Abstract
The instant blood-mediated inflammatory reaction (IBMIR) is a major barrier to successful intraportal islet engraftment. Bivalirudin, a direct thrombin inhibitor, could mitigate IBMIR by suppressing thrombin activity and platelet activation, yet its role in islet transplantation remains insufficiently defined. Using a portal vein islet transplantation model with rat islets, we compared bivalirudin treatment (BT) with heparin treatment (HT). In recipient Wistar rats, both BT and HT increased circulating antithrombin and prolonged activated partial thromboplastin time (APTT) versus saline. BT more effectively reduced serum β-thromboglobulin (β-TG) and thrombin–antithrombin (TAT) complexes, and it decreased peri-graft fibrin deposition to a greater extent than HT. BT and HT also attenuated hepatic NF-κB activation, lowered serum TNF-α, and diminished inflammatory-cell infiltration within grafts, with more pronounced effects under BT. In BALB/c-nu recipient mice, BT and HT similarly reduced graft inflammatory infiltration. Functionally, a higher proportion of diabetic recipients achieved normoglycemia after BT or HT than after saline, and BT yielded a greater normoglycemia rate with a shorter time-to-normoglycemia than HT. Collectively, BT and HT alleviate coagulation activation and inflammatory infiltration characteristic of IBMIR, thereby improving post-transplant islet survival. BT demonstrated greater anti-thrombotic and anti-inflammatory activity than HT in this setting, supporting bivalirudin as a promising adjunct anticoagulant strategy for intraportal islet transplantation and potentially other cell-therapy applications.

Keywords
Introduction
Islet transplantation can restore endogenous insulin secretion and improve glycemic control in selected patients with diabetes 1 . However, long-term outcomes remain suboptimal. A principal barrier is the instant blood-mediated inflammatory reaction (IBMIR) that follows intraportal islet infusion2–4. Triggered by direct contact between islets and blood, IBMIR rapidly activates the coagulation and complement cascades, induces platelet activation, recruits inflammatory cells, and promotes fibrin deposition around the graft5,6. These events lead to perivascular microthrombi and local inflammation, destroying up to ~60% of transplanted islet cells within hours and compromising engraftment7,8. Although unfractionated heparin is commonly used to attenuate IBMIR, it may paradoxically augment platelet and complement activation, carries a clinically relevant bleeding risk, and has been reported to inhibit angiogenesis9,10. By contrast, bivalirudin—a direct thrombin inhibitor—does not impair vascular endothelial growth factor signaling and has been used in systemic anticoagulation without hindering tissue growth or repair11,12. Because robust revascularization is essential for durable islet function, there is a compelling need for anticoagulation strategies that mitigate IBMIR while preserving angiogenesis. Evaluating bivalirudin as an alternative to heparin may, therefore, provide a safer and more effective approach to enhance islet engraftment and improve long-term transplant outcomes.
Bivalirudin is a direct thrombin inhibitor that exerts anticoagulant effects by selectively engaging the enzyme’s catalytic site, resulting in rapid onset, reversible inhibition, and a comparatively low bleeding liability. Clinical analyses in elderly patients with acute myocardial infarction undergoing percutaneous coronary intervention have shown that, relative to unfractionated heparin, bivalirudin improves net clinical outcomes by simultaneously optimizing coagulation profiles, reducing bleeding complications, and lowering circulating inflammatory markers13,14. In extracorporeal membrane oxygenation (ECMO) settings, meta-analytic data further suggest that, compared with heparin, alternative bivalirudin can be associated with lower risks of thrombosis, in-hospital mortality, and bleeding, with corresponding gains in ECMO survival15,16. Moreover, bivalirudin suppresses thrombin- and collagen-induced platelet activation in the absence of antithrombin III, underscoring its antithrombin-independent mechanism and addressing a key limitation of heparin-based strategies 17 . Despite its broad adoption in cardiovascular medicine and interventional practice, the role of bivalirudin in islet transplantation remains insufficiently characterized, and systematic experimental evidence is lacking. Rigorous preclinical and clinical studies are, therefore, warranted to define its efficacy and safety in mitigating IBMIR and improving long-term graft function after intraportal islet infusion. Although the protective role of bivalirudin treatment (BT) in cardiovascular disease and interventional therapy has been widely recognized, it is still unclear whether BT protects intrahepatic islet transplants that mimic clinical islet transplantation environments. The focus of this study is to evaluate the effect of BT on IBMIR-induced pancreatic islet death and understand the molecular mechanisms of anticoagulant and anti-inflammatory effects of BT.
Although bivalirudin is well established in cardiovascular and interventional settings, its capacity to protect intraportal transplanted islets within a clinically relevant intrahepatic milieu remains undefined. This study was designed to test whether BT mitigates IBMIR-associated early islet loss and to elucidate the molecular underpinnings of its anticoagulant and anti-inflammatory actions. Specifically, we evaluate the impact of BT on IBMIR-driven coagulation and inflammatory responses in an intraportal islet transplantation model that recapitulates clinical conditions, with the goal of clarifying its therapeutic potential for improving graft survival and function.
Materials and methods
Animals and reagents
Male Wistar rats aged 5 to 6 weeks, male Sprague Dawley (SD) rats aged 6 to 8 weeks, and BALB/c-nu mice aged 7 to 8 weeks were purchased from Changsha Tianqin Biotechnology Co., Ltd. (Changsha, China). The rats and mice were acclimatized and fed in a specific pathogen-free facility for 1 week before being used in this experiment. All experimental procedures were carried out strictly in accordance with the guidelines for the care and use of laboratory animals at Guangxi University of Chinese Medicine, and were approved by the Laboratory Animal Welfare and Ethics Committee of Guangxi University of Chinese Medicine (Approval Number: DW20231115-263). Bivalirudin was purchased from Ruiweier (Changsha, China) and mixed with normal saline to form an injection solution, and heparin sodium for injection was purchased from Shanghai Pharmaceuticals Holding Co. Ltd.
