Abstract
Circulating microRNAs (miRNAs) can be biomarkers for diagnosis and progression of several pathophysiological conditions. In a cohort undergoing total pancreatectomy with islet autotransplantation (TPIAT) from the multicenter Prospective Observational Study of TPIAT (POST), we investigated associations between a panel of circulating miRNAs (hsa-miR-375, hsa-miR-29b-3p, hsa-miR-148a-3p, hsa-miR-216a-5p, hsa-miR-320d, hsa-miR-200c, hsa-miR-125b, hsa-miR-7-5p, hsa-miR-221-3p, hsa-miR-122-5p) and patient, disease and islet-isolation characteristics. Plasma samples (n = 139) were collected before TPIAT and miRNA levels were measured by RTPCR. Disease duration, prior surgery, and pre-surgical diabetes were not associated with circulating miRNAs. Levels of hsa-miR-29b-3p (P = 0.03), hsa-miR-148a-3p (P = 0.04) and hsa-miR-221-3p (P = 0.01) were lower in those with genetic risk factors. Levels of hsa-miR-148a-3p (P = 0.04) and hsa-miR-7-5p (P = 0.04) were elevated in toxic/metabolic disease. Participants with exocrine insufficiency had lower hsa-miR-29b-3p, hsa-miR-148a-3p, hsa-miR-320d, hsa-miR-221-3p (P < 0.01) and hsa-miR-375, hsa-miR-200c-3p, and hsa-miR-125b-5p (P < 0.05). Four miRNAs were associated with fasting C-peptide before TPIAT (hsa-miR-29b-3p, r = 0.18; hsa-miR-148a-3p, r = 0.21; hsa-miR-320d, r = 0.19; and hsa-miR-221-3p, r = 0.21; all P < 0.05), while hsa-miR-29b-3p was inversely associated with post-isolation islet equivalents/kg and islet number/kg (r = −0.20, P = 0.02). Also, hsa-miR-200c (r = 0.18, P = 0.03) and hsa-miR-221-3p (r = 0.19, P = 0.03) were associated with islet graft tissue volume. Further investigation is needed to determine the predictive potential of these miRNAs for assessing islet autotransplant outcomes.
Introduction
Total pancreatectomy with islet autotransplantation (TPIAT) is performed to treat chronic pancreatitis (CP) and recurrent acute pancreatitis (RAP), characterized by intractable pain, progressive inflammation and fibrosis of the pancreas 1 –3 . Eligible patients undergo TPIAT only after other interventions including medical management, endoscopic therapies or other surgical procedures have failed to alleviate pain and improve quality of life, and following a multidisciplinary evaluation 2,4 –6 . Although increasingly offered at multiple institutions with varied success, there is no consensus on timing of surgery, criteria for patient selection, or definition of associated functional measures 1,2 . Optimal islet yield from an inflamed pancreas is an important factor determining islet graft function post-transplantation. Although current diagnostic practices (meal tolerance tests, hemoglobin A1c, fasting glucose, and C-peptide) provide a clear picture of glycemic control before surgery, they do not offer direct insight into extent or status of islet stress/damage. Thus, there is a need for simple biomarkers to assess disease progression, which could enhance timing of surgery, and predict islet isolation outcomes, to improve post-procedural functional measures 1,2 .
Circulating miRNA biomarkers are attractive candidates to assess pancreatic and islet damage due to their stability in circulation (free form or packaged in extracellular vesicles called exosomes) and ease of detection using PCR technologies. Our recent literature review suggested that a panel of miRNAs in circulation might be useful in landscaping disease progression in diabetes 7 . Thus, miRNA signature panels focusing on inflammatory and metabolic states may be especially useful in understanding disease progression in CP.
Using Hi-Seq analysis and RT-PCR, we previously reported elevated levels of hsa-miR-375, hsa-miR-148a-3p, hsa-miR-29b-3p, hsa-miR-216a-5p, and hsa-miR-200c-3p in islet culture media and in circulation in patients undergoing TPIAT at a single center 8 –11 . Further comprehensive analysis of circulating miRNAs in CP patients before TPIAT surgery using small RNA sequencing revealed distinct circulating miRNA profiles in CP patients in comparison to healthy controls (data unpublished). In this cohort of 18 CP patients and 6 healthy controls, out of 804 miRNAs analyzed, 43 miRNAs were significantly expressed in comparison to healthy controls. Of these, we identified hsa-miR-375, hsa-miR-148a-3p, hsa-miR-221-3p, hsa-miR-122-5p, hsa-miR-99b-5p, and let-7e-5p as readily detectible in plasma with existing technologies. In an independent study of CP patients, Xin and colleagues reported elevated levels of hsa-miR-221-3p in early CP and reduced levels of circulating hsa-miR-320d in late CP 12 . In two other independent CP studies, hsa-miR-148a-3p was also elevated, as was hsa-miR-122-5p in a subset of patients 12,13 . Circulating miRNAs may also be elevated in other pancreatic conditions including severe acute pancreatitis episodes (hsa-miR-7-5p) 14 and pancreatic ductal adenocarcinoma (hsa-miR-125b-5p, hsa-miR-200c-3p) 13,15 .
The multicenter Prospective Observational Study in TPIAT (POST) presents an opportunity to explore these biomarkers using prospectively obtained samples in a larger and more diverse well-phenotyped cohort of patients with CP or RAP undergoing TPIAT across the United States. Based on our previous observations and these independent reports, we included miRNAs identified in conditions of islet stress and damage, miRNAs identified in CP and miRNAs identified in other pancreatic conditions (acute pancreatitis and pancreatic cancer) in this study. This panel consisting of 10 miRNAs (hsa-miR-375, hsa-miR-148a-3p, hsa-miR-29b-3p, hsa-miR-216a-5p, hsa-miR-200c-3p, hsa-miR-221-3p, hsa-miR-122-5p, hsa-miR-320d, hsa-miR-125b-5p, and hsa-miR-7-5p) will provide an overall picture of circulating miRNAs in chronic pancreatitis. The main objective of our study was to study associations between circulating miRNA signatures and clinical measures of disease progression and islet autotransplantation outcomes.
Methods
Study Design
The POST study represents a multi-center consortium of 13 clinical institutions performing TPIAT in the United States and a data and coordinating center (DCC), formed to address research gaps in TPIAT, define predictive factors to determine timing of surgical intervention, determine patient and disease features associated with optimal pain and diabetes outcomes, and assess cost effectiveness of TPIAT 2 . Patients of any age scheduled for TPIAT for an indication of CP or RAP were eligible for inclusion. The POST protocol was reviewed and approved by the institutional review board at each participating institution (Supplementary Table 1 gives IRB protocol numbers). Informed consent or parental consent and child assent were obtained from each study participant as indicated by institutional protocols.
