Abstract
A significant proportion of patients with relapsed/refractory diffuse large B-cell lymphoma (R/R DLBCL) exhibit no response to chimeric antigen receptor (CAR) T-cell therapy or suffer from disease progression thereafter. This study investigated the efficacy and safety of salvage therapy with PD-1 inhibitor-based combination treatment and the patient selection after their CAR T-cell therapy. Twenty-one patients with R/R DLBCL and a high tumor burden were treated with CAR T-cell therapy, a treatment that has shown promising results in clinical trials and has been approved by the Food and Drug Administration (FDA) for use in DLBCL. Patients who achieved complete response (CR) with the CAR T-cell therapy received salvage therapy when their disease progressed again. Patients who obtained partial response (PR) or stable disease (SD) with the CAR T-cell therapy received salvage therapy immediately. Salvage therapy consisted of single PD-1 inhibitors or PD-1 inhibitors combined with chemotherapy. We observed the overall response rate (ORR), overall survival (OS), CAR T-cell amplification, the expression of PD-1, CD3+ T cells, cytokines, and the adverse events. For instance, in a clinical trial of LCAR-B38M CAR T-cell therapy, an 88% ORR was observed, with 74% of patients achieving CR and a median duration of response (DOR) of 16 months. The ORR and CR of the salvage therapy were 28.57% and 19.05%, respectively. The ORR and CR were 38.46% and 30.77% in the 13 patients who achieved PR/SD with the CAR T-cell therapy and received salvage therapy 2 months after CAR T-cell infusion. But the ORR and CR were only 12.5% and 0%, respectively, in patients who achieved CR with the CAR T-cell therapy and received salvage therapy when they experienced disease re-progression. The ratio of CAR-T cells on day 7/day 14 was lower in the PR in CAR-T (effective to PD-1) group. Before salvage therapy, the percentage of CD3+ T cells was higher in the PR in CAR-T (effective to PD-1) group. There was no difference in the Common Terminology Criteria for Adverse Events (CTCAE) grades among the four groups in the salvage therapy. PD-1 inhibitor-based salvage therapy in patients with R/R DLBCL following the CAR T-cell therapy could be an effective and safe treatment, especially in patients who achieved PR after the CAR T-cell therapy and received this salvage therapy immediately.
Keywords
Introduction
Anti-CD19 chimeric antigen receptor (CAR) T-cell therapy has been approved as an effective salvage strategy for relapsed/refractory (R/R) diffuse large B-cell lymphoma (DLBCL), high-grade B-cell lymphoma, mantle cell lymphoma, and follicular lymphoma (FL)1–5. Despite significantly enhancing response rates and survival in R/R B-cell lymphoma patients, anti-CD19 CAR T-cell therapy achieves long-term remission in only 30%–40% of R/R DLBCL cases6–8. Some patients do not respond to this therapy, and some patients experience disease progression again. In the ZUMA-1 study 9 , which involved 101 patients with R/R large B-cell lymphomas, the overall response rate (ORR) and complete response rate (CR) were 83% and 58%, respectively, for those treated with axicabtagene ciloleucel. While the 2-year progression-free survival (PFS) for CAR T-cell therapy stands at 39%1,9. Therefore, there is a need to explore salvage therapies for patients with R/R DLBCL who do not respond to anti-CD19 CAR T-cell therapy or those who relapse.
In recent years, programmed cell death 1 (PD-1) inhibitors have shown satisfactory effects in some malignancies10–12. Studies have shown that overexpression of PD-1 and PD-L1 proteins in DLBCL is associated with increased tumor aggressiveness and poor prognosis13,14. As such, PD-1 inhibitors present a promising immunological target for patients with R/R DLBCL. Our previous studies demonstrated the synergistic effects of PD-1 inhibitors in combination with chemotherapy regiments containing platinum-based chemotherapy agents on Pfeiffer cells in vitro and in vivo 15 . In a study, pembrolizumab demonstrated efficacy in treating patients with relapsed or refractory (R/R) primary mediastinal large B-cell lymphoma (PMBCL), as evidenced by FDA approval for this indication 16 . Pembrolizumab with rituximab demonstrated an ORR of 80% with a CR of 60% in R/R FL 17 . Nivolumab demonstrated a 40% response rate, and most responses were durable for up to 2 years in R/R FL 18 . But PD-1 inhibitors as single-agent therapy had limited efficacy against DLBCL18,19. Therefore, the combination with PD-1 inhibitors for R/R DLBCL should be further explored.
In our study, 21 patients with R/R DLBCL, and high tumor loads were treated with salvage therapy following anti-CD19 CAR T-cell therapy. The objective of this study was to assess the efficacy and safety of salvage therapy utilizing PD-1 inhibitors alone or in combination with chemotherapy, administered following anti-CD19-CAR T-cell therapy.
Patients and methods
Patients enrolled in the study
Between September 2019 and December 2021, a total of 44 patients with R/R DLBCL with a high tumor burden were admitted to our center, all of whom received anti-CD19-CAR T-cell therapy. None of the patients had undergone hematopoietic stem cell transplantation (HSCT) before this study. Among the 44 patients, 21 were selected for our study and underwent salvage therapy subsequent to the anti-CD19-CAR T-cell therapy. Patients with R/R DLBCL who achieved CR through CAR T-cell therapy subsequently experienced disease progression. Patients who had achieved partial response (PR) or stable disease (SD) with CAR T-cell therapy subsequently underwent this salvage therapy. The cutoff date for our study was 31 December 2024. Follow-up was performed from the day of anti-CD19 CAR T-cell infusion to the cutoff date or the death date.