Islet isolation and counting
The islet isolation method is as described previously 18 . In brief, SD male rats were euthanized, and then an abdominal laparotomy was performed. And then, 8 ml of collagenase V (1 mg/ml, Sigma, MO, USA) was injected into the pancreas through the common bile duct. The enlarged pancreas was removed, and the conical tubes were stirred in a 37°C water bath for 5 to 6 min for digestion. Subsequently, the isolated islet cells were purified by density gradient centrifugation using sample density separation solution (TBD, China) and Histopaque 1077 (Sigma, USA). The islets were further purified by manual harvesting and cultured in RPMI-1640 (Servicebio, Wuhan, China) medium containing 10% fetal bovine serum at 37°C in a humidified atmosphere with 5% CO2. The islets were observed under an optical microscope, and the islet equivalent (IEQ) was determined by counting the number of islets stained with DTZ.
Diabetes rats induced and islet transplantation
Diabetes was induced in Wistar rats by a single intraperitoneal injection of streptozotocin (70 mg/kg, Beyotime, China) and in BALB/c-nu mice by a single intraperitoneal injection of streptozotocin (200 mg/kg, Beyotime). Three days later, blood glucose levels were monitored through the tail vein using a glucose monitoring system (Sinocare, Changsha, China). Only the receptors with blood glucose levels higher than 300 mg/dl for two consecutive days were selected as recipients19,20.
The diabetes recipients were anesthetized by continuous inhalation of isoflurane (Rayward, Shemzhen, China), the inhalation dose of which was 2 ml/min for Wistar rats and 1 ml/min for BALB/c-nu mice, respectively, and then the anesthesia state was maintained by inhalation of isoflurane (1 ml/min for rats, 0.5 ml/min for nude mice). The receptor was placed supine on the experimental platform, and its abdominal wall was disinfected. Then, an appropriate amount of lidocaine was injected into the midline of the receptor’s abdomen for local anesthesia. Then, the upper abdomen was cut open to expose the superior mesenteric vein and portal vein. In the Wistar rat receptor, the superior mesenteric vein was punctured into the liver using a 27G syringe, and pancreatic islet cells (2000IEQ/100 g) were injected into the liver through the syringe. 21 . Similarly, in the BALB/c-nu mouse, 300 islets were injected into the liver using a 30G syringe. The sham group received saline implants. For the administration method, 24G intravenous infusion was administered through the tail vein in Wistar rats; 26G intravenous infusion was administered via tail vein in BALB/c-nu mice22,23.
The grouping of Wistar rat receptors is as follows (n = 8): sham surgery group: 0.5 ml of physiological saline was injected intravenously before intraportal islet transplantation (BIT), followed by infusion of 5 ml (kg · h) of physiological saline until 1 h after intraportal islet transplantation (AIT); model group: 0.5 ml of physiological saline was injected BIT, followed by 5 ml (kg · h) of saline infusion until 1 h AIT; BT group: 50 mg/kg bivalirudin injection (0.5 ml) was injected BIT, followed by bivalirudin injection at a rate of 70 mg/(kg · h) until 1 h AIT; 12 and heparin treatment (HT) group: heparin sodium for injection (0.5 ml) was injected at a dose of 200U/kg BIT, and then heparin sodium for injection was infused at a rate of 300U/ (kg · h) through until 1 h AIT.24,25.
The grouping of BALB/c-nu mouse receptors is as follows (n = 8): model group: 50 μL of physiological saline was intravenously injected BIT, followed by 5 ml (kg · h) of saline infusion until 1 h AIT; BT group: 80 mg/kg bivalirudin injection (0.5 ml) was injected BIT, and then bivalirudin injection was injected at a rate of 120 mg/ (kg · h) until 1 h AIT;26,27 and HT group: heparin sodium for injection (50 μl) was injected at a dose of 400U/kg BIT, and then heparin sodium for injection was infused at a rate of 500U/ (kg · h) through until 1 h AIT12,26.
Anticoagulation function assessment
After anesthetizing the rats with isoflurane, blood was collected through the inferior vena cava venipuncture using a 3.2% trisodium citrate solution in a 9:1 ratio. The plasma was separated by centrifugation at 3000×g for 20 min. According to the manufacturer’s protocol, the activity of each group was analyzed using the colorimetric anti-factor IIa and anti-Xa assays (Valukine, Shanghai). The coagulation functions of the rats in each group were measured using the Automatic Coagulation Analyzer (RAC-1830; Rayto, Shenzhen, China), including activated partial thromboplastin time (APTT) and prothrombin time (PT).
ELISA assay
According to the manufacturer’s instructions, the concentrations of thrombin–antithrombin (TAT), β-thromboglobulin (β-TG), and tumor necrosis factor (TNF)-α in the receptor serum were determined using a specific ELISA kit (Fine Tset, Wuhan, China). All values were measured by absorbance spectrophotometry at 450 nm using a microplate reader (Infinite 200 PRO; TECAN, Switzerland).
Immunohistochemical staining
The rat liver tissue was harvested 3 h AIT and the mouse liver tissue was harvested 6 h AIT. They were fixed with paraformaldehyde, then dehydrated, followed by xylene clearing. The tissue was embedded in paraffin and cut into 5 μm thin sections using a microtome. The paraffin sections were dehydrated, hydrated in xylene, stained with hematoxylin-eosin (HE), sealed, and sections were prepared.
For immunohistochemical staining, anti-insulin (ab181547; Abcam, Cambridge, UK), anti-CD11b (ab133357; Abcam), anti-fibrin (A31837; Nature Biosciences), anti-Ly6G (GB11229; Servicebio), and anti-F4/80 (GB11027; Servicebio) were incubated overnight at 4°C, and then stained with BAD working solution. The pathological changes of liver tissue were observed under a microscope (EVOS XL Core, Thermo Fisher, MA, USA) and images were captured.
Western blot analysis
After removing the entire liver tissue, it is immediately frozen in liquid nitrogen, then ground into a uniform powder, and the total protein is extracted to ensure that the analyzed protein can represent the expression of the entire liver. According to the manufacturer’s guidelines, total protein was extracted using the RIPA lysis and extraction buffer (AIWB-012; Affinibody, Wuhan, China). After electrophoresis and membrane transfer, the membranes were incubated in specific antibodies targeting fibrin (1:1000, A31837; Nature Biosciences), NF-κB (1:500, AF0246; Beyotime), and p-NF-κB (1:500, AF5881; Beyotime) at 4°C overnight. Then, they were incubated with horseradish peroxidase–labeled secondary antibodies for 2 h. Finally, the membrane was detected using the automatic chemiluminescence image analysis system (Clinx, Shanghai, China) and GAPDH was used as the internal control.
