Abstract
The impact of early tacrolimus (TAC) blood levels on acute graft-versus-host disease (aGVHD) and transplant outcomes in adults undergoing haploidentical hematopoietic stem cell transplantation (haplo-HSCT) with posttransplant cyclophosphamide (PTCy) is incompletely investigated. We retrospectively analyzed 161 T-cell–replete haplo-HSCT with PTCy, TAC, and mycophenolate-mofetil. TAC trough levels from weeks 1–2 (w1/2) and weeks 3–4 (w3/4) posttransplant were categorized as “Low” or “High” using a threshold of 10 ng/ml. Outcomes assessed included grade III–IV acute graft-versus-host-disease (aGVHD), nonrelapse mortality (NRM), relapse, and overall survival (OS). Multivariate analyses controlled for relevant patient and transplant factors. Higher w1/2 TAC (≥10 ng/ml) in weeks 1–2 had no association with aGVHD III/IV (35% vs. 35%, P = 0.71). Higher TAC levels during w3/4 were associated with a trend toward decreased aGVHD III–IV incidence (8% vs. 20%; P = 0.09). Multivariate analysis confirmed w3/4 TAC levels as protective against aGVHD III/IV (sub-Hazard Ratio [sHR] = 0.83, 95% CI: 0.70–0.98, P = 0.03) and NRM (sHR = 0.82, 95% CI: 0.71–0.95, P = 0.01), while w1/2 TAC levels had no significant impact on the above outcomes. TAC levels did not significantly impact OS or relapse. We conclude that following PTCy-based haplo-HSCT, higher TAC levels during weeks 3–4 may decrease aGVHD and NRM.

Keywords
Introduction
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is a potentially curative therapy for many hematologic malignancies and other hematologic disorders 1 . However, its success is limited by the risk of graft-versus-host disease (GVHD) and other transplant-related toxicities 1 . GVHD, particularly acute GVHD (aGVHD), is a leading cause of morbidity and nonrelapse mortality (NRM) after transplant 2 . Historically, the lack of suitably HLA-matched donors posed a significant barrier to allogeneic transplant for a substantial fraction of patients. Haploidentical donors (usually a partially HLA-mismatched family member) greatly expand the donor pool, but early attempts at haploidentical HSCT were associated with unacceptably high rates of graft rejection, severe GVHD, and treatment-related mortality 1 .
The introduction of posttransplant cyclophosphamide (PTCy) as GVHD prophylaxis has revolutionized haploidentical transplantation, effectively overcoming the HLA barrier 3 . High-dose PTCy selectively eliminates proliferating alloreactive T cells shortly after infusion, thereby preventing GVHD while sparing hematopoietic stem cells and nondividing memory T cells. Clinical studies have demonstrated that haploidentical HSCT with PTCy (often combined with additional immunosuppression) can achieve rates of acute and chronic GVHD that are as low as or lower than those observed in HLA-matched transplants 4 . Reported incidences of grade II–IV aGVHD in T cell–replete haplo-HSCT with PTCy range from approximately 15 to 40%, and chronic GVHD rates range from ~0 to 30%, depending on conditioning intensity and graft source 4 . Outcomes after haplo-HSCT with PTCy have approached those of matched donor transplantation, with comparable GVHD-free survival in some analyses 2 .
A standard GVHD prophylaxis platform for haploidentical HSCT incorporates PTCy (typically given on days +3 and +4 posttransplant) followed by a calcineurin inhibitor (most commonly tacrolimus) and mycophenolate mofetil (MMF) starting on day +5. Tacrolimus (TAC) is a calcineurin inhibitor that binds to FK506-binding protein and inhibits T-lymphocyte activation by blocking IL-2 transcription. In combination with MMF (an antimetabolite that inhibits T and B cell proliferation via guanine nucleotide depletion), tacrolimus plays a central role in preventing GVHD after allografting. Tacrolimus has a narrow therapeutic index, and its optimal dosing requires careful monitoring of blood concentrations to balance efficacy and toxicity. Supratherapeutic TAC levels can result in significant toxicities, including nephrotoxicity, thrombotic microangiopathy, neurotoxicity (e.g. seizures, posterior reversible encephalopathy syndrome), and increased infection risk. Conversely, subtherapeutic levels may fail to adequately prevent GVHD. In conventional matched donor HSCT (e.g. with tacrolimus/methotrexate prophylaxis), target trough concentrations of tacrolimus are often 5–15 ng/ml (some protocols 10–20 ng/ml), based on studies showing better GVHD prevention within this range. For instance, tacrolimus with methotrexate or sirolimus has well-established target levels in matched sibling or unrelated donor transplants. In the haploidentical PTCy setting, however, the optimal tacrolimus target level is not well defined. Some centers use a flat low-dose tacrolimus approach post-PTCy, hypothesizing that PTCy allows for lower calcineurin inhibitor exposure without increasing GVHD 3 .
