Abstract
NPM1-mutated acute myeloid leukemia (AML) is a distinct entity with specific biological and clinicopathological features. Prognosis varies depending on the associated mutations and clinical characteristics. This study evaluated the prognostic factors of NPM1-mutated AML in patients who underwent allogeneic hematopoietic stem cell transplantation (allo-HSCT). We enrolled 139 patients with NPM1-mutated AML who underwent allo-HSCT. The median age at allo-HSCT was 47 years with a median follow-up of 1,134 days. The 3-year cumulative incidence of relapse (CIR), leukemia-free survival (LFS), and overall survival (OS) rates were 15.1%, 78.4%, and 78.0%, respectively. FLT3-ITD mutations showed higher CIR (22.6% vs 5.3%, P = 0.006), lower LFS (70.9% vs 88.1%, P = 0.019), and reduced OS (70.8% vs 87.5%, P = 0.036). FLT3-ITD and DTA mutations indicated adverse prognosis, while IDH1/2 mutations showed improved outcomes. Matched sibling donor (MSD) allo-HSCT patients had higher CIR and lower LFS and OS than unrelated donor (URD) or haploidentical-related donor (HRD) recipients. Pre-HSCT MRD status affected the outcomes, with MRD+ and nCR patients showing worse outcomes. Multivariable analysis identified FLT3-ITD, MSD, and pre-MRD status as significant predictors. In conclusion, FLT3-ITD and DTA mutations are associated with an adverse prognosis, whereas HRD or URD transplants may offer better outcomes than MSD allo-HSCT.
Introduction
The nucleophosmin1 (NPM1) gene encodes the nucleolar phosphoprotein nucleophosmin. NPM1 gene mutations are the most common genetic changes in acute myeloid leukemia (AML), present in 30%–35% of adult cases1 –3. NPM1 mutations behave as gatekeepers for leukemogenesis 4 and frequently co-occur with other genetic aberrations, such as FLT3-ITD, DNMT3A, TET2, RAS, and IDH mutations 2 . NPM1-mutated AML has been identified as a distinct leukemia category in both the World Health Organization AML classification and the International Consensus Classification owing to its unique biological and clinical features5,6. Although NPM1-mutated AML is generally associated with a favorable prognosis 7 , the presence of additional mutations or specific clinical features can result in variability in patient outcomes. Many patients still require hematopoietic stem cell transplantation; however, even after undergoing this procedure, some patients remain at risk of relapse and mortality. This highlights the need for a more nuanced understanding of the prognostic factors affecting this patient population.
At diagnosis, NPM1-mutated AML is typically characterized by high blast percentages and increased white blood cell and platelet counts and is frequently linked to a normal karyotype 8 . Currently, there is no consensus on the impact of clinical biology on the prognosis of NPM1-mutated patients undergoing allogeneic hematopoietic stem cell transplantation (allo-HSCT). To better understand this area and enhance clinical decision-making, we conducted a comprehensive evaluation of prognostic factors in AML patients with NPM1 mutations who underwent allo-HSCT. This study aimed to elucidate the connections among genetic mutations, clinical characteristics, and treatment outcomes in this specific group of patients.
Materials and methods
Patients
The study included adult patients (age ≥18 years) with de novo AML who underwent their first allo-HSCT at the First Affiliated Hospital, Zhejiang University School of Medicine, from March 1, 2016, to December 31, 2023. The indications for allo-HSCT in these patients were as follows: (1) ELN-defined favorable-risk AML with persistent minimal residual disease (MRD) positivity, (2) intermediate- or high-risk stratification based on ELN criteria, (3) primary refractory AML (failure to attain complete remission (CR) after two consecutive induction chemotherapy cycles), or (4) disease status of ≥ second CR or nonremission. Transplantation procedures, including the conditioning regimen, graft-versus-host disease (GVHD) prophylaxis regimen, and supportive care, were conducted in accordance with previously established methodologies 9 . The study was approved by the Ethics Review Committee of the First Affiliated Hospital, Zhejiang University School of Medicine, and adhered to the Declaration of Helsinki. All participants provided written informed consent authorizing the use of their data.
