Abstract
This study examines extracellular matrix (ECM) protein (ECM) expression in chronic pancreatitis (CP) patients and its correlation with graft function after total pancreatectomy with islet autologous transplantation (TPIAT). Pancreatic sections from 29 CP patients undergoing TPIAT were analyzed for ECM including pan-laminin, laminin alpha 5 (LAMA5), collagen IV, and Perlecan by immunohistochemistry and scored by the percentage positive staining area within the whole tissue area. Graft function was monitored by blood glucose and C-peptide levels. Laminin alpha 5 levels in blood plasma were greater in CP. Laminin alpha 5 expression was significantly higher in all CP patient etiological categories including alcoholic, hereditary, idiopathic, Oddi dysfunction, and pancreatic divisum compared to healthy controls. The overall expression of LAMA5 positively correlated with expression of the ECM proteins pan-laminin (R = 0.63, P < 0.001), collagen IV (R = 0.67, P < 0.001), and Perlecan (R = 0.71, P < 0.001). Increased LAMA5 expression was observed within islet endothelial vascular tissue and the peri-islet basal membrane. Increased LAMA5 expression in the pancreas correlated with poor islet isolation yield and posttransplant islet function after 3 months. Increased endothelial expression of LAMA5 and ECM proteins is indicative of progressive damage to the pancreas and correlates with poor graft function after TPIAT.

Introduction
Pancreatitis is characterized by inflammation and injury to the pancreas associated with pain in the upper abdominal area that may radiate to the back. Pancreatitis can be acute or chronic, and both circumstances cause serious health complications, poor quality of life, and a shorter life expectancy1–3. Approximately 275,000 people are hospitalized with acute pancreatitis in the United States each year. Acute pancreatitis occurs suddenly and may last for several days, but many patients recover within a few days of treatment. In more severe cases, patients with nausea, vomiting, fever, and a swollen or tender abdomen may require longer hospitalization. Recurrent acute pancreatitis can develop into chronic pancreatitis (CP) with permanent pancreatic damage. As time progresses, patients report worsening symptoms and eventually may experience hyperglycemic or hypoglycemic episodes due to pancreatic endocrine insufficiency.
In CP patients who do not improve with medication or surgical intervention, total pancreatectomy with islet autologous transplantation (TPIAT) is required. In this complex procedure, the pancreas undergoes chemical digestion by collagenase enzyme and mechanical digestion in a closed chamber under constant monitoring. A delicate balance of temperature, enzyme concentration, and time can produce a single cell suspension of exocrine acinar separate from endocrine islets that remain intact. This suspension of purified islets is infused into the portal vein of the liver, as this is a suitable environment for islet revascularization and signaling. Multiple factors contribute to islet graft function, and thus TPIAT success depends on the pretransplant pancreatic condition, isolation methods, and the posttransplant environment. Here, we were interested in the pancreas condition pre-TPIAT.
During CP, changes in basement membrane components of the extracellular matrix (ECM) lead to fibrosis of the pancreatic exocrine tissue 4 . Fibrous components of the ECM include α-smooth muscle actin (SMA), elastins, fibronectins, laminins, and collagen. Laminins are essential to the formation, stability, and repair of the basement membrane 5 and are necessary to support cell differentiation, proliferation, migration, and survival through specific interactions with integrins, dystroglycans, and other receptors6,7. Laminin subunit alpha 5 (LAMA5) is a key component of the pancreatic ECM, and in vitro studies have shown its role in support of beta cell survival, cell signaling regulation, insulin secretion, and stem cell renewal or differentiation 8 .
Laminin expression is regulated in different ways by numerous signals, including growth factors, cytokines, and proteases. In studies of pancreatic tumor tissue, there is a reported increase in laminin expression that diminishes with progressive dedifferentiation of the tumor9,10. Similar laminin expression patterns have been observed in colorectal cancer 11 and breast cancer 12 . In vitro studies have shown that LAMA5 expression is increased in endothelial cells after stress treatments including lipopolysaccharide, interleukin-1β, and tumor necrosis factor-α 13 . In CP, we hypothesize that LAMA5 production increases over time in an attempt to repair parenchymal damage caused by increased exocrine enzyme secretions. Furthermore, we wanted to analyze whether LAMA5 expression in CP patients could predict TPIAT outcomes. In this study, we explored the change in LAMA5 expression due to CP and evaluated whether there are correlations with islet function after TPIAT.
