Abstract
Subcutaneous islet transplantation is a promising treatment for severe diabetes; however, poor engraftment hinders its prevalence. We previously revealed that a gelatin hydrogel nonwoven fabric (GHNF) markedly improved subcutaneous islet engraftment. We herein investigated whether the addition of adipose tissue–derived stem cells (ADSCs) to GHNF affected the outcome. A silicone spacer sandwiched between two GHNFs with (AG group) or without (GHNF group) ADSCs, or a silicone spacer alone (Silicone group) was implanted into the subcutaneous space of healthy mice at 6 weeks before transplantation, then diabetes was induced 7 days before transplantation. Syngeneic islets were transplanted into the pretreated space. Intraportal transplantation (IPO group) was also performed to compare the transplant efficiency. Blood glucose, intraperitoneal glucose tolerance, immunohistochemistry, and inflammatory mediators were evaluated. The results in the subcutaneous transplantation were compared using the Silicone group as a control. The results of the IPO group were also compared with those of the AG group. The AG group showed significantly better blood glucose changes than the Silicone and the IPO groups. The cure rate of AG group (72.7%) was the highest among the groups (GHNF; 40.0%, IPO; 40.0%, Silicone; 0%). The number of vWF-positive vessels in the subcutaneous space of the AG group was significantly higher than that in other groups before transplantation (P < 0.01). Lectin angiography also showed that the same results (P < 0.05). According to the results of the ADSCs tracing, ADSCs did not exist at the transplant site (6 weeks after implantation). The positive rates for laminin and collagen III constructed around the transplanted islets did not differ among groups. Inflammatory mediators were higher in the Silicone group, followed by the AG and GHNF groups. Pretreatment using bioabsorbable scaffolds combined with ADSCs enhanced neovascularization in subcutaneous space, and subcutaneous islet transplantation using GHNF with ADSCs was superior to intraportal islet transplantation.
Keywords
Introduction
Pancreatic islet transplantation in which isolated islet grafts are injected into the liver through the portal vein has become an effective clinical treatment option for severe type 1 diabetes 1 . However, intraportal islet transplantation has several problems2,3, including damage to islet grafts due to strong innate immune reactions4,5, risks of bleeding and embolism6,7, and difficulty in monitoring and removing islet grafts when needed. Thus, alternative extrahepatic transplant sites should be investigated.
The subcutaneous space has many advantages as a transplant site for islet grafts 8 . It is easily accessible, minimally invasive and easy to monitor or remove islet grafts if necessary9,10. However, subcutaneous islet transplantation requires a large number of islets to control diabetes because the subcutaneous space is a severe environment as the vessels there are a poor source of nutrition and oxygen for transplanted islets 11 . Accordingly, optimization of the subcutaneous environment for transplantation is essential for improving subcutaneous islet engraftment.
Adipose tissue–derived stem cells (ADSCs) can be obtained in large quantities with a relatively simple and noninvasive procedure12,13 and have been reported to improve the revascularization process by secreting anti-apoptotic and angiogenic cytokines14,15. Moreover, various anti-inflammatory factors and growth factors induce extracellular matrix (ECM) composition and improve islet graft survival and function16,17.
We previously reported the usefulness of recombinant peptide (RCP) for improving islet engraftment in subcutaneous transplantation 18 . The main drawback of the RCP device was that it is hardly absorbed in the recipient body and must therefore be removed at the time of islet transplantation, which may destroy newly constructed vessels and ECM. Thus, we next focused on a gelatin hydrogel nonwoven fabric (GHNF) 19 , which is a biodegradable scaffold made from gelatin. It was previously reported to be gradually degraded and absorbed in vivo and replaced by host cells and ECM; thus, removal is unnecessary19,20. We previously reported that preimplantation of GHNF sheets could improve the outcomes of subcutaneous islet engraftment 21 . We also recently reported that the duration of GHNF pretreatment apparently influenced the outcomes of subcutaneous islet transplantation and that 6 weeks appeared to be the ideal duration 22 . However, no significant increase in new vessels in the subcutaneous space was detected.
Accordingly, we investigated whether the addition of ADSCs in combination with GHNF sheets could induce more effective vascularization at the transplant site and improve subcutaneous islet engraftment using a marginal-dose islet graft in a syngeneic mouse transplantation model.
Materials and Methods
Animals
All animals were handled in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health 23 . All experimental protocols (protocol ID: 2022 MdA-022) were approved by the animal experimental committee at Tohoku University. C57BL/6 mice (male; age, 6-14 weeks) (Japan SLC Inc., Shizuoka, Japan) were used as donors and recipients.
C57BL/6 transgenic mice (CAG-EGFP) (male; age, 7–8 weeks) (Japan SLC Inc.) were used for in vivo ADSC tracing. ADSCs were isolated from Green fluorescent protein-transgenic (GFP-Tg) mice.
All surgical operations were performed under general anesthesia. All efforts were made to minimize suffering.
The Induction and Diagnosis of Diabetes in the Recipients
Diabetes was induced by the intravenous injection of streptozotocin (STZ [170 mg/kg]) (Sigma–Aldrich, Inc., MO, USA). Mice whose nonfasting blood glucose levels were ≥400 mg/dL on two consecutive measurements were considered diabetic. Recipients whose nonfasting blood glucose levels were <200 mg/dL on two consecutive measurements were considered to be cured.
Islet Isolation
Islet isolation and culturing were performed as previously described 24 . The islets were cultured in Roswell Park Memorial Institute-1640 medium containing 5.5 mmol/L glucose and 10% fetal bovine serum at 37°C in 5% CO2 overnight before transplantation.
The Isolation and Culture of ADSCs
ADSCs were isolated from C57BL/6 mice (7–8 weeks of age) or GFP-Tg mice, as reported previously 12 . Adipose tissues were obtained from the subcutaneous fat around the lower abdomen. ADSCs at passage 2 to 4 were used for experiments.
In Vivo Tracing of GFP-Positive ADSCs Seeded on GHNF
ADSCs (1.0 × 106) isolated from GFP-Tg mice were seeded on GHNF sheets in ADSC growth medium (ADSC-1; Kojin-Bio Co., Ltd., Sakado, Japan) and incubated overnight at 37°C in 5% CO2 25 . GHNF sheets containing ADSCs were implanted in the subcutaneous space of C57BL/6 mice. To confirm the presence of ADSCs on the GHNF, we obtained pathological samples at 0, 7, 14, and 42 days after implantation and evaluated them using anti-GFP antibody (ab290; Abcam, Cambridge, UK).
Preparation of GHNF With or Without ADSCs and Islet Transplantation
A GHNF (KYOTO MEDICAL PLANNING Co., Ltd., Kyoto, Japan) was prepared using a previously reported method 19 and processed into a circular sheet (diameter, 11 mm; thickness, 0.5 mm). ADSCs were trypsinized with 0.25% trypsin and 0.53 mM ethylenediaminetetraacetic acid for 3 min at 37°C, counted and resuspended in ADSC-1. Then 5.0 × 105 ADSCs were seeded on one sheet of GHNF (total 1.0 × 106 cells for two sheets). GHNF sheets seeded with ADSCs were cultured overnight at 37°C in 5% CO2. Two GHNF sheets sandwiching a silicone spacer were placed into the left dorsal subcutaneous space at 6 weeks before islet transplantation (ADSCs with GHNF: AG group), and GHNF sheets without ADSCs were placed in the same way (GHNF group).
In the Silicone group, a silicone spacer without GHNF was placed into the left dorsal subcutaneous space. To observe the effect of GHNF with or without ADSCs in the subcutaneous islet transplantation, the results were compared using the Silicone group as a control. GHNF sheets were implanted into healthy mice, and then the mice were injected with STZ 7 days before islet transplantation.
After the removal of the silicone spacer, 180 islet equivalents (IEQs) of syngeneic mouse islets were transplanted into the pretreated space using a gastight syringe (Hamilton Co., Reno, NV, USA) in the AG, GHNF, and control groups 18 . According to our previous report, 270 IEQs of islets using GHNF cured the 60% of diabetic mice 22 . Therefore, in this study, we used lower dose of islets to observe the additional effect of ADSCs. We also compared the results of the AG group with those of the intraportal transplantation group (IPO group), that is the conventional method for islet transplantation. In the IPO group, 180 IEQs of islets in a total volume of 300 µL were infused through the portal vein using a 27-gauge Surshield (TERUMO, Inc., Tokyo, Japan) 24 . Recipients were followed by measuring nonfasting blood glucose levels every 3 to 4 days throughout the study period (60 days after islet transplantation).
Intraperitoneal Glucose Tolerance Test
An intraperitoneal glucose tolerance test (IPGTT) was performed 60 to 65 days after islet transplantation, as described previously 26 . The blood glucose curves were generated, and the area under the curve (AUC) was compared.