Definitions
Neutrophil engraftment was defined as the first absolute neutrophil count exceeding 0.5 × 109/L for a minimum of 3 consecutive days, whereas platelet engraftment was identified as the first platelet count surpassing 20 × 109/L for 7 consecutive days without platelet transfusion. Relapse was characterized as the recurrence of ≥ 5% blasts in bone marrow, the reappearance of blasts in the blood, or the identification of extramedullary disease. MRD status was assessed using reverse transcription quantitative polymerase chain reaction (RT-qPCR). MRD positivity (MRD+) was defined as NPM1 mutant copies per ABL1 copy > 2%, core-binding factor (CBF) > 0.1%, and WT1 > 0.6% measured in bone marrow samples10,11. Cytogenetic risk categories were assigned based on the Medical Research Council (MRC) criteria 12 . Acute GVHD (aGVHD) and chronic GVHD (cGVHD) were diagnosed and graded according to the established criteria13,14.
Endpoints
The day of transplantation was considered day 0. The primary endpoint of this study was cumulative incidence of relapse (CIR). The secondary endpoints were leukemia-free survival (LFS), overall survival (OS), nonrelapse mortality (NRM), and GVHD. CIR was determined by calculating the time from allo-HSCT to relapse. LFS refers to the period from allo-HSCT to relapse or death from any cause. OS was measured from the date of allo-HSCT to either the date of death or last follow-up. NRM was defined as the time from allo-HSCT to death from any cause without prior hematologic relapse or progression.
Statistical analysis
Patient characteristics are presented as frequencies and percentages for categorical variables and medians with interquartile ranges (IQR) for continuous variables. Chi-square tests were used to compare categorical variables, and Mann–Whitney U tests were used to compare continuous variables. The cumulative incidence function was employed to estimate CIR, NRM and GVHD using a competing risk model. Relapse and NRM were considered competing risks. Relapse and death were treated as competing events for GVHD. The Kaplan–Meier estimator and log-rank test were used to calculate the probabilities of OS and LFS. The Cox proportional hazards regression model was used to calculate the hazard ratio (HR) and 95% confidence interval (CI) for associated outcome factors. Statistical analyses were performed using IBM SPSS (version 22) and R (version 4.2.3; https://www.r-project.org). Statistical significance was defined as a two-tailed P-value < 0.05.
Results
Patient characteristics
A total of 139 consecutive patients with NPM1-mutated AML, who underwent their first allo-HSCT, were included in the analysis. The median age at the time of allo-HSCT was 47 years (IQR = 36–52), and the median follow-up time was 1,134 days (range, 214–3,038). Table 1 presents the baseline patient characteristics. The most common co-mutations identified were FLT3-ITD (56.1%) and DNMT3A (41.0%), followed by IDH2 (19.4%), NRAS (15.1%), TET2 (11.5%), and IDH1 (10.8%) (Fig. 1).
Demographic and clinical characteristics of study population.

IQR: interquartile ranges; WBC: white blood cell; BM: bone marrow; ELN: European LeukemiaNet; FAB: French-American-British; CR1: first complete remission; CR2 second complete remission; nCR: noncomplete remission; MRD: measurable residual disease; URD: unrelated donor; HRD: haploidentical-related donor; MSD: matched sibling donor; MNC: mononuclear cells.

Distribution of co-mutations in patients with NPM1-mutated AML.
Transplantation outcomes
In the entire cohort, the 3-year CIR, LFS, and OS following allo-HSCT were 15.1% (95% CI = 8.9%–21.3%), 78.4% (95% CI = 71.3%–85.5%), and 78.0% (95% CI = 70.7%–85.3%), respectively. The cumulative incidence of grade II–IV and grades III–IV acute GVHD at day 100 was 11.5% (95% CI, 6.2%–16.8%) and 5.0% (95% CI, 1.4%–8.7%), respectively. The 3-year cumulative incidence of moderate-to-severe cGVHD was 17.3% (95% CI, 10.6%–23.9%). The 3-year cumulative incidence of NRM was 6.5% (95% CI, 2.4%–10.6%).