The current study included participants [n = 139—pediatric (<18 years of age, n = 48) and adult (≥18 years of age, n = 91)] of all ages, races, ethnicities and sexes, from 9 POST centers that had stored plasma aliquots obtained before surgery and had completed TPIAT surgery. Baseline data collected from medical records or participant interview included demographics, anthropometric measures, and disease clinical characteristics including disease etiology, disease duration, procedure history (ERCP history, celiac plexus block), surgical history, pre-existing diabetes mellitus (with islet function), and pancreatic exocrine insufficiency. Diagnosis of diabetes mellitus or pancreatic exocrine insufficiency were based on physician diagnosis at the treating center. Risk factors for pancreatitis, including genetic mutations, are abstracted from medical records data. Biorepository specimens are collected within a 90 day window before TPIAT surgery. The majority of specimens were collected immediately before surgery, with a median of 3 days (IQR 0, 5 days) between specimen collection and TPIAT. Fasting glucose, fasting C-peptide and hemoglobin A1c (HbA1c) levels were measured before TPIAT. Islet isolation results include islet mass expressed as islet equivalents (IEQ) or IEQ/kg, islet number (IN and IN/kg), and tissue volume. Plasma was collected in EDTA tubes, processed and aliquoted using the same standard operating procedures across sites, and stored frozen at −80°C until analyses.
Circulating miRNA analysis
The investigator was blinded to patient information and all samples were coded before miRNA analyses. Plasma miRNA was extracted using miRNeasy Serum/Plasma advanced kit (Qiagen, Germantown, MD, USA) following the manufacturer’s instructions. Briefly, plasma samples were stabilized using MS2 RNA (bacteriophage MS2, Millipore Sigma, St. Louis, MO, USA) and an exogenous spike-in control, Unisp6 (Qiagen, Germantown, MD, USA). Following lysis, plasma proteins were precipitated and centrifuged to remove debris. After isopropanol precipitation, nucleic acids were bound to spin-column and washed thrice, using series of ethanol solutions, and miRNA was eluted in nuclease free water containing RNAse inhibitor. Then miRNA (2 µl/sample) was converted to cDNA using the miRCURY LNA RT kit (Qiagen, Germantown, MD, USA) following the manufacturer’s instructions. Quantitative real-time qPCR was performed using the commercially available miRCURY LNA miRNA PCR assay system. Absolute quantification of miRNAs was performed using an miRNA standard curve (miRCURY LNA miRNA mimics, Qiagen, Germantown, MD, USA). We used UniSp6 cycle threshold values, 18 ± 0.2, as within acceptable range for inclusion in the analysis. In all assays, we did not exclude any sample from analysis as all data fell within this acceptable range. All samples were analyzed in triplicate, with cDNA dilution at 1:40.
Statistical analysis: All analyses were done using the R system (v. 4.0.1) 16 . A very small number of measurements were below the limit of detection; these were inputed as 0.9 times the lowest measurement of the same miRNA measure. Preliminary analyses found that the miRNA measures had distributions skewed to the right (i.e., had a long upper tail); the Box-Cox procedure suggested analyzing their logarithms, so all analyses use the common log (log to base 10) of the miRNA measures.
Two-sample t-tests were used to estimate and test the association of log10miRNA levels with binary characteristics (e.g., pre-operative exocrine insufficiency), and simple linear regression was used to estimate and test the association of log10miRNA levels with measures on continuous scales (e.g., C-peptide or islet-yield measures). Plots of the latter associations describe the association using a scatterplot smoother (R package ggplot2, v. 3.3.1 17 , functions geom_smooth() and geom_point() with default settings). Effect sizes (ESs) for t-tests are described as differences between groups, while effect sizes for regressions are described using Pearson’s correlation (r).
Results
Preoperative Patient Characteristics
Preoperative patient characteristics are in Table 1. The indication for proceeding to TPIAT, as determined by the surgical center, was CP in 67 (48%), RAP in 25 (18%), and both in 47 (34%) participants, with an average disease duration of 6.8 years. Table 1 summarizes risk factors for pancreatitis, with genetic risk factors being common in this cohort (77% of participants). Genetic risk factors included PRSS1 (31.8%), SPINK1 (21.5%), CFTR (37.4%), and CTRC (8.4%). In this study population, 18 patients (13%) had diabetes before TPIAT while 47 (34%) had diagnosed pancreatic exocrine insufficiency. The average HbA1c, fasting glucose and fasting C-peptide levels were 5.7%, 98.6 mg/dl and 1.96 ng/ml, respectively. After islet isolation, the average tissue volume of islet cell products was 8.9 ml, with islet yields at an average of 3896 IEQ/kg (median 2995 IEQ/kg; IQR 2018 to 4995).
Pre-Operative Patient Characteristics.
