Abstract
Hematological toxicity is a severe adverse event (AE) in anti-CD19 chimeric antigen receptor (CAR) T cell therapy for relapsed/refractory (R/R) diffuse large B-cell lymphoma (DLBCL). However, the pathophysiological mechanism underlying prolonged cytopenia and the relationship between persistent cytopenia, efficacy, and AEs after anti-CD19 CAR T cell therapy are unknown. Therefore, this study explored whether persistent cytopenia after anti-CD19 CAR T cell therapy in patients with R/R DLBCL can predict therapeutic efficacy and AEs. Thirty-eight patients with R/R DLBCL were enrolled in an anti-CD19 CAR T cell therapy clinical trial. Patients received lymphodepleting chemotherapy with fludarabine and cyclophosphamide before CAR T cell therapy. The degree and duration of cytopenia, clinical response, proportion of CAR T cells, interleukin-6 (IL-6) levels, AEs, and follow-up were observed after therapy. Grades 3–4 persistent cytopenia occurred in 14 patients with R/R DLBCL, who recovered 8–18 weeks after CAR T cell infusion. These patients achieved an objective response rate (ORR) for anti-CD19 CAR T cell therapy. In patients who achieved ORR, the incidence of Grades 3–4 persistent cytopenia was higher in patients with a high tumor load than in those without a high tumor load. The mean peaks of IL-6 and anti-CD19 CAR T cells and the cytokine release syndrome grade in patients with Grades 3–4 persistent cytopenia were higher than those in patients without persistent cytopenia. Anti-CD19 CAR T cells were observed 21 and 28 days after infusion, and patients had Grades 3–4 persistent cytopenia. Progression-free and overall survival were higher in patients with Grades 3–4 persistent cytopenia than in those without cytopenia. Therefore, persistent cytopenia after anti-CD19 CAR T cell therapy in patients with R/R DLBCL can predict therapeutic efficacy and AEs, allowing clinicians to determine the efficiency of CD-19 CAR T cell therapy and the associated AEs.
Introduction
Diffuse large B-cell lymphoma (DLBCL) is a common non-Hodgkin lymphoma. Combination chemotherapy based on rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone achieves a high objective response rate (ORR) in DLBCL; however, many patients develop relapsed/refractory (R/R) DLBCL after standard combination chemotherapy1–3. Anti-CD19 chimeric antigen receptor (CAR) T cell therapy is an effective salvage therapy for R/R DLBCL4–7. However, the use of anti-CD19 CAR T cell therapy is hampered by adverse events (AEs). These AEs include cytokine release syndrome (CRS), immune effector cell-associated neurotoxic syndrome (ICANS), hematological toxicity, and B cell aplasia8–14.
Hematological toxicity is associated with secondary infection after anti-CD19 CAR T cell therapy. In the long-term activity and safety outcomes of the ZUMA-1 study, less than 20% of patients had Grade 3 or worse cytopenia 3 months after therapy 4 . None of these patients experienced severe clinical sequelae owing to persistent cytopenia15–17. Risk factors associated with persistent cytopenia include CRS/ICANS grade, baseline cytopenia, prior allogeneic stem cell transplantation, and higher peak C-reactive protein or ferritin levels 18 . However, the pathophysiological mechanism underlying prolonged cytopenia remains unclear, and whether persistent cytopenia can be explained by lymphodepleting chemotherapy with fludarabine or cyclophosphamide alone is unknown. Furthermore, the relationship between persistent cytopenia, therapeutic efficacy, and AEs after anti-CD19 CAR T cell therapy is unknown. Therefore, this study explored whether persistent cytopenia after anti-CD19 CAR T cell therapy in patients with R/R DLBCL can be used to predict therapeutic efficacy and AEs.
Materials and Methods
Research Ethics and Patient Consent
This study was approved by the Medical Ethics Committee of Tianjin First Center Hospital (Tianjin, China, approval nos. 2015002X and 2018N105KY). Informed consent was obtained from all participants, and all patients agreed to participate in the clinical trials. Patients agreed to use their data for the study. All data and materials were obtained in accordance with the Declaration of Helsinki and conformed to relevant aspects of the ARRIVE guidelines. This clinical trial was registered at http://www.chictr.org.cn/index.aspx as ChiCTR-ONN-16009862 and ChiCTR1800019622.
Medical History of Patients Enrolled in This Study
Thirty-eight patients with R/R DLBCL were admitted to our hospital and enrolled in the anti-CD19 CAR T cell therapy clinical trial (ChiCTR-ONN-16009862 and ChiCTR1800018059) between January 2017 and May 2021. All patients were diagnosed with R/R DLBCL and had undergone hematopoietic stem cell transplantation before or after the study. Follow-up was performed from the day of CAR T cell infusion to the cutoff date or death. The cutoff date for the study was June 30, 2022.
Anti-CD19 CAR T Cell Therapy
The lentiviral vector encoding the humanized CD19 CAR constructs used in this study was provided by GenBase Biotechnology (Shanghai, China, patent owner of CD19 CAR). Before anti-CD19 CAR T cell therapy, all patients received lymphodepleting chemotherapy with fludarabine (30 mg/m2) and cyclophosphamide (400 mg/m2) from day −4 to day −2. All 38 patients with R/R DLBCL received autologous peripheral blood mononuclear cells as anti-CD19 CAR T cell therapy. After 12–15 days of cell culture in vitro, the transduction efficiency of anti-CD19 CAR T cells was analyzed using flow cytometry (FCM). Anti-CD19 CAR T cells were infused on day 0 (2 × 106 cells/kg) into all patients with R/R DLBCL.
Clinical Response Criteria
From the date of anti-CD19 CAR T cell infusion, follow-up was performed until death. One and 2 months after anti-CD19 CAR T cell therapy, the therapeutic efficacy was evaluated in all 38 patients with R/R DLBCL. Responses to anti-CD19 CAR T cell therapy were evaluated using computed tomography (CT) or positron emission tomography-CT. A high tumor load was defined as having at least one tumor with a maximum diameter of ≥7.5 cm19. Disease status was defined as complete response (CR), partial remission (PR), stable disease (SD), or disease progression (PD) according to the Lugano Revised Criteria for Response Assessment 19 . Progression-free survival (PFS) and overall survival (OS) were observed after anti-CD19 CAR T cell therapy.
AEs of Anti-CD19 CAR T Cell Therapy
AEs were observed for >120 days after anti-CD19 CAR T cell infusion. The CRS grade was determined according to the National Cancer Institute Common Terminology Criteria for AE (version 4.03) 20 . Neurotoxicity syndrome was determined according to ICANS 21 .