Immunohistochemical Analyses
After 6 weeks of subcutaneous implantation of GHNF sheets before islet transplantation, recipient subcutaneous tissue was obtained, fixed with 4% paraformaldehyde. In addition, at 7 days after transplantation, tissue was obtained from the site of subcutaneous islet transplantation. Immunohistochemical staining was performed using anti-insulin (ab181547; Abcam), anti-von Willebrand factor (vWF) (ab6994; Abcam), anti-collagen III (ab7778; Abcam), anti-collagen IV (ab6586; Abcam), and anti-laminin (ab11575; Abcam) antibodies. The EnVision + System- HRP labeled polymer anti-rabbit (4003; DAKO, Glostrup, Denmark) was used as a secondary antibody. For the evaluation of neovascularization, the vWF-positive cells in the capsule touching the silicone spacer were counted 27 . In collagen III, collagen IV, and laminin staining, “positive” was defined as marked immunopositivity that was detectable in the fibrous capsule around the islets 28 . More than six sections from each experimental group before islet transplantation and four sections after islet transplantation were evaluated by a pathologist using a blind method (AG; n = 5, GHNF; n = 4, Silicone; n = 5).
Evaluation of Transplanted Islets
All insulin-positive islets at 7 days after transplantation were evaluated. Gross morphology was qualitatively assessed by scoring the islets for shape (irregular border vs. well-rounded) and staining uniformity (not uniform vs. perfectly uniform)29,30. Each parameter was graded from 0 to 1 with 0 equaling the worst and 1 the best score, so that all islets were categorized Grade 1 to 3. The size of islets was also evaluated.
Lectin Angiography
Lectin angiography was performed in the AG (n = 5), GHNF (n = 5), and Silicone (n = 5) groups at 6 weeks after pretreatment 31 . Mice were sedated, and 100 μg/body DyLight® 488 Lycopersican esculentum agglutinin (LEA, tomato lectin; Vector Labs, Burlingame, CA) was injected via the tail vein. Five minutes after lectin injection, animals were perfused through the heart using Na-PO4 buffered 4% paraformaldehyde. Following perfusion, tissues were washed with phosphate-buffered saline and transferred to 30% sucrose.
Tissues were cut using a microtome (thickness, 50 μm). Sections were examined under a confocal microscope (LSM780; Carl-Zeiss, Oberkochen, Germany). The same controls for brightness and exposure times were used among groups. Images were imported into Zeiss IMARIS (Carl-Zeiss, Germany) for volumetry. The vascular volume in subcutaneous capsules surrounding the silicone spacer or GHNF was calculated. To determine the density of blood vessels, the vascular volume was divided by the capsular volume. The brightness value of the vascular area was defined as >20, and the minimum volume was <5000 μm3 to extract artifacts.
Real-Time PCR Using TaqMan Arrays
RNA was extracted from the recipient subcutaneous fibrous capsules surrounding the silicone spacer at 6 weeks after pretreatment 32 (AG; n = 5, GHNF; n = 5). The relative gene expression was determined using a TaqMan array 96-well FAST plate (4413257, Applied Biosystems; Bedford, Massachusetts, US). A TaqMan array plate contains 46 target genes and two assays for candidate endogenous control genes (Table 1). Samples were analyzed using a StepOnePlus Real-Time polymerase chain reaction (PCR) System (Applied Biosystems). The results were analyzed using ExpressionSuite Software ver. 1.3 (Applied Biosystems). The relative quantification (RQ) was calculated using the comparative computed tomography (CT) method. To determine the relative gene expression in the AG group, the samples in the GHNF group were designated as a calibrator. 18S was used as a housekeeping gene.
The List of Analyzed Target Genes.

Immunoassay for the Protein Expression of Inflammatory Mediators and Islet-Protective Factors
The recipient subcutaneous fibrous capsule surrounding the silicone spacer at 6 weeks after pretreatment was obtained and the protein extraction was performed (AG; n = 5, GHNF; n = 5, Silicone; n = 5) 33 . Total protein concentrations in the supernatant were measured using a Pierce BCA Protein Assay kit (Thermo Fisher Scientific Inc., Waltham, MA, USA). Cytokines in the supernatant were measured using a Bio-Plex Pro Mouse Cytokine 23-plex Assay (#M60009RDPD; Bio-Rad, California, USA). Insulin-like growth factor 2 (IGF-2) concentrations in the supernatant were measured using a Quantikine ELISA Mouse/Rat/Porcine/Canine IGF-2 Immunoassay Kit (R&D Systems, Minneapolis, MN, USA).
Statistical Analyses
All data are expressed as the mean ± standard deviation. All statistical analyses were performed using the JMP pro 16 software program (SAS Institute Inc., Cary, NC, USA). Changes in the blood glucose levels were analyzed by mixed-effect model analysis. To compare the data among groups more than three, one-way analysis of variance (ANOVA) was used for analysis. The islet grade was evaluated by Fisher’s exact test. The immunopositive rate in ECM staining was analyzed by Pearson’s chi-squared test. Kaplan–Meier curves were compared using the log-rank test. P values of <0.05 were considered statistically significant.
Results
Characterization of ADSCs
Flow cytometry revealed that ADSCs were negative for CD31 (2.5%) and CD45 (0.1%), whereas were positive for the Sca-1 in 99.0%, CD44 in 86.8%, and CD90 in 99.4% (Fig. 1).
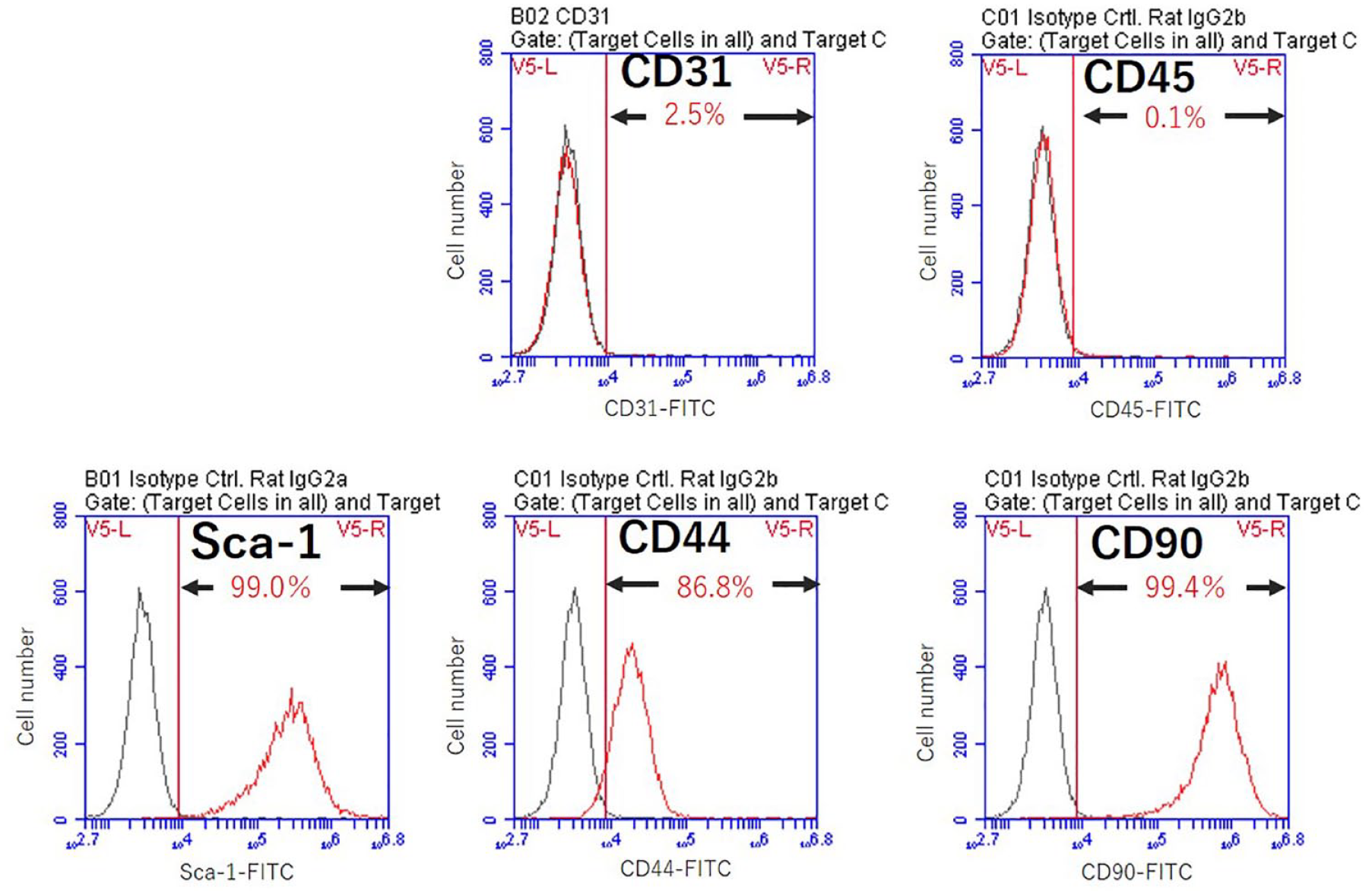
Characterization of ADSCs. Flow cytometry revealed that ADSCs were negative for CD31 (2.5%) and CD45 (0.1%), whereas were positive for the Sca-1 in 99.0%, CD44 in 86.8%, and CD90 in 99.4%.