There is an increasing interest in whether tacrolimus blood concentration influences GVHD outcomes in the context of PTCy-based regimens. In HLA-matched transplant settings (without PTCy), several studies have suggested that higher tacrolimus levels early posttransplant are associated with a reduced risk of aGVHD. Ganetsky et al. 5 reported that achieving higher tacrolimus troughs in the first two weeks after reduced-intensity HSCT was associated with a reduction in grade II–IV aGVHD incidence. Similarly, Huynh et al. 6 found that maintaining TAC >14 ng/ml in the first 2 weeks correlated with a significantly lower GVHD rate in unrelated donor transplants. Conversely, patients with low early tacrolimus exposure have been shown to experience higher GVHD rates. For example, a recent analysis in matched donor HSCT showed that CYP3A5 high-metabolizer genotype (which leads to lower tacrolimus troughs) was associated with significantly increased incidence of aGVHD: 61.5% versus 24.5% for grade II–IV aGVHD in fast metabolizers versus poor metabolizers (P = 0.008) 7 . These findings underscore that inadequate TAC immunosuppression can predispose to GVHD in the absence of other GVHD-suppressing interventions.
In haploidentical HSCT with PTCy, it remains unclear whether achieving higher tacrolimus levels provides additional protection against GVHD, or if PTCy’s effect makes tacrolimus levels less critical. It is conceivable that if tacrolimus exposure is too low, GVHD might still emerge even with PTCy, whereas excessively high levels might only increase toxicity without further reducing GVHD. Only a few studies have directly examined tacrolimus level-effects in PTCy-based prophylaxis. Yao et al. 3 recently analyzed a PTCy/tacrolimus-based cohort (mixed haploidentical and mismatched unrelated donors) and found no significant difference in acute or chronic GVHD incidence between patients with initial tacrolimus steady-state levels <10 ng/ml versus ≥10 ng/ml. Notably, higher tacrolimus levels (≥10) in that study were associated with increased infectious complications (especially viral infections) without improvement in GVHD or survival 3 . Another report suggested that while a tacrolimus trough ≥10 ng/ml in week 1 post-HSCT may reduce aGVHD, maintaining levels >11–12 ng/ml beyond the first week could increase relapse risk 8 . Given these mixed observations, further investigation is needed to clarify the impact of tacrolimus level modulation on transplant outcomes in the haploidentical setting.
Objective: in this study, we aimed to evaluate the influence of tacrolimus levels during the early posttransplant period on GVHD and other outcomes in haploidentical HSCT recipients. Specifically, we compared tacrolimus exposure in the first 2 weeks versus the last 2 weeks of the first month posttransplant, and analyzed how TAC levels in these intervals correlated with the incidence of aGVHD, as well as with NRM, overall survival (OS), and relapse.
Method
Study design and patients
We conducted a retrospective cohort study of adult patients who underwent haploidentical allogeneic HSCT at our center. Inclusion criteria were (1) patients aged ≥18 years; (2) receipt of a haploidentical bone marrow or peripheral blood stem cell transplant for a hematologic malignancy; (3) GVHD prophylaxis consisting of PTCy on days +3 and +4, followed by tacrolimus and MMF; (4) availability of complete tacrolimus level data for the first 4 weeks posttransplant; and (5) engraftment achieved. We excluded patients who did not engraft or who died or relapsed before day +28. Patient data were obtained from institutional databases and chart review with appropriate ethics committee approval (Nb.1037/2025, ethics committee upper Austria). The study population consists of transplantation cases that were carried out at our center between 1 January 2015 and 31 July 2024 while the follow-up data would be collected until 31 December 2024.
GVHD prophylaxis and tacrolimus monitoring
All patients received high-dose cyclophosphamide 50 mg/kg on days +3 and +4 posttransplant as GVHD prophylaxis, per the Baltimore PTCy protocol. Tacrolimus (usually starting after the first PTCy dose on the evening of day +3) was administered orally. The initial dosing of tacrolimus was weight-based (2 × 0.08 mg/kg/day). Dosing was adapted according to TAC trough levels, which were performed three times weekly, to achieve a target level of 10–12 ng/ml during the first month. MMF was given orally at 15 mg/kg three times daily from day +5 through day +35 in the absence of GVHD. For the purpose of this analysis, we collected all tacrolimus trough concentrations measured during the first 28 days posttransplant.
We then calculated mean weekly TAC levels for each patient for week 1 (defined as days +0 to +7 posttransplant; note tacrolimus typically started on day +3, so week 1 values reflect days +4 to +7), week 2 (days +8 to +14), week 3 (days +15 to +21), and week 4 (days +22 to +28). From these, we derived aggregate mean TAC levels over the first 2 weeks (weeks 1–2) and the last 2 weeks of the first month (weeks 3–4).