Impact of co-mutations on outcomes
Among 139 patients, 78 (56.1%) had concomitant FLT3-ITD mutations at diagnosis. Patients with FLT3-ITD exhibited a notably higher 3-year CIR (22.6% vs 5.3%; P = 0.006), lower LFS (70.9% vs 88.1%, P = 0.019), and reduced OS (OS = 70.8% vs 87.5%, P = 0.036) than those without FLT3-ITD mutation (Fig. 2a–c).

Prognostic value of FLT3-ITD, DTA and IDH1/2 mutations status in the entire cohort. (a) CIR, (b) LFS and (c) OS of NPM1-mutated AML patients by FLT3-ITD mutations. (d) CIR, (e) LFS, and (f) OS of NPM1-mutated AML patients by FLT3-ITD and DTA mutation status. (g) CIR, (h) LFS, and (i) OS of NPM1-mutated AML patients by IDH1/2 mutation status.
The presence of DNMT3A, TET2, or DTA (DNMT3A, TET2, or ASXL1) mutations did not significantly affect the CIR, LFS, or OS (Supplemental Figure S1). Although patients with DTA had a higher CIR and a lower 3-year LFS and OS, these differences were not statistically significant (CIR = 20.1% vs 10.4%, P = 0.085; LFS = 72.9% vs 83.7%, P = 0.086; OS = 72.2% vs 83.3%, P = 0.127).
When examining outcomes based on combinations of FLT3-ITD and DTA mutations. Patients with both FLT3-ITD and DTA mutations had the highest 3-year CIR (29.0%, 95% CI = 14.1%–43.9%) and the worst LFS (63.3%, 95% CI = 47.8%–78.8%) and OS (63.4%, 95% CI = 47.1%–79.7%). The next worst outcomes were observed in patients with only the FLT3-ITD mutation, followed by those with only the DTA mutations. Patients without FLT3-ITD and DTA mutations had the lowest CIR (3.5%, 95% CI = 0%–10.2%) and the best LFS (90.3%, 95% CI = 79.9%–100%) and OS (89.5%, 95% CI = 78.1%–100%) (Fig. 2d–f).
Patients with IDH1/2 mutations had a significantly lower 3-year CIR than those with wild-type IDH1/2 (4.8% vs 19.6%; P = 0.033; Fig. 2g). Furthermore, patients with IDH1/2 mutations had a markedly higher 3-year LFS and OS than those with wild-type IDH1/2 (LFS = 90.4% vs 73.1%, P = 0.032; OS = 89.8% vs 72.8%, P = 0.043; Fig. 2h, i).
We also examined the effects of RAS pathway mutations (KRAS, NRAS, PTPN11, NF1, and BRAF) and secondary-type gene mutations (STM) (ASXL1, RUNX1, EZH2, BCOR, SF3B1, SRSF2, STAG2, U2AF1, and ZRSR) on posttransplant outcomes. However, these genetic alterations did not significantly affect clinical outcomes following allo-HSCT (Supplemental Figure S2).