Comparison of Islet Engraftment After Marginal Islet Mass Transplantation Among the AG, GHNF, Silicone, and IPO Groups
For subcutaneous transplantation, the results were compared using the Silicone group as a control. Blood glucose levels after islet transplantation in the AG group (n = 11) were significantly better than those in the Silicone group (n = 10) (AG; P < 0.001, GHNF; P = 0.369) (Fig. 2A). Blood glucose changes in the AG group were also significantly better than those in the IPO group (n = 10) (P = 0.003) (Fig. 2B). The cure rate of diabetic mice at 60 days after islet transplantation was significantly higher in the AG and GHNF groups (AG, 72.7% [8/11]; GHNF, 40.0% [6/15]; Silicone, 0% [0/10]; AG; P < 0.001, GHNF; P = 0.027) (Fig.2C). Although the difference between the AG and IPO groups was nonsignificant, the cure rate of the AG group was higher than that of the IPO group (AG, 72.7% [8/11]; IPO, 40.0% [4/10], P = 0.329) (Fig.2D).
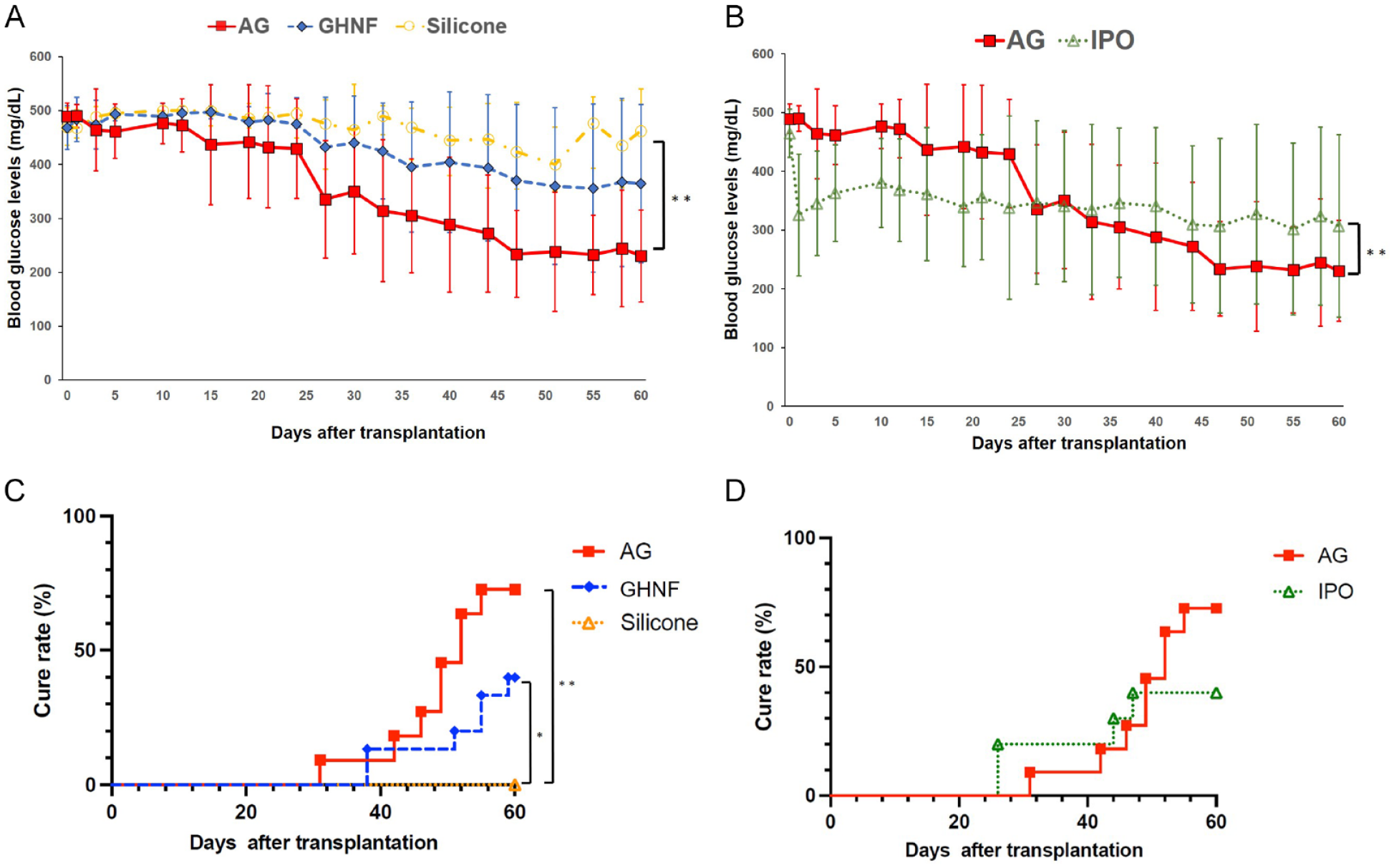
The outcome of islet engraftment after marginal islet mass transplantation (180 IEQs). (A) Changes in blood glucose levels after islet transplantation in the AG (filled square, n = 11), GHNF (filled rhombus, n = 15), and Silicone (open circle n = 10) groups. The AG group significantly showed significantly better glucose changes than the Silicone group (**; P < 0.01). (B) Changes in blood glucose levels after islet transplantation in the AG the IPO (open triangle, n = 10) groups. The AG group showed significantly better glucose changes than the IPO group (**; P < 0.01). (C) The cure rate curve of diabetic mice after islet transplantation in each group. The cure rate at 60 days after islet transplantation in the AG group (72.7%) was significantly higher than that in the Silicone (0%) group (**AG vs. Silicone; P < 0.01, *GHNF vs. Silicone; P < 0.05). (D) The cure rate curve in the AG and IPO groups. The cure rate of the AG group (72.7%) was higher than that of the IPO group (40.0%) (P = 0.329), but the difference did not reach statistical significance.
Intraperitoneal Glucose Tolerance Test
The blood glucose changes of the AG group in the IPGTT were significantly better than those in the Silicone group (AG; P = 0.038, GHNF; P = 0.950) (Fig.3A). There was no significant difference between the AG and IPO groups (P = 0.346) (Fig. 3B). Although the difference did not reach statistical significance, the AUC in the AG group was lower than that in the other groups (AG, 36,466 ± 10,141; GHNF, 41,430 ± 11,526; Silicone, 44,681 ± 5,131; P = 0.201) (Fig. 3C). There was no significant difference between the AG and IPO groups in the AUC (AG, 36,466 ± 10,141; IPO, 41,736 ± 9,929; P = 0.283) (Fig. 3D).
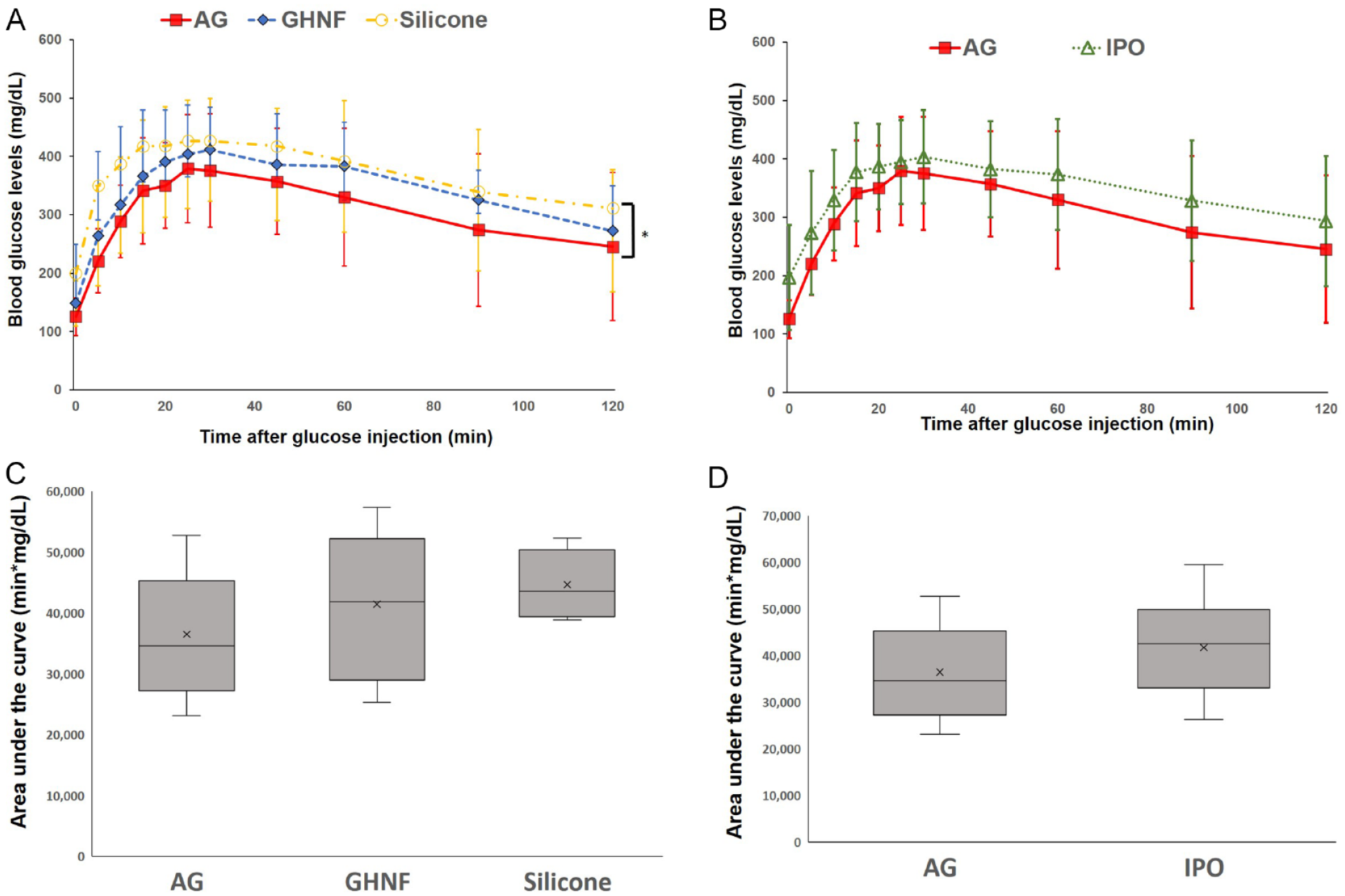
The glucose tolerance profiles of the AG, GHNF, IPO, and Silicone groups. (A) The results of the intraperitoneal glucose tolerance test (IPGTT) in the AG (filled square, n = 11), GHNF (filled rhombus, n = 12), and Silicone (open circle n = 9) groups at approximately 60 days after islet transplantation. The AG group showed significantly better glucose changes than the Silicone group (*, P < 0.05). (B) The results of the IPGTT in the AG and IPO (open triangle, n = 9) groups. (C) The area under the curve (AUC) of the IPGTT in each group is shown. Although the difference did not reach statistical significance, the AUC of the AG group was lower than that of the other groups (P = 0.202). (D) The AUC of the IPGTT in the AG and IPO groups. No significant difference was observed between the two groups (P = 0.283).