We chose to examine these two time intervals to differentiate the very early (w1/2) post-PTCy period (when tacrolimus is just starting and PTCy is exerting its effect) from the early (w3/4) peri-engraftment period within the first month. For each interval, patients were classified into “Low TAC” or “High TAC” level groups based on a cutoff of 10 ng/ml. This threshold approximately corresponded to the median tacrolimus trough concentration in our cohort during the first 4 weeks.
Outcomes and definitions
AGVHD was classified according to the Glucksberg or modified Glucksberg criteria9 –11. We focused on grade II–IV aGVHD (grades III–IV being severe aGVHD) as clinically meaningful acute GVHD events Secondary outcomes included the following:
NRM: death from any cause without prior relapse of the primary disease. Relapse/progression was treated as competing risks for NRM.
NRM associated with aGVHD (grades II–IV): death from any cause without prior relapse of the primary disease in patients with grades II–IV aGVHD, with relapse/progression treated as competing risks for NRM.
Relapse incidence: relapse or progression of the underlying hematologic malignancy. NRM was the competing risk for relapse.
OS: time from transplantation to death from any cause. Patients alive at last follow-up were censored.
The relevant clinical and laboratory data were extracted from electronic medical records and stored in a pseudonymized, password-protected database. The primary exposure was weekly Tacrolimus blood levels, with three measurements per week used to calculate the average values for the first two weeks and for the third and fourth weeks. Additional covariates included recipient and donor age, CMV serostatus, conditioning regimen intensity, the use of Methotrexate (MTX) compared with other medications, and HLA mismatch status.
Disease risk classification used currently accepted scoring systems. Patients with malignant disease in first complete remission or with low-risk myelodysplastic syndrome (MDS) or myeloproliferative neoplasia (MPN) were classified as the low-risk group. The intermediate-risk group included patients in the second complete remission or those with intermediate-risk MDS/MPN. High-risk patients were those with active disease at the time of transplantation, high-risk MDS/MPN, or relapsed disease following a prior allo-HSCT.
Statistical analysis
We first performed descriptive analyses of the cohort. Continuous variables were summarized by median and range, and categorical variables by counts and percentages. Baseline characteristics were compared between the Low TAC versus High TAC groups (for each time interval) using the Mann-Whitney U test for continuous variables and chi-square/Fisher’s exact tests for categorical variables.
Time-to-event outcomes were analyzed with appropriate censoring and consideration of competing risks. The cumulative incidence of aGVHD III–IV, relapse, NRM, and chronic GVHD was estimated accounting for competing risks (e.g. treating death or relapse as competing events for GVHD, and vice versa) using the method of Fine and Gray. The Gray’s test was used to compare cumulative incidence curves between TAC level groups for these outcomes. Overall survival was estimated using the Kaplan–Meier method, and survival between groups was compared using the log-rank test.
To examine the effect of early (day 0–14) and late (day 15–28) TAC exposure without introducing immortal-time bias, landmark analyses were set at days +14 and +28. For each endpoint, we first excluded patients who had already reached that specific endpoint- or had died—before the landmark (e.g. prior grade III–IV aGVHD for the GVHD model, prior relapse for the relapse model); other events were not grounds for exclusion. All remaining patients were then classified as Low (≤10 ng/ml) or High (>10 ng/ml) according to their mean TAC trough in the relevant preceding interval, and outcomes were analyzed from the landmark forward.
We performed univariate analyses for each outcome using Cox proportional hazards models for OS, and Fine-Gray subdistribution hazard models for aGVHD III–IV, NRM, relapse, and cGVHD (subdistribution hazard ratios, HRs, reported for the latter). Tacrolimus level was examined both as a categorical variable (High vs. Low, with Low as reference) and as a continuous variable (per 1 ng/ml increment in mean TAC level)
In the continuous models, TAC level was log-transformed if necessary to check for linearity of hazard assumption; however, results are presented per 1 ng/ml for ease of interpretation given an approximately linear effect in the range observed. We then constructed multivariate models to adjust for potential confounders identified a priori or those that differed between TAC groups. A stepwise selection was used, ensuring that the main predictor (TAC group) remained in all models. Results are reported as HRs or subdistribution HR with 95% confidence intervals (CI). All statistical analyses were performed using R version 4.4.2 and Packages survival, survminer, ggplot2, cmprsk, MASS and dplyr12 –18.
Results
The cohort characteristics are summarized in Table 1. The results for the univariate and multivariate analyses are summarized in Tables 2 and 3.
Baseline characteristics of patients undergoing haploidentical HSCT with posttransplant cyclophosphamide-based GVHD prophylaxis (N = 161).
