Abstract
Haploidentical-related donor transplantation using posttransplant cyclophosphamide (PTCy-haplo) and cord blood transplantation (CBT) are valid alternatives for patients with hematological malignancies when HLA-matched donor transplantation (MDT) is unavailable. However, the effects of graft-versus-host disease (GVHD) on outcomes after these transplants have not been fully elucidated. Therefore, we evaluated the effects of acute and chronic GVHD on transplant outcomes after PTCy-haplo transplants and compared them with CBT and MDT. We included a total of 914 adult patients with hematological malignancies in the Kyoto Stem Cell Transplantation Group registry who received PTCy-haplo (N = 120), CBT (N = 402), and MDT (N = 392), and achieved neutrophil engraftment. A multivariate analysis revealed that grade I–II acute GVHD improved of overall survival (OS) after PTCy-haplo [hazard ratio (HR) = 0.39, P = 0.018] and CBT (HR = 0.48, P < 0.001), but not after MDT (HR = 0.80, P = 0.267) compared with patients without acute GVHD. Grade I–II acute GVHD had a trend toward reducing the risk of nonrelapse mortality (NRM) after PTCy-haplo (HR = 0.13, P = 0.060) and this positive effect was significant after CBT (HR = 0.39, P = 0.003). A negative impact of grade III–IV acute GVHD on NRM was observed after CBT and MDT, but not after PTCy-haplo. Limited chronic GVHD had a positive impact on OS after CBT and MDT, but not after PTCy-haplo. In conclusion, mild acute GVHD improved outcomes after PTCy-haplo and CBT, and limited chronic GVHD improved outcomes after CBT and MDT. These data indicated that the effects of GVHD on transplant outcomes depended on transplant platforms.
Keywords
Introduction
HLA-haploidentical hematopoietic stem cell transplantation (HSCT) procedure with posttransplant cyclophosphamide (PTCy-haplo) has spread rapidly worldwide 1 . It prevents graft-versus-host disease (GVHD) by expanding functional myeloid-derived suppressor cells and indirectly influencing regulatory T cells 2 . A series of previous studies have demonstrated that this strategy is feasible and safe with low incidences of acute and chronic GVHD and NRM3–7. Graft-versus-host disease is a serious transplant complication that substantially affects NRM after allogeneic HSCT, whereas the graft-versus-malignancy (GVM) effect is expected to be accompanied by a risk of GVHD8–11. Recent studies have suggested that the GVM effect from haploidentical donors is similar or superior to the GVM seen with HLA-identical donors for patients with acute leukemia12–14. However, little evidence is available regarding the impact of GVHD on transplant outcomes after PTCy-haplo transplantation.
Cord blood transplantation (CBT) allows multiple HLA mismatches with lower incidences of acute and chronic GVHD15–23. Grade I–II acute GVHD and limited chronic GVHD have been shown to have positive effects after CBT, indicating that mild GVHD not only reduces the risk of relapse but also lowers both nonrelapse and overall mortality24–26. This could provide a wider therapeutic window between the reduction of relapse and increase in transplant-related mortality associated with the development of GVHD.
Therefore, we performed a multicenter retrospective analysis to reveal the effects of acute and chronic GVHD on outcomes after PTCy-haplo transplant and compared them with CBT and matched donor transplantation (MDT). In addition, we evaluated the impact of cyclophosphamide dose on outcomes after PTCy-haplo transplant and its subsequent lymphocyte recovery. Identifying the impact of GVHD on PTCy-haplo helps achieve optimal anti-tumor effects while having enough immunosuppression to prevent severe GVHD.
Materials and Methods
Data Source
All transplantation data were submitted to the Japanese Data Center for Hematopoietic Cell Transplantation (JDCHCT) after all patients at each center gave their written informed consent. These data were redistributed upon request from the Kyoto Stem Cell Transplantation Group (KSCTG), which is a multicenter group of 18 transplantation centers in Japan. We included 1,140 adult patients aged 16 to 75 years with hematological malignancies who received a first allogeneic HSCT, including PTCy-haplo, CBT, and MDT between 2015 and 2021. We excluded 204 patients who lacked data on HLA compatibility (except CBT, n = 155), neutrophil engraftment (n = 31), survival status (n = 5), or GVHD (n = 13). We also excluded eight patients with myeloma, 12 patients who did not achieve neutrophil engraftment, and two patients with a 3 HLA antigen mismatch after CBT. Patients receiving double umbilical cord units were not included in the analyses as they are only performed on clinical trials in Japan. Graft-versus-host disease prophylaxis with PTCy is only applied after haploidentical transplantation and is not used after CBT or MDT in Japan. Ultimately, 914 patients were eligible for this study. This study was approved by the institutional review board of each center and conducted in accordance with the Declaration of Helsinki.