Statistics
Graft survival was plotted using GraphPad Prism 10.1.2. and differences were compared by a log rank test. The data were tested for normal distribution using the D’Agostino and Pearson combined normality test. For normally distributed data, one-way analysis of variance (ANOVA) was used to compare the differences among three or more groups; Otherwise, we utilize Kruskal–Wallis test. All statistical calculations were performed using GraphPad Prism software version 10.1.2.
Results
Effect of BT on inhibiting thrombin activity
We first verified thrombin selective target engagement by BT in Wistar recipients. At 1 h after intraportal islet infusion 1 h AIT, plasma anti-IIa activity was significantly higher in the BT group than in the model group and HT group (Fig. 1a), consistent with potent thrombin inhibition. Because no additional BT or HT was administered beyond 1 h AIT, anti-IIa activity subsequently declined in both treatment arms (Fig. 1a).
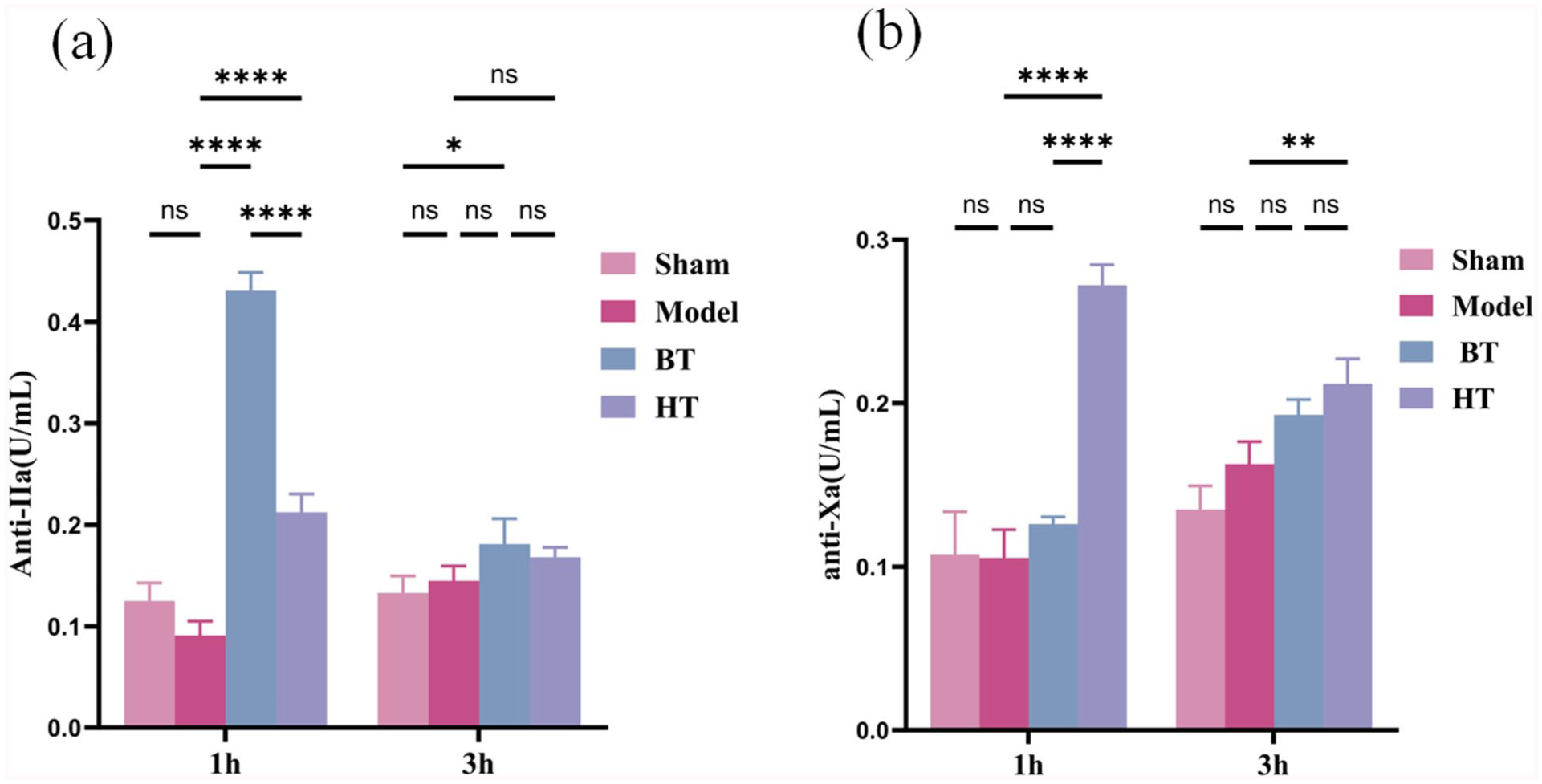
BT can significantly inhibit thrombin activity. (a) The anti-IIa activity of the BT group (n = 4) was the highest at both (model group: n = 4, HT group: n = 4) 1 h and 3 h. (b) The anti-Xa activity of the HT group (n = 4) was the highest at both (model group: n = 4, HT group: n = 4) 1 h and 3 h AIT (The differences between the above groups were analyzed by one-way ANOVA. ****P < 0.0001, **P < 0.01, *P < 0.05).
Mechanistically, HT produced a stronger inhibition of factor Xa12,13, whereas BT exerted minimal anti-Xa activity during dosing (Fig. 1b), in line with their distinct modes of action. After treatment cessation at 1 h AIT, both anti-IIa and anti-Xa activities trended downward toward baseline in the respective groups (Fig. 1b).
Effect of BT on coagulation time
Pharmacologic inhibition of thrombin (factor IIa) and factor Xa is expected to prolong the activated APTT12,28,29. We, therefore, measured APTT and PT at 1 and 3 h AIT in Wistar recipients. In the saline model group, the 1 h APTT was shorter than the rat reference interval (15–18 s), consistent with a hypercoagulable state (Fig. 2a). In contrast, both BT and HT significantly prolonged APTT at 1 h AIT, with a larger effect under BT; values in both treatment arms exceeded the reference range, yet no intraoperative bleeding events or bleeding-related deaths occurred. By 3 h AIT, APTT values declined across all groups; the BT group had returned to the physiological range (Fig. 2a). PT showed no significant changes at either 1 or 3 h AIT in any group (Fig. 2b).