Effect of donor type and other clinical factors on outcomes
Patients who received matched sibling donor (MSD) allo-HSCT exhibited significantly higher 3-year CIR of 42.1% (95% CI = 19.2%–65.0%) than unrelated donor (URD) recipients at 13.3% (95% CI = 0.0%–30.9%) and haploidentical-related donor (HRD) recipients at 9.9% (95% CI = 4.0%–15.8%) (P = 0.001; Fig. 3a). Patients in the MSD group also showed significantly lower LFS (47.4%, 95% CI = 24.9%–69.9%) than those in the URD (86.7%, 95% CI = 69.5%–100.0%) and HRD (83.4%, 95% CI = 76.1%–90.7%) groups (P < 0.001; Fig. 3b). Similarly, OS was significantly lower in the MSD group (47.4%, 95% CI = 24.9%–69.9%) than in the URD (86.7%, 95% CI = 69.5%–100.0%) and HRD (83.6%, 95% CI = 76.2%–91.0%) groups (P < 0.001; Fig. 3c). At day 100 after allo-HSCT, the cumulative incidence of grades II–IV aGVHD was 10.5% (95% CI, 4.6–16.4%) in HRD recipients and 26.3% (95% CI, 5.9%–46.7%) in MSD recipients (P = 0.047), with no cases observed in the URD group. The corresponding rates for grades III–IV aGVHD were 2.9% (95% CI, 0%–6.1%), 21.1% (95% CI, 2.2%–39.9%; P = 0.002), and 0%, respectively. The 3-year cumulative incidence of moderate-to-severe cGVHD was 16.8% (95% CI = 9.2–24.5%) in patients receiving HRD transplants, 21.1% (95% CI = 2.0%–40.1%) in those with MSD, and 17.3% (95% CI = 0%–40.3%) in URD recipients. Pairwise comparisons revealed no statistically significant differences between the donor types (HRD vs MSD, P = 0.574; HRD vs URD, P = 0.921; MSD vs URD, P = 0.613). The 3-year NRM was 6.7%, 10.5%, and 0% in HRD, MSD, and URD recipients, respectively. No significant difference was observed between HRD and MSD recipients (P = 0.527).

Prognostic value of pre-HSCT donor type and disease status in the entire cohort. (a) CIR, (b) LFS and (c) OS of NPM1-mutated AML patients by donor type. (d) CIR, (e) LFS and (f) OS of NPM1-mutated AML patients by pre-HSCT disease status.
The 3-year CIR for patients with pre-HSCT MRD-, MRD+, and noncomplete remission (nCR) were 9.2%, 30.0%, and 67.3%, respectively (P < 0.001; Fig. 3d). The 3-year LFS rates were 84.0%, 70.0%, and 21.8%, respectively (P < 0.001; Fig. 3e). The 3-year OS rates were 84.0%, 70.0%, and 21.8%, respectively (P < 0.001; Fig. 3f).
Among the 78 patients with FLT3-ITD mutations, 46 received prophylactic/preemptive (pro/pre) sorafenib or gilteritinib, while 7 patients, due to early relapse or death (within 100 days posttransplantation), were not eligible for such treatment. Compared with those without pro-/pre-sorafenib or gilteritinib (n = 25), FLT3-ITD-mutated patients who received these treatments (n = 46) exhibited a lower CIR, with a 3-year CIR of 13.9% (95% CI = 3.4%–24.4%) compared with 20.2% (95% CI = 4.0%–36.4%), although the difference was not statistically significant (P = 0.350). They showed a significantly higher 3-year LFS of 86.1% (95% CI = 75.7%–96.5%) compared with 63.3% (95% CI = 44.4%–82.5%) (P = 0.012), and a 3-year OS of 84.6% (95% CI = 73%–96.2%) versus 62.0% (95% CI = 42.2%–81.8%) (P = 0.009). Patients with FLT3-ITD mutations who received pro-/pre-sorafenib or gilteritinib demonstrated a prognosis comparable with those without FLT3-ITD mutations. Specifically, the 3-year CIR was 13.9% compared with 5.3% (P = 0.170), the 3-year LFS was 86.1% compared with 88.1% (P = 0.909), and the 3-year OS was 84.6% compared with 87.5% (P = 0.988).
Univariable analysis did not demonstrate any significant effects of gender, donor or recipient age, WBC count, cytogenetic abnormalities, French-American-British (FAB) classification, or conditioning regimen on transplantation outcomes (Fig. 4).