The Expansion of Anti-CD19 CAR T Cells and Cytokine Levels
The expansion proportions of anti-CD19 CAR T cells in CD3+ T cells in the peripheral blood were determined using FCM on days 0, 7, 14, 21, 28, and 60 after CAR T cell infusion. Humanized CD19 CAR antibodies were provided by GenBase Biotechnology (patent owner of CD19 CAR). Interleukin-6 (IL-6) levels were measured on days 0, 7, 14, 21, and 28 after CAR T cell infusion using an enzyme-linked immunosorbent assay.
Criteria for CAR T Cell Therapy-Mediated Hematotoxicity and Persistent Cytopenia
The criteria for CAR T cell therapy-mediated hematotoxicity were graded according to the joint American Society of Clinical Oncology/Infectious Diseases Society of America (ASCO/IDSA) consensus guidelines for cancer-related infection risk22,23. Persistent cytopenia was defined as Grades 3–4 cytopenia and hematotoxicity lasting >56 days after CAR T cell infusion. Because patients were evaluated once every 2 weeks, AEs other than hematotoxicity resolved 21–45 days after CAR T cell infusion; therefore, persisting hematological toxicity beyond 56 days (8 weeks) was considered as the time persistent cytopenia was evaluated. Patients with Grades 3–4 persistent cytopenia underwent immediate bone marrow smear tests and blood count evaluations once per week or less.
Management of Persistent Cytopenia
Patients with Grades 3–4 persistent cytopenia received treatment, including the application of granulocyte-colony stimulating factor (150–300 µg/day), thrombopoietin (25–50 mg/day) receptor (eltrombopag), or a blood or platelet transfusion until Grades 3–4 persistent cytopenia recovered.
Statistical Analyses
SPSS version 17.0 (SPSS Inc., Chicago, IL, USA) was used for all statistical analyses. The t-test was used to compare data conforming to a normal distribution, and the rank sum test was used to compare data not conforming to a normal distribution. The chi-square test was used to analyze the data. All values were expressed as the mean ± standard deviation. PFS and OS were analyzed using univariate analysis. P < 0.05 was considered statistically significant.
Results
Patient Characteristics
Baseline characteristics of the 38 patients with R/R DLBCL enrolled in the clinical trial are listed in Table 1. Except for the maximum tumor diameter, C-reactive protein levels, and lactate dehydrogenase levels, no differences were observed in characteristics between patients with or without Grades 3–4 persistent cytopenia after anti-CD19 CAR T cell therapy (Table 2).
Patients Baseline Characteristics.
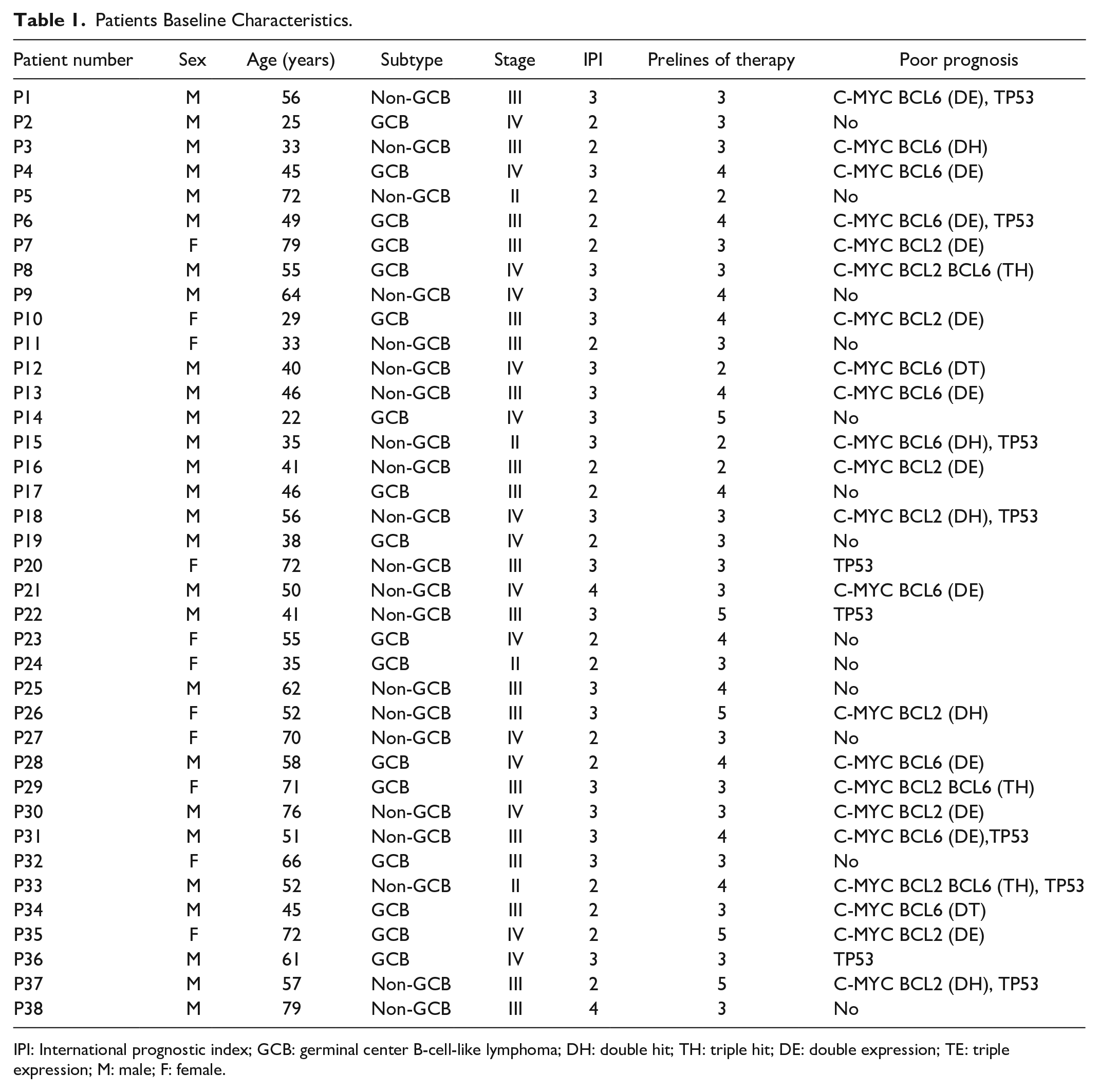
IPI: International prognostic index; GCB: germinal center B-cell-like lymphoma; DH: double hit; TH: triple hit; DE: double expression; TE: triple expression; M: male; F: female.
Comparison of Baseline Characteristics Between the Two Groups.