Endpoints
The primary end point of the study was the impact of acute GVHD on overall survival (OS) for each donor type. Other end points included the impact of acute GVHD on relapse and NRM and the impact of chronic GVHD on OS, relapse, and NRM for each donor type.
Definitions
The diagnosis and grading of acute and chronic GVHD were based on traditional criteria27,28. We classified the intensity of conditioning regimens as myeloablative conditioning (MAC) if total body irradiation >8 Gy, oral busulfan ≥9 mg/kg, intravenous busulfan ≥7.2 mg/kg, melphalan >140 mg/m2, or thiotepa ≥10 mg/kg was used, and otherwise classified it as reduced intensity conditioning (RIC) 29 . Compatibility at HLA-A, HLA-B, HLA-C, and HLA-DRB1 loci between patients and donors was determined using standard serologic techniques and high-resolution DNA typing. We defined HLA-matching based on HLA-A, HLA-B, and HLA-DR antigen levels in cord blood and sibling donors (PTCy-haplo and HLA-matched donors) and on HLA-A, HLA-B, HLA-C, and HLA-DRB1 allele levels in HLA-matched unrelated donors. The refined disease risk index (rDRI) was used in risk stratification analyses 30 . Furthermore, adult T-cell leukemia-lymphoma (ATL) in complete or partial remission was defined as intermediate risk, and that in nonremission was considered very high risk 31 .
Statistical Analysis
Patients, diseases, and transplant characteristics were compared between groups using a chi-square test or Fisher’s exact test for categorical variables and the Kruskal–Wallis test for continuous variables. The Cox proportional-hazards model was used to evaluate the effects of GVHD and confounding variables on OS, relapse, and NRM. In these regression models, the development of acute and chronic GVHD was treated as a time-dependent covariate. We treated the development of acute or chronic GVHD at their worst severity as a time-varying covariate. When we evaluated the effect of GVHD on transplant outcomes, acute GVHD and chronic GVHD were analyzed separately. In the analysis of acute GVHD, patients were assigned to the “no acute GVHD” group at the time of transplantation and transferred to the “grade I-II acute GVHD” group or the “grade III-IV acute GVHD” group at the onset of acute GVHD, not considering the occurrence of chronic GVHD. The analysis of chronic GVHD included patients who survived at least 100 days. Patients were assigned to the “no chronic GVHD” group at the time of transplantation and then the “limited chronic GVHD” group or the “extensive chronic GVHD” group at the onset of chronic GVHD. In the analysis of the effect of chronic GVHD, the prior history of acute GVHD (no, grade I–II or grade III–IV acute GVHD) was included in the multivariate analysis. The impact of GVHD on survival was graphically illustrated by Simon–Makuch plots24,32.
Adjusted covariates were patient sex, age (<50 or ≥50 years old), performance status (PS, 0–1 or >1), rDRI (low/intermediate or high/very high), and the conditioning regimen intensity (reduced intensity or myeloablative). We then added the occurrence of GVHD to the final model. All tests were two-sided and P values <0.05 were considered statistically significant. All statistical analyses were performed with EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan) 33 .
Results
Patient Characteristics
A schematic workflow of the inclusion and exclusion of our study patients is shown in Supplementary Fig. 1. Ultimately, we enrolled 914 patients who received allogeneic HSCT in 17 centers from 2015 to 2021. Table 1 shows the patient characteristics. Among these 914 patients, 120 received PTCy-haplo transplant [116 peripheral blood stem cell (PBSC) and four bone marrow grafts], 402 received CBT, and 392 received MDT (174 matched sibling donors and 118 matched unrelated donors). The median age at HSCT was 55.5, 57, and 51.5 years in PTCY-haplo, CBT, and MDT, respectively (P < 0.001). The performance status was similar among the donor types (P = 0.478). A higher proportion of PTCy-haplo was RIC compared with CBT and MDT (P < 0.001). Cases with high or very high rDRI were more frequently included in CBT (P < 0.001). In PTCy-haplo, 51.7% of the patients received 50 mg/kg of Cy on days 3 and 4, and 48.3% received 40 mg/kg of Cy on days 3 and 4, and the median duration of tacrolimus and mycophenolate mofetil (MMF) use was 182 days (8–1115) and 34 days (4–121), respectively. The median number of CD34+ cells of PBSCs infused (×106/kg) was 4.1 (0.61–15.0) in PTCy-haplo units. In CBT, the median total nucleated cells (TNCs) that were cryopreserved and infused (×107/kg) and CD34+ cells infused (×105/kg) were 3.00 (2.00–6.00) and 0.74 (0.20–4.43), respectively.
