Abstract
The conventional approach for addressing bone defects and stubborn non-unions typically involves the use of autogenous bone grafts. Nevertheless, obtaining these grafts can be challenging, and the procedure can lead to significant morbidity. Three primary treatment strategies for managing bone defects and non-unions prove resistant to conventional treatments: synthetic bone graft substitutes (BGS), a combination of BGS with bioactive molecules, and the use of BGS in conjunction with stem cells. In the realm of synthetic BGS, a multitude of biomaterials have emerged for creating scaffolds in bone tissue engineering (TE). These materials encompass biometals like titanium, iron, magnesium, and zinc, as well as bioceramics such as hydroxyapatite (HA) and tricalcium phosphate (TCP). Bone TE scaffolds serve as temporary implants, fostering tissue ingrowth and the regeneration of new bone. They are meticulously designed to enhance bone healing by optimizing geometric, mechanical, and biological properties. These scaffolds undergo continual remodeling facilitated by bone cells like osteoblasts and osteoclasts. Through various signaling pathways, stem cells and bone cells work together to regulate bone regeneration when a portion of bone is damaged or deformed. By targeting signaling pathways, bone TE can improve bone defects through effective therapies. This review provided insights into the interplay between cells and the current state of bioceramics in the context of bone regeneration.
Introduction
Bone tissue serves multiple crucial functions in the body, including providing support for muscles, enabling movement and locomotion, safeguarding internal organs, housing bone marrow, and regulating mineral balance and various endocrine functions 1 . Bone tissue exhibits a hierarchical structure, encompassing cancellous and cortical bone at the macroscopic level (Fig. 1). These two types of bone coexist in varying proportions within each bone type 2 . The structural disparities between cortical and trabecular bone extend to their porosity and metabolic activity. Trabecular bone displays higher porosity and lower density, rendering it more flexible but weaker than cortical bone. Furthermore, the trabecular bone exhibits greater metabolic activity. This structural divergence mirrors their respective functions 3 . The sub-microstructure consists of lamellae composed of mineralized collagen fiber sheets arranged in a planar configuration. These crystals can be intra-fibrillar or extra-fibrillar, depending on their location relative to collagen fibrils 4 . Bone tissue qualifies as specialized connective tissue, characterized at the molecular level by an organic phase comprising approximately 90% type I collagen and roughly 5% non-collagenous proteins 5 . Bone diseases encompass various skeletal disorders, some of which severely impact mobility and survival. Common conditions like osteosarcoma and metastatic bone cancer lack effective treatments, necessitating the development of novel drugs and drug delivery systems that balance treatment efficacy with side effects. Bone loss poses a significant challenge in orthopedic and dental contexts, including periodontitis6,7, osteonecrosis, and osteoporosis, incurring substantial financial and social costs 8 . Large bone defects resulting from accidents, bone tumors, and other factors necessitate intervention therapy for recovery. However, the use of autogenous bone grafts or autografts, considered the “gold standard,” is limited due to supply constraints 9 . As a result, researchers have developed innovative tissue engineering (TE) strategies for regenerating bone tissue. Cell adhesion and proliferation are enhanced by scaffolds, which provide a three dimensional (3D) environment 10 . Ceramic materials have proven successful in addressing bone tissue defects, falling under the umbrella of bioceramics. Bioceramics encompass inorganic, non-metallic materials with biocompatibility, high melting points, electrical resistance, and corrosion resistance 11 . While ceramic scaffolds offer promise for various applications in oral and maxillofacial surgery, periodontal treatments, and orthopedics, their brittle nature restricts their use to non-load-bearing applications12,13. The resemblance between calcium phosphate (CP) like hydroxyapatite (HA) and tricalcium phosphate (TCP) and mineral bones makes them the most popular bioceramics for bone grafting 14 . Silicate bioceramics have also gained attention for hard tissue regeneration. The ability to adjust mechanical properties, bioactivity, and degradation rate through variations in chemical composition contributes to the versatility of bioceramics. To enhance scaffold performance, composite materials combine bioceramics and polymers. Bioceramic scaffolds can also be incorporated with growth factors (GFs) that promote osteogenesis and angiogenesis, and these scaffolds can serve as carriers for stem cells that help to accelerate bone repair by promoting osteogenesis and angiogenesis (Fig. 2) 15 . Moreover, these scaffolds can be tailored to exhibit macrostructures, microstructures, and nanostructures, enabling cell adhesion and nutrient transport and promoting bone tissue ingrowth. Nanostructures and microstructures enhance surface roughness and area, facilitating osteoblast adhesion to scaffold surfaces 16 . Reviewing the current state of the research field is imperative for several reasons. First, as technology and scientific understanding advance, new bioceramic materials are continually developed and introduced into the field of bone regeneration. Understanding the specific cellular interactions with these contemporary bioceramics is crucial for optimizing their application and enhancing bone regeneration outcomes. Second, advancements in in vitro, in vivo, and clinical studies provide valuable insights into the performance and effectiveness of bioceramics in different contexts. A thorough review of the existing literature allows researchers to stay abreast of the latest findings, methodologies, and challenges, enabling them to build upon existing knowledge and contribute to the refinement of bioceramic-based bone-regeneration strategies. Lastly, by reviewing the current state of the research field, gaps in knowledge, inconsistencies, and potential areas for improvement can be identified. This critical assessment sets the foundation for future research directions, guiding scientists toward addressing unanswered questions and refining experimental approaches. Overall, a comprehensive review ensures that the research community is well informed, facilitating the progression and innovation within the field of bioceramics and bone regeneration. The aim of this review article is to investigate and comprehensively analyze the cross talk between cells and contemporary bioceramics in the context of bone regeneration. This investigation will encompass in vitro, in vivo, and clinical studies, aiming to elucidate the intricate cellular interactions with the current generation of bioceramic materials employed in bone-regeneration processes.
