Abstract
Multipotent mesenchymal stem cells (MSCs) have high self-renewal and multi-lineage differentiation potentials and low immunogenicity, so they have attracted much attention in the field of regenerative medicine and have a promising clinical application. MSCs originate from the mesoderm and can differentiate not only into osteoblasts, cartilage, adipocytes, and muscle cells but also into ectodermal and endodermal cell lineages across embryonic layers. To design cell therapy for replacement of damaged tissues, it is essential to understand the signaling pathways, which have a major impact on MSC differentiation, as this will help to integrate the signaling inputs to initiate a specific lineage. Hedgehog (Hh) signaling plays a vital role in the development of various tissues and organs in the embryo. As a morphogen, Hh not only regulates the survival and proliferation of tissue progenitor and stem populations but also is a critical moderator of MSC differentiation, involving tri-lineage and across embryonic layer differentiation of MSCs. This review summarizes the role of Hh signaling pathway in the differentiation of MSCs to mesodermal, endodermal, and ectodermal cells.
Introduction
Multipotent mesenchymal stem cells (MSCs) are a type of adult stem cell that can be isolated from various tissues in the body, such as bone marrow 1 , adipose tissue1–3, peripheral blood, umbilical cord tissue1,2, Wharton’s jelly 1 , amniotic fluid 4 , gingiva, dental pulp 2 , and placental tissue 1 . The International Society for Cellular Therapy (ISCT) has established the minimum criteria to identify MSCs: they must be capable of adhering to plastic surfaces during culture, differentiating into three mesodermal cell lineages (such as osteoblasts, chondrocytes, and adipocytes), and expressing certain cell surface antigens 5 . Human mesenchymal stem cells (hMSCs) express positive markers, such as CD29, CD44, CD73, CD90, and CD105, while negative markers, such as CD11b, CD14, CD19, CD34, CD45, CD79, and human leukocyte antigen DR (HLA-DR) are absent 5 . Due to their self-renewal, multi-lineage differentiation, and immunomodulatory capabilities, MSCs have great potential for applications in regenerative medicine and inflammatory disorders 6 . Depending on the induction signals provided and microenvironment in which MSCs reside, they can differentiate not only into mesodermal lineages (such as adipocyte, chondrocyte, osteoblast, and myocytes) but also into ectodermal lineages (such as epithelial and neural cells) and endodermal lineages (such as hepatocytes, pancreatic islet cells, and lung epithelial cells)7–9 although MSCs originate from the mesoderm (Fig. 1).
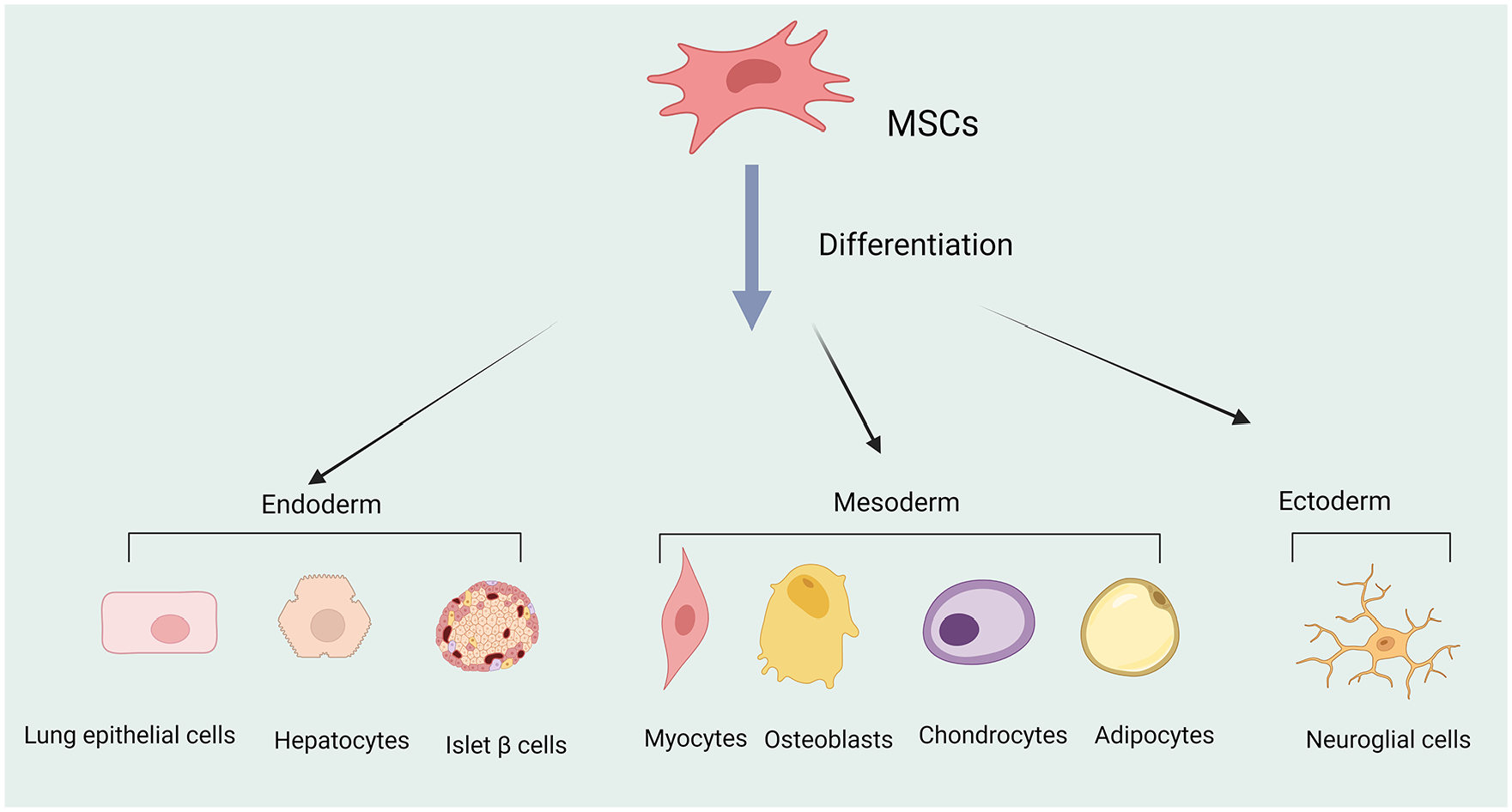
Multipotent mesenchymal stem cells (MSCs) can differentiate into different cell lineages, such as mesodermal cells, cross-embryonic differentiation into endodermal and ectodermal cells (figure was created with BioRender.com).
