Abstract
Considering the limitations of intraportal transplantation (Tx), we sought to establish an alternative approach for it—transplanting islets onto the liver surface (LS) by optimizing adipose tissue-derived stem cell (ADSC) co-Tx procedures with a gelatin hydrogel nonwoven fabric (GHNF). In the in vivo study, we examined the use of the GHNF, the effectiveness of islet covering materials, and preferred procedures for ADSC co-Tx using a syngeneic rat model. Immunohistochemical staining was performed to evaluate the extracellular matrix (ECM) expression and angiogenesis. In the in vitro study, we analyzed the culture supernatants to identify crucial factors secreted from ADSCs in different ADSC co-Tx procedures. It was shown that the GHNF should be used to cover the islets but not to embed internally (encapsulate) them. Utilization of the GHNF in LS Tx resulted in significantly better glucose changes (P = 0.0002) and cure rate of diabetic recipients (P = 0.0003) than the use of a common adhesion barrier. Although neovascularization was comparable among groups, ECM reconstitution tended to be higher when the GHNF was used. ADSC co-Tx further enhanced ECM reconstitution only when ADSCs were cultured in the GHNF before islet Tx. Leptin, vascular endothelial growth factor (VEGF), epidermal growth factor (EGF), and several chemokines were identified as candidate factors for enhancing ECM reconstitution (P < 0.001). The inhibition assay using antagonist suggested that leptin might be at least in part responsible for the difference in transplant efficiency in distinct ADSC co-Tx methods. This study showed that the GHNF effectively improved the outcomes of LS islet Tx, mainly due to ECM reconstitution around the islets. Furthermore, we established a novel method of LS islet Tx by combining a GHNF with ADSCs, which is equally effective as intraportal Tx.

Keywords
Introduction
Pancreatic islet transplantation (Tx), in which isolated islets are commonly infused into the portal vein, has become a promising therapy since the development of the Edmonton protocol 1 . Nevertheless, instant blood-mediated inflammatory reaction (IBMIR), characterized by the innate immune chain activation of both coagulation and complement cascades, is recognized as a major cause of a poor long-term graft survival2–5. Several researchers have investigated alternative transplant sites that might prevent IBMIR, such as the renal subcapsular, intraperitoneal, and subcutaneous spaces. Among these, the subcutaneous space stands out as a potent extrahepatic candidate owing to its minimal invasiveness and easy accessibility. However, the subcutaneous space is a challenging environment for islets, owing to factors such as hypoxia and hypovascularity 6 . The renal subcapsular space has been experimentally recognized as a favorable transplant site because it avoids direct islet-blood contact. However, it is deemed impractical for clinical therapy because type 1 diabetes patients tend to have diabetic nephropathy.
The liver surface (LS) offers several advantages over portal vein, including substantial vascularization and oxygen supply, without concerns related to intraportal (IPO) transplant procedures, such as IBMIR, portal hypertension, portal thrombosis 7 , and hemorrhage8–10. Furthermore, this site provides a sufficiently large transplant space that enables us to perform repeated Txs with minimal invasiveness via laparoscopic surgery. We previously reported that LS Tx using syngeneic keratinocyte sheets is an attractive method for avoiding islet-blood contact 11 . However, in addition to the lot-to-lot variation problems 12 , severe adhesion occurred between the diaphragm and LS; thus, large amounts of islet grafts are required to cure diabetic animals, suggesting a need for alternative materials to effectively cover the transplanted islets.
One possible alternative to keratinocyte sheets is adhesion barrier (AB) film, which was originally produced to prevent adhesion among several tissues. It is already clinically available, and lot-to-lot variation issues may be solved using the established production procedures. However, to our knowledge, there are no reports on the favorable interactions between AB film and transplanted islets. Another possible alternative is a gelatin hydrogel nonwoven fabric (GHNF), which was originally developed for culturing mesenchymal stromal cells (MSCs) 13 . The GHNF is a biodegradable three-dimensional (3D) gelatin fiber sheet that has been stably industrialized with strong tissue affinity 14 . Notably, it has been clinically used to treat patients with Bell’s palsy by combining it with basic fibroblast growth factor. We recently reported that GHNF pretreatment effectively improves the outcomes of subcutaneous islet Tx 15 and that 6 weeks is the ideal duration for GHNF pretreatment 16 . As one of the main mechanisms of the beneficial effects of the GHNF is extracellular matrix (ECM) reconstitution for transplanted islets, this fabric may be useful as not only a covering material but also a suitable scaffold for islet grafts, even in LS Tx.
Adipose tissue-derived stem cells (ADSCs) are MSCs that possess self-renewal and multidifferentiation potential 17 . Many studies have reported that ADSCs secrete several growth factors, anti-inflammatory cytokines, and anti-apoptotic factors 18 . ADSC co-Tx promotes engraftment of islets18,19 and hepatocytes 20 . However, in the case of IPO islet Tx, which is considered the gold standard worldwide, ADSC co-Tx may carry a risk of inducing portal embolism and/or thrombosis, owing to the sticky characteristics of ADSCs. In this regard, ADSC co-Tx may be a safe and attractive strategy for improving the outcomes of LS islet Tx.
In this study, we evaluated the effects of the GHNF on the outcomes of LS islet Tx. Furthermore, we sought to establish a novel method for islet Tx at the LS by optimizing ADSC co-Tx procedures in combination with the GHNF.
Materials and methods
Animals
All animals were treated in accordance with the “Guide for the Care and Use of Laboratory Animals” published by the National Institutes of Health, as well as the guidelines for animal experiments and related activities established at Tohoku University (approved protocol ID:2022 MdA-080). Male Lewis rats (Japan SLC Inc., Shizuoka, Japan), 9–13 weeks old, were used as donors, recipients, and resource of ADSCs. All surgical procedures were performed under isoflurane anesthesia (Viatris Inc., Tokyo, Japan), and all efforts were made to minimize suffering.
The induction and diagnosis of diabetes in the recipients
Diabetes was induced by an intravenous infusion of 70 mg/kg streptozotocin (Sigma-Aldrich, Inc., St. Louis, MO, USA). Rats whose non-fasting blood glucose (BG) levels were ≥400 mg/dL on two consecutive measurements were considered as diabetic. Serial BG levels were determined, and recipients whose non-fasting BG levels were <200 mg/dL on two consecutive measurements were considered to reach normoglycemia.