Immunohistochemical Analyses
The number of vWF-positive vessels in the capsule around the silicone spacer was counted and divided by the capsular area to examine neovascularization before islet transplantation (Fig. 4A). The number of vWF-positive vessels in the AG group was significantly higher than that in the GHNF or Silicone groups (AG vs. GHNF; P = 0.003, AG vs. Silicone; P < 0.001, GHNF vs. Silicone; P < 0.001) (Fig. 4B). To examine the influence of ADSCs and GHNF pretreatment on the ECM components in subcutaneous capsules, the rate of immunopositive sections, in terms of collagen III, collagen IV, and laminin at the fibrous capsule around the islet grafts, was evaluated (Fig. 5A). Although the rate of collagen IV positivity in the GHNF group was significantly lower than that in the other groups (AG vs. GHNF; P = 0.034, GHNF vs. Silicone; P = 0.023), the rates of collagen III and laminin positivity showed no significant differences (Fig. 5B).
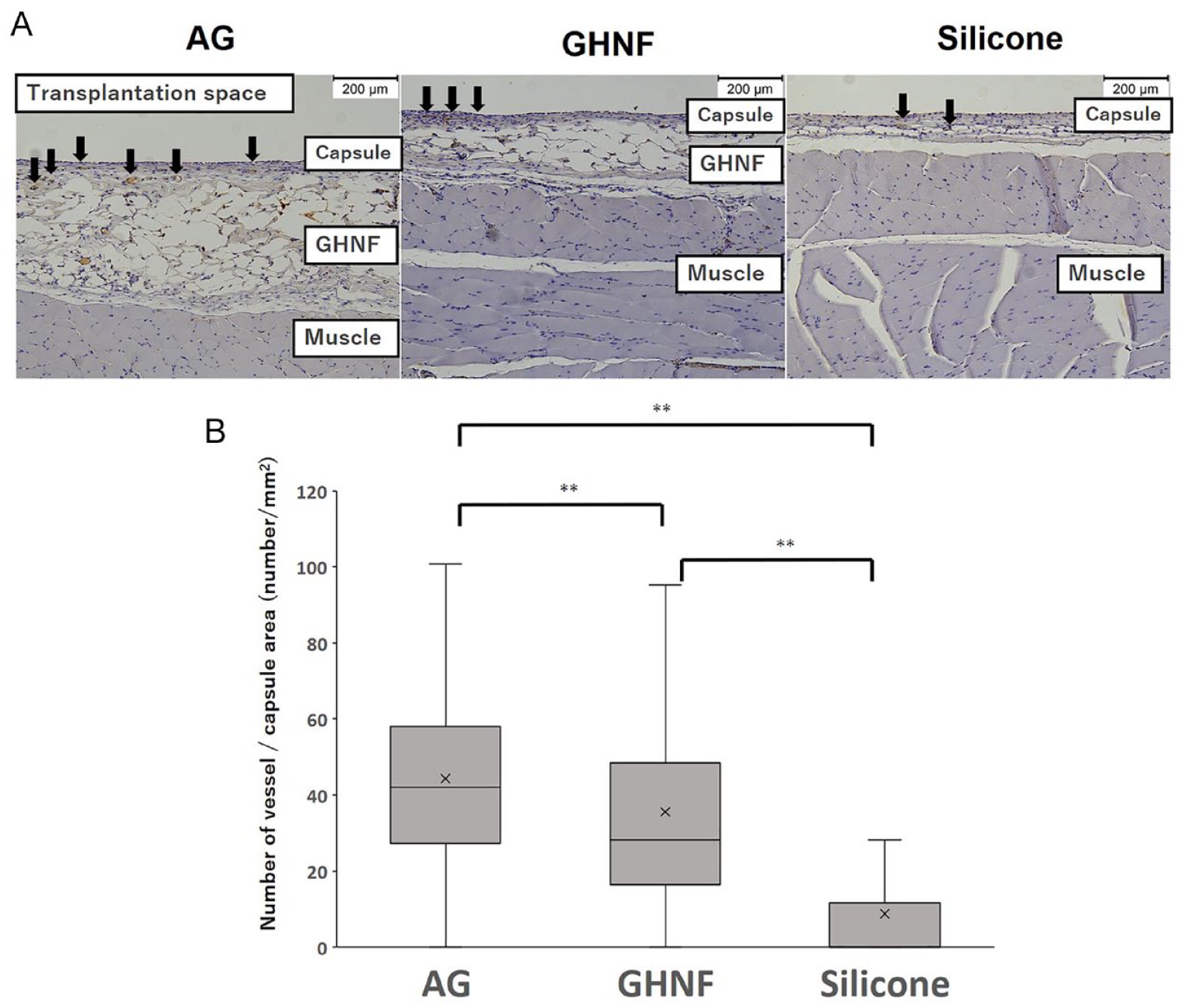
Immunohistochemical analyses of von Willebrand factor (vWF)-positive vessels. (A) Photomicrographs of vWF staining before islet transplantation. The vWF-positive vessels (black arrows) in the capsule around the silicone spacer were counted. Magnification: ×100. Calibration bars: 200 µm. (B) The number of vWF-positive vessels in the AG group was significantly higher than that in the other groups (**, P < 0.01). Black arrows represent the vWF-positive vessels.
