Abstract
Posaconazole (POS) tablets were approved for prophylaxis of invasive fungal disease (IFD) in patients with hematological disorders undergoing haploidentical allogeneic stem cell transplantation (haplo-HSCT). There is limited research on drug–drug interactions (DDIs) between POS, cyclosporine A (CsA), and mycophenolate mofetil (MMF), as well as the impact of POS on acute graft versus host disease (aGVHD) in haplo-HSCT patients receiving POS for secondary antifungal prophylaxis (SAP). This study aims to investigate the DDI between POS, CsA, and MMF, as well as the incidence of aGVHD in haplo-HSCT patients with prior-IFD. This is a single-arm, open-label, prospective trial. Plasma concentrations of POS, mycophenolic acid (MPA), and CsA were monitored within 30 days post-transplantation. In addition, the incidences of aGVHD and IFD were observed. Forty-six patients with prior-IFD were enrolled. POS reached a steady state by week 2, with the mean through blood concentration (TBC) of 0.54 ± 0.07 μg/mL. MPA achieved target levels by week 3 without dose adjustment, with a mean TBC of 0.84 ± 0.08 mg/L. The CsA TBC levels required individualized dose modifications. The incidences of grade II–IV and grade III–IV aGVHD were 47.83% and 21.74%, respectively. The breakthrough rate of SAP was 4.35% at 100 days and 10.86% at 6 months. The conclusions indicate that POS tablets do not require dose adjustments for MMF; however, CsA dosing must be individualized. POS tablets appear to be effective and well-tolerated for SAP in haplo-HSCT (the Chinese Clinical Trial Registry: www.chictr.org.cn (ChiCTR2200059472)).
Keywords
Introduction
Haploidentical allogeneic hematopoietic stem cell transplantation (haplo-HSCT) is an effective therapeutic approach for the treatment of hematological malignancies. Approximately 94% of haplo-HSCT procedures in China follow the “Beijing Protocol,” which consists of T-replete therapy with granulocyte colony-stimulating factor and anti-thymocyte globulin (ATG). The prophylaxis regimen for acute graft versus host disease (aGVHD) includes cyclosporine A(CsA), methotrexate (MTX), and mycophenolate mofetil (MMF) 1 . The approach significantly reduces the risk of aGVHD following transplantation while potentially increasing the risk of infection.
Posaconazole (POS) tablets are a second-generation broad-spectrum antifungal agent. Numerous guidelines1–3 recommend POS for preventing of invasive fungal disease (IFD) in patients undergoing haplo-HSCT. Secondary antifungal prophylaxis (SAP) has been reported to improve the prognosis of patients with prior-IFD in haplo-HSCT 4 . According to the latest European Conference on Infections in Leukemia (ECIL) guideline 2 , POS tablets are recommended for SAP of IFD in haplo-HSCT recipients.
The absorption, metabolism, and excretion of CsA can be influenced by factors such as food intake, liver and kidney function, and genetics, leading to significant interindividual variability in CsA levels5,6. As a substrate of cytochrome P450 (CYP) 3A4/5 and P-glycoprotein (P-gp), CsA interacts with multiple drugs, including antibiotics, antifungals, and glucocorticoids. POS is both a substrate and an inhibitor of P-gp efflux transporters, which may increase the CsA levels by inhibiting P-gp activity, and it is also a potent inhibitor of CYP3A4 expression, thereby reducing CsA metabolism 5 . Significant drug–drug interactions (DDIs) have been reported between MMF and CsA. The active metabolite of MMF, mycophenolic acid (MPA), shows different exposure levels with CsA compared with tacrolimus, with a 40% reduction in MPA area under the curve when combined with CsA 7 . The combination requires dose adjustments to minimize the risk of aGVHD.
The interaction between POS and CsA raises concerns regarding its effect on MPA levels. Currently, there is not enough data to confirm the presence of DDI between POS, CsA, and MMF. The study aims to evaluate the DDI of the regimen to provide clinical guidance for safe medication use and to assess its impact on aGVHD and patient outcomes.