Islet isolation
Rat islet isolation and culture were performed as described previously 21 . Islets were cultured overnight in Roswell Park Memorial Institute-1640 (RPMI; Thermo Fisher Scientific Inc., Tokyo, Japan) medium containing 5.5 mmol/L glucose and 10% fetal bovine serum (FBS) at 37°C in 5% CO2 and humidified air before Tx.
Isolation and culture of ADSCs
Rat ADSCs were isolated as previously reported20,22. The medium was changed daily, and when the cells reached 80% to 90% confluence, they were detached from the dishes using 0.05% trypsin-0.53 mmol/L ethylenediaminetetraacetic acid (EDTA; Thermo Fisher Scientific Inc.) at 37°C for 5 min, suspended in CELLBANKER 1plus (Takara-Bio, Shiga, Japan), and cryopreserved in liquid nitrogen. ADSCs were used at Passage 3. Viability was assessed using the trypan blue exclusion assay and was confirmed to be >90% at the time of passaging and Tx. To analyze the phenotype, flow cytometry was performed. ADSCs were labeled with fluorescein isothiocyanate-conjugated anti-rat cluster of Differentiation (CD) 29 antibody (Becton, Dickinson and Company, Franklin Lakes, NJ, USA), anti-rat CD31 antibody (Thermo Fisher Scientific Inc., Waltham, MA, USA), anti-rat CD44 antibody (Thermo Fisher Scientific Inc.), anti-rat CD45 antibody (Thermo Fisher Scientific Inc.), and anti-rat CD90 antibody (Becton, Dickinson and Company). The data acquired with the BD AccuriTM C6 Flow Cytometer (BD Biosciences, Franklin Lakes, NJ, USA) were analyzed using BD AccuriTM C (BD Biosciences). The phenotypes of rat ADSCs were strongly positive for CD 29, CD44, and CD90, and negative for CD31 and CD45.
Islet Tx
Islets were precisely quantified using a 20-gauge injection needle sheath (Surflo®; TERUMO BCT, Inc., Tokyo, Japan) before islet Tx. In LS and intraperitoneal Tx, islets were transplanted using an airtight syringe (Hamilton Co., Reno, NV, USA). For IPO Tx, islets were infused into the portal vein using a 25-gauge winged needle (TERUMO BCT Inc.).
Experimental groups of the in vivo model
In the encapsulating method (EM 10 group, n = 10), 10 islet equivalents (IEQs)/g of body weight (BW) were suspended in 700 μl of RPMI containing 10% FBS and dropped onto swollen GHNF (KYOTO MEDICAL PLANNING Co., Ltd., Kyoto, Japan) using a pipette tip (ART™ Wide Bore Filtered Pipette Tips 2069G; Thermo Fisher Scientific Inc.). In this method, the islets are embedded inside the GHNF. The GHNF was placed on the right hepatic lobe, and an anti-adhesive barrier composed of sodium hyaluronate and carboxymethyl cellulose (Seprafilm®; Genzyme Corp., Cambridge, MA, USA), with a thickness of approximately 0.05 mm and cut to a 2 cm diameter, was applied over it to prevent postoperative adhesion (Fig. 1a). In the covering method (CM 10 group; n = 10), islets were directly placed on the right hepatic lobe, spread thinly using a glass rod, and then covered with the GHNF swollen with islet medium. Subsequently, they were also covered with AB film (Fig. 1a).

(a) This figure illustrates the experimental design of this study. In the EM 10 group (n = 10), 10 IEQs/g of body weight were dropped onto the GHNF, and then the GHNF encapsulating islet grafts was placed on the right hepatic lobe, followed by covering with AB film. In the CM 10 group (n = 10), the same number of islets was directly placed on the right hepatic lobe and then covered with the GHNF and AB film. (b, c) The changes in the BG levels and body weight after islet Tx in the CM 10 (filled circle, n = 10) and EM 10 (filled square, n = 10) groups. The CM 10 group showed significantly better glucose and body weight changes than the EM 10 group (**P < 0.01). (d) The cure rate curve of diabetic rats after islet Tx in each group. The cure rate at 56 days after islet Tx in the CM 10 group (90%) was significantly higher than that in the EM 10 group (50%) (*P < 0.05). (e) The glucose tolerance profiles of the CM 10 and EM 10 groups. The results of the IVGTT in the CM 10 (filled circle, n = 9) and EM 10 (filled square, n = 10) groups at approximately 56 days after islet Tx. (f) The AUC of the IVGTT in each group is shown. The AUC in the CM 10 group was significantly lower than that in the EM 10 group (*P < 0.05). AB: Adhesion barrier, AUC: area under the curve, BG: blood glucose, CM: covering method, EM: encapsulating method, GHNF: gelatin hydrogel nonwoven fabric, IEQs: islet equivalents, IVGTT: intravenous glucose tolerance test, Tx: transplantation.
To investigate the utility of the GHNF in LS Tx (Fig. 2a), the CM was used as an optimized procedure according to the abovementioned study. In the group receiving islet Tx onto the LS (LS 8.5 group, n = 7), 8.5 IEQs/g of BW were simply placed on the right hepatic lobe. In the group receiving islet Tx onto the LS along with GHNF (GHNF 8.5 group, n = 7), the same number of islets was placed on the right hepatic lobe and then covered with the GHNF. In the group receiving islet Tx onto the LS with AB film (AB 8.5 group, n = 7), the same number of islets was placed on the right hepatic lobe and then covered with AB film. In the group receiving islet intraperitoneal Tx (IP 8.5 group, n = 7), the same number of islets was placed into the abdominal cavity, excluding the LS.