Patient Characteristics.

PTCy-haplo: posttransplant cyclophosphamide haploidentical; CBT: cord blood transplantation; MDT: matched donor transplantation; AML: acute myeloid leukemia; ALL: acute lymphoblastic leukemia; MDS: myelodysplastic syndrome; CML: chronic myeloid leukemia; NHL: non-Hodgkin lymphoma; HL: Hodgkin lymphoma; ATL: adult T-cell leukemia; MPN: myelodysplastic neoplasm; ECOG PS: Eastern Cooperative Oncology Group performance status; rDRI: refined disease risk index; GVHD: graft-versus-host disease; CI: confidence interval; MTX: methotrexate; MMF: mycophenolate mofetil; Cy: cyclophosphamide; HLA: human leukocyte antigen.
Effect of Acute GVHD on Overall Survival
The median day of onset of acute GVHD of any grade after transplantation was 34.00 [interquartile range (IQR) = 22.50–59.50], 30.00 (IQR = 21.00–42.75), and 29.50 (19.25–42.00) in PTCy-haplo, CBT, and MDT, respectively (P = 0.123). Acute GVHD of grades I–II and III–IV occurred in 45 (37.5%) and 7 (5.8%) in PTCy-haplo, 148 (36.8%) and 66 (16.4%) in CBT, and 152 (38.8%) and 54 (13.8%) in MDT, respectively (P = 0.047). The types of organ involvement associated with acute GVHD were shown in Supplementary Fig. 2. Skin involvement in grade III–IV acute GVHD was less frequent after PTCy-haplo (20%) than after CBT (73%) or MDT (61%) (P = 0.037). Intestinal involved in grade III–IV acute GVHD was less frequently observed after MDT (30%) than after PTCy (100%) or CBT (88%) (P < 0.001).
The effect of acute GVHD on overall survival was illustrated with reference to three categories: no, grade I–II, and grade III–IV acute GVHD (Fig. 1A–C). A multivariate analysis that treated acute GVHD as a time-dependent covariate revealed a positive impact of grade I–II acute GVHD in PTCy-haplo and CBT [hazard ratio (HR) = 0.39, 95% confidence interval (CI) = 0.18–0.85, P = 0.018 in PTCy-haplo, and HR = 0.48, 95% CI = 0.32–0.72, P < 0.001 in CBT], whereas there was no impact in MDT (HR = 0.80, 95% CI = 0.55–1.19, P = 0.267) (Table 2). An adverse impact of grade III–IV acute GVHD on OS compared with no GVHD was observed only in CBT (HR = 1.93, 95% CI = 1.34–2.79, P < 0.001), although a trend for worse outcomes with grade III–IV acute GVHD was also observed in the MDT group (HR = 1.54, 95% CI = 0.97–2.46, P = 0.070).