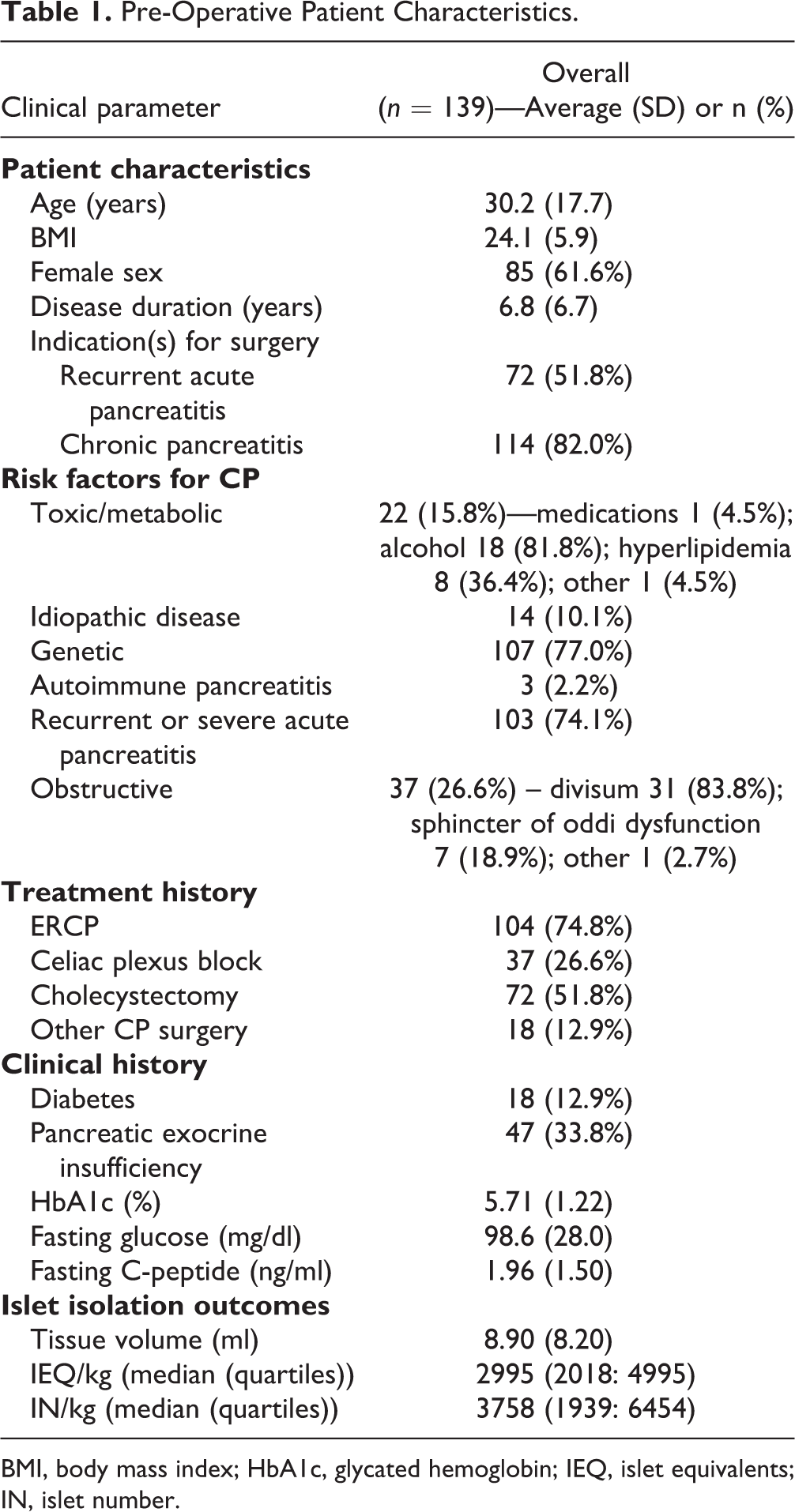
BMI, body mass index; HbA1c, glycated hemoglobin; IEQ, islet equivalents; IN, islet number.
Association of Circulating miRNAs with Patient Characteristics
Of the 10 miRNAs analyzed, hsa-miR-221-3p exhibited a significant association with age (r = 0.18, P = 0.03) and hsa-miR-148a-3p was associated with BMI (r = 0.18, P = 0.04) in the entire study cohort. Dividing the cohort into age groups (pediatric (<18 years of age) vs adult (≥18 years of age), hsa-miR-148a-3p (P = 0.03, effect size (ES) = adult average minus pediatric average 0.17, standard error [SE] 0.08), hsa-miR-200c-3p (P = 0.02, ES 0.20 SE 0.09) and hsa-miR-221-3p (P = 0.009, ES 0.23 SE 0.09) were significantly elevated in adults compared to pediatric patients (Fig. 1A-C). Disease duration, symptoms, history of surgery and type of pancreatitis (recurrent acute or chronic) were not significantly associated with circulating miRNA levels.
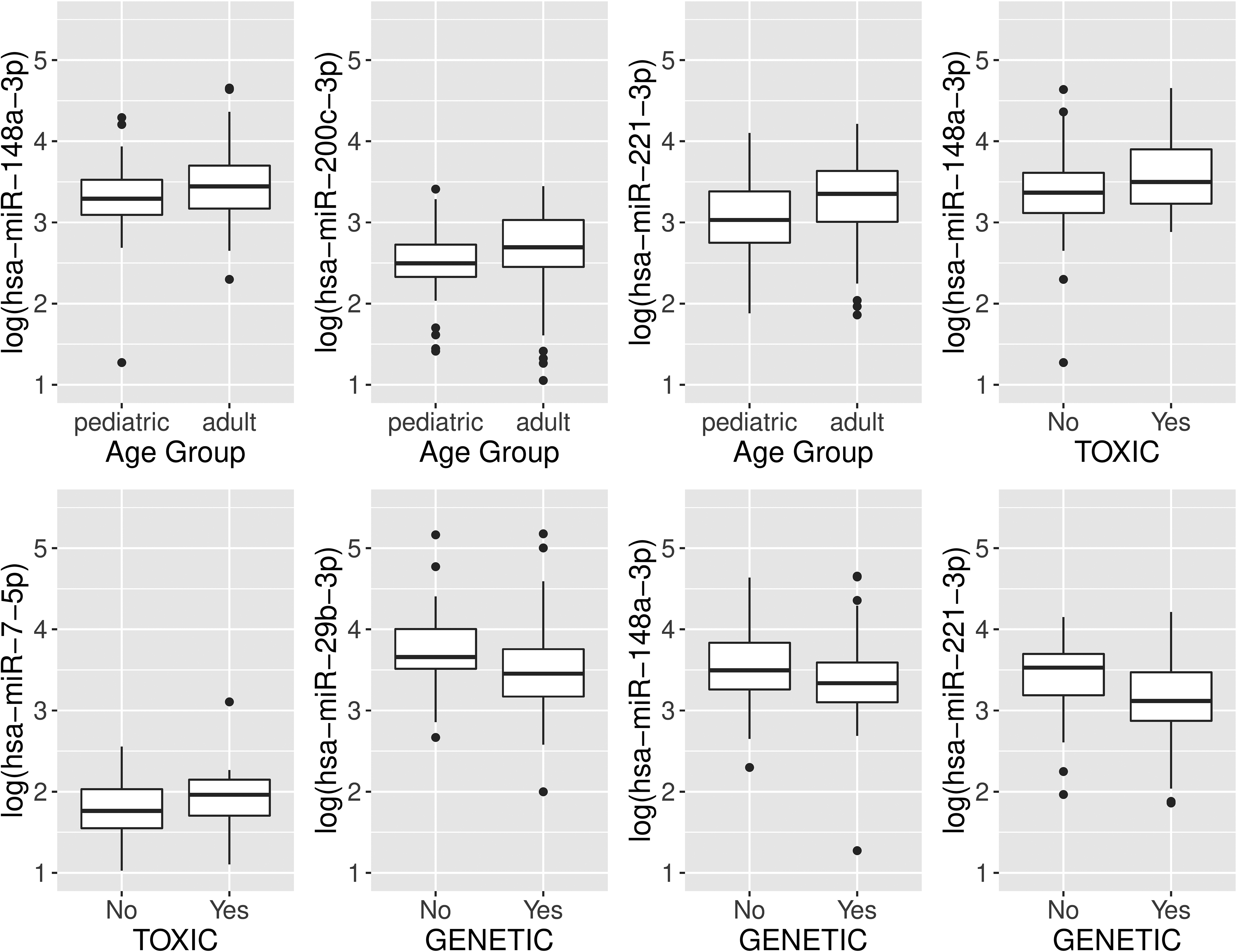
Association of Circulating miRNAs with Pancreatitis Risk Factors and Disease Complications (Diabetes and Exocrine Insufficiency)
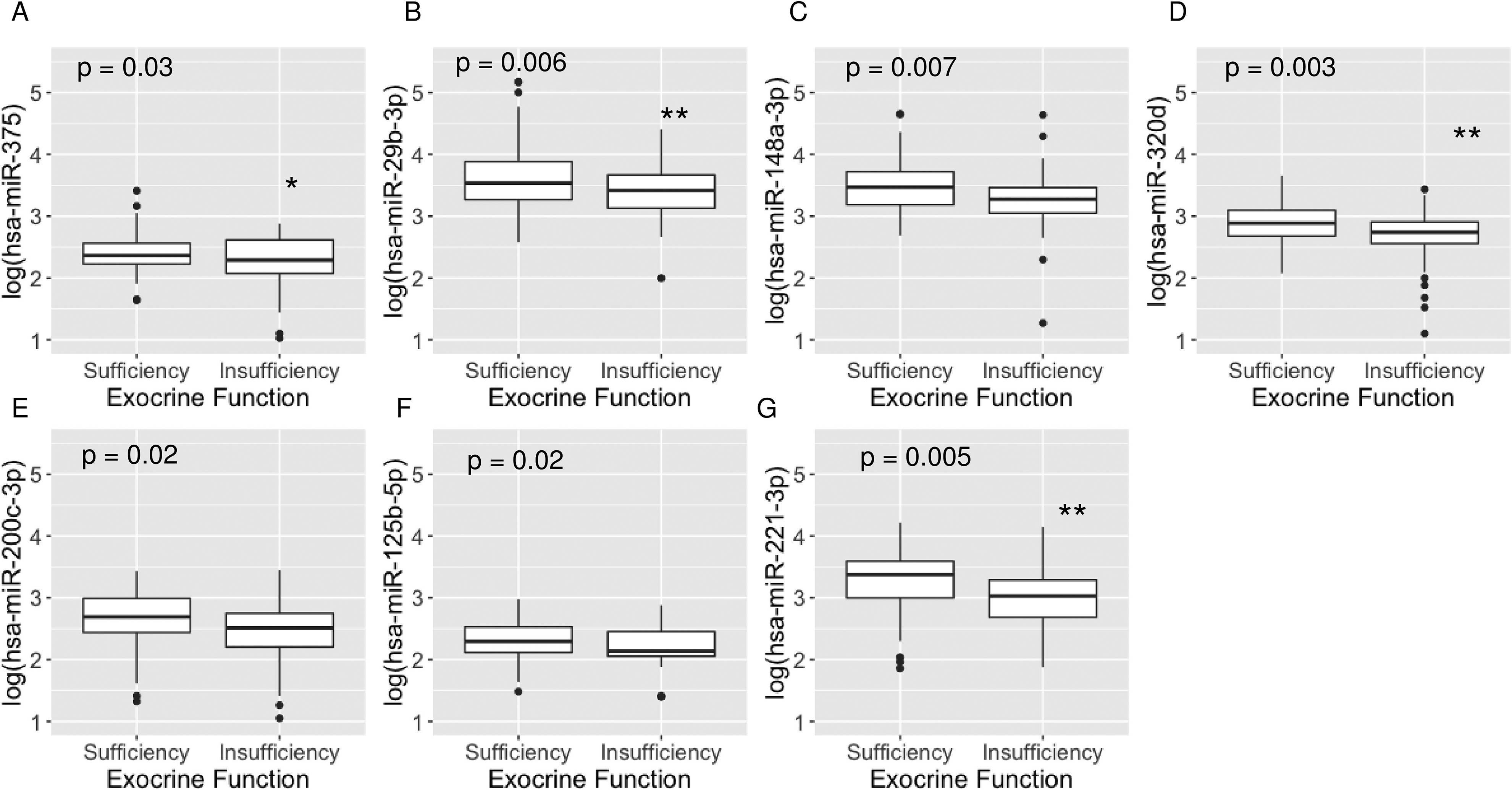
We tested whether miRNA biomarkers were associated with these pancreatitis risk factors (1) toxic/metabolic disease; (2) pancreatitis-predisposing genetic mutations; and (3) obstructive disease. In these