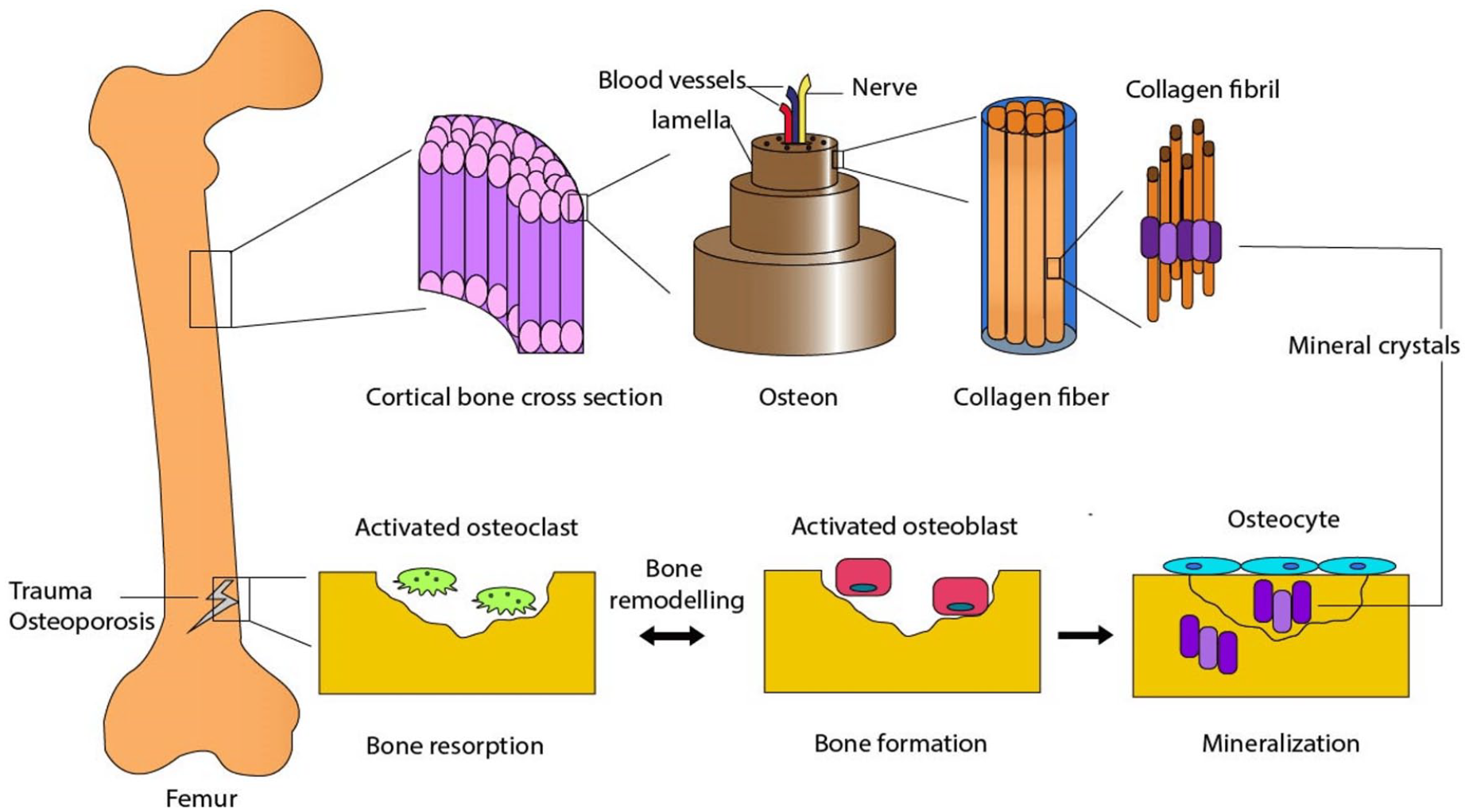
Bone formation and remodeling: hierarchical structure and mechanisms 17 .
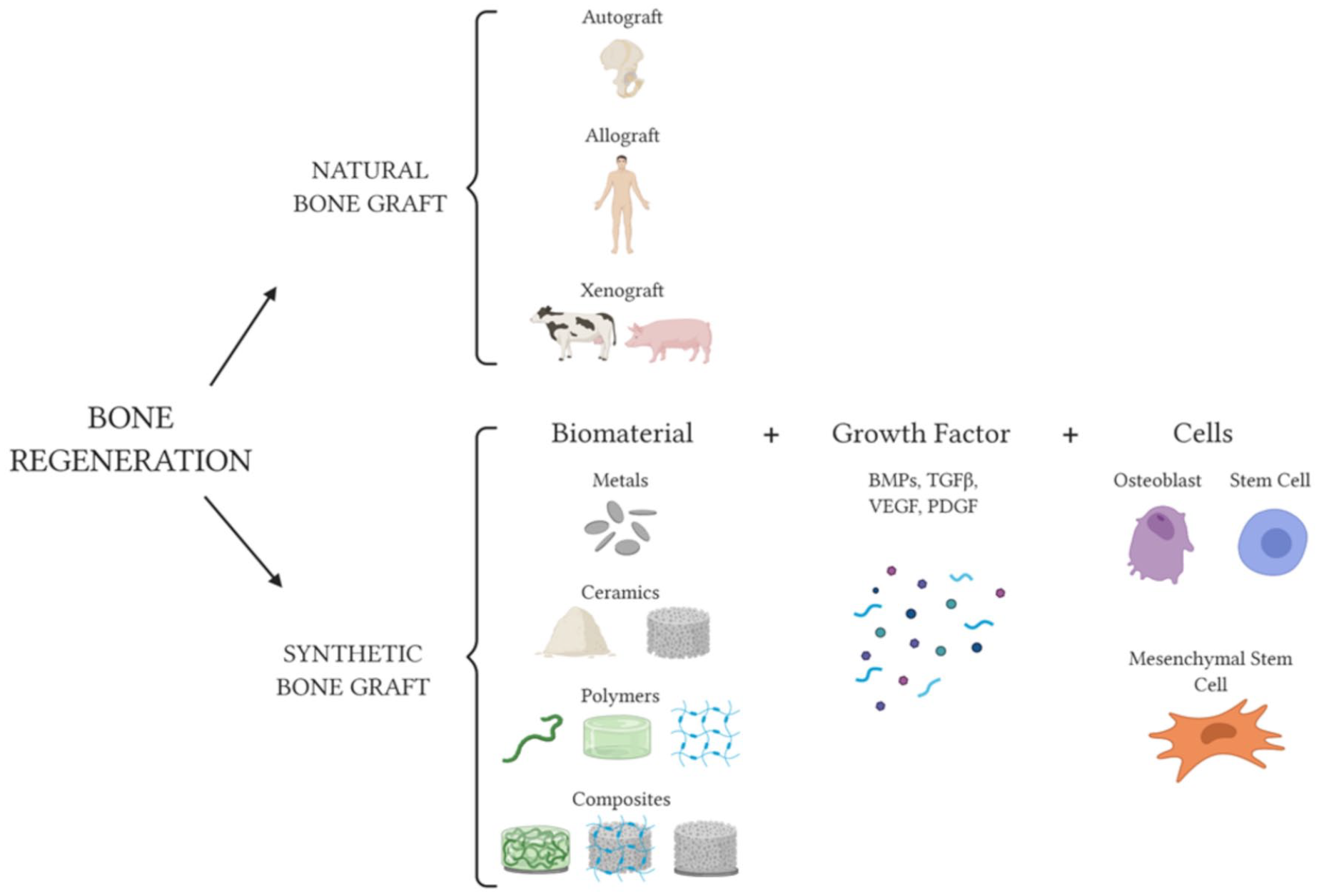
Regeneration options for bone tissue are shown in a representative scheme. An autograft or allograft derived from a human donor, as well as a xenograft derived from an animal, are all-natural bone grafts 5 .
Study Design
Databases PubMed and Web of Science were used in the present review using the following keywords “Differentiation,” “Bioceramics,” “Epigenetics,” “Bone Regeneration,” “Growth Factor Delivery,” “Bioniert,” “Calcium phosphate,” “Tricalcium phosphate,” “HA,” “Bioresorbable,” “Bioactive,” and “Bioglass.” Those articles were not relevant due to emphasis on the delivery system, or used repeated information or non-documented data were rejected. English-language articles were selected for this review article.
Bioceramic Materials and Properties
Biomedical composites incorporate ceramic reinforcements due to their inherent weakness and brittleness. These reinforcements typically take the form of particulates, and various materials, such as CPs, aluminum phosphates, zinc phosphates, glass ceramics, and bone minerals, have been utilized for this purpose. TCP and HA find widespread application in fields like orthopedics and dentistry. They are employed as coatings on metal implants 18 or in combination with other substances, earning the designation of “bioactive ceramics” because they stimulate osseointegration through specific biological responses19,20. Bioactive glass (BG) can also be incorporated into a polymeric matrix to create composite materials 21 . In dentistry, glasses have been paired with acrylic resins, while for enhancing X-ray image contrast in biocomposites, barium sulfate powder is often added. Alternatively, radiopaque metallic wires or markers serve as an alternative to radiopaque powder 22 . In a wide range of applications, bioactive synthetic materials with seamless integration have gained significant interest. Assays for bone cells in vitro, resorbable cements in vivo, coatings for implants that enhance bonding between the natural bone and the implant 23 , and agents for bone repair are included. When such materials are applied in vivo, they promote optimal healing by stimulating osteogenic activity within the surrounding bone tissues, as well as allowing osteoclasts to gradually resorb bone during the remodeling process. They provide standardized laboratory substrates for assessing and quantifying bone cell restoration and mineralization in vitro. Osteoclasts resorb bone minerals from these substrates by interacting with them within the biological environment 24 . Naturally derived bioceramics, sourced abundantly from inorganic materials found in marine life such as corals, fish bones, and sponges, hold great promise for tissue replacement and regeneration, offering a wide range of practical applications in this field 22 . In the following sections, various basic research findings of the interactions between CP-based bioceramics and various biological processes are discussed, such as redox balance, epigenetic control, or cell signaling of cellular activity. Doped HA interacts with HA, with a focus on cellular outputs as shown in Fig. 3 25 .
