Abstract
The therapeutic efficacy of bone marrow mesenchymal stem cells (BMSCs) in myocardial infarction (MI) is hindered by poor cell survival. This study explored the role of N6-methyladenosine (m6A) regulation, specifically YTHDC1, in improving BMSC transplantation for MI. By screening m6A-related regulators in hypoxia and serum deprivation (HSD)-induced BMSC apoptosis, YTHDC1 was found to be downregulated. Overexpression of Ythdc1 in BMSCs reduced apoptosis markers, reactive oxygen species (ROS) release, and improved cell survival under HSD conditions. Conversely, Ythdc1 knockdown enhanced apoptosis. In rat MI models, transplantation of Ythdc1-overexpressing BMSCs improved cardiac function and reduced myocardial fibrosis. Mechanistically, YTHDC1 interacts with nuclear factor kappa B (NF-κB) inhibitor-alpha mRNA, suggesting its involvement in BMSC survival pathways. This study identifies YTHDC1 as a potential target to enhance BMSC efficacy in MI therapy.
Keywords
Introduction
The prevalence and mortality associated with cardiovascular disease (CVD) have been steadily increasing 1 . Among the various types of CVD, coronary heart disease (CHD) has emerged as the leading cause of death worldwide. Myocardial infarction (MI) is the most common and severe form of CHD 2 . MI triggers the structural and functional changes in cells in various tissues and organs. This is often results in cell death. Alongside conventional surgical, intervention, and pharmacological interventions, stem cell therapy has emerged as a promising approach to the treatment of many diseases, especially ischemic diseases, such as MI 3 , hypoxic/ischemic cerebral palsy 4 , ischemia-reperfusion kidney injury 5 , and oxygen-induced retinopathy 6 . This therapeutic modality harnesses the regenerative potential of stem cells, which can secrete trophic factors, undergo directed differentiation, and help restore damaged vascular and tissue 7 . Mesenchymal stem cells (MSCs) transplantation has demonstrated safety and therapeutic benefit in the treatment of MI. This has been attributed to their ability to undergo multi-directional differentiation and exhibit low immunogenicity 8 . Despite these inherent advantages, transplanted MSCs survival after MI is often compromised by the adverse ischemic and hypoxia micro-environment within the infarct myocardium 9 . Consequently, achieving optimal therapeutic efficacy through stem cell transplantation for the treatment of MI is significantly challenged by the limited survival and subsequent apoptosis of MSCs.
N6-methyladenosine (m6A) RNA modification serves as a pivotal regulator in post-transcriptional RNA metabolism and various biogenesis processes. YT521-B homology-domain-containing protein 1 (YTHDC1), as the only nucleus-localized m6A recognition protein in the YT521-B homology domain family of methyl-binding proteins 10 , has a relatively unique role in selective splicing and nuclear export 11 , as well as regulation of RNA stability and degradation12–15. Moreover, YTHDC1 plays a critical and extensive role in the regulation of disease initiation and progression. Recent evidence suggests that YTHDC1 influences cellular processes related to stem cell death and survival. These processes include cell proliferation and apoptosis16–21. For instance, studies have been reported that increased YTHDC1 expression is associated with the malignant proliferation and maintaining tumor stemness 19 . In addition, another investigation revealed that reduced YTHDC1 levels led to the degradation of sequestome 1 (Sqstm1) mRNA in the nucleus, while interacting with ELAV-like RNA-binding protein 1 (ELAVL1) to modulate cell apoptosis in human carcinoma cell lines treated with elevated glucose levels 22 . However, the precise role of YTHDC1 in regulating the survival of bone marrow mesenchymal stem cells (BMSCs) in the challenging local micro-environment of ischemia and hypoxia following MI remains unclear.
In this study, a significant downregulation of Ythdc1 was observed under hypoxia and serum deprivation (HSD) conditions in BMSCs. Notably, the expression of YTHDC1 gradually decreased with increasing duration of HSD treatment. Based on these findings, we hypothesized that YTHDC1 might be an excellent candidate for promoting BMSCs survival and reducing apoptosis in a hostile ischemic and hypoxia environment. Here, we aimed to investigate the effects and underlying mechanism of Ythdc1 overexpression on BMSCs survival under HSD conditions and its potential therapeutic implications for MI. Our findings provide valuable insights and potential alternative treatment option for stem cell transplantation in the context of MI.
Materials and Methods
Animals
Sprague-Dawley (SD) rats of SPF grade were obtained from Hunan Slake Jingda Experimental Animal Co., Ltd. [Animal License SCXK (Xiang) 2019-0004]. All animal care, surgical interventions, and handling procedures were performed according to the guidelines and regulations set by the Institutional Animal Care and Use Committee of Zunyi Medical University. The study protocol was approved by the Medical Ethics Committee of Zunyi Medical University, with approval number KLLY(A)-2021-054.
BMSCs Isolation, Culture, and Identification
BMSCs were isolated from healthy male SD rats aged approximately 3 to 4 weeks old, and weighing 70–100 g, and then cultured in low-glucose Dulbecco’s modified Eagle’s medium (L-DMEM) (Gibco, USA). Flow cytometry was used for the identification of surface markers in BMSCs. And the osteogenic differentiation potentiality of BMSCs was analyzed using OriCell® kit for rat bone marrow MSCs osteogenic differentiation (#RAXMX-90021, Cyagen Biosciences, China) according to the manufacturer’s instructions. Detailed description is given in the Supplementary Material methods.
Exposure of BMSCs to HSD
To mimic the micro-environment of the ischemic myocardium, an in vitro model of BMSC apoptosis was established as previously described by exposing the cells to HSD (1% O2, 5% CO2, 94% N2, 37℃)23,24. BMSCs were transferred into serum deprivation medium and exposed to a hypoxia environment consisting for 3, 6, 9, 12, or 24 h after being rinsed 2–3 times with phosphate-buffered saline (PBS). For the control group, BMSCs were cultured under normal oxygen conditions (21% O2, 5% CO2, 37°C), with 10% fetal bovine serum (FBS), and allowed to reach 80%–90% confluence before initiating the HSD treatment. Lentivirally transfected BMSCs required only 12 h of culture period under HSD conditions.
Western Blot
The prepared BMSCs were digested with trypsin and resuspended in RNA immunoprecipitation (RIP) assay (RIPA buffer; #P0013B, Beyotime, China) supplemented with a 1 × protease inhibitor cocktail mixture (Epizyme, China), and the total protein concentration was quantified using a BCA assay Kit (#PC0020, Epizyme, China). 20 μg of protein was loaded for separation by SDS-PAGE and then transferred to 0.22 μm PVDF membranes (#ISEQ00010, Epizyme, China). After blocking with 5% skim milk (Epizyme, China), the membranes were incubated for 8 h at 4℃ with the specific primary antibodies for 1 h with HRP-conjugated secondary antibody (#SA00001-2, Proteintech, China). Finally, the proteins were visualized using ChemiDoc Imaging System (Bio-Rad, USA) after the addition of ECL Western blotting substrate (Epizyme, China). Protein expression was quantified using ImageJ software and normalized to the housekeeping protein β-actin. The Western blot antibodies used in this study are listed (S Tab. 1).
CCK-8 Assay
Cell viability was assessed by means of the CCK-8 assay (Biotechwel, USA) according to the manufactures’ instructions. Briefly, the BMSCs from the different subgroups were digested, counted, and reseeded into a 96-well plate (1,000 cells/well) for 12 h under standard conditions. The BMSCs were then incubated with 10 μl CCK-8 reagent per well for a further 3 h at 37°C. The absorbance value (OD) was detected at 450 nm using a microplate reader (ThermoFisher Scientific, Inc., USA) with the wells without cells as blanks.
Annexin V-FITC/Propidium Iodide Staining
Annexin V-FITC/propidium iodide (PI) staining was performed to assess apoptosis using an Annexin V-FITC/PI apoptosis kit (4A Biotech, China). Briefly, both the culture medium and cell suspension of BMSCs digested with trypsin without ethylenediaminetetraacetic acid (EDTA) were harvested into one 15-ml reaction tube. The supernatant was then removed after centrifugation at 1200 rpm for 5 min, and the cell precipitate was resuspended in 100 μl 1× binding buffer without Ca2+ by mixing 10 μl Annexin V-FITC and 5 μl PI reagent for 15 min at room temperature and protected from light. The samples were analyzed by flow cytometry and the apoptosis rate was calculated as the percentage of cells stained with Annexin V-FITC.
Total RNA Extraction and Real-Time Quantitative Polymerase Chain Reaction
Total RNA was extracted from the cultured BMSCs using the RNA iso Plus Kit (#9108, Takara, Japan) according to the manufacturer’s instructions. The extracted RNA was then reverse transcribed into cDNAs using the PrimeScript RT Kit (#RR036A, Takara, Japan) according to the supplied protocol. For real-time quantitative polymerase chain reaction (RT-qPCR), the cDNAs were amplified using SYBR Green dye (#RR820A, Takara, Japan) on a CFX96 real-time PCR system (Bio-Rad Laboratories, Inc., CA, USA). Relative mRNA expression levels were determined using the 2-△△Ct quantification method, with β-actin serving as a housekeeping gene for normalization. Primer sequences used for RT-qPCR are listed (S Tab. 2).