Materials and Methods
Human Subjects and Ethical Concerns
The study protocol was approved by the Institutional Review Board at Baylor Scott & White Research Institute, Dallas. TPIAT was indicated in patients with unmanageable abdominal pain, chronic narcotic dependence, and poor quality of life. A detailed patient history was obtained before proceeding to TPIAT. Potential outcomes and complications during and after surgery were explained to the patients and their legal guardians. Pancreas samples from non-CP patients were defined as healthy and were obtained through Southwest Transplant Alliance. Healthy donor samples were an average age of 55 ± 5.7 years, average body mass index (BMI) of 28.9 ± 5.4 kg/m2, and cause of death was stroke.
Study Design
In this retrospective cohort study, human pancreatic tissue samples from 29 CP patients who underwent TPIAT at Baylor University Medical Center, Dallas, Texas, were obtained from the resected pancreas head. Pretransplant assessments including age, gender, BMI, duration of symptoms, fasting blood glucose, hemoglobin A1c (HbA1c), glucose-stimulated C-peptide, and previous surgical interventions were recorded. Patients’ first follow-up is minimum 10 days and ideally at 1 month posttransplant for clinical data collection including body weight, c-peptide, blood glucose, HbA1c, diabetes treatment and insulin dosage, pain score and narcotics dosage. Consecutive follow-up occurred on month 3.
Human Pancreatic Islet Isolation and Tissue Sample Collection
CP patients’ baseline characteristics were documented and using previously reported techniques14,15 islet isolation was carried out in our center’s Islet Cell Processing Laboratory, which complies with Current Good Tissue Practice. Briefly, islets were isolated using the modified Ricordi method. If the tissue volume exceeded 20 mL, the islets were purified with a COBE 2991 cell processor (CaridianBCT) and an iodixanol-based continuous density gradient. The final islet tissue preparation was suspended in 200 mL of CMRL 1066 culture medium (Mediatech, Inc) per infusion bag, with a final concentration of 2.5% human serum albumin, 25 mM HEPES (4-[2-hydroxyethyl]-1-piperazineethanesulfonic acid), and 70 U/kg patient body weight heparin. TPIAT samples were collected in optical coherence tomography (OCT) compound molds (Fisher Healthcare) and stored at −80°C.
Tissue Sample Processing
The tissue samples were retrieved, and 5 µm sections were obtained using a cryostat microtome (Leica CM 3050S). The pancreatic sections were deparaffinized and rehydrated and analyzed for ECM including P-laminin, LAMA5, collagen IV, and perlecan using a standard immunofluorescence procedure16,17.
Immunofluorescence Staining
Tissue sections were immunofluorescent tagged to determine the location of LAMA5, pan-laminin, insulin, glucose, c-kit, CD31, collagen IV, and perlecan. Antibodies were prepared in 0.01% Triton X-100 in phosphate-buffered saline (PBS) at dilutions specified by the manufacturer. Sections were first incubated in blocking buffer (PBS + 5% bovine serum albumin + 0.1% Triton X 100) for 30 minutes at room temperature. Tissue was then incubated overnight at 4°C in primary antibody and for 2 hours at room temperature in secondary antibody. Tissue was washed three times in washing buffer (PBS + 0.1% bovine serum albumin + 0.1% Triton X 100) between each step. Vectashield with 4′,6-diamidino-2-phenylindole (DAPI) was used to mount the cover glass. The tissue sections were assessed by ZEISS Axio Imager 2 microscope (Zeiss, Jena, Germany) and scored by the percentage of the positive staining area within the whole tissue section using ImageJ software.
Quantification of Basement Membrane Proteins and Other Parameters
To assess the correlation between the CP patients’ islet outcomes and LAMA5 expression, plasma samples of TPIAT patients were quantified for LAMA5. LAMA5 expression was measured in plasma samples using a human laminin alpha 5 enzyme-linked immunosorbent assay kit (LifeSpan BioSciences, Inc.).
Statistical Analysis
Statistical significance was determined by one-way analysis of variance using the Pearson method performed using R package and GraphPad Prism. Differences were considered significant when P values were less than 0.05.