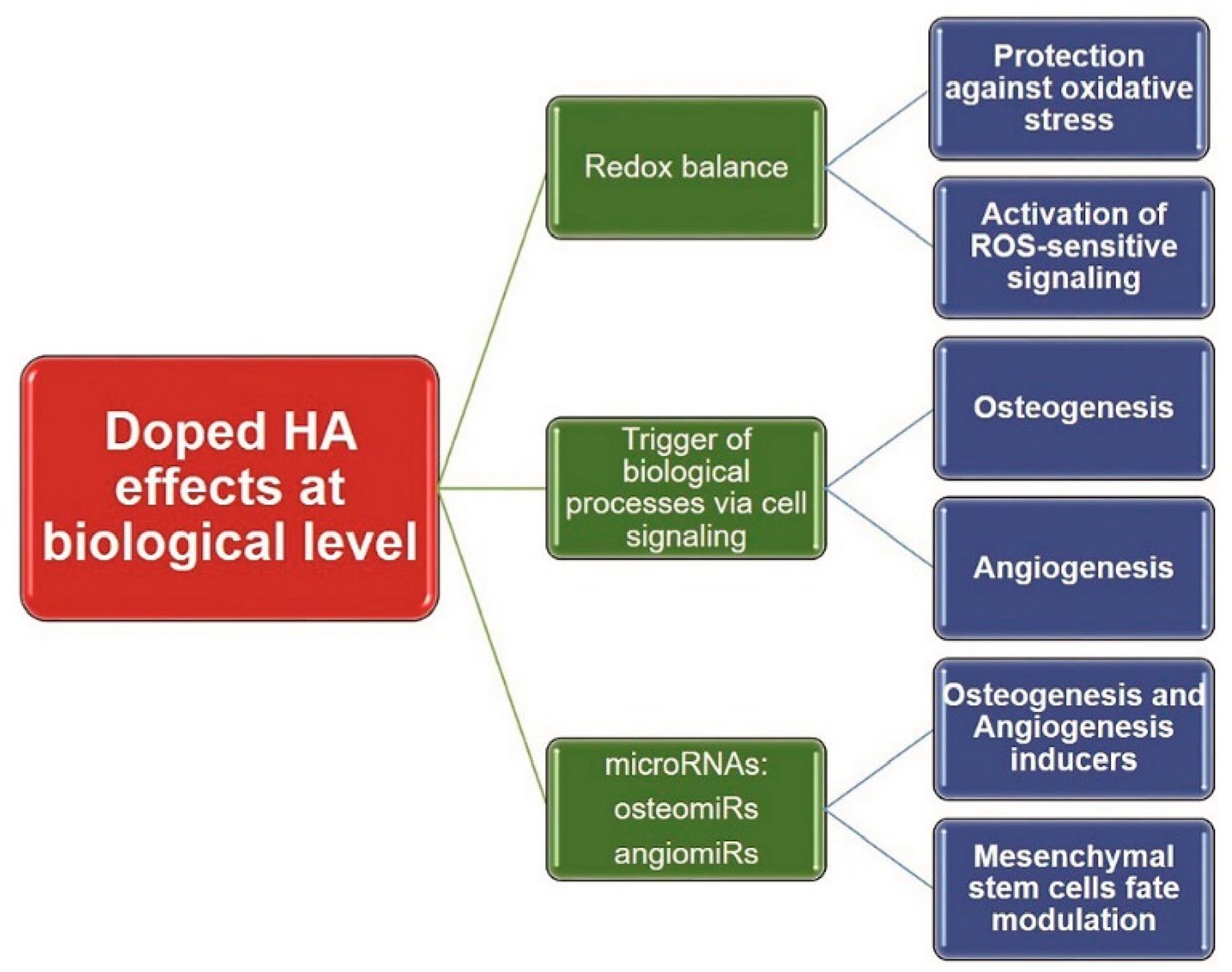
Biochemical interactions and cellular outputs of doped HA with several main biological processes 25 .
Biocompatibility of a material denotes its capacity to interact harmoniously with bodily tissues. One of the primary advantages of biocompatible materials lies in their non-toxic nature when introduced into the body. This characteristic is of utmost importance in the realm of biomaterials, as they must facilitate cellular processes while avoiding any undesirable local or systemic reactions from the host to achieve complete tissue regeneration 26 . Biocompatibility, contingent upon both the bulk and surface composition of scaffolds, encompasses a material’s ability to effectively fulfill its intended purpose, which includes exhibiting an appropriate degradation profile suitable for its designated application. CP-based biomaterials and their end products have been shown to possess short- and long-term biocompatible attributes 27 . In terms of biocompatibility, naturally occurring biomaterials hold the most promise 28 . To facilitate effective bone replacement, the scaffold must possess osteoconductive properties, allowing bone cells to securely adhere, proliferate, migrate, and integrate with it. CP-based scaffolds exhibit excellent osteoconductivity due to their chemical similarity to natural bone. Notably, enhancing the microporosity of bioceramic scaffolds further enhances their osteoconductive capabilities 29 . Osteoinductivity pertains to the capacity of biomaterials to incite osteogenesis in precursor cells. While CP-based bioceramics typically do not possess osteoinductivity as a primary characteristic, some are capable of inducing bone formation in vivo without the need for external osteogenic factors, a phenomenon referred to as “intrinsic” osteoinductivity 30 . Moreover, the osteoinductive potential can be attributed to the combined effects of bioceramics’ topography, composition, and microporosity/macroporosity, allowing for the entrapment of osteoprogenitor cells and bone morphogenetic proteins (BMPs) in vivo 31 . The osteoinductivity of CP-based bioceramics can be enhanced by incorporating magnesium within the 5–10 wt% range, which promotes adhesion and osteogenic differentiation of bone marrow–derived stem cells (BMSCs) 32 . Nevertheless, various strategies for augmenting osteoinductivity have demonstrated positive effects on promoting bone repair 33 . For a bone scaffold to effectively serve its purpose, it must possess mechanical characteristics akin to those of the natural tissue it replaces. Beyond simply ensuring temporary mechanical stability, these scaffolds play a crucial role in facilitating the transmission of mechanical forces to the healing site, thanks to their stiffness being on par with the adjacent natural tissue. When a scaffold’s strength falls short of that of the host bone, it becomes incapable of supporting the necessary physiological loads after implantation. Conversely, overly rigid scaffolds can result in stress shielding and osteolysis of the surrounding tissues 34 . Stress shielding occurs when the robust scaffold structure absorbs the majority of mechanical stresses, leaving the neighboring native tissue insufficiently stimulated for proper bone health. This, in turn, leads to bone resorption and compromised bone regeneration, ultimately resulting in implant failure due to tissue degradation around the implant site 35 . These properties collectively contribute to the effectiveness of bioceramic materials in various biomedical applications, including bone implants, dental materials, and TE (Table 1)10,15,17,36–41.