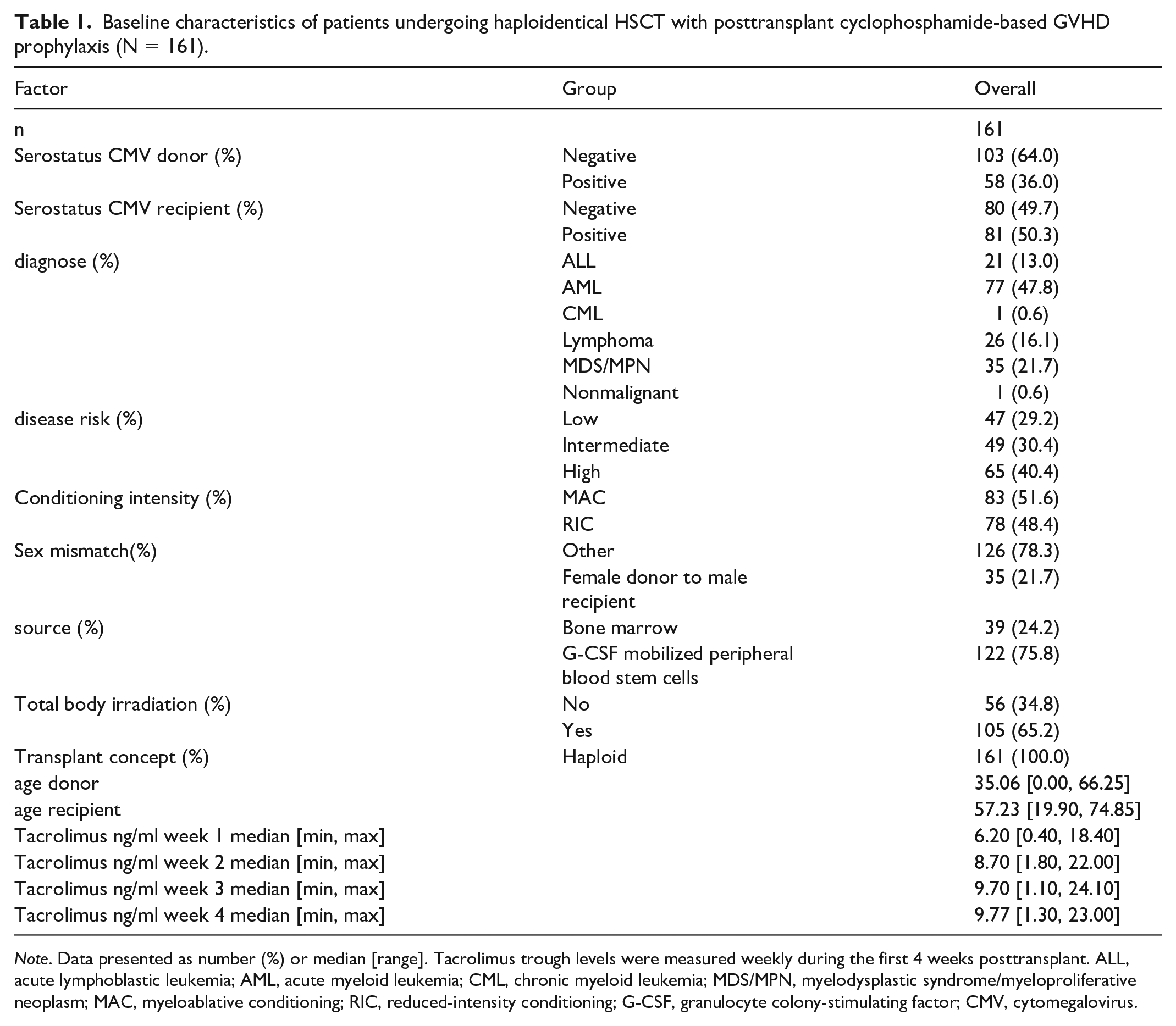
Note. Data presented as number (%) or median [range]. Tacrolimus trough levels were measured weekly during the first 4 weeks posttransplant. ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; CML, chronic myeloid leukemia; MDS/MPN, myelodysplastic syndrome/myeloproliferative neoplasm; MAC, myeloablative conditioning; RIC, reduced-intensity conditioning; G-CSF, granulocyte colony-stimulating factor; CMV, cytomegalovirus.
Univariate analysis of transplant outcomes based on tacrolimus (TAC) trough levels during early (days 1–14) and late (days 15–28) posttransplant periods.

Note. Outcomes shown as cumulative incidences or survival probabilities at day 100 posttransplant. OS, overall survival; CIR, cumulative incidence of relapse; aGVHD, acute graft-versus-host disease grades II–IV; NRM, nonrelapse mortality.