BT can prolong APTT with almost no impact on TT. (a) The APTT of the BT group (n = 4) was the longest at 1 h AIT, and the APTT of the BT group was longer than that of the model group (n = 4) and the HT group (n = 4) at 3 h AIT. (b) There was no statistical difference in PT among the model group, BT group, and HT group. (The area between the two black dotted lines in this graph represents the normal value range of APTT and PT. The differences between the above groups were analyzed by one-way ANOVA. ****P < 0.0001, ***P < 0.001, **P < 0.01, *P < 0.05).
Effect of BT on inhibiting platelet activation and reducing TAT and fibrin deposition in the rat receptor liver tissue
β-TG is a specific protein secreted by platelets and is a specific indicator of platelet activation 25 . We found that the concentration of β-TG in the plasma of the NS group was significantly higher than that of the sham group, while the concentration of β-TG in the plasma of the BT group was lower than that of the NS group and the HT group (Fig. 3a) in Wistar rats. After the withdrawal of BT, that is, at 3 h AIT, there was no statistically significant difference in the β-TG concentration between the BT group, the HT group, and the NS group (Fig. 3a).
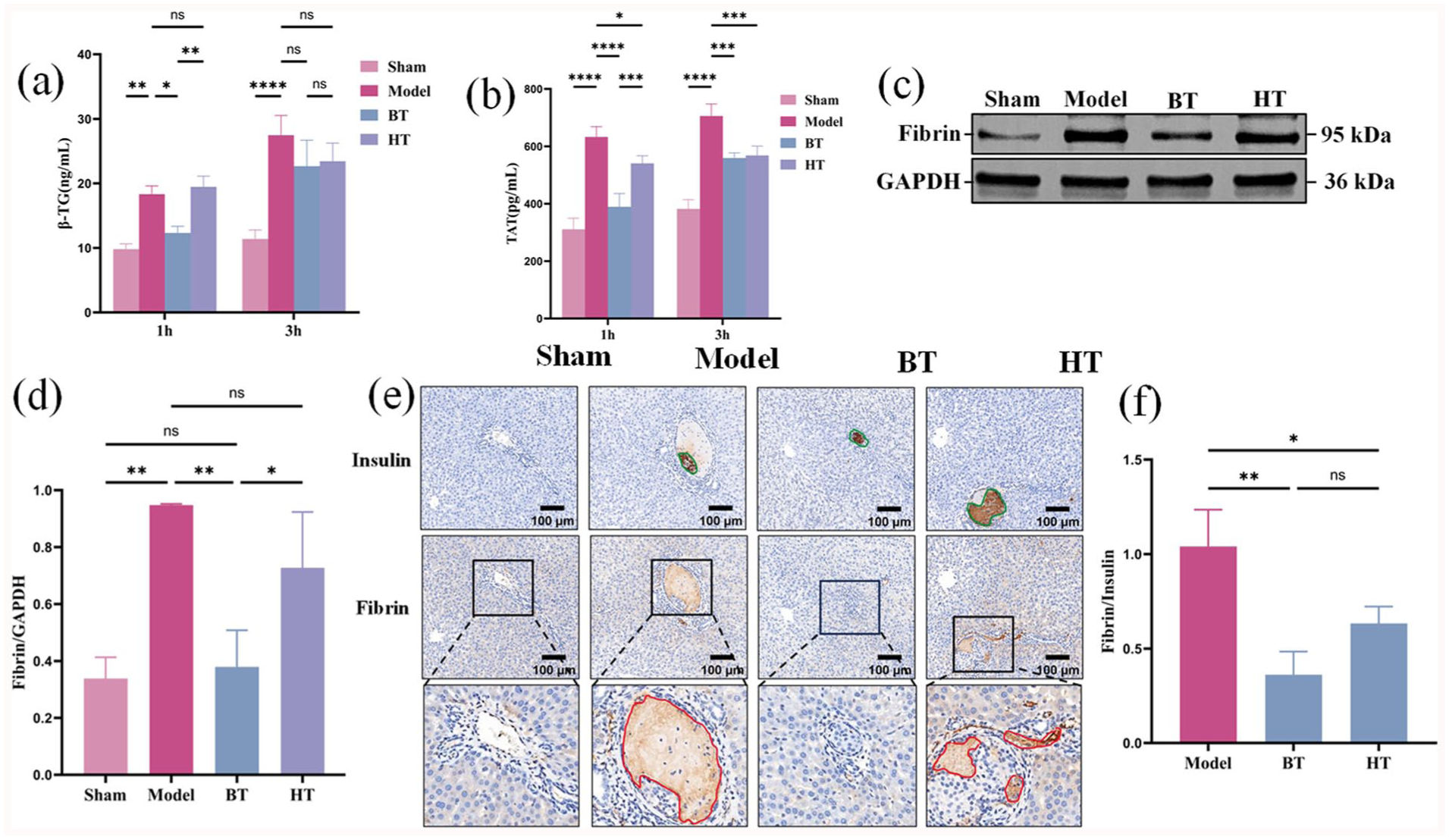
BT is more effective than HT in inhibiting platelet activation and reducing TAT and fibrin deposition in islet grafts. (a) The plasma β-TG activation in the BT group at 1 h and 3 h AIT were lower than those in the model group and the HT group. (b) The plasma TAT levels in the BT group at 1 h and 3 h AIT were lower than those in the model group and the HT group. (c) Western blot analysis was used to detect the representative immunoblots of fibrin and GAPDH in the liver tissue of each group at 3 h AIT. (d) The ratios of fibrin to GAPDH in the western blot analysis were quantified using the density measurement method. (e) Immunohistochemical staining was used to detect representative microscopic images of insulin and fibrin in the liver of 3 h AIT (200× magnification). Serial paraffin sections of an islet graft were stained with anti-insulin and anti-fibrinogen. Green line outlines pancreatic islet transplant based on insulin staining. The red line outlines fibrin deposition on islet graft. (f) The average optical density value of fibrin immunohistochemical staining was divided by the density value of insulin grafts in the corresponding group by using Image-Pro Plus 6.0. 56 Model islets, 55 BT islets and 91 BT islets. (The differences between the groups in Fig. 3a, b, d were analyzed by one-way ANOVA. The differences between the groups in Fig. 3f were analyzed by Kruskal–Wallis test. ****P < 0.0001, ***P < 0.001, **P < 0.05, *P < 0.05.).