Compared with MSCs, pluripotent stem cells (PSCs), including human embryonic stem cells (hESCs) 10 and induced pluripotent stem cells (iPSCs), 11 have broad differentiation potential and theoretically can generate any cell type in the body 12 . Although several phase I clinical trials based on PSCs for diseases, such as corneal disease, amyotrophic lateral sclerosis, and Parkinson’s disease have been launched in Japan, China, and Israel12–15. The use of PSCs still faces a series of challenges, including carcinogenicity, safety, and ethical issues. From a clinical translation perspective, MSCs have many advantages compared with embryonic stem cells (ESCs), including ease isolation, low immunogenicity (due to the low expression of major histocompatibility complex [MHC] class I molecules in MSCs 10 ) and almost no ethical restrictions. Therefore, MSCs are a promising source for stem cells that can be used for various regenerative medicine purposes and have become the most widely used stem cell type in clinical practice6,16–18.
In addition to their regenerative properties, MSCs have also shown potential for immune regulation. They participate in innate and adaptive immune regulation19,20, and have been shown to inhibit the activation and proliferation of T cells19,21, B cells19,22, and natural killer (NK) cells19,23, as well as the migration and maturation of antigen-presenting cells (APCs)19,24,25. Therefore, MSCs have also been used for the treatment of acute, chronic, and severe immune disorders, such as autoimmune diseases 26 , acute lung injury (ALI)7,27,28, chronic graft-versus-host disease (GVHD)29,30, and rejection reactions in tissue and organ transplantation31,32. These studies have demonstrated that MSCs can regulate the immune function of the body and reduce inflammation through the secretion of cytokines and interaction with the microenvironment of injured tissue. However, recent research suggests that apoptotic and metabolically inactivated MSCs also have immunomodulatory potential, with regulatory T cells and monocytes playing a critical role in this process 20 .
Regenerative medicine is a rapidly developing field of medical science aimed at repairing, regenerating, or replacing damaged, diseased, or missing cells, tissues, and organs to restore their normal function. With the multipotential differentiation ability, MSCs can be used to replace or repair damaged or diseased cells and tissues, making them an ideal material for regenerative medicine. In fact, the application of MSCs through a variety of therapeutic mechanisms (Fig. 2) has shown great potential in treating a variety of diseases, including immune and autoimmune diseases (multiple sclerosis), heart disease, diabetes, neurological disorders (Alzheimer’s disease, Parkinson’s disease, spinal cord injury, stroke, and traumatic brain injury), musculoskeletal injuries, various skin conditions (burns, wounds, and ulcers), as well as bone, cartilage, and joint-related diseases. Numerous studies have shown that MSCs can improve tissue and organ function and reduce the risk of death6,17,18,28,33. So far, MSCs from various sources have been used in registered clinical trials. Most preclinical studies have been conducted using bone marrow-derived mesenchymal stem cells (BM-MSCs). However, in recent years, MSCs derived from adipose tissue (AD-MSCs) and umbilical cord blood (UC-MSCs) have also received widespread attention 4 . Overall, MSCs have been demonstrated with good tolerability, safety, and significant therapeutic effects in many clinical diseases, leading some countries to approve their use. Currently, there are over 900 studies in clinical trials involving various sources of MSCs 34 , and this number is constantly increasing. Therefore, MSCs have emerged as one of the most promising sources for regenerating injured tissue cells, and their potential for future clinical applications is enormous.