(a) This figure shows the experimental design of this study. In the islet Tx onto the LS group (LS 8.5 group, n = 7), 8.5 IEQs/g of body weight were placed on the right hepatic lobe. In the islet Tx onto the LS with GHNF group (GHNF 8.5 group, n = 7), the same number of islets was placed on the right hepatic lobe and then covered with the GHNF. In the islet Tx onto the LS with AB film group (AB 8.5 group, n = 7), the same number of islets was placed on the right hepatic lobe and then covered with AB. In the islet intraperitoneal Tx (IP 8.5) group (n = 7), the same number of islets was placed into the abdominal cavity, excluding the LS. (b, c) The outcomes of islet engraftment after transplantation (approximately 1,700 IEQs). The changes in the BG levels and body weight after islet Tx in the LS 8.5 (filled circle, n = 7), GHNF 8.5 (open square, n = 7), AB 8.5 (filled triangle, n = 7), and IP 8.5 (filled downward triangle, n = 7) groups. The GHNF 8.5 group showed significantly better glucose changes than the other groups (*P < 0.05, **P < 0.01). (d) The cure rate curve of diabetic rats after islet Tx in each group. The cure rate at 28 days after islet Tx in the GHNF 8.5 group (100%) was significantly higher than that in the other groups (**P < 0.01). (e) The glucose tolerance profiles of the LS 8.5, GHNF 8.5, AB 8.5, and IP 8.5 groups. The results of the IVGTT in the LS 8.5 (filled circle, n = 7), GHNF 8.5 (open square, n = 7), AB 8.5 (filled triangle, n = 7), and IP 8.5 (filled downward triangle, n = 7) groups at approximately 28 days after islet Tx. (f) The AUCs of the IVGTT in each group are shown. The AUCs tended to be lower in the GHNF 8.5 group than the other groups. AB: Adhesion barrier, AUC: area under the curve, BG: blood glucose, CM: covering method, EM: encapsulating method, GHNF: gelatin hydrogel nonwoven fabric, IEQs: islet equivalents, IP: intraperitoneal, IVGTT: intravenous glucose tolerance test, LS: liver surface, Tx: transplantation.
To examine the additional effects of ADSCs on transplant outcomes, we compared two different approaches using islets at a marginal dose of 5 IEQs/g BW (Fig. 3a). In the first procedure, ADSCs (5.0 × 105) were suspended in medium for ADSCs (ADSC-1; Kojin-Bio Co., Ltd., Sakado, Japan) and cultured overnight on the GHNF prior to islet Tx. Subsequently, the islets were placed on the right hepatic lobe and covered with ADSC-preimplanted GHNF (ADSC-preimplanted GHNF 5.0 group, n = 13). In the other approach, the islets were co-transplanted with ADSCs (5.0 × 105) onto the LS and then covered with the GHNF (ADSC co-Tx with GHNF 5.0 group, n = 13). GHNF group (n = 13) Islet IPO Tx (IPO 5.0 group, n = 9) was also performed to compare transplant efficiency with that of an optimized LS Tx procedure.

(a) This figure illustrates the experimental design of this study. In the islet Tx onto the LS with GHNF group (GHNF 5.0 group, n = 13), 5 IEQs/g of body weight were placed on the right hepatic lobe and then covered with the GHNF. To investigate the additional effects of adipose tissue-derived stem cells (ADSCs) on transplant outcomes, ADSCs (5.0 × 105 cells) were cultured on the GHNF overnight prior to islet Tx. The same number of islets was placed on the right hepatic lobe and then covered with ADSC-preimplanted GHNF (ADSC-preimplanted GHNF 5.0 group, n = 13). In an alternate procedure, the same number of islets was co-transplanted with ADSCs (5.0 × 105 cells) onto the LS and then covered with the GHNF (ADSC co-Tx with GHNF 5.0 group, n = 13). In the islet IPO Tx group (IPO 5.0 group, n = 9), the same number of islets was infused into the portal vein. (b, c) The changes in the BG levels and body weight after islet transplantation (Tx) in the GHNF 5.0 (open square, n = 13), ADSC-preimplanted GHNF 5.0 (filled diamond, n = 13), ADSC co-Tx with GHNF 5.0 (open circle, n = 9), and IPO 5.0 (open downward triangle, n = 9) groups. The IPO 5.0 group showed favorable outcomes for both body weight increase and BG reduction (*P < 0.05). Likewise, in the ADSC-preimplanted GHNF 5.0 group, there was a favorable trend in BG levels and body weight. In contrast, the ADSC co-Tx with GHNF 5.0 group showed a worse trend than the GHNF 5.0 group. (d) The cure rate curve of diabetic rats after islet Tx in each group. The cure rate at 56 days after islet Tx in the ADSC-preimplanted GHNF 5.0 group (100%) was higher than that in the IPO (88.9%), GHNF 5.0 (77.8%), and ADSC co-Tx with GHNF 5.0 (55.6%) groups (*P < 0.05). (e, f) The glucose tolerance profiles of the GHNF 5.0 (open square, n = 13), ADSC-preimplanted GHNF 5.0 (filled diamond, n = 13), ADSC co-Tx with GHNF 5.0 (open circle, n = 13), and IPO 5.0 (open downward triangle, n = 9) groups. There were no significant differences among the groups. ADSC: Adipose tissue-derived stem cell, AUC: area under the curve, BG: blood glucose, CM: covering method, EM: encapsulating method, GHNF: gelatin hydrogel nonwoven fabric, IEQs: islet equivalents, IPO: intraportal, IVGTT: intravenous glucose tolerance test, Tx: transplantation.
Experimental groups of the in vitro model
To examine the influence of ADSC-preimplantation using the GHNF, in vitro assays were performed (Fig. 4a). To mimic the ADSC-preimplanted GHNF group in vivo model, ADSCs (7.5 × 105) were seeded onto the GHNF and cultured overnight. The following day, the GHNF with seeded ADSCs was washed three times with serum-free RPMI, placed on the upper insert of an EZ-BindShut® II Microplate 6Well (AGC TECHNO GLASS Co., Ltd., Shizuoka, Japan), and cultured for 3 days (Pre group, n = 9). To mimic the ADSC co-Tx with GHNF group in vivo model, ADSCs (7.5 × 105) were scattered in the well and cultured for 3 days (Co-Tx group, n = 9). Cultured supernatants were collected from both groups.