Methods
Study design and participants
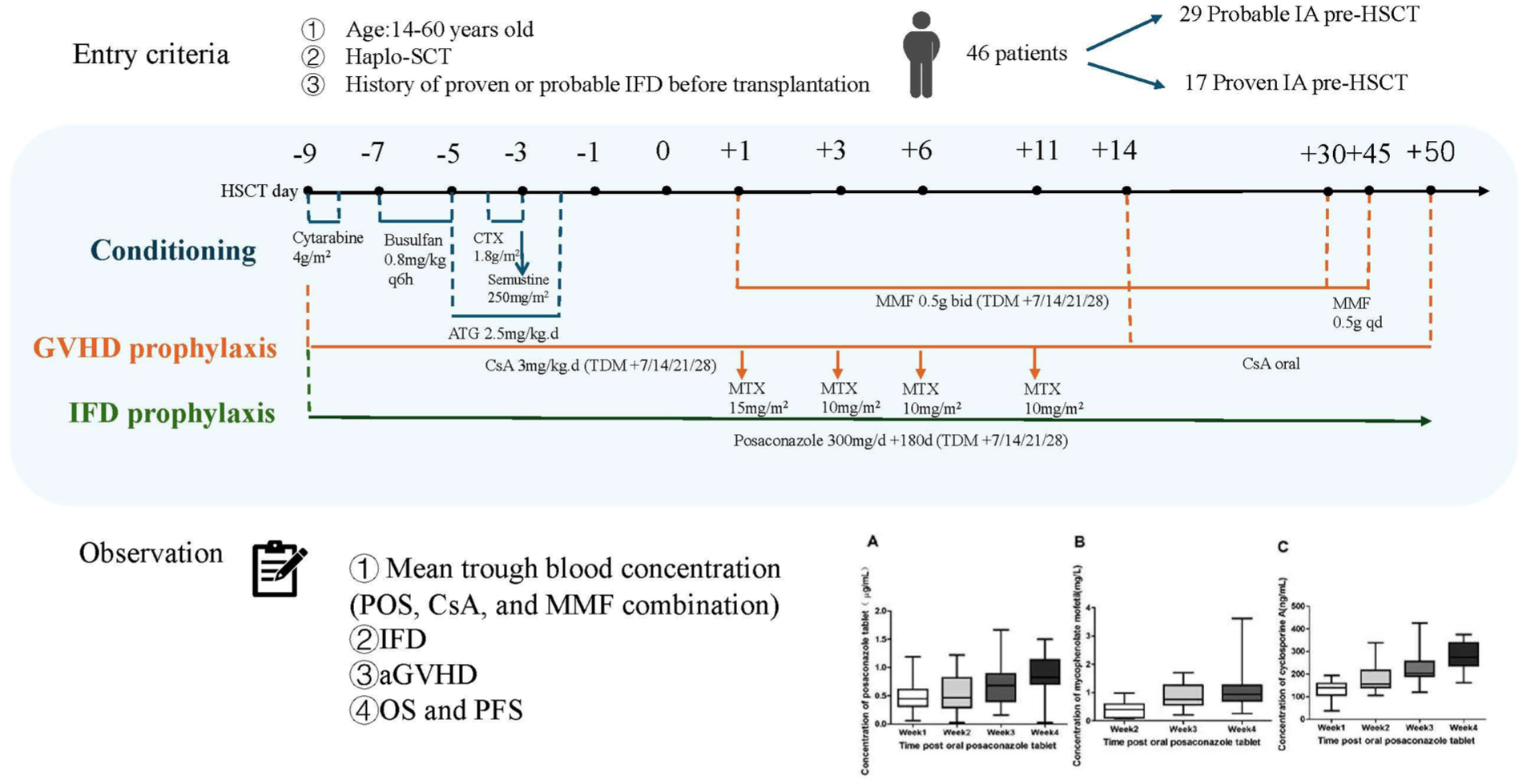
This was a prospective, open-label, single-center study (ChiCTR2200059472). Inclusion criteria were as follows: (1) Age ≥14 and ≤60 years. (2) Patients undergoing haplo-HSCT. (3) The aGVHD prophylaxis regimen consists of CsA combined with MTX and MMF. (4) Patients with a proven or probable IFD history before transplantation who achieved a complete response (CR), partial response (PR), or stable disease (SD). The exclusion criteria as follows: (1) Liver dysfunction (bilirubin or transaminase level >2× normal) or renal impairment (creatinine >97 μmol/L). (2) Use of medications that are known to interact with azoles and potentially induce life-threatening effects within 24 hours, excluding astemizole, which was not administered within the preceding 10 days, or β-hydroxy-β-methylglutaryl-coenzyme A reductase inhibitors metabolized by CYP 3A4 enzyme within 24 hours from the commencement of the study.