Lentivirus Infection
The lentiviral particles used in this experiment were obtained from HanHeng Biological Company (Shanghai, China). Selected BMSCs were digested, counted, and reseeded into flasks for overnight culture. The BMSCs were then infected with lentiviruses at a multiplicity of infection (MOI) of 50 for 24 h. After 24 h of virus infection, the virus-containing supernatants were replaced with fresh medium, and the cells were further cultured for a further 48 h. To select for lentivirally stable strains, puromycin was added to the culture medium at a concentration of 6 μg/ml for 48 h, when the cells reached 60%–70% confluence. The virus volume was calculated as follows: virus volume = MOI × cell number/virus concentration × 1000. The specific interference sequences used are listed
ROS Detection
Cellular ROS production was detected using a 2ʹ,7ʹ-dichlorodihydrofluorescein diacetate (DCFH-DA) staining kit (#D6883; Sigma-Aldrich, USA) according to the manufacturer’s instructions. Briefly, selected BMSCs from different experimental groups were rinsed 2–3 times with PBS when they reached 80%–90% confluence. The cells were then incubated in L-DMEM without phenol red at 37°C, protected from light, and mixed with 10 μM DCFH-DA reagent for 30 min. After the incubation, the cells were washed and digested with trypsin without phenol red, and then resuspended in PBS. Cells without DCFH-DA reagent were used as a blank control group. Samples were analyzed by flow cytometry to measure the level of cellular ROS.
MI Model and BMSCs Transplantation
Fifty-five male SD rats weighing between 200 and 250 g were randomly allocated into two groups: the experimental group (MI surgery group, comprising five subgroups with five SD rats each) and the control group (sham surgery group, comprising five subgroups with six SD rats each). Prior to the surgery, the rats were anesthetized to ensure their well-being and minimize pain and discomfort. Briefly, a midline incision was made in the chest, and the heart was exposed. The left anterior descending coronary artery (LAD) was ligated using a suture to induce localized ischemia in the myocardium. Successful ligation was confirmed by observing an elevated ST segment in the limb electrocardiogram (ECG), a characteristic feature of MI 25 . Then, the BMSCs used for transplantation were suspended in 100 μl of PBS at a concentration of 1 × 106 cells per rat. Intramyocardial injection was performed using a microsyringe to deliver the BMSCs to the border zone of the infarct area26,27. In the control group, an equivalent volume of PBS was injected in a similar manner. The control group underwent a sham surgery procedure to serve as a baseline comparison. The rats in this group received a similar surgical procedure, including anesthesia and chest incision, but without LAD ligation. Instead, they were administered an equivalent volume of PBS to mimic the transplantation procedure performed in the experimental group.
2,3,5-Triphenyltetrazolium Chloride Staining
The heart tissue was cut into five thin slices from the ligature to the apex of the heart, with approximately 2 mm apart between each slice. The slices were placed in 2% 2,3,5-triphenyltetrazolium chloride (TTC; #G3005, Solarbio, China) at 37°C for 30 min under light protection 28 . The sections were washed three times with ddH2O and fixed with 4% paraformaldehyde for 20 min. The sections were photographed using a digital camera.
Echocardiography
After injection of BMSCs or PBS for 4 weeks, the rats were anesthetized, and transthoracic echocardiography was performed to assess cardiac function 29 . M-mode echocardiography scanning was completed within three cardiac cycles, and cardiac function was manifested by left ventricular ejection fraction (LVEF), left ventricular fraction shortening (LVFS), left ventricular end-diastolic diameter (LVEDd), left ventricular end-systolic diameter (LVED). Images were obtained using a Vevo 2100 ultrasound system (Visual Sonics, Canada).
Masson’s Trichrome (Masson) Staining
Rat myocardial tissue was fixed in 4% paraformaldehyde, dehydrated through a graded ethanol series, routinely paraffin-embedded, and sectioned at 5 μm thickness. Sections were stained with Masson’s trichrome staining (#G1346, Solarbio, China) according to the manufacturer’s instructions. The image was captured by light microscopy. Collagen fibers were stained blue and the normal myocardial tissue was stained red. The ratio of the fibrotic area was quantified using Image J software.
RNA Sequencing (RNA-seq)
RNA-seq was performed by NewCore Biotechnology (Shanghai, China). Briefly, total RNAs (>100 mg) was extracted from BMSCs lentivirally transfected with knockout empty vactor or Ythdc1; bound RNA was eluted and extracted with TRIzol reagent. Ribo-off rRNA Depletion Kit (#N406-02, Vazyme, China) was used for ribosome depletion, and the extracted and purified RNA was library constructed using NEBNext Ultra II Directional RNA Library Prep Kit (#E7760, NEB, USA) and sequenced on NovaSeq 6000 platform.
RNA Immunoprecipitation
The relationship between YTHDC1 protein and NfƙBiα mRNA was further confirmed by RIPA 30 . Briefly, BMSCs were harvested and washed twice with PBS. After centrifugation, the pellets were resuspended in IP lysis buffer and kept on ice for 5 min. Magnetic beads Protein A/G were incubated with 5 μg antibody at 4℃ overnight supplemented with 100 μl sample lysate, using normal rabbit IgG as a negative control RIP reaction. The protein A/G complex was resuspended with RIPA buffer after the samples were centrifuged at 4000 rpm for 1 min, repeated for 5–10 times. The beads were then resuspended with Proteinase K buffer and incubated at 55℃ for 30 min. TRIzol was used for RNA purification and RIP-qPCR was performed according to the manufacturer’s protocol. RNA enrichment was normalized to the input control. Relative mRNA expression levels were determined using the 2-△△Ct quantification method, with β-actin serving as a housekeeping gene for normalization. Primer sequences used for RT-qPCR are listed (S Tab. 3).
Statistical Analysis
Representative experiments were repeated at least three times for in vitro experiments and five times for in vivo experiments. All data are presented as mean and standard deviation (SDs), and were analyzed using GraphPad Prism 7.0 software (San Diego, CA, USA). Statistical differences between two data sets were analyzed by Student’s t-test, and multiple groups were analyzed by one-way analysis of variance (ANOVA) method with a post hoc pairwise comparison. Differences for which P < 0.05 were considered statistically significant.
Results
HSD-Induced Apoptosis in BMSCs and Downregulation of YTHDC1
To simulate the micro-environment of MI, BMSCs were cultured under HSD conditions. To evaluate the effect of different HSD treatment time conditions on BMSCs activity, cell viability was assessed using the CCK-8 assay. The CCK-8 assay demonstrated a gradual decrease in BMSC viability with prolonged HSD treatment (Fig. 1A). According to the result, we found that the cell activity of BMSCs gradually decreased with increasing HSD treatment time. Notably, the most pronounced reduction in BMSCs activity was noted after 12 h of HSD exposure, while the decline in cellular activity commenced to attenuate after 24 h of HSD treatment. Based on this finding, the optimal condition for BMSC treatment in this experiment was determined to be 12 h of HSD exposure, as it demonstrated the most pronounced effects on cell viability. Subsequently, the Western blot analysis was performed to examine the expression of apoptotic proteins in BMSCs. Compared with the control group, BMSCs cultured under HSD conditions for 12 h exhibited decreased levels of the anti-apoptotic protein Bcl-2, while the levels of the pro-apoptotic proteins Bax and cleaved caspase 3 were increased (Fig. 1B, C).