Results
Human Subjects’ Epidemiological and Basic Information
The characteristics of CP patients analyzed for this study are outlined in Table 1. Of the 29 patients, 12 were men and 17 were women. The BMI range of the patients was 25 to 33 kg/m2. Patients with pancreatic divisum or hereditary etiology had the longest reported symptoms, ranging from 8 to 9 years, while patients with CP of other etiologies had symptoms for 2 to 6 years. C-peptide levels were within a normal healthy range (1.0–2.8 ng/mL) and basal glucose ranged from 92 to 114 mg/dL which is considered prediabetic by the American Diabetes Association standard of care. The pancreas weight after final trimming during the islet isolation procedure was between 81 and 90 g. The islet yield was higher in patients with a greater BMI. HbA1c levels were less than 6% and were considered normal.
Patients’ Demographic and Disease Characteristics.

Values reported as mean ± standard deviation.
The formula of SUITO index is fasting C-peptide (ng/mL)/(fasting blood glucose – 63 [mg/dL]) × 1,500.
Quantification of Laminin Alpha 5 in Patient Plasma
On the day of islet infusion (Day 0), circulating levels of LAMA5 in plasma were notably greater in patients receiving an islet yield of less than 5000 IEQ/g of the pancreas compared to patients receiving an islet yield greater than 5000 IEQ/g of the pancreas (Fig. 1). Compared to both TPIAT recipient groups, the concentration of LAMA5 was lower in healthy control patients (Fig. 1). These results indicate that LAMA5 levels found in blood plasma are greater in CP patients and even more so in patients in which a suboptimal islet quantity was retrieved and retransplanted, possibly due to a more diseased pancreas.

Laminin alpha 5 (LAMA5) present in the plasma of patients undergoing total pancreatectomy with islet autologous transplantation (TPIAT) and normal patients on the day of transplant and on posttransplant days 1, 30, and 90 in TPIAT patients. TPIAT patients are compared by the islet dosage received in transplant. Student’s t test was calculated for statistical significance. ****p < 0.0001.
Laminin Alpha 5 Staining in Tissues of Patients With CP
LAMA5 expression in pancreases used for this study was analyzed by immunofluorescent staining of pancreas sections. We were interested in the pretransplant LAMA5 expression in pancreases of different CP etiologies. Frozen pancreatic tissue sections were stained by LAMA5, pan-laminin, and DAPI. The percentage of the LAMA5-positive area per total section was calculated and compared by CP etiology. LAMA5 staining was greatest in CP pancreatic tissue of autoimmune and Oddi dysfunction followed by idiopathic, hereditary, and alcoholic CP conditions (Fig. 2). All sections with CP had a greater area of LAMA5 expression than pancreases from normal organ donors (healthy controls) and ranged from 10% to 25% area expression.

Immunofluorescent imaging of laminin alpha 5 (LAMA5) expression in the pancreatic tissue of patients with chronic pancreatitis undergoing total pancreatectomy with islet autologous transplantation. Merged image of DAPI/LAMA5/P.LAM at (A) 10× and (B) 20× magnification. (C) LAMA5-positive area (%) comparison by etiology of chronic pancreatitis.
High expression of LAMA5 was observed in areas surrounding exocrine and endocrine cells as well as in intra-islet substructures. LAMA5 was expressed in islet basement membranes that formed intra-islet substructures between alpha cells and beta cells (Fig. 3). This expression was similar to c-kit–positive hematopoietic stem cells and CD31-positive endothelial cells (Fig. 4). These data show LAMA5-positive substructures contribution to an intra-islet microenvironment that is necessary to provide nutrients and support the regeneration of islets.

Intra-islet laminin alpha 5 (LAMA5) excludes alpha and beta cells: (A) Pancreatic tissue section stained with DAPI. (B) Positive staining area of LAMA5. (C) Positive staining area of insulin within the islets. (D) Glucagon-positive staining area in the islets. (E) Merged image of LAMA5 and insulin stains. (F) Merged image of LAMA5 and glucagon stains. (G) Merged image of DAPI, LAMA5, insulin, and glucagon stains.

Intra-islet laminin alpha 5 (LAMA5) related to c-kit–positive hematopoietic stem cells and CD31-positive endothelial cells. First row: 10× magnification of representative islet: (A) DAPI, (B) LAMA5, (C) c-kit, (D) merged image of LAMA5 and c-kit. Second row: 20× magnification of representative islet: (E) DAPI, (F) LAMA5, (G) CD31, (H) merged image of LAMA5 and CD31.
