Abstract
Acute graft versus host disease (aGvHD) is the major contributor of nonrelapse mortality in alloHSCT. It is associated with an inflammatory immune response manifesting as cytokine storm with ensuing damage to target organs such as liver, gut, and skin. Prevention of aGvHD while retaining the beneficial graft versus leukemia (GvL) effect remains a major challenge. Withania somnifera extract (WSE) is known for its anti-inflammatory, immune-modulatory, and anticancer properties, which are appealing in the context of aGvHD. Herein, we demonstrated that prophylactic and therapeutic use of WSE in experimental model of alloHSCT mitigates aGvHD-associated morbidity and mortality. In the prophylaxis study, a dose of 75 mg/kg of WSE offered greatest protection against death due to aGvHD (hazard ratio [HR] = 0.15 [0.03-0.68], P ≤ .01), whereas 250 mg/kg was most effective for the treatment of aGvHD (HR = 0.16 [0.05-0.5], P ≤ .01). WSE treatment protected liver, gut, and skin from damage by inhibiting cytokine storm and lymphocytic infiltration to aGvHD target organs. In addition, WSE did not compromise the GvL effect, as alloHSCT with or without WSE did not allow the leukemic A20 cells to grow. In fact, WSE showed marginal antileukemic effect in vivo. WSE is currently under clinical investigation for the prevention and treatment of aGvHD.
Keywords
Introduction
Allogeneic hematopoietic stem cell transplantation (AlloHSCT) is often the only curative treatment option for several hematological disorders including leukemias 1 . Worldwide, every year approximately 30,000 patients undergo alloHSCT. Despite advances in treatment and supportive care, 30% to 70% of alloHSCT recipients develop acute graft versus host disease (aGvHD), the most common cause of death following transplantation 1 . aGvHD mainly causes the destruction of skin, gut, and liver due to an overarching immune response of donor cells against these host tissues 2 . Standard GvHD prophylactic and therapeutic regimens are not always effective 3 , thereby highlighting the need to develop novel pharmacological agents against aGvHD.
Medicinal plants have often attracted the attention of medical scientists due to their safety profile and short development time4–6. In recent years, many plant products such as curcumin 7 , extract of Tripterygium hypoglaucum 8 , green tea catechin epigallocatechin gallate 9 , and polyphenolic extract from olive oil 10 , have been tested against aGvHD in preclinical models due to their anti-inflammatory and immunomodulatory efficiency. Withania somnifera (Indian ginseng or Ashwagandha) also has anti-inflammatory, immune-modulatory, and antiproliferative properties 11 – 13 . The use of Withania somnifera for aGvHD, a condition orchestrated by an inflammatory milieu and a hyperactive immune response 14 , is therefore appealing. In this study, a root extract of Withania somnifera extract (WSE) comprising a mixture of withanoloids standardized for 4.5% withaferin-A w/w was investigated for its utility against aGvHD, both for prophylaxis and treatment. In addition, we also elucidated the role of WSE in GvHD target organ protection, cytokine storm, and graft versus leukemia (GvL) effects.
Materials and Methods
Experimental Animals
In the study, BALB/c (H-2Kd) female mice and C57BL/6 (H-2Kb) male mice of 8- to 10-week old having a weight of 20 ± 2 g were used. Institutional animal ethics committee of Advanced Center for Treatment, Research and Education in Cancer (ACTREC) approved this study (project no. 19/2022). All experimental animals were acclimatized for at least 7 days before the initiation of experiments. All animals were housed in the laboratory animal facility of ACTREC. Standard chow and water was given ad libitum. 55% ± 15% humidity, 22°C to 25°C temperature with a 12-h light/dark cycle were maintained in the facility. All animal procedures were carried out in compliance with the committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) and ARRIVE guidelines.
Induction of aGvHD
Development of aGvHD model was carried out according to the previously published study by our group 13 . Briefly, aGvHD was induced by performing allogeneic transplantation between donor C57BL/6 (H-2Kb) and recipient BALB/c (H-2Kd) mice. One day before transplantation, BALB/c mice were exposed to total body myeloablative 6.5 Gy of irradiation. Transplantation was carried by intravenous injection of 15 × 106 splenocytes and 5 × 106 bone marrow cells obtained from C57BL/6 mice. After transplantation, all mice were closely monitored for aGvHD symptoms and the clinical score (CS) of aGvHD was recorded based on the following six criteria: fur texture, skin integrity, posture, activity, weight loss, and diarrhea. Clinical scoring method was adopted from Lai et al. 15 and an earlier published report by our group 13 .
Drug Administration for Prevention and Treatment of GvHD
Two separate experiments were carried out to investigate the utility of WSE in the prophylaxis and treatment of aGvHD. WSE was suspended in 0.5% sodium corboxymethylcellulose (CMC). In the prophylaxis study, WSE was administered by oral gavage from day +1 to day +21 of transplantation. The following four groups were employed: (1) GvHD control, (2) 25 mg/kg of WSE, (3) 75 mg/kg of WSE, and (4) 250 mg/kg of WSE. For therapeutic purpose, WSE was administered from day +7 to day +27 of transplantation and the four study groups used were (1) GvHD control, (2) 75 mg/kg WSE, (3) 250 mg/kg WSE, and (4) 500 mg/kg WSE.
GvHD control group in both experiments received vehicle (CMC) only. WSE used in this study was a mixture of compounds containing 15% withanoloids. Out of 15%, it was standardized for 4.5% withaferin-A w/w. The remaining 10.5% consisted of other withanoloids such as withanolide A and 12-Deoxy-withastramonolide.