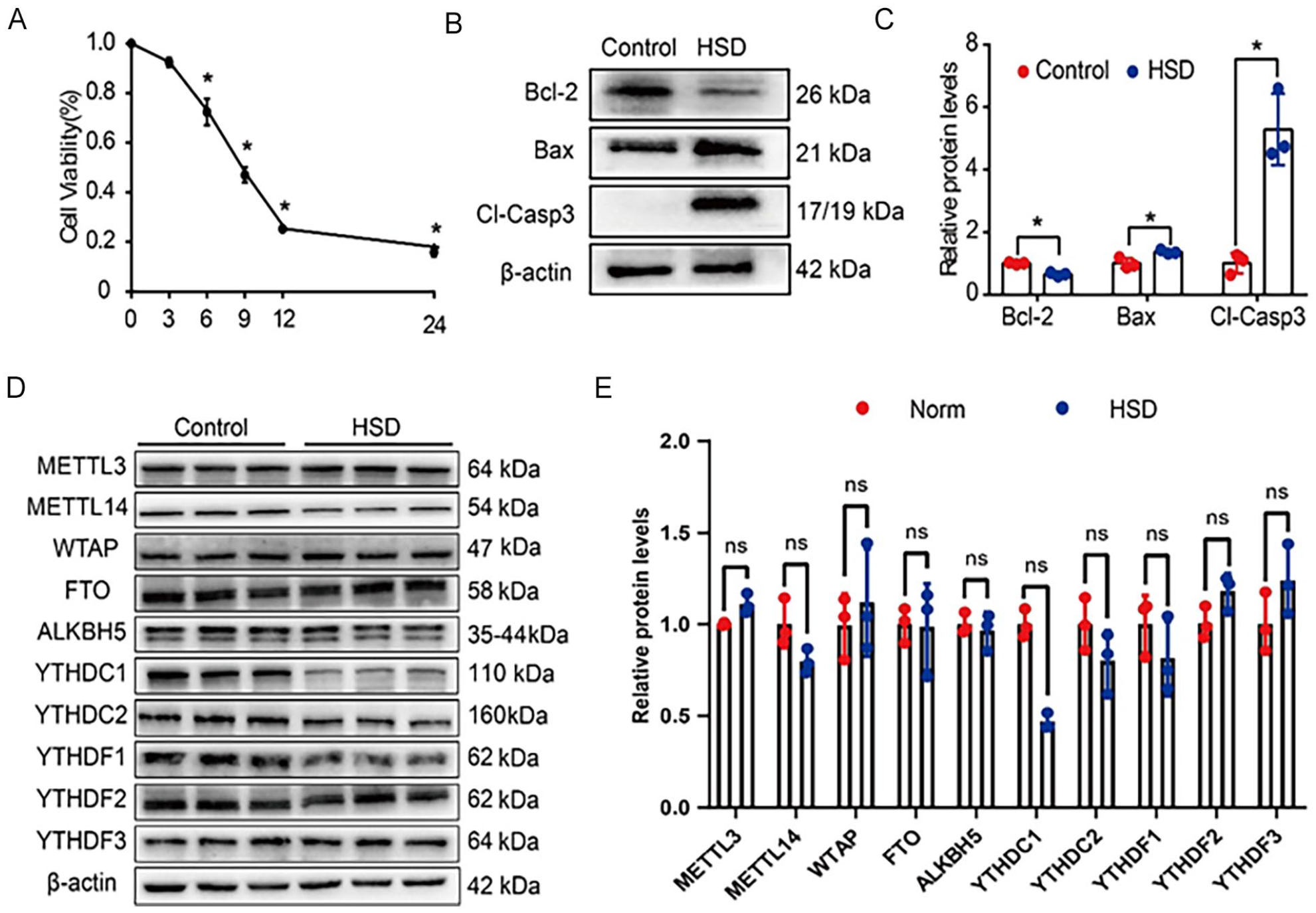
HSD-induced apoptosis in BMSCs and downregulation of YTHDC1. (A) Quantitative analysis of the CCK8 assay showing the HSD condition on the viability of BMSCs at different time points. (B-C) Western blot analysis of Bcl-2, Bax, and cleaved caspase 3 expression in BMSCs treated for 12 h under control conditions (21% O2, 5% CO2, 37℃) with 10% FBS or under hypoxia and serum deprivation conditions (1% O2, 5% CO2, 94% N2, 37℃). Quantification results of protein expression are shown (n = 3). The relative expression level was normalized by β-actin. (D-E) Western blot analysis of m6A methyltransferase, demethylase and methyl-binding enzyme expression levels after 12 h of BMSCs in control and HSD groups. Quantification results of protein expression are shown. Relative expression level was normalized by β-actin. Data are presented as mean ± SDs (n = 3) and analyzed by one-way ANOVA with Fisher’s LSD post hoc analysis (A-C) or Student’s t-test (D–E). *P < 0.05.
Furthermore, the expression levels of enzymes associated with m6A RNA modification were evaluated through Western blot analysis. The expression of several enzymes involved in m6A modification, including methyltransferase like 3 (METTL3), methyltransferase like 14 (METTL14), Wilms’ tumor associating protein (WTAP), fat mass and obesity-associated gene (FTO), AlkB homolog 5 (ALKBH5), YTH domain-containing 1/2 (YTHDC1/2), and YTH domain family 1/2/3 (YTHDF1/2/3)31–33, was examined. Remarkably, the protein levels of YTHDC1 were obviously reduced under HSD conditions (Fig. 1D, E). These observations suggest that HSD treatment induces apoptosis in BMSCs and is associated with the downregulation of YTHDC1. Thus, YTHDC1 may potentially mediate the apoptotic response of BMSCs under HSD conditions.
Ythdc1 Overexpression Reduces HSD-Induced Apoptosis in BMSCs In Vitro
To investigate the role of Ythdc1 in HSD-induced BMSC apoptosis, we conducted experiments to examine whether Ythdc1 overexpression can reduce the level of apoptosis in BMSCs. Lentivirus-mediated infection was utilized to achieve sustained overexpression of Ythdc1. BMSCs were infected with lentiviruses at a MOI of 50 for 24 h (S Fig. 2A). After 24 h of viral infection, the virus-containing supernatants were replaced with fresh medium, and the cells were further cultured for an additional 48 h. Puromycin was added to the culture medium at a concentration of 6 μg/ml for 48 h to select for lentivirally stable strains (S Fig. 2B) when the cells reached 60%–70% confluence. The efficiency of lentivirus-mediated overexpression was confirmed (Fig. 2A, B).
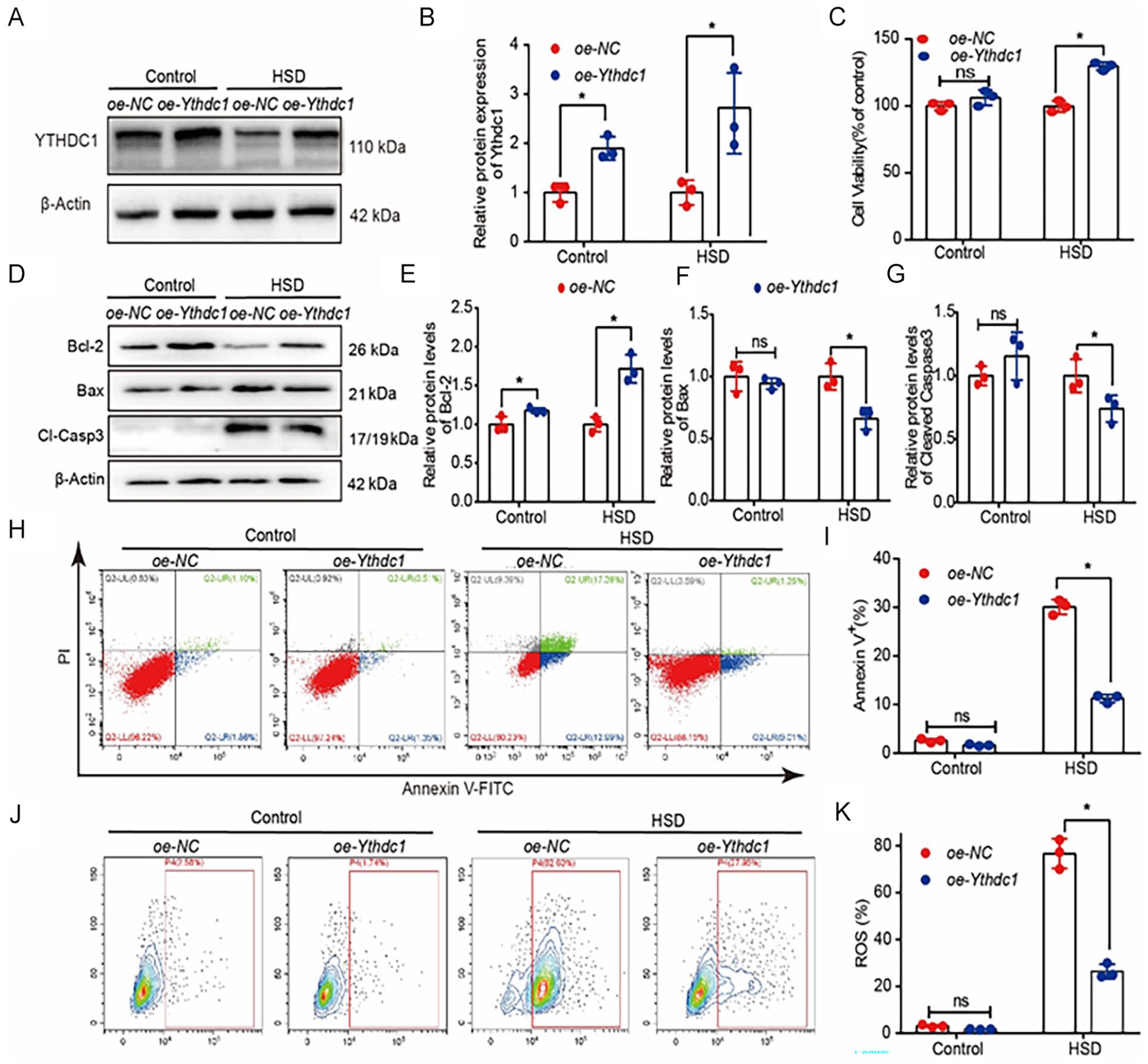
Ythdc1 overexpression reduces HSD-induced apoptosis in BMSCs in vitro. (A-B) Representative Western blot image and quantification of YTHDC1 protein levels in lentivirus overexpression (oe-Ythdc1) and control lentivirus (oe-NC) BMSCs under control and HSD conditions. The relative expression level was normalized to β-actin. (C) Cell viability of Ythdc1 overexpressing BMSCs and control BMSCs under control and HSD conditions. (D-G) Representative Western blot image and quantification of Bcl-2, Bax, and cleaved caspase 3 protein levels in oe-Ythdc1 and oe-NC BMSCs under control and HSD conditions. The relative expression level was normalized to β-actin. (H-I) Representative Annexin V-FITC/PI staining images (scale bar = 20 μm) and quantification of the rate of apoptosis. (J-K) Representative 2ʹ,7ʹ-dichlorodihydrofluorescein diacetate (DCFH-DA) staining images and quantification of the cellular ROS production. Data are presented as mean ± SDs (n = 3) and analyzed by Student’s t-test. *P < 0.05.