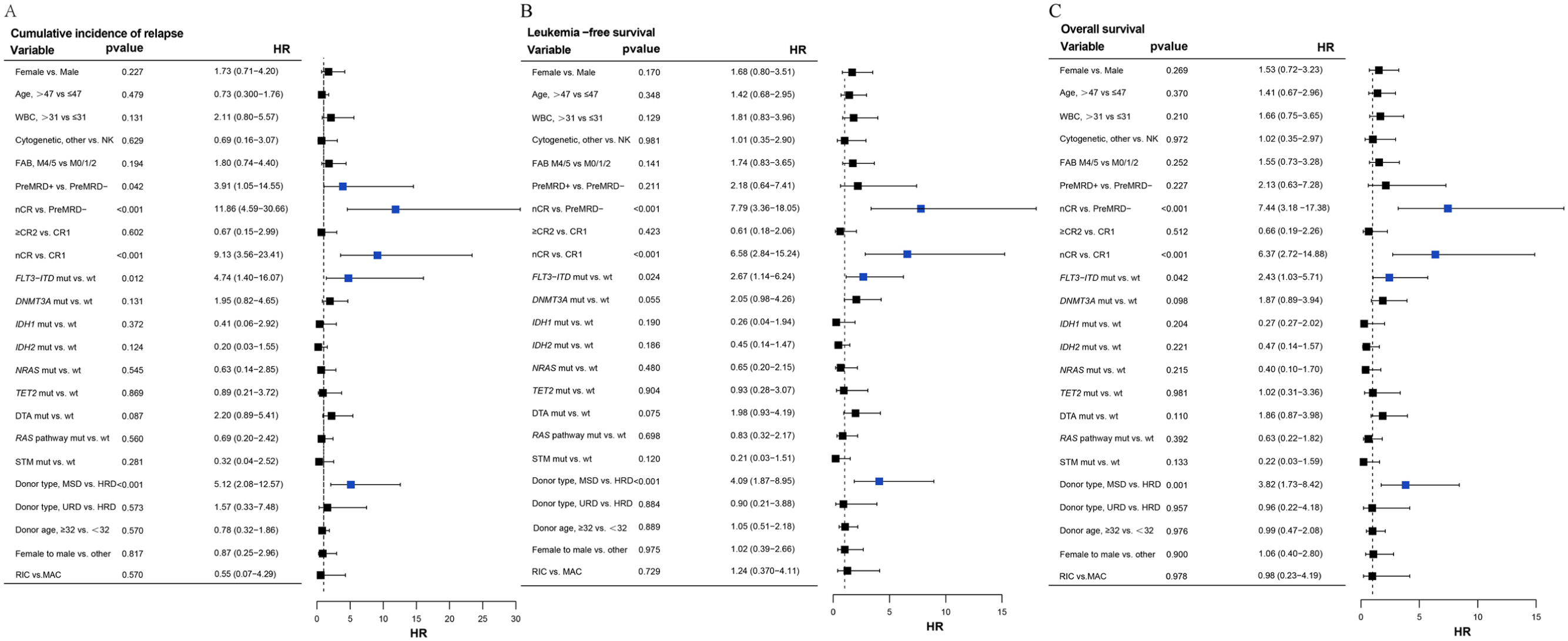
Forest plots of CIR, LFS, and OS (univariable analysis) in our patient cohort. Each row shows the HR with 95% CI of the variable. (a) Cumulative incidences of relapse. (b) Leukemia-free survival. (c) Overall survival. Blue, P < 0.05 and HR > 1.
Predictors of outcomes in NPM1-mutated AML after allo-HSCT
Multivariable analysis (MVA) for CIR revealed that patients with FLT3-ITD at diagnosis had a 4.76 times higher relapse risk (HR = 4.76, 95% CI = 1.09–20.84, P = 0.038), while recipients with MSD had a 7.85 times higher relapse risk compared with those with HRD (HR = 7.85, 95% CI = 2.98–20.67, P < 0.001). Patients who were pre-MRD positive and nCR exhibited a 9.62-fold and 7.62-fold higher risk of relapse posttransplantation, respectively, compared with pre-MRD negative patients (P < 0.001; P = 0.005). WBC count, FAB type, IDH1/2 mutations, and DTA mutations showed no significant increases in the risk of relapse (Table 2).
Multivariable analysis of transplant outcomes of all patients.