Generation of anti-CD19-CAR T cells and the transduction efficiency
All the CD3+ T cells (3 × 106) were isolated from the peripheral blood mononuclear cells (PBMCs) of the 44 patients with R/R DLBCL. The cells were transduced with a lentiviral vector encoding humanized CD19 CAR constructs (10 µg, lenti-CD19-2rd-CAR; 41BB co-stimulatory domain, Shanghai Genbase Biotechnology Co., Ltd., Shanghai, China). On the 12th–14th days of cultivation, the transduction efficiency of anti-CD19-CAR was analyzed by flow cytometry (FCM).
Anti-CD19-CAR T-cell therapy
All 21 patients with R/R DLBCL received anti-CD19 CAR T-cell therapy (ChiCTR1800019622). This study was approved by the Ethics Committee of Tianjin First Central Hospital. All patients provided written informed consent before therapy. Fludarabine (30 mg/m2/day) and cyclophosphamide (400 mg/m2/day) were administered from days −4 to −2 as lymphodepleting chemotherapy. The anti-CD19-CAR T-cell infusion dose was 2 × 106 cells/kg on day 0 in all the 21 patients with R/R DLBCL.
Salvage therapy of PD-1 inhibitors or PD-1 inhibitors combined with chemotherapy
In our study, all patients with R/R DLBCL who achieved CR with the anti-CD19 CAR T-cell therapy were observed for their efficacy without any maintenance therapy. Salvage therapy was initiated when the disease progressed. The other patients with R/R DLBCL who achieved PR or SD with the anti-CD19 CAR T-cell therapy received salvage therapy 2 months after CAR T-cell infusion. This salvage therapy was another clinical trial of our center (ChiCTR1900025310). It was approved by the Ethics Committee of Tianjin First Central Hospital.
If the patient’s neutrophil count was lower than 1.0 × 109/L, the patient received 200 mg of PD-1 inhibitors every 3 weeks as a single-drug salvage therapy. To prevent severe hematological toxicity caused by chemotherapy after anti-CD19 CAR T-cell therapy, the other patients with R/R DLBCL who had neutrophil counters higher than 1.0 × 109/L were treated with single PD-1 inhibitors/PD-1 inhibitors combined with chemotherapy alternately every 3 weeks as salvage therapy. The ifosfamide-carboplatin-etoposide (ICE) chemotherapy regimen was selected as the combined salvage therapy with PD-1 inhibitors (Fig. 1). In salvage therapy, the efficacy was evaluated once every two cycles. All patients were scheduled to receive more than two cycles of salvage therapy until they achieved a CR or experienced grades 3–4 of adverse events (AEs) associated with PD-1 inhibitors and disease progression.

Design of anti-CD19 CAR T-cell therapy and PD-1 inhibitor-based salvage therapy.
None of the 21 patients with DLBCL was treated with PD-1 inhibitors before the anti-CD19 CAR T-cell therapy. They previously used ICE/DICE (dexamethasone-ICE), but the efficacy evaluation was SD/PD.
Expression of anti-CD19-CAR T cells, PD-1, CD3+ T cells, and cytokines
We categorized 21 patients with R/R DLBCL into four groups to monitor anti-CD19-CAR T-cell expansion and PD-1 expression: CR following CAR T-cell therapy (patients experiencing disease progression post-treatment), PR in CAR T-cell therapy with PD-1 responsiveness, PR in CAR T-cell therapy without PD-1 responsiveness, and SD in CAR T-cell therapy groups. The transfection rate of anti-CD19-CAR and the amplification rate of anti-CD19-CAR T cells were determined using FCM. During anti-CD19-CAR T-cell therapy and salvage therapies, the proportion of CD3+ T cells in the peripheral blood and the expression of anti-CD19-CAR T cells and PD-1 in CD3+ T cells were observed using FCM. Levels of interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α) were measured on days 0, 7, 14, 21, and 28 in anti-CD19-CAR T-cell and salvage therapies by enzyme-linked immunosorbent assay (ELISA).
Adverse events
In the context of anti-CD19-CAR T-cell therapy, the severity of cytokine release syndrome (CRS) and immune-effector cell-associated neurotoxicity syndrome (ICANS) were was assessed using the National Cancer Institute’s Common Terminology Criteria for Adverse Events (CTCAE) version 4.03 20 . Similarly, the grading of neurotoxicity was determined in accordance with the Immune Effector Cell-Associated Encephalopathy (ICE) scale 21 , as detailed in the CAR T-cell treatment toxicity management guidelines. For the salvage therapy with PD-1 inhibitors, the grades of AEs were evaluated according to the CTCAE 5 .