HLA haploidentical transplantation refers to donors who are related to the recipient and share at least 3 out of 6 HLA loci (typically HLA-A, HLA-B, HLA-C, HLA-DRB1, HLA-DQB1, and HLA-DPB1), corresponding to 6 to 11 matched alleles out of 12.
The study was conducted in accordance with the amended Helsinki Declaration, and the protocol received approval from the ethical review boards of the First Affiliated Hospital of Xi’an Jiaotong University (NO. XJTU1AF2022LSK-207). All adult patients or authorized representatives of minors provided written informed consent.
Primary and secondary endpoint
The primary endpoints were to investigate whether POS affects the time to reach the target TBC of CsA and MPA, as well as the dose adjustment of CsA and MMF after POS reaches the steady concentration. The secondary endpoint involved assessing the impact on aGVHD, overall survival (OS), and progression-free survival (PFS) in haplo-HSCT patients with prior IFD.
Conditioning
All patients received a myeloablative conditioning regimen based on the modified BuCy2 protocol, which consists of cytarabine 4 mg/m2 on days −9 to −8, busulfan 0.8 mg/kg every 6 hours on days −7 to −5, cyclophosphamide 1.8 mg/m2 on days −4 to −3, semustine 250 mg/m2 on day −3, and ATG (Thymoglobulin, Genzyme, Cambridge, MA, USA) administered at 2.5 mg/kg on days −5 to −2.
GVHD prophylaxis
CsA combined with MMF and a short course of MTX was administered for aGVHD prophylaxis. CsA is administered intravenously at a dose of 3 mg/g per day starting from the conditioning regimen and is then switched to oral administration on day +14. The dose is gradually reduced starting from day +50, with discontinuation depending on the presence of aGVHD. MTX is given intravenously at a dose of 15 mg/m2 on day +1, followed by 10 mg/m2 on days+3, +6, and +11. MMF is initiated on day +1 at a dosage of 0.5 g twice daily. After 1 month, the dose is reduced to 0.5 g once daily and discontinued 45 days after transplantation. The grading criteria for aGVHD were referenced from the “Chinese consensus of allogeneic hematopoietic stem cell transplantation for hematological disease (III)—acute graft-versus-host disease (2020)”8,9
SAP and definition
Based on guidelines and relevant studies references1–3,10–12, SAP was initiated on the first day of the conditioning regimen and continued until 100 days after transplantation, unless there was a breakthrough of IFD, an adverse event requiring discontinuation of the study medication, or death. The antifungal prophylaxis agent was reinstated in patients with chronic GVHD or those receiving long-term systemic corticosteroids. POS tablets 300 mg/day twice for the first day, followed by 300 mg per day as SAP.
The diagnosis of IFD was made in accordance with the revised European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group; National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) criteria 13 and BMT-CTN Infection Grading System13,14. IFD was classified as proven, probable, or possible according to EORTC/MSG13,14. Grade 1 fungal infections often do not have a significant impact on clinical outcomes, and some may resolve spontaneously. Grade 2 fungal infections are of moderate severity, may require the administration of intravenous or oral anti-infective agents, and may require hospitalization or low-risk procedures, but generally are not lethal. Grade 3 fungal infections are potentially life-threatening or associated with a substantial risk of morbidity 14 .
Responses to treatment were categorized as CR, PR, SD, progressive disease (PD), and death. Failure of SAP was defined by any of the following cases: PD of previous IFD or new onset of different types of fungal infections according to microbiology 15 . Upon failure of SAP, we would modify the dosage of POS or consider alternative broad-spectrum antifungal agents (voriconazole, caspofungin, amphotericin B, or a combination of voriconazole and caspofungin).