analyses, hsa-miR-148a-3p (P = 0.04, ES = average of those with toxic etiology minus average of those not having toxic etiology = 0.22 SE 0.11, Fig. 1D) and hsa-miR-7-5p (P = 0.04, ES 0.20 SE 0.10, Fig. 1E) were elevated significantly in patients with pancreatitis having toxic etiologies compared to patients without such an etiology. Also, hsa-miR-29b-3p (P = 0.03, ES = average of those with genetic etiology minus average of those not having genetic etiology = −0.23 SE 0.10, Fig. 1F), hsa-miR-148a-3p (P = 0.04, ES −0.19 SE 0.09, Fig. 1G) and hsa-miR-221-3p (P = 0.01, ES −0.26 SE 0.10, Fig. 1H) were lower in patients with genetic versus non-genetic etiology. Circulating miRNAs were not associated with obstructive disease or pre-existing diabetes (data not shown). Compared to patients with preserved exocrine function, patients with exocrine insufficiency had lower levels of hsa-miR-375 (P = 0.03, ES = average of those with exocrine insufficiency minus average of those who are exocrine-sufficient −0.14 SE 0.06, Fig. 2A), hsa-miR-29b-3p (P = 0.006, ES −0.25 SE 0.09, Fig. 2B), hsa-miR-148a-3p (P = 0.007, ES −0.22 SE 0.08, Fig. 2C), hsa-miR-320d (P = 0.003, ES −0.19 SE 0.06, Fig. 2D), hsa-miR-200c-3p (P = 0.02, ES −0.21 SE 0.09, Fig. 2E), hsa-miR-125b-5p (P = 0.02, ES −0.13 SE 0.06, Fig. 2F), and hsa-miR-221-3p (P = 0.005, ES −0.25 SE 0.09, Fig. 2G).
