Abstract
Introduction
The aim of this study was to determine the effect of a new type of Mg-3Nd-1Gd-0.3Sr-0.2Zn-0.4Zr (abbreviated to Mg-Nd-Gd-Sr) alloy on bone marrow mesenchymal stem cell (BMSCs) function derived from SD rats.
Methods
BMSCs were first isolated and cultured in vitro using the whole bone marrow adherence method, and identified by BMSC surface biomarkers and osteogenic induction. The in vitro biological safety of the Mg-Nd-Gd-Sr alloy was studied by cytotoxicity and apoptosis experiments, and the in vitro biological functions were studied by cell adhesion and cell proliferation experiments.
Results
The results showed that high-purity BMSCs were isolated using the whole bone marrow adherence method. Flow cytometry showed high expression of CD44 and CD90, and low expression of CD31 and CD45 in the BMSCs. Osteogenic induction showed that the BMSCs differentiated into osteoblasts, and mineralized nodules were observed. The cytotoxicity of the Mg-Nd-Gd-Sr alloy to SD rat BMSCs was 0–1 grade, suggesting that the Mg-Nd-Gd-Sr alloy had no significant cytotoxic effect on SD rat BMSCs; compared with the control group, there was no significant cell apoptosis in any of the experimental groups (p > 0.05). Cell adhesion experiments showed that the number of adherent cells increased with the duration of culture with the exception of the 100% concentration group; compared with the control group, the 75% concentration group had the highest number of adherent cells at the 1st, 3rd, 5th, and 7th hours (p < 0.05). Cell proliferation experiments showed that the number of cells in all experiment groups was higher than the control group (p < 0.05) on the 1st, 3rd, 5th, and 7th days, with the highest number of cells in the 75% concentration group (p < 0.05).
Conclusion
Our data indicate that the extracts of new type of Mg-Nd-Gd-Sr alloy has no apparent cytotoxicity to BMSCs, does not affect cell apoptosis, and has good biocompatibility. Different concentrations of Mg-Nd-Gd-Sr alloy extracts promoted the adhesion and proliferation of BMSCs. The alloy had good biological functions, and is thus a promising bone repair material.
Keywords
Introduction
There are various kinds of biological materials used in orthopedics, including metal, organic polymer, and ceramic materials; however, these materials have shortcomings. Metal materials are not degradable and require removal by the second operation. In addition, elastic modulus is very different from the normal human bone cortex, thus, a stress shielding effect may occur. There is friction between metal and surrounding tissues and body fluids during long-term implantation of metal materials that can induce a micro-electrolyte reaction in which toxic metal cations are released and cause damage to human tissues. Organic polymer materials have poor biomechanical properties, are non-degradable or the degraded products are unnecessary elements for the human body and likely induce an inflammatory response, 1 which limits their application. Ceramic materials are rarely used in bone repair due to poor toughness, high brittleness, and unfavorable biomechanical properties. In view of these findings, there is an urgent need to develop a new type of biomedical material with good biocompatibility, degradability, non-toxic reactivity, and good mechanical properties.
Magnesium alloys have become a promising bone repair material due to multiple advantages. Magnesium alloys are degradable and absorbable, thus, avoiding the need for a secondary surgical procedure. 2 The elastic modulus of magnesium alloys is closer to that of human cortical bone, and the stress shielding effect can be largely reduced.3–5 Magnesium alloys have good biocompatibility and biomechanical properties, and promote cell proliferation and adhesion under appropriate concentration. Specifically, magnesium alloys promote osteogenic induction and new bone formation.6,7 In addition, magnesium alloys have good specific strength and low specific stiffness. 8 Although magnesium alloys have the above advantages and satisfy the criteria for ideal bone repair materials, the degradation rate of magnesium alloys in vivo and in vitro is fast, and the degradation and bone repair rates cannot be well-matched. Additionally, during the degradation process, magnesium alloys release gas that prevents the adhesion between surrounding tissues and implanted materials. There are no magnesium alloys that have been used in clinical practice to date. To adjust the rate of degradation and absorption, enhance mechanical properties, and optimize biocompatibility, we prepared a new type of Mg-3Nd-1Gd-0.3Sr-0.2Zn-0.4Zr (abbreviated to Mg-Nd-Gd-Sr) alloy on the basis of our previous work using a gravity casting method and adding appropriate proportions of elements, including Gd, Nd, Zr, Sr, and Zn. 9
As a bone repair material, Mg-Nd-Gd-Sr alloy is in direct contact with human tissues and body fluids after implantation. Medullary mesenchymal stem cells are the key cells for osteogenesis, have multidirectional differentiation potential,10,11 and can be induced into osteoblasts. 12 Medullary mesenchymal stem cells are the best cells for evaluating an Mg-Nd-Gd-Sr alloy as bone repair materials in vitro.13–15 Therefore, we cultured BMSCs in medium containing the Mg-Nd-Gd-Sr alloy extract, then used cytotoxicity and apoptosis experiments to study the in vitro biological safety of the Mg-Nd-Gd-Sr alloy. We also studied the in vitro biological functions of the Mg-Nd-Gd-Sr alloy by cell adhesion and cell proliferation experiments. Our study provides experimental data for the clinical application of a new type of magnesium alloy.