Therapeutic drug monitoring
POS, MMF, and CsA samples were tested with a validated ultra-performance liquid chromatography fluorescence assay, and an independent quality control center was employed for comprehensive quality control management throughout the entire process. Exposure metrics were defined as TBC (Cmin; 24 h ± 2 h post-dose on days 7, 14, 21, and 28). Samples were collected in the morning on days 7, 14, 21, and 28 after transplantation and were collected in ethylenediaminetetraacetic acid (EDTA)-containing anticoagulant tubes. The pharmacokinetic (PK) parameters for total POS concentrations were estimated using both compartmental and non-compartmental methods. CsA doses were adjusted to achieve target TBC 200–300 ng/mL and to prevent renal dysfunction 16 . The MPA plasma levels in haplo-HSCT are below the therapeutic range recommended for solid organ transplantation (total target TBC 1.0–3.5 mg/L) 17 .
Follow-up and outcomes
Follow-up assessments were conducted weekly during the first 3 months and at biweekly intervals from months 3 to 6 after transplantation. All patients were followed up until February 28= 2025. IFD was regularly monitored through various assessments, including clinical symptoms, chest, sinuses, and abdomen CT scans (monthly for the first 3 months and every 2–3 months from 3 to 6 months), as well as bronchoscopy, bronchoalveolar lavage, serological tests for Aspergillus galactomannan and (1,3)-B-
Statistical analysis
Continuous variables are presented as medians, whereas categorical variables are shown as percentages. The proven and probable groups were compared using the chi-square test for categorical data and the Wilcoxon rank-sum test for quantitative data. Survival curves for OS and PFS were generated with the Kaplan–Meier method. Statistical analysis was performed with SPSS version 16.0 (IBM, Chicago, IL, USA), and competing risks analysis was conducted using R version 3.6.0 (http://abc.e-ghijklm.opq) software. A P value of less than 0.05 was regarded as statistically significant.
Results
Patient characteristics
Seventy-two patients were screened between May 1, 2022, and May 1, 2023. Forty-six patients were included for analysis, and a flow diagram is presented in Fig. 1, as indicated in Table 1. Seventeen patients (36.96%) had a previously proven IFD, primarily diagnosed through culture and tissue nucleic acid testing, including 10 cases of Aspergillus species and 7 cases of Candida species.

Flow chart of patients.
Characteristics of patients who received posaconazole tablets.

The TBCs of POS, CsA, and MPA
The mean TBC of POS at weeks 1, 2, 3, and 4 were 0.48 ± 0.06 µg/mL, 0.54 ± 0.07 µg/mL, 0.75 ± 0.08 µg/mL, and 0.90 ± 0.08 µg/mL (Fig. 2a). POS did not achieve 0.5 µg/mL until week 2. The mean TBC of MPA at weeks 2, 3, and 4 were 0.41 ± 0.06 mg/L, 0.84 ± 0.08 mg/L, and 1.08 ± 0.13 mg/L (Fig. 2b). After both TBCs of POS and CsA reached steady-state levels, the TBC of MPA gradually increased. MPA reached a steady state at week 4, and the steady-state TBC reached the targeted range. During the entire observation period, no cases had their MPA dosage adjusted.

The TBC of (a) POS, (b) MMF, and (c) CsA within 30 days post-transplantation.
The mean TBC of CsA at weeks 1, 2, 3, and 4 were 133.04 ± 0.13 ng/mL, 179.53 ± 11.19 ng/mL, 225.02 ± 13.80 ng/mL, and 278.61 ± 13.61 ng/mL (Fig. 2c). At week 1, 26 cases of CsA mean TBC were below 150 ng/mL, and the dosage was increased by 25% of the original dose. At week 4, 15 cases of CsA mean TBC were above 250 ng/mL, and the dosage was decreased by 25%. It was observed that in 4 out of 15 cases, as the TBC of POS increased, the TBC of CsA also significantly increased, exceeding the safe therapeutic window 18 and necessitating a dose adjustment.