(a) To identify variations in the humoral factors secreted by ADSCs depending on culture conditions, in vitro analyses were conducted. A comprehensive analysis of 30 factors, including those promoting islet engraftment, was performed (Table 1). In the Pre group, ADSCs (7.5 × 105 cells) were seeded on the GHNF and cultured overnight. The following day, the ADSC-preimplanted GHNF was placed on the upper insert and cultured for 3 days (n = 9). In the Co-Tx group, ADSCs (7.5 × 105 cells) were scattered in the well and cultured for 3 days (n = 9). In both groups, the cultured supernatants were collected and analyzed. (b) The summary of the distinct factors in the cultured supernatants between the Pre (gray box, n = 9) and Co-Tx (black box, n = 9) groups. Leptin, EGF, VEGF, fractalkine, GROα, and IL-2 levels were significantly higher in the Pre group, while IP-10 and RANTES showed significantly elevated levels in the Co-Tx group (*P < 0.05, **P < 0.01). ADSC: Adipose tissue-derived stem cell, epidermal growth factor (EGF), GHNF: gelatin hydrogel nonwoven fabric, GROα: growth-related oncogene-alpha, IL-2: interleukin-2, IP-10: γ-interferon inducible protein-10, RANTES: regulated upon activation, normal T-cell expressed and secreted, VEGF: vascular endothelial growth factor.
Intravenous glucose tolerance test (IVGTT)
The IVGTT was conducted 28 to 35 or 56 to 64 days after islet Tx, as described previously 23 . The area under the curve (AUC) was calculated from the BG values at each time point.
Immunohistochemical staining
At 33 to 38 days after islet Tx, liver tissue was procured from the recipient rats (LS 8.5, GHNF 8.5, AB 8.5, and ADSC-preimplanted GHNF 8.5 groups, n = 7 each). In the IP 8.5 group, engrafted islets could not be identified. Immunohistochemical staining was performed using anti-insulin (ab181547; Abcam, Cambridge, UK), anti-von Willebrand factor (vWF) (ab6994; Abcam), anti-collagen III (ab7778; Abcam), anti-collagen IV (ab6586; Abcam), anti-laminin (ab11575; Abcam), anti-fibronectin (ab2413; Abcam), and anti-vitronectin (ab45139; Abcam) antibodies. The secondary antibody reaction was performed using EnVision+ System-HRP-labeled polymer anti-rabbit (K4003; DAKO, Glostrup, Denmark). For heparan sulfate (HS) staining, an anti-HS antibody (370260-S; Amsbio, Abingdon, England) was used as the primary antibody, and the secondary antibody reaction was performed using Goat Anti-Mouse IgG2b heavy chain (ab97250; Abcam). Color development was performed using DAB (4800-30-07; Trevigen Inc., Gaithersburg, MD, USA). To evaluate the neovascularization of the LS around the islets, the number of vWF-positive vessels was counted. In the GHNF 8.5 group, insulin staining was performed to assess the site and condition of the engrafted islets. Figure 5a showed the location and status of the engrafted islets. The number of vWF-positive blood vessels around the islets was counted (Fig. 5b). Regarding the expression of collagen III 24 , collagen IV 25 , laminin26,27, fibronectin 28 , vitronectin 29 , and HS30–33, we defined “positive” as marked immunopositivity that was detectable (Fig. 6a) 34 . Two slices were prepared for each sample, and the count was conducted using one or more slices to confirm islet engraftment. All counts were performed by a pathologist under blinded conditions.
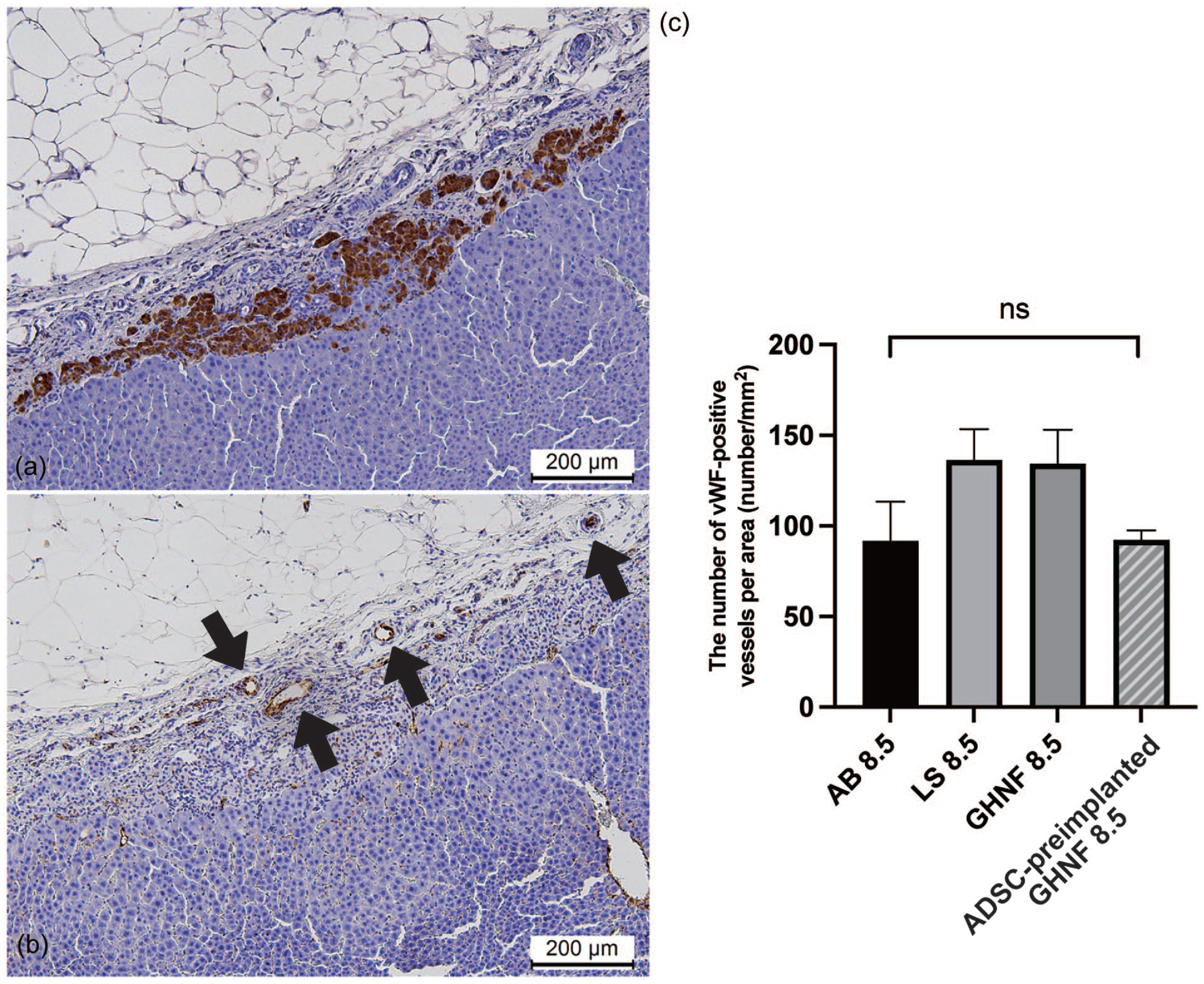
Immunohistochemical analyses of vWF-positive vessels. (a) Photomicrographs of insulin staining 35 days after islet transplantation. (b) The vWF-positive vessels (black arrows) on the liver surface around the islets were counted. Magnification: ×200. Calibration bars: 200 μm. (c) The number of vWF-positive blood vessels was comparable in the AB 8.5, LS, and GHNF 8.5 groups but lower in the ADSC-preimplanted GHNF 8.5 group (*P < 0.05). AB: Adhesion barrier, ADSC: Adipose tissue-derived stem cell, vWF: von Willebrand factor, GHNF: gelatin hydrogel nonwoven fabric, LS: liver surface.