Results
Anti-CD19-CAR T-cell therapy
Baseline characteristics before anti-CD19-CAR T-cell therapy
All patients with R/R DLBCL with high tumor bulk had at least one tumor with a maximum diameter ≥7.5 cm or at least two extra-nodal lesions before the anti-CD19-CAR T-cell therapy. Before anti-CD19-CAR T-cell and salvage therapies with PD-1 inhibitors, we compared the baseline characteristics of patients with R/R DLBCL. Except for the primary lines, there were no differences in age, sex, stage, or International Prognostic Index scores between the CR an PR/SD in the anti-CD19-CAR T-cell therapy group (Table 1). Baseline characteristics of all the 44 patients with R/R DLBCL are shown in Supplementary Table 1.
Comparison of baseline characteristics between the two groups.
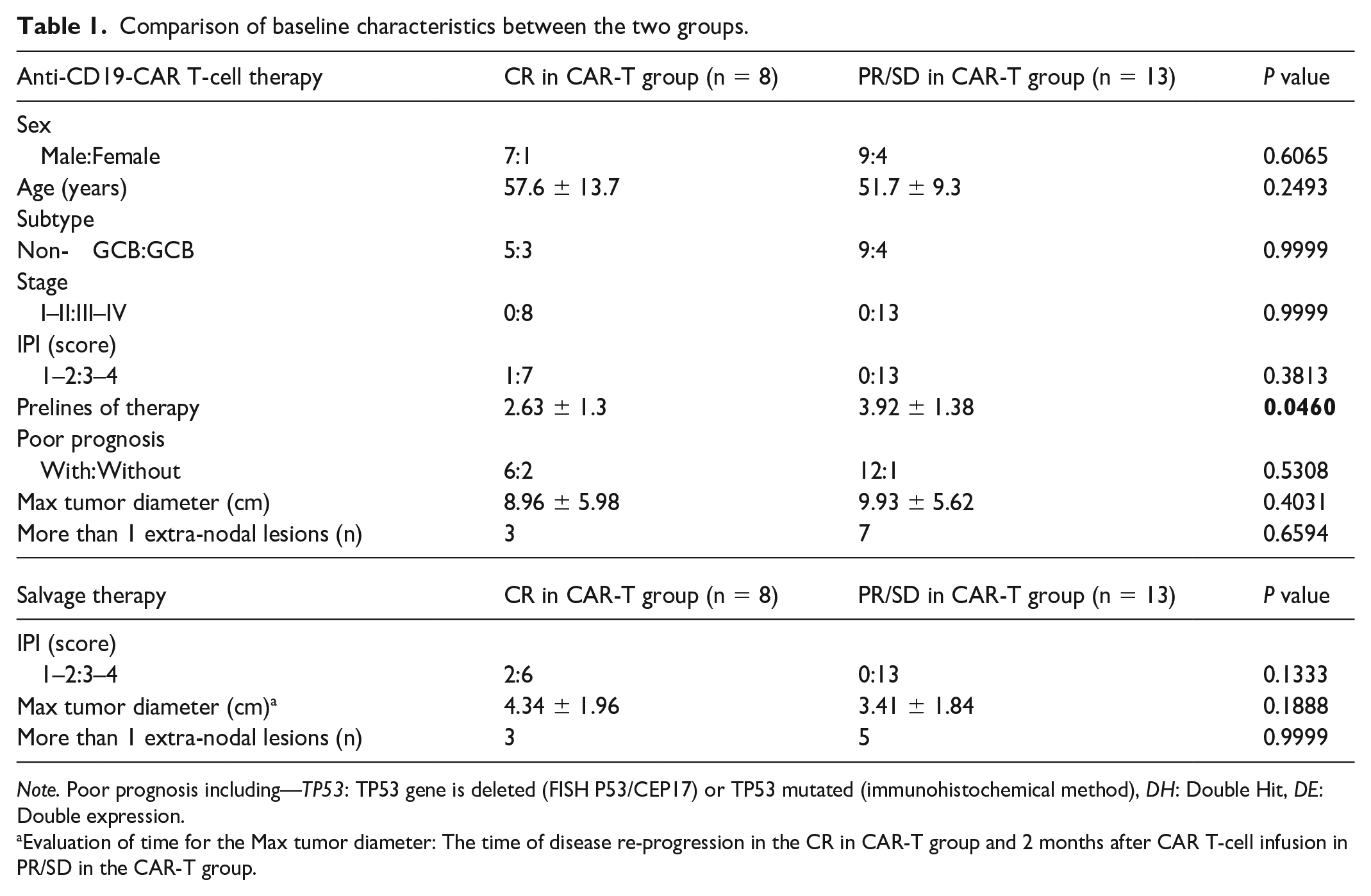
Note. Poor prognosis including—TP53: TP53 gene is deleted (FISH P53/CEP17) or TP53 mutated (immunohistochemical method), DH: Double Hit, DE: Double expression.
Evaluation of time for the Max tumor diameter: The time of disease re-progression in the CR in CAR-T group and 2 months after CAR T-cell infusion in PR/SD in the CAR-T group.
Transduction efficiency of anti-CD19-CAR and infusion dose
On the harvest date, the mean transduction efficiency of anti-CD19-CAR in all enrolled patients was 49.07 ± 8.37%. On the day of infusion, all 21 patients intravenously received 2.19 ± 0.36 × 106 cells/kg autologous anti-CD19-CAR T cells (CAR-positive cells).