Correlation of Laminin Alpha 5 and ECM Protein Expression
In CP pancreatic tissue, LAMA5 area expression positively correlated with the expression area of other pancreatic basement membrane proteins. LAMA5 and pan-laminin were co-expressed in some areas of the tissue, and their expression was positively correlated (p = 0.00005). The LAMA5-positive area was also positively correlated with the collagen IV-positive area (p = 0.00001) and perlecan-positive area (p = 0.0039) of the pancreas (Fig. 5). In CP pancreatic tissue, positive staining of multiple ECM proteins increased similar to LAMA5 protein.

Laminin alpha 5 (LAMA5) expression correlates with the expression of key extracellular matrix proteins such as P-laminin, collagen IV, and perlecan. (A) Merged and individual stains of DAPI (blue), LAMA5 (green), and pan-laminin (red) in pancreatic tissue. (B) LAMA5 and pan-laminin percentage of positive area. (C) Positive stained area of collagen IV merged with DAPI. (D) Correlation between positive staining area as determined by scatterplot documentation of multiple sections. (E) Positive stained area of perlecan. (F) LAMA5 and perlecan percentage of positive area.
Laminin Alpha 5 Expression and Patient Outcomes
TPIAT patients returned for follow-up laboratory tests around day 10 and then again at month 3 after islet infusion. Laboratory analysis included fasting blood sugar and exogenous insulin usage, and at 3 months HbA1c levels were recorded. LAMA5 area expression was positively correlated with fasting blood glucose levels (p = 0.0018) and HbA1c (p = 0.0039) (Fig. 6A, B). There was a negative correlation between LAMA5 area and IEQ/mL tissue infused (p = 0.02) (Fig. 6C). There was no correlation between LAMA5 area expression and insulin dosage posttransplant (Fig. 6D). The results indicate that greater area expression of LAMA5 correlates with poor islet isolation and posttransplant islet function.
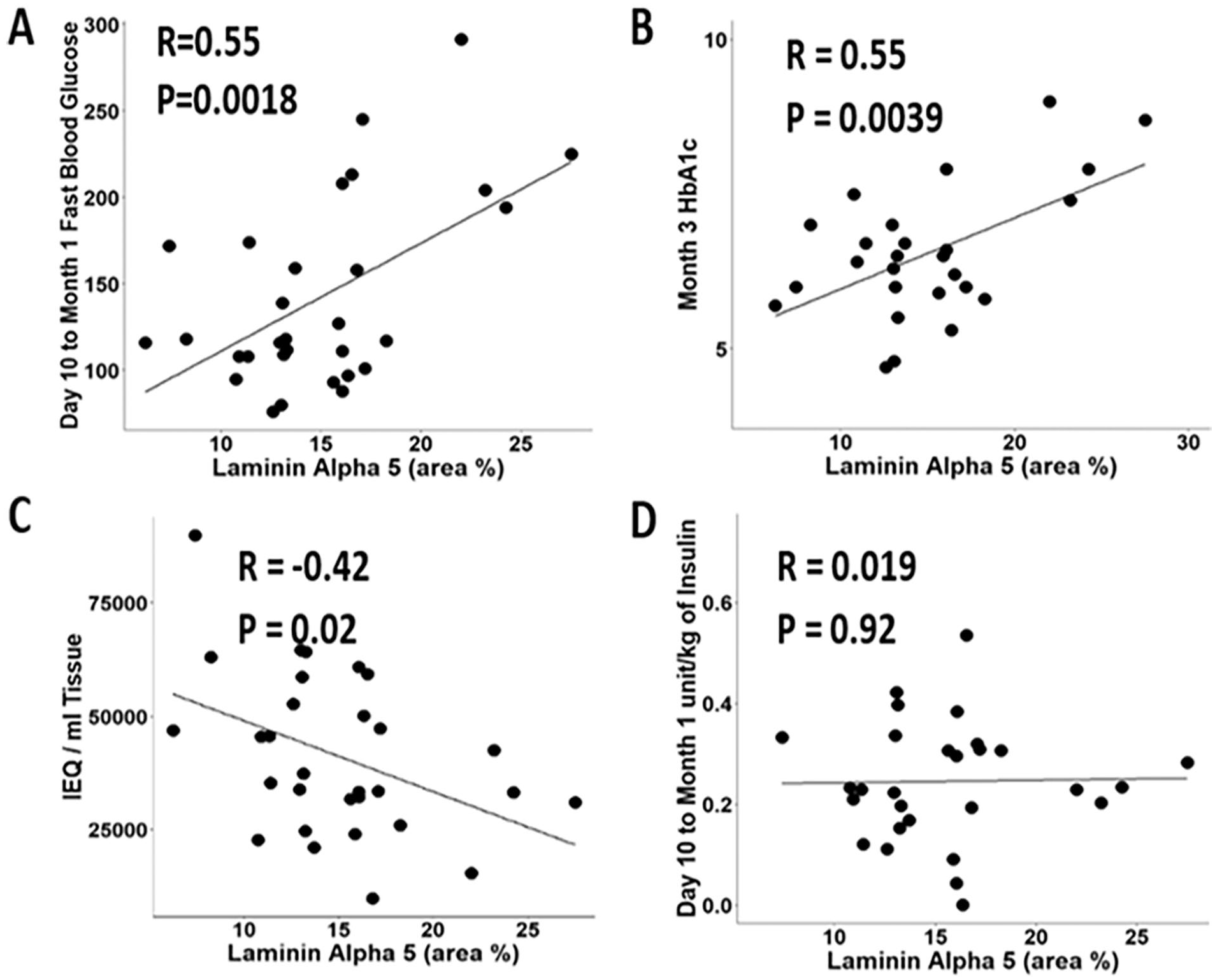
High expression of laminin alpha 5 (LAMA5) does not support islet function after the TPIAT procedure. Graphs show the correlation between the LAMA5-positive area percentage in the tissue sample and (A) fasting blood glucose measurements taken 10–31 days posttransplant; (B) Hemoglobin A1c at 3 months posttransplant; (C) tissue acquired (IEQ/mL); and (D) Insulin (unit/kg) dosage administered 10–31 days posttransplant.
Correlation of Laminin Alpha 5 and Extracellular Matrix Proteins With Clinical Outcomes
To determine the correlation between other pancreatic tissue ECM protein expression and islet autotransplant function, a heatmap correlation analysis was performed with clinical parameters documented in patient follow-up (Fig. 7). Of significance, CA19-9 expression negatively correlated with poor isolation outcomes. Two cell types that contribute to fibrogenesis and inflammation are pancreatic stellate cells and collagen, which are identified by alpha-SMA, and picosirius red (PSR), respectively. Interestingly, SMA and PSR saw opposite results and correlated with poor isolation outcomes. Further correlation analysis demonstrated that LAMA5 expression had a negative correlation with total IEQ infused into the portal vein. These results indicate that high LAMA5 expression correlates with poor islet graft function in TPIAT.