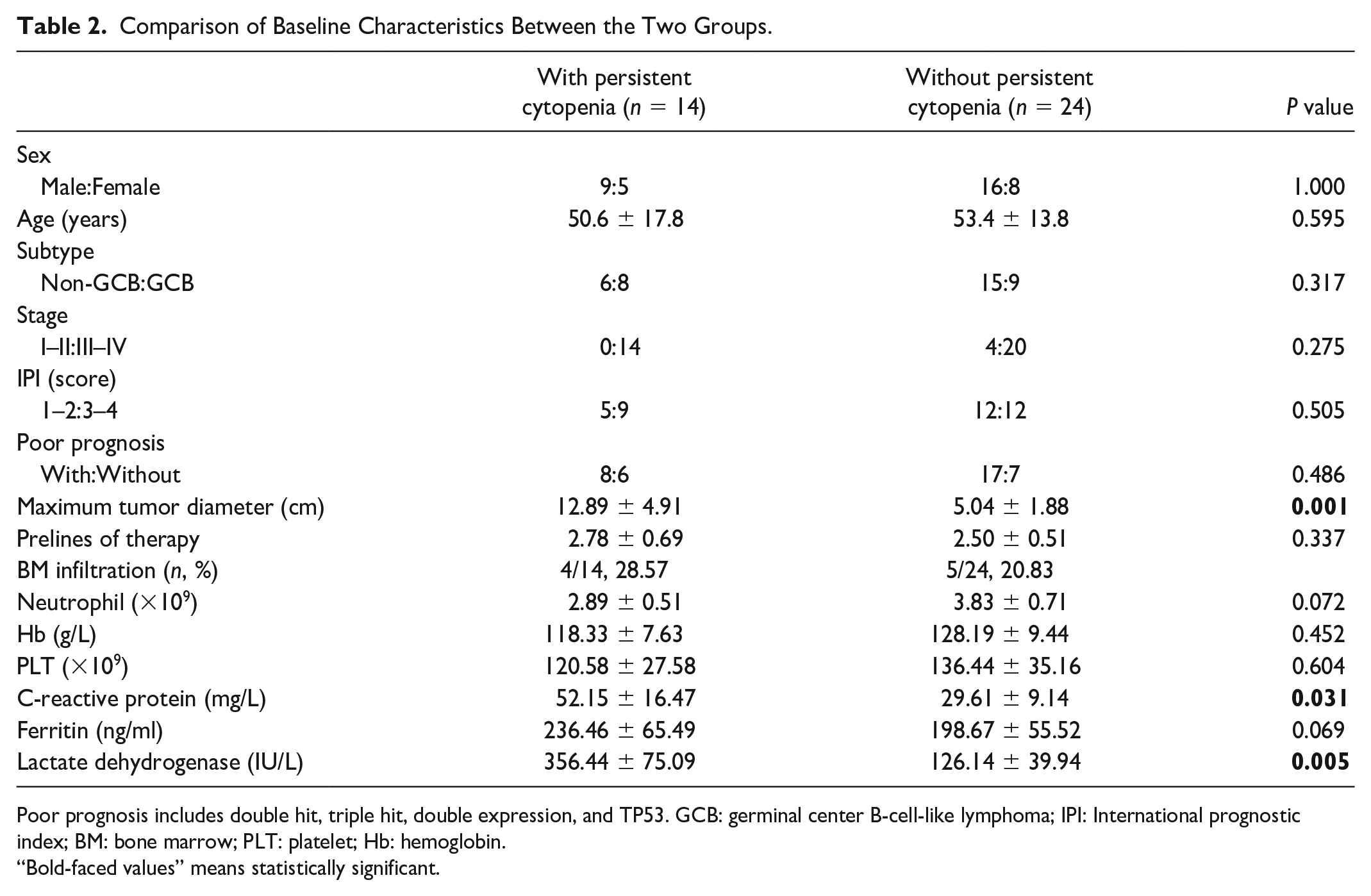
Poor prognosis includes double hit, triple hit, double expression, and TP53. GCB: germinal center B-cell-like lymphoma; IPI: International prognostic index; BM: bone marrow; PLT: platelet; Hb: hemoglobin.
“Bold-faced values” means statistically significant.
Transduction Efficiency, Amplification, and Anti-CD19 CAR T Cell Infusion
The mean anti-CD19 CAR transduction efficiency in the final products of the 38 patients with R/R DLBCL was 49.85 ± 13.35%. All 38 patients received a dose of 2.19 ± 0.42 × 106 cells/kg anti-CD19 CAR T cell infusion on day 0 after lymphodepleting chemotherapy.
Hematological Toxicity of Anti-CD19 CAR T Cell Therapy
Hematological toxicity was diagnosed in Grades 0–4 persistent cytopenia after anti-CD19 CAR T cell therapy. Hematological toxicity began 4–10 days after anti-CD19 CAR T cell infusion. Thirteen patients had Grades 3–4 neutropenia, 9 had Grades 3–4 anemia, and 11 had Grades 3–4 thrombocytopenia (Fig. 1A). Grades 3–4 persistent cytopenia occurred in 14 of the 38 patients with R/R DLBCL (hematotoxicity lasting >8 weeks after CAR T cell infusion). The different types and persistent times of cytopenia in these 14 patients with Grades 3–4 cytopenia are shown in Fig. 1B. The blood cell changes in patients with Grades 3–4 neutropenia, anemia, and thrombocytopenia are shown in Fig. 1C–E. The trend curve of Grades 3–4 neutropenia had two valleys in seven patients with persistent neutropenia (Patients 2, 4, 14, 16, 21, 32, and 34). The mean value of the first valley of neutropenia was 0.66 ± 0.21 × 109/L on day 7 after CAR T infusion, whereas the second valley of neutropenia was 0.21 ± 0.11 × 109/L at 28–56 days after CAR T infusion (Fig. 1C). This trend was also observed in three patients (Patients 17, 21, and 34) with persistent thrombocytopenia (Fig. 1D) but not in patients with persistent anemia.