Immunohistochemical analyses of extracellular matrix. (a) Representative photomicrographs of collagen III, collagen IV, fibronectin, HS, laminin, and vitronectin staining. “Positive” for collagen III, collagen IV, fibronectin, HS, laminin, and vitronectin indicates that distinct immunopositivity was detectable on the liver surface around the islets. “Negative” indicates that immunopositivity was undetectable. Magnification: ×200. Calibration bars: 200 μm. (b) The rates of collagen III, collagen IV, fibronectin, HS, laminin, and vitronectin positivity in the AB 8.5 (black box), LS 8.5 (light gray box), GHNF 8.5 (deep gray box), and ADSC-preimplanted GHNF 8.5 (stripe box) groups. The immunopositivity rate of the AB 8.5 group was significantly lower than that of the other groups (*P < 0.05, **P < 0.01). The positivity rates of all extracellular matrices examined in this study tended to increase in the ADSC-preimplanted GHNF 8.5 group. AB: adhesion barrier, ADSC: adipose tissue-derived stem cell, GHNF: gelatin hydrogel nonwoven fabric, HS: heparan sulfate.
The immunoassay for the protein expression of inflammatory mediators and islet-protective factors
We analyzed the concentration of several proteins in the culture supernatant obtained from the aforementioned in vitro models. The concentrations of insulin-like growth factor (IGF)-2, hepatocyte growth factor (HGF), and stromal cell-derived factor 1 (SDF-1) were measured using a Mouse/Rat/Porcine/Canine IGF-2 Immunoassay Quantikine ELISA Kit (R&D Systems, Minneapolis, MN, USA), Mouse/Rat HGF Quantikine ELISA kit (R&D Systems), and Rat SDF-1 ELISA kit (KE1347; ImmunoWay Biotechnology Co., Suzhou, China), respectively. For the measurement of other inflammatory mediators, we utilized the Milliplex MAP Rat Cytokine/Chemokine 27-plex Magnetic Bead Panel-premixed 27 plex (Merck KGaA, Darmstadt, Germany), and the Bio-Plex200 System (Bio-Rad, Hercules, CA, USA).
Inhibition of candidate factor in the ADSC co-culture supernatant
According to the results of abovementioned in vitro immunoassay (2.7 and 2.10), we focused on the contribution of leptin to the beneficial effects in the Pre group because a sharp difference was observed in the leptin levels between the two groups. Freshly isolated islets (approximately 130 IEQs) were collected in 700 μl of 3 types of ADSC co-culture supernatants as follows (each group has an in vitro experimental number of n = 12); (1) Co-Tx group, (2) Pre group, or (3) Pre group supernatant with 35 ng/mL Super Rat Leptin Antagonist (SRLA) (SLAN-4, Protein Laboratories Rehovot Ltd., Rehovot, Israel) (Pre + SRLA group). The islets were then cultured for 26 hours in a 24-well plate (Ultra-Low Attachment Surface, Corning) and islet viability and function were evaluated using a fluorescein diacetate (FDA; 10 mg/ml, F-7378; Sigma-Aldrich)/propidium iodide (PI; 0.5 mg/ml, Calbiochem 537059, Sigma-Aldrich) assay (Supplementary Fig. A, B) 35 and a respiratory activity assay (Supplementary Fig. C) as described previously36,37. In the FDA/PI assay, 20 islets with diameters ranging from 150 to 200 μm were randomly selected from each group for analysis. In the respiratory activity assay, five islets with an approximate diameter of 200 μm were randomly selected from each group for analysis.
Statistical analyses
All data are presented as the mean ± standard error. All statistical analyses were performed using the JMP Pro 16 software program (SAS Institute Inc., Cary, NC, USA). Trends in BW, BG levels, and the IVGTT results were analyzed using mixed-effect models, with the Tukey–Kramer test used for post-hoc comparisons. Comparisons of the AUC in the IVGTT were conducted using a one-way analysis of variance (ANOVA) (t-test for comparison between two groups), followed by the Tukey–Kramer test. Diabetes cure rates were plotted using Kaplan–Meier curves, followed by a log-rank test for comparison. In cases involving more than three groups, the Bonferroni test was applied for post-hoc analysis. The number of positive vessels on immunohistochemical staining, FDA/PI ratio, and respiratory index were assessed using a one-way ANOVA, followed by the Tukey–Kramer test. The concentrations of inflammatory mediators in the culture supernatants were compared using Wilcoxon’s test. Statistical significance was set at P < 0.05.