Several experiments were performed to test our hypothesis. The results demonstrated that BMSCs with Ythdc1 overexpression exhibited higher cell viability under HSD conditions (Fig. 2C). In addition, the expression of the anti-apoptotic protein Bcl-2 was increased, while the levels of the pro-apoptotic proteins Bax and cleaved caspase 3 were lower than those in the control group. However, no significant differences were observed under control conditions, except for the expression of Bcl-2 (Fig. 2D, G). Further validation experiments revealed that Ythdc1 overexpression could reduce the rate of apoptosis and decrease the level of cellular ROS production under HSD conditions (Fig. 2H, K). These results indicate that while Ythdc1 overexpression may not significantly affect the function of BMSCs under control conditions, it significantly reduces the level of apoptosis induced by HSD conditions, thereby enhancing the tolerance of BMSCs to HSD.
Ythdc1 Knockdown Exacerbates HSD-Induced Apoptosis in BMSCs In Vitro
To further investigate the role of Ythdc1 in BMSCs apoptosis, we conducted experiments to examine the effect of Ythdc1 knockdown. Lentiviral-mediated Ythdc1 knockdown was performed, and the most efficient knockdown sequence was selected based on Western blot analysis (S Fig. 3A, B) and evaluated under both control and HSD conditions (Fig. 3A, B).
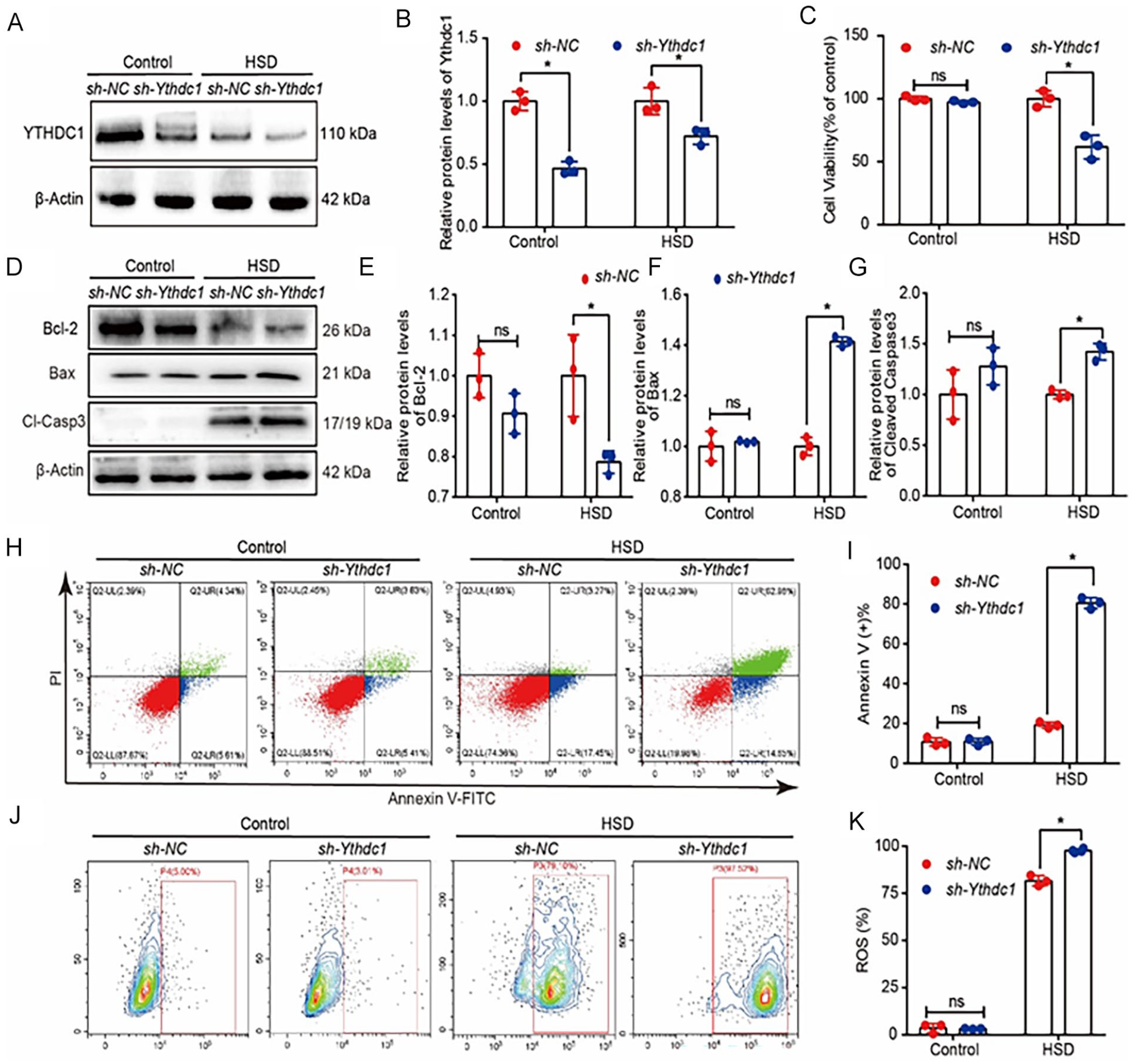
Ythdc1 knockdown exacerbates HSD-induced apoptosis in BMSCs in vitro. (A-B) Representative Western blot image and quantification of YTHDC1 protein levels in knockdown lentivirus (sh-Ythdc1) and control lentivirus (sh-NC) BMSCs under control and HSD conditions. The relative expression level was normalized by the β-actin. (C) Cell viability of Ythdc1 knockdown BMSCs and control BMSCs under control and HSD conditions (D-G) Representative Western blot image and quantification of Bcl-2, Bax, and cleaved caspase 3 protein levels in sh-Ythdc1 and sh-NC BMSCs under control and HSD conditions. The relative expression level was normalized by β-actin. (H-I) Representative Annexin V-FITC/PI staining images (scale bar = 20 μm) and quantification of the rate of apoptosis. (J-K) Representative 2ʹ,7ʹ-dichlorodihydrofluorescein diacetate (DCFH-DA) staining images and quantification of the cellular ROS production. Data are presented as mean ± SDs (n = 3) and analyzed by Student’s t-test. *P < 0.05.
CCK8 assays revealed that Ythdc1 knockdown reduced the viability of BMSCs under HSD conditions but not under control conditions (Fig. 3C). Western blot analysis demonstrated that the expression of the anti-apoptotic protein Bcl-2 was lower under HSD conditions, while the levels of the pro-apoptotic proteins Bax and cleaved caspase 3 were higher in Ythdc1 knockdown BMSCs compared with the control group. However, no significant differences were observed under control conditions (Fig. 3D–G). Further validation experiments showed that Ythdc1 knockdown increased the rate of apoptosis in BMSCs and elevated the level of cellular ROS production only under HSD conditions (Fig. 3H–K). These findings indicate that although Ythdc1 knockdown may not significantly affect the expression of Bcl-2, Bax, cleaved caspase 3, and cellular ROS production under control conditions, it significantly enhances the level of apoptosis, thereby reducing the tolerance of BMSCs and decreasing cell viability under HSD conditions.