Materials and methods
Materials
Chemical composition of the Mg-Nd-Gd-Sr alloy.

The Sprague-Dawley (SD) rat were provided by Chinese Academy of Science Type Culture Collection (China).
Methods
This study is a randomized controlled cell experiment in vitro.
Preparation of the magnesium alloy extracts
The Mg-Nd-Gd-Sr alloy extracts were prepared according to the ISO10993-12 standard. 16 The experimental Mg-Nd-Gd-Sr alloy sample was put in a 15-mL centrifuge tube, and SD rat BMSC-specific medium was used for the extracts. SD rat-specific culture medium was added at a 1.25:1 ratio of the Mg-Nd-Gd-Sr alloy surface area (cm2) to the medium (mL), and incubated at 37°C in 5% CO2 for 24 h to obtain the raw extracts. After filtering through a microporous membrane, the medium was diluted into 25%, 50%, 75%, and 100% extract concentrations.
Isolation, culture and identification of BMSCs from SD rats
Isolation and culture of BMSCs from SD rats
The whole bone marrow adherence method was used to isolate and culture primary BMSCs from SD rats in vitro. SD rats were sacrificed by air embolism, disinfected by immersion in 75% alcohol, and the femur and tibia were separated aseptically on an ultra-clean bench. The femur and tibia metaphyses were excised with ophthalmic scissors. SD rat BMSC-specific medium (10 mL) was added to another sterile petri dish. The bone marrow cavity was repeatedly rinsed with the medium. The medium was then collected to a 10-mL centrifuge tube and centrifuged at 1000 r/min at 4°C for 5 min. The pellet was resuspended and inoculated into a culture flask, and the medium was changed after 48 h. Cell morphology was observed under an inverted microscope. The medium was changed every 2 days. The isolated primary cells were cultured until reaching 85% confluence, then passaged at a 1:2 ratio.
Identification of BMSCs from SD rats
The third-generation cells were collected. The cells were washed with PBS, digested with 0.25% trypsin, and centrifuged (1000 r/min) at 4°C for 5 min. The supernatants were discarded, and the cells were resuspended in PBS and centrifuged at 1000 r/min for 5 min. The supernatants were discarded, then the cells were thrice-washed with PBS and resuspended. The cell solution (1×105 cells) was added to 5 PE tubes. One tube was set as a blank control in which an equal amount of PBS was added; 10 μL of PE-labeled CD90, PE-labeled CD44, APC-labeled CD45, or FITC-labeled CD31 was added to one of the four remaining PE tubes. After a 30-min culture, the unlabeled cells were washed with PBS, and the expression of corresponding antigens was tested by flow cytometry.
The third-generation cells were collected and inoculated into 6-well plates at 1×104/mL. Each plate had three induction and three control wells. After the cells reached 90% confluence, 1 mL of osteogenic induction medium was added to the induction wells, and 1 mL of medium was added to the control wells. Cell morphology was observed after 24 h and the medium was changed every 2 days. The cells were stained with alkaline phosphatase after 14 days of osteogenic induction and stained with Alizarin red after 21 days of osteogenic induction.
Cytotoxicity experiments
Classification of the cytotoxicity.

Cell apoptosis experiments
The Annexin V-FITC/PI assay was used to study the effect of Mg-Nd-Gd-Sr alloy on SD rat BMSC apoptosis. There were five groups, including the control group that did not contain Mg-Nd-Gd-Sr alloy extract and four experimental groups that contained 25%, 50%, 75%, or 100% of the Mg-Nd-Gd-Sr alloy extracts. Each group had six replicate wells. The 3rd-generation cells in good growth condition were digested with 0.25% trypsin, inoculated into 48-well plates at 1×104/mL, and cultured for 24 h. The experimental group media were replaced by medium containing 25%, 50%, 75%, or 100% of the Mg-Nd-Gd-Sr alloy extracts. The medium was replaced every 2 days. After a 5-day culture, the cells were digested with 0.25% trypsin, and centrifuged at 1800 r/min for 5 min. The supernatants were discarded. The cells were washed with PBS and resuspended in elution buffer, then aliquoted into 5 PE tubes with 1×105 cells each tube. Annexin-V (5 μL) was added to each tube, mixed evenly, and incubated at 37°Cin 5% CO2 for 15 min. PI (2.5 μL) was then added to the above tubes and mixed evenly. After a 10-min incubation, 200 μL of elution buffer was added. Cell apoptosis was detected by flow cytometry.
Cell adhesion experiment
The effect of the Mg-Nd-Gd-Sr alloy on BMSC adhesion from SD rats was evaluated using a CCK-8 assay. The 3rd-generation cells in good growth condition were inoculated in four 96-well plates at 1×104/mL. Each plate included the following five groups: control group containing no Mg-Nd-Gd-Sr alloy extract; and four experimental groups containing 25%, 50%, 75%, or 100% of the Mg-Nd-Gd-Sr alloy extracts. Each group had six replicate wells. The cells were cultured at 37°C in 5% CO2. Medium and non-adherent cells were aspirated at the 1st, 3rd, 5th, and 7th days and 10 μL of CCK-8 solution was added and incubated for 2 h in a dark environment. The OD value at 450 nm was measured.