Results
Optimization of the LS islet Tx method using the GHNF
The increase in BW was significantly greater (P = 0.0003) (Fig. 1b) in the CM 10 group than in the EM 10 group, and the BG also showed a significant decrease (P = 0.0028) (Fig. 1c). In the CM 10 group, 90% of the recipients achieved normoglycemia (CM 10 group: 9/10 vs. EM 10 group: 5/10, P = 0.0107) (Fig. 1d). In the IVGTT, the CM 10 group demonstrated significantly better BG trends (P = 0.0274) (Fig. 1e) and a lower AUC (CM 10 group: 22,503.9 ± 1,544.9 vs. EM 10 group: 28,549.3 ± 1,465.6, P = 0.0113) (Fig. 1f) than the EM 10 group.
Effect of the GHNF on LS islet Tx
The rate of weight gain was highest in the GHNF 8.5 group, although no significant difference was observed (Fig. 2b). The BG level was significantly lower in the GHNF 8.5 group than in the AB 8.5 group (P = 0.0002), IP 8.5 group (P = 0.0002), and LS 8.5 group (P = 0.0453) (Fig. 2c). The cure rate was significantly higher in the GHNF 8.5 group than in the other groups (GHNF 8.5 group: 100% [7/7] vs. LS 8.5 group: 43% [3/7] vs. AB 8.5 group: 0% [0/7] vs. IP 8.5 group: 0% [0/7], P = 0.0003) (Fig. 2d). In the IVGTT, the GHNF 8.5 group showed a significant decrease in BG compared with the AB 8.5 group (P = 0.0056) and IP 8.5 group (P = 0.0005) (Fig. 2e). The AUC, like the IVGTT, was also significantly lower in the GHNF 8.5 group (Fig. 2f).
Comparison of protein levels of inflammatory mediators and islet-protective factors in cultured supernatants between the Pre and Co-Tx groups
We analyzed 30 inflammatory mediators and islet-protective factors using multiplex assays and ELISA kits (Table 1). Between the Pre and Co-Tx groups, leptin (P = 0.0001), epidermal growth factor (EGF) (P = 0.0053), vascular endothelial growth factor (VEGF) (P = 0.0003), fractalkine (P = 0.013), growth-related oncogene-alpha (GROα) (P = 0.0003), and interleukin-2 (IL-2) (P = 0.0397) levels were significantly higher in the Pre group, whereas γ-interferon inducible protein-10 (P = 0.0003) and regulated upon activation, normal T-cell expressed and secreted (P = 0.001) levels were significantly higher in the Co-Tx group (Fig. 4b).
Comparison of protein levels of inflammatory mediators and islet-protective factors in cultured supernatants between the Pre and Co-Tx groups.

*P < 0.05. **P < 0.01.
Validation of ADSC co-Tx method using the GHNF
The ADSC-preimplanted GHNF 5.0 group tended to show better outcomes than the GHNF 5.0 group (Fig. 3b, c). In contrast, ADSC co-Tx with GHNF 5.0 group showed similar outcomes to the GHNF 5.0 group. Likewise, the cure rate of the ADSC-preimplanted GHNF 5.0 group was 92.5%, while the ADSC co-Tx with GHNF 5.0 group achieved only 53.8% (GHNF 5.0 group: 61.5% [8/13] vs. ADSC co-Tx with GHNF 5.0 group: 53.8% [7/13] vs. ADSC-preimplanted GHNF 5.0 group: 92.5% [12/13] vs. IPO 5.0 group: 88.9% [8/9], P = 0.0004) (Fig. 3d). Regarding glucose tolerance profiles, no significant differences were observed among the groups (Fig. 3e, f).
Immunohistochemical staining of the LS around the islets
Insulin staining was utilized to identify engraftment sites of islet grafts (Fig. 5a), and the number of vWF-positive blood vessels around the islets (Fig. 5b) was quantified. The number of vWF-stained vessels in the LS around the islet grafts tended to be lower in the AB 8.5 group than in the LS 8.5 group, although this difference was not statistically significant. The LS 8.5 and GHNF 8.5 groups exhibited similar vWF-positive vessel counts (LS 8.5 group: 133.6 ± 15.3/mm2, GHNF 8.5 group: 137.1 ± 9.9/mm2, AB 8.5 group: 86.7 ± 14.0 /mm2, ADSC-preimplanted GHNF 8.5 group: 91.7 ± 13.5/mm2, P = 0.0943) (Fig. 5c).
In the islet engraftment region, we investigated the proportions of collagen III, collagen IV, laminin, fibronectin, vitronectin, and HS-positive sections (Fig. 6a) to evaluate the degree of ECM reconstitution. The immunopositivity rate of the AB 8.5 group was significantly lower than that of the other groups, whereas the ADSC-preimplanted GHNF 8.5 group consistently showed a tendency toward an increase in all ECM components compared to the other groups (collagen III, P = 0.0001; collagen IV, P = 0.0003; laminin, P < 0.0001; fibronectin, P = 0.0345; vitronectin, P < 0.0001; and HS, P < 0.0001) (Fig. 6b).
Evaluation of the islet viability and function by inhibiting leptin in the ADSC co-culture supernatant
The islet injury rate, as indicated by FDA/PI assay, was significantly lower in the Pre group compared to the Co-Tx and Pre + SRLA groups (Co-Tx group: 20.9% ± 3.2% vs. Pre group: 11.4% ± 1.7% vs. Pre + SRLA group: 19.7% ± 1.8% respectively, P = 0.0091, Supplementary Fig. A, B).
In the respiratory activity assay, although the differences did not reach statistical significance, the Pre group exhibited the highest respiratory index, while the Co-Tx group showed the lowest index. Moreover, the Pre group tended to approximate the Co-Tx group when the SRLA was added (Co-Tx group: 2.5% ± 0.15% vs. Pre group: 3.0% ± 0.14% vs. Pre + SRLA group: 2.7% ± 0.19% respectively, P = 0.085, Supplementary Fig. C).
Discussion
In this study, we demonstrated for the first time that the GHNF effectively improved the outcomes of LS islet Tx. These beneficial effects are likely based on ECM reconstitution around the islets rather than on neovascularization (Figs. 5 and 6). The present data show that the GHNF can be used to cover the islet grafts in LS islet Tx but should not encapsulate them. Furthermore, we successfully established a novel method for LS islet Tx by combining GHNF with ADSCs. Notably, the transplant outcome of this novel approach was comparable to that of IPO Tx, which is the current gold standard worldwide. Interestingly, unlike islets, ADSCs appeared to secrete favorable factors for islet engraftment when cultured by the GHNF compared to when covered by the GHNF. Corroborating this in vitro observation, the in vivo transplant outcome was better when ADSCs were pre-implanted into the GHNF prior to LS islet Tx than when ADSCs were co-transplanted with islets onto the LS.