TAT is a sensitive indicator of coagulation activation5,30. We found that BT effectively inhibited the increase in plasma TAT levels 1 h and 3 h after AIT, and was more effective than HT (Fig. 3b). We also measured the fibrin deposition, which is usually triggered by the activation of thrombin and is harmful to pancreatic islet grafts. As shown in Western blot analysis, the liver tissue in the BT group showed a significant reduction in fibrin expression 3 h AIT (Fig. 3c, d) compared with the model group. Although the liver fibrin expression in the HT group was relatively lower than that in the model group, there was no statistical difference between the two groups (Fig. 3d). For the immunohistochemistry of liver tissue fibrin, an apparent decrease of fibrin staining was observed in islet grafts from the BT group compared with grafts from the model group 3 h AIT (Fig. 3e, f). These data further confirmed the anti-thrombotic effect of BT in IBMIR.
Effective of BT on reducing NF-κB activation and inflammatory infiltration in liver tissue
The activation of NF-κB was investigated in liver tissue harvested 3 h AIT (Fig. 4a). A significant inhibition of NF-κB activation was observed in the liver tissue of rats treated with bivalirudin (Fig. 4b) compared with the model group (Fig. 4b). Although HT could also inhibit NF-κB activation, there was no statistical difference compared with the NS group. Notably, the inhibition of NF-κB activation was more significant in the BT group compared with the HT group (Fig. 4b).
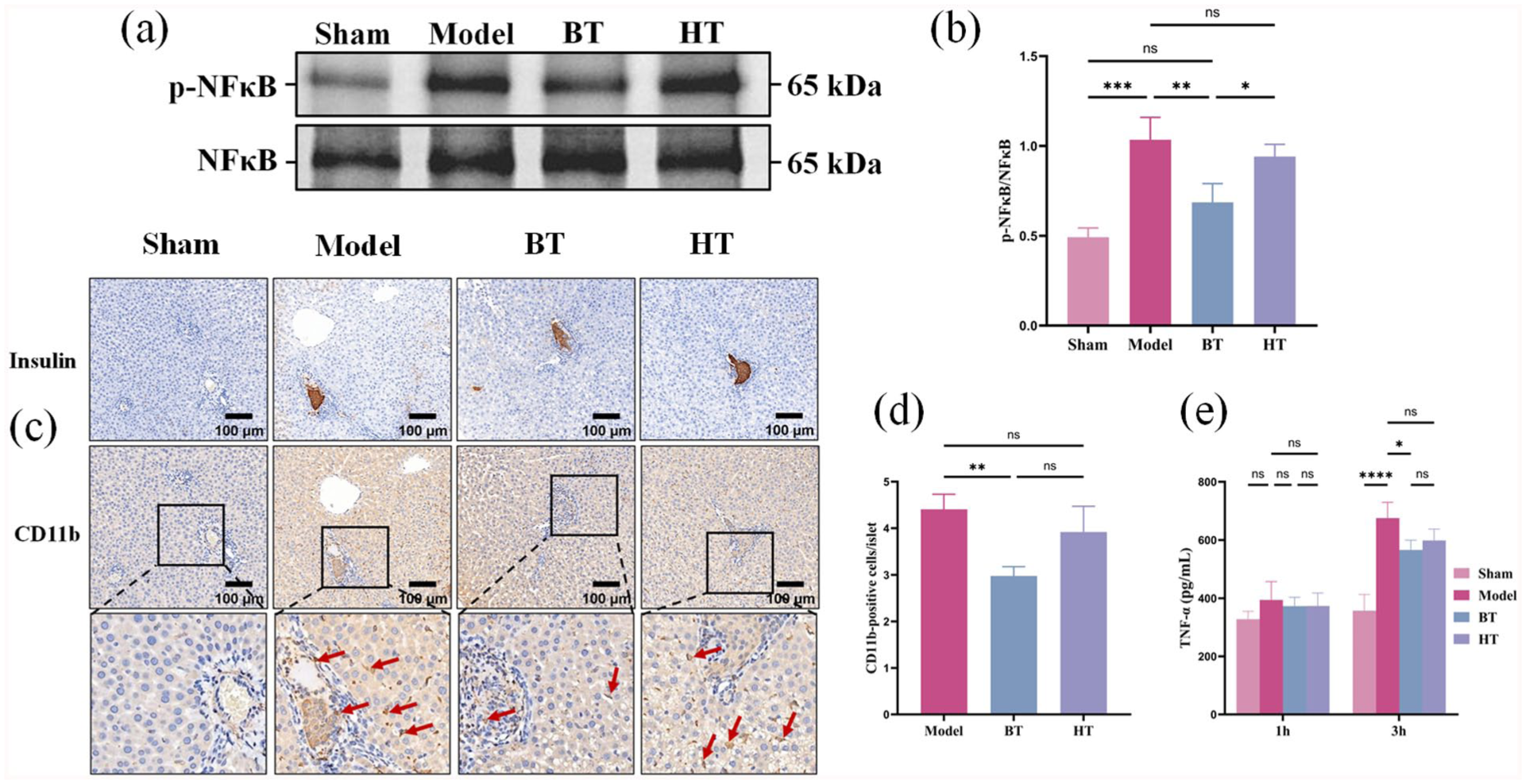
BT is more effective than heparin in reducing the activation of NF-κB and inflammatory infiltration in liver AIT. (a) Representative western blot of NF-κB and p-NF-κB in liver tissue 3 h AIT. (b) The ratios of p-NF-κB to NF-κB were quantified using the density measurement method. (c) The plasma levels of TNF-α by ELISA at 1 h and 3 h AIT. (d) Immunohistochemical staining was used to detect representative microscopic images of CD11b in the liver of 3 h AIT (200× magnification). (e) Numbers of inflammatory cells within the islet were lower in the BT group compared with those from NS group and HT group 3 h AIT. 55 Model islets (average size: 2,443.18 ± 327.52 µm2), 50 BT islets (average size: 2,050.65 ± 60.83 µm2) and 49 BT islets (average size: 1838.82 ± 137.49 µm2) were counted. The differences between these groups were analyzed by Kruskal–Wallis test.
One of the main characteristics of IBMIR after portal vein transplantation is that inflammatory cells are recruited to the site of islet implantation 31 . BT can indirectly reduce the infiltration of inflammatory cells around the islet graft by inhibiting thrombin. To verify our hypothesis, liver tissue samples from the recipients were collected 3 h AIT for histological analysis.