SAP and efficacy
The SAP failure rate was 4.35% at 100 days and 10.86% at 6 months. Four patients with SAP failure received amphotericin B treatment, and one received combination therapy with voriconazole and caspofungin. The breakthrough IFD was grade 2. None of the patients died due to IFD.
aGVHD and survival
Twenty-two cases developed aGVHD (3 cases of grade III and 7 cases of grade IV), including 7 cases of intestinal and skin aGVHD, and 15 cases of skin aGVHD. The incidence of aGVHD was 47.8%, with 21.7% classified as grade III–IV. The endpoint of the last follow-up for all surviving patients was February 28, 2025. During the median follow-up of 274 days (range 133–408 days), there were nine deaths attributed to relapse (4 cases), GVHD (4 cases), and myocarditis (1 case). The 1-year OS and PFS rates were 77.4 % ± 7.51% (95% CI: 64.0%–93.6%) and 74.2% ± 7.86% (95% CI: 60.3%–91.3%), respectively. The expected 3-year OS and PFS rates were 77.4% ± 7.51% (95% CI: 64.0%–93.6%) and 70.5% ± 8.30% (95% CI: 56.0%–88.8%), respectively (Fig. 3).
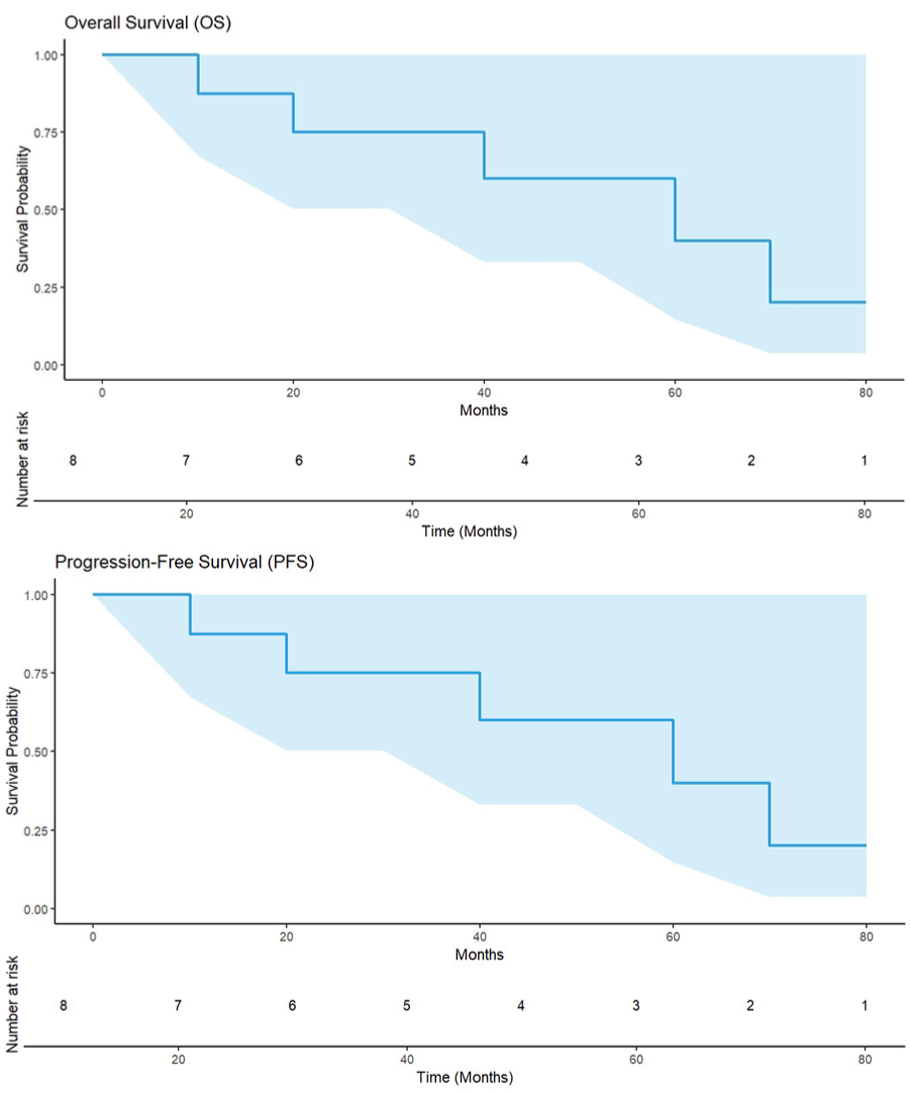
OS and PFS of patients.