Effect of WSE on Graft Versus Leukemia (GvL) Response
To investigate the effect of WSE on anti-leukemic response of the graft, 28 mice were divided equally into the following four groups: (1) A20, (2) A20 + BMT (bone marrow transplantation), (3) A20 + WSE, and (4) A20 + BMT + WSE. Briefly, after exposing animals to 6.5 Gy total body radiation, 3 × 106 A20 cells were injected with or without BMT according to group allocation. WSE was administered prophylactically to groups 3 and 4 at a dose of 75 mg/kg. The GvL effect was monitored according to earlier published method13,16,17. Leukemic death was defined by the presence of hind leg paralysis and the presence of tumor nodules in the liver. On the contrary, GvHD deaths were characterized by the presence of GvHD symptoms but absence of hind-leg paralysis and tumor nodules in liver.
Analysis of Donor Cell Engraftment Using Flow Cytometry
The presence of donor-positive cell (H-2Kb) in recipient mice were evaluated as per earlier published method 13 . Briefly, peripheral blood was collected from recipient mice through retro-orbital puncture. Following RBC lysis and washing, cells were incubated with fluorochrome labeled antibodies against donor H-2Kb (FITC) and recipient H-2Kd (PE) MHC class I antigen and acquired using flow cytometry (attune NxT [Thermofisher, USA]).
Histopathology of aGvHD Target Organs
In a separate set of experiment, on day +7 and +14 of transplantation, aGvHD target organs (liver, gut, skin, and lung) were harvested and processed for H&E. A trained pathologist evaluated and scored the severity of aGvHD and lymphocytic infiltration to the target organs as per method published earlier 13 . The pathological evaluation was carried out in a blinded fashion.
In vivo Cytokine Measurement
In the same cohort of animals used for histopathology, blood from recipient mice was collected on day +7 and +14 of transplantation. Serum was separated and Th1 (interleukin [IL]-2, interferon [IFN]-γ, and tumor necrosis factor [TNF]-α), Th2 (IL-4, IL-6, and IL-10), and Th17 (IL17A) cytokines were measured using cytometric beads array as per manufacture’s instruction and earlier published method 13 .
Reagents and Antibodies
H-2Kb (cat: 562002) and H-2Kd (cat: 553566) antibodies were procured from BD Biosciences. Pharmanza Herbal Pvt. Ltd. provided WSE. Cytokine measurements was performed using the BD CBA mouse Th1/Th2/Th17 cytokine kit (cat: 560485). A20 cell lines were obtained from ATCC (cat: ATCC-TIB-208).
Statistical Analysis
Mean ± standard error of the mean (SEM) was used to represent the data. Comparison between two groups was done using unpaired t-test. Time to event data such as survival and onset of leukemia were analyzed using Kaplan-Meier plots and Mantel-Cox log-rank test. P ≤ .05 was considered statistically significant. Data analysis was done using GraphPad Prism version 8.0.
Results
Prophylactic Administration of WSE Alleviates aGvHD-Associated Morbidity and Mortality
All mice were monitored periodically for aGvHD sign and survival. The median clinical score on day 14 in all experimental groups was 9, 3.5, 2, and 2.5, respectively in the GvHD control, 25, 75, and 250 mg/kg WSE (Fig. 1A). Furthermore, continuous weight loss was observed in the GvHD control and the 25 mg/kg WSE group, the other groups had initial weight loss but was paused over a period of treatment (Fig. 1B). Survival analysis suggested a significant increase in survival in all the three treatment arms compared to GvHD control (Fig. 1C). Survival was significantly higher in 75 mg/kg WSE (hazard ratio [HR] = 0.15 [0.03-0.68], P ≤ .01) and 250 mg/kg WSE (HR = 0.16 [0.03-0.73], P ≤ .01) group compared to the GvHD control and of 25 mg/kg WSE arm (HR = 0.26 [0.06-1.03], P ≤ .05). Median survival in GvHD control and 25 mg/kg WSE was 14 and 28.5 days, respectively; however, it was not reached in 75 and 250 mg/kg WSE group (Fig. 1C). Furthermore, we analyzed donor cell engraftment in WSE-treated animals and found the complete presence of donor-positive cells in the host body, suggesting that WSE does not interfere with the engraftment (Supplementary Fig. 1). Based on these observation 75 and 250 mg/kg WSE was found to be equally efficacious; therefore, further prophylactic experiments was conducted at 75 mg/kg WSE dose.