Clinical responses to anti-CD19-CAR T-cell therapy
Two months after CAR T-cell infusion, the 44 patients with R/R DLBCL received the anti-CD19-CAR T-cell therapy, 20 patients (45.45%, 20/44) obtained CR, 13 patients (29.95%, 13/44) obtained PR, 4 (9.09%, 4/44) patients obtained SD, and 7 patients (15.91%, 7/44) obtained PD only. Despite 4 of the 13 patients having normal lymph node sizes, indicative of a PR, PET-CT scans revealed a positive radiation concentration, suggesting ongoing cancer activity. These four patients did not receive salvage therapy and achieved CR 3 months after CAR T-cell infusion. The other nine patients evaluated for PR were those whose tumor volume did not reduce or increase 2 months after CAR T-cell infusion.
Anti-CD19-CAR T-cell amplification in anti-CD19-CAR T-cell therapy
In the anti-CD19-CAR T-cell therapy, the proportion of anti-CD19-CAR T cells in the peripheral blood was detected 0, 7, 14, 28, and 60 days after CAR T-cell infusion in all 21 patients (Fig. 2a). The levels of anti-CD19-CAR T cells were significantly elevated in the PR group of patients who responded to PD-1 inhibition compared to those in the PR group with no response to PD-1 and the SD group, as indicated by a P value of 0.0005 and less than 0.0001, respectively, but there was no difference in the peaks of CAR T cells between the CR in CAR-T and PR in CAR-T (effective to PD-1) groups (Fig. 2b). There was no difference in the percentage of CAR-T cells 7 days after infusion among the four groups (Fig. 2c). But 14 days after the infusion, the percentage of CAR-T cells in the PR in CAR-T (effective to PD-1) group was higher than that in the CR in CAR-T group (P = 0.5232). But it was higher in the PR in CAR-T (effective to PD-1) group than that in the PR in CAR-T (no response to PD-1) and SD in CAR-T groups (0.0305 and 0.0202, respectively; Fig. 2d). We calculated the ratio of CAR-T cells in each group on day 7/day 14. This ratio was lower in the PR in CAR-T (effective to PD-1) group than in the PR in CAR-T (no response to PD-1) and SD in CAR-T groups (P = 0.0436 and 0.0448, respectively). However, there was no difference in this ratio between the PR in CAR-T (no response to PD-1) and the SD in CAR-T groups (P = 0.9463; Fig. 2e).

Anti-CD19-CAR T-cell amplification. (a) The proportion of anti-CD19-CAR T cells was detected 0, 7, 14, 28, and 60 days in anti-CD19-CAR T-cell therapy. (b) The peaks of anti-CD19-CAR T-cells were higher in the PR in CAR-T (effective to PD-1) group than those in the PR in CAR-T (no response to PD-1) group and in the SD in CAR-T group. (c) There was no difference of the percentage of CAR-T cells at 7 days after infusion between the four groups. (d) The percentage of CAR-T cells in the PR in CAR-T (effective to PD-1) group was higher 14 days after CAR T-cell infusion. (e) The ratio of the proportion of CAR-T cells on day 7 to day 14 was lower in the PR in CART (effective to PD-1) group.
Expression of PD-1 and CD3+ T cells before and during anti-CD19-CAR T-cell therapy
Before the anti-CD19-CAR T-cell therapy, there was no difference in PD-1 expression in CD3+ T cells among the four groups (Fig. 3a). Before the PD-1 inhibitor-based salvage therapy, there was no difference in PD-1 expression in CD3+ T cells among the four groups (Fig. 3b). After the initial salvage therapy, the disease burden was significantly reduced in all 21 patients with R/R DLBCL, with the specific measure of disease activity decreasing to less than 1.0%. This reduction was sustained throughout subsequent salvage therapies.

Expression of PD-1 and CD3+ T cells. (a and b) There was no difference of PD-1 expression in four groups before CAR T-cell therapy and before salvage therapy. (c) There was no difference in the percentage of CD3+ T cells in four groups before CAR T-cell therapy. (d) In CAR T-cell therapy, the peak percentage of CD3+ T cells was higher in the PR in CAR-T (effective to PD-1) group and in the CR in CAR-T group than that in the other two groups. (e and f) Before the first- and second-time salvage therapy, the percentage of CD3+ T cells was higher in the PR in CAR-T (effective to PD-1) group than that in the other three groups.
We observed the percentage of CD3+ T cells in the peripheral blood before anti-CD19-CAR T-cell therapy and at the time of CAR T-cell expression peak in anti-CD19-CAR T-cell therapy. There was no difference in CD3+ T-cell expression among the four groups before the anti-CD19-CAR T-cell therapy (Fig. 3c). At the time of CAR T-cell expression peak in anti-CD19-CAR T-cell therapy, the percentage of CD3+ T cells was higher in the PR group receiving CAR T-cell therapy effective against PD-1 than in the PR group showing no response to PD-1 and SD in CAR-T groups (P = 0.0012 and 0.0002, respectively) in anti-CD19-CAR T-cell therapy. However, there was no difference between the CR in CAR-T group and PR in CAR-T (effective to PD-1) group (Fig. 3d).