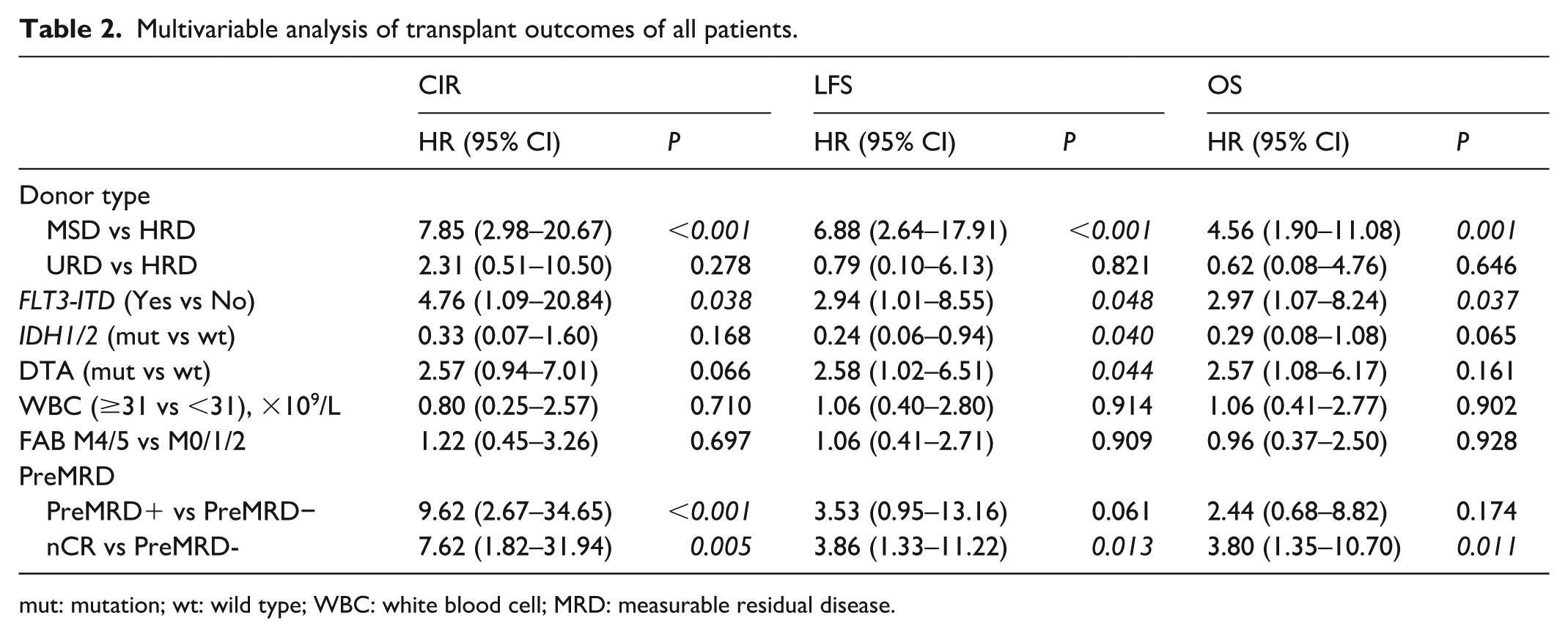
mut: mutation; wt: wild type; WBC: white blood cell; MRD: measurable residual disease.
Multivariable analysis for LFS identified that IDH1/2 mutations correlated with a 76% decreased risk of death or relapse (HR = 0.24; 95% CI, 0.06–0.94; P = 0.040). Patients with FLT3-ITD (HR = 2.94; 95% CI = 1.01–8.55, P = 0.048) or DTA (HR = 2.58, 95% CI = 1.02–6.51, P = 0.044) at diagnosis were associated with poor LFS. Recipients with MSD associated with poor LFS than those with HRD (HR = 6.88, 95% CI = 2.64–17.91, P < 0.001). nCR patients have a 3.86-fold increased risk of relapse/death after transplantation compared with pre-MRD negative patients (P = 0.013) (Table 2).