Regarding LS islet Tx in a rat model, some attempts have been made using chitin films 38 and keratinocyte sheets 11 . However, all of these approaches caused robust adhesion between the liver and diaphragm, subsequently requiring a substantial number of islets for diabetes treatment (chitin film: 12–14 IEQs/g, keratinocyte sheet: 8 IEQs/g). However, in this study, adhesion was limited, and only 5 IEQs/g were needed to cure a diabetic rat, suggesting that one diabetic recipient can theoretically be achieved normoglycemia by islets obtained from a single donor. Another method for islet Tx onto the LS involves using islet cell sheets attached to a 16% gelatin gel, as reported in previous studies 39 . Furthermore, recent technological innovations have enabled researchers to perform co-Tx strategies using materials with islet-protective properties, for example, one involving pretreatment of human amniotic membrane covered with silk threads on the mouse LS, followed by islet Tx into the space created after removal of silk thread 40 and another involving the cultivation of islets on ADSC sheets for subsequent co-Tx 19 . Compared to these procedures, our method using GHNF has several advantages, including less invasiveness because it does not require pretreatment, effective utilization of the LS area without occupying non-transplant sites due to pretreatment, and uniform products. Taken together, our method seems to be simpler and more stable than the other methods.
The superior performance of the CM group compared to the EM group can be explained by previous reports suggesting that the favorable transplant outcomes may be attributed to islet contact with the parenchyma of the solid organs41,42. Indeed, we previously reported that hepatocyte engraftment was improved by islet co-Tx, which has the characteristic of penetrating the renal parenchyma 43 . We also observed that functioning islets penetrated beyond the liver capsule and infiltrated the hepatic parenchyma in the LS Tx model 11 . However, almost no islets infiltrated the hepatic parenchyma in both the CM and EM groups. As the transplant outcome of the LS group was considerably better than that of the IP group, we can speculate that islet contact with the LS through the liver capsule is also crucial for islet engraftment in the LS model. However, there are reports of islets forming clusters and infiltrating into the liver parenchyma in LS Tx 38 , and since it is unclear what exactly occurs on the LS, it is necessary to consider other possibilities, such as the infiltration of islets due to remodeling of the LS. As it is difficult to address the mechanisms behind this in this study, further investigation is awaited.
The GHNF not only induces angiogenesis and ECM formation44,45 but also serves as a substrate with inherent biodegradability upon cell proliferation13,14. In subcutaneous islet Tx, where poor vascularity and low oxygen tension cause suboptimal outcomes, we reported that the GHNF pretreatment improves transplant outcomes through angiogenesis promotion 16 and ECM reconstitution15,16. Likewise, various reports have highlighted the ability of ADSCs to facilitate engraftment and provide protective effects to islets18,19,46,47. These effects are mediated by a number of factors, including VEGF, interleukin-6, and interleukin-10, as well as anti-inflammatory effects attributed to transforming growth factor-β1, HGF, and SDF-148,49. Therefore, the combination of the GHNF and ADSCs is anticipated to enhance Tx outcomes through increased angiogenesis and ECM reconstitution 50 . As expected, synergistic effects were also observed. However, angiogenesis was unexpectedly not enhanced by the GHNF or ADSC introduction.
Regarding the ECM, the positive rates of collagen III, collagen IV, laminin, vitronectin, and HS increased with the introduction of the GHNF, and further improvement was observed with the addition of ADSCs. HS is known to play a crucial role in maintaining the islet function; thus, its increased expression due to the GHNF and ADSCs may have significantly contributed to the observed beneficial results. These findings are consistent with our previous report 15 , indicating that ECM reconstitution is one of the main mechanisms at work in GHNF pretreatment in subcutaneous islet Tx.
However, why the addition of ADSCs did not promote but rather attenuated angiogenesis in this study remains unclear. While ADSCs are commonly reported to exert anti-inflammatory effects, under the conditions of chronic inflammation in diabetes, ADSCs have been found to actively secrete inflammatory cytokines, shifting from an anti-inflammatory state to a pro-inflammatory one 51 . This may be a possible explanation as to why ADSCs attenuated angiogenesis in this study. In addition, we should keep in mind that environmental factors are completely different between liver and subcutaneous tissues. Of note, the number of vWF-positive blood vessels in the LS Tx model, irrespective of GHNF introduction, in this study (ranging 130–140/mm2) was much higher than that of the subcutaneous Tx model in our previous report 16 (46.1 ± 3.9/mm2). In the present LS Tx model, the addition of ADSCs lowered the vWF-positive blood vessel number (range: 80–90/mm2), but this number was still almost two times higher than that of the subcutaneous Tx model. This unique characteristic may at least partially explain why ECM reconstitution was more significant than angiogenesis in the LS Tx model.
We comprehensively examined 30 factors, including cytokines known for their representative islet-protective and engraftment-promoting effects. As a result, leptin, VEGF, EGF, IL-2, GROα, and fractalkine were considered potential candidate factors in this study. In ADSC co-Tx with islets, cell-to-cell interactions and actions mediated by soluble factors are known to play a complex role 52 . In this study, the ADSC-preimplanted GHNF group showed better outcomes than the ADSC co-Tx with GHNF group. Detecting precise cell contacts in vivo is challenging; however, based on the above results, ADSCs may work mainly by the release of soluble factors. Leptin, a peptide hormone derived from adipocytes, is known to influence BG homeostasis 53 . Leptin-treated cells have been reported to show the upregulated expression of genes related to various ECM components, including HS 54 and fibronectin 55 , in the context of epithelial mesenchymal transition of malignant tumors56,57. Although the effect of leptin on islet grafts in an in vivo model remains unknown, it is plausible that leptin promotes islet engraftment by stimulating ECM production. In support of this speculation, an inhibition assay in the present in vitro model revealed that leptin inhibition counteracted the beneficial effects of ADSC co-culture supernatant obtained from the Pre group on islets, suggesting that leptin might be at least in part responsible for the difference of transplant efficiency observed in distinct ADSC co-Tx methods. VEGF and leptin have also been reported to stimulate ECM generation in liver stellate cells58,59, whereas IL-2 regulates ECM production through T cells 60 .