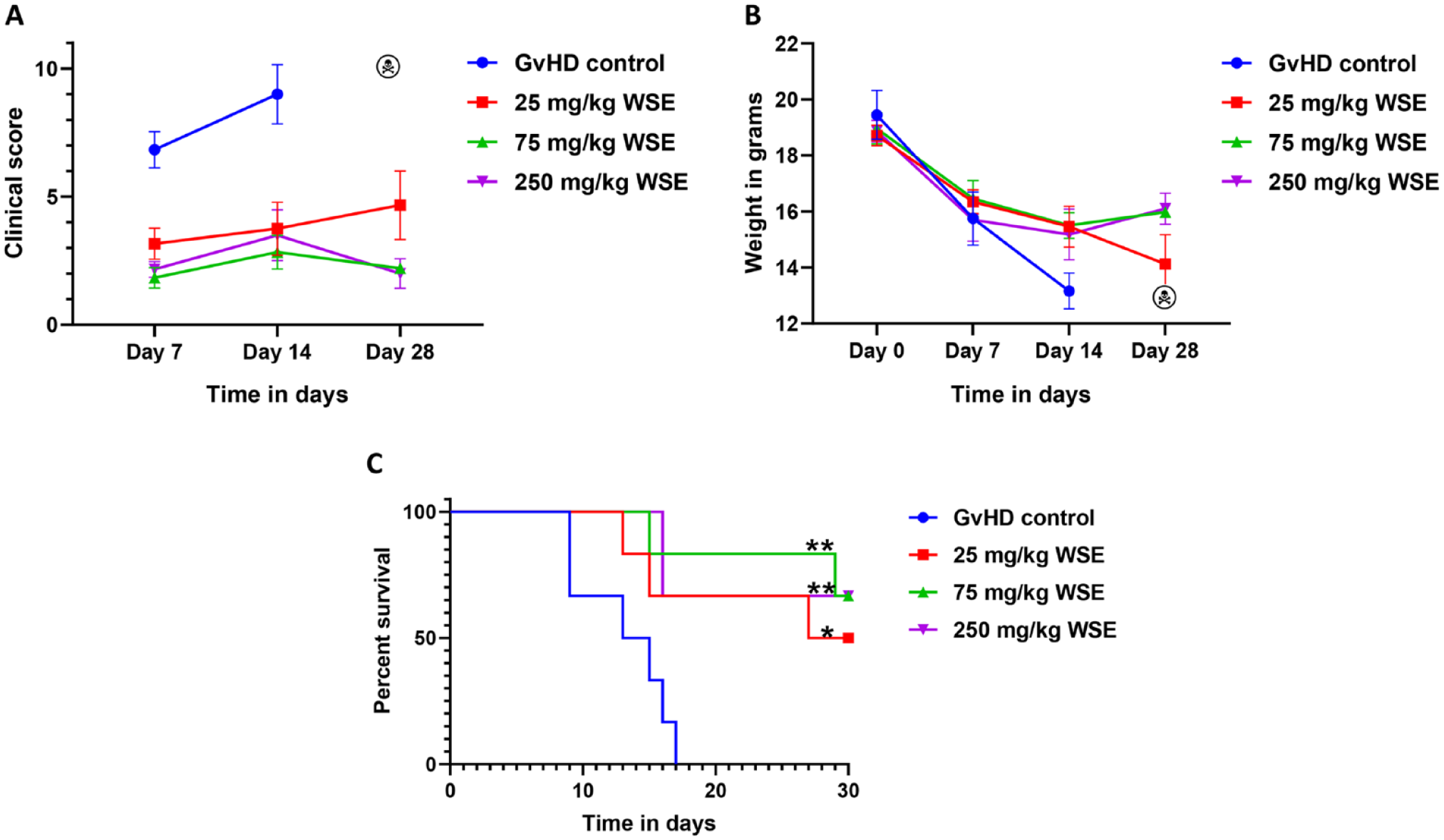
Effect of WSE on aGvHD prophylaxis. Following radiation and transplantation, mice were divided into GvHD control and WSE treatment (25, 75, and 250 mg/kg doses) groups. (A) The severity of aGvHD was monitored all along the study and CS was documented. (B) Body weight alteration in all experimental arms were also recorded throughout the study. (C) At the end of the study, overall survival was analyzed in all experimental arms using Kaplan–Meier plot and Mantel-Cox log rank test. Data is represented as mean ± SEM. n = 6 mice/group. aGvHD: acute graft versus host disease; GvHD: graft versus host disease; SEM: standard error of the mean; WSE: Withania somnifera extract. *P ≤ .05; **P ≤ .01.
WSE Averts aGvHD Target Organ Damage
In a separate cohort of mice at day +7 and day +14 of transplantation, aGvHD target organ from GvHD control and WSE-treated arms were harvested and evaluated histologically. Mice in GvHD control arm showed mild chronic inflammation in the liver and lung, vacuolar degeneration and desquamation of skin, mild-moderate chronic inflammation, ulceration, crypt loss, fibrosis, and sloughing of the small intestine and colon (Fig. 2). In contrast, WSE-treated group showed marked protection of these organs (Fig. 2). Histopathological scoring based on the extent of damage to organs in GvHD control versus treatment arm is shown in Supplementary Fig. 2. In adition, the same tissue was evaluated for lymphocyte infiltration into these organs based on earlier published method 13 . Lymphocyte infiltration in GvHD control group were higher compared to WSE arm, suggesting protection of these organs possibly via inhibition of lymphocyte infiltration (Supplementary Fig. 3).
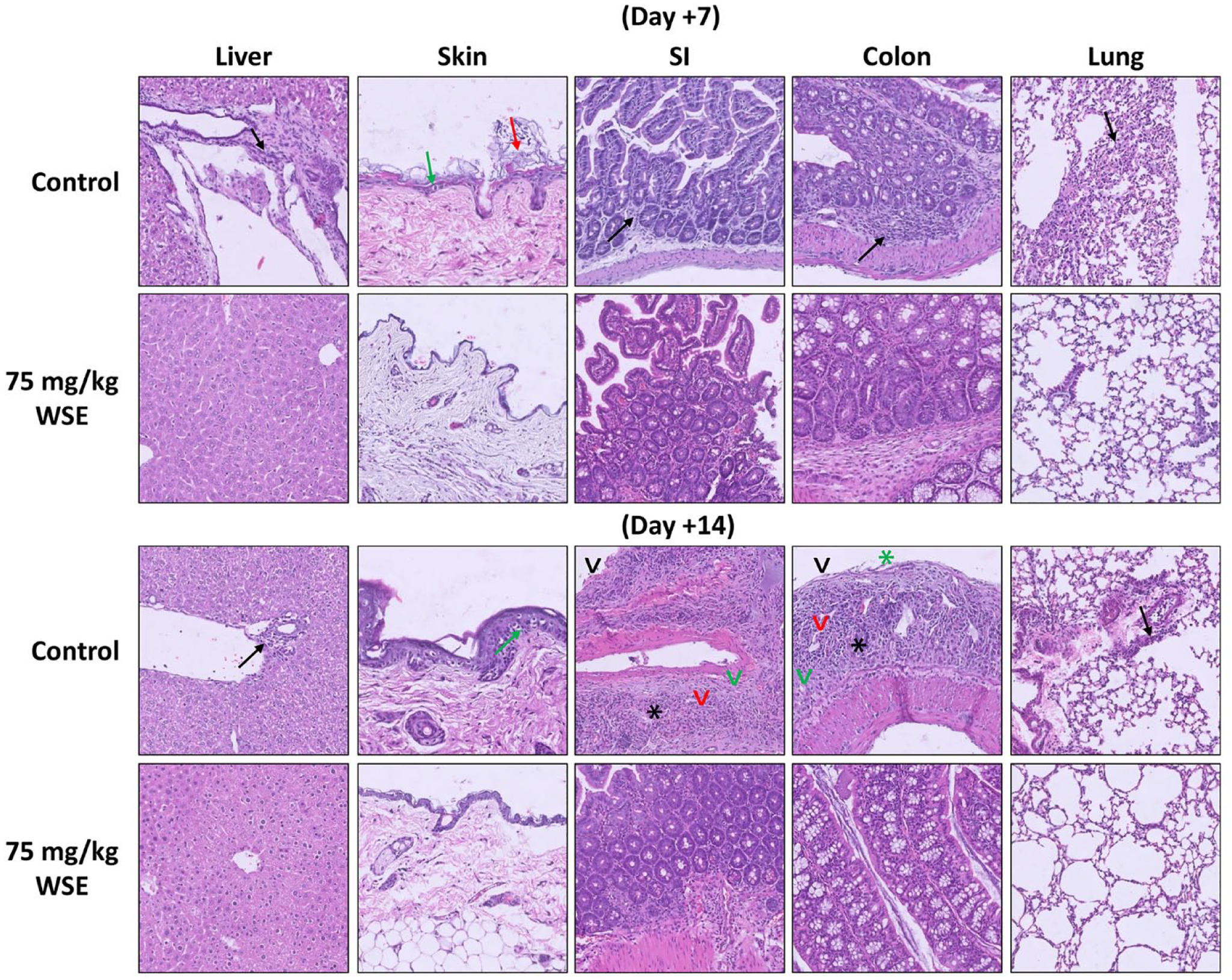