Cell proliferation experiment
The CCK-8 assay was used to study the effect of Mg-Nd-Gd-Sr alloy on the proliferation of BMSCs from SD rats. The 3rd-generation cells in good growth condition were inoculated in 4 of 96-well plates at 1×104/mL. Each plate included the following five groups: control group containing no Mg-Nd-Gd-Sr alloy extract; and four experimental groups containing 25%, 50%, 75%, or 100% of the Mg-Nd-Gd-Sr alloy extracts. Each group had six replicate wells. After a 24-h culture, the media in the experimental groups were changed with the media containing 25%, 50%, 75%, or 100% of the Mg-Nd-Gd-Sr alloy extracts. The medium in the control group was changed with an equal amount of SD rat-specific culture medium. The medium was replaced every 2 days. After 1, 3, 5, and 7 days of culture, the supernatants were removed and 20 μL of CCK-8 solution was added and cultured for an additional 2 h. The OD values at 570 nm were measured at the above time points.
Statistical analysis
SPSS18.0 (SPSS, Inc., Chicago, IL, USA) was used to analyze the data. The differences between the groups were compared using one-way analysis of variance (ANOVA). Statistical significance was defined as P < .05.
Results
Isolation, culture, and identification of SD rat BMSCs
Morphology
A large number of floating red blood and hematopoietic stem cells were observed immediately after inoculation. The cell composition was complex. After a 48-h culture and replacement of the medium, the number of floating cells was reduced; and a small number of cells were attached to the bottom of the dish in the shape of a spindle (Figure 1(a)). After 3 days, a small number of non-specific cells were noted (Figure 1(b)), and after 5 days, only a small number of floating cells remained (Figure 1(c)). After 10 days, colonies of tightly connected, spindle-shaped attached cells were noted (Figure 1(d)). The morphologies of bone marrow mesenchymal stem cells isolated from SD rats and cultured after (a) 2 days, (b) 3 days, (c) 5 days, and (d) 10 days (inverted phase contrast microscope, ×10).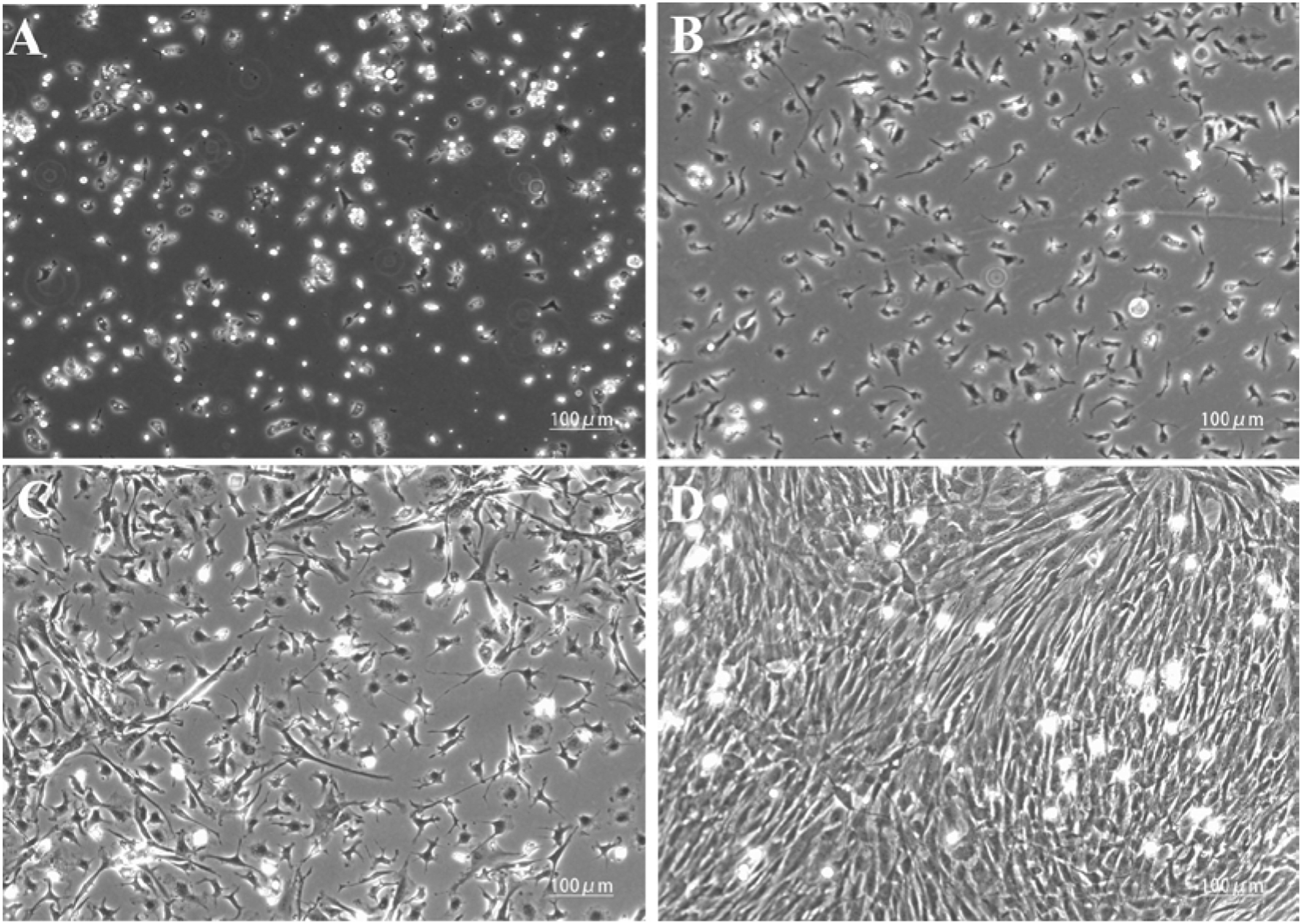
The passaged cells grew rapidly and were fully attached within 24 h. The passaged cells had a similar spindle shape, and were arranged spirally or radially (Figure 2). The morphology of the third generation of bone marrow mesenchymal stem cells (inverted phase contrast microscope, ×10).