Multivariate analyses evaluating the impact of tacrolimus (TAC) levels on transplant outcomes.

Note. Hazard ratios (HR) and subdistribution hazard ratios (sHR) with 95% confidence intervals (CIs) indicate the effect per unit increase in TAC trough level during early (days 1–14) and late (days 15–28) posttransplant periods. Significant results (P < 0.05) highlighted. OS, overall survival; NRM, nonrelapse mortality; CIR, cumulative incidence of relapse; aGVHD, acute graft-versus-host disease grades III–IV.
Patient characteristics
A total of 161 haploidentical HSCT recipients met the inclusion criteria. Table 1 summarizes the baseline demographics and transplant characteristics, including CMV serostatus, disease types, conditioning, and donor/recipient ages. The majority had AML (47.8%), followed by ALL (13.0%), lymphoma (16.1%), MDS/MPN (21.7%), and others (CML, nonmalignant). Approximately 75.8% received PBSC grafts, while 24.2% received BM. Conditioning regimens varied among MAC or RIC. The median donor age was 35. and the median recipient age was 57.2. Tacrolimus levels in the first four weeks are also shown (TAC week 1 through TAC week 4).
Acute GVHD incidence
On univariate analysis (Table 2), patients with High w1/2 TAC (≥10 ng/ml) had an aGVHD III–IV incidence of 35%, compared with 35% in those with Low w1/2 TAC (<10 ng/ml) (P = 0.71, Fig. 1a). During the w3/4 period (days 15–28), the incidence of aGVHD III–IV was lower in the High TAC group (8%) compared with 20% in the Low TAC group (P = 0.09, Fig. 1b).

Cumulative incidence of grade III–IV acute GVHD, stratified by tacrolimus levels (≥10 ng/ml vs. <10 ng/ml). (a) Early tacrolimus exposure (days 1–14). (b) Late tacrolimus exposure (days 15–28). Curves were estimated using the Fine-Gray method, with Gray’s test for between-group comparisons. Numbers at risk are shown below each plot.
In multivariate models (Table 3) treating TAC as a continuous variable, w1/2 TAC was not associated with aGVHD III–IV risk (sHR = 1.01; 95% CI: 0.88–1.18; P = 0.85), but higher w3/4 TAC levels were significantly protective (sHR = 0.83; 95% CI: 0.70–0.98; P = 0.03). Thus, while w1/2 tacrolimus exposure (weeks 1–2) did not significantly affect aGVHD III–IV incidence, maintaining adequate TAC levels in weeks 3–4 was associated with lower aGVHD III–IV rates.
Overall, tacrolimus trough levels in the first month did not significantly impact aGVHD III–IV incidence. Only a few patients developed very early aGVHD prior to day 14, and those low-TAC cases were too small in number to draw firm conclusions. Landmark and intent-to-treat analyses yielded similar findings.
In an interaction model between time and TAC level, the SHR of 0.87 (P = 0.08) for the interaction term indicates that for each unit increase in the average TAC level, the risk of aGVHD III–IV in weeks 3 and 4 decreases by approximately 13% more compared with its effect in weeks 1 and 2.
NRM
As shown in Table 2, the cumulative incidence of NRM at 1 year was 9% versus 13% (P = 0.91) in the Low versus High TAC groups for the w1/2 period, and 12% versus 8% (P = 0.24) for the late period. In multivariate analysis (Table 3), w1/2 TAC did not significantly influence NRM (sHR = 1.06, 95% CI: 0.90–1.25; P = 0.47). However, w3/4 TAC was independently associated with a reduced NRM risk (sHR = 0.82, 95% CI: 0.71–0.95; P = 0.01).
When specifically examining NRM associated with aGVHD (grades II–IV), results were similar: higher w3/4 TAC levels (weeks 3–4) correlated with a significantly lower cumulative incidence of aGVHD-related NRM (Fig. 2b), whereas w1/2 TAC did not (Fig. 2a).

Cumulative incidence of nonrelapse mortality (NRM) associated with grade II–IV acute GVHD, stratified by tacrolimus levels (≥10 ng/ml vs. <10 ng/ml). (a) Early tacrolimus exposure (days 1–14). (b) Late tacrolimus exposure (days 15–28). Curves were estimated using the Fine-Gray method, with Gray’s test for between-group comparisons. Numbers at risk are shown below each plot.
Relapse incidence
The cumulative incidence of relapse (CIR) at 1 year was 23% versus 14% (P = 0.49) for w1/2 Low versus High TAC. w3/4 TAC levels also did not significantly affect relapse (21% vs. 21%; P = 0.64). In multivariate analysis, tacrolimus levels in weeks 1–2 were not significantly associated with CIR (sHR = 0.89, 95% CI: 0.75–1.05, P = 0.16), and tacrolimus levels in weeks 3–4 also showed no significant association with CIR (sHR = 1.07, 95% CI: 0.97–1.18, P = 0.21).