The sharp difference between the two types of ADSC co-Tx procedures in this study may be explained by the culture environment of ADSCs. ADSCs may secrete favorable factors due to the 3D environment 61 when implanted into the GHNF, subsequently improving the outcomes of LS islet Tx. The second suggestion appears more logical than the first.
It may be presumed in this study that the number of ADSCs in the ADSC-preimplanted GHNF group, where ADSCs were cultured on the GHNF overnight, was higher than in the ADSC co-Tx with GHNF group when the in vitro study was initiated. However, increasing the number of ADSCs or extending the culture period on the GHNF did not affect the in vivo outcomes, suggesting that the impact was limited (data not shown). Owing to the fragility of islets, it was challenging to fully replicate the spatial relationship between islets, ADSCs, and GHNF observed in in vivo experiments within the in vitro setting. Therefore, as an alternative approach to mimic the in vivo experiments, we measured the differences in cytokine secretion between culturing ADSCs on GHNF and culturing ADSCs alone. The in vitro experimental results of this study are presented exclusively in the Supplementary Figure. Furthermore, it cannot be ruled out that elevated leptin in the ADSC-preimplanted GHNF group may have directly contributed to BG reduction, as leptin is known to improve insulin sensitivity 53 . Given that the leptin concentration in the recipient blood may not be very high, the impact of this possibility is marginal. Conducting leptin inhibition tests in the in vivo model is a crucial next step for future investigations. Finally, the number of ADSCs and the size of the GHNF should be optimized before clinical application. Thus, further investigations using larger animal models are required.
In conclusion, this study showed that the GHNF, but not common AB film, effectively improved the outcomes of LS islet Tx, mainly due to ECM reconstitution around the islets. Furthermore, we established a novel method for LS islet Tx by combining the GHNF with ADSCs, which is comparable to IPO Tx. Considering the number of islets required to maintain stable BG levels, this novel approach may be used as a substitute for IPO Tx when it is not suitable for reasons such as coagulation abnormalities and portal hypertension.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251328419 – Supplemental material for A novel method of pancreatic islet transplantation at the liver surface using a gelatin hydrogel nonwoven fabric
Supplemental material, sj-docx-1-cll-10.1177_09636897251328419 for A novel method of pancreatic islet transplantation at the liver surface using a gelatin hydrogel nonwoven fabric by Yukiko Endo Kumata, Akiko Inagaki, Yasuhiro Nakamura, Takehiro Imura, Ryusuke Saito, Takumi Katano, Shoki Suzuki, Kazuaki Tokodai, Takashi Kamei, Michiaki Unno, Kimiko Watanabe, Yasuhiko Tabata and Masafumi Goto in Cell Transplantation
Footnotes
Acknowledgements
The authors thank Kozue Maya and Megumi Goto (Division of Transplantation and Regenerative Medicine, Tohoku University) for their technical assistance. The authors also acknowledge the support of the Biomedical Research Core of Tohoku University, Graduate School of Medicine, and Tohoku Advanced Medical Research and Incubation Center (TAMRIC).
Abbreviations
ADSC: adipose tissue-derived stem cell; ANOVA: analysis of variance; AUC: area under the curve; BG: blood glucose; BW: body weight; CM: covering method; CD: cluster of differentiation; ECM: extracellular matrices; EGF: epidermal growth factor; EM: encapsulated method; FDA: fluorescein diacetate; GROα: growth-related oncogene-alpha; GHNF: gelatin hydrogel nonwoven fabric; HGF: hepatocyte growth factor; HS: heparan sulfate; IBMIR: instant blood-mediated inflammatory reaction; IEQs: islet equivalents; IGF-2: insulin-like growth factor 2; IL-2: interleukin-2; IP: intraperitoneal; IPO: intraportal;IVGTT: intravenous glucose tolerance test; LS with GHNF: liver surface with GHNF; LS: liver surface; LS with AB: liver surface with adhesion barrier; MSCs: mesenchymal stromal cells; PI: propidium iodide; RPMI: Roswell Park Memorial Institute-1640 medium; SDF-1: stromal cell-derived factor-1; SRLA: Super Rat Leptin Antagonist; STZ: streptozotocin; 3D: three-dimensional; Tx: transplantation; VEGF: vascular endothelial growth factor; vWF: von Willebrand factor.
Ethical Considerations
This study was approved by Tohoku University (approved protocol ID:2022 MdA-080).
Authors’ Contributions
Y.E.K. participated in the research design, the performance of the research, and the writing of the paper. A.I. participated in the performance of the research and the writing of the paper. Y.N. participated in pathological analysis in the research. T.I. participated in the performance of the research. R.S. participated in the performance of the research. T.K. participated in the performance of the research. S.S. participated in the performance of the research. K.T. participated in the performance of the research. T.K. participated in the writing of the paper. M.U. participated in the writing of the paper. K.W. participated in the writing of the paper. Y.T. provided technological advice on gelatin hydrogel nonwoven fabrics. M.G. participated in the research design, research performance, and writing of the paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author is a research fellow at the Japan Society for the Promotion of Science (JSPS). This study was supported by a Japanese Grant-in-Aid for Scientific Research (A) (grant no. 18H04056), Research (B) (grant no. 22H03133), and KAKENHI (grant no. 23KJ0134) from JSPS. It is important to note that the funders had no role in the study design, data collection and analysis, decision to publish, or manuscript preparation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare no conflicts of interest in association with this study, although this study was performed according to a patent application agreement with KYOTO MEDICAL PLANNING Co., Ltd.
Statement of Human and Animal Rights
All animals were treated in accordance with the “Guide for the Care and Use of Laboratory Animals” published by the National Institutes of Health, as well as the guidelines for animal experiments and related activities established at Tohoku University (approved protocol ID:2022 MdA-080).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Data Availability Statement
All relevant data included in the paper are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