Identification
Flow cytometry of the 3rd-generation BMSCs (Figure 3) showed that the levels of CD90 and CD44 expression were 99.8% and 99.5%, and the levels of CD45 and CD31 expression were 0.4% and 0.004%, respectively, indicating that the attached cells isolated from the bone marrow of SD rats were BMSCs. The levels of (a) CD90, (b) CD44, (c) CD45 and (d) CD31 expression in bone marrow mesenchymal stem cells from SD rats.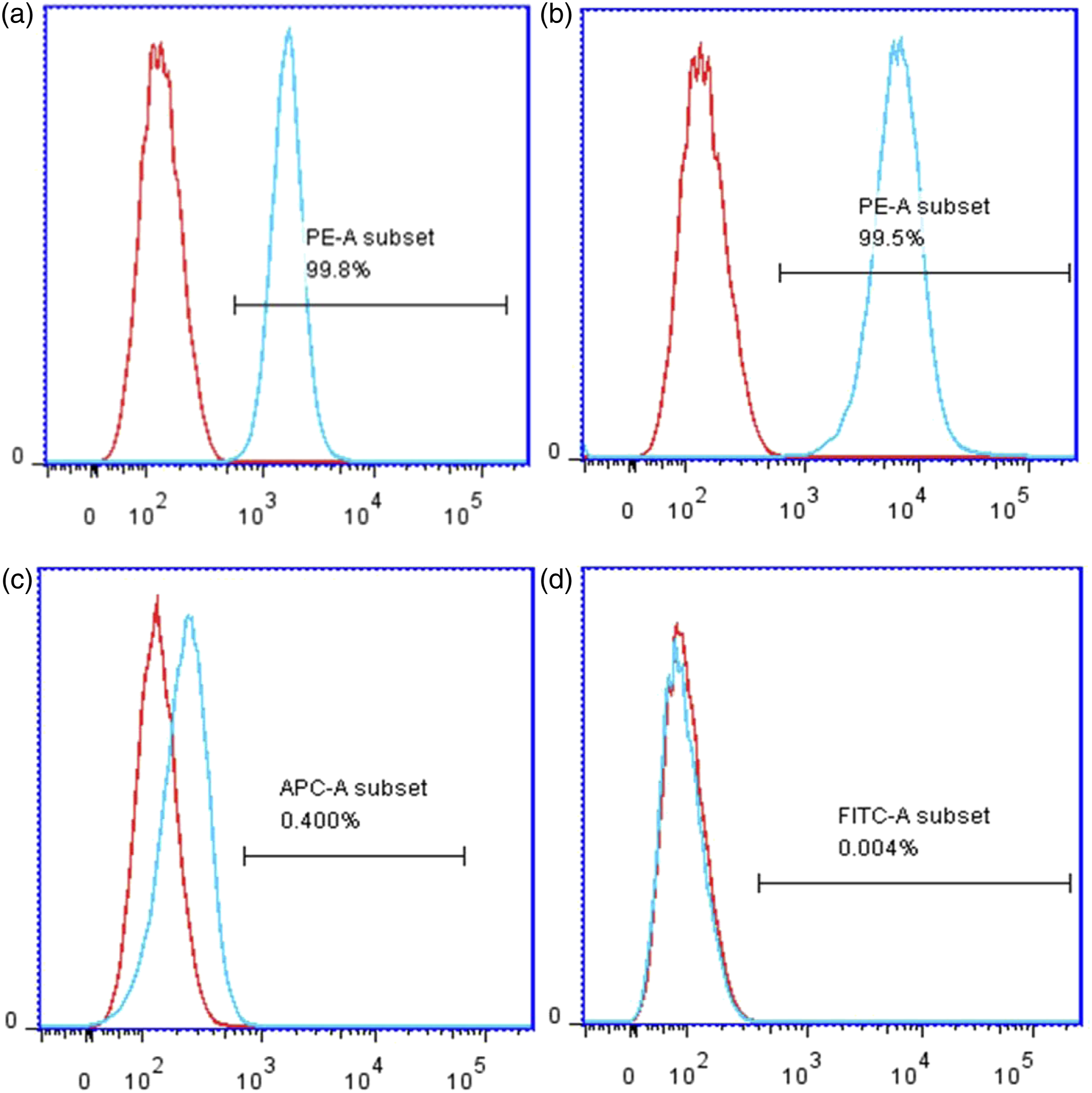
Alkaline phosphatase staining
After a 14-day osteogenic induction, the active part of alkaline phosphatase was blue-stained and the nucleus was red-stained (Figure 4). The result of alkaline phosphatase staining in bone marrow mesenchymal stem cells from SD rat (fluorescence microscopy, ×10).