RNA-seq Identifies NfƙBiα as a Potential Target of YTHDC1
To elucidate the potential mechanisms of HSD-induced apoptosis in BMSCs mediated by YTHDC1, we performed RNA-seq analysis on RNA extracted and purified from the BMSCs, infected with either sh-NC or sh-Ythdc1 lentivirus, and subsequently treated under HSD conditions for 12 h. The RNA-seq-based heatmap illustrated differential gene expression, including 171 downregulated genes and 229 upregulated genes (Fig. 4A). Enrichment analysis of these differentially expressed genes was then performed using Metascape (http://metascape.org). The results revealed that the upregulated genes were primarily associated with biological processes such as oxygen sensing, oxidative stress, inflammatory response, and nuclear factor-kappa-B (NF-κB) signaling pathway regulation (Fig. 4B). Conversely, the downregulated genes were predominantly linked to the inflammatory response and amino acid metabolism (S Fig. 5A). The NF-κB signaling pathway notably functions to inhibit cell apoptosis34–36. Studies have demonstrated that NF-κB is closely linked to various processes, such as tumor initiation, growth, and metastasis37–42. Mutations in NF-κB family genes are frequently observed in human cancers, particularly in malignant lymphoid tumors43–45. Since NF-κB downstream targets include cyclinD1 and c-Myc, its persistent activation promotes cell growth, leading to uncontrolled cell proliferation46–48. Integrating these findings with the apoptosis phenotype, we identified four potential apoptosis-related target mRNAs from the 229 upregulated genes: NF-κB inhibitor alpha (NfƙBiα), endoplasmic reticulum-to-nucleus signaling 1 (Ern1), neurotrophic tyrosine receptor kinase 1 (Ntrk1), and cell cycle and apoptosis regulator 1 (Ccar1). To validate the RNA-seq results, we examined the mRNA expression levels of NfƙBiα, Ern1, Ntrk1, and Ccar1 in BMSCs treated with normoxic and HSD conditions, following the overexpression or knockdown of Ythdc1. Notably, the expression levels of these four mRNAs consistently mirrored the sequencing results in HSD following Ythdc1 knockdown. However, under HSD conditions, only the expression pattern of NfƙBiα mRNA exhibited opposite trends in the cases of Ythdc1 overexpression and knockdown (Fig. 4C–F). These findings suggest that NfƙBiα mRNA may be a potential anti-apoptotic target gene of Ythdc1, which warrants further investigation.
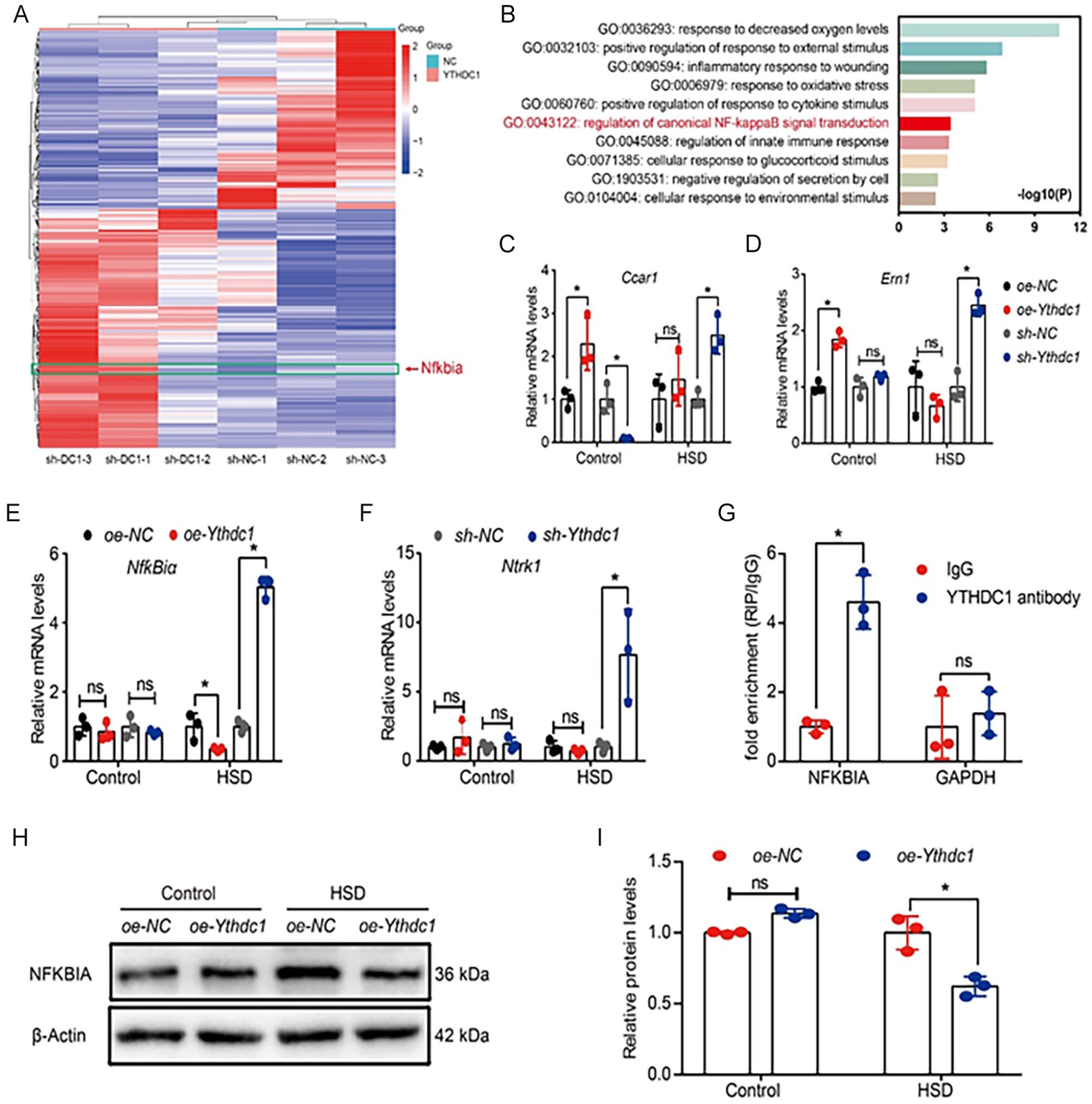
To gain further insight into the relationship between YTHDC1 and NfƙBiα, we examined the effect of YTHDC1 on using previous data from the cross-linking immunoprecipitation high-throughput sequencing (CLIP-seq) database. Notably, a binding peak was identified between YTHDC1 (UniProtID: Q96MU7) and exon 3 of NfƙBiα mRNA (NCBI: NM_020529.3) was identified (S Fig. 5B). Subsequently, an RIPA using a YTHDC1 antibody targeting the region of NfƙBiα mRNA (chr14: 35872376-35872418) confirmed the binding of YTHDC1 to NfƙBiα in BMSCs. The fold enrichment value of 4.67 indicates specific binding of YTHDC1 to NfƙBiα mRNA (Fig. 4G). Furthermore, our results show that HSD conditions led to an upregulation of NfƙBiα, which could be reversed by Ythdc1 overexpression (Fig. 4H, I).
YTHDC1 Regulates HSD-Induced Apoptosis in BMSCs Dependent on NfƙBiα mRNA
Given the potential role of YTHDC1 in modulating NfƙBiα mRNA expression, we sought to investigate the relationship between YTHDC1 and NfƙBiα mRNA, as well as the functional significance of NfƙBiα in regulating apoptosis under HSD conditions. To this end, we performed NfƙBiα knockdown experiments in the context of YTHDC1 knockdown, and successfully verified of NfƙBiα knockdown by Western blot analysis (Fig. 5A, B).
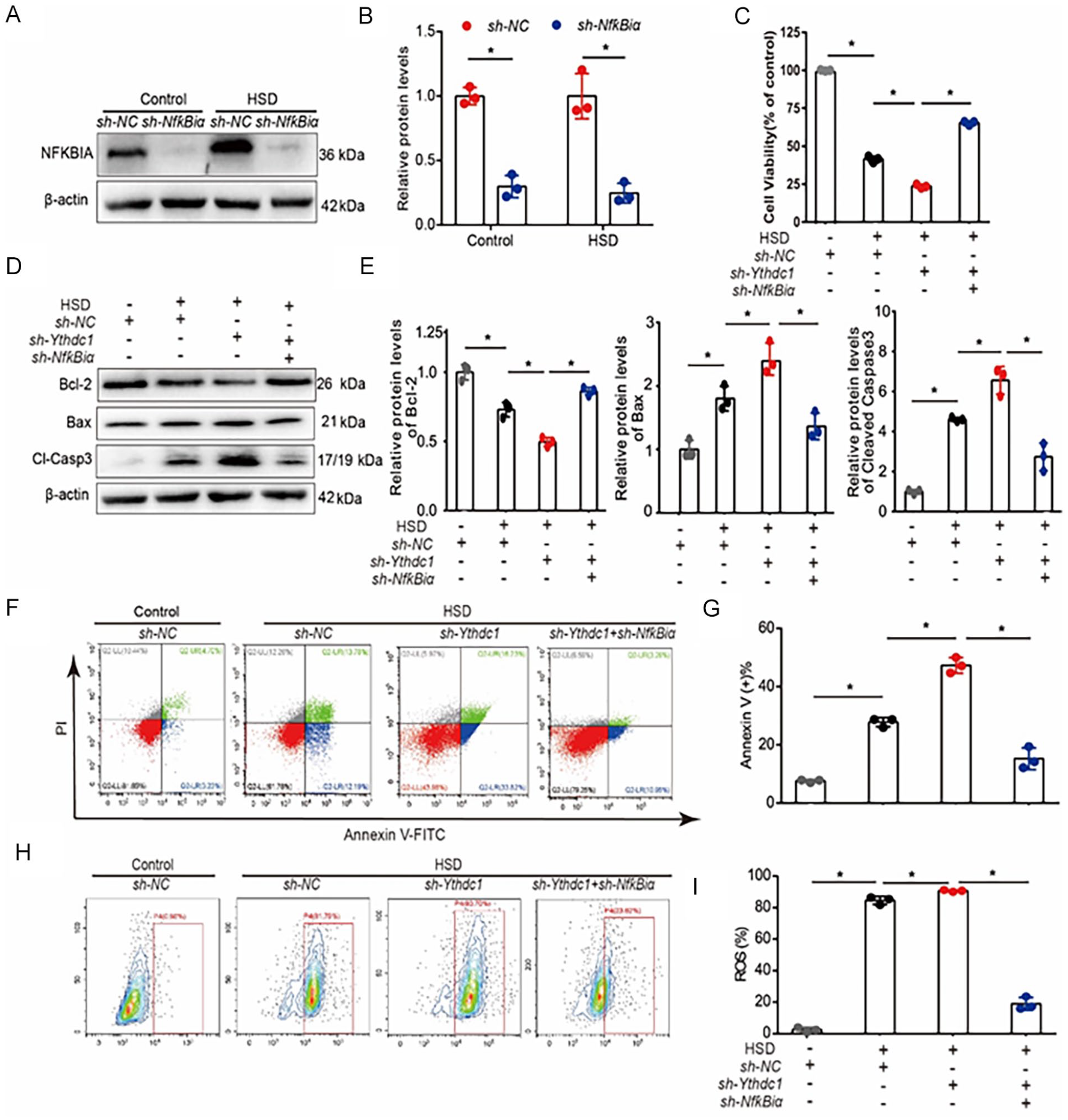
YTHDC1 regulates HSD-induced apoptosis in BMSCs dependent on NfƙBiα mRNA. (A-B) Representative Western blot image and quantification of NFKBIA protein levels in knockdown lentivirus (sh-NfƙBiα) and control lentivirus (sh-NC) BMSCs under control and HSD conditions. The relative expression level was normalized to β-actin. (C) Cell viability of NfƙBiα knockdown BMSCs and control BMSCs under control and HSD conditions. (D-E) Representative Western blot image and quantification of Bcl-2, Bax, and cleaved caspase 3 protein levels in sh-NfƙBiα and sh-NC BMSCs under control and HSD condition. The relative expression level was normalized to β-actin. (F-G) Representative Annexin V-FITC/PI staining images (scale bar = 20 μm) and quantification of the rate of apoptosis. (H-I) Representative 2ʹ,7ʹ-dichlorodihydrofluorescein diacetate (DCFH-DA) staining images and quantification of the cellular ROS production. Data are presented as mean ± SDs (n = 3) and analyzed by Student’s t-test. *P < 0.05.