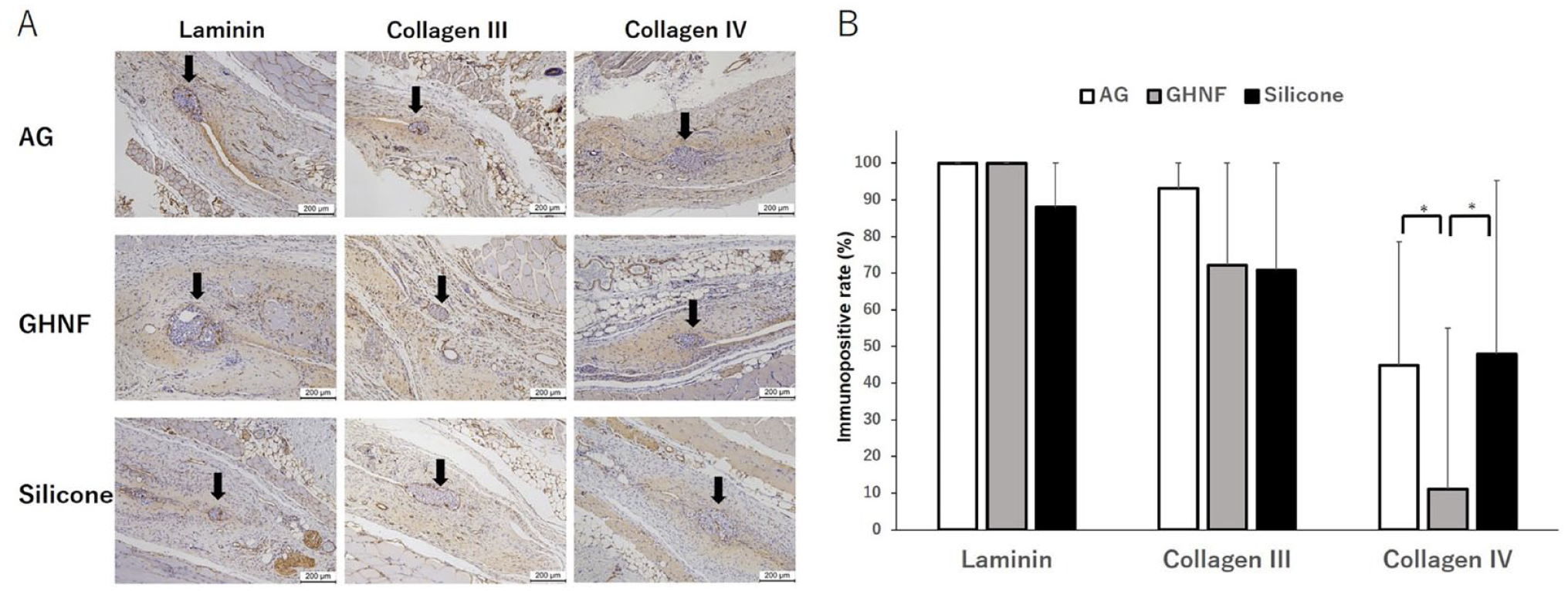
Immunohistochemical analyses of extracellular matrix (ECM). (A) Representative photomicrographs of laminin, collagen III, and collagen IV staining. Black arrows represent the transplanted islets. “Positive” for laminin, collagen III, and collagen IV indicates that distinct immunopositivity was detectable in the fibrous capsule around the islets. Magnification: ×200. Calibration bars: 200 µm. (B) The rates of laminin, collagen III, and collagen IV immunopositivity in the AG (white box), GHNF (gray box), and Silicone (black box). Although the rate of collagen IV positivity in the GHNF group was significantly lower than that in the other groups (*, P < 0.05), there was no significant difference in the rates of collagen III or laminin positivity among the groups.
Evaluation of Transplanted Islets
All insulin-positive islets in a pathological specimen were evaluated at 1 week after islet transplantation (Fig.6A). The percentage of Grade 2 and 3 islets in the AG and GHNF groups was higher than that in the Silicone group (AG: 96.4%, GHNF: 94.4%, Silicone: 77.7%, P = 0.097) (Fig. 6B). The average size of islets in the AG group was significantly bigger than that in the Silicone group (AG: 32,578 ± 40,763 vs. GHNF: 30,979 ± 40,998 vs. Silicone: 12,921 ± 9,947 μm2, respectively, P = 0.047) (Fig. 6C).
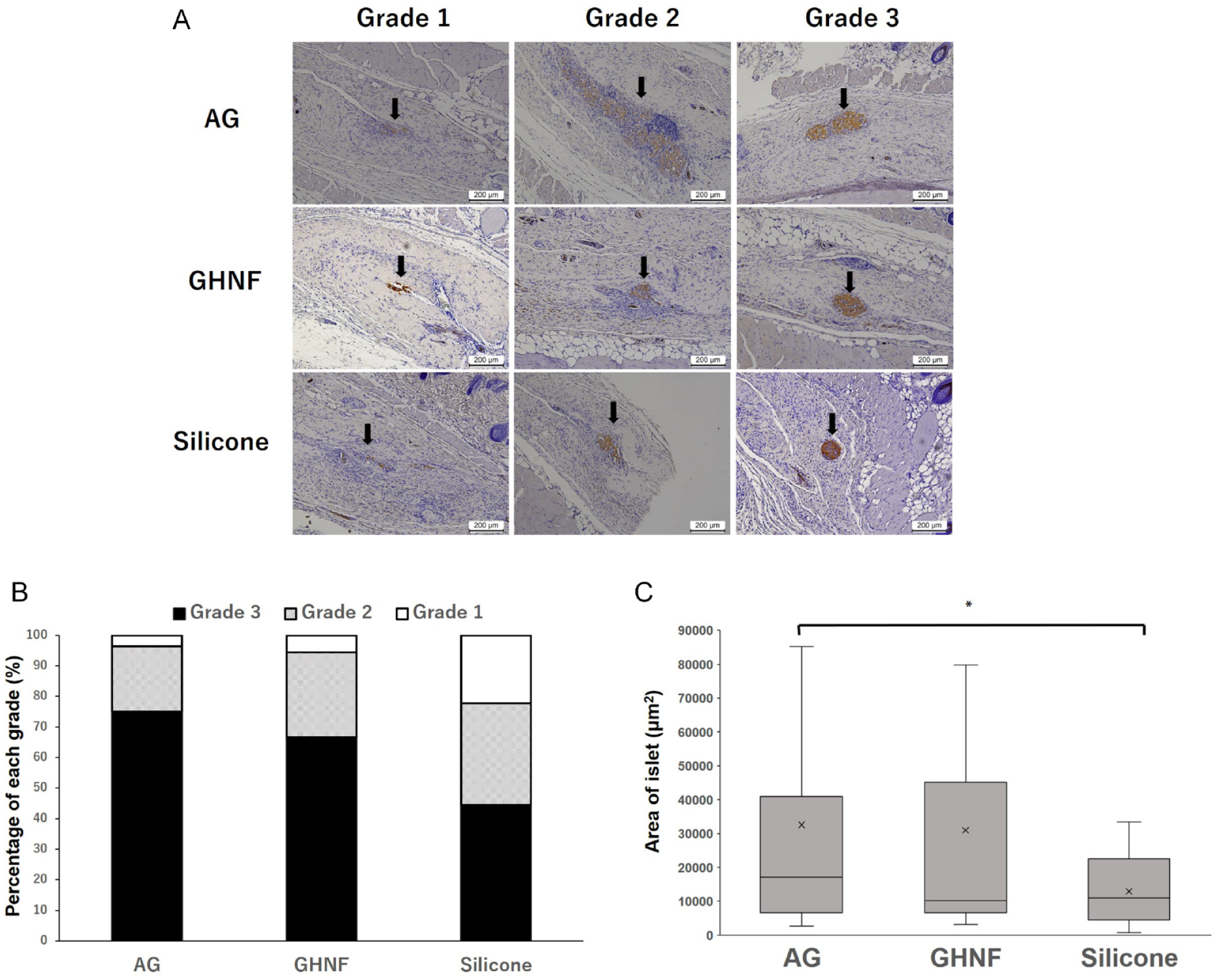
Immunohistochemical analyses of insulin-positive islets at 1 week after transplantation (A). Photomicrographs of islets. Magnification: ×100. Calibration bars: 200 µm. Insulin-positive islets were categorized to Grade 1 to 3 according to their shape and uniformity of insulin staining. (B) The percentage of Grade 2 and 3 islets in the AG and GHNF groups was higher than that in the Silicone group (AG: 96.4%, GHNF: 94.4%, Silicone: 77.7%, P = 0.097). (C) The average size of islets in the AG group was significantly bigger than that in the Silicone group (AG: 32,578 ± 40,763 vs. GHNF: 30,979 ± 40,998 vs. Silicone: 12,921 ± 9,947 μm2, respectively. *, P < 0.05).
In Vivo Tracing of GFP-Positive ADSCs Seeded on GHNF
GFP-positive ADSCs in the GHNF were counted before implantation (0 week) and at 1, 2, and 6 weeks after subcutaneous implantation of GHNF (Fig. 7A). The number of GFP-positive ADSCs at 0 or 1 week after implantation was significantly higher than that at 2 or 6 weeks after implantation (0 weeks, 248.5 ± 2.0; 1 week, 235.6 ± 50.2; 2 weeks, 136.9 ± 55.9; 6 weeks, 7.9 ± 6.2 number of cells in five fields). The number of GFP-positive ADSCs decreased in a time-dependent manner, and few cells were detected at 6 weeks after implantation (P < 0.001) (Fig. 7B).