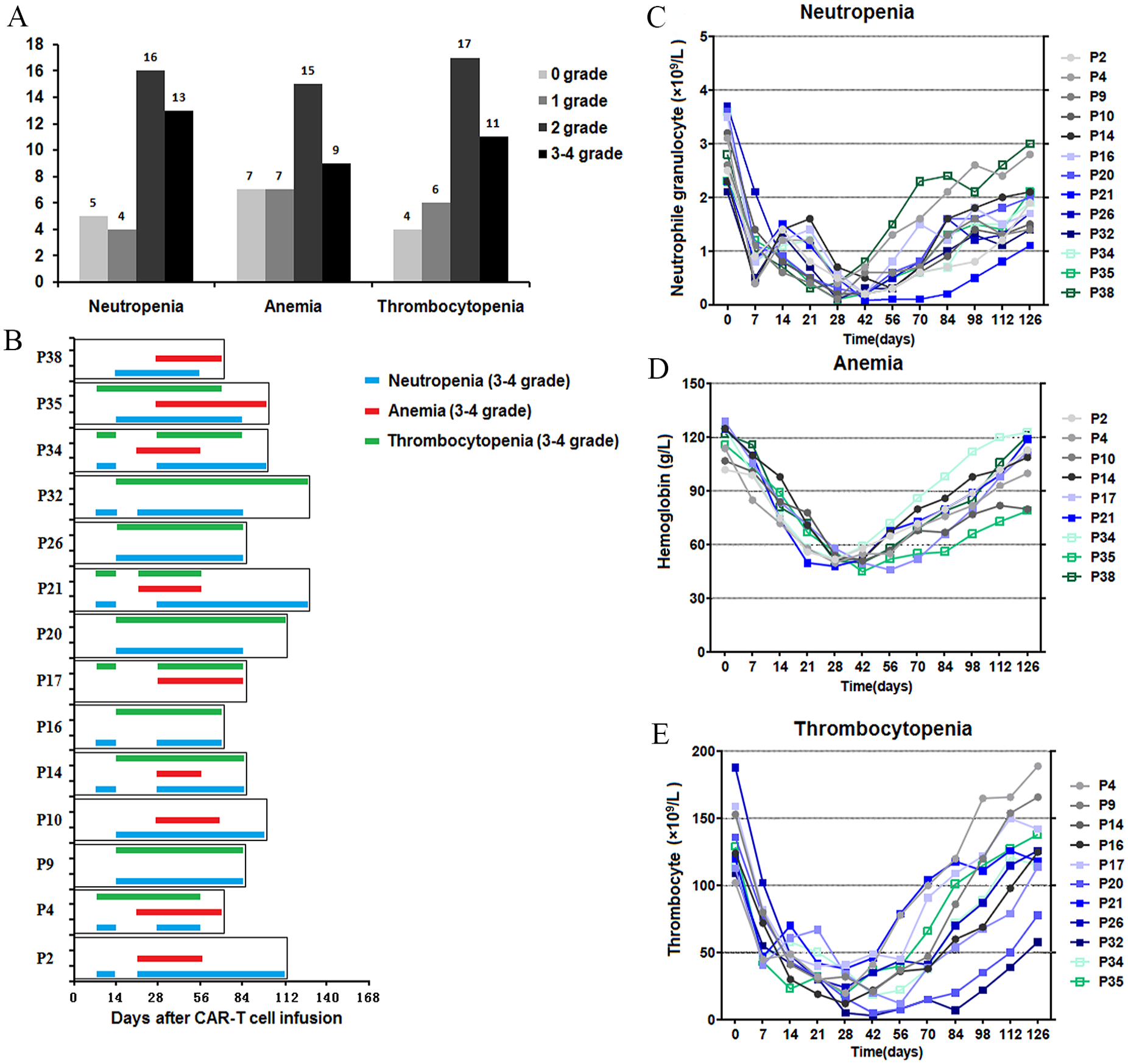
Hematological toxicity of anti-CD19 CAR T cell therapy. (A) Grades 0–4 neutropenia, anemia, and thrombocytopenia were diagnosed after anti-CD19 CAR T cell therapy. (B) The different types and persistent times of cytopenia in the 14 patients who had Grades 3–4 persistent cytopenia. (C–E) Blood cell changes in patients with Grades 3–4 neutropenia, anemia, and thrombocytopenia. CAR: chimeric antigen receptor.
Severe marrow dysplasia or relapse was not observed in the bone marrow smear 56 days after CAR T cell infusion in any of the 14 patients with Grades 3–4 persistent cytopenia. These patients received red blood cell transfusions, cytokines, and other supportive therapies to recover from persistent cytopenia. Persistent cytopenia recovered 8–18 weeks after CAR T cell infusion.
After anti-CD19 CAR T cell infusion, three patients diagnosed with Grade 3 or 4 neutropenia and one patient diagnosed with Grade 2 neutropenia had bacterial infections. None of these patients had been previously diagnosed with an invasive fungal disease. Only one patient without neutropenia (Patient 10) died of bacterial septicemia when the disease progressed.
The Clinical Response of CAR T Cells
Two months after anti-CD19 CAR T cell infusion, the therapeutic efficacy in all 38 patients with R/R DLBCL was evaluated. Eighteen patients (47.37%) achieved CR, whereas eight patients (21.05%) achieved PR with anti-CD19 CAR T cell therapy. The ORR was 68.42% (26/38). Six patients (15.79%) had SD and PD, respectively. All 14 patients with Grades 3–4 persistent cytopenia achieved CR and PR, whereas no patient who achieved SD and PD had Grades 3–4 persistent cytopenia after anti-CD19 CAR T cell therapy (Fig. 2A).