Alizarin red staining
After a 21-day osteogenic induction, mineralized nodules were visualized under an inverted microscope. Alizarin red staining was positive (Figure 5). The result of alizarin red staining in bone marrow mesenchymal stem cells from SD rats after a 21-day osteogenic induction (inverted phase contrast microscope, ×10).
Cytotoxicity experiments
Cell morphology
The SD rat BMSCs were co-cultured with different concentrations of Mg-Nd-Gd-Sr alloy extracts, and the changes in cell morphology were observed on culture days 1, 3, 5, and 7. On the 1st day, there was a small number of spindle-shaped cells and nearly no dead cells (Figure 6). On the 3rd day, the cell density and number increased significantly and cells were spindle-shaped. On the 5th day, the cells were tightly connected, and on the 7th day, the spindle-shaped cells were arranged like a whirlpool or shoal of fish and had a normal morphology (Figure 7). There was no significant change in cell morphology between the experimental and control groups on culture days 1, 3, 5, and 7. The cell morphology in different groups on the 1st day: (a) Control group; (b) 25% extracts; (c) 50% extracts; (d) 75% extracts; (e) 100% extracts (inverted phase contrast microscope, ×20). The cell morphology in different groups on the 7st day: (a) Control group; (b) 25% extracts; (c) 50% extracts; (d) 75% extracts; (e) 100% extracts (inverted phase contrast microscope, ×10).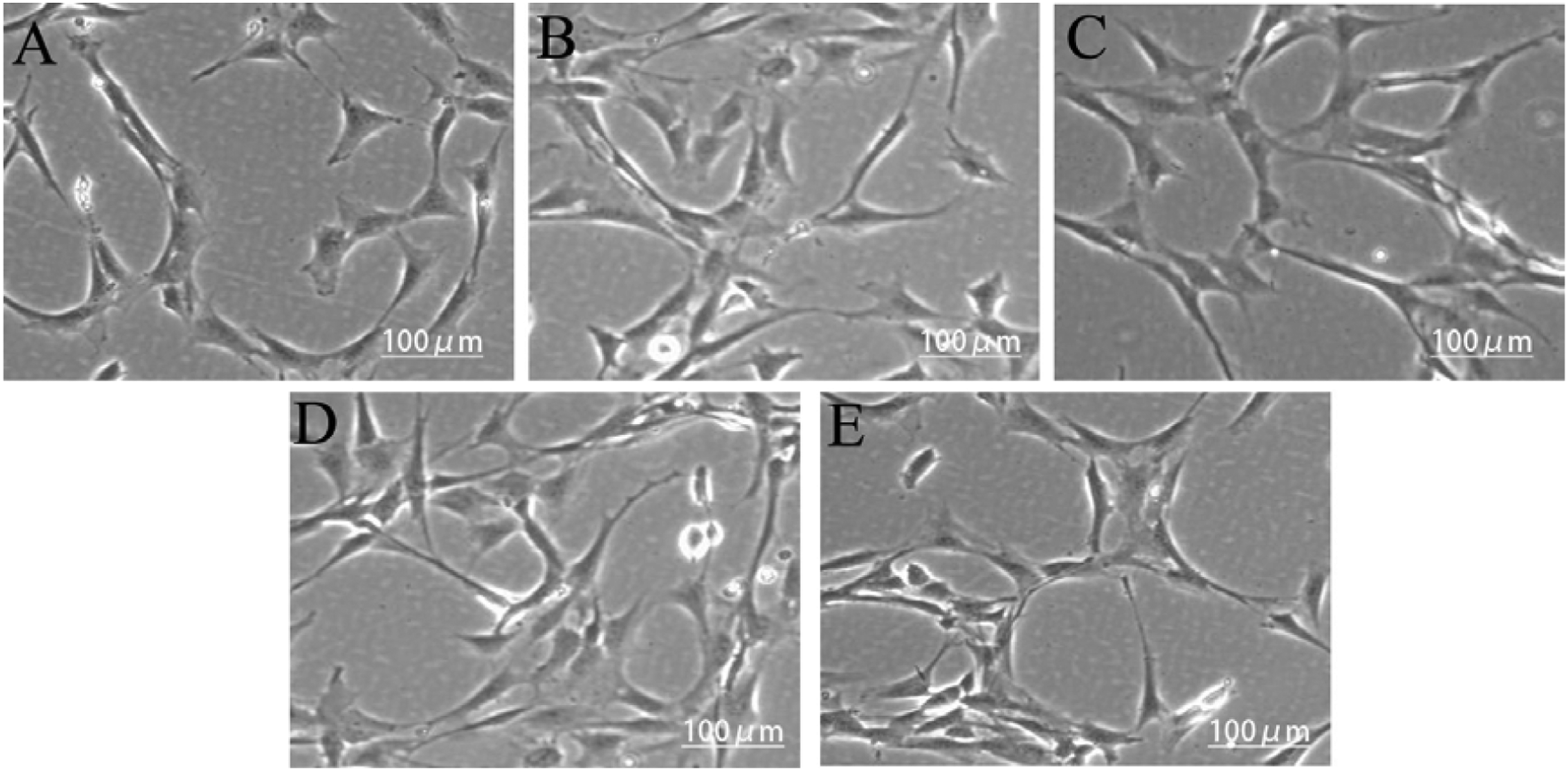

Cytotoxicity experiments
SD rat BMSCs were cultured in medium containing 25%, 50%, 75%, or 100% Mg-Nd-Gd-Sr alloy extracts for 1, 3, 5, and 7 days. The CCK-8 assay showed that the number of rat BMSCs was highest in the 75% extract (Figure 8). (a) The optical density (OD) and (b) relative cell growth rate (RGR) in different groups.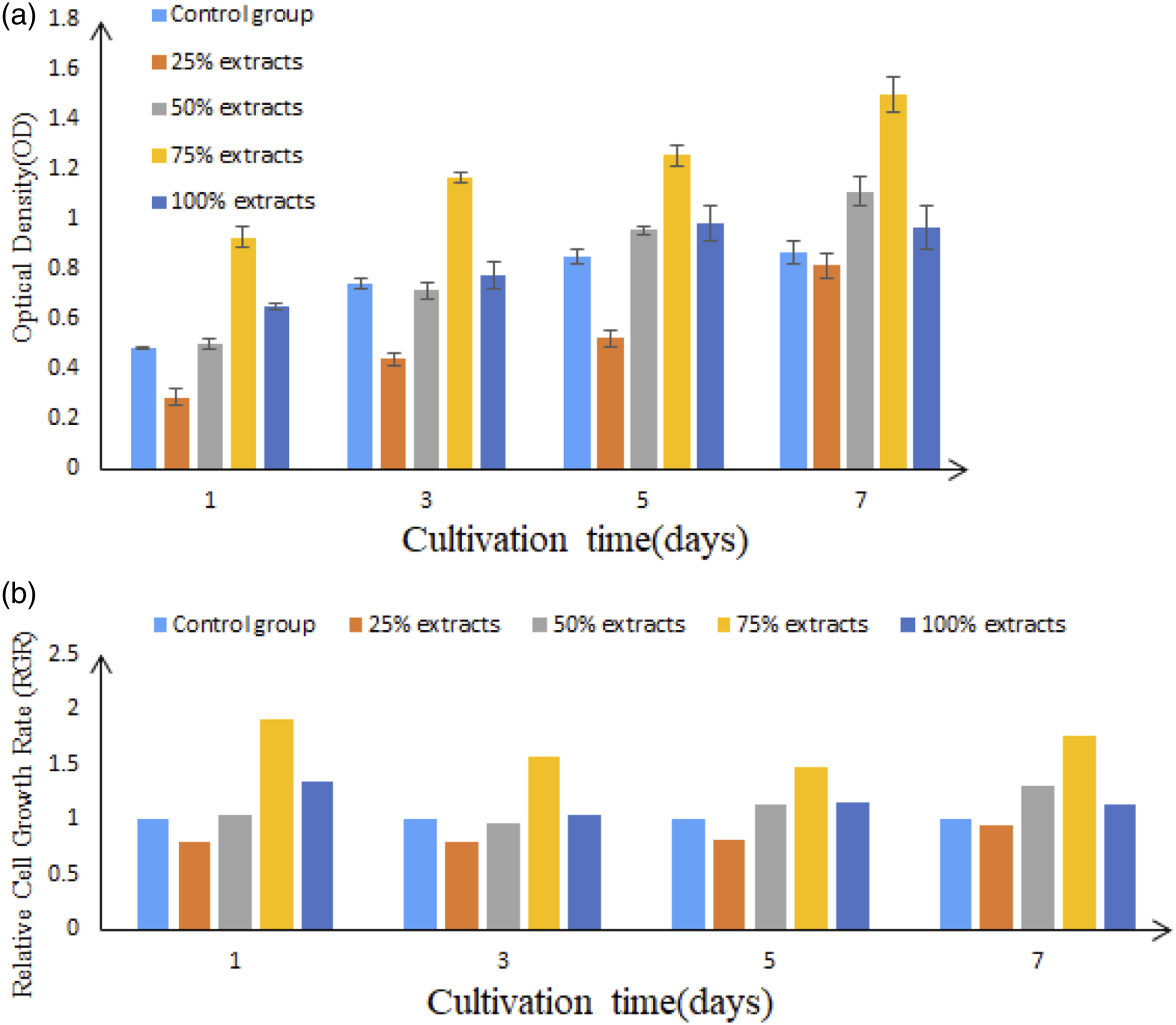
The cytotoxicity of the Mg-Nd-Gd-Sr alloy to SD rat BMSCs was 0–1 grade, suggesting that the Mg-Nd-Gd-Sr alloy had no significant cytotoxic effect on SD rat BMSCs.