OS
On univariate analysis, patients with low versus high tacrolimus (<10 vs. ≥10 ng/ml) had 1-year OS rates of 77% versus 79% (P = 0.51) for the w1/2 landmark (Fig. 3a) and 75% versus 79% (P = 0.25) for the w3/4 landmark (Fig. 3b), indicating no statistically significant difference. In multivariate analysis, higher w1/2 TAC levels at weeks 1–2 (sHR = 0.90, 95% CI: 0.78–1.04, P = 0.13) and w3/4 (sHR = 0.91, 95% CI: 0.82–1.01, P = 0.08) were not significantly associated with improved survival. Thus, tacrolimus exposure did not emerge as a significant predictor of OS in either univariate or multivariate models.

Kaplan–Meier estimates of overall survival (OS), stratified by tacrolimus levels (≥10 ng/ml vs. <10 ng/ml). (a) Early tacrolimus exposure (days 1–14). (b) Late tacrolimus exposure (days 15–28). Numbers at risk are shown below each plot. Survival differences were compared using the log-rank test.
Discussion
Our retrospective study examined how tacrolimus trough levels at different early posttransplant time points influence aGVHD and transplant outcomes in adult haploidentical HSCT with PTCy-based GVHD prophylaxis. In our multivariate analysis using tacrolimus as a continuous variable, early tacrolimus exposure (weeks 1–2) was not significantly associated with aGVHD risk; however, higher late tacrolimus exposure (weeks 3–4) reduced the risk of severe aGVHD (grades III–IV) and associated NRM. These findings suggest a critical impact of tacrolimus levels specifically during weeks 3–4 posttransplant. Tacrolimus levels also showed no clear impact on OS in our cohort.
Haplo-HSCT with PTCy: our observation that maintaining higher tacrolimus levels did not markedly reduce aGVHD in the haploidentical setting aligns with reports from other PTCy-based protocols. Yao et al., analyzing initial steady-state tacrolimus levels measured around day +7 post-HSCT, found no significant difference in grade II–IV aGVHD incidence between patients with lower tacrolimus levels (<10 ng/ml) and those with higher levels (≥10 ng/ml). They reported comparable rates of aGVHD across tacrolimus level groups on multivariable analysis, suggesting that escalating tacrolimus beyond standard ranges offers no additional GVHD protection in this context 3 . Similarly, an analysis of haplo-HSCT recipients by Trepte et al., 19 who measured tacrolimus levels weekly from day +7 through day +35 (weeks 1–4), concluded that no protective benefit of higher tacrolimus concentrations during weeks 1–2 was seen in haploidentical HSCT with PTCy for aGVHD.
Tacrolimus Levels and GVHD Risk: prior studies suggest subtherapeutic tacrolimus can predispose to GVHD even if supratherapeutic levels provide no further benefit 20 . For example, Cheung et al. 21 observed that maintaining tacrolimus trough levels consistently within the therapeutic range (~7–10 ng/ml) specifically during days 1–21 post-HSCT was associated with lower aGVHD incidence. This implies there is a minimum effective concentration needed for GVHD prophylaxis, but once that threshold is met, additional tacrolimus may yield diminishing returns in PTCy-based regimens. Indeed, Gao et al. 22 reported a ~29% grade II–IV aGVHD incidence in haplo-HSCT with tacrolimus maintained in the 10–15 ng/ml range, a rate comparable to our findings. Taken together, the evidence suggests that standard-range tacrolimus exposure is sufficient when used alongside PTCy, and pushing levels higher does not significantly enhance GVHD prevention in adults undergoing haploidentical transplant.
A potential explanation for this time-dependent effect is that the potent immunosuppressive action of PTCy during the first 1–2 weeks posttransplant may overshadow any incremental benefit from higher tacrolimus levels. As PTCy’s influence wanes in weeks 3–4, tacrolimus exposure becomes more crucial for modulating residual T-cell alloreactivity, thereby affecting aGVHD and NRM more prominently
Conventional (non-PTCy-based) Matched Donor Transplants: In contrast to the haploidentical / PTCy-based experience, studies in the mainly HLA-matched transplant setting without PTCy have shown a clearer correlation between tacrolimus levels and GVHD outcomes. In contrast to the haploidentical experience, studies in HLA-matched transplant settings have shown a clearer correlation between tacrolimus levels and GVHD outcomes. For instance, Ganetsky et al., evaluating weekly tacrolimus trough levels specifically during weeks 1–4, found that higher tacrolimus troughs (>12 ng/ml) specifically in week 1 post-HSCT were significantly associated with reduced aGVHD rates (grades II–IV) without increasing relapse risk in matched related and unrelated donor transplants. Their data suggested roughly a one-third reduction in GVHD risk when tacrolimus levels were ~14–15 ng/ml early on, without an increase in relapse, leading some to recommend targeting the upper end of the therapeutic range during this critical period5,19. See also Nikoloudis et al. 23 for CSA-based prophylaxis during weeks 1–2. Our results did not mirror this pattern in the haplo-PTCy setting. Differences between PTCy-based haploidentical grafts and conventional platforms may partly explain these divergent results. PTCy effectively eliminates alloreactive T cells, which may reduce the reliance on very high calcineurin inhibitor levels to prevent GVHD. Thus, while tacrolimus exposure is crucial in any transplant, its optimal therapeutic window may be lower in PTCy-supported haplo-HSCT compared with transplants without PTCy.