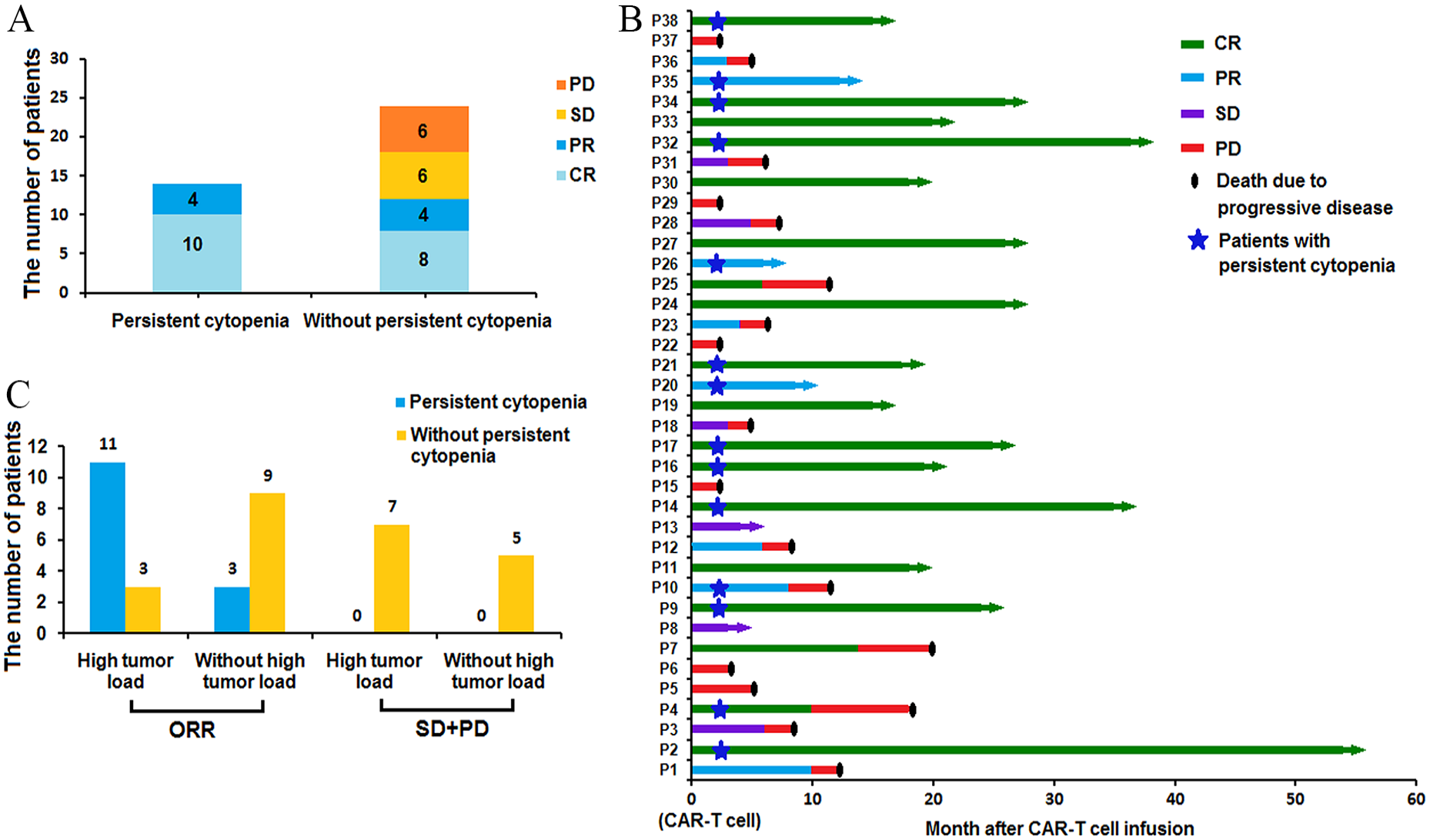
(A) All 14 patients with Grades 3–4 persistent cytopenia obtained CR and PR, whereas no patient who obtained SD and PD had cytopenia during CAR T cell therapy. (B) The clinical response to anti-CD19 CAR T cell therapy, PD, and survival time of all 38 patients. (C) The incidence of Grades 3–4 persistent cytopenia was higher in 26 patients who obtained ORR with high tumor load than in patients with lower tumor loads. CAR: chimeric antigen receptor; CR: complete response; PR: partial remission; SD: stable disease; PD: disease progression; ORR: objective response rate.
In all 18 patients with R/R DLBCL who achieved CR, 15 survived in the CR state until the cutoff date, and the other 3 patients died of disease relapse (Patients 7 and 25 with persistent cytopenia and Patient 4 without persistent cytopenia). Only three patients who achieved PR (Patients 22, 26, and 35 with persistent cytopenia) and two patients who achieved SD (Patients 8 and 13 without persistent cytopenia) survived until the cutoff date. All patients who achieved PD died due to PD (Fig. 2B).
Of the 26 patients with R/R DLBCL who achieved ORR, 14 had a high tumor load, and 12 had no high tumor load. Among these 26 patients, Grades 3–4 persistent cytopenia occurred in 11 (78.57%) patients with a high tumor load and 3 (25.00%) without a high tumor load. The incidence of Grades 3–4 persistent cytopenia was higher in patients with a high tumor load than in patients without a high tumor load in these 26 patients who obtained ORR (P = 0.006; Fig. 2C).
AEs of Anti-CD19 CAR T Cell Therapy
AEs were observed for >120 days following anti-CD19 CAR T cell infusion. Apart from hematological toxicity, no difference in the characteristics between patients with or without Grades 3–4 persistent cytopenia was observed after anti-CD19 CAR T cell therapy (Table 3). Except for hematological toxicity, the AEs disappeared 14–21 days after CAR T cell infusion.
Adverse Events During Anti-CD19 CAR T Cell Therapy.
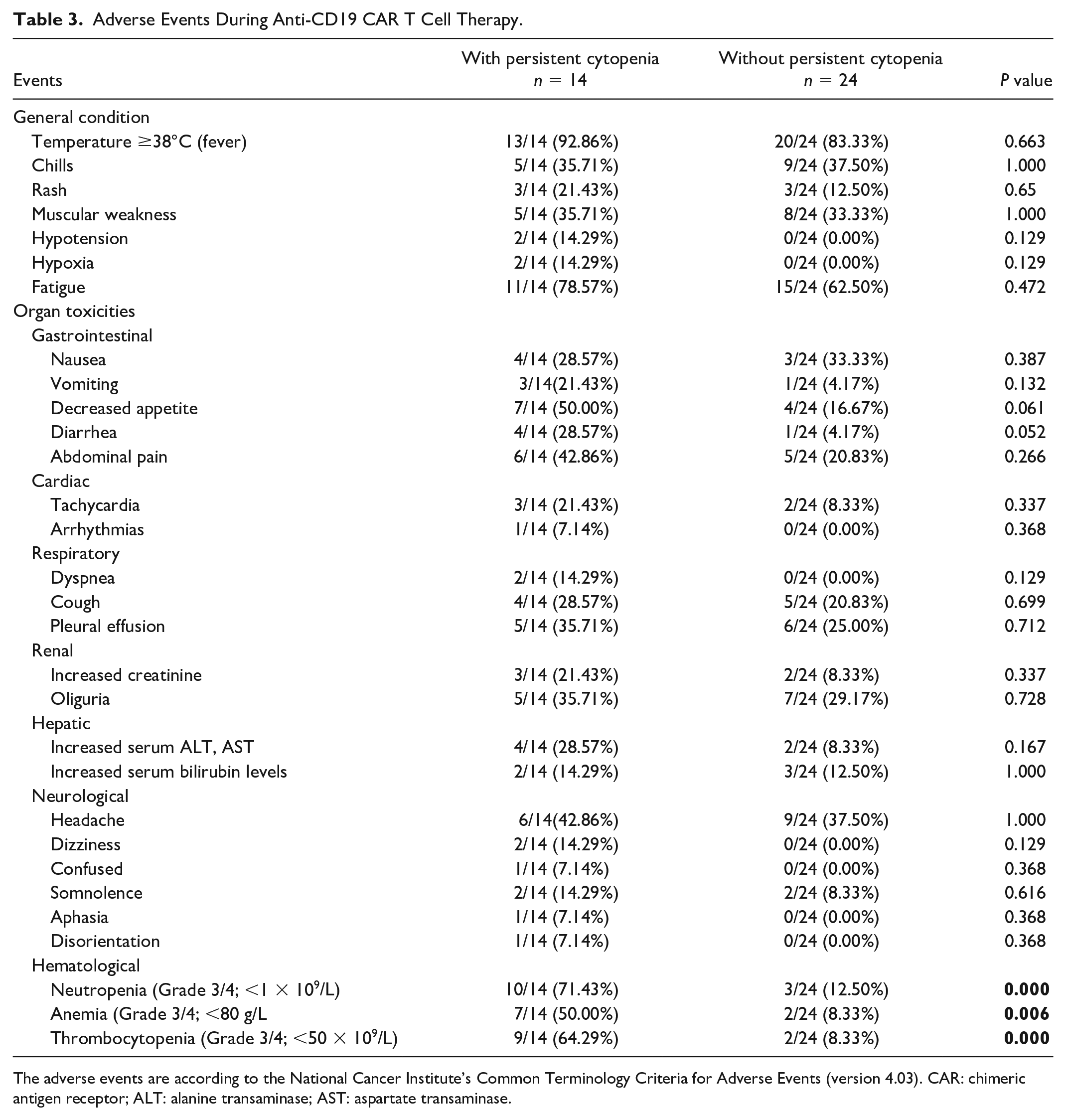
The adverse events are according to the National Cancer Institute’s Common Terminology Criteria for Adverse Events (version 4.03). CAR: chimeric antigen receptor; ALT: alanine transaminase; AST: aspartate transaminase.