The Effect of Bioceramics in Delivering, Differentiation, and Epigenetics
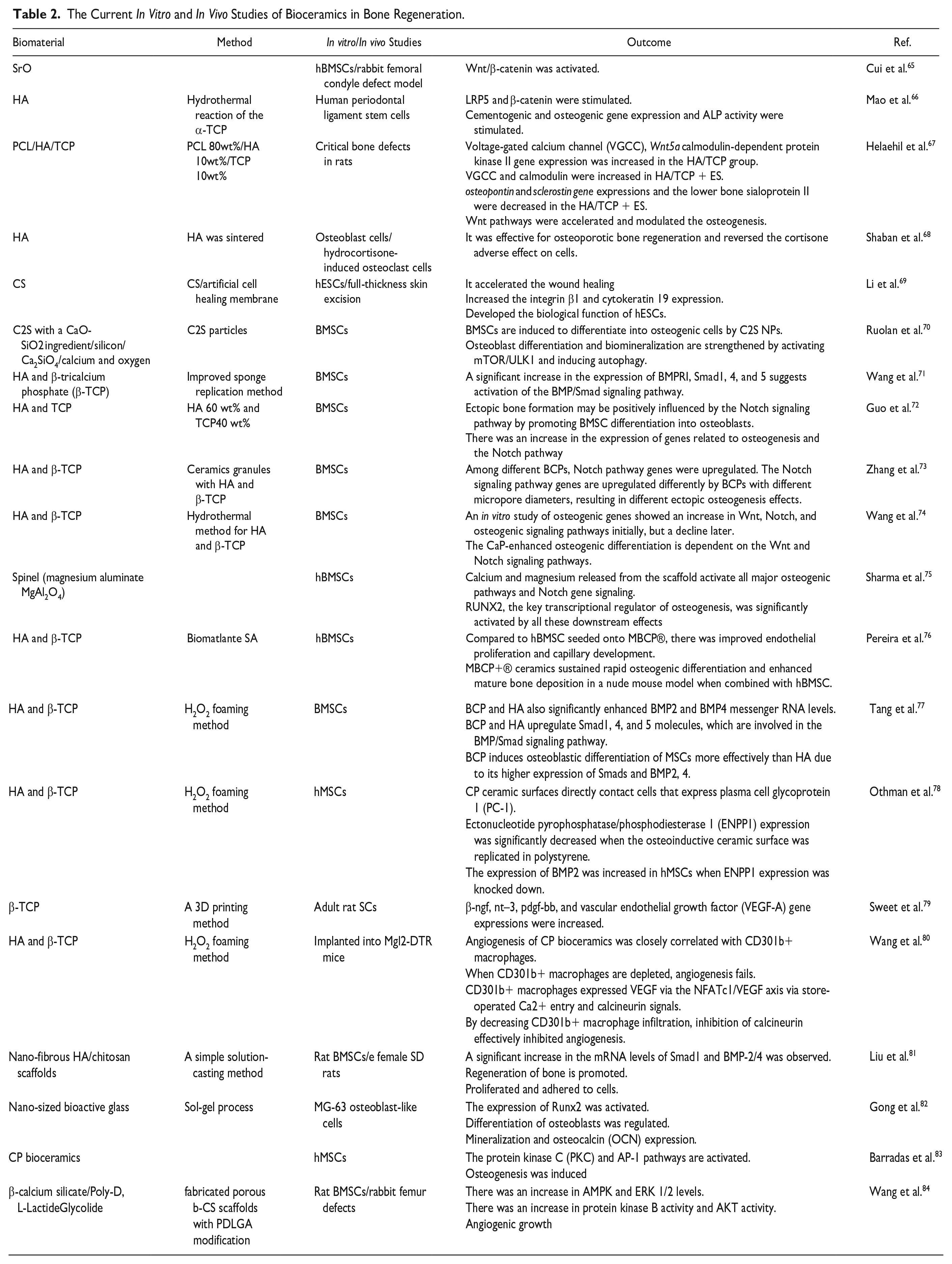
To facilitate effective bone regeneration, a collaborative interplay among cells, proteins, genes, and biophysical signals is essential. These elements combine synergistically and are naturally distributed in native tissues, relying on extracellular matrix (ECM) components. Researchers are developing biomimetic, biodegradable materials that have ECM-like properties for releasing regenerative factors at injury sites. Both clinical and translational perspectives are discussed in this section. In addition, it explores potential associated risks, considering the increasing use of biomaterials for delivering therapeutic agents 42 . Biomaterials can facilitate stem cell delivery, offering a promising avenue for therapeutic applications. Traditional direct injections of cells into saline or media have been found to pose challenges, with cells facing diminished chances of survival and engraftment due to harsh environmental factors 43 . The incorporation of biomaterials as substitutes for native ECM presents a solution, enabling encapsulated cells to adhere and thrive, thus preventing anoikis. Furthermore, biomaterials can be tailored to enhance cellular regeneration and provide immunological protection44,45. This approach, exemplified by the use of hydrogels, has demonstrated potential as a minimally invasive means of delivering stem cells to address craniofacial defects arising from trauma or disease. Notably, injectable composite hydrogels have shown promise in fostering regenerative potential in rats with critical cranial defects46,47. However, it is worth acknowledging that injectable hydrogel systems do have limitations, particularly their lack of 3D structural and mechanical stability, which can be a significant drawback in craniofacial regeneration, especially for cases involving substantial bone loss, such as traumatic injuries or cancer. Clinical trials have explored the use of stem cells and progenitor cells sourced from adult tissues as alternatives to pluripotent stem cells, which have limited clinical application due to ethical concerns and technical challenges associated with embryonic stem cells and induced pluripotent stem cell transplants. In this regard, therapies employing mesenchymal stem cells (MSCs) have gained traction. The advantages of MSCs over other cell types include their ability to differentiate, their immunomodulatory properties, and their low toxicity. MSCs are widely used to restore organ function and treat inflammation since they lack co-stimulatory molecules, are less immunogenic, and exhibit strong differentiation capacity48,49. MSCs are incorporated into several commercially available bone graft materials and among these products, Trinity Evolution Matrix, Allostem, and Map3 are notable options50,51. In addition, other bone graft materials, including collagen sponges, TCP, collagen-TCP composites, and nano-hydroxyapatite (nano-HA) collagen carriers, have shown promise as carriers for MSCs51,52. Despite persistent concerns related to long-term safety and efficacy, the use of biomaterials for stem cell delivery to oral and maxillofacial structures holds significant potential for regeneration 53 . Gene therapy could enhance brain protein synthesis in a precise and time-sensitive manner. Despite challenges such as limited bioactivity, in vivo stability issues, and rapid hepatic and renal clearance of GFs, this strategy aims to achieve localized and sustained gene expression at the site of the defect54,55. Biomaterials are often favored over viral vectors due to their reduced immunogenicity, enhanced safety profile, ease of manipulation, and reduced risk of mutagenesis despite maintaining high transfection efficiency56,57. Various materials and techniques have been explored for gene delivery, including lipids, peptides, and polymers 57 . However, biomaterial-based vectors must overcome a spectrum of obstacles at systemic, extracellular, and subcellular levels58,59. While numerous gene therapy clinical trials have been conducted, a majority of them have employed viral vectors to treat a wide range of disorders 60 . Promising outcomes have been observed in both in vitro and in vivo studies concerning nonviral biomaterial-based gene delivery. Nonetheless, achieving clinical success remains a challenge 56 . Researchers like Bonadio and colleagues investigated the effectiveness of this approach in a beagle tibia critical defect model, wherein Parathyroid hormone (PTH) 1-34/BMP-4-encoding cDNAs were physically entrapped61,62. Multifunctional therapeutic benefits can be obtained through the direct delivery of DNA complexes by biomaterial carrier systems. Future clinical applications of biomaterial-mediated gene delivery technologies hold promise for treating bone loss63,64. Some current studies are summarized in Table 2.