The multivariable model for OS indicated that FLT3-ITD was associated with a 2.97-fold increased risk of death (P = 0.037). Concomitant mutations in IDH1/2 were associated with a decreased risk of death, although this did not reach statistical significance (HR = 0.29; 95% CI = 0.08–1.08; P = 0.065). Recipients with MSD had a 4.56-fold increased risk of death compared with those with HRD (HR = 4.56; 95% CI = 1.90–11.08, P = 0.001). Patients with nCR exhibit a 3.80 times higher mortality risk posttransplant than those with pre-MRD negative status (HR = 3.80, 95% CI = 1.35–10.70, P = 0.011) (Table 2).
Discussion
This retrospective study examined the clinical characteristics and outcomes of adult patients with NPM1-mutated AML who underwent allo-HSCT at our institution and assessed the influence of various clinico-biological and clinical factors on their prognosis. We identified that MSD donor, FLT3-ITD, concurrent FLT3-ITD and DTA mutations, and nCR status at allo-HSCT were associated with poorer outcomes.
Among the various genetic mutations implicated in the development of AML, NPM1 and FLT3-ITD mutations are the most commonly identified in adult AML patients. In nontransplant settings, the combination of NPM1 mutations and FLT3-ITD is associated with a significantly poorer prognosis 15 . Mutations in DNA methylation regulators are prevalent in NPM1-mutated AML and occur in approximately 70% of AML cases. Notably, DNMT3A mutations are detected in approximately 50% of these cases, whereas TET2, IDH1, and IDH2 mutations occur in approximately 15% of cases1,2,16,17. Oñate et al. 18 reported that DNMT3A mutations in intermediate-risk cytogenetics NPM1-mutated AML did not significantly affect overall outcomes. Triple mutations involving NPM1, FLT3-ITD, and DNMT3A were associated with higher CIR and lower LFS 19 . In contrast, co-mutations of NPM1 and IDH1/2 have been associated with enhanced LFS and OS20–22. Rivera et al. 23 found that co-mutations with RAS did not significantly alter outcomes in comparison to RAS wild-type AML.
In the context of transplantation, Zhou et al. found that NPM1-mutated patients co-occurring with FLT3-ITD mutations had a 2-year CIR of 17.8% (95% CI = 5.7%–30.0%), which was higher than the 10% (95% CI = 0%–21.0%) reported in FLT3-ITD wild-type patients, although this difference was not statistically significant. According to the European Society for Blood and Marrow Transplantation (EBMT), IDH mutations are associated with improved prognosis in AML patients undergoing allo-HSCT 24 . Our study found that NPM1-mutated AML patients with FLT3-ITD or both FLT3-ITD and DTA mutations exhibited increased CIR and decreased LFS and OS. Patients co-occurring with IDH1/2 mutations exhibited notably lower CIR and higher LFS and OS than those with IDH1/2 wild-type. Mutations in DNMT3A, TET2, DTA, NRAS, or RAS-pathway alone did not modify the prognosis of NPM1-mutated AML.
Chan et al. 25 found that NPM1-mutated AML with mutations highly specific to secondary AML (secondary-type gene mutations) had a poorer prognosis. To date, ELN2022 has not categorized AML with STM concurrent with NPM1 mutations as an adverse effect. Eckardt et al. 26 found that STM mutations did not influence posttransplantation outcomes in patients with NPM1 mutations. Similarly, we did not find any impact of STM on the clinical outcomes of patients with NPM1-mutated AML undergoing allo-HSCT. The presence of adverse-risk cytogenetic abnormalities in NPM1-mutated AML is now classified as an adverse risk according to the ELN20227,27. However, in the context of allo-HSCT, we did not observe any effect of adverse-risk cytogenetic abnormalities on the patient outcomes.
Our study revealed that patients with NPM1-mutated AML undergoing MSD allo-HSCT had significantly higher CIR and reduced LFS and OS than those receiving URD or HRD transplants. Notably, baseline characteristics, including ELN risk stratification, pretransplant MRD status, disease status, and FLT3-ITD, were comparably distributed among the MSD, URD, and HRD cohorts. The inferior outcomes associated with MSD-allo-HSCT may be attributed to a less pronounced graft-versus-leukemia (GVL) effect compared with URD or HRD transplants, potentially due to reduced efficacy in eradicating residual leukemic cells28,29. For patients with NPM1 mutations who require transplantation, choosing not to use a matched sibling donor may be associated with improved outcomes.