Cytokine secretion in anti-CD19-CAR T-cell therapy
The secretion of IL-6 and TNF-α during the anti-CD19-CAR T-cell therapy is shown in Fig. 4a, c. In the CAR T-cell therapy, the mean peak of IL-6 and the mean peak of TNF-α in the PR in CAR-T (effective to PD-1) group were higher than those of the SD in CAR-T group (P = 0.0315 and P = 0.0081, respectively). However, there was no difference between the PR in CAR-T (effective to PD-1), CR in CAR-T, and PR in CAR-T (no response to PD-1) groups (Fig. 4b, d).

Cytokine secretion in anti-CD19-CAR T-cell therapy and salvage therapy. (a and c) The secretion of IL-6 and TNF-α in anti-CD19-CAR T-cell therapy. (b and d) In anti-CD19-CAR T-cell therapy, there was no difference in the mean peak of IL-6 or TNF-α between the three groups except SD in CAR-T group. (e and g) In salvage therapy, the secretion of IL-6 and TNF-α in first-time salvage therapy. (f) In salvage therapy, the mean peak of IL-6 in the PR in CAR-T (effective to PD-1) group was higher than that in the other three groups. (h) In salvage therapy, there was no difference in the mean peak of TNF-α between the PR in CAR-T (effective to PD-1) group and the CR in CAR-T group and the PR in CAR-T (no response to PD-1) group.
AEs in anti-CD19-CAR T-cell therapy
All patients with R/R DLBCL who experienced AEs during the anti-CD19-CAR T-cell therapy recovered 12–23 days after CAR T-cell infusion. There was no difference in the AEs in the CR in CAR-T and PR/SD in CAR-T groups after the anti-CD19-CAR T-cell therapy (Table 2). None of the patients who received the CAR T-cell therapy died of bacterial infections, and none were diagnosed with invasive fungal diseases. There was no difference in the CRS grades in the CR in CAR-T, PR in CAR-T (effective to PD-1), and PR in CAR-T (no response to PD-1) groups. The CRS grade in the SD-CAR-T group was lower than that in the PR-CAR-T group (effective against PD-1) (Fig. 5a, b). However, there was no difference in the ICANS grades among the four groups (Fig. 5a, c). Only patients diagnosed with grade 3 CRS received tocilizumab (4 mg/kg per day, for 1–3 days), and two patients diagnosed with grade 1 ICANS received dexamethasone (5 mg, only once).
Grades 3 and 4 of AEs in CAR T-cell therapy and salvage therapy (first two cycles).

Note. The AEs in CAR T-cell therapy were according to the National Cancer Institute’s Common Terminology Criteria for Adverse Events (version 4.03). The AEs in PD-1 salvage therapy were evaluated according to the CTCAE. The causal relationship between study drugs and AEs was determined by the investigator.

AEs in anti-CD19-CAR T-cell therapy and salvage therapy. (a) The CRS and ICANS grades in anti-CD19-CAR T-cell therapy; the CTCAE grades in PD-1 inhibitor-based salvage therapy. (b) There was no difference in the CRS grades in the three groups except in the SD in CAR-T group. (c) There was no difference in the ICANS grades between the four groups. (d) There was no difference in the CTCAE grades between the four groups.
Salvage therapy
Clinical responses to salvage therapy
Of the 44 patients, 21 who received salvage therapy were selected: eight achieved CR but experienced disease progression after CAR T-cell therapy, nine achieved PR, and four achieved SD. Eight patients with R/R DLBCL (Patients 1–8) who achieved CR after CAR T-cell therapy experienced disease re-progression at 2–9 (6.0 ± 2.1) months after CAR T-cell infusion. All the other 13 patients with R/R DLBCL (Patients 9–21) who achieved PR or SD after CAR T-cell therapy received salvage therapy 2 months after CAR T-cell infusion (Fig. 6a). The treatment options (single PD-1 inhibitors or PD-1 inhibitors combined with ICE chemotherapy) and cycles of salvage therapy were shown in Fig. 6.

Clinical responses to anti-CD19 CAR T-cell therapy and salvage therapy. (a) The details of salvage treatment in all patients. In the CR from CAR-T group, only one patient obtained PR in salvage therapy after their disease re-progression. In the PR/SD from CAR-T group, four patients obtained CR, and one patient kept PR in their salvage therapy. (b) The ORR of this salvage therapy was 28.57%; CR rate was 19.05% in all the patients. The ORR and CR rate was 38.46% and 30.77%, respectively, in the PR/SD in CAR-T group. (c) The OS was higher in the PR/SD in CAR-T group than that in the CR in CAR-T group.
Of the eight patients who achieved CR after the anti-CD19-CAR T-cell therapy and experienced re-progression of their disease, only one patient (Patient 4) achieved PR in salvage therapy. The patient received allogeneic HSCT and survived until the cutoff date. None of the other seven patients (Patients 1–3, 5–8) responded to the salvage therapy. Five patients (Patients 13, 16, 18, 19, and 21) who achieved PR in CAR T-cell therapy achieved CR in their salvage therapy. Four patients who achieved PR and four patients who achieved SD in their CAR T-cell therapy died of progressive disease in their salvage therapy (Fig. 6a).