The Current In Vitro and In Vivo Studies of Bioceramics in Bone Regeneration.
The Cross Talk Between Bioceramics and Cells in Bone Regeneration
Wnt signaling
Development, organogenesis, and tissue maintenance are all dependent on Wnt signaling. Wnt ligands interact with 19 secreted glycoproteins to initiate intracellular signaling 85 . Cells use this signaling cascade to proliferate, differentiate, grow, survive, develop, regenerate, and self-renew 86 . Wnt signaling is believed to contribute to the healing process at different stages, influencing a wide array of cell fate decisions related to osteogenesis. Canonical Wnt ligands bind to frizzled receptors (Fzd), along with lipoprotein receptor-related protein 5 (LRP5) and LRP6 receptors associated with low-density lipoproteins (LDLs) 87 . These frizzled proteins mediate signal transmission through both independent and dependent pathways. In response to Wnt3a, adult human MSCs undergo proliferation and osteogenic differentiation 88 . In skeletal homeostasis, Wnt3a, Frizzled, and Lrp5 can form a complex with chitosan, activating the canonical Wnt pathway. Food and water contain fluorine (F), which stimulates osteoblasts 89 . In experiments, the fluorine and chitosan groups exhibited downregulated RANKL expression, facilitating osteogenic differentiation 90 . Chitosan-conjugated AuNPs induced osteogenic differentiation in human adipose-derived mesenchymal stem cells (hADMSCs) 91 . The Wnt/β-catenin signaling pathway regulates adipogenic differentiation, redirecting cells from adipocytes to osteoblasts 92 . In vivo assessments of HPDCs revealed their ability to form ectopic bones within HA-based scaffolds, driven by BMP/Wnt signaling 93 . Another study by Cui et al. evaluated bone cement containing borosilicate glass particles with varying strontium (Sr) concentrations. The inclusion of Sr in the glass resulted in a slower cement setting. hBMSCs demonstrated enhanced proliferation, mineralization, and differentiation in the presence of Sr2+. Among the formulations, SrO-substituted cement (BG6Sr) exhibited the most optimal osteogenic properties. When compared to cement with 0 mol% or 9 mol% SrO, BG6Sr cement significantly promoted peri-implant bone formation in rabbit femoral condyles. In the osteogenic differentiation of hBMSCs, a possible explanation for this effect was attributed to the activation of the Wnt/β-catenin signaling pathway. BG6Sr cement offers a minimally invasive solution for healing bone defects due to its favorable physicochemical properties and biological performance 65 . Shaban et al. introduced a microwave-assisted technology (MAT) furnace for synthesizing and sintering HA bioceramics cost-effectively and without the need for additives. Conventional sintering at the target temperature (1100°C) required 1 to 2 h, whereas microwave hybrid sintering at 100% microwave power achieved the same results in just 20–30 min. This method led to an increase in osteogenesis markers and a decrease in markers associated with cell resorption in response to HA. In addition, HA nanorods were found to reverse the effects of cortisone on death markers and cell differentiation, as well as the effects of beta-agonists. Moreover, microwave hybrid-sintered HA counteracted cortisone’s adverse effects on osteoblast cell death through both canonical and non-canonical pathways, offering promise for osteoporotic bone regeneration 68 . Li et al. conducted a study that examined the therapeutic potential of chondroitin sulfate (CS) in wound healing and re-epithelialization. They observed that CS accelerated wound healing in vivo and enhanced the expression of cytokeratin 19 and integrin b1, indicative of improved stemness in human embryonic stem cells. In vitro studies further revealed that human embryonic stem cells responded positively to CS, possibly through the activation of the EGF/EGFR/ERK signaling pathway. By stimulating the Epidermal Growth Factor (EGF)/Epidermal growth factor receptor (EGFR)/Extracellular-signal Regulated Kinase (ERK) pathway, CS promoted re-epithelialization of human embryonic stem cells and enhanced their biological function 69 . BMSC differentiation into osteoblasts was studied by Ruolan et al. using C2S nanoparticles. BMSCs were tested for osteogenic gene and protein expression using C2S NPs. It was observed that C2S NPs facilitated BMSC differentiation into osteoblasts when autophagy was active. Treatment with C2S NPs significantly upregulated the expression of LC3 and Beclin, while downregulating P62, an autophagy substrate. Although C2S NP treatment had no significant effect on alizarin red staining (ARS) activity, it did enhance ARS activity. Notably, the effects of C2S NPs were partially reversed by 3-methyladenine (3-MA). The study revealed that C2S NPs inhibited mammalian target of rapamycin (mTOR) and promoted ULK1 expression, thereby activating autophagy. This activation of autophagy by C2S NPs promoted osteogenic differentiation and bone formation in BMSCs through the mTOR/ULK1 pathway and subsequent activation of the WNT/β-catenin pathway, ultimately enhancing osteoblast differentiation and biomineralization 70 . Both diffusible signals and proper vascularization are necessary for bone formation, bone regeneration, and bone healing. Consequently, cation- and anion-substituted HA bioceramics are often investigated along with their osteoinductivity. As a result, the same signals (Fig. 4) promote osteodifferentiation, proliferation, and angiogenesis, depending on the type of receiver cell 25 .