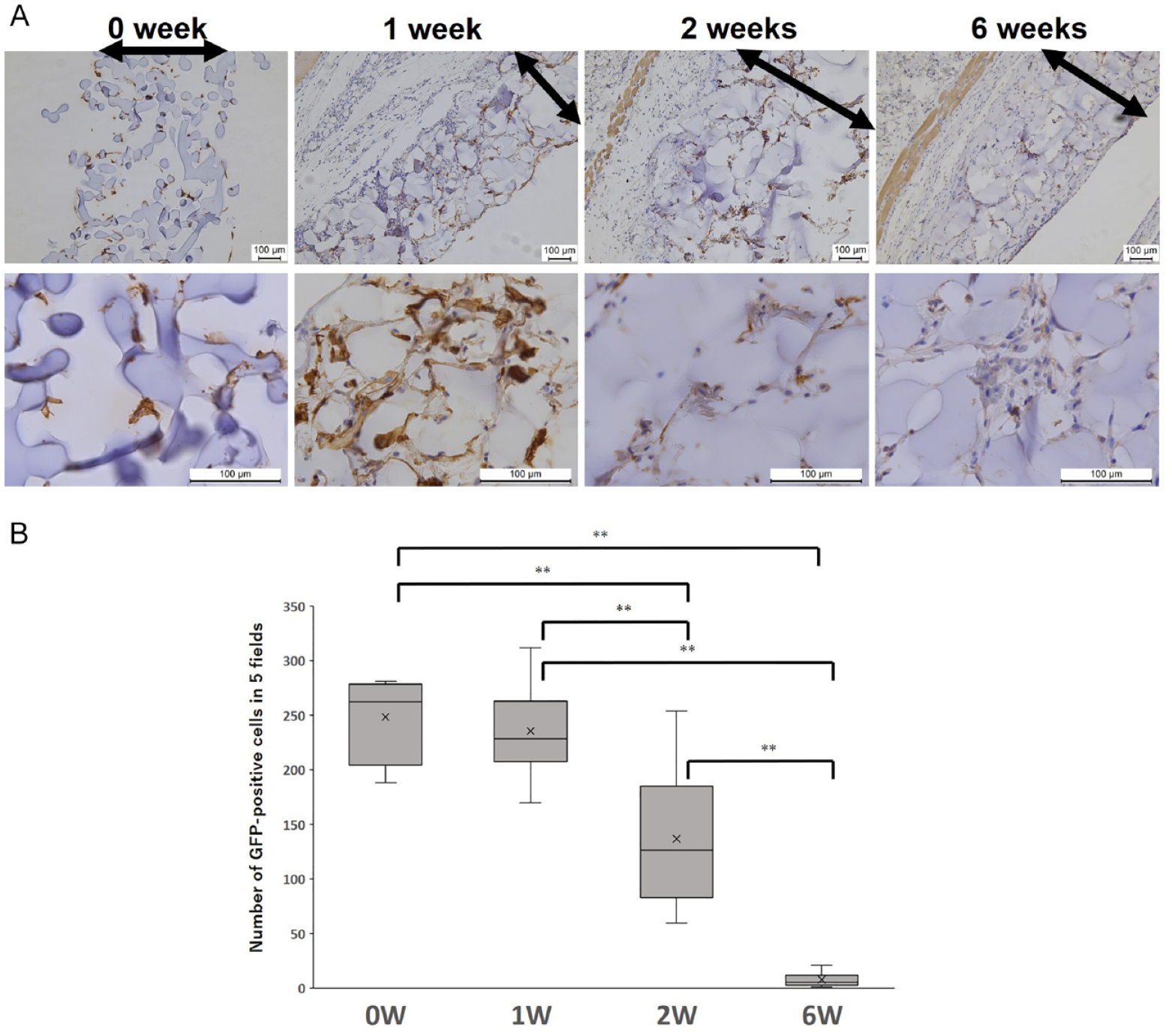
Immunohistochemical analyses of GFP-positive ADSCs. (A) Photomicrographs of GFP staining in the GHNF. Magnification: ×100 (upper row) and ×200 (lower row). Calibration bars: 100 µm. Black arrows represent the GHNF. (B) The mean number of GFP-positive ADSCs in the GHNF. The number of GFP-positive ADSCs at 0 or 1 week after implantation was significantly higher than that at 2 or 6 weeks after implantation (P < 0.01). The number of GFP-positive ADSCs decreased in a time-dependent manner, and only a limited number of them were detected at 6 weeks after implantation (**, P < 0.01).
Quantification of the Vascular Volume Using Lectin Angiography
To determine the density of blood vessels, the vascular volume (Fig. 8A, left) and capsular volume (Fig. 8A, right) were calculated in each group. Lectin angiography revealed that the blood vessel density in the AG group was significantly higher than that in the other groups (AG, 0.21 ± 0.23; GHNF, 0.10 ± 0.14; Silicone, 0.03 ± 0.02, AG vs. GHNF; P = 0.046, AG vs. Silicone; P = 0.009) (Fig. 8B).
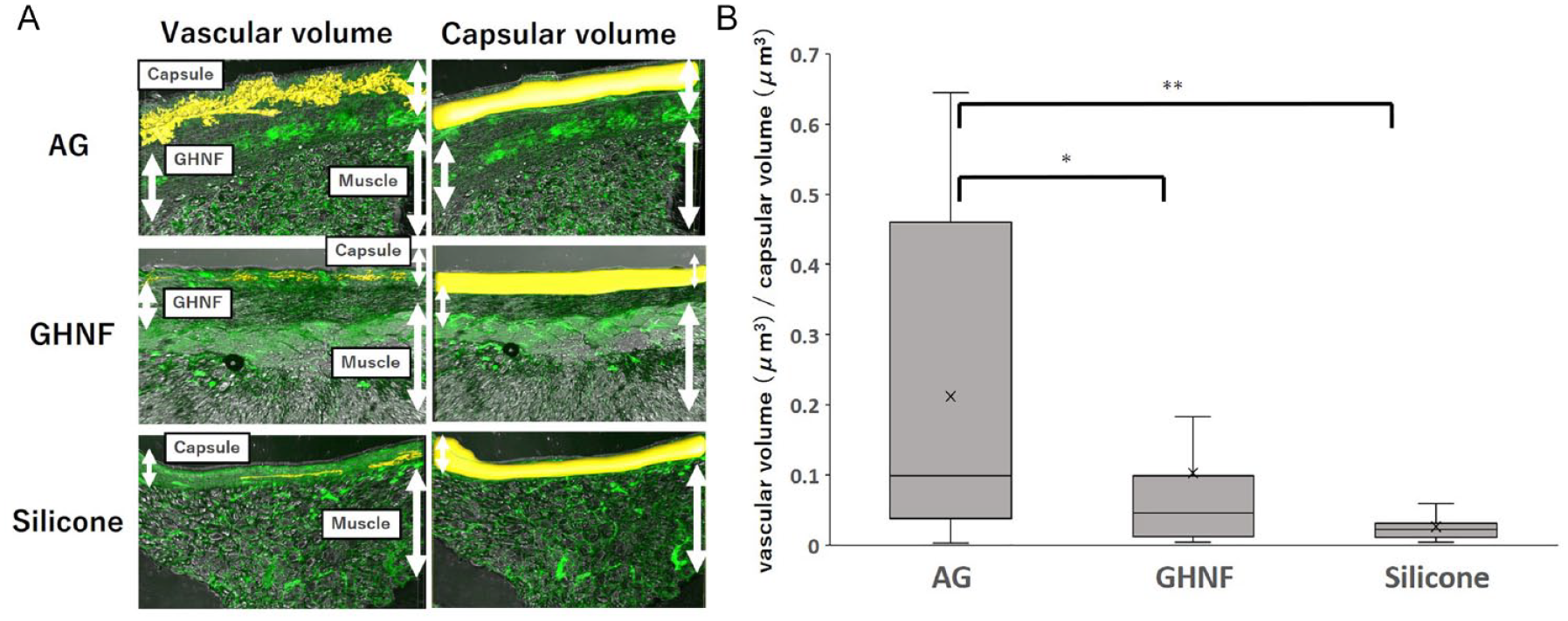
Quantification of the vascular volume using lectin angiography in the capsule at the transplant site of the AG, GHNF, and Silicone groups. (A) Image of lectin angiography. The yellow structure on the left side of the picture shows the vascular volume constructed in the capsule, and the yellow structure on the right side of the picture shows the total capsular volume. (B) Summary of vascular density among the groups. The results represent the vascular volume (μm3)/capsular volume (μm3), and the density of vessels in the AG group was significantly higher than that in the GHNF and Silicone groups (*, P < 0.05, **, P < 0.01).
Relative Gene Expression
To identify genes upregulated in subcutaneous capsules in the AG group, 46 target genes were analyzed using TaqMan arrays (Table 1). The relative gene expression in the AG group toward the GHNF group is shown in Fig. 9. The gene expression analyses revealed that 7 target genes, vascular cell adhesion molecule-1 [VCAM-1], FMS-like tyrosine kinase 1 [FLT1], heparan sulfate (glucosamine) [HS3ST3A1], vasohibin 2 [VASH2], vascular endothelial growth factor A [VEGFA], versican [VCAN], and platelet/endothelial cell adhesion molecule 1 [PECAM1], were significantly upregulated in comparison to the GHNF group (P < 0.05).