Protection of aGvHD target organs by WSE. In a separate cohort of mice, on days +7 and +14 of transplantation, aGvHD target organs were collected from GvHD control and WSE-treated mice. aGvHD-associated damage were assessed by a pathologist in a blinded manner. Mice that received WSE prophylaxis showed intact tissue histology; however, mice in GvHD control arm had marked damage of target organs. n = 6 mice/group/time point. Black arrow—Mild-moderate chronic inflammation; green arrow—vacuolar degeneration; red arrow—desquamation; black arrowhead—ulceration; green arrowhead—fibrosis; black star—crypt loss; green star—sloughing. aGvHD: acute graft versus host disease; GvHD: graft versus host disease; WSE: Withania somnifera extract.
An inversely proportional relationship between spleen size and GvHD severity has been established earlier 18 . Therefore, we also measured the spleen size and weight, which was found to be significantly decreased in GvHD control group compared to WSE group (supplementary Fig. 4A, B).
WSE Modulates In Vivo Serum Cytokine Storm
Excess cytokine secretion is one of the hallmarks of GvHD and inhibiting their release is critical for the prevention of GvHD 19 . Herein, we evaluated the effect of WSE treatment on cytokine levels at day +7 and +14 of transplantation and found a significant decrease in inflammatory cytokines such as IL-2, IFN-γ, TNF-α, IL-6, and IL-17A in WSE-treated group compared to GvHD control (P ≤ .05) (Fig. 3A, B). IL-10, an anti-inflammatory cytokine, was found to increase in mice treated with WSE compared to the GvHD control (P ≤ .05) (Fig. 3A).
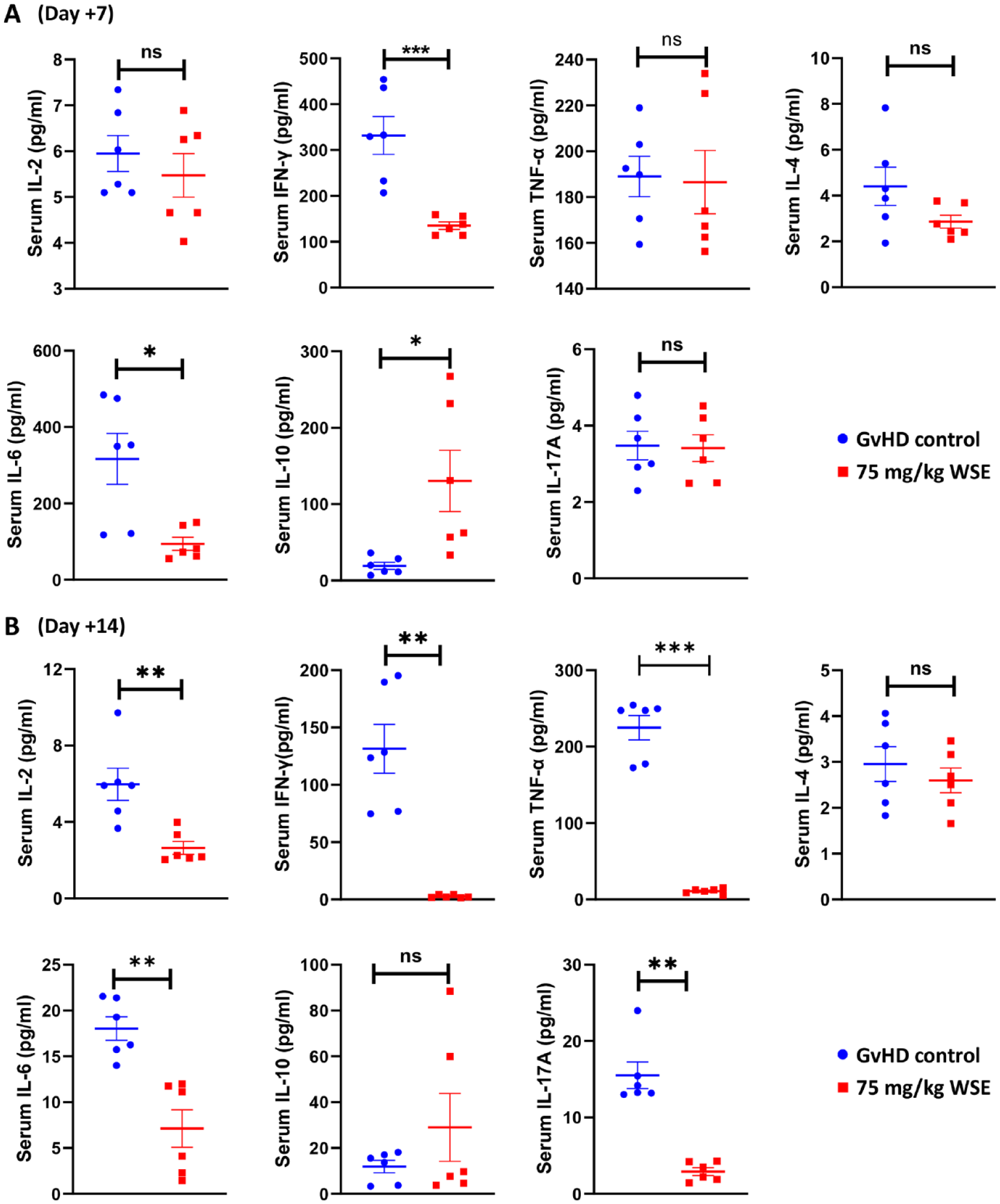
Effect of WSE treatment on in-vivo cytokine secretion. Serum Th1 (IL-2, IFN-γ, TNF-α), Th2 (IL-4, IL-6, IL-10) and Th17 (IL17A) cytokine levels from GvHD control and WSE-treated arms were measured at (A) day +7 and (B) day +14 of transplantation using cytometric beads array. Data is represented as mean ± SEM. n=6 mice/group/time point. GvHD: graft versus host disease; SEM: standard error of the mean; WSE: Withania somnifera extract. *P ≤ .05; **P ≤ .01; ***P ≤ .001.
WSE Does Not Hamper GvL Effect