Association of Circulating miRNAs with Glycemic Measures Before TPIAT
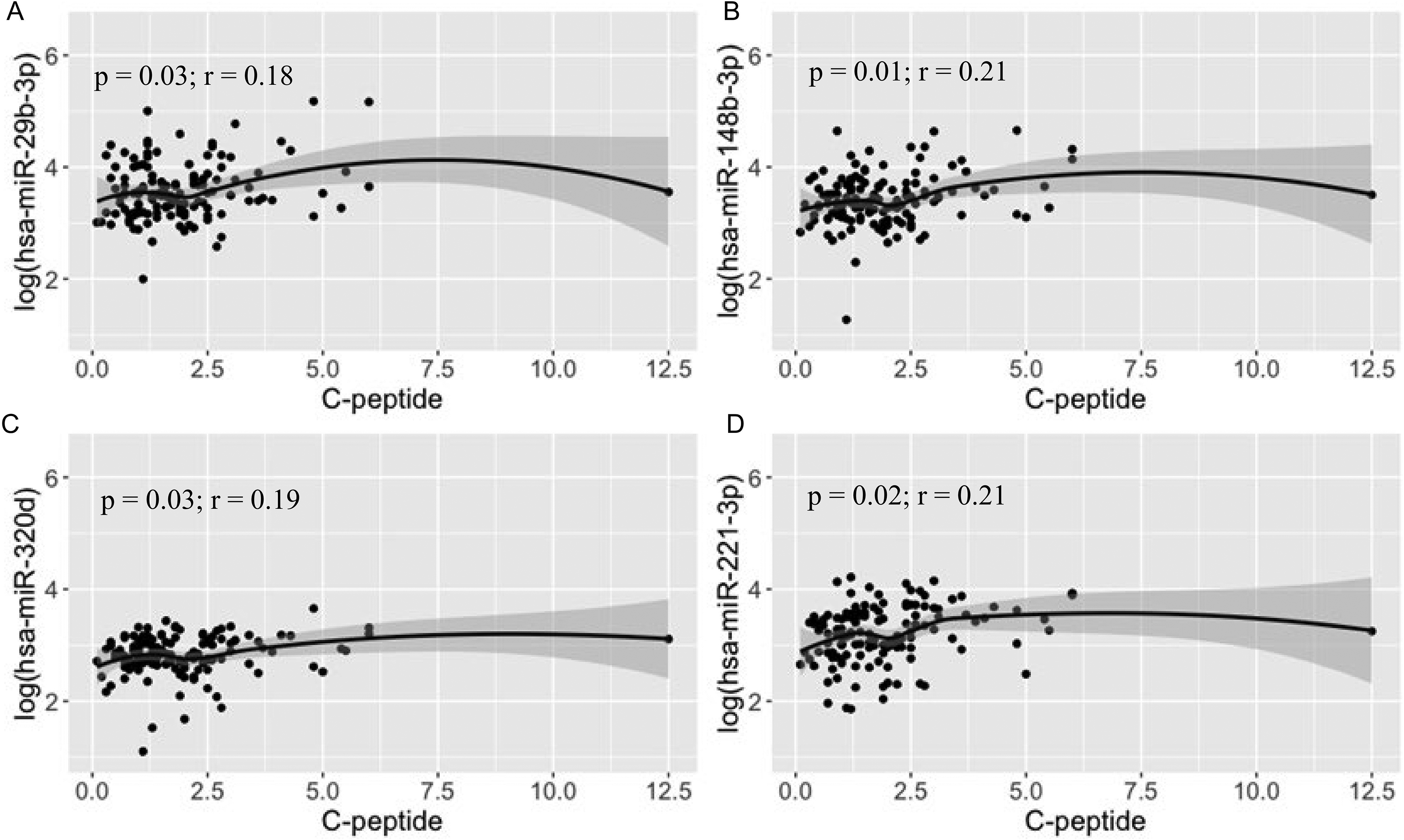
Circulating miRNAs were not associated with HbA1c and fasting glucose levels (Table 2). However, hsa-miR-29b-3p (r = 0.18, P = 0.03, Fig. 3A), hsa-miR-148a-3p (r = 0.21, P = 0.01, Fig. 3B), hsa-miR-320d (r = 0.19, P = 0.03, Fig. 3C), and hsa-miR-221-3p (r = 0.21, P = 0.02, Fig. 3D) were significantly associated with fasting C-peptide levels (Table 3).
Association of Circulating miRNAs with Preoperative Metabolic Measures.
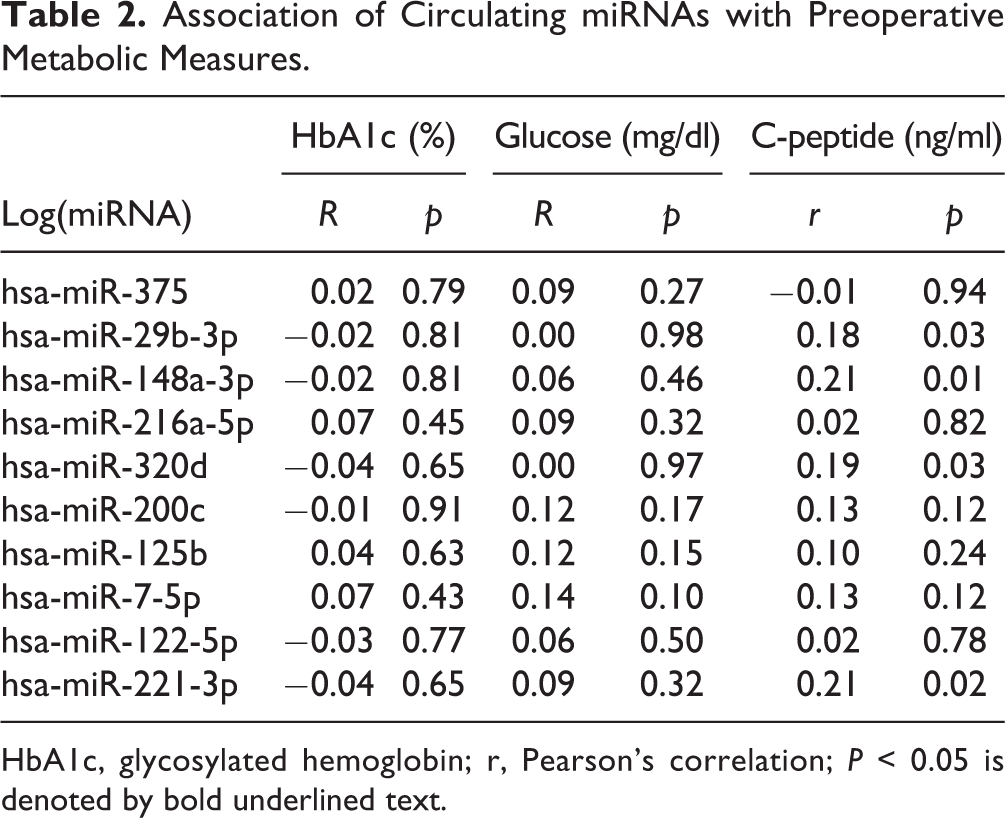
HbA1c, glycosylated hemoglobin; r, Pearson’s correlation; P < 0.05 is denoted by bold underlined text.