Discussion
POS has DDI with CsA, and CsA has DDI with MMF. However, there are limited data regarding the effects of POS on CsA and MMF, and how to adjust the dosage. This study first evaluates the DDI among POS, CsA, and MMF, and their impact on aGVHD in patients undergoing haplo-HSCT.
A pharmacokinetics study 18 of POS tablets in the Chinese population showed that in the sparse sampling subgroup, POS tablets reached steady concentration by the eighth day, with a mean TBC of 1410 ng/mL. The TBC reached its highest level on the 15th day. Our study revealed that POS tablets achieved 0.5 µg/mL by week 2, and TBC gradually increased, reaching its highest concentration at week 4. The mean TBC of POS reached effective prophylactic concentrations; however, it was not as high as reported in the phase III study 19 . This discrepancy may be resulting from the high prevalence of vomiting and diarrhea in haplo-HSCT patients, who are often on multiple comorbid medications.
The systematic review20,21 indicates that the dosage of CsA should be reduced when initiating POS tablets therapy, and TBC levels of the immunosuppressant should be monitored throughout the course of POS tablets therapy to allow for appropriate dose adjustments. Other studies18,21 found a significant elevation in CsA levels within 1 week of initiating POS therapy, and the highest concentration-to-dose ratio was observed 9 to 12 days after POS initiation. Our study also found that after 1 week of administering POS, the CsA concentration increased significantly in 43.48% of patients, and approximately 56.52% of patients required dose adjustment. MMF is often combined with CsA as part of a maintenance immunosuppressive regimen. In haplo-HSCT patients, various factors can affect the pharmacokinetics of MPA, causing fluctuations in MPA exposure22,23. CsA inhibits MRP2-mediated enterohepatic circulation of MPA glucuronide, causing a decrease in MPA exposures. During the initial post-transplant period, haplo-HSCT patients often suffer from diarrhea, which reduces the reabsorption of MPA in the gastrointestinal tract and leads to lower MPA levels 24 . A phase I/II study 25 investigating the combination of MMF with CsA for GVHD showed that dose escalation of MMF is necessary to achieve the target concentration.
MPA is a substrate of P-gp, while POS is both a substrate and an inhibitor of P-gp. P-gp is found on the canalicular surface of hepatocytes as well as on the apical surfaces of renal tubular epithelial cells and intestinal interstitial cells. This transporter can facilitate the removal of drugs from cells. When P-gp is inhibited, drug exposure for its substrates increases. Conversely, inducing P-gp reduces drug exposure. Therefore, MPA may accumulate when administered with POS 26 . In this study, we conducted a more detailed analysis of the blood concentrations of these three drugs when used in combination. The combination of POS and CsA does not significantly decrease the TBC of MPA. Instead, it slightly increases the TBC, indicating that there is no need to adjust the dose of MMF. This reduces the complexity of medication management and lowers the risk of adverse reactions associated with MMF. Therefore, the addition of POS ensures a relatively stable TBC of all three drugs. This treatment regimen greatly benefits patients and enhances medication safety.
A prospective randomized trial 27 found that using a combination of CsA and MMF prophylactic regimens led to incidences of 48% for grade II–IV aGVHD and 63% for chronic GVHD in myeloablative allogeneic bone marrow transplantation. Our study showed a similar incidence of aGVHD compared with the above study. This indicates that POS may have no impact on aGVHD when combined with appropriate TBC of CsA and MMF.