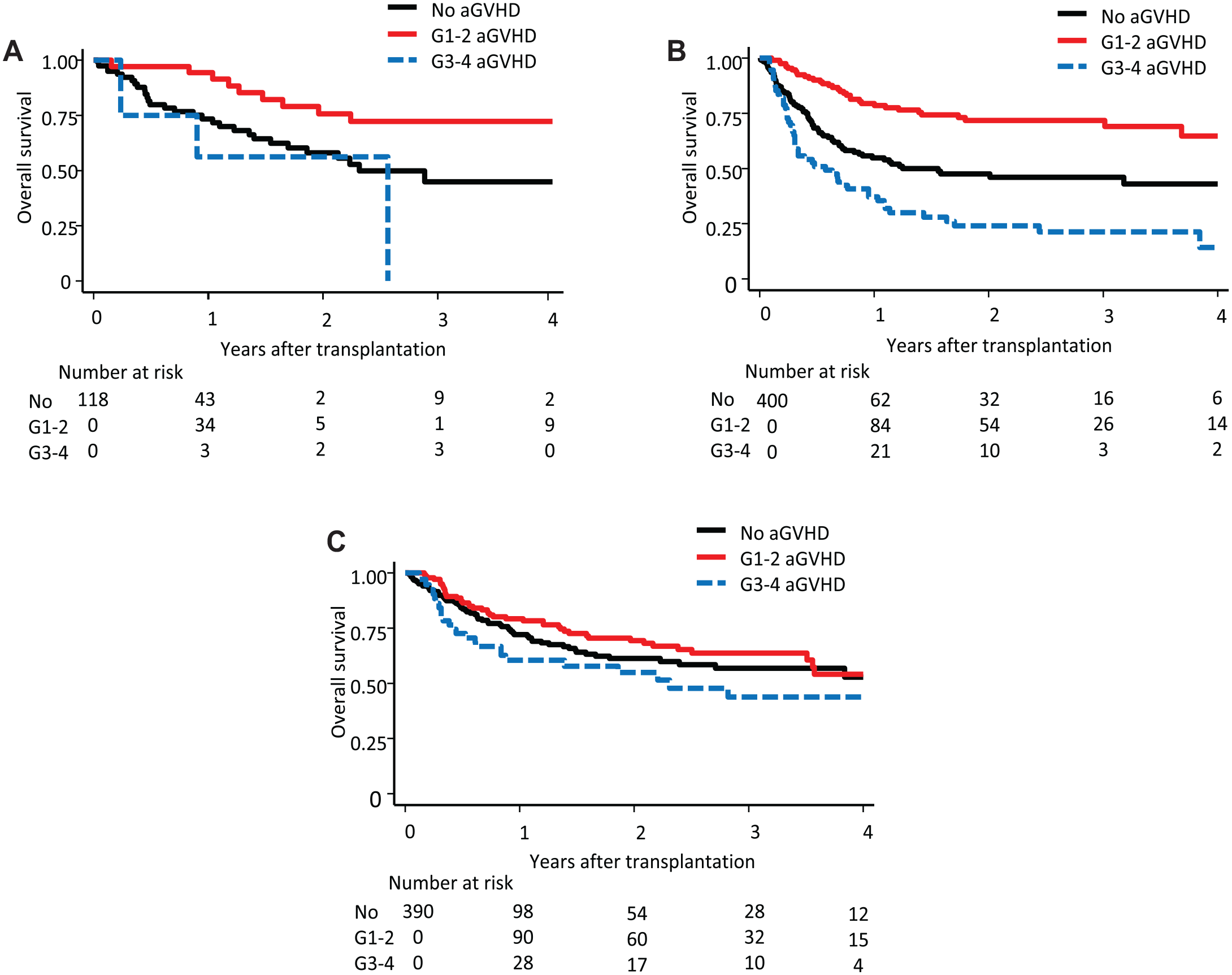
Effect of acute GVHD on overall survival according to the donor type. The effects of aGVHD on overall survival in (A) PTCy-haplo transplantation, (B) cord blood transplantation, and (C) HLA-matched donor transplantation are illustrated by Simon-Makuch plots. GVHD: graft-versus-host disease; aGVHD: acute graft-versus-host disease; PTCy-haplo: posttransplant cyclophosphamide haploidentical; HLA: human leukocyte antigen.
Impact of Acute GVHD on Overall Survival, Relapse, and Nonrelapse Mortality According to the Donor Type.

Other variables included for adjustment were patient age, patient sex, performance status, conditioning intensity, and disease status.
GVHD: graft-versus-host disease; PTCy-haplo: posttransplant cyclophosphamide haploidentical; CBT: cord blood transplantation; MDT: matched donor transplantation; HR: hazard ratio; CI: confidence interval; OS: overall survival; aGVHD: acute graft-versus-host disease; G I–II: grade I–II; G III–IV: grade III–IV.
Effects of Acute GVHD on Relapse and NRM
We next evaluated the effects of acute GVHD on relapse and NRM. The effects of acute GVHD on relapse (Fig. 2A–C) and NRM (Fig. 3A–C) are illustrated with reference to three categories: no, grade I–II, and grade III–IV acute GVHD. A multivariate analysis revealed that the risk of relapse was comparable in the presence of grades I–II and III–IV acute GVHD compared with no acute GVHD in PTCy (grade I–II acute GVHD: HR = 0.78; 95% CI = 0.35–17.3; P = 0.534; grade III–IV acute GVHD: HR= 1.34; 95% CI = 0.30–6.00; P =0.706; Table 2). Grades I–II and III–IV acute GVHD also had no effect on relapse in CBT and MDT (Table 2). These results were consistent in analyses that excluded patients with chronic GVHD (data not shown).

Effect of acute GVHD on relapse according to the donor type. The effects of aGVHD on relapse in (A) PTCy-haplo transplantation, (B) cord blood transplantation, and (C) HLA-matched donor transplantation are illustrated by Simon-Makuch plots. GVHD: graft-versus-host disease; aGVHD: acute graft-versus-host disease; PTCy-haplo: posttransplant cyclophosphamide haploidentical; HLA: human leukocyte antigen.