To determine whether NfƙBiα knockdown could reverse the apoptosis induced by Ythdc1 knockdown, we performed several apoptosis-related experiments to evaluate the functional role of NfƙBiα in BMSCs. The results showed that NfƙBiα knockdown increased the cell viability (Fig. 5C) and the expression of Bcl-2, while decreasing the expression of Bax and cleaved caspase 3 under HSD conditions, thereby counteracting the apoptosis induced by Ythdc1 knockdown (Fig. 5D, E). Furthermore, the rate of apoptosis, as detected by Annexin V-FITC/PI staining, and cellular ROS production, were reduced in NfƙBiα knockdown BMSCs under HSD conditions. However, no significant differences were observed under control conditions (Fig. 5F–I).
Taken together, these results indicate that the decrease in YTHDC1 contributes to increased apoptosis in BMSCs under HSD condition, which can be reversed by NfƙBiα knockdown. In conclusion, our data suggest that Ythdc1 knockdown affects the biological functions of BMSCs by modulating NfƙBiα expression, resulting in decreased cell viability, increased apoptosis rates, and increased cellular ROS production.
Effect of BMSCs Transplantation on Cardiac Remodeling Following MI
Given the therapeutic potential of BMSCs transplantation for the treatment of MI, we next tested whether BMSCs overexpression or knockdown of Ythdc1 would be differentially effective in improving cardiac morphology and function. First, we confirmed the successful establishment of MI models in rats, which were induced by ligation of the LAD after being anesthetized. Immediate ST-segment elevation on the ECG after limb elevation indicated successful establishment of the MI model. This was further confirmed by TTC staining on day 7 and Masson’s trichrome staining on day 21 (S Fig. 6).
To investigate the in vivo effects of BMSCs with Ythdc1 overexpression or knockdown in response to MI, we performed BMSCs transplantation into the injured hearts of rats with MI. The cardiac function of sham-operated or MI rats was assessed after injection of lentivirus-infected BMSCs for 4 weeks, evaluating parameters such as ejection fraction (EF), fractional shortening (FS), left LVEDd, and LVEDs. In the MI group, both EF and FS values were obviously reduced compared with the sham group, while LVEDd and LVEDs were increased. Notably, in the MI groups, transplantation of BMSCs overexpressing Ythdc1 resulted in significantly improved cardiac function compared with the transplantation of PBS or the empty vector overexpressing BMSCs. Conversely, transplantation of Ythdc1 knockdown BMSCs resulted in significantly worse cardiac function compared with the transplantation of PBS or the empty vector knockdown BMSCs. However, no significant differences were observed in the sham-operated group (Fig. 6A, B).
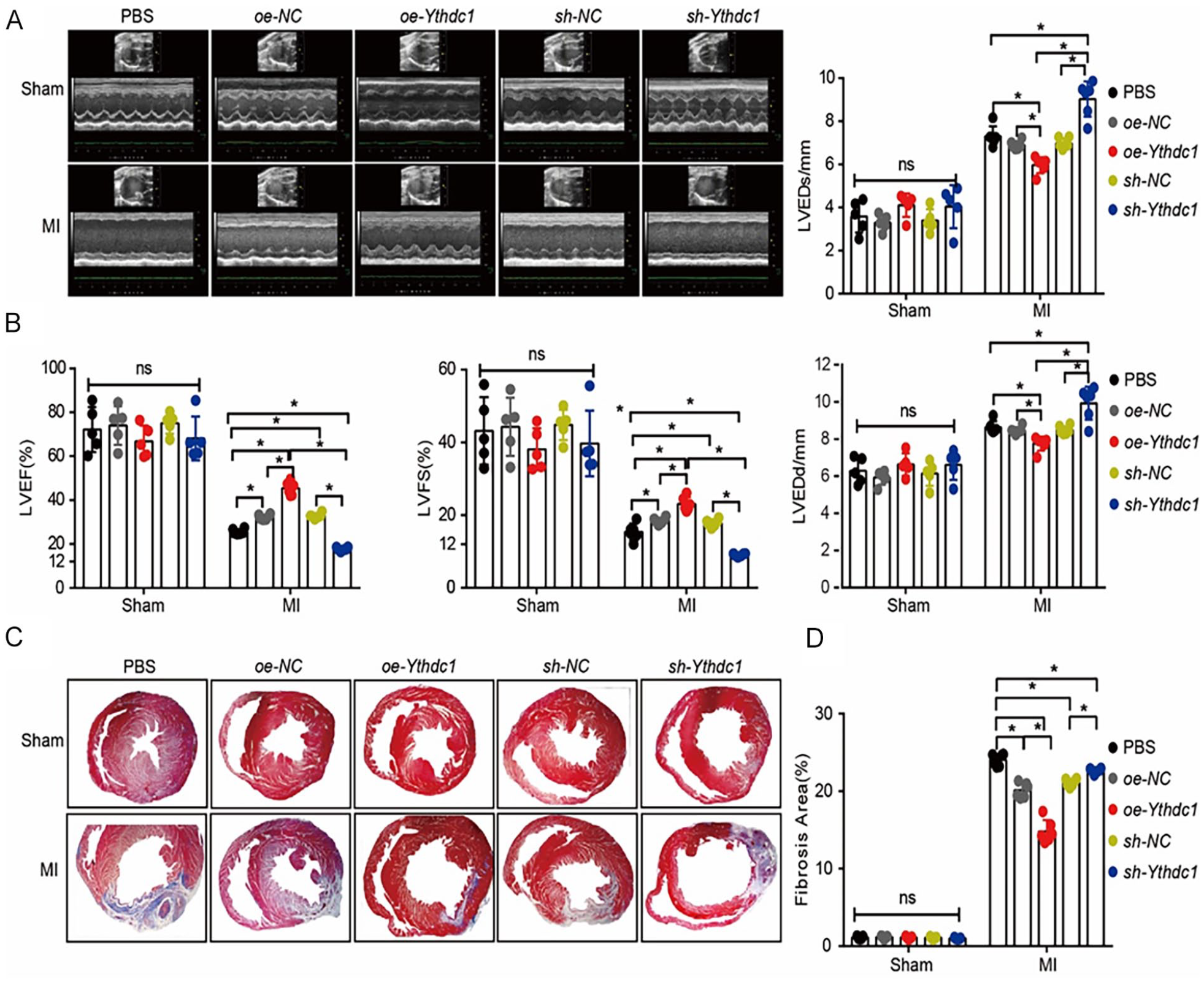
Effect of BMSCs transplantation on cardiac remodeling following MI. (A) Representative echocardiographic images of the left ventricle in M-mode on 4 weeks after LAD ligation followed by BMSCs transplantation. (B) Left ventricular ejection fraction (LVEF), left ventricular fraction shortening (LVFS), left ventricular end-diastolic diameter (LVEDd), left ventricular end-systolic diameter (LVEDs) were quantified. (C) Representative Masson’s trichrome staining of hearts 28 days after BMSCs transplantation. (D) Infarct size was calculated as the sum of the ratio of the collagen fiber relative to the total normal myocardial tissue. Data are presented as mean ± SDs (sham: n = 5/group, MI: n = 6/group) and analyzed by one-way ANOVA with Fisher’s LSD post hoc analysis. *P < 0.05.