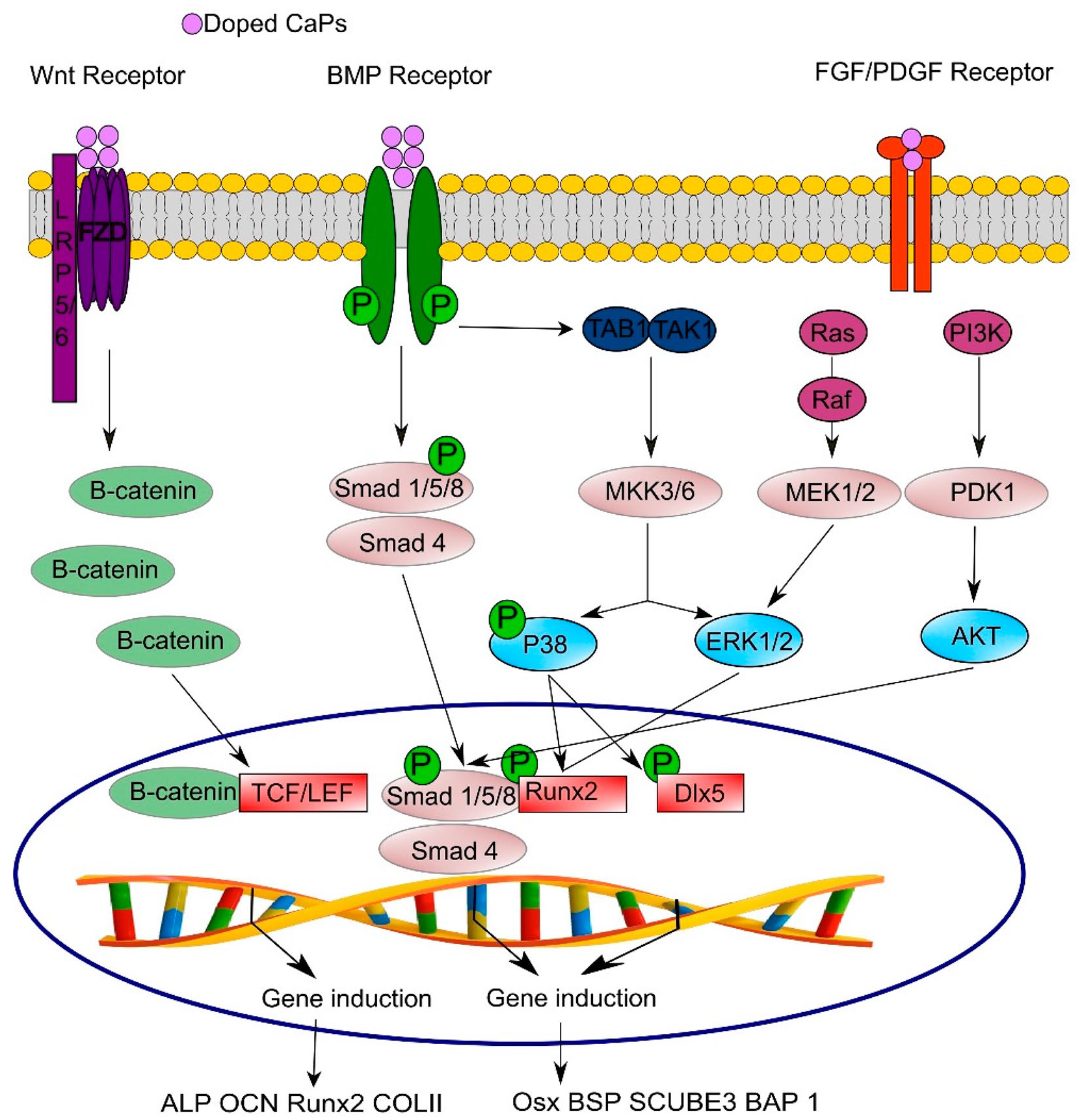
In osteogenesis and angiogenesis models, signaling pathways are activated. This pathway converges and cross talks with RUNX2 to guide the differentiation of bone and new vessels 25 .
Notch signaling
The Notch signaling pathway represents a fundamental cellular communication system with profound implications for determining cell fate, self-renewal capacity, and programmed cell death. This intricate pathway hinges on Notch receptors, which belong to a class of transmembrane proteins characterized by expansive extracellular domains. These receptors interact with specific ligands, namely Jagged and Delta, initiating their activation. Upon activation, the Notch receptor undergoes cleavage mediated by γ-secretase, liberating the notch intracellular domain (NICD). Subsequently, the NICD translocates into the cell nucleus, where it engages with transcriptional regulators from the CBF1/RBPJ-kappa in vertebrates (CSL) and the transcriptional coactivator, mastermind-like-1 (MAML) families, forming a complex that orchestrates the activation of Notch target genes, notably Hey/Hes, which function to inhibit Runx2. It is worth noting that Notch 2 plays a pivotal role in BMSCs 94 . Activation of the Notch signaling pathway in human BMSCs initially triggers early osteogenic differentiation. As a result, Runx2 activity is restricted to prevent the formation of mature osteoblasts. BMP and Wnt pathways can also be modulated by Notch2 signaling. In osteoblastic genes, Hey1 overexpression impedes Runx2 transcriptional activity induced by BMP2. It also inhibits osteogenesis triggered by Wnt. Among its impressive amino acid composition is silk fibroin, derived from silk cocoons 95 . This fibrous protein bears a striking similarity to collagen I, a prominent component of the ECM 96 . Silk proteins are also employed as biomaterials to promote osteogenesis97,98. Guo and colleagues conducted a research investigation aimed at deciphering the intricacies of ectopic bone formation, focusing primarily on the Notch signaling pathway. Their study involved the cultivation of BMSCs in the presence of biphasic calcium phosphate (BCP) ceramics, both with and without the Notch signaling inhibitor RO4929097. In the presence of BCP ceramics, they noted a significant increase in the expression of osteogenesis-related genes and genes associated with the Notch signaling pathway when compared to a control group lacking BCP ceramics. To corroborate these findings, the researchers conducted intramuscular ectopic implantation experiments in beagle dogs. The results from q-PCR experiments echoed the outcomes of the in vitro study. Histological and histomorphometric analyses indicated that RO4929097 had a retarding effect on bone formation. These results underscore the pivotal role of the Notch signaling pathway in BCP-induced bone formation, suggesting that this pathway positively influences ectopic bone formation by facilitating the differentiation of BMSCs into osteoblasts 72 . In another study by Zhang et al., they observed the upregulation of Notch pathway genes in various types of BCPs. Their investigation revealed that BCPs with different micropore diameters had varying impacts on ectopic osteogenesis, resulting in differential upregulation of Notch signaling pathway genes 73 . BMSCs cultured on CP ceramics were evaluated for their response to Wnt and Notch signal pathways by Wang and colleagues. These pathways, as well as osteogenesis-related genes, were polled in vitro using real-time reverse-transcription PCR. CP ceramics were also implanted in vivo to assess new bone formation. In vitro experiments showed that Wnt and Notch signaling pathways and osteogenesis-related genes increased initially, then declined. Reduced expression levels were observed when DKK1, a Wnt pathway inhibitor, was introduced. Ceramic CP ceramics exhibited significant bone formation in vivo. Calcium phosphate (CaP)-enhanced osteogenic differentiation involves both Wnt and Notch signaling pathways. It remains unknown, however, how they synergize 74 .