Effect of acute GVHD on nonrelapse mortality according to the donor type. The effects of aGVHD on nonrelapse mortality in (A) PTCy-haplo transplantation, (B) cord blood transplantation, and (C) HLA-matched donor transplantation are illustrated by Simon–Makuch plots. GVHD: graft-versus-host disease; aGVHD: acute graft-versus-host disease; PTCy-haplo: posttransplant cyclophosphamide haploidentical; HLA: human leukocyte antigen.
Notably, the development of grade I–II acute GVHD tended to lower the risk of NRM compared with no acute GVHD in PTCy (HR = 0.13, 95% CI = 0.02–1.09, P = 0.060) and the effect was significant in CBT (HR = 0.39, 95% CI = 0.21–0.72, P = 0.003) (Table 2). However, there was no positive effect of grade I–II acute GVHD in MDT (HR = 1.36, 95% CI = 0.76–2.44, P = 0.301) (Table 2). The development of grade III–IV acute GVHD was significantly associated with higher NRM compared with no acute GVHD in CBT and MDT (HR = 2.65, 95% CI = 1.66–4.24, P < 0.001 in CBT and HR, 2.57, 95% CI = 1.25–5.28, P = 0.010 in MDT), whereas it was comparable in PTCy (HR = 1.09, 95% CI = 0.12–10.1, P = 0.941) (Table 2).
The primary cause of death according to no GVHD, grade I–II GVHD, and grade III–IV in each donor type is shown in Supplementary Table 1. The most common cause of death was relapse in patients who did not develop acute GVHD or who developed grade I–II acute GVHD for each donor type. In the grade I–II acute GVHD group, GVHD-associated death was not observed in PTCy or CBT, whereas it was 13.3% in MDT. In the grade III–IV acute GVHD group, GVHD-associated death was observed in 33.3%, 15.2%, and 8.0% in PTCy, CBT, and MDT, respectively.
Effects of Chronic GVHD on Overall Survival, Relapse, and NRM
Chronic GVHD was evaluated in 777 patients who survived at least 100 days after transplantation. The median number of days until the occurrence of chronic GVHD after transplantation was 176.00 [interquartile range (IQR) = 108.50–260.50 days], 113.50 (IQR = 100.00–140.00 days), and 127.00 (IQR = 103.00–191.00 days) in PTCy, CBT, and MDT, respectively (P < 0.001). Limited chronic GVHD and extensive chronic GVHD occurred in 18 (15.0%) and 17 (14.2%) in PTCy, 50 (12.4%) and 32 (8.0%) in CBT, and 54 (13.8%) and 79 (20.2%) in MDT, respectively (P < 0.001). The effect of chronic GVHD on overall survival was illustrated with reference to three categories: no, limited, and extensive chronic GVHD (Fig. 4A–C). A multivariate analysis that treated chronic GVHD as a time-dependent covariate revealed that limited chronic GVHD had a positive effect on overall survival compared with no chronic GVHD in CBT and MDT (HR = 0.45, 95% CI = 0.22–0.94, P = 0.033 in CBT and HR = 0.37, 95% CI = 0.18–0.79, P = 0.010 in MDT), whereas there was no impact in PTCy (HR = 0.35, 95% CI = 0.08–1.51, P = 0.158; Table 3).

Effect of chronic GVHD on overall survival according to the donor type. The effects of cGVHD on overall survival in (A) PTCy-haplo transplantation, (B) cord blood transplantation, and (C) HLA-matched donor transplantation are illustrated by Simon–Makuch plots. GVHD: graft-versus-host disease; cGVHD: chronic graft-versus-host disease; PTCy-haplo: posttransplant cyclophosphamide haploidentical; HLA: human leukocyte antigen.
Impact of Chronic GVHD on Overall Survival, Relapse, and Nonrelapse Mortality According to the Donor Type.

Other variables included for adjustment were patient age, patient sex, performance status, conditioning intensity, disease status, and prior history of acute GVHD.
GVHD: graft-versus-host disease; PTCy-haplo: posttransplant cyclophosphamide haploidentical; CBT: cord blood transplantation; MDT: matched donor transplantation; HR: hazard ratio; CI: confidence interval; OS: overall survival; cGVHD: chronic graft-versus-host disease.
Could not be calculated because of the low incidence of relapse in the “limited cGVHD group” after PTCy-haplo transplantation.
The effects of chronic GVHD on relapse (Supplementary Fig. 3A–C) and NRM (Supplementary Fig. 4A–C) are shown with reference to three categories: no, limited, and extensive chronic GVHD. Limited and extensive chronic GVHD were not associated with a lower risk of relapse in this study, except that extensive chronic GVHD in MDT had a lower risk of relapse compared with no chronic GVHD (HR = 0.29, 95% CI = 0.09–0.97, P = 0.044; Table 3). Limited and extensive chronic GVHD were not associated with a lower risk of NRM (Table 3).