The ORR and CR of salvage therapy were 28.57% and 23.81%, respectively. The ORR and CR were only 12.5% and 0%, respectively, in the eight patients who achieved CR with the CAR T-cell therapy and received their salvage therapy when they experienced disease re-progression. However, in the 13 patients who achieved PR/SD with the CAR T-cell therapy and received salvage therapy 2 months after CAR T-cell infusion, the ORR and CR were 38.46% and 38.46%, respectively (Fig. 6b). The OS was higher in the PR/SD in CAR T-cell therapy group than that in the CR in CAR T-cell therapy group after their inhibitor-based salvage therapy (P = 0.0028) (Fig. 6c).
Unfortunately, the number of patients who received single PD-1 inhibitor salvage therapy was very small, and we did not compare the difference in responses between patients with and without PD-1 inhibitors combined with chemotherapy.
Anti-CD19-CAR T-cell amplification in salvage therapy
Of the 21 patients with R/R DLBCL who achieved salvage therapy when their disease failed or progressed after the anti-CD19-CAR T-cell therapy, only Patient 13 showed re-amplification of CAR-T cells at 3.5% after the first-time PD-1 inhibitors monotherapy. The percentage of CAR-T cells progressively decreased in subsequent salvage therapies (Fig. 7a). The patient achieved PR two months after anti-CD19-CAR T-cell infusion and achieved CR twelve months after CAR-T cell infusion (Figure 7b-e).
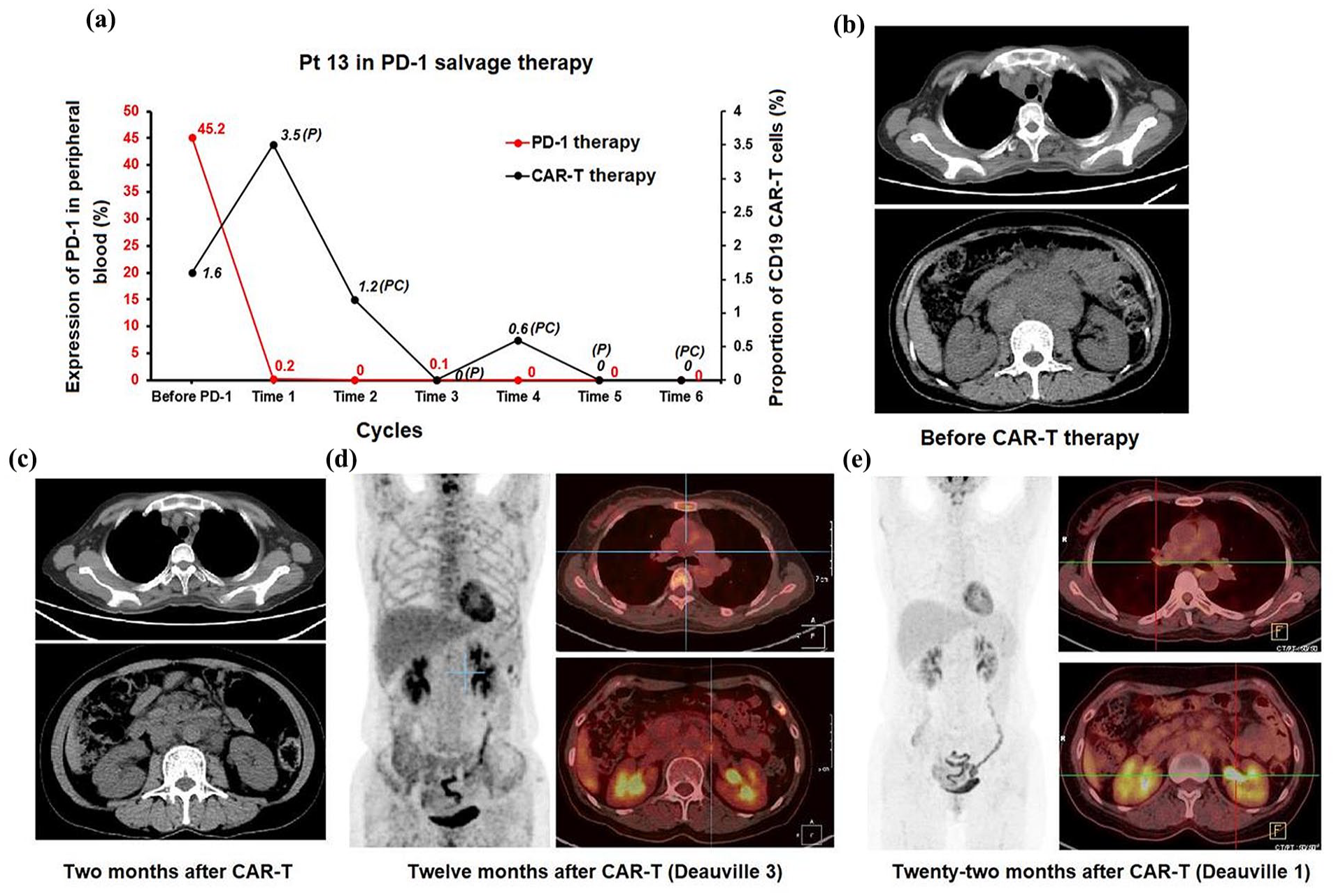
CAR-T cells and PD-1 expression, CT and PET-CT results of Patient 13. (a) Patient 13 had a re-amplification of percentage of CAR-T cells at 3.5% after her first-time PD-1 inhibitors monotherapy. (b) CT results before CAR-T therapy. (c) CT results 2 months after CAR-T therapy. (d) PET-CT results 12 months after CAR-T therapy. (e) PET-CT results 22 months after CAR-T therapy.