Cell apoptosis experiments
The effect of Mg-Nd-Gd-Sr alloy on SD rat BMSC apoptosis was tested by flow cytometry (Figure 9). Cell apoptosis rate in different groups on the 5st day: (a) Control group; (b) 25% extracts; (c) 50% extracts; (d) 75% extracts; (e) 100% extracts.
Compared with the control group, the 25%, 50%, 75%, and 100% concentration groups had no significant differences in the cell apoptosis rate (p > 0.05).
Cell adhesion experiments
The CCK-8 assay was used to evaluate the effect of the Mg-Nd-Gd-Sr alloy on BMSC adhesion. SD rat BMSCs were co-cultured with 25%, 50%, 75%, or 100% Mg-Nd-Gd-Sr alloy extracts for 1, 3, 5 and 7 h, the OD values of the experimental and control groups are shown in Figure 10. The number of adherent cells (OD value) in different groups.
With the exception of the 100% group, the number of adherent cells increased with the duration of culture. Compared with the control group at various time points, the 75% group had the highest number of adherent cells at the 1st, 3rd, 5th, and 7th h (p < .05). The number of adherent cells was higher in the 25% and 50% concentration groups than the control group at the 1st, 3rd, and 5th h (p < .05). The number of adherent cells decreased in the 25%, 50%, and 100% groups compared with the control group at the 7th h (p < .05). SD rat BMSCs had the strongest adhesion in the 75% group; the higher concentration did not facilitate cell adhesion.
Cell proliferation experiments
The CCK-8 assay was used to evaluate the effect of the Mg-Nd-Gd-Sr alloy on the proliferation of SD rat BMSCs. The OD values in various groups after 1, 3, 5, and 7 days of culture with different concentrations of the Mg-Nd-Gd-Sr alloy extracts are shown in Figure 11. Cell proliferation expressed as absorbance values in different groups after 1, 3, 5 and 7 days incubation in osteogenic medium (*p < .05).
The OD values increased as the duration of culture increased. The number of cells in all experimental groups was higher than the control group on the 1st, 3rd, 5th, and 7th days, with the highest number in the 75% concentration group (p < .05). The Mg-Nd-Gd-Sr alloy extracts promoted the proliferation of SD rat BMSCs; the 75% concentration yielded the greatest effect.
Discussion
Bone defect is a difficult disease in orthopedics, and there is no satisfactory repair method at present. Bone repair, the process of bone formation, is regulated by associated transcription factors and osteogenesis-related genes. BMSCs have multi-directional differentiation potential and are the key cells for osteogenic formation. 11 BMSCs can be directionally induced to differentiate into osteoblasts, 12 and are considered as the best tool for evaluating bone repair materials in vitro.13–15 In the current study, we used the whole bone marrow adherence method to isolate BMSCs from SD rats. After a 48-h culture, attached spindle-shaped cells were observed, and after an 8–10 days culture, the attached cells were colonized and tightly connected. When passaged to the 3rd generation, the BMSCs exhibited a unique morphology, high purity, and good activity. BMSCs usually express surface markers, including CD90, CD44, CD29, CD105, and SH3, but not CD45, CD31, and CD14. 17 We used flow cytometry to detect surface CD molecules in BMSCs, and showed that CD44 and CD90 were positive, while CD45 and CD31 were negative. We induced the well-growing 3rd-generation BMSCs to differentiate into osteoblasts, and identified the characteristics of osteoblasts, such as type I collagen, alkaline phosphatase, and mineralized nodules.18–20 Alizarin red staining revealed positive mineralized nodules, and alkaline phosphatase staining was also positive, suggesting successful osteoblast differentiation of BMSCs. We further co-cultured BMSCs derived from SD rats with the Mg-Nd-Gd-Sr alloy extracts and detected the cytotoxicity and apoptosis-related indicators. The Mg-Nd-Gd-Sr alloy extracts did not impose a significant impact on BMSC apoptosis, had no obvious cytotoxicity, and displayed good biocompatibility to BMSCs derived from SD rats.