Association of Circulating miRNAs with Islet Isolation Outcomes.
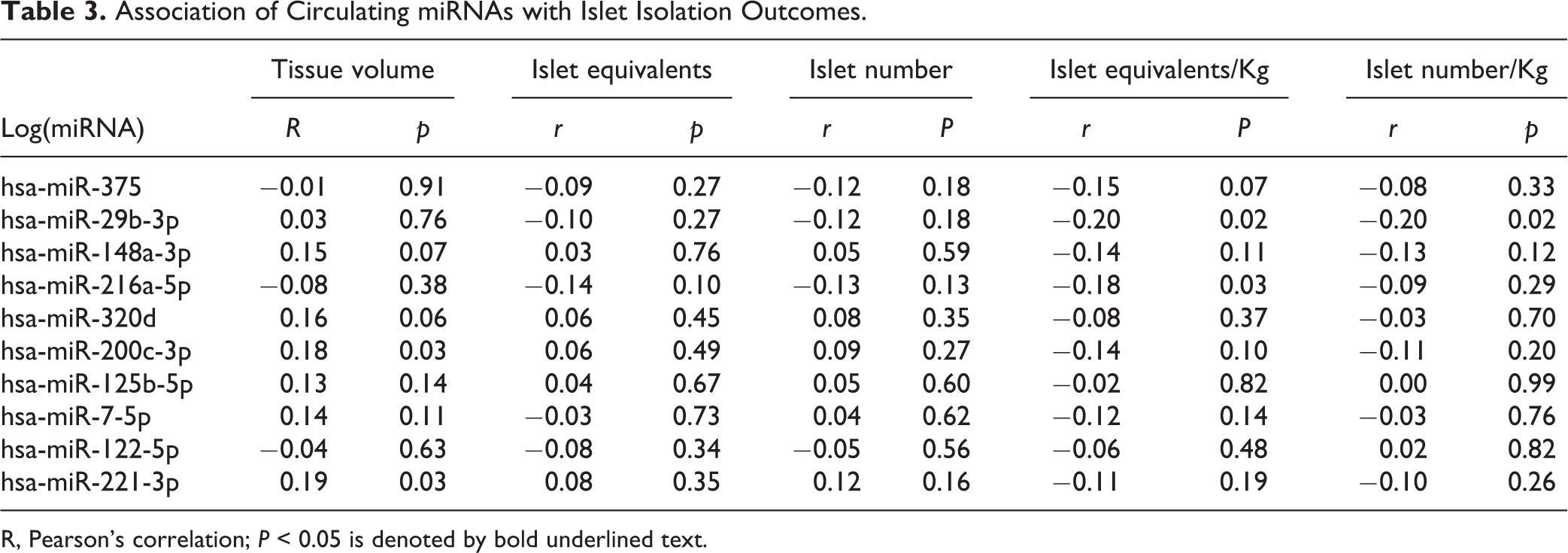
R, Pearson’s correlation; P < 0.05 is denoted by bold underlined text.
Association of Circulating miRNAs with Islet Isolation Outcomes
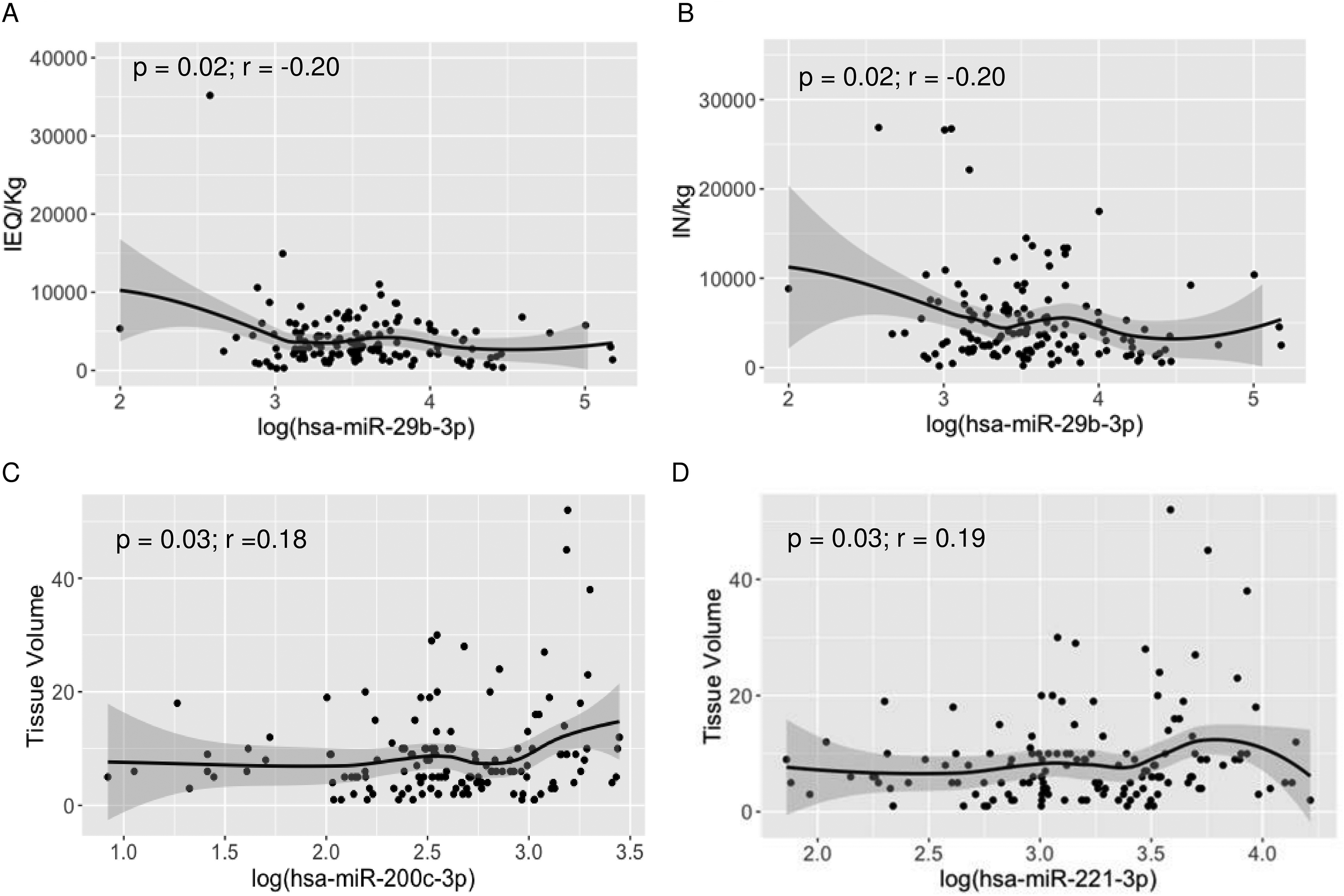
Circulating miRNAs were not associated with total islet yield (IEQ, IN) (Table 3). However, hsa-miR-29b-3p was inversely associated with islet yield/kg body weight (IEQ/kg and IN/kg) (r = −0.20, P = 0.02, Table 3, Fig. 4A, B) and hsa-miR-216a-5p was inversely associated with IEQ/kg (r = −0.18, P = 0.03) but not with IN/kg. Circulating hsa-miR-200c-3p (r = 0.18, P = 0.03, Fig. 4C) and hsa-miR-221-3p (r = 0.19, P = 0.03, Fig. 4D) levels were associated with post isolation tissue volume, a measure that reflects both islet mass and exocrine tissue contamination of the islet product.