BMP/TGF-β pathway
GFs and cytokines are included in the BMP family of GFs. These receptors have two distinct types, type I receptors and type II receptors, which are similar to TβR. Mothers against decapentaplegic (SMAD) proteins are phosphorylated by type II receptors 99 . These SMADs are complex entities responsible for transmitting signals and binding to specific DNA sequences like the smad binding element (SBE) within the nucleus 100 . SMAD proteins comprise regulatory-SMADs, inhibitory-SMADs, and common-SMADs 101 . Biomaterials, particularly nano-fibrous HA/chitosan scaffolds, have been demonstrated to activate BMP signaling pathways, thereby promoting MSC proliferation and cell adhesion 102 . In vivo experiments involving rat BMSCs seeded on HA/chitosan scaffolds exhibited increased mRNA levels for Smad1, Runx2, BMP-2/4, Alkaline phosphatase (ALP), collagen I, integrin subunits, and myosins. Activation of R-SMAD/co-SMAD complexes within the BMP pathway led to Runx2 activation, elevated pSmad1/5/8 protein levels, and increased osteocalcin levels, indicating the potential of HA/chitosan composite nanofibers in bone TE 81 . A novel nanoparticulate mineralized collagen-glycosaminoglycan (MC-GAG) scaffold integrates both organic and inorganic components of the ECM and exhibits remarkable osteogenic properties for primary human MSCs and rabbit BMSCs103,104. This innovative approach holds promise for clinical applications in bone TE, mitigating the unforeseen effects associated with high-dose GF stimulation. CPs, closely resembling the mineral composition of bone, serve as constructs to induce osteogenesis at injury sites 105 . These ceramics can balance various physicochemical properties to optimize osteoconductive and osteoinductive behavior in the host tissue. In vitro studies have demonstrated the positive impact of calcium ions (Ca2+) on osteogenesis in preosteoblasts and osteoblasts106,107. BCP and nHA-coated BCP scaffolds were observed by Wang et al. Trabecular bone-like structures were observed on both scaffolds, but surface characteristics and grain sizes varied. Cell adhesion and osteogenic differentiation of BMSCs were significantly enhanced by nano-hydroxyapatite (nHA)-coated BCP scaffolds. In turn, osteogenic genes were expressed, alkaline phosphatase activity was elevated, and osteocalcin was produced. In the nHA-coated BCP group, higher Smad5, Smad4, Smad1, and BMPRI levels were evidence of activation of BMP/Smad signaling. In addition, the nHA-coated BCP scaffold stimulated ectopic bone growth in a rabbit model, unlike the BCP substrate that rapidly degraded and did not promote osteogenesis 71 . In a study conducted by Sharma et al., the focus was on gene interactions among a scaffold, BMP2, and MSCs. They cultured human bone marrow MSCs in three distinct groups: a control group with BMSCs alone, BMSCs treated with BMP2, and BMSCs subjected to both scaffold and BMP2. Gene expression analysis via microarray revealed intriguing results. Group 3 (comprising scaffold and BMP2) upregulated 3,222 genes compared to the 2,158 genes observed in group 2 (BMP2 alone). Group 3 exhibited pronounced activation of numerous osteogenic pathways, including TGF, SMAD, BMP2, Notch, and WNT signaling. These pathways were influenced by calcium and magnesium released from the scaffold, culminating in a significant activation of RUNX2, a critical regulator of osteogenesis. Network analysis further unveiled the intricate interactions among these factors, implying that the scaffold, when combined with BMP2, could enhance MSC differentiation for osteogenesis, thereby holding substantial potential for applications in bone TE 75 . Pereira et al. reported a pivotal finding that MBCP+® displayed a notably higher rate of calcium ion release in vitro than MBCP® (Keystone Dental Group, Burlington, MA, USA). In co-culture experiments, hBMSCs seeded onto MBCP+® exhibited enhanced vascular endothelial growth factor (VEGF) secretion, which, in turn, led to increased endothelial cell proliferation and the formation of capillaries compared to hBMSCs seeded onto MBCP®. In a nude mouse model with hBMSCs, MBCP+® demonstrated expedited osteogenic differentiation and the deposition of mature bone, aided by a rich host vasculature. These effects were linked to the activation of the calcium-sensing receptor protein (CaSr) on the surface of hBMSCs and elevated BMP-2 protein expression. These findings provided valuable insights into the mechanisms underlying ossification and angiogenesis triggered by calcium ions released from CP scaffolds 76 . Chen et al. delved into the impact of Zr ions on human osteoblasts in culture. Their findings revealed that Zr ions fostered the expression of genes associated with osteoblast differentiation and osteoblast proliferation. Remarkably, Zr ions, at specific concentrations, encouraged CP deposition. This phenomenon was accompanied by heightened expression levels of BMP2 and BMP receptors, along with the phosphorylation of SMAD1/5. Significantly, the inhibition of BMP signaling by Noggin counteracted the osteogenic effects of Zr ions in osteoblasts 97 .
PDGF signaling
The skeletal system depends heavily on Platelet-derived growth factor (PDGF), an extracellular factor 98 . The factors that increase the number of MSCs are their angiogenic, mitogenic, and proliferative properties. Depletion of the β-PDGFR gene in MSCs promotes osteocalcin, osteox, BMP2, and Runx2 expression at mRNA levels 108 . Inhibiting PDGFR pharmacologically reduces MSC proliferation without affecting osteoblast differentiation. In a wound, platelets last less than 5 days, and their GFs last for only a few hours 98 . The primary mechanisms by which PDGF promotes bone regeneration and healing are as follows. First, it stimulates the proliferation of BMSCs and activates their differentiation into osteoblasts, which then release TGF-β. Second, macrophage chemotaxis and activation are more prominent and crucial mechanisms 109 . Colcemid-induced metaphase arrests and [3H] proline incorporation into collagen are enhanced by PDGF’s mitogenic activity, especially in osteoblast-rich central bones. A PDGF molecule is a dimeric glycoprotein composed of two A (-AA) chains or two B chains (-BB). BMP-Smad1/5/8-Runx2/Osterix pathway is activated by PDGF-AA, which promotes MSC differentiation into osteoblasts 110 . A multicenter clinical study found that platelet-derived GF (PDGF-BB) delivered with TCP promoted ankle fusions with comparable fusion rates, less pain, and fewer side effects than autografts 111 .
IGF signaling
IGF signaling regulates osteoblast growth and specialization. The only members of the IGF family are IGF2 and IGF1, which exhibit similar biological functions in osteoblasts. Bone tissue contains high levels of IGF2. The GFs regulate collagen and non-collagenous protein expression, induction of osteoblast differentiation, and bone matrix deposition in bone biology 112 . In primary mouse osteoblasts, IGF1 induces the expression of mRNA-binding protein LARP6, which is essential for osteogenic differentiation and collagen I alpha 2 synthesis 113 . In addition, IGF-1 stimulates osteogenic differentiation of stem cells derived from adipose tissue when combined with BMP-2. A study suggests that combining BMP-6 with IGF-1 may offer a more promising alternative to BMP-2 for promoting bone formation and enhancing mineralization in orthopedic implants. A poly-lactide-co-glycolide acid (PLGA) membrane and a TCP osseous graft were used concurrently in clinical studies to achieve improved clinical outcomes using VEGF and IGF-I. Compared to IGF-I or VEGF alone, these outcomes include enhanced pocket reduction, increased clinical attachment, and improved bone fill114,115.
Ca2+ signaling
Within bone tissue, Ca2+ serves a dual purpose. They play a crucial structural role, while also acting as key players in the signaling pathways that drive osteoblast differentiation 83 . During bone remodeling, Ca2+ is continuously released into the extracellular environment, making it readily available to osteoblasts and osteoprogenitors for various biological functions. Runx2 mediates calcium channel entry into cells. Ca2+ is released from intracellular stores as a result of inositol-1,4,5-trisphosphate (IP3) and phospholipase C (PLC) signaling. The transcription factor AP-1 is upregulated, binding to the promoter regions of mechanosensitive genes and ultimately regulating GFs 116 . A comprehensive understanding of Ca2+ signaling mechanisms holds the potential to enhance the application of osteogenesis in bone regeneration. Previous research has demonstrated the positive influence of Ca2+ on the osteogenesis of various cell types, such as preosteoblasts, osteoblasts 117 , HPDCs, and macrophages 118 . Furthermore, in the context of bone TE, CP biomaterials serve as scaffolds for delivering MSCs to fracture sites, facilitating new bone formation 119 . It is believed that the release of Ca2+ from these biomaterials plays a substantial role in their bioactive properties 120 . CP crystals have been found to activate Ca2+ channels, resulting in increased expression levels of bone sialoprotein, osteopontin, and ALP. In addition, there is evidence of cross talk between Ca2+, MAPK, and Notch signaling pathways 121 .