We observed that there was a significant infiltration of inflammatory cells around the grafts in the model group, which was consistent with previous similar reports31,32. BT significantly reduced the infiltration of inflammatory cells into the islets compared with the model group (Fig. 4c, d). Although HT also reduced the infiltration of inflammatory cells into the islets, the difference was not statistically significant compared with the model group (Fig. 4c, d).
TNF-α is one of the key regulatory factors in IBMIR and plays a crucial role in post-transplant inflammation and pancreatic islet cell death33–35. We further measured the TNF-α levels in the serum of each group. There was no statistically significant difference in the serum TNF-α levels among the groups of rats 1 h AIT (Fig. 4e). However, the TNF-α level in the model group significantly increased, and both BT and HT reduced the serum TNF-α level 3 h AIT (Fig. 4e).
Effect of BT on functional outcome of intrahepatic islet grafts
We further studied the protective effect of BT in the xenogeneic islet transplantation model. In this model, the marginal mass islets (n = 300–350) of SD rats were infused into the liver of the nude mouse diabetes receptor. At 15 days post-transplantation, only 3 in 8 (37.5%) of mice in the model group reached normoglycemia, compared with 6 in 8 (75%) in the BT group (n = 10, P = 0.05 vs. Model, log rank test, Fig. 5a) and 4 in 8 (50%) in the HT group (n = 8, P = 0.43 vs. Model). In mice with normoglycemia, average days needed to reach normoglycemia was 11.3 ± 1.2 days in Model, 8.0 ± 1.6 days in BT and 9.7 ± 1.0 days in HT groups, respectively (Fig. 5b). At 14 days post-transplantation, we performed IPGTT in BT mice, HT mice, and controls that were normoglycemic.

BT and HT improve islet survival and function in a xenogeneic mouse intraportal islet transplantation model. (a) Kaplan-Meier analysis shows significantly higher percentages of post-transplantation normoglycemia in diabetic mice that received BT or HT. (b) AAT treated mice reached normoglycemia faster than model mice and HT mice. (c) IPGTT shows that BT enhances blood glucose disposal 14 days AIT. (d) Area under the curve (AUC) of IPGTT.
Mice that received BT showed faster glucose clearance (Fig. 5c) and a significantly smaller area under the curve (AUC) after the glucose challenge compared with model (Fig. 5d). However, there was no significant difference between the HT group and the model group. These data provided strong evidence that BT is more effective in improving graft survival and function in the intrahepatic islet transplantation model.
BT inhibit platelet activation, plasma TAT levels and fibrin deposition in the BALB/c-nu mouse receptor
We measured the β-TG levels and TAT levels in plasma 6 h AIT in order to verify the anticoagulant effect of BT in BALB/c-nu mice. We found that β-TG levels in the plasma of the BT group was significantly lower than that of the model group and HT group (Fig. 6a). BT effectively suppressed the elevated plasma TAT level observed in the model group within 6 h AIT (Fig. 6b).
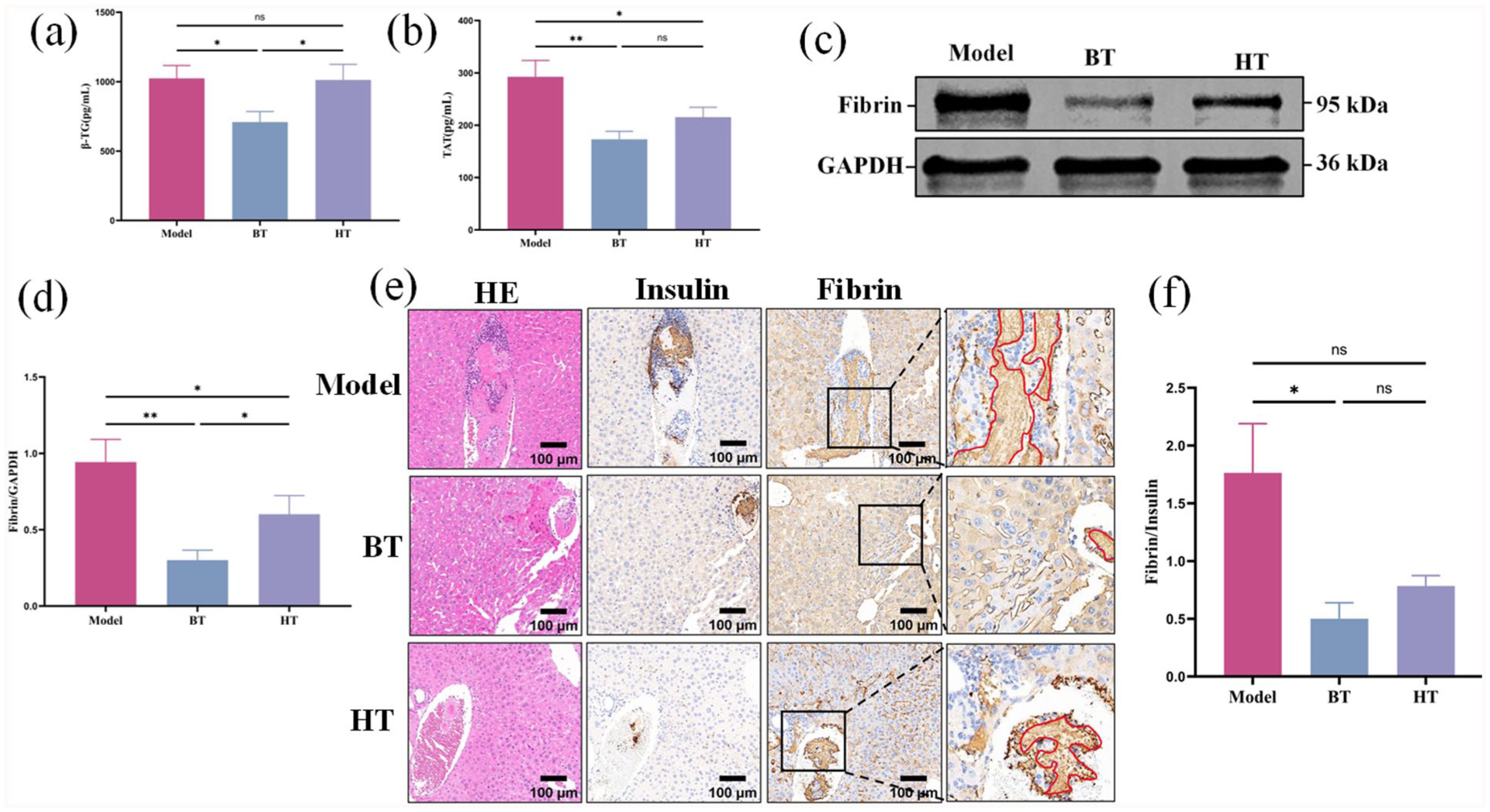
Effect of BT on anti-thrombotic in BALB/c-nu mice 6 h AIT. (a) The plasma β-TG level in the BT group was lower than those in the model group and the HT group. (b) The plasma TAT level in the BT group was lower than those in the model group and the HT group. (c) Western blot analysis was used to detect the representative immunoblots of fibrin and GAPDH in the liver tissue of each group at 6 h AIT. (d) The ratios of fibrin to GAPDH in the Western blot analysis were quantified using the density measurement method. (e) Immunohistochemical staining was used to detect representative microscopic images of insulin and fibrin in the liver of 6 h AIT (×200). Serial paraffin sections of an islet graft were stained with anti-insulin and anti-fibrin. The red line outlines fibrin deposition on islet graft. (f) The average optical density value of fibrin immunohistochemical staining was divided by the density value of insulin grafts in the corresponding group by using Image-Pro Plus 6.0. 143 Model islets, 74 BT islets and 64 BT islets. (The differences between the groups in Fig. 6a, b, d were analyzed by one-way ANOVA. The differences between the groups in Fig. 6f were analyzed by Kruskal–Wallis test. **P < 0.05, *P < 0.05.).