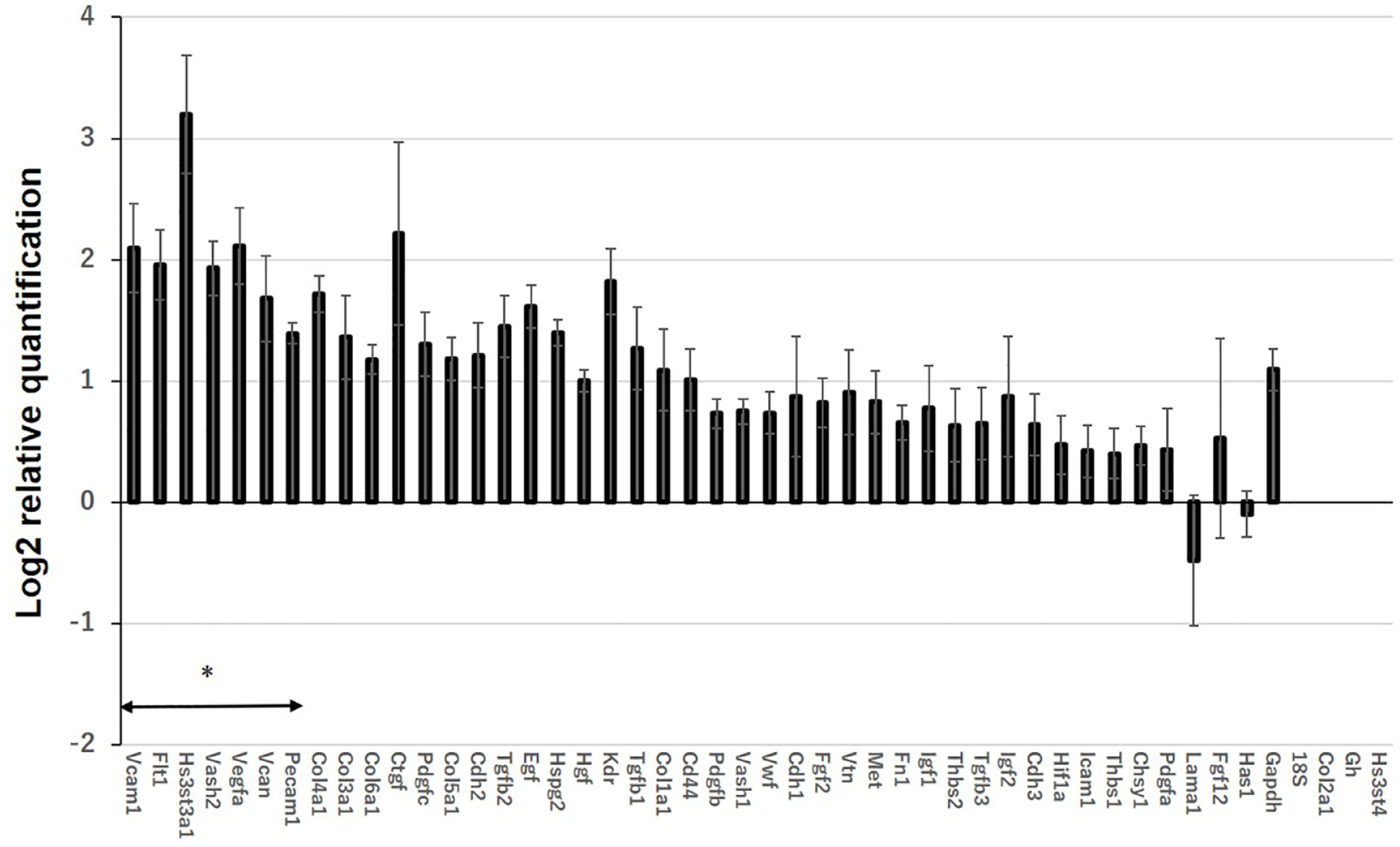
Upregulated and downregulated genes at subcutaneous capsules in the AG group (n = 5) in comparison to the GHNF group (n = 5). RNA was extracted from the recipient subcutaneous capsules surrounding the silicone spacer 6 weeks after pretreatment. Values represent the mean log2 relative quantification (RQ). Error bars represent the standard error on a log2 RQ-based scale. The +1 or −1 values represent a two-fold increase or decrease threshold in the gene expression. The gene expression analyses showed that seven target genes in the AG group were significantly upregulated in comparison to the GHNF group (*, P < 0.05).
The Protein Expression of Inflammatory Mediators and Islet-Protective Factors
To evaluate the inflammatory status in each group, 23 inflammatory mediators in the subcutaneous capsules were measured. Interestingly, the expression of proinflammatory mediators, including IL-1α, IL-1β, IL-6, granulocyte colony stimulating factor (G-CSF), interferon-γ (IFN-γ), and tumor necrosis factor-α (TNF-α), was highest in the Silicone group, and tended to be lower in the AG and GHNF groups (Fig. 10A–F). Regarding the islet-protective factors, the IGF-2 expression in the AG and GHNF groups was higher than that in the Silicone group (P = 0.347) (Fig. 10G).
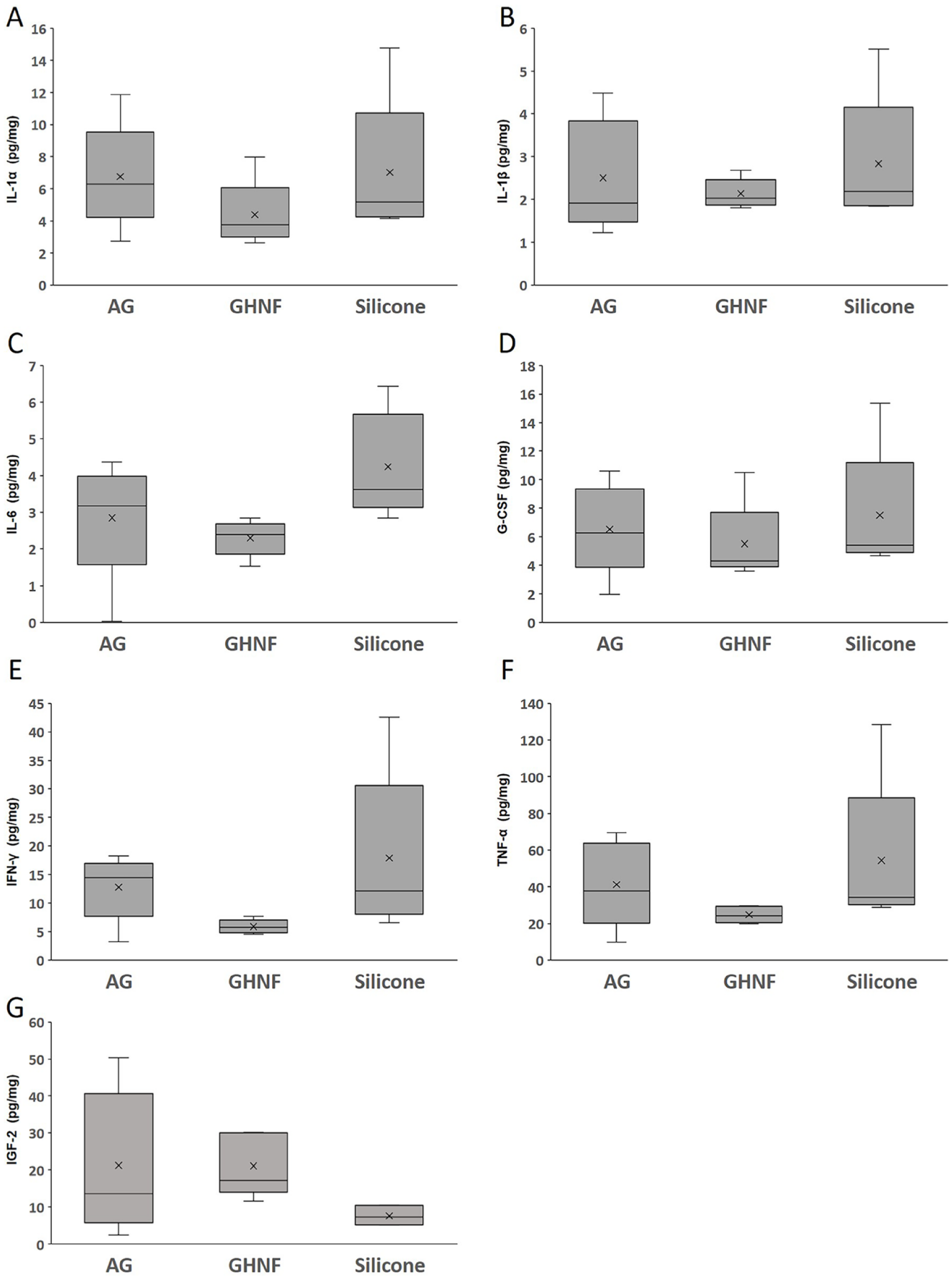
The concentration of inflammatory mediators and islet protective growth factor in the supernatant of the homogenized subcutaneous fibrous capsule surrounding the silicone spacer in each group (AG, n = 5; GHNF, n = 5; Silicone, n = 5). The concentrations of (A) interleukin-1α (IL-1α), (B) interleukin-1β (IL-1β), (C) interleukin-6 (IL-6), (D) granulocyte colony stimulating factor (G-CSF), (E) interferon-γ (IFN-γ), (F) tumor necrosis factor-α (TNF-α), and (G) insulin-like growth factor-2 (IGF-2).
Discussion
This study demonstrated that GHNF pretreatment together with ADSCs apparently enhanced neovascularization in the subcutaneous space and dramatically improved the outcomes of subcutaneous islet transplantation, although pretreatment with GHNF alone was also effective in comparison to the subcutaneous islet transplantation without pretreatment21,22. To our knowledge, this is the first report demonstrating that pretreatment using a bioabsorbable device combined with ADSCs synergistically induced sufficient neovascularization for islet engraftment and subsequently improved the outcome of subcutaneous islet transplantation. In fact, only 180 IEQs of islets (an extremely small number in comparison to previous studies) were transplanted and 72.7% [8/11] of diabetic mice in the AG group were cured (Fig. 2C). In comparison to the results of the IPO (the current standard procedure of islet transplantation) group, the blood glucose changes of the AG group were significantly better, and its cure rate was higher (AG, 72.7%; IPO, 40.0%) (Fig. 2B, D). Furthermore, the glucose tolerance of the AG group was better than that of the IPO group (Fig. 3B, D). These data clearly showed that subcutaneous islet transplantation using GHNF combined with ADSCs is more effective than intraportal islet transplantation, at least in a mouse model.