Effects of Lymphocyte Recovery and Cyclophosphamide Dose on OS After PTCy-Haplo Transplantation
Since lymphocyte recovery is an important clinical factor for transplant outcomes, we analyzed the impact of lymphocyte recovery on OS in PTCy-haplo transplantation and its association with the occurrence of GVHD (no, grade I–II or grade III–IV acute GVHD) and the PTCy dose (reduced dose; 40 mg/kg on days 3 and 4 or standard dose; 50 mg/kg on days 3 and 4). The data regarding lymphocyte counts in peripheral blood were collected on days 0, 30, 60, 100, and 180 after PTCy-haplo transplantation. Lymphocyte counts according to the incidence of GVHD are shown in Supplementary Table 2. Although lymphocyte recovery at days 30, 60, 100, and 180 was comparable between the “no acute GVHD” group and the “grade I-II acute GVHD” group, the lymphocyte count at day 180 in the “grade III-IV acute GVHD” group (median 325.5/μL, quantile value 257.25–393.75/μL) was significantly lower than that in the “no acute GVHD” group (median 1181.5/μL, quantile value 776.25–1792/μL, P = 0.033). Notably, 2-year OS in patients with lymphocyte counts <1,000/μL at day 180 (59.3%, 95% CI = 38.3%–75.3%) was significantly lower than that in patients with day 180 counts ≥1,000/μL (84.5%, 95% CI = 71.3%–92.0%, P = 0.021) (Supplementary Fig. 5).
Lymphocyte counts according to cyclophosphamide (Cy) dose are shown in Supplementary Table 3. The decision regarding the dose of Cy was based on the physician’s discretion. Lymphocyte counts at days 60, 100, and 180 in the “reduced-dose” group (median 732/μL, 936/μL, and 1295/μL, respectively) were significantly higher than those in the “standard-dose” group (median 493/μL, 625/μL, and 1120.5/μL, respectively) (P = 0.039, 0.030, and 0.033, respectively). The effects of acute GVHD on OS, relapse, and NRM according to the Cy dose are shown in Supplementary Table 4. Grade I–II acute GVHD had a positive effect on OS in the “reduced-dose” group (HR = 0.27, 95% CI = 0.07–1.00, P = 0.049), but there was no significant effect in the “standard-dose” group (HR = 0.49, 95% CI = 0.18–1.38, P = 0.177). Furthermore, 2-year OS in the “standard-dose” group (74.9%, 95% CI = 61.8–84.1%) tended to be lower than that in the “reduced-dose” group (85.4%, 95% CI = 72.8%–92.4%, P = 0.059) (Fig. 5A). Although 2-year relapse rates were comparable between the two groups (“standard-dose” group: 31.8%, 95% CI = 20.3%–43.9% and “reduced-dose” group: 30.0%, 95% CI = 18.6%–42.3%, P = 0.673, Fig. 5B), 2-year NRM rates tended to be higher in the “standard-dose” group (17.2%, 95% CI = 8.7%–28.2%) than that in the “reduced-dose” group (3.9%, 95% CI = 0.7%–12.1%) (P = 0.065, Fig. 5C). The cause of death according to Cy dose was shown in Supplementary Table 5. While death from hemorrhage was not observed in the “reduced-dose” group, the rate of hemorrhage was 11.1% in the “standard-dose” group.

Impact of cyclophosphamide dose on outcomes after PTCy-haplo transplantation. Kaplan-Meier estimates of (A) overall survival, (B) relapse, and (C) nonrelapse mortality according to the dose of Cy (reduced dose: 40 mg/kg on day 3 and day 4 or standard dose: 40 mg/kg on day 3 and day 4). PTCy-haplo: posttransplant cyclophosphamide haploidentical.
Effects of Acute GVHD on OS, Relapse, and NRM in Patients Who Survived for More Than 100 Days
We also assessed the effects of acute GVHD on OS, relapse, and NRM in patients who survived for more than 100 days after transplantation because patients who died within 100 days might be less likely to have GVHD, resulting in a bias that patients with a poor prognosis would be more likely to be included in the “no acute GVHD” group than in the “grade I-II acute GVHD” group. However, as in the whole cohort, grade I–II acute GVHD had a positive impact after PTCy-haplo (HR = 0.36, 95% CI = 0.16–0.84, P = 0.018) and CBT (HR = 0.56, 95% CI = 0.35–0.89, P = 0.015), but not after MDT (HR = 0.90; 95% CI = 0.59–1.37, P = 0.613) (Supplementary Table 6).