A prerequisite for any pharmacotherapy to be developed against GvHD is that the drug should not compromise the beneficial GvL effect of the graft 20 . Therefore, we evaluated the effect of WSE treatment on GvL. Mice in all experimental arms were monitored daily for any sign of leukemia (based on hind-leg paralysis) or aGvHD. Phenotype of leukemia or aGvHD in each group is represented in Fig. 4A. Mice in A20 alone and A20 + WSE group showed signs of leukemia (Fig. 4A, B). Mice in A20 + BMT and A20 + BMT + WSE group did not showed any sign of leukemia, suggesting no disruption of GvL effect by WSE (Fig. 4A, B). Mice in A20 + WSE group showed delayed onset of leukemia (Fig. 4B), and better overall survival (Fig. 4C) compared to A20 alone group (HR = 0.55 [0.18-1.65], P = .06). Although this difference is not statistically significant, the trend possibly hints at anti-leukemic effect of WSE. The median survival in the experimental groups was 16, 17, and 14 days, respectively, in the A20, A20 + WSE, A20 + BMT groups. However, the median survival was not reached in the A20 + BMT + WSE group (Fig. 4C).
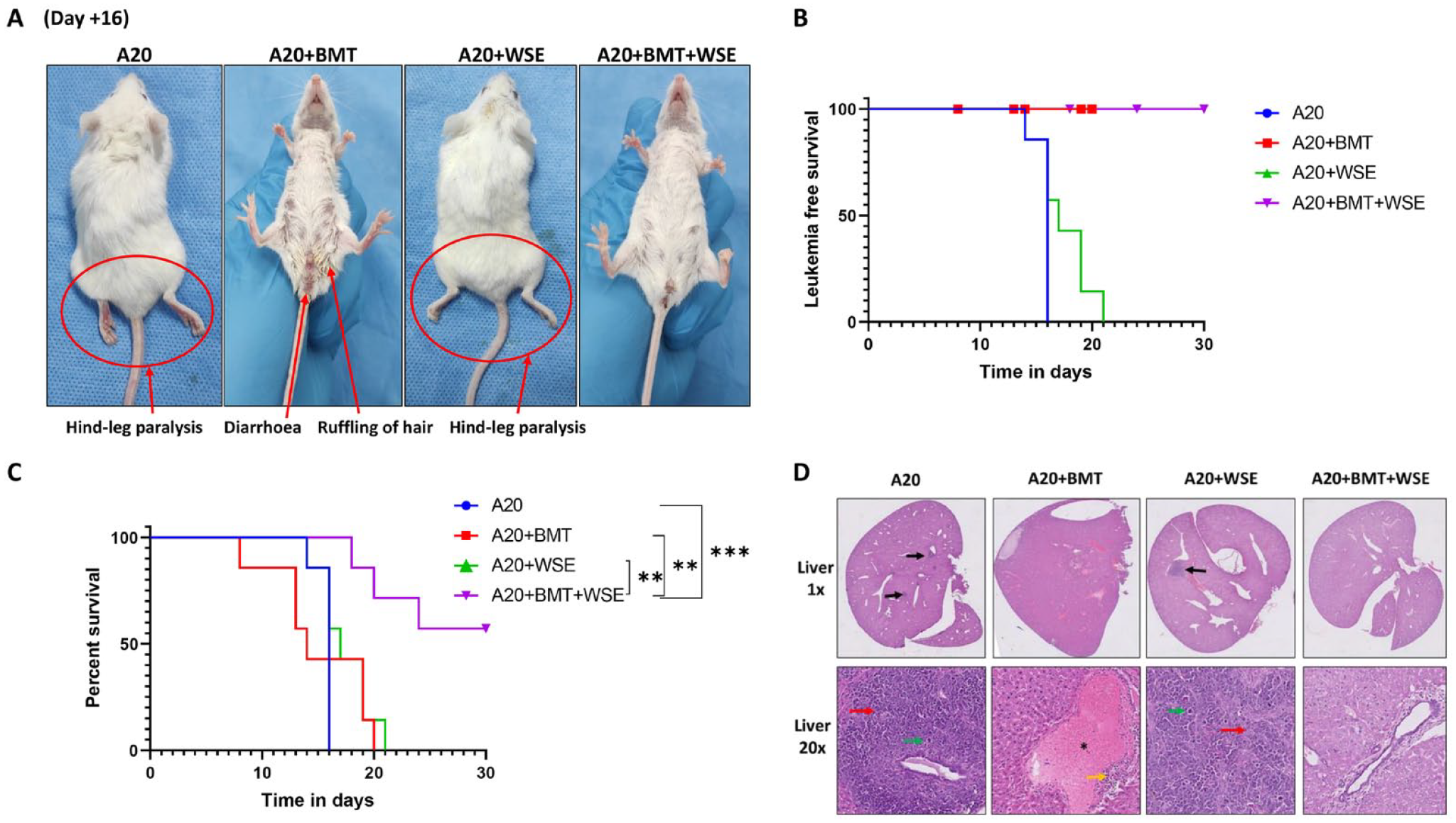
Effect of WSE treatment on GvL. Following radiation, mice were divide into (1) A20, (2) A20 + BMT, (3) A20 + WSE, (4) A20 + BMT + WSE groups. (A) All mice were closely monitored for any signs of leukemia (based on hind leg paralysis) or aGvHD throughout the study. (B) Time-to-event data such as leukemia free survival and (C) overall survival were analyzed in all experimental arms using Kaplan–Meier plot and Mantel-Cox log-rank test. (D) As and when mice became morbid either due to leukemia or GvHD, they were humanely sacrificed. Liver was collected for microscopic examination for the presence of tumor nodules or features of aGvHD. In Fig. 2B, death due to non-leukemic causes were censored. Censored points are represented with blue circle, red square, green triangle and inverted purple triangle in A20, A20+BMT, A20+WSE and A20+BMT+WSE groups respectively. Data is represented as mean ± SEM. n = 7 mice/group. Black arrow—tumor nodules; red arrow—nuclear atypia; green arrow—mitosis; yellow arrow—moderate chronic inflammation; black star—portal vein congestion. (H and E) magnification 1x and 20x. aGvHD: acute graft versus host disease; BMT: bone marrow transplantation; GvHD: graft versus host disease; GvL: graft versus leukemia; WSE: Withania somnifera extract. *P ≤ .05; **P ≤ .01; ***P ≤ .001.