The mean peak of IL-6 in patients with R/R DLBCL with Grades 3–4 persistent cytopenia (54.44 ± 28.96 pg/ml) was higher than that of patients without persistent cytopenia (17.06 ± 8.22 pg/ml; P < 0.0001; Fig. 3A).
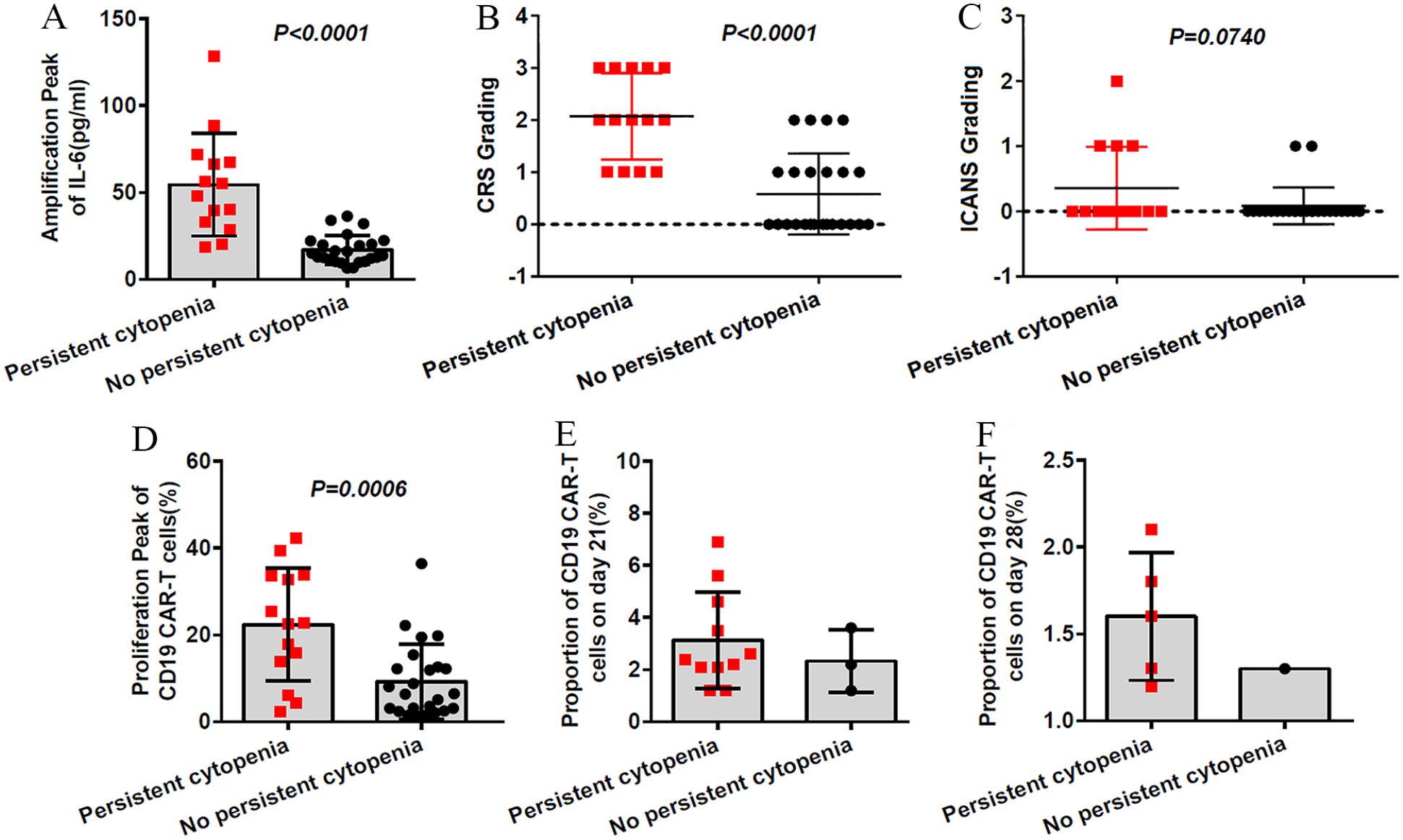
Adverse events of anti-CD19 CAR T cell therapy. (A) The mean peak of IL-6 levels in patients with Grades 3–4 persistent cytopenia was higher than in patients without Grades 3–4 persistent cytopenia. (B) CRS was higher in patients with Grades 3–4 persistent cytopenia than in patients without persistent cytopenia. (C) No difference was observed in Grades 3–4 persistent cytopenia between different ICANS grades. (D) The median amplification peak of CAR T cells was higher in patients with Grades 3–4 persistent cytopenia than in patients without persistent cytopenia. (E, F) Most patients whose CAR T cells were observed at 21 and 28 days after CAR T cell infusion had Grades 3–4 persistent cytopenia. CAR: chimeric antigen receptor; IL: interleukin; CRS: cytokine release syndrome; ICANS: immune effector cell-associated neurotoxic syndrome.