Transplant Outcomes and Toxicities: Consistent with prior haplo-HSCT studies, we found no significant impact of tacrolimus level on OS or relapse. Yao et al. 3 likewise reported similar 2-year survival between tacrolimus <10 and ≥10 ng/ml groups. They did note a trend toward higher relapse in the high-tacrolimus cohort (35% vs. 22% relapse at 2 years), hinting that excessive immunosuppression might marginally impair graft-versus-leukemia effects, though this difference was not significant after adjustment. Our analysis did not demonstrate a clear relapse difference, but the possibility that lower tacrolimus exposure could preserve anti-tumor immunity without exacerbating GVHD remains an important consideration. However, higher tacrolimus levels in our study were associated with expected toxicities, in line with known pharmacology. Prior reports show that tacrolimus >10–15 ng/ml can increase infections and organ toxicity. Yao et al. 3 observed significantly more viral infections (e.g. cytomegalovirus reactivation) in patients with tacrolimus ≥10 ng/ml. We similarly noted trends toward greater infection and renal impairment in patients with higher tacrolimus exposure (though detailed toxicity data were outside this study’s primary scope). These findings reinforce the importance of balancing immunosuppression and toxicity: maintaining tacrolimus in a moderate therapeutic range appears sufficient and safer for haploidentical HSCT with PTCy, as excessive levels confer more harm than benefit.
Our results and those of others highlight a nuanced interplay of factors that can influence how tacrolimus levels correlate with GVHD and outcomes. Several considerations could explain the consistencies and discrepancies observed across studies.
Differences in cohort makeup (adult vs. pediatric patients, malignant vs. nonmalignant diseases) and transplant protocols could impact GVHD incidence independent of tacrolimus. For example, pediatric transplants from parent donors tend to have higher GVHD risk than sibling adult donors 24 , potentially overshadowing the effect of drug levels. Similarly, graft source (bone marrow vs. peripheral blood), conditioning intensity, and HLA disparity vary between studies and can modulate baseline GVHD risk. Our cohort’s specific features (e.g. predominantly peripheral blood grafts and high-risk diagnoses) may differ from those in other reports, contributing to divergent findings.
How tacrolimus is administered and monitored can markedly influence achieved drug levels. Some centers use flat dosing (a fixed starting dose) while others use weight-based dosing; this was shown to affect early tacrolimus steady-state levels 3 . Frequent therapeutic drug monitoring with rapid dose adjustments (as in our practice) might keep most patients within target range, whereas in studies with less intensive monitoring, greater variability in levels could reveal stronger associations with GVHD. The target therapeutic range itself varies between institutions (often 5–15 ng/ml, but some use 5–10 ng/ml or higher upper limits), which can change the interpretation of “low” versus “high” tacrolimus. Timing of tacrolimus initiation is another factor: in PTCy-based haplo transplants, tacrolimus typically starts on day +5 (after cyclophosphamide), whereas in conventional grafts it starts before or at transplant. This delay in the haplo setting means early posttransplant immune reactions occur under minimal tacrolimus coverage, possibly making PTCy the dominant factor in early GVHD prevention. Such protocol differences in drug timing and level monitoring could explain why matched transplant studies (tacrolimus on board sooner) saw a bigger impact of tacrolimus levels on GVHD 19 , whereas PTCy-haplo studies (tacrolimus introduced later) did not. The analytical approaches across studies are heterogeneous. Some analyses used tacrolimus as a continuous variable (e.g. calculating HRs per 1 ng/ml increase 19 ), while others dichotomized patients by trough thresholds (such as 7 or 10 ng/ml) 21 . The definition of aGVHD end points also differed: certain studies focused on grade III–IV GVHD (severe GVHD) versus grade II–IV (any moderate-to-severe GVHD), which can yield different incidence comparisons. Our study considered clinically significant (grade II–IV) aGVHD, similar to most, but we performed multivariable competing-risk analyses to account for death as a competing event. In contrast, a simpler Kaplan–Meier or univariate approach in a smaller sample might detect or exaggerate associations due to random imbalances. For example, Yao et al. 3 observed a significant difference in relapse with tacrolimus level in univariate analysis that disappeared after adjustment, indicating how confounding factors (disease risk, donor type, etc.) can influence results. Therefore, disparities in statistical rigor, sample size, and covariate adjustment between studies likely contribute to whether a tacrolimus-GVHD relationship is found to be significant. Pharmacokinetics