Xuan et al. 30 conducted an open-label, multicentre, randomized, phase 3 trial and found that patients with NPM1 and FLT3-ITD co-mutations who received sorafenib maintenance post-allo-HSCT exhibited improved 5-year OS and CIR compared with those in the nonsorafenib maintenance group. Patients with mutated NPM1 and FLT3-ITD benefited significantly from sorafenib maintenance posttransplantation 31 . A prospective study by Levis et al. 32 indicated that relapse-free survival after allo-HSCT was higher in the gilteritinib group than in the placebo group, although the difference was not statistically significant. Our analysis revealed that patients with co-mutations of NPM1 and FLT3-ITD who underwent pre/pro sorafenib or gilteritinib treatment appeared to achieve outcomes closer to those of NPM1-mutated patients without FLT3-ITD mutations, and better than those who did not receive these treatments. However, given the small sample sizes and retrospective design, these findings are hypothesis-generating and warrant validation in prospective trials with larger cohorts. Mathew et al. 33 discovered that sorafenib enhances graft-versus-leukemia effects by stimulating IL-15 production in FLT3-ITD-mutated leukemia cells. Other studies have indicated that sorafenib is effective in patients without FLT3 mutations34,35. In the future, the use of sorafenib or gilteritinib as maintenance therapy for high-risk relapse populations may improve posttransplant outcomes.
This study has several inherent limitations. First, its retrospective, single-center design introduces inevitable selection bias. Second, the overall cohort is small and markedly imbalanced by donor type, substantially limiting the precision and reliability of comparisons for GVHD and relapse. Third, because of its retrospective nature, detailed information was not available, such as specific NPM1 mutation types and variant allele frequency (VAF) levels. Despite these limitations, our findings underscore the importance of understanding the complex interplay between genetic mutations and clinical features in predicting outcomes in patients with NPM1-mutated AML undergoing allo-HSCT. Further large-scale studies are warranted to validate these results and refine the prognostic models for this patient population.
Conclusion
This study retrospectively analyzed the clinical characteristics and outcomes of 139 adult patients with NPM1-mutated AML who underwent allo-HSCT. We identified several key prognostic factors, including FLT3-ITD and DTA mutations and pre-HSCT MRD status, which were associated with poorer outcomes. Notably, the graft-versus-leukemia effect may be more pronounced in URD/HRD transplants than in MSD-allo-HSCT. Our findings underscore the importance of personalized therapeutic strategies that consider genetic profiles and donor types in patients with NPM1-mutated AML undergoing allo-HSCT. The data from this study can serve as a foundation for future prospective trials aimed at refining prognostic models and optimizing treatment approaches for this leukemia subtype.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251414205 – Supplemental material for Prognostic impact of clinico-genetic characteristics and donor types in NPM1-mutated AML undergoing allogeneic hematopoietic stem cell transplantation
Supplemental material, sj-docx-1-cll-10.1177_09636897251414205 for Prognostic impact of clinico-genetic characteristics and donor types in NPM1-mutated AML undergoing allogeneic hematopoietic stem cell transplantation by Xiaolin Yuan, Yibo Wu, Tingting Yang, Panpan Zhu, Xiaoyu Lai, Lizhen Liu, Luxin Yang, Jimin Shi, Jian Yu, Yanmin Zhao, Weiyan Zheng, Jie Sun, Wenjun Wu, Yi Zhao, Zhen Cai, He Huang, Shanshan Pei and Yi Luo in Cell Transplantation
Footnotes
Acknowledgements
Not applicable.
Ethical Approval
This study was approved by the Ethics Review Committee of the First Affiliated Hospital of Zhejiang University School of Medicine in China (Approval No. IIT20241018A; 4 September 2024).
Consent to Participate
Informed consent was obtained from patients in accordance with the Declaration of Helsinki.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China (Grant No. 82170205).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The study data that support the findings can be obtained from the corresponding author upon reasonable request.
AI Declaration
The authors declare that they have not use AI‑generated work in this manuscript.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