Although many studies have reported that co-transplantation of ADSCs and islets improved islet viability and transplant outcomes16,17, few studies have reported that ADSCs were seeded at the transplant site prior to islet transplantation as a preconditioning element to optimize the subcutaneous environment. In this context, Fumimoto et al 34 . previously reported that ADSCs were useful for preconditioning the subcutaneous environment for islet transplantation. In this study, they found that ADSC pretreatment combined with minced adipose tissues 4 weeks before islet transplantation improved (to some extent) the diabetic conditions of recipients, whereas ADSCs alone or adipose tissue alone did not work at all. In this report, unlike this study, ADSCs with or without minced adipose tissues were directly seeded into the subcutaneous space. Considering the clear differences in outcomes, minced adipose tissues might have worked as an effective scaffold for ADSCs. This suggests that ADSCs should be combined with ideal scaffolds when used as a preconditioning element for the subcutaneous transplant space. Corroborating this observation, Makarevich et al 35 . also reported that the direct injection of ADSCs alone into the subcutaneous site was ineffective for inducing neovascularization, subsequently resulting in poor outcomes in comparison to ADSC implantation together with scaffolds. Given that GHNF, which was shown to effectively proliferate cells 25 , was originally produced as a specific scaffold for mesenchymal stem cells (similar to ADSCs), the outstanding outcomes in this study make sense.
It was previously reported that most ADSCs implanted into the subcutaneous space disappeared within 3 weeks 36 . Likewise, in this study, GFP-positive ADSCs implanted subcutaneously halved at 2 weeks after implantation and were hardly observed at the time of islet transplantation (6 weeks after ADSCs implantation) (Fig. 7), suggesting that the beneficial effects of ADSCs were not due to direct cell-to-cell contact between ADSCs and islets, but ADSCs might have worked as preconditioning factors to optimize the subcutaneous environment.
The main drawback of subcutaneous islet transplantation is that there are few vessels that nourish transplanted islets 11 . It reportedly takes approximately 14 days for the construction of new vessels that support islet grafts and the vessel maturation is likely to take much longer time37,38. Therefore, it is crucial to create new vessels in the subcutaneous space in advance when islets are transplanted into the subcutaneous space. The number of vessels in the capsule around the silicone spacer significantly increased when GHNF was used for pretreatment and further increased when ADSCs were added onto the GHNF (Fig. 4B). Corroborating the immunohistochemical findings, the relative gene expression in the AG group also showed amplification of angiogenic genes, including VASH2 39 , VCAM-1 40 , and VEGFA 41 (Fig. 9). Interestingly, we observed a dramatical decrease of the blood glucose levels at 4 weeks after transplantation in the AG group (Fig. 2A). This finding may be explained by the hypothesis that it took approximately 4 weeks to have the mature vessels induced by ADSCs in this condition, which subsequently supported the islet survival and increased the insulin secretion. It was previously reported that the smallest visible vessels detectable by microcomputed tomography had a diameter of 50 μm42. Of particular note, lectin angiography can theoretically detect retinal vasculature of <10 μm in diameter 43 , subsequently enabling us to identify micro subcutaneous structures (Fig. 8A). In fact, in this study, lectin angiography revealed that the subcutaneous blood vessel volume in the GHNF group tended to be higher than that in the Silicone group, whereas CT showed no difference between the groups in our previous study 21 . One possible explanation for this discrepancy is that small vessels of <50 μm in diameter were not counted due to the poor resolution of CT in our previous study. The outcome trend of lectin angiography was consistent with that of vWF staining, which is regarded as the current standard procedure in the field of islet transplantation (Figs. 4B, 8B). Considering that lectin angiography can evaluate blood vessels three-dimensionally and does not require specific evaluation by pathologists, lectin angiography may be a useful alternative to conventional immunohistochemical staining for correctly evaluating vessels in the subcutaneous space.
Previous studies reported that ECM surrounding islets in the pancreas was lost in islet isolation 44 . Compensation of the ECM reportedly improved the islet function45,46. In this study, the immunohistochemical expression of collagen IV in the fibrous capsule surrounding the islet grafts in the GHNF group was unexpectedly lower than that in the other groups, but there was no difference in collagen III and laminin positivity among the three groups (Fig. 5). The reason for the lower collagen IV expression in the GHNF group remains unclear, but it may be concluded that synergistic effects of the AG group were at least not dependent on ECM compensation in this study.
The release of islet protective growth factors from the indwelling host cells would also have a crucial role in islet engraftment. Notably, IGF-2 protein expression in the subcutaneous fibrous capsule in the AG and GHNF groups tended to be higher than that in the Silicone group (Fig. 10G). Likewise, in our previous study, marked IGF-2 elevation in subcutaneous tissues was observed after GHNF pretreatment 21 . IGF-2 has been reported to maintain islet viability during the culture period and improve islet engraftment due to anti-apoptotic effects 47 . GHNF is also reported to attract host cells into the gaps of GHNF 19 . Furthermore, we recently reported that M2 macrophages, which have anti-inflammatory properties and promote islet engraftment 48 , accumulated among the gap in GHNF 22 . These findings may explain why the inflammatory mediators in the Silicone group were substantially higher than those in the AG and GHNF groups. Corroborating these observations, the percentage of islets with better grade in the AG and GHNF groups was higher than that in the Silicone group, and the islet size in the AG and GHNF groups was also twice as large as that in the Silicone group (Fig. 6).
Unexpectedly, inflammatory mediator expression levels, including IL-1α, IL-1β, IL-6, G-CSF, IFN-γ, and TNF-α, in the AG group were relatively higher in comparison to the GHNF group, although the inflammatory mediators of both groups tended to be suppressed in comparison to the Silicone group (Fig. 10A–F). It was previously reported that ADSCs were usually responsible for anti-inflammatory reactions 49 . However, under inflammatory or metabolic syndrome settings, including diabetes, adipocytes actively secrete proinflammatory cytokines, and ADSCs contribute even more to enhancing inflammatory loops, subsequently switching their behavior from anti-inflammatory to proinflammatory status50,51. This could clearly explain our unexpected observations. Considering that ADSCs may—to some extent—induce inflammation at the transplant site, beneficial effects of ADSCs, mainly based on angiogenesis, could have a strong impact on islet engraftment. From another perspective, this finding may suggest that a substance which specifically targets angiogenesis without inducing inflammation, such as an angiogenic peptide combined with GHNF, could potentially further improve the outcomes of subcutaneous islet transplantation. Therefore, this is a topic of interest for our next study.
Since the ADSC function may depend on the density of ADSCs at the transplant site, the number of ADSCs added onto the GHNF should be optimized according to the size of the GHNF when applied in large animal models, including clinical trials for humans. Further studies using large animal models are warranted as next steps.
In conclusion, this study revealed that pretreatment using bioabsorbable scaffolds (GHNFs) combined with ADSCs enhanced neovascularization in the subcutaneous space and improved the outcomes of subcutaneous islet transplantation in comparison to intraportal islet transplantation. This beneficial effect may be mainly due to neovascularization at the transplantation site and protection of islet viability by various growth factors, rather than compensation by the ECM.
Footnotes
Acknowledgements
The authors thank Kozue Maya and Megumi Goto (Division of Transplantation and Regenerative Medicine, Tohoku University) for their excellent technical assistance. The authors also acknowledge the support of the Biomedical Research Core of Tohoku University, Graduate School of Medicine and TAMRIC (Tohoku Advanced Medical Research and Incubation Center).
Author Contributions
RS and MG participated in the research design, performance of the research and writing of the paper. AI participated in the performance of the research and the writing of the paper.
YN participated in pathological analysis in the research. TI, NK, HM, YEK, TKat, SS, and KT participated in the performance of the research. TKam, MU, and KW participated in the writing of the paper. YT participated in technological advice on gelatin hydrogel nonwoven fabrics.
Availability of data and material
All data generated or analyzed in this study were included in this published manuscript.
Ethical Approval
All experimental protocols used in this study (protocol ID:2020 MdA-022) were approved by the Animal Experimental Committee of Tohoku University. All surgical operations were performed under anesthesia, and efforts were made to minimize suffering.
Statement of Human and Animal Rights
All animals used in this study were handled in accordance with the Guide for the Care and Use of Laboratory Animals, published by the National Institutes of Health.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare no conflicts of interest in association with the present study, although this study was performed according to the patent application agreement with KYOTO MEDICAL PLANNING Co., Ltd.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Japanese Grant-in-Aid for Scientific Research (A) (grant no. 18H04056) and Research (B) (grant no. 22H03133) from the Japan Society for the Promotion of Science, and by AMED under grant no. JP19bm0404043. The funders played no role in the study design, the collection and analysis of the data, the decision to publish, or the preparation of the manuscript.