Discussion
Patients with CP are considered for TPIAT when other interventions have failed to alleviate persistent pain and improve quality of life. While total pancreatectomy improves chronic pain and opioid dependence, islet autotransplantation is important to prevent brittle diabetes after surgery 2,18,19 . One challenge in achieving optimal islet function is inability to predict disease stage and islet isolation outcomes before TPIAT. In the POST study, in addition to studying associations of patient and disease characteristics with favorable pain and health-related quality of life outcomes 2 , we aimed to identify distinctive molecular signatures of pancreatic damage, especially islet cell stress and damage, before surgery. The present study, using a cohort of 139 patients recruited across 9 institutions, highlights the associations of specific circulating miRNAs with patient, disease and islet isolation outcomes in CP and RAP patients undergoing TPIAT (Table 4).
Summary of Associations of Circulating miRNAs with Patient and Disease Characteristics and Islet Isolation Outcomes

Direct or inverse associations are denoted as + or –, respectively. Increased or decreased levels are denoted as upward or downward arrows, respectively. Red indicates positive association, green indicates negative association. * Age category indicates adults vs pediatrics (<18 years of age).
We selected miRNA biomarkers based on our prior work and external data suggesting tissue specificity for islet or pancreas tissue and correlation with islet or acinar damage or stress (Table 4). Compared to pediatric patients, circulating hsa-miR-148a-3p, hsa-miR-200c-3p, and hsa-miR-221-3p were significantly higher in adults. Of these, hsa-miR-148a-3p exhibited a significant positive association with BMI while others showed a non-significant trend toward positive association with BMI. In those with genetic risk factors for pancreatitis, circulating levels of hsa-miR-29b-3p, hsa-miR-148a-3p, and hsa-miR-221-3p were significantly lower compared to patients with non-genetic pancreatitis; conversely, patients with toxic/metabolic disease had elevated levels of hsa-miR-148a-3p and hsa-miR-7-5p, strikingly opposite compared to patients with genetic pancreatitis. Our observation of elevated hsa-miR-7-5p levels in toxic/metabolic disease is particularly significant because of similar observations in an independent study of patients with severe acute pancreatitis 14 . We could not determine whether the differences in circulating miRNA levels in pediatric and adult patients are due to age or etiology because all of our pediatric patients had at least one genetic risk factor for pancreatitis.
Patients with exocrine insufficiency showed a striking pattern of reduced levels of multiple circulating miRNAs (except hsa-miR-216a-5p, hsa-miR-7-5p, and hsa-miR-122-5p) compared to patients without diagnosed exocrine insufficiency. In a cohort of patients with late stage CP (with exocrine insufficiency), levels of circulating hsa-miR-320a-d and hsa-miR-221-3p were reduced compared to patients with early stage CP (without exocrine insufficiency) 12 . Together with our current study, these observations suggest that severity of disease leading to exocrine insufficiency and possibly reduced pancreatic tissue volume may reduce pancreatic miRNA content for release into circulation. This hypothesis should be validated in future studies using pancreatic tissue biopsies procured during TPIAT.
One of the most important factors determining islet graft function in patients after TPIAT procedure is the mass of transplanted islets 19,23 . Currently, status of endocrine cell stress/damage and islet isolation outcomes generally cannot be predicted before surgery, necessitating research into predictive tools. In our cohort, circulating miRNAs were not associated with HbA1c (%) or fasting blood glucose levels before surgery. However, circulating hsa-miR-29b-3p, hsa-miR-148a-3p, hsa-miR-320d, and hsa-miR-221-3p were associated with fasting C-peptide levels before TPIAT. C-peptide levels are a marker of residual beta cell mass and decrease with advanced chronic pancreatitis 24 . In contrast, elevated fasting C-peptide level in a non-diabetic individual indicates insulin resistance. Previous metabolic and epidemiologic studies for diabetes risk in CP suggest that insulin resistance is a factor in the pathogenesis of pancreatogenic DM 25,26 . Thus we hypothesize that in patients with CP or RAP who are also insulin resistant (and thus have higher fasting C-peptide), elevated pancreatic islet miRNA levels are a marker for metabolic islet stress.
This is in line with other reports of elevated levels of these miRNAs in non-diabetic autoantibody positive, pre-diabetic and diabetic individuals 27 –35 . Elevated circulating hsa-miR-29b-3p has been reported in insulin-resistant conditions including obesity 27 , before onset of gestational diabetes 28 and reduced insulin sensitivity index 29 ; while elevated circulating hsa-miR-148a-3p levels have been associated with islet damage in early type 1 diabetes pathogenesis 30 –35 and in pre-diabetes 32 . In our study, hsa-miR-148a-3p was positively associated with both BMI and fasting C-peptide, also suggesting a state of insulin resistance.
In the context of CP and islet autotransplantation, we have previously observed inverse association of preoperative hsa-miR-375 levels with ΔC-peptide (stimulated C-peptide minus basal C-peptide in a glucose tolerance test) and post-isolation islet counts in a cohort of 31 patients undergoing TPIAT 11 . Further analysis of associations between circulating miRNA levels and islet isolation outcomes found inverse association of hsa-miR-29b-3p with islet yield (IEQ/kg, IN/kg body weight). Note that hsa-miR-29b-3p showed opposite associations with fasting C-peptide levels and islet yield in our cohort, warranting further validation for better understanding of this circulating miRNA in CP patients. Also, hsa-miR-200c-3p and hsa-miR-221-3p levels were associated with post-isolation islet tissue volume, a measure that reflects both islet mass and (predominantly) residual exocrine tissue. Notably, these two measures were also higher in adults (vs children) and overall significantly lower in patients with exocrine insufficiency. Exocrine insufficiency manifests in chronic disease due to severe fibrosis and necro-inflammation 3,21 . We hypothesize that hsa-miR-200c-3p and hsa-miR-221-3p may be elevated in circulation during disease progression when pancreas tissue volume is sufficient to contribute to circulating miRNA levels, or that the association of hsa-miR-200c-3p and hsa-miR-221-3p with tissue volume may reflect the higher tissue volumes seen in adult patients. Our current study design permits follow-up after transplantation for investigation of circulating miRNA levels and their associations with post-transplantation functional measures.