Heatmap correlation analysis of laminin alpha 5 (LAMA5) and other pancreatic proteins to clinical variables of islet autotransplantation patients.
Discussion
While islet transplantation is an effective procedure to maintain glycemic control, reduce pain, and increase quality of life, hurdles remain to increase the function of the islet graft posttransplant. To improve the success of the islet graft, efforts are ongoing to determine the best timing for TPIAT intervention, changes to the isolation procedure to reduce stress, and posttransplant treatment plans to reduce inflammation and its signaling cascade 18 .
In this study, we asked whether a better understanding of the pretransplant ECM status in the pancreas could predict TPIAT outcomes. Previous studies have determined that the basement membrane of the ECM provides structural support, divides tissues into discrete compartments, and regulates cellular function19,20. Islets are characterized by a double basement membrane: vascular and peri-islet. Unlike the exocrine compartment of the pancreas and most other epithelial cell types, endocrine islet cells cannot form their own vasculature 21 .
Islet integrity is maintained by surrounding ECM proteins in the pancreas. The vascular basement membrane ensures that islets are in contact with blood vessels22,23. The peri-islet basement membrane protects the islets from immune cell infiltration and separates the endocrine islet cells from the exocrine compartment of the pancreas 24 . We hypothesized that ECM molecules help regulate islet graft function and survival post-TPIAT. In fact, some groups have reported that supplementation with ECM molecules enhances graft function and survival 25 . Here we explored the role of ECM proteins, specifically LAMA5, and their contribution to islet homeostasis during the progression of CP.
Circulating LAMA5 levels were higher in the plasma of patients in which less than 5000 IEQ/g pancreas was isolated, indicating a factor of suboptimal isolation outcomes. The exacerbated laminin expression in CP tissue, and heightened expression in extreme CP cases, may be explained by increased pancreatic stress signaling. We hypothesize that basement membrane laminin protein is generated to compensate for damage and repair processes in an attempt to salvage islet cell function, and it accumulates during the progression of CP. While this is a possibility, soluble LAMA5 levels may be resultant of multiple organs, and we cannot conclude these levels are only resultant of the pancreas.