We also measured the generation of fibrin deposition, which is normally caused by the activation of thrombin, and is detrimental to islet grafts. As shown by Western blot and immunohistochemistry, an apparent decrease of fibrin expression was detected in the liver tissue and fibrin staining was observed in islet grafts from the BT group compared with grafts from the model group 6 h AIT (Fig. 6c, d, e). These data further confirmed the anti-thrombotic effect of BT in IBMIR.
Effect of BT on neutrophil and macrophage infiltration in BALB/c-nu mouse receptor
One of the major characteristics of IBMIR after intraportal transplantation is the recruitment of neutrophils and macrophages to sites of islet engraftment 36 . To test our hypothesis that BT protects islet grafts from IBMIR, livers of recipients were collected 6 h AIT for histological analysis. We observed a substantial neutrophil infiltration in livers from the model group, which was consistent with previous reports of a similar assay 37 . In contrast, BT significantly reduced neutrophil infiltration into islets compared with model (Fig. 7a, c). Similarly, fewer macrophages (F4/80+ cells) were seen at the site of islet grafts in BT mice compared with those from model (Fig. 7b, d). Overall, BT inhibited neutrophil and macrophage infiltration in transplanted islets. We further measured serum TNF-α in mice. Administration of BT significantly decreased the serum TNF-α level (Fig. 7e).

BT suppresses neutrophil and macrophage infiltration, and serum TNF-α production. (a) Representative immunohistochemistry images of Ly6G expression in mice liver tissue from model group (n = 3), BT group (n = 3), and HT group (n = 3). Red arrows indicate neutrophils. (b) Numbers of neutrophils within the islet were lower in the BT group compared with those from model group and HT group 6 h AIT. 150 Model islets (average size: 34,090.52 ± 53,176.21 µm²), 64 BT islets (average size: 2,427.20 ± 309.76 µm²), and 64 HT islets (average size: 2,346.14 ± 382.49 µm²) were counted. (c) Representative immunohistochemistry images of F4/80 expression in mice liver tissue from model group (n = 3), BT group (n = 3), and HT group (n = 3). Red arrows indicate macrophages. (d) Numbers of neutrophils within the islet were lower in the BT group compared with those from model group and HT group 6 h AIT. 83 Model islets (average size: 3,125.54 ± 255.37 µm²), 71 BT islets (average size: 2,565.57 ± 254.71 µm²), and 104 HT islets (average size: 3,588.02 ± 1,177.92 µm²) were counted. The differences between the above groups were analyzed by Kruskal–Wallis test. (e) Serum TNF-α levels measured by ELISA are higher in serum of model group (n = 4) than BT group (n = 4) and HT group (n = 4) 6 h AIT. The differences between the groups were analyzed by one-way ANOVA. *P < 0.05 AAT vs. Ctrl, Student’s t test. (**P < 0.05, *P < 0.05.).
Discussion
IBMIR is a frequent early complication of clinical intraportal islet transplantation and remains a major target for intervention to minimize immediate post-infusion islet loss38,39. Bivalirudin, a direct and specific thrombin inhibitor with rapid and reversible activity, is well established in cardiovascular and interventional medicine, but its efficacy in vivo in the context of islet transplantation has not been defined. Here, we present the first in vivo evaluation of BT in a portal vein islet transplantation model and delineate its anticoagulant and anti-inflammatory actions. We further benchmarked BT against HT—the agent most commonly used during clinical intraportal islet infusion. Our data demonstrate that BT attenuates IBMIR more effectively than HT, resulting in improved engraftment and functional outcomes after allogeneic portal vein islet transplantation, including in BALB/c-nu mouse recipients.
At 1 h AIT, BT produced a greater increase in plasma anti-IIa activity than HT, indicating more potent thrombin blockade and corresponding with a more pronounced prolongation of the activated APTT. No bleeding events or bleeding-related deaths were observed during the dosing and short follow-up intervals. Nevertheless, the limited exposure and observation window preclude definitive conclusions regarding hemorrhagic risk; extended-duration, dose–response, and longitudinal safety studies are warranted to delineate the bleeding liability and therapeutic window of BT in the islet transplantation setting.
By 3 h AIT, APTT values in both the BT and HT groups declined in parallel with waning anti-IIa activity (Fig. 2a), consistent with cessation of anticoagulation at 1 h AIT. In particular, the short plasma half-life of BT (~25 min) predicts near-complete offset by 3 h, accounting for the observed normalization of coagulation parameters40,41. Collectively, these findings indicate that BT provides highly titratable, rapidly reversible anticoagulation, enabling precise peritransplant control of coagulation intensity.