Four weeks after MI induction, infarct size was measured and quantified by histo-morphometric analysis of Masson’s trichrome–stained myocardial sections. In the MI group, the myocardial infarct size was obviously reduced in rats receiving transplanted BMSCs compared with the rats receiving transplanted PBS. Remarkably, transplantation of BMSCs overexpressing Ythdc1 resulted in the greatest reduction in infarct size, whereas transplantation of BMSCs with Ythdc1 knockdown resulted in increased infarct size. However, no significant differences were observed in the sham group (Fig. 6C, D). Taken together, these results suggest that BMSCs overexpressing Ythdc1 exhibit superior myocardial repair potential.
Discussion
The aim of the present study was to evaluate the viability and tolerance of BMSCs overexpressing Ythdc1 in the harsh micro-environment and their improvement of cardiac function in a murine model of MI compared with PBS and BMSCs alone. Stem cell–based transplantation therapy, particularly using MSCs, has demonstrated efficacy in the treatment of MI49,50. MSCs have significant regenerative potential and hold promise for clinical applications in various diseases. MI is the most common and severe form of CVD and is the leading cause of mortality worldwide. Acute MI induces structural and functional changes in various tissues and organs, leading to cell death and posing a serious threat to the well-being of individuals 51 . Nevertheless, the hypoxic and ischemic conditions prevailing in the infarcted region resulted in a significant decrease in the survival rate of transplanted stem cells, thereby reducing the therapeutic efficacy of stem cell transplantation. Numerous efforts have been undertaken to enhance the efficacy of MSCs-based therapies for MI, including prolyl hydroxylase inhibition 52 , catalpol pretreatment 53 and the application of novel functional materials 54 , among others. Consequently, improving the survival of transplanted BMSCs is emerging as a pressing clinical need that warrants attention.
In recent years, a growing body of research has focused on epigenetics-related molecules in the context of cardiovascular and cerebrovascular disease, demonstrating promising results in the treatment of ischemic disease55–58. Among these molecules, Ythdc1 has garnered significant attention due to its potential role in various biological functions and disease progression, including cell proliferation and death59–61. Notably, Ythdc1 is the only YTH family methyl-binding protein that is located in the nucleus and is involved in the m6A recognition. By influencing the nuclear localization, stability, decay and splicing of RNA, Ythdc1 regulates a range of RNA metabolic reactions, thereby influencing disease development. Previous studies have suggested potential beneficial effects of Ythdc1 in cardiomyopathies62,63. For instance, deletion of Ythdc1 in mice was found to induce dilated cardiomyopathy, characterized by significant left ventricular enlargement, severe systolic function, reduced contractility of myocardial cell contractility and disorganized sarcomere arrangement. High-throughput sequencing has showed that Ythdc1 affects myocardial contraction through aberrant splicing of m6A-modified myosin mRNA 62 . In the context of osteosarcoma therapy, microRNA-451a has been observed to increase the stability of inositol 3-phosphate-dependent protein kinase 1 via Ythdc1-mediated m6A modification, leading to the inhibition of protein kinase B phosphorylation and promotion of osteosarcoma cell apoptosis. Furthermore, Ythdc1 has been associated with a favorable prognosis by regulating mRNA stability and suppressing tumor growth and metastasis 59 . Specifically, Ythdc1 facilitates the generation of mature miR-30d by stabilizing its precursor miR-30d, thereby inhibiting tumor growth 16 . Deletion of Ythdc1 results in increased nuclear residence time of m6A mRNA and nuclear sequestration of transcripts, hampering their translation into functional proteins in the cytoplasm11,64. Collectively, these findings highlight the significance of Ythdc1 in cell survival and apoptosis. Nevertheless, there is currently no available literature investigating whether Ythdc1 can inhibit the apoptosis in BMSCs, which could potentially play a critical role in enhancing the survival of transplanted BMSCs.
Our study has showed that Ythdc1 expression was reduced in BMSCs subjected to HSD conditions, which was associated with an increase in BMSCs apoptosis. This observation suggests that restoring Ythdc1 function could potentially alleviate cell apoptosis and enhance the survival of BMSCs. However, the precise apoptosis pathway triggered by Ythdc1 downregulation remains poorly understood. To address this knowledge gap, we performed a comprehensive analysis of cellular ROS production, cell viability, cell apoptosis and the expression of key apoptotic regulators, including Bcl-2, Bax, cleaved caspase 3. Our results confirmed that BMSCs overexpressing Ythdc1 exhibited reduced apoptosis rates and lower levels of cellular ROS production, thereby enhancing cell viability under HSD conditions. Notably, no significant differences were observed in BMSCs under control conditions. These results highlight the potential of Ythdc1 overexpression to ameliorate the adverse effects of HSD on BMSCs. However, how YTHDC1 is involved in the regulation of HSD-induced apoptosis in MSCs has not been reported.
The results of RNA-seq analysis suggested that nuclear factor-kappa-B inhibitor-alpha (NfκBiα) mRNA could be a potential target of YTHDC1 in the regulation of BMSCs apoptosis. NfκBiα is an inhibitor of the transcription factor NF-κB, which is known to play a role in various cellular processes including inflammation, cell proliferation, differentiation, and apoptosis 65 . NfƙBiα is a transcription factor inhibitor that regulates NF-ƙB activity by regulating the localization and stability of NF-ƙB 66 . Under normal conditions, NfκBiα keeps NF-κB in the cytoplasm, preventing its activation and limiting its ability to stimulate gene expression. However, under oxidative stress conditions, NfκBiα is degraded, leading to the release and translocation of NF-κB to the nucleus, where it activates target genes involved in cell survival and other processes 67 . Previous studies have shown that NfκBiα is a direct target of m6A modification in colonic epithelial cells, and its stability is regulated by METTL14, an m6A methyltransferase. This m6A modification of NfκBiα mRNA affects its stability and thereby modulates NF-κB signaling. NF-κB activation is known to promote cell survival and inhibit apoptosis, while its inhibition can induce apoptosis.
Related studies suggest that NfƙBiα is involved in regulating cell damage. As far as stem cell is concerned, NF-κB signaling pathway has been linked to several pathological processes in the apoptosis including, ischemia/reperfusion of cardiac stem cells 68 , pulmonary MSCs in mild cases of COVID-19 69 , and MSCs influence megakaryocytopoiesis in patients with immune thrombocytopenia 70 . These reports indicate an important role of NfƙBiα in stem cell apoptosis and to further investigate whether YTHDC1 exerts resistance to HSD conditions induced apoptosis by regulating NfƙBiα. To investigate the potential role of YTHDC1 in regulating NfκBiα and apoptosis, an RIPA was performed using a YTHDC1 antibody targeting a specific region of NfκBiα mRNA. The assay confirmed the binding of YTHDC1 to NfκBiα in BMSCs. In addition, it was demonstrated that knockdown of NfκBiα could increase cell viability and the expression of the anti-apoptotic protein Bcl-2, while decreasing the expression of the pro-apoptotic proteins Bax and cleaved caspase 3 under HSD conditions. This suggests that reducing NfκBiα levels counteracts the apoptosis induced by YTHDC1 knockdown.
In summary, this study provides evidence that YTHDC1 can regulate the ratio of apoptosis and ROS production by inhibiting NfκBiα during HSD conditions. However, the precise mechanism by which YTHDC1 regulates NfκBiα was not investigated in this experiment, representing a limitation. Further exploration of the factors influencing cell viability and apoptosis of BMSCs under HSD conditions will be crucial to optimize future strategies for BMSC therapy in ischemia and hypoxia-induced heart disease.