Due to the limited availability of large prospective studies, two retrospective studies have reported the breakthrough incidence of IFD in patients with prior IFD undergoing haplo-HSCT as follows: 42.9% used itraconazole as SAP, 31.3% used voriconazole, 0% to 55.5% used liposomal amphotericin B, and 13.8% to 16.7% used caspofungin4,10. In all, 23.1% in POS oral suspension for SAP 11 ; however, the concentrations in the POS tablets are higher and are not affected by food, gastrointestinal function, or other factors 28 . Therefore, the failure rate of SAP within 6 months in our study was only 10.84%, which is lower than that of other antifungal drugs used for SAP. The burden of IFD in HSCT patients is substantial, characterized by high morbidity and mortality rates, diagnostic challenges, and significant economic impact 29 . The lower incidence of IFD observed in our study significantly reduces morbidity and mortality rates in HSCT patients 30 . With insurance reimbursement for POS tablets in China, HSCT patients can incur lower expenses, thereby reducing hospital stays and healthcare costs associated with IFD 31 .
A meta-analysis 32 showed that the main pathogens causing prophylaxis failure with POS were Aspergillus and Fusarium. Consequently, for the five patients who experienced breakthrough IFD, we switched to liposomal amphotericin B for treatment. Among these patients, one had a high G test result, prompting the initiation of combination therapy. Due to the small number of patients with SAP prevention failure, it is not possible to assess the associated risk factors. Literature 33 reports indicate that the incidence of pulmonary mixed infections in allo-HSCT patients is as high as 50.5%, with an associated increased risk of mortality 34 . The primary pathogens include bacterial, fungal, and viral. Since viral infections often present with atypical clinical symptoms and limited treatment options, we conducted routine viral load monitoring for common viruses such as cytomegalovirus (CMV), Epstein-Barr virus (EBV), BK virus (BKV), and COVID-19 throughout the transplantation period. Viral loads were monitored weekly for the first 100 days and biweekly from days 100 to 200. The incidence rates were 43.5% for CMV viremia, 13.0% for EBV viremia, and 8.7% for BKV-related hemorrhagic cystitis in our study. All patients received preemptive treatment promptly upon positive viral DNA results, and no deaths due to progression of mixed infections were reported. Our study also suggests that early prophylaxis with POS tablets during transplantation may reduce the risk of mixed infections of fungal and other pathogens, particularly in those with aGVHD, severe mucositis, colitis, diarrhea, nausea, or emesis associated with recurrent or new systemic fungal diseases.
Limitations
Our study has several limitations. First, it is a single-center study with a small sample size. In addition, the study did not include a comparison with other drugs utilized for SAP. Second, the monitoring of drug concentrations was limited to 1 month after transplantation, and further investigation is necessary to understand the subsequent DDI and their specific mechanisms.
Conclusion
In summary, our study supports the use of CsA, MMF, and POS tablet in combination for haplo-HSCT patients following the “Beijing protocol.” Our findings indicate that the dosage of MMF may not need to be adjusted, while the dosage of CsA should be personalized and adjusted based on therapeutic drug monitoring. POS tablets appear to be effective and well-tolerated for SAP in IFD after haplo-HSCT. Given the limitations mentioned above, further investigation through multicenter prospective trials is required.
Footnotes
Acknowledgements
This study would not have been possible without the cooperation of all the patients, medical and nursery staff, and data managers involved in the project. Supported in part by a research grant from Investigator-Initiated Studies Program of MSD. The opinions expressed in this paper are those of the authors and do not necessarily represent those of MSD.
Ethical Considerations
The study was performed in accordance with the modified Helsinki Declaration, and the protocol was approved by the ethical review boards of the First Affiliated Hospital of Xi’an Jiaotong University (NO.XJTU1AF2022LSK-207). This clinical trial was also registered at the Chinese Clinical Trial Registry (ChiCTR2200059472).
Author Contributions
Concept, design, and drafting of the manuscript: WXN. Acquisition of data, analysis, and interpretation of data: WYQ. Writing, review, and/or revision of the manuscript: HPC.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Medical Basic Clinical Integration Innovation Project of Xi’an Jiaotong University (YXJLRH2022007), and National Key R&D Program of China (2022YFC2502700).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and analyzed in this study can be obtained from the corresponding author upon reasonable request.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
All procedures in this study, including the study intervention, data analysis, and blood sample collection, were conducted after obtaining written informed consent. Patients were fully informed about the study intervention and its experimental nature, as well as potential adverse events, including possible drug dosage adjustments and adverse events mentioned in the prescribing information.