As for the pancreatic origin of these circulating miRNAs, we previously reported elevated levels of these miRNAs in circulation during digestion of pancreas, islet infusion and immediately after transplantation 8 –10 . In our ex vivo islet studies, we observed release of miRNAs (hsa-miR-375, hsa-miR-148a-3p, hsa-miR-29b-3p, hsa-miR-216a-5p, hsa-miR-200c-3p, hsa-miR-125b-5p) under proinflammatory and hypoxic culture conditions 10 . We did not observe any striking relationships between clinical measures and either hsa-miR-216a-5p, an acinar cell specific miRNA (undetected in circulation in healthy conditions) 36 , or hsa-miR-122-5p, a liver specific miRNA 22 . We included liver-specific miRNA in our analysis because our unpublished observations found elevated levels at 3 months after intraportal infusion of islets in TPIAT patients (Vasu et al, unpublished). Most importantly, given the reported alterations of some of these circulating miRNAs (hsa-miR-200c-3p, hsa-miR-125b-5p) in pancreatic cancer 13,20 , data should be interpreted in light of clinical history. We acknowledge that other tissues may contribute to circulating miRNA levels and influence our interpretation. Changes in circulating miRNA levels may reflect underlying pathological conditions and thus, studying circulating miRNA panels consisting of multiple miRNAs will help establish their trends and inter-relationships in specific disease conditions. Nevertheless, further studies using an expanded panel of miRNAs and larger cohorts are important in establishing miRNA associations with CP.
This is the first study to arise from the POST cohort using biorepository specimens and highlights the future potential for research from this consortium. In these first exploratory analyses, strength of correlations were weak (r < 0.25 in magnitude), reflecting the heterogeneity of the cohort and noise in the assays, and thus necessitating further studies using large cohorts and stringent analysis for establishing strong associations. The current study’s aim was to explore potential associations and results have not yet been adjusted for multiple comparisons or adjusted for potential confounders (age, BMI). We are as yet unable to investigate specific subgroups (for example, only those with a specific cause of pancreatitis) because such subgroup analyses would further cut sample size and reduce power. These steps should be considered for future research questions informed by these preliminary studies.
Our findings in this first study represent an interim analysis and are limited by partial enrollment of the POST cohort and reliance on clinically available data for pancreatitis history. Genetic testing for pancreatitis risk factors is performed as part of clinical care, and may be incomplete for some adult patients, leading to a risk of underestimating genetic disease in this cohort. Diabetes outcomes will not be analyzed until enrollment is complete and thus this initial study cannot measure the association of miRNA biomarkers with post-TPIAT diabetes outcomes in this initial study. Also of note, because the POST study is observational and collects clinically available data, the classification of exocrine insufficiency relied on physician diagnosis, and could be susceptible to under-reporting or misdiagnosis. Apart from patient characteristics, factors including sample handling and differences in islet isolation process across institutions may influence the associations analyzed in this study. Because sample sizes are small at individual sites, subanalyses for site-specific effects is not feasible at this point, but could be explored in the future with a larger cohort size. Although the current POST study does not collect control biospecimens, future explorations could include comparisons of miRNA levels with non-pancreatitis control groups in more participants to assess specificity.
Overall, this first exploratory multi-center study highlights the potential associations of specific circulating miRNAs with pancreatic exocrine insufficiency, preoperative metabolic measures and islet isolation outcomes. Future studies will focus on confirming preliminary results, and on follow-up analysis and predictive potential of these circulating miRNAs regarding islet mass yield, and metabolic and clinical outcomes.
Supplemental Material
Supplemental Material, sj-pdf-1-cll-10.1177_0963689721999330 - Circulating miRNA in Patients Undergoing Total Pancreatectomy and Islet Autotransplantation
Supplemental Material, sj-pdf-1-cll-10.1177_0963689721999330 for Circulating miRNA in Patients Undergoing Total Pancreatectomy and Islet Autotransplantation by Srividya Vasu, Jiemin M. Yang, James Hodges, Maisam A. Abu-El-Haija, David B. Adams, Appakalai N. Balamurugan, Greg J. Beilman, Srinath Chinnakotla, Darwin L. Conwell, Martin L. Freeman, Timothy B. Gardner, Betul Hatipoglu, Varvara Kirchner, Luis F. Lara, Katherine A. Morgan, Jaimie D. Nathan, Andrew Posselt, Timothy L. Pruett, Sarah J. Schwarzenberg, Vikesh K. Singh, Martin Wijkstrom, Piotr Witkowski, Bashoo Naziruddin and Melena D. Bellin in Cell Transplantation
Footnotes
Abbreviations
Acknowledgments
The study investigators would like to acknowledge the contributions of collaborators and coordinators at the participating centers.
Ethical Approval
Ethical approval for this study was obtained from each individual participating institution’s IRB (IRB#s available in supplement).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the protocols approved by each institutional review board.
Statement of Informed Consent
Written informed consent was obtained from participants for their study participation and for anonymized publication of aggregate data.
Minnesota - Jayne Pederson, Peggy Ptacek
Baylor - Rehma Shabbir, Jessica Clark
Cincinnati Children’s - Jyoti Patel, Amanda Schreibeis
Dartmouth - Penny Doughty
Johns Hopkins - Mahya Faghih
Pittsburgh - Rita Johnson
Chicago - Lindsay Basto, Piotr Bachul
South Carolina - Jason Hirsch
Ohio State - Jill Buss
UCSF - Joanne Kwan
Louisville - Mechelle Kaufman
Cleveland - Amy Orasko
Data & Coordinating Center, Minnesota - Leslie Long-Simpson, Rebecca Mitchell, Helen Voelker.
We also acknowledge input from Dr. Syed Ahmad at the University of Cincinnati.
Supporting information statement
Additional supporting information may be found online in the Supporting Information section at the end of the article. Deidentified data may be accessed upon request to the study investigators.
Declaration of Conflicting Interests
M. Bellin discloses research funding from Viacyte and Dexcom, and medical advisory role (DSMB) for Insulet. The authors of this manuscript otherwise have no conflicts of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by NIDDK R01-DK109124 (PI Bellin).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