A20 cells tend to form tumor nodules in the liver. We further confirmed the death due to leukemia or GvHD by histological analysis of liver (Fig. 4D). Liver of mice in A20 and A20 + WSE showed multiple nodular deposits of a high-grade malignant tumor composed of large cells with moderate to marked nuclear atypia, brisk mitosis including atypical forms and scant eosinophilic cytoplasm. Consistent with leukemia-free survival, A20 + WSE group had less tumor burden than A20-alone group. A20 + BMT group showed signs of aGvHD in the form of moderate chronic inflammation and portal vein congestion. However, A20 + BMT + WSE group neither showed leukemic nor signs of aGvHD (Fig. 4D). These results suggest that WSE do not hamper GvL effect while mitigating aGvHD.
WSE in the Treatment of Established GvHD
We next evaluated the anti-GvHD efficacy of WSE, wherein we first allowed mice to develop GvHD and then WSE treatment was initiated from day +7 of transplantation. The median CS after 2 weeks of treatment with WSE was 6.5, 3, 1, and 3, respectively, in the GvHD control, 75 mg/kg of WSE, 250 mg/kg of WSE and 500 mg/kg of WSE (Fig. 5A). Continuous loss of weight was observed in the GvHD control group, however, treatment with WSE at all doses halted weight loss (Fig. 5B). Furthermore, overall survival was found to increase significantly in treatment groups compared to the GvHD control arm (P ≤ .05). Hazard ratio (HR) in comparison with GvHD control was 0.32 (0.10-0.95) for 75 mg/kg of WSE, 0.16 (0.05-0.5) for 250 mg/kg of WSE, and 0.25 (0.08-0.75) for 500 mg/kg of WSE. The median survival was 21.5, 30, 30 days respectively in the GvHD control, 75 mg/kg and 500 mg/kg WSE groups (Fig. 5C), whereas it was not reached in 250 mg/kg WSE group (Fig. 5C).