Conclusion
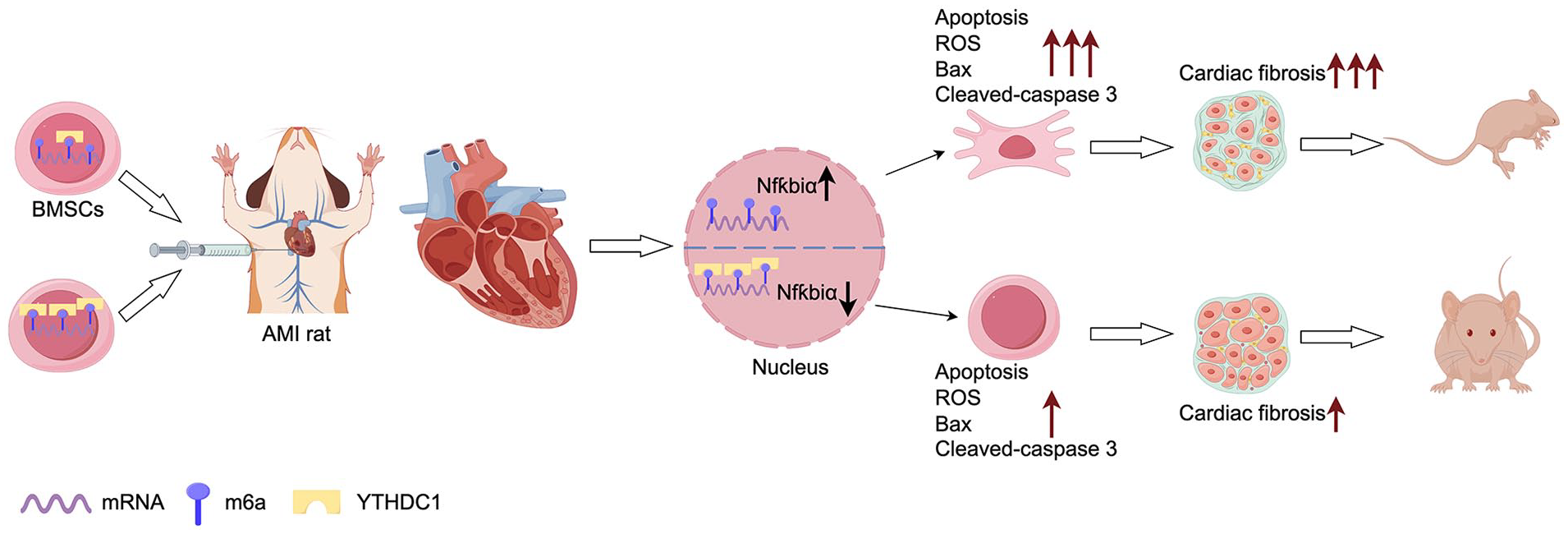
In this study, we investigated the effect of Ythdc1 expression on BMSCs under conditions simulating MI, including HSD in vitro. We observed a downregulation of YTHDC1 protein in BMSCs when subjected to HSD conditions. To explore the potential therapeutic role of Ythdc1, we overexpressed Ythdc1 in BMSCs and observed a protective effect against apoptosis, along with a reduction in cellular ROS production specifically under HSD conditions. Conversely, when Ythdc1 was knocked down in BMSCs, we also investigated the effects of Ythdc1 augmentation in rat models of MI by transplanting BMSCs overexpressing Ythdc1. Our results showed improved cardiac function, as evidenced by increased EF and FS, along with a reduction in infarct size, suggesting that Ythdc1 augmentation enhances the efficacy of BMSCs transplantation in rat models of MI. To gain insight into the underlying mechanisms, we investigated the role of NfƙBiα in mediating the anti-apoptotic effect of YTHDC1. Our data demonstrated a significant association between YTHDC1 and NfƙBiα, further supporting the involvement of this pathway in the observed effects. Overall, our study provides valuable insights into a novel approach to improve the efficacy of stem cell transplantation in the treatment of MI and highlights the potential of YTHDC1 as a therapeutic target (Fig. 7).
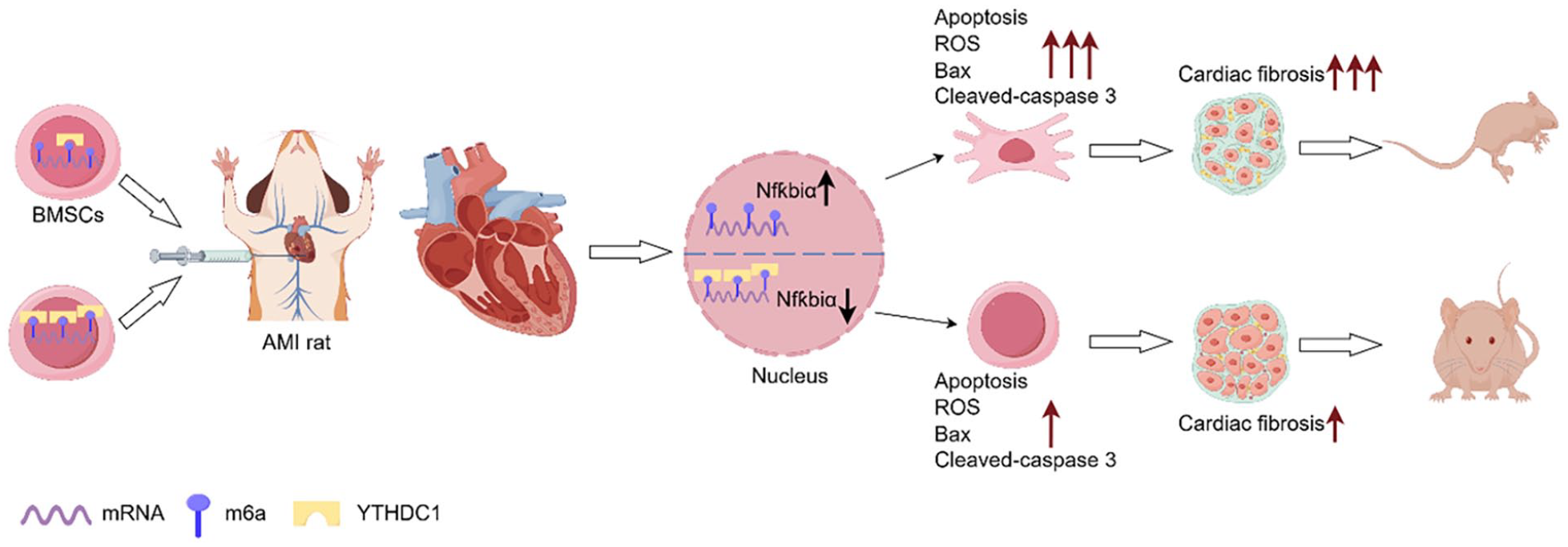
YTHDC1 regulates BMSC apoptosis via NfƙBiα inhibition, bolstering cardiac function post-MI. Element from Figdraw (<https://www.figdraw.com/>). In experimental rat models mimicking MI, the overexpression of YTHDC1 within BMSCs was found to significantly bolster cardiac function recovery and mitigate myocardial fibrosis post-transplantation. This therapeutic benefit stems from YTHDC1’s ability to suppress the expression of NfkBiα, a pivotal factor contributing to BMSC demise in the adverse milieu. Consequently, this downregulation curtails the loss of transplanted BMSCs within the ischemic-hypoxic niche, thereby amplifying their regenerative potential and enhancing the overall efficacy of BMSC-based therapy for myocardial infarction.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241290910 – Supplemental material for YTHDC1 Mitigates Apoptosis in Bone Marrow Mesenchymal Stem Cells by Inhibiting NfƙBiα and Augmenting Cardiac Function Following Myocardial Infarction
Supplemental material, sj-docx-1-cll-10.1177_09636897241290910 for YTHDC1 Mitigates Apoptosis in Bone Marrow Mesenchymal Stem Cells by Inhibiting NfƙBiα and Augmenting Cardiac Function Following Myocardial Infarction by Weiyu Han, Weidong Xiong, Weixing Sun, Weiwei Liu, Yu Zhang, Chaofu Li, Ning Gu, Youcheng Shen, Zhimei Qiu, Chaozhong Li, Yongchao Zhao and Ranzun Zhao in Cell Transplantation
Footnotes
Acknowledgements
I would like to extend my heartfelt thanks to Youyang Huang, Xinyu Xu, and Yajie Hou for their invaluable assistance and dedication during the experimental phase of this project, particularly in the realm of Hoest33342 immunofluorescence experiments. While the specific outcomes of those experiments did not ultimately find their place in the final manuscript, their contributions and efforts were instrumental in shaping the broader research direction and informing our understanding of the underlying mechanisms. Their expertise, guidance, and willingness to share their insights, even on experiments that did not directly contribute to the final version, reflect their commitment to advancing scientific knowledge and supporting the research community. I am deeply grateful for their time, energy, and the invaluable lessons learned through our collaboration, which have undoubtedly enriched my own understanding and approach to scientific inquiry.
Author Contributions
WY Han, YC Zhao, and RZ Zhao designed the study. WY Han, WD Xiong, WW Liu, Y Z, and WX Sun completed the experimental process. CF Li, Ning Gu, YC Shen, and WD Xiong participated in part of experiments. WD Xiong, CF Li, ZM Qiu, and CZ Li analyzed the data and performed statistical analysis. RZ Zhao, YZ Zhao, and WY Han wrote and revised the manuscript with contributions upon all listed authors. All authors reviewed and approved the final manuscript.
Availability of Data and Material
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethical Approval
All animal studies were approved by the Animal Care and Utilization Committee of Affiliated hospital of Zunyi Medical University On December 31, 2021, China. Ethical check number: KLLY(A)-2021-054.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants from the National Natural Science Foundation of China (81960066 and 82200290), the Basic Research Program of the Department of Science and Technology of Guizhou Province (Qiankehe Foundation-ZK [2022] General 653 and 671), and the Science and Technology Plan project Fund of Zunyi City (Zunshi Kehe HZ (2022) 341 and 366). The funding bodies played no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
