derived macrophages from resident brain parenchymal microglia and their relative degree of involvement in the M2-M1 phenotype shift in the brain, using a bone marrow chimeric technique in models of experimental autoimmune encephalomyelitis (EAE), Ajami et al. showed that significant monocyte recruitment into the brain was found only in animals that already displayed considerable disease activity and that progression to severe EAE was strongly correlated with the extent of this myelomonocytic infiltration 59 . Additionally M2 microglia (i.e. resident brain parenchymal macrophages) have been reported to show a greater tendency to maintain their M2 status than monocyte derived M2 macrophage in vitro 60 . Taken together, the spleen appears to play an important role in the acute influx of M1 phenotypic immune cells into the brain after stroke, which is at least partly the underlying mechanism of the M2-M1 phenotype shift in the brain that leads to disruption of the BBB, and might be a target for intravenously administered hAMSCs 61 , although it cannot be denied that hAMSCs directly suppress intraparenchymal inflammation in the brain and disruption of BBB leading to a reduction in acute M1 myelomonocytic infiltration. This emphasizes the importance of evaluating the efficacy of administering hAMSCs intravenously.
As confirmed by other studies 39,54 , we demonstrated that the subacute intravenous administration of hAMSCs (a day after MCAO induction), ameliorated long-term neurological deficits in a mouse model following cerebral infarction. Furthermore, for the first time we report that the effect of amnion derived mesenchymal stem cells is dose dependent. These findings could distinguish the cell therapy by intravenous administration of hAMSCs from conventional reperfusion therapies in the clinical setting, when considering that it could still be effective in the acute phase even after the failure of hyperacute conventional reperfusion therapies via definitely different mechanisms by suppressing apoptosis as well as neuroinflammation.
However, several previous studies have suggested that the delayed administration of hAMSCs after the stroke-induced brain injury has already established itself (1–3 d following the onset of stroke) may still effectively enhance long-term functional recovery 54 . This recovery seems to be partly due to the promotion of M2 macrophage polarization by the amnion derived stem cells 51 , further resulting in the release of anti-inflammatory cytokines (IL-10, IL-6) and growth factors, including angiogenic factors 20 , which assist in the brain repair process 62 . Moreover, it has been suggested that amnion derived stem cells may differentiate toward a neural lineage in vivo 21 . The current study is beset with certain limitations. One such limitation is that we did not assess whether the brain parenchyma or spleen was more important in terms of the target point for intravenously administered hAMSCs, even though we demonstrated the possibility that a certain number of stem101-positive hAMSCs migrated into the brain parenchyma. Another limitation is that the effects of hAMSCs on the anti-inflammatory, neuro-protective, and neuro-regenerative processes in the subacute and chronic phases were not evaluated.
In regard to future studies, it may be helpful to investigate the mechanisms underlying the differentiation of hAMSCs into a neuronal lineage, which strongly supports the need to administer the hAMSCs themselves, instead of conditioned medium obtained from hAMSCs.
Conclusion
In conclusion, the current study demonstrates that administering hAMSCs improves neurological deficits in a mouse model of ischemic stroke. The underlying mechanisms of such improvements appears to be the suppression of pro-inflammatory cytokines and the shift from a M2 to a M1 phenotype, resulting in a reduction in the breakdown of the BBB and reduced apoptosis in peri-infarct areas. The effects of administering hAMSCs on delayed and chronic phases, including anti-inflammatory, neuro-protective, and neuro-regenerative effects, warrant further investigation.
Supplemental Material
Supplemental Material, sj-docx-1-cll-10.1177_09636897211024183 - Intravenous Administration of Human Amniotic Mesenchymal Stem Cells in the Subacute Phase of Cerebral Infarction in a Mouse Model Ameliorates Neurological Disturbance by Suppressing Blood Brain Barrier Disruption and Apoptosis via Immunomodulation
Supplemental Material, sj-docx-1-cll-10.1177_09636897211024183 for Intravenous Administration of Human Amniotic Mesenchymal Stem Cells in the Subacute Phase of Cerebral Infarction in a Mouse Model Ameliorates Neurological Disturbance by Suppressing Blood Brain Barrier Disruption and Apoptosis via Immunomodulation by Yasunori Yoshida, Toshinori Takagi, Yoji Kuramoto, Kotaro Tatebayashi, Manabu Shirakawa, Kenichi Yamahara, Nobutaka Doe and Shinichi Yoshimura in Cell Transplantation
Footnotes
Availability of data and materials
The authors confirm that all data underlying the findings are fully available without restriction. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors Contribution
YY conducted experiments, analyzed the data, and drafted the manuscript. TT participated in experimental design and the manuscript revision. YK, KT and MS participated in experimental design, data analysis and interpretation, and manuscript preparation. KY isolated, expanded, and formulated the hAMSCs as an investigational drug. ND made substantial contributions to the analysis of behavioral tasks. SY made substantial contributions to the study conception and design, approved the final version of the article to be published, and agreed to be accountable for all aspects of the work. All authors have read and approved the final manuscript.
Acknowledgements
The authors thank Yuki Takeda and Makiko Murase for supporting our experiments. The authors also thank the staff in Joint-Use Research facilities, Hyogo College of Medicine, for allowing us to use their resources, such as quantitative PCR equipment.
Ethical Approval and Consent to Participate
The study protocol was approved by the Animal Care Committee of Hyogo College of Medicine (Approval number: 17-034).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the ‘Guide for the Care and Use of Laboratory Animals’ published by the National Academy of Science of the USA. All fetal membranes from human donors were obtained according to the protocol approved by our institutional ethic committee (approval number 325).
Statement of Informed Consent
Written informed consent was obtained from the donors of fetal membranes for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grant-in-Aid for Scientific Research (C) [Grant Number 18K16601].
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