Except for the five patients who developed Grade 3 CRS, all other patients developed Grades 0–2 CRS during anti-CD19 CAR T cell therapy. Two patients developed Grade 2 ICANS, seven developed Grade 1 ICANS, and the other patients developed Grade 0 ICANS during anti-CD19 CAR T cell therapy. CRS grades were higher in patients with Grades 3–4 persistent cytopenia than in those without persistent cytopenia (PCRS < 0.0001). However, this finding was not observed in patients with different ICANS grades (PICANS = 0.0740; Fig. 3B, C). None of the patients died of CRS or ICANS during anti-CD19 CAR T cell therapy. Only patients who developed Grade 3 CRS and Grade 2 ICANS received glucocorticoids or tocilizumab as an anti-CD19 CAR T cell therapy.
Anti-CD19 CAR T Cell Amplification
The proportion of anti-CD19 CAR T cells in the peripheral blood was observed after CAR T cell infusion. The interquartile range (IQR) of the anti-CD19 CAR T cell amplification peak in CD3+ T cells was 14.1% (IQR 3.3–21.6) at 4 or 7 days after CAR T cell infusion. The median amplification peak of anti-CD19 CAR T cells was higher in patients with Grades 3–4 persistent cytopenia [24.1% (IQR 16.3–33.5)] than in patients without persistent cytopenia [8.2% (IQR 2.5–12.2); P = 0.0006; Fig. 3D]. The proportion of anti-CD19 CAR T cells (>1%) was observed 21 days after CAR T cell infusion in 14 patients. Anti-CD19 CAR T cells were observed 28 days after CAR T cell infusion in six patients. The results of patients whose CAR T cells could be observed at 21 and 28 days after CAR T cell infusion were associated with Grades 3–4 persistent cytopenia. However, because the number of cases was relatively small, a statistical analysis was not performed (Fig. 3E, F). All 20 patients with R/R DLBCL whose CAR T cells were observed at 21 and 28 days after CAR T cell infusion achieved CR or PR during therapy.
Follow-up
By the cutoff date, the median PFS and OS in the 38 patients were 14.32 (IQR 3–54) and 15.11 (IQR 2–54) months, respectively. In patients with Grades 3–4 persistent time of hematological toxicity, the 1-year median PFS and OS were 18 and 18.5 months, respectively. However, in patients without toxicities, the 1-year median PFS and OS were 5 and 6.5 months, respectively. The PFS and OS were longer in patients with Grades 3–4 persistent cytopenia than in patients without persistent cytopenia [95% (confidence interval) CI: 9–35 months, PPFS = 0.0019 and 95% CI: 12–35 months, POS = 0.0006; Fig. 4A, B].
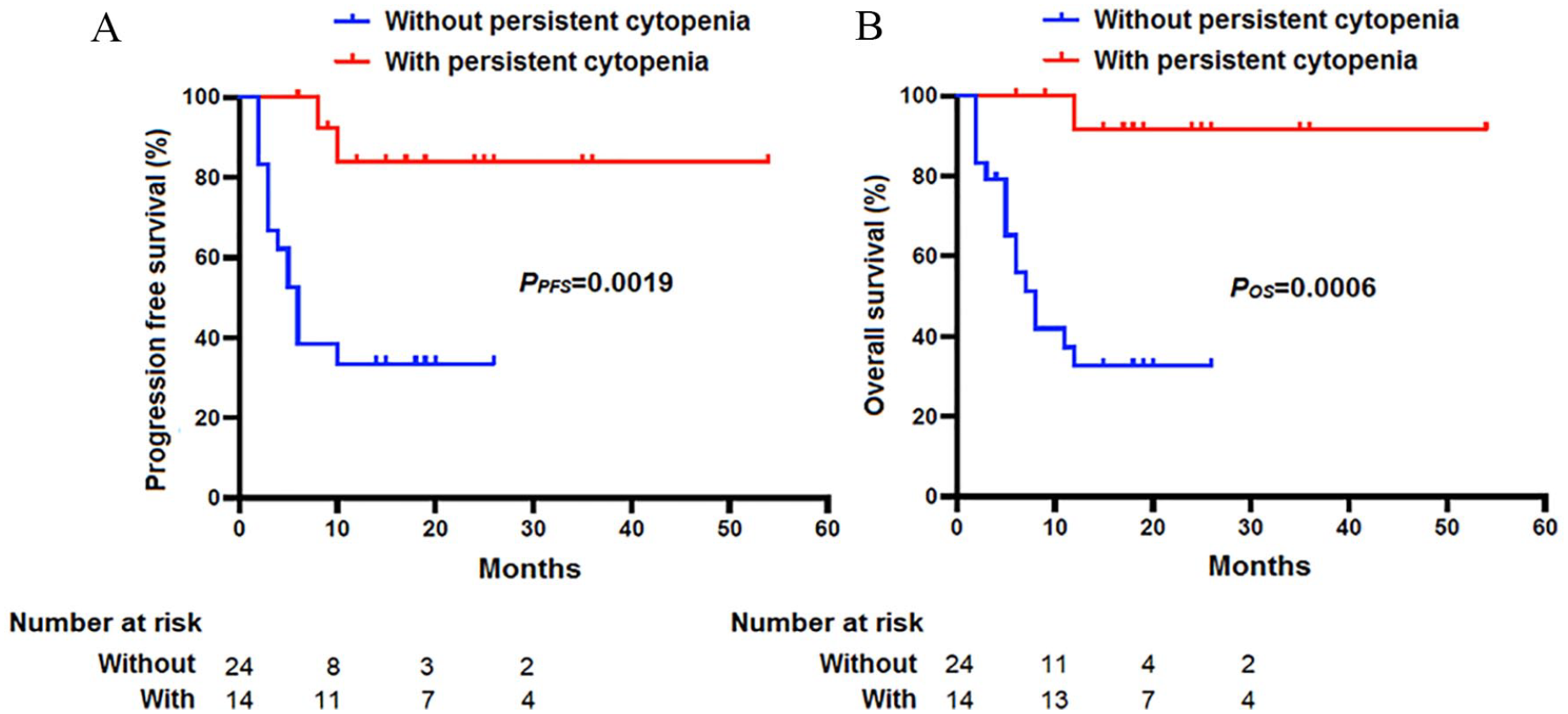
Follow-up after anti-CD19 CAR T cell therapy. (A, B) The progression-free and overall survival were higher in patients with Grades 3–4 persistent cytopenia than in those who did not have persistent cytopenia. CAR: chimeric antigen receptor.
None of the patients died from Grades 3–4 persistent cytopenia. In the Grades 3–4 persistent cytopenia group, two patients (Patients 4 and 10) died of PD. In the group without Grades 3–4 persistent cytopenia, 15 patients died owing to PD.