Grade I–II acute GVHD had a positive effect on relapse after MDT (HR = 0.57, 95% CI = 0.33–0.98, P = 0.042) compared with that after no GVHD (Supplementary Table 5). Grades I–II and III–IV acute GVHD did not affect relapse in PTCy-haplo or CBT. The development of grade I–II acute GVHD tended to lower the risk of NRM compared with no acute GVHD in PTCy (HR = 0.16, 95% CI = 0.02–1.36, P = 0.093) and CBT (HR = 0.55, 95% CI = 0.27–1.11, P = 0.093), whereas there was an adverse trend in MDT (HR = 1.95, 95% CI = 0.97–3.91, P = 0.059) (Supplementary Table 6).
Discussion
This study evaluated the effects of acute and chronic GVHD on transplant outcomes in PTCy-haplo, CBT, and MDT. There were three major findings: (1) grade I–II acute GVHD improved OS after PTCy-haplo and CBT, but not after MDT, (2) this result can be explained by the observation that grade I–II acute GVHD reduced NRM after PTCy-haplo and CBT, and (3) limited chronic GVHD improved OS only after CBT and MDT.
In the conventional transplant strategy, the benefit of GVM effect was considered to be counter-balanced by the adverse effect of GVHD11,34. However, our study demonstrated that mild GVHD in the absence of an increase in severe GVHD improved OS after PTCy-haplo transplantation, which is consistent with a previous report 35 . In that report, grade II acute GVHD after PTCy-haplo transplantation improved progression-free survival with a lower risk of relapse 35 . Our study is unique in that the positive effects of mild GVHD were due to its ability to reduce NRM, whereas the previous study indicated that improved OS is due to a reduction in the risk of relapse and without reductive effects on NRM. The reason for this finding is not clear, as the distribution of the causes of death and lymphocyte recovery were similar between the no acute GVHD and grade I–II acute GVHD groups. A recent study reported that better CD4-positive T-cell reconstitution at GVHD onset was associated with lower mortality 36 .
Patients who experienced grade I–II acute GVHD may achieve better immune reconstitution without inducing any profound immunosuppression. The positive effect of mild GVHD was consistently observed in the analysis that only included patients who survived for more than 100 days. This suggests that a long-term change in the immune-state after GVHD influences outcomes.
In addition, because a reduced dose of Cy resulted in better lymphocyte recovery and a better trend of OS compared with a standard dose of Cy, and mild acute GVHD was only observed to have a positive effect after administration of a reduced dose of Cy, a reduced dose of Cy is a valid option in PTCy-haplo transplantation. The difference of Cy dose may explain the discrepancy between our study and a previous study demonstrating that grade II acute GVHD was associated with higher NRM in acute myeloid leukemia patients after PTCy-haplo transplantation 37 .
We demonstrated different incidences and impacts of GVHD according to the three donor types. Severe acute GVHD was observed less frequently after PTCy-haplo than after CBT and MDT. Because the risk of NRM was significantly higher after the development of grade III–IV acute GVHD in CBT and MDT, severe acute GVHD should be avoided after transplantation. In this context, PTCy-haplo is beneficial by reducing the risk of severe acute GVHD and retaining a similar incidence of grade I–II acute GVHD, which improved OS. On the contrary, CBT reduced the risk of extensive chronic GVHD with a similar incidence of limited chronic GVHD as in PTCy-haplo and MDT, which is consistent with previous studies7,22,38,39. Because chronic GVHD is a serious complication that affects the quality of life of long-term survivors after HSCT, this advantage of CBT should be noted. In this study, the positive impact of limited chronic GVHD on OS was observed after CBT and MDT, but not after PTCy-haplo. It is unclear whether there is no association or the result was not statistically significant due in part to a low number of events after PTCy-haplo. The occurrence of chronic GVHD was delayed after PTCy-haplo compared with CBT and MDT. The delayed occurrence of chronic GVHD after PTCy-haplo transplantation might reflect specific immune reconstruction and be partly responsible for the result. Further studies are warranted to elucidate the impact of chronic GVHD after PTCy-haplo transplant.