Ideal bone repair materials should have good biocompatibility, be non-toxic after implantation, possess mechanical properties and elastic modulus matched or close to bone tissue, and give rise to sufficient strength. Owing to the good biomechanical properties, stiffness and strength, as well as elastic modulus close to human bone, make magnesium alloy better meet clinical needs. Moreover, a second surgical procedure to remove the implant is not necessary with magnesium alloy as a medical implant due to its unique degradability. The Mg-Nd-Gd-Sr alloy has excellent biocompatibility. Rare earth elements can significantly increase the corrosion resistance of magnesium alloy. Removing rare earth impurities and forming rare earth oxides can increase membrane stability. Rare earth elements can also bind to the surface hydroxides, and interact with chloride ions to weaken its erosion to magnesium alloy. Tsao 21 observed that the addition of neodymium (Nd) to amorphous Mg-Ni alloy immersed in 3% NaCl solution not only increased the oxygen species but also greatly inhibited the infiltration of harmful anions (Cl-). Gadolinium ions (Gd3+) in the Mg-Gd alloy are incorporated into the hydroxide layer by replacing Mg2+ to improve corrosion resistance. Zinc ions and strontium ions are not only trace elements in the human body and participate in normal metabolism, but also regulate the expression of early osteogenic differentiation markers, such as the COL-I and ALP genes. The addition of zinc ions enhances the ductility and plasticity of magnesium alloy, thus the alloy can form complex shapes and contours to fit anatomic structures. 22 Strontium ions promote the expression of late osteoblast differentiation biomarkers, such as osteopontin and osteocalcin, induce the formation of mineralized nodules, further improve the corrosion resistance of magnesium alloy in a physiologic environment, and exhibit in vitro biocompatibility.23–25 Zirconium is a non-toxic and -carcinogenic element. The addition of zirconium can effectively refine magnesium alloy. It is usually used in combination with zinc, calcium and rare earth elements to improve the corrosion resistance of magnesium alloy. 26
In this study, the in vitro biological safety of the Mg-Nd-Gd-Sr alloy was studied by cytotoxicity and apoptosis experiments; and the in vitro biological functions were studied by cell adhesion and cell proliferation experiments. Biosafety of an implant can be well reflected in an in vitro cytotoxicity test, as the weaker the cytotoxicity is in vitro, the weaker the cytotoxicity is in vivo.27–29 CCK-8 was used for cytotoxicity detection in BMSCs that were cultured with 25%, 50%, 75%, and 100% Mg-Nd-Gd-Sr alloy extracts for 1, 3, 5, and 7 days. The OD value of BMSC s was significantly increased after 1, 3, 5, and 7 days of culture with different Mg-Nd-Gd-Sr alloy extracts. Results from the cytotoxicity test indicate that the Mg-Nd-Gd-Sr alloy extract has no toxic effect on BMSCs regardless of its concentration, and the Mg-Nd-Gd-Sr alloy extract within a certain concentration range can promote the proliferation of BMSCs. According to the ISO10993 standard, 30 the RGR of BMSCs was over 100% in different Mg-Nd-Gd-Sr alloy extract groups. This indicates that the Mg-Nd-Gd-Sr alloy extract has no obvious toxic effect on BMSCs, but instead promotes the proliferation of MC3T3-E1 cells. Meanwhile, the Annexin V-FITC/PI assay was used to detect cell apoptosis by flow cytometry, thereby evaluating the influence of Mg-Nd-Gd-Sr alloy extracts on BMSC apoptosis. On the 1st and 3rd days of cell culture, there was no obvious apoptosis in each experimental group compared with the control group. On the 5th day of cell culture, the apoptotic rate was significantly increased in the 50%, 75%, and 100% extract groups compared with the control group. However, the Mg-Nd-Gd-Sr alloy extract at an excessive concentration or in poor conditions may have a certain apoptosis-inducing effect on the cells.
The biological function of implant materials is also evaluated based on their effects on cell adhesion and cell proliferation. Cell adhesion affects the fate of BMSCs, and the ability of cell proliferation, differentiation, and long-term survival. It is important to understand the effect of bone repair materials on cell adhesion. 31 The number of adherent cells in the experimental groups increased significantly in an approximate dose-dependent manner compared to the control group. The 75% concentration group had the highest increase of adherent cells, which may result from the release of magnesium ions from the alloy. Previous studies have shown that an appropriate concentration of magnesium ions is conducive to cell adhesion. 32 The experimental group also had a higher cell number than the control group at various time points, with the highest number in the 75% concentration group, suggesting that the Mg-Nd-Gd-Sr alloy extracts promoted the proliferation of BMSCs derived from SD rats. The reason why the 75% concentration had the greatest effect may be that an appropriate amount of magnesium ions can be released at this concentration to promote proliferation.33,34 In addition, the Mg-Nd-Gd-Sr alloy also contained zinc and zirconium ions. Zinc ions are involved in the early metabolism of cells and promote cell proliferation, 35 while zirconium ions form ZrO2, which can create a more suitable environment for cell growth and adhesion. 36
However, the effect of magnesium alloy on BMSCS osteogenic differentiation and its specific mechanism were not further discussed in this study. In addition, the research object of this study is rats, which is still far from the clinical application of magnesium alloy.
Conclusion
In summary, the new type of Mg-Nd-Gd-Sr alloy has no apparent cytotoxicity to BMSCs, does not affect cell apoptosis, and has good biocompatibility. Different concentrations of Mg-Nd-Gd-Sr alloy extracts promoted the adhesion and proliferation of BMSCs. The alloy had good biological functions, and is thus a promising bone repair material.
Footnotes
Acknowledgements
This work was supported by The First Hospital of Lanzhou University and The First Clinical Medical College of Lanzhou University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from Ethics Committee of The First Hospital of Lanzhou University (LDYYLL2020-40).
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (No.: 81960398), Natural Science Foundation of Gansu Province (No.: 20JR5RA369), Scientific research projects of central universities (No.: 31920200021) and Scientific Research Projects of Gansu Province Health Industry (No.: GSWSKY 2021 - 009).