We did not observe the amplification of CAR-T cells in other patients during the PD-1 inhibitor-based salvage therapy.
Expression of CD3+ T cells before salvage therapy
We observed the percentage of CD3+ T cells in the peripheral blood before the first and second salvage therapies. Prior to the initial and subsequent salvage therapies, the peak percentage of CD3+ T cells was elevated in the CAR-T (effective against PD-1) group compared to that in the other three groups, as depicted in Fig. 3e, f.
Cytokine secretion during salvage therapy
The secretion of IL-6 and TNF-α during the initial salvage therapy is detailed in Fig. 4e, g, which is consistent with findings in Myasthenia Gravis patients where these cytokines’ levels are indicative of disease progression and prognosis. The mean peak of IL-6 in the PR in CAR-T (effective to PD-1) group was higher than that in the other three groups (P = 0.0009, P = 0.0007, and P < 0.0001, respectively; Fig. 4f). The mean peak of TNF-α in the PR in CAR-T (effective to PD-1) group was higher than that in the SD in CAR-T group (P = 0.0073) during the salvage therapy. However, there was no difference between the mean peak of TNF-α in the PR in CAR-T (effective to PD-1), CR in CAR-T, and PR in CAR-T (no response to PD-1) groups (Fig. 4h).
AEs in salvage therapy
The 21 patients with R/R DLBCL exhibited symptoms including fever, fatigue, rash, nausea, elevated serum transaminase and bilirubin levels, cough, and hematological toxicity during the salvage therapy (Table 2). No significant difference was observed in symptoms between patients achieving CR and those with PR or SD in the CAR-T groups. Six patients experienced grade 1 CTCAE toxicity, with only one patient (Patient 19) experiencing grade 2 toxicity (Fig. 5a). There was no difference in the CTCAE grades among the four groups (Fig. 5d). Antipyretic drugs and symptomatic treatments were selected to alleviate AEs. Patient 13 and Patient 19 were administered methylprednisolone to manage AEs. None of the 21 patients with R/R DLBCL discontinued the salvage therapy with PD-1 inhibitors because of side effects. Grade 1 of hypothyroidism occurred in only one patient (Patient 21) who achieved CR after his salvage therapy. The patient underwent seven cycles of PD-1 inhibitors monotherapy, followed by seven cycles of PD-1 inhibitors in combination with ICE chemotherapy. After the completion of PD-1 inhibitor-based salvage therapy, the patient’s hypothyroidism symptoms spontaneously resolved without specific treatment.
Discussion
The ORR of patients with R/R DLBCL with high tumor loads shows a poor response to the anti-CD19-CAR T-cell therapy22–24. Salvage therapies for patients with R/R DLBCL who failed to achieve CR or experienced disease progression following anti-CD19-CAR T-cell therapy remain under investigation7,25. In a salvage therapy study, 100 patients with R/R DLBCL with PD after the anti-CD19-CAR T-cell therapy subsequently received salvage therapy. Salvage therapies included checkpoint inhibitors, lenalidomide, chemotherapy, or radiotherapy 26 . In this study 27 , the ORR was 29%, CR was 17%, and median PFS was 55 days; the efficacy needs to be improved. In a single PD-1 inhibitor salvage therapy study, 12 patients with R/R B-cell lymphomas after the anti-CD19-CAR T-cell therapy received pembrolizumab 200 mg every 3 weeks. The best ORR after pembrolizumab treatment was 25% (8.3% CR and 16.7% PR), indicating that pembrolizumab was well tolerated 27 . In another single PD-1 inhibitor salvage therapy study, five patients with R/R DLBCL with disease progression following the CAR T-cell therapy received a single PD-1 inhibitors salvage therapy. Two of the five patients obtained CR 28 . Recent studies have investigated the therapeutic efficacy and potential side effects of PD-1 inhibitors as a rescue therapy for patients who have experienced CAR-T therapy failure. However, the initiation time of this salvage therapy, the PD-1 inhibitor-based combination therapy, and the patient selection have not yet been reported.
The efficacy of the single PD-1 inhibitors salvage therapy must be improved. Therefore, we opted for the combination of PD-1 inhibitors and chemotherapy for R/R DLBCL patients with a high tumor burden. In our study, we selected PD-1 inhibitors combined with ICE combination chemotherapy in patients with R/R DLBCL who had disease progression or did not achieve CR after the CAR T-cell therapy. The ORR in our study was 28.57%, and the CR was 19.05%. We compared the efficacy and side effects of the salvage therapy in patients with R/R DLBCL with different disease states and obtained interesting results. In all patients who achieved CR with the anti-CD19-CAR T-cell therapy and experienced disease re-progression, the ORR and CR of the salvage therapy were only 12.5% and 0%, respectively. However, in the 13 patients who did not achieved CR with the anti-CD19-CAR T-cell therapy, the ORR and CR rates were 38.46% and 30.77%, respectively. The initial time of receiving the salvage therapy was different: 6.0 ± 2.1 months after CAR T-cell infusion in the CR in CAR T-cell group and 2 months after CAR T-cell infusion in the PR/SD in CAR T-cell group. It was reported that pembrolizumab was administered to a refractory DLBCL patient who failed to respond to anti-CD19-CAR T-cell therapy at 26 days after CAR T-cell infusion. Following pembrolizumab treatment, the patient showed a clinically significant antitumor response and expansion of CAR-T cells 29 . However, in our study, except of patient 13, we did not observe the amplification of CAR-T cells in other patients undergoing the PD-1 inhibitor-based salvage therapy.