Discussion
Hematological toxicity is a common condition after anti-CD19 CAR T cell therapy. Most (90.4%) cases of persistent cytopenia occur between days 31 and 180 after CAR T cell infusion 24 . In the ZUMA-1 study6,12,25, Grade 3 or higher cytopenia frequently occurred in the first 30 days after anti-CD19 CAR T cell infusion and was likely associated with lymphodepleting chemotherapy with fludarabine and cyclophosphamide. Patients with chronic lymphocytic leukemia generally recover from cytopenia within several weeks, even after six cycles of fludarabine and cyclophosphamide 26 . However, in the ZUMA-1 study, cytopenia persisted for 3 months or longer without evidence of marrow dysplasia or relapse 4 . Therefore, hematological toxicities unrelated to lymphodepleting chemotherapy may exist.
The definitions of cytopenia prolongation and severity vary in different studies13,27–29. A high incidence of persistent Common Terminology Criteria for Adverse Events Grade ≥3 neutropenia (30–38%), thrombocytopenia (21–29%), and anemia (5–17%) after 21 days has been reported. Similarly, in our study, 14 of the 38 patients with R/R DLBCL developed Grades 3–4 persistent cytopenia after anti-CD19 CAR T cell therapy. To distinguish between persistent cytopenia and hematological toxicity caused by lymphodepleting chemotherapy with fludarabine and cyclophosphamide, persistent cytopenia was defined as hematotoxicity lasting >56 days after CAR T cell infusion. Apart from hematotoxicity, most AEs disappeared 21–28 days after CAR T cell infusion.
Notably, in this study, the trend curve of Grades 3–4 neutropenia had two valleys in seven patients with persistent neutropenia at 7 and 28–56 days after CAR T cell infusion. This Grade 3 or 4 persistent cytopenia recovered 8–18 weeks after CAR T cell infusion with supportive therapy. The first valley of Grades 3–4 neutropenia may be related to lymphodepleting chemotherapy with fludarabine and cyclophosphamide. However, the cause of the second valley in patients with Grades 3–4 neutropenia is unclear. In the present study, most pathogenic infections after anti-CD19 CAR T cell therapy occurred after neutropenia. However, no evidence of severe marrow dysplasia or relapse in the bone marrow in the second valley of Grades 3–4 neutropenia was observed in these patients. Therefore, the second valley of Grades 3–4 neutropenia was not caused by lymphodepleting chemotherapy, infections, marrow dysplasia, or relapse but was most likely caused by anti-CD19 CAR T cell therapy.
The mechanism underlying persistent cytopenia after anti-CD19 CAR T cell therapy remains unclear. Cytotoxic therapies and autologous or allogeneic hematopoietic stem cell transplantation have been hypothesized to cause cytopenia10,28,30. In the present study, no difference was observed in the baseline characteristics between patients with and without Grades 3–4 persistent cytopenia, except that the tumor load, C-reactive protein, and lactate dehydrogenase before CAR T cell therapy were higher in patients with Grades 3–4 persistent cytopenia. An increasing grade of CRS and ICANS, baseline cytopenia, CAR constructs, and higher peak C-reactive protein or ferritin levels are associated with persistent cytopenia for more than 1 month 18 . Another study demonstrated the same result that Grade ≥4 CRS was associated with delayed hematopoietic recovery 30 . Anti-CD19 CAR T cell-induced cytolysis of CD19-positive lymphoma cells facilitates antigenic spreading, which may be associated with severe CRS31–33. Furthermore, severe neutropenia until 60 days after anti-CD19 CAR T cell therapy is not associated with the incidence and severity of CRS, ICANS, or peak cytokine levels but is associated with hematopoietic reserves and baseline inflammation 23 . The mechanism of persistent cytopenia after anti-CD19 CAR T cell therapy has been explored previously 23 . However, the current study identified a substantially increased frequency of clonally expanded CX3CR1hi cytotoxic T cells, expressing high interferon-γ and cytokine signaling gene sets associated with prolonged cytopenia.
In a long-term follow-up study of patients after anti-CD19 CAR T cell therapy, 16% of patients with persistent CR sustained persistent cytopenia after infusion 30 , similar to the results of this study. The PFS and OS were longer in patients with Grades 3–4 persistent cytopenia than in those without persistent cytopenia. Grade 3 or 4 persistent cytopenia was associated with high tumor load, higher IL-6 and CAR T cell levels, and higher CRS grades. Moreover, higher IL-6 and CAR T cell peaks and CRS grades were associated with higher CR ratios and longer PFS and OS. Therefore, sustained cell reduction after CAR T cell therapy may be related to prolonged PFS and OS.
However, the present study has several limitations. First, only a limited number of patients were included.
In conclusion, patients with R/R DLBCL who had Grades 3–4 persistent cytopenia after anti-CD19 CAR T cell therapy achieved ORR. Grade 3 or 4 persistent cytopenia was associated with a high tumor load, higher IL-6 and CAR T cell peaks, higher CRS grades, and a higher PFS and OS. The results of this study indicate that persistent cytopenia after anti-CD19 CAR T cell therapy in patients with R/R DLBCL can be used as a predictor of therapeutic efficacy and AEs. However, further studies are required to confirm these conclusions.
Footnotes
Acknowledgements
The authors thank all the patients for their participation in the clinical trial. They thank Shanghai Genbase Biotechnology Co., Ltd. for providing them with anti-CD19 CAR T cells.
Author Contributions
L.J.Y. and D.Q. contributed to the study’s conception and design. D.Q. and M.J. contributed to supervision. L.J.Y., W.J., L.X., and L.Q. wrote the manuscript and acquired funding. W.J. and J.Y.L. collected the data and performed the methods. L.J.Y. contributed to the validation and software usage. All the authors have approved the final manuscript.
Availability of Data and Material
All data and materials were obtained in accordance with the Declaration of Helsinki and conformed to relevant aspects of the ARRIVE guidelines.
Ethical Approval
This study was approved by the Medical Ethics Committee of Tianjin First Center Hospital (Tianjin, China; approval nos. 2015002X and 2018N105KY).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Informed consent was obtained from all participants. All patients agreed to participate in the clinical trials. Patients agreed to use their data for the study.
Trial Registration
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Tianjin Health Research Project (grant no. TJWJ2023ZD003) and the Chinese Society of Clinical Oncology Beijing Xisike Clinical Oncology Research Foundation (grant nos. Y-SY2021QN-0184, Y-Young2022-0209, Y-NCJH202201-0027, and Y-2022YMJN/MS-0001).