are notoriously variable between patients due to genetic polymorphisms and drug interactions. Populations with a higher prevalence of the CYP3A5*1 allele (which confers faster tacrolimus metabolism) will have more patients with low trough levels unless doses are increased 20 . If one study cohort had many rapid metabolizers (e.g. a different ethnic composition or younger patients with robust liver enzyme activity), subtherapeutic tacrolimus exposure might have occurred more frequently, leading to more GVHD and a clearer correlation with levels. In contrast, another study (or our own) might include routine adjustments or preemptive dosing for such patients, blunting any association. In addition, co-medications like azole antifungals (voriconazole/posaconazole), often used for prophylaxis, can elevate tacrolimus levels via CYP3A inhibition. Variations in antifungal use between centers could mean some cohorts had uniformly higher tacrolimus concentrations, potentially masking a low-level GVHD risk. In our center, nearly all patients received azole prophylaxis, which may partially explain the consistently therapeutic tacrolimus levels achieved. Such pharmacogenetic and pharmacological differences highlight why tacrolimus exposure-outcome relationships are not universal—the metabolic context matters. Tailoring tacrolimus dosing to patient-specific metabolism (through genotyping or frequent level checks) could standardize exposure and thereby reduce the detectable variability in GVHD outcomes attributable to tacrolimus.
In summary, our study contributes to a growing body of evidence that in haploidentical HSCT with PTCy, tacrolimus trough levels within the conventional therapeutic range are generally sufficient for GVHD prophylaxis. We did not find a compelling advantage to maintaining higher tacrolimus levels, which is consistent with other haplo-PTCy reports and likely stems from the dominant GVHD-protective effect of PTCy. These findings underscore the importance of context when interpreting immunosuppressant level data. They also suggest that future efforts could focus on personalizing tacrolimus therapy—for instance, using pharmacogenetic-guided dosing or AUC-based monitoring—to ensure patients achieve the necessary exposure to prevent GVHD without undue toxicity 24 . Refining tacrolimus management in this way may further optimize haploidentical transplant outcomes by balancing effective GVHD prevention with the minimization of drug-related complications.
Our study is limited by its retrospective, single-center design, which may reduce the generalizability of our findings and introduce selection bias. Although we measured tacrolimus trough levels frequently, we did not evaluate more comprehensive pharmacokinetic parameters (e.g. area under the curve) or incorporate pharmacogenetic analyses, which could yield deeper insights into individual variability in drug metabolism. Furthermore, our relatively modest sample size may have limited our power to detect smaller but clinically relevant differences in outcomes. Despite these constraints, our data provide a useful perspective on tacrolimus management in haploidentical HSCT with PTCy and form a basis for future prospective and multicenter investigations.
Conclusion
Overall, our results support a strategy of maintaining tacrolimus in the mid-therapeutic range in haplo-HSCT with PTCy, which appears to provide adequate GVHD control and favorable transplant outcomes in most patients. This approach is consistent with the experience of comparable cohorts and reinforces that extreme tacrolimus levels (either too low or too high) should be avoided. Continued investigation in larger, multicenter studies—incorporating pharmacogenetic profiling and standardized monitoring protocols Yao et al., in a large cohort of haploidentical and mismatched transplants with PTCy, found no significant difference– will be valuable to confirm these conclusions and guide best practices for immunosuppression in the haploidentical transplant setting.
Footnotes
Acknowledgements
The authors express their gratitude to all nurses, physicians, and technicians at the transplantation, cell processing and transfusion medicine facility in Linz, Austria
Research ethics and patient consent
This study was approved by the institutional ethics committee of Upper Austria (No. 1037/2025) and conducted in accordance with the Declaration of Helsinki. All patients signed an informed consent.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Author contributions
Alexander Nikoloudis and Johannes Clausen wrote the manuscript. Alexander Nikoloudis, Veronika Buxhofer-Ausch, Ameya Kunte, Christina Groiss, Lorenz Mair, Christoph Aichinger, Michaela Binder, Petra Hasengruber, Emine Kaynak, Dagmar Wipplinger, Robert Milanov, Irene Strassl, Olga Stiefel, Sigrid Machherndl-Spandl, Andreas Petzer, Ansgar Weltermann, Holger Rumpold, and Johannes Clausen analyzed data; Johannes Clausen edited the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