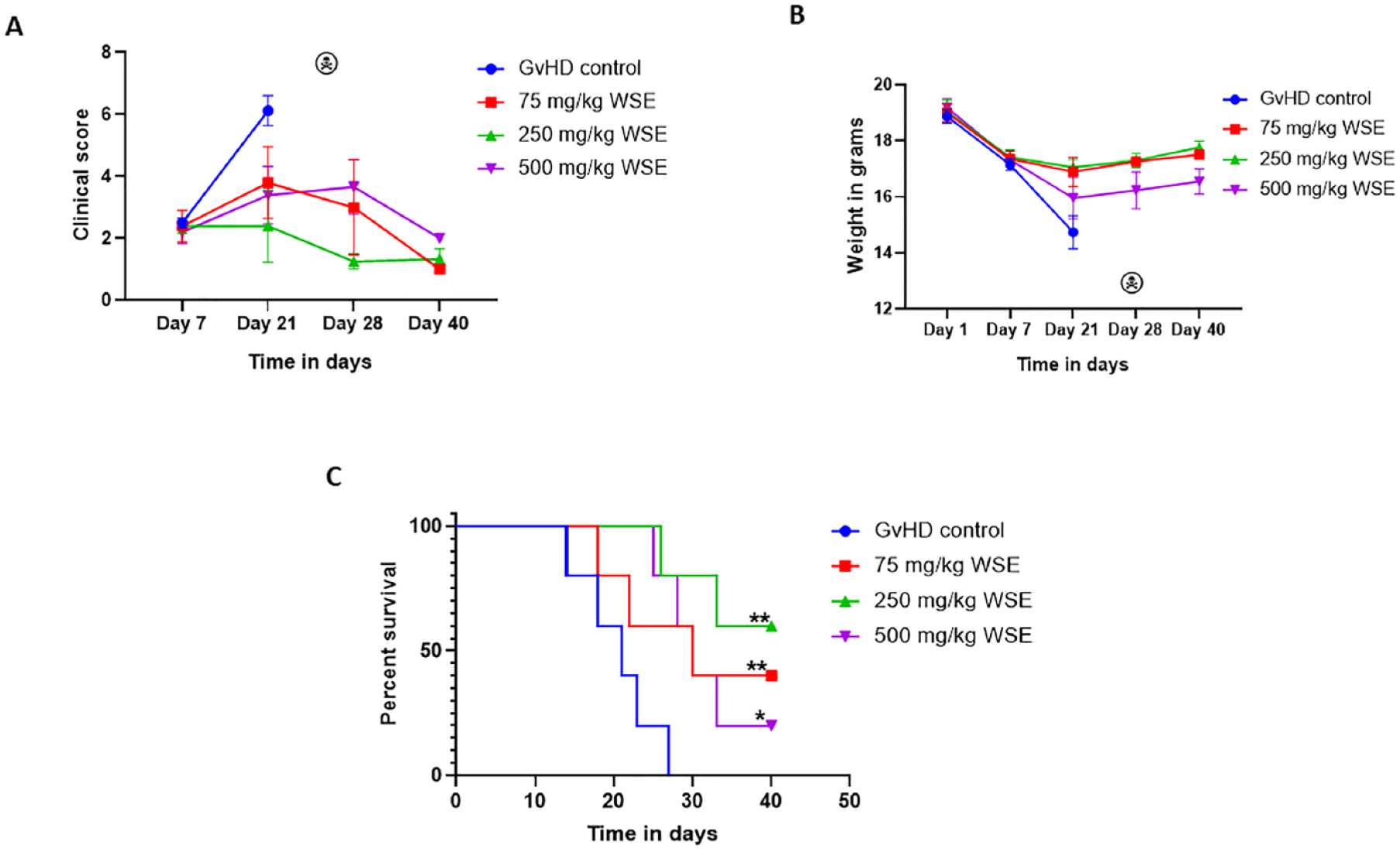
Anti-GvHD efficacy of WSE. To evaluate the anti-GvHD efficacy of WSE, posttransplantation mice were allowed to develop GvHD and WSE treatment at three different dose levels (75, 250, and 500 mg/kg) was started from day +7. (A) aGvHD clinical score and, (B) body weight were recorded at different time points as shown. (C) Overall survival among study groups was analyzed using Kaplan–Meier plot and Mantel-Cox log rank test. Data is represented as mean ± SEM. n = 5 mice/group. aGvHD: acute graft versus host disease; GvHD: graft versus host disease; SEM: standard error of the mean; WSE: Withania somnifera extract. *P ≤ .05; **P ≤ .01.
Discussion
The development of novel interventions to prevent and treat aGvHD remains a challenge 21 . Herein, we demonstrated the utility of WSE against aGvHD. Our finding suggests that early (prophylactic) or late (therapeutic) administration of WSE to mice can mitigate the occurrence or severity of aGvHD and improve survival significantly. In addition, WSE could also inhibit inflammatory cytokine secretion and protect aGvHD target organs. We also demonstrated that WSE does not obstruct the beneficial GvL effect.
Earlier, we established the inhibition of lymphocyte proliferation and cytokine secretion from mouse lymphocytes treated ex vivo with withaferin-A (WA), a principle component of WSE 22 . Next, we established that ex vivo graft manipulation with WA could prevent aGvHD 23 . Furthermore, to bring WA one-step closure to clinic, we established safety, pharmacokinetics and anti-GvHD efficacy of oral WA13,24. Pharmacodynamically, WA was shown to regulate JAK/PI3K/NF-kB/Akt/mTOR signaling and exert potent immune-modulatory response 13 . Thus, WA is an attractive candidate for development as an anti-GvHD agent. However, development of a pure compound as a drug is a long-drawn process associated with huge costs. On the contrary, extracts of Withania somnifera containing WA are available as nutraceuticals around the world and therefore could be an alternative to pure WA should they have comparable activity. In fact, WSE has been tested in several human trials 12 , and its safety is well established up to doses equivalent to 216 mg of WA per day 25 . For these reasons, we went on to test the efficacy of WSE in the mouse model of aGvHD.
WSE is the main component of tradition Rasayana and serves as a remedy against a plethora of human diseases 26 . It is well known for its ability to enhance age, vitality, and protection against numerous diseases 27 . In addition to this, WSE also has anticancer, anti-inflammatory, and immune modulatory properties28,29. Turrini et al. reported the antileukemic activity of WSE 30 . Our observations that WSE resulted in a slight increase in survival and reduced tumor burden in mice transplanted with leukemic cells (A20) corroborates these findings. This antitumor efficiency of WSE may synergise with antileukemic activity of the graft in patients with leukemia who undergo transplantation, and could decrease the chance of disease relapse as well. The immune-modulatory ability of WSE was reported by Singh et al. through inhibition of NFkB and AP-1 proteins 31 . Furthermore, Singh et al. and Naidoo et al. reported inhibition of inflammatory cytokine secretion from human peripheral blood mononuclear cells following ex vivo treatment with WSE31,32. A recent study by Kaur et al. reported the inhibition of inflammatory molecules including PPARγ, MCP-1, iNOS, IL-1β, TNFα, and IL-6 by WSE via inhibition of JAK2-STAT3 and NFkB signaling 33 . Consistent with these studies, we also found a significant decrease in pro-inflammatory cytokine secretion in mice treated with WSE. In addition, we also reported increased IL-10 levels, an anti-inflammatory cytokine. We further reported that mice treated with WSE showed protection of gut, liver, and skin from GvHD induced destruction of these tissues. In line with our results, several preclinical studies have demonstrated the protection of the gut, liver, and skin by WSE in different experimental settings other than GvHD34,35.