The combination regimen of PD-1 inhibitors with chemotherapy has not yet been extensively adopted in the treatment of R/R DLBCL. A large multicenter retrospective analysis has demonstrated that post-checkpoint blockade therapy (CBT) regimens, which include chemotherapy, targeted therapy, clinical trials, transplant conditioning, and CAR T-cell therapy, significantly enhance the objective response rate and prolong the duration of response (DOR) 30 . This is in line with findings from other studies, such as the efficacy of PD-1 inhibitors combined with radiotherapy in treating oligometastatic nasopharyngeal carcinoma and the improved ORR and PFS observed with PD-1 inhibitors plus chemotherapy in recurrent/metastatic NPC. CBT remains systemically active for several months, potentially constituting one of the mechanisms underlying these clinical observations.al studies 31 . However, the underlying mechanism of combining CBT with chemotherapy has yet to be fully explored.
In our study, we observed the expression of PD-1 and CD3+ T cells in the anti-CD19-CAR T-cell and salvage therapy groups. Before the CAR T-cell therapy and the subsequent salvage therapy, there was no difference in PD-1 expression in CD3+ T cells among the four groups. The CR in CAR-T and PR in CAR-T (effective to PD-1) groups had higher peaks of the percentage of CD3+ T cells in anti-CD19-CAR T-cell therapy. Our research showed that the PR in CAR-T (effective to PD-1) group had higher peaks. Consider the percentage of CD3+ T cells prior to the salvage therapy. High expression of CD3+ T cells might be an effective mechanism for the PD-1 inhibitor-based salvage therapy in patients following the anti-CD19-CAR T cell therapy. The IL-6 and TNF-α secretion increased again during the salvage therapy, especially the IL-6 levels in the PR in CAR-T (effective to PD-1) group. This might also explain the best response to salvage therapy in this group.
PD-1 inhibitor-based therapy could serve as a promising salvage option for patients with R/R DLBCL after anti-CD19-CAR T-cell therapy, particularly for those who achieved a PR during CAR T-cell therapy and promptly underwent this salvage approach. For patients with R/R DLBCL who experience CAR T-cell therapy failure or disease re-progression, PD-1 inhibitors or combination chemotherapy initially and a therapeutic schedule of salvage therapy remain to be explored further. All patients with progressive or ineffective disease in their anti-CD19-CAR T-cell therapy have received salvage therapy. To mitigate the impact of confounding factors, we opted not to include an untreated control group. Instead, we focused on increasing the case count and maintaining ongoing observation and exploration.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251338713 – Supplemental material for An exploration of the initiation time and patient selection of PD-1 inhibitors/PD-1 inhibitors combined with chemotherapy as salvage therapy in R/R DLBCL patients after anti-CD19-CAR T-cell therapy
Supplemental material, sj-docx-1-cll-10.1177_09636897251338713 for An exploration of the initiation time and patient selection of PD-1 inhibitors/PD-1 inhibitors combined with chemotherapy as salvage therapy in R/R DLBCL patients after anti-CD19-CAR T-cell therapy by Xin Li, Juan Mu, Jia Wang, Qing Li, Yili Jiang, Jingyi Li and Qi Deng in Cell Transplantation
Footnotes
Ethical Considerations
This study was approved by the Medical Ethics Committee of the Department of Hematology, Tianjin First Center Hospital (Tianjin, China). (approval no. of the ethic committee: 2018N105KY and 2019N062KY). The patient gave their written informed consent in accordance with the Declaration of Helsinki. The Clinical trial in our study was registered at ChiCTR1800019622 and ChiCTR1900025310.
Consent for Publication
All the patients agreed to the use of their specimens and data for our study.
Author Contributions
Concept and design: DQ and LX. Drafted or revised the manuscript: MJ, LQ, and WJ. Acquisition of data: JYL. Analysis and interpretation of data: MJ and LJY. Writing, review, and/or revision of manuscript: LX. Study supervision: DQ.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was sponsored by the Tianjin Health Research Project (TJWJ2023ZD003); Chinese Society of Clinical Oncology Beijing Xisike Clinical Oncology Research Foundation (Y-NCJH202201-0027 and Y-2022YMJN/MS-0001).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Provenance and Peer Review
Not commissioned, externally peer-reviewed.
Data Availability Statement
Data are available on reasonable request.
Open Access
This is an open-access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, and build upon this work non-commercially and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See ![]() .
.
Supplemental Material
Supplemental material for this article is available online. This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names, and drug dosages) and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