Further investigation indicated that expression of other ECM proteins such as pan-laminin, collagen IV, and perlecan were positively correlated with LAMA5 expression in CP patients. The pancreatic ECM proteins are helpful for survival and maintain the physiological functions of human islets 26 . Proteomics has been widely employed in translational research to investigate pathophysiology, identify biomarkers, and screen drug targets 27 . The ECM proteomic profile of human pancreas contains a wide range of proteins such as Collagens (type I, III, IV, V, VI), Laminins, Fibronectin, Proteoglycans (Heparan sulfate proteoglycans, Decorin, Biglycan), and other glycoproteins (Tenascin-C, Nidogen, Osteopontin) 28 . The in-depth genomic and proteomic analysis of ECM profiles is necessary to understand the expression profile of pancreatic enzymatic precursors, stress response proteins, and inflammatory mediators in CP patients when compared with normal human pancreatic tissue.
We employed multiple established techniques including analysis of plasma or serum, histopathology/immunohistochemistry, or immunofluorescence of tissue samples. Developing technologies show even greater potential for in depth analysis of ECM profiles to build upon what we know. These include single-cell RNA sequencing (Sc-RNA seq) and single-nucleus RNA sequencing (Sn-RNA-seq), proteomic profiling by liquid chromatography-mass spectrometry (LC-MS/MS), isotope-coded affinity tags (iTRAQ), and matrisome analysis by using tools like matrisome analyzer R and matrisome DB29–33. This type of investigation will contribute to the molecular mechanisms driving CP and other illness progression and prognosis.
LAMA5 has also been reported to promote angiogenesis in colon cancer due to induction by inflammation. It is a possibility that the inflammatory environment of CP could trigger greater LAMA5 promotion for islet vascularization 34 . Perlecan plays an important role in angiogenesis, tissue development, and stabilization of the ECM within tissues and contributes to the mechanosensory properties through interactions with fibrillin, collagen, and elastin 35 . Overall, ECM proteins are primarily produced to support islet survival and function. One explanation for poor TPIAT outcomes associated with greater ECM protein expression may be the additional strain on the islets that develops with an overproduction of the ECM proteins and the consequent fibrosis that compromises islet function and yield.
Interestingly, when the TPIAT outcomes were correlated with individual ECM proteins, there were mixed results, as depicted in the heatmap analysis. Another explanation for poor TPIAT outcomes may be generation of LAMA5 in an attempt to salvage islets, which is ineffective in combination with other ECM protein generation. Future testing is required to determine the effects of individual ECM protein accumulation on islet function. Additional limitations of this study include a small number of patient samples which limits the statistical analysis power. Furthermore, long-term follow-up of patients did not occur past 1 year and therefore needs to be improved.
Conclusion
Patients with CP have high levels of LAMA5 expression in plasma and pancreatic tissue compared to normal cohorts. LAMA5 forms intra-islet substructures that relate to endothelial cells and c-kit–positive hematopoietic stem cell expression, possibly supporting beta cell regeneration in the pancreas in response to CP stress. Highly expressed LAMA5 does not produce successful islet grafts in the TPIAT procedure, possibly due to the aggregation of ECM proteins in disease conditions. Although our data introduce LAMA5 as a marker of poor TPIAT outcomes, with further testing, it has the potential to be a predictive marker to determine ideal TPIAT interventions.
Footnotes
Acknowledgements
The authors acknowledge the Baylor Scott & White Research Institute imaging core members for their technical support in immunofluorescent analysis.
Author Contributions
J.K.: Writing—original draft, Investigation, Formal analysis, Data curation, Conceptualization. C.M.D.: Formal analysis. Y.L.: Methodology, Investigation, Data curation, Conceptualization. M.L.: Review & editing, Conceptualization. J.S.D.: Review & editing, Conceptualization. B.N.: Review & editing, Supervision, Project administration, Formal analysis, conceptualization.
Ethical Approval
Ethical approval to conduct and report this study was provided by the Baylor Scott & White Research Institute Institutional Review Board (IRB 010-237).
Statement of Human and Animal Rights
All procedures reported in this article were conducted in accordance with approved protocols of the Baylor Scott & White Research Institute Institutional Review Board.
Statement of Informed Consent
All patients enrolled in this study provided written informed consent for the publication of data without disclosing their personal identification information.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support from Baylor University Medical Center is gratefully acknowledged.