MAPK signaling
Mitogen-activated protein kinases (MAPKs), a family of serine/threonine kinases, primarily orchestrate cell proliferation in response to external stimuli. Beyond this core function, MAPKs play diverse roles specific to different tissues and are ubiquitous across eukaryotic organisms. In the context of MAPK signaling pathways, namely p38, ERK, and JNK, they play a pivotal role in the intricate signaling cascade within osteoblasts 122 . Using ceramics, bone regeneration can be enhanced in vivo by stimulating osteogenic differentiation and angiogenesis123,124. By inducing BMP-mediated Runx2 expression, akermanite bioceramics promote osteoblast proliferation and differentiation in ovariectomized rats. Consequently, alkaline phosphatase activity and critical bone markers were elevated 125 . In addition to fostering osteoblast formation, akermanite bioceramics also facilitated angiogenesis, a fundamental step in the bone-regeneration process, by enhancing the expression of angiogenic factors like ANG-1 and VEGF. The ceramics achieved this by activating key signaling components, including ERK, AKT, and STAT3, which further induced Runx2 expression. This cross talk between osteogenesis and angiogenesis was substantiated by the sustained activation of the MAPK pathway, particularly ERK, AKT, p38, and STAT3 signaling pathways 126 . In akermanite bioceramics, magnesium and silicon ions play an important role in osteoporosis bone regeneration. Biomaterials based on calcium silicates are primarily used for BGs 127 and have garnered attention for their remarkable biocompatibility, osteogenic properties, and bioactivity. BG substrates stimulate osteoblast proliferation and differentiation by releasing Si and Ca ions 128 . Consequently, BGs emerge as a promising treatment option for bone-related disorders. In recent developments, composites combining ceramics and polymers, such as poly(glycolic acid) (PGA)/β-TCP, poly(96L/4D-lactide)/β-TCP, poly(D, L-lactide-co-glycolide) (PDLGA)/HA, and Poly(lactic acid) (PLA)/BG, have been engineered to enhance scaffold mechanical properties and bioactivity129,130.
Limitations
Bioceramics may have limited ability to promote cell adhesion and communication due to their surface properties. The surface roughness, chemistry, and charge of bioceramic materials play a crucial role in determining cell attachment and signaling. While bioceramics are generally biocompatible, some materials may still trigger an immune response or inflammation in the surrounding tissues. This immune response can affect the communication between cells and hinder the success of implant integration. The ability of cells to adhere to and proliferate on bioceramic surfaces is essential for tissue regeneration. Limitations in promoting cell adhesion and proliferation can impede the formation of functional tissues around the implant. Bioceramics are often used in orthopedic applications for bone regeneration. The limitations in their ability to induce and conduct bone formation (osteoinductivity and osteoconductivity) can impact the integration of the implant with the surrounding bone tissue. Some bioceramics are bioresorbable, meaning they can be gradually broken down and replaced by natural bone tissue. However, the rate of biodegradability may not always match the rate of tissue regeneration, leading to issues in tissue remodeling and integration. The mechanical properties of bioceramics, such as stiffness and strength, should match the mechanical requirements of the surrounding tissues. Mismatch in mechanical properties can lead to stress shielding and implant failure and hinder cell signaling. Inflammatory responses to bioceramic implants can affect the microenvironment around the implant, influencing cell signaling and communication. Chronic inflammation can lead to fibrous tissue formation instead of proper tissue regeneration. Research and development in the field of biomaterials are ongoing to address these limitations and improve the performance of bioceramics in promoting positive cell cross talk for effective tissue regeneration.
Conclusion and Future Perspectives
Cross talk between cells and bioceramics is an important aspect in the field of biomaterials and TE. Bioceramics are materials that are compatible with biological systems and often used in medical applications due to their biocompatibility and bioactivity. The interaction between cells and bioceramics plays a crucial role in the success of implant materials, tissue regeneration, and other biomedical applications. Bioceramics have achieved significant success in the fields of orthopedic and dental surgery. Their potential, however, extends beyond these applications to address various TE needs, particularly in restoring damaged Osteochondral (OC) tissue to its natural state and function. Despite their advantageous qualities such as biocompatibility, resistance to chemical reactivity in biological environments, and hardness, bioceramics have a limitation in terms of tensile strength. Current research endeavors focus on enhancing their mechanical durability upon implantation by exploring the chemistry, composition, microstructure, and nanostructure of these materials. In addition, researchers are working on achieving the right level of porosity to facilitate cellular adhesion, proliferation, and differentiation. While there have been notable strides in TE, there remains a necessity to develop materials precisely tailored for specific biomedical purposes. This involves gaining a deep understanding of how bioceramics interact with tissue, establishing a hierarchical structure for long-term functionality, and assessing mechanical strength, especially their resistance to fatigue from recurring external stresses. Biomaterials and innovative design strategies have ushered in a new era of bioengineered bone tissue, incorporating cells, drugs, and biochemical cues to encourage bone regrowth. Recent advances in 3D bio-fabrication technologies promise patient-specific surgical implants designed to ensure efficient vascularization and rapid integration with the host tissue. In addition, manipulating the inherent properties of biomaterials to influence stem cell fate decisions is a pioneering approach. Using soluble factors or biochemical inducers not only advances bone regeneration but also mitigates side effects. Furthermore, biomaterials can accelerate bone regeneration by harnessing physical, chemical, biological, and engineering processes to replace complex craniofacial deformities. Bioceramics should support cell adhesion, where cells attach themselves to the surface of the material. This initial interaction is vital for subsequent cellular responses. In the context of bone regeneration, bioceramics are often designed to be osteoconductive, meaning they facilitate the ingrowth of bone tissue. This involves interactions with osteoblasts and other bone-related cells. Bioceramics can influence cell behavior by releasing ions or other bioactive substances. This can affect cell proliferation, differentiation, and other cellular activities. The interaction between cells and bioceramics can also influence the inflammatory response. Ideally, bioceramics should promote a favorable immune response to prevent rejection or adverse reactions. Bioceramics are used in TE to create scaffolds that support tissue regeneration. The cross talk between cells and these scaffolds is crucial for the successful formation of functional tissues. As for prospects, ongoing research is focused on enhancing the understanding of cell-bioceramic interactions and developing advanced biomaterials with improved properties.
Moving forward, the goal should be creating materials that can respond to the cellular environment and adjust their properties accordingly for better integration with surrounding tissues; expanding the use of bioceramics beyond bone regeneration to include applications in soft tissues and organs; developing innovative surface modifications to further improve cell adhesion, proliferation, and differentiation; tailoring bioceramics to individual patient needs for better outcomes in terms of biocompatibility and tissue integration; and incorporating sensors into bioceramics for real-time monitoring of cellular responses and overall performance in vivo. Research in this field is dynamic, and ongoing advancements in materials science, cell biology, and TE are likely to shape the future of cross talk between cells and bioceramics, leading to improved medical implants and therapeutic strategies.
Footnotes
Acknowledgements
The authors would like to acknowledge colleagues who gave valuable comments.
Author Contributions
Conceptualization: M.A., S.K., M.M., A.B., and D.K.; Methodology: M.A., S.K., M.M., D.K., A.B.O., A.B., M.G., K.A., R.S.S., L.K.H, Ar.H., Ah.H., and H.T.; Investigation: M.A., S.K., M.M., D.K., A.B.O., A.B., M.G., K.A., R.S.S., Ar.H., Ah.H., and H.T.; Data curation: D.K., M.A., S.K., M.M., A.B.O., and A.B.; Writing—original draft preparation: M.A., S.K., M.M., D.K., A.B.O., A.B., M.G., K.A., R.S.S., Ar.H., Ah.H., and H.T.; Writing—review and editing: M.A., S.K., M.M., D.K., A.B.O., A.B., M.G., K.A., R.S.S., L.K.H., Ar.H., Ah.H., and H.T.; Visualization: M.A., S.K., M.M., D.K., A.B.O., A.B., M.G., K.A., R.S.S., L.K.H., Ar.H., Ah.H., and H.T.; Supervision: Ar.H., Ah.H., and H.T.; Project administration: Ar.H., Ah.H., and H.T. All authors have reviewed and accepted the final version of the text.
Data Availability Statement
Not applicable.
Ethical Approval
Not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