In this study, we tested WSE at three dose levels (25, 75, and 250 mg/kg) for the prophylaxis experiment, wherein we found that 75 and 250 mg/kg of WSE provided equal protection. Therefore, for further prophylactic experiments 75 mg/kg WSE dose was chosen. On the contrary, for treatment of established GvHD experiments, 75, 250, and 500 mg/kg WSE doses were selected and we found highest efficacy at 250 mg/kg of WSE. A three-fold higher effective dose of WSE for GvHD therapy compared to prophylaxis could be explained by lower absorption associated with GvHD of the gut.
A limitation of this study is we could not measure residual leukemic cells in our GvL model. Generally, in preclinical mouse experiments where human-derived leukemic cells are injected (xenotransplantation), leukemia burden or residual leuekemic cells in mice are measured by assessing the percentage of human CD45+ (hCD45+) cells using flow cytometry. The syngeneic transplantation model of leukemia used in this study (A20 cells derived from BALB/c transplanted into BALB/c), precludes the flow cytometry-based detection of residual leukemic cells. Owing to these reasons, we opted for the traditional and acceptable method of measuring GvL response based on onset of hind leg paralysis and tumor nodules in the liver13,16,17. The current standard of care for GvHD is associated with significant clinical and/or financial toxicities36,37. Therefore, a safe and cost-effective intervention for the management of GvHD is appealing. WSE has gained popularity in the nutra-health sector, it being the 12th bestselling nutraceutical of 2020 38 , which is also a reflection of its affordability. Safety of the product used in our study is already established up to 2000 mg/kg/day in nonclinical acute and subacute toxicity studies 39 , and up to 4,800 mg/day in a phase 1 clinical trial 25 . Recently, pharmacokinetics of WSE in rats demonstrated oral bioavailability of withanolides such as withanoside IV, WA, 12-deoxy-withastramonolide, and withanolide A 40 . Based on our extensive preclinical safety, efficacy, pharmacokinetics, and pharmacodynamic investigations, and the safety of WSE, a pilot clinical study trial (CTRI/2023/07/055361) for the treatment of aGvHD is currently under investigation at our center.
In conclusion, in this study, we explored the prophylactic and therapeutic efficacy of WSE against aGvHD. WSE mitigated the occurrence and severity of aGvHD and improved survival of mice by protecting target organ damage and inhibiting the inflammatory milieu without compromising the beneficial GvL effect. Based on these findings and its compelling safety profile, WSE is currently under investigation in a phase 2 clinical trial for the treatment of aGvHD at our center.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241226573 – Supplemental material for Withania Somnifera Extract Mitigates Experimental Acute Graft versus Host Disease Without Abrogating Graft Versus Leukemia Effect
Supplemental material, sj-docx-1-cll-10.1177_09636897241226573 for Withania Somnifera Extract Mitigates Experimental Acute Graft versus Host Disease Without Abrogating Graft Versus Leukemia Effect by Saurabh Kumar Gupta, Dievya Gohil, Mohd Bashar Momin, Subhash Yadav, Akanksha Chichra, Sachin Punatar, Anant Gokarn, Sumeet Mirgh, Nishant Jindal, Lingaraj Nayak, Lal Hingorani, Navin Khattry and Vikram Gota in Cell Transplantation
Footnotes
Acknowledgements
The authors express their gratitude to Dr. Jayant Goda and his team for helping them during the animal irradiation. They thank the laboratory animal house facility-ACTREC for supply and maintenance of the animals. They are also thankful to Mr. Jayraj Kasale from photography department of ACTREC, for helping them prepare the publication standard images.
Author Contributions
SKG: Conceptualization; Writing-original draft; Data curation; Formal analysis; Investigation; Methodology; Software; Validation; Visualization. DG: Data curation; Investigation; Validation; Visualization. MBM: Data curation; Validation; Visualization. SY: Data curation; Investigation; Software, Visualization. AC: Conceptualization; Investigation; Methodology. SP: Conceptualization; Investigation; Methodology. AG: Conceptualization; Investigation; Methodology; SM: Conceptualization; Investigation; Methodology. NJ: Conceptualization; Investigation; Methodology. LN: Conceptualization; Investigation; Methodology. LH: Resources; Writing-review and editing. NK: Conceptualization; Project administration; Supervision; Validation; Investigation; Methodology; Writing-review and editing. VG: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Supervision; Validation; Visualization; Writing-review and editing.
Availability of Data and Materials
Data will be made available upon request to the corresponding author.
Ethical Approval
The study was approved by the Institutional Animal Ethics Committee ACTREC (project no. 19/2022).
Statement of Human and Animal Rights
All animal procedures were carried out in compliance with the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) and ARRIVE guidelines.
Statement of Informed Consent
This research does not involve human subject, and therefore informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LH is the managing director of Pharmanza Herbal Pvt. Ltd. which is licensed to manufacture WSE for human use as a nutraceutical. He had no role in the design or conduct of the study, nor in the interpretation of results. Other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are thankful to Tata Memorial Center—Intramural Annual Scientific Funding (Grant no.: 4598) for supporting this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