BT exerted a pronounced inhibitory effect on platelet activation 42 . Serum β-TG was significantly lower in the BT group than in the HT group, indicating superior suppression of platelet activation; by contrast, HT was associated with higher β-TG levels, consistent with prior reports of heparin-related platelet activation. TAT complexes, a clinical marker of thrombin generation 5 , were reduced at 1 h and 3 h AIT in both BT and HT animals relative to saline controls. In an additional arm, rivaroxaban yielded even lower TAT levels than HT. Histological assessment at 3 h AIT demonstrated less peri-graft fibrin deposition with BT than with HT, indicating more effective mitigation of microthrombus formation. Collectively, these findings show that BT effectively dampens IBMIR-associated thrombosis by curbing thrombin generation and platelet activation, thereby limiting the deleterious impact of excessive thrombin activity on islet engraftment.
Consistent with its anticoagulant activity, BT attenuated inflammatory signaling AIT. At 3 h AIT, hepatic NF-κB activation was significantly reduced in BT-treated rats, accompanied by a marked decrease in inflammatory-cell infiltration within the graft; both effects were greater with BT than with HT. Serum TNF-α concentrations were likewise significantly lower in the BT group. Given that BT directly and selectively inhibits thrombin, whereas HT acts indirectly via antithrombin to inhibit both thrombin and factor Xa, the superior anti-inflammatory profile observed with BT may reflect targeted suppression of thrombin-driven platelet activation and microthrombosis, thereby dampening downstream inflammatory cascades. These findings indicate that BT exerts anti-inflammatory effects in addition to anticoagulation in the early post-transplant period14,43,44.
Based on the above results, we further investigate that BT significantly suppressed elevated plasma TAT levels, reduced fibrin deposition within 6 h AIT in BALB/c-nu mouse recipients. Also, BT decreased circulating TNF-α levels and leukocyte infiltration in the islet grafts compared with the model group and HT group. All of these results translated into less islet cell damage and better blood glucose control 15 days AIT in recipients treated with BT. Although HT has a certain effect on reducing IBMIR, there is no significant difference compared with saline treatment. The evidence shows that BT inhibits IBMIR, which results in improved outcome of intraportal islet transplantation in mice and the effect is more significant than HT. Based on the above results, we further investigate that BT significantly suppressed elevated plasma TAT levels and reduced fibrin deposition within 6 h AIT in BALB/c-nu mouse recipients. Also, BT decreased circulating TNF-α levels and leukocyte infiltration in the islet grafts compared with the model group and HT group. All of these results translated into less islet cell damage and better blood glucose control 15 days AIT in recipients treated with BT. Although HT has a certain effect on reducing IBMIR, there is no significant difference compared with saline treatment. The evidence shows that BT inhibits IBMIR, which results in improved outcome of intraportal islet transplantation in mice and the effect is more significant than HT.
Our experiments have shown that HT can inhibit thrombin and factor Xa through antithrombin III. While it can effectively reduce IBMIR by inhibiting the inflammatory cascade activated by coagulation, its irreversible anticoagulant effect, risk of platelet activation (such as heparin-induced thrombocytopenia (HIT)), and lack of direct anti-inflammatory properties may increase the risk of bleeding and immune reactions12,45,46. However, as a direct thrombin inhibitor, bivalirudin may have the following advantages in IBMIR: (1) reversibility: it has a short half-life (25 min)17,47,48, allowing for more controllable anticoagulant effects and potentially reducing bleeding risks; (2) anti-inflammatory potential, thrombin is a key mediator of the inflammatory response (e.g. activating endothelial cells and the complement system), and directly inhibiting thrombin may be more effective than HT in blocking the initial steps of IBMIR; and (3) inhibition of platelet activation: It does not rely on antithrombin III, avoiding heparin-related thrombocytopenia or activation, while inhibiting platelet activation. In addition, numerous clinical trials in percutaneous coronary intervention have demonstrated that BT carries a lower bleeding risk than HT49,50. Therefore, BT theoretically may achieve anticoagulation and anti-inflammatory effects with superior safety.
Although the results of this study are highly desirable, there are still some limitations that need to be addressed. First, the sample size of this study is relatively small, and larger multicenter clinical trials are needed to verify the reliability of the results. Second, as this study mainly evaluated the effect of bivalirudin 1 h, 3 h, and 15 days AIT, there is a lack of assessment of bivalirudin’s effect on islet function and transplantation success rate over a longer period (such as 30 days or longer). Moreover, although the administration methods of HT in cardiovascular diseases and interventional treatments are mature12,48, the optimal dose and administration route in portal vein islet transplantation still need further exploration and validation.
Conclusion
This study is the first to investigate the effect of BT in the IBMIR of portal vein islet transplantation and its effect on improving short-term outcomes of portal vein islet transplantation. It was compared with the clinical use of heparin. We found that bivalirudin effectively alleviated the IBMIR AIT through multiple mechanisms. It can inhibit thrombin, reduce platelet activation, decrease thrombosis formation, and regulate inflammatory responses, and thus improve short-term outcomes of pancreatic islet transplantation. Compared with the traditional anticoagulant HT used in clinical practice, BT shows superior effects in reducing IBMIR and may become a safer and more effective anticoagulation strategy in future islet transplantation.
Footnotes
Acknowledgements
The authors express gratitude to Guangxi Clinical Research Center for Kidney Diseases of Integrated Traditional Chinese and Western Medicine and the Science Experiment Center of Guangxi University of Chinese Medicine for their support.
Ethical Considerations and Consent to Participate
This research has been approved by the Experimental Animal Welfare and Ethics Committee of Guangxi University of Chinese Medicine (Approval Number: No. DW20231115-263).
Consent for Publication
Not Applicable.
Author Contributions
M.L. participated in the research design, implementation, and writing of the paper. Z.Z. participated in the implementation, and writing of the paper. Y.L. participated in the data analysis of this study. X.Y. participated in the performance of this research. C.C. participated in the performance of this research. S.C. participated in the performance of this research. Y.Y participated in the performance of this research. P.W. and L.W and H.G. participated in the research design, fund support, and paper writing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Innovation Project of Guangxi Graduate Education (grant number YCSW2024422), the Guangxi Key Research and Development Program (grant number GuikeAB24010077), the Project for Enhancing Young and Middle-aged Teacher’s Research Basis Ability in Colleges of Guangxi(grant number 2024KY0298), and the Western Medicine for Kidney Diseases (grant number GuikeAD22035122).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