This study has several limitations. First, our analysis is inherently limited by its retrospective nature and also by the low number of grade III–IV acute GVHD and chronic GVHD events after PTCy-haplo transplantation. This limitation warrants larger studies to corroborate our results and elucidate the effect of grade III–IV acute GVHD and chronic GVHD after PTCy-haplo transplantation. Second, the heterogeneity of the disease in this study made it difficult to clarify the relevant effects of GVHD on relapse and NRM, because the risk depends on the disease type10,34,40. In fact, previous studies have demonstrated that grade I–II acute GVHD and limited chronic GVHD after CBT reduce the risk of relapse for patients with acute leukemia or myelodysplastic syndrome24,26,41. Furthermore, a recent study has shown that grade I–II acute GVHD was associated with better OS in CBT, but not with PTCy-haplo transplantation 42 . Third, information on the day when each grade of GVHD occurred was not fully available. Therefore, we treated the development of acute or chronic GVHD at their worst severity as a time-varying covariate. A recent registry study has demonstrated that grade I acute GVHD was associated with better survival but this was not the case for grade II acute GVHD in patients with AML after PTCy-haplo transplantation 43 . The impact of grade I and grade II acute GVHD should be investigated separately in the future studies. In addition, the treatment intervention and response for acute and chronic GVHD were not considered in this analysis, which could affect the transplant outcomes and lymphocyte recovery. Fourth, information on minimal residual disease (MRD) was not available in this study. Previous studies suggested a different impact of acute GVHD on survival between AML patients in complete response (CR) and not in CR37,43. Finally, it has been suggested that immunotherapy approaches can enhance GVM effects without exacerbating GVHD 44 . The benefit of GVM effects may not be offset by the adverse effect of GVHD in future strategies.
In conclusion, our study demonstrated that mild acute GVHD carried a survival benefit by reducing NRM in PTCy-haplo and CBT. Severe GVHD should be prevented because of its association with a high risk of NRM after CBT and MDT, although the incidence of severe GVHD itself was lower in PTCy-haplo transplant. Limited chronic GVHD was associated with better OS after CBT and MDT. Further improvement of these transplant platforms should be explored to promote well-tolerated mild GVHD in the absence of an increase in severe GVHD.
Supplemental Material
sj-docx-1-cll-10.1177_09636897231194497 – Supplemental material for Mild Acute Graft-Versus-Host Disease Improves Outcomes After HLA-Haploidentical-Related Donor Transplantation Using Posttransplant Cyclophosphamide and Cord Blood Transplantation
Supplemental material, sj-docx-1-cll-10.1177_09636897231194497 for Mild Acute Graft-Versus-Host Disease Improves Outcomes After HLA-Haploidentical-Related Donor Transplantation Using Posttransplant Cyclophosphamide and Cord Blood Transplantation by Fumiya Wada, Junya Kanda, Kimimori Kamijo, Masashi Nishikubo, Satoshi Yoshioka, Takayuki Ishikawa, Yasunori Ueda, Takashi Akasaka, Yasuyuki Arai, Kiyotaka Izumi, Hirokazu Hirata, Takashi Ikeda, Akihito Yonezawa, Naoyuki Anzai, Mitsumasa Watanabe, Kazunori Imada, Kazuhiro Yago, Naoki Tamura, Mitsuru Itoh, Yuki Masuo, Akane Kunitomi, Tomoharu Takeoka, Toshiyuki Kitano, Nobuyoshi Arima, Masakatsu Hishizawa, Kohsuke Asagoe, Tadakazu Kondo and Akifumi Takaori-Kondo in Cell Transplantation
Footnotes
Acknowledgements
The authors thank Maki Sakurada, Miki Shirasu, Mieko Hatae, and Tomoko Okuda for their expert data management and secretarial assistance, all of the physicians and data managers at the institutes who provided important data on transplantation, and the Kyoto Stem Cell Transplantation Group (KSCTG), which consists of Hyogo Prefectural Amagasaki General Medical Center, Japanese Red Cross Osaka Hospital, Japanese Red Cross Otsu Hospital, Japanese Red Cross Wakayama Medical Center, Kansai Electric Power Hospital, Kitano Hospital, Kobe City Medical Center General Hospital, Kokura Memorial Hospital, Kurashiki Central Hospital, Kyoto-Katsura Hospital, Kyoto University Hospital, Shiga General Hospital, Shinko Hospital, Kyoto City Hospital, Shizuoka Cancer Center, Shizuoka General Hospital, Takatsuki Red Cross Hospital, and Tenri Hospital
Author Contributions
F.W. and J.K. designed the research; J.K., T.K. and A.T.-K. organized the project; F.W. and J.K. performed the statistical analysis; F.W. wrote the first draft; and all of the other authors critically reviewed the draft and approved the final version for publication.
Ethical Approval
This study was approved by the institutional review board of each center.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Takeda Science Foundation (J.K.) and JSPL KAKENHI Grant number 18K08325/21K08391 (J.K.).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