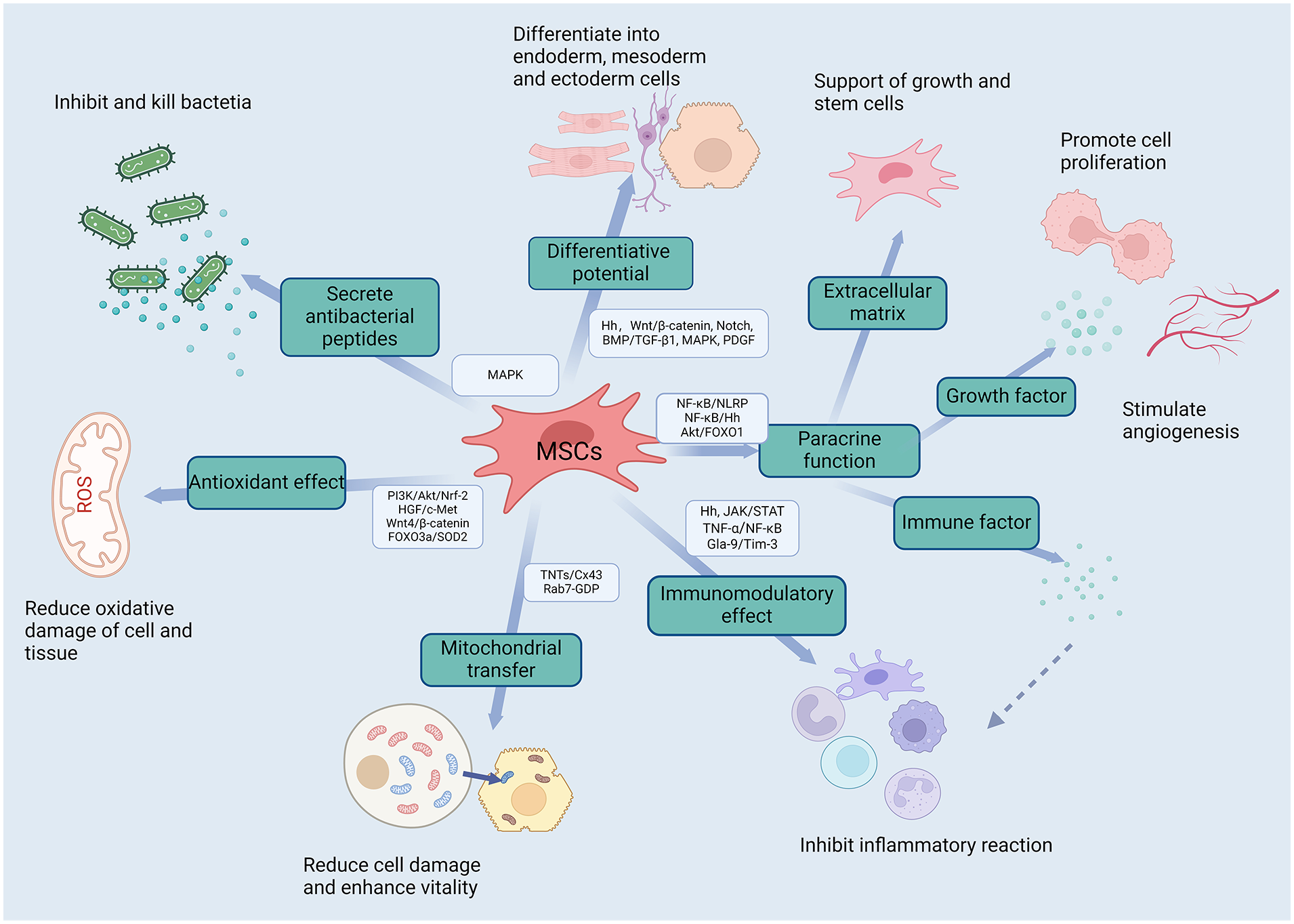
Multipotent mesenchymal stem cells play therapeutic roles in diseases through various therapeutic mechanisms, which involve multiple signaling pathways (figure was created with BioRender.com).
The therapeutic effects of many MSCs can be attributed to intercellular signal transduction mechanisms, where extracellular vesicles derived from MSCs (MSC-EVs) act as key effectors
35
. In regenerative medicine methods for repairing tissue damage, the application of MSC-EVs has shown great feasibility, and data from animal models and the first human trials indicate that MSC-EVs can exert therapeutic potential like their source cells
36
. MSC-EVs are membrane structures that load proteins, lipids, nucleic acids and signaling molecules, maintain pluripotency, induce regenerative phenotypes, inhibit apoptotic reactions, regulate immune responses, and promote regeneration and repair of damaged or diseased cells
35
. The therapeutic potency of MSC-EVs appears to be equivalent to that of MSCs, and the mechanism of action of MSCs can also be reproduced in MSC-EVs therapy, indicating that MSC-EVs can effectively simulate the therapeutic effects of MSCs in preclinical animal models of diseases35–37. However, some studies have shown that MSCs are more effective than MSC-EVs37,38. Direct cell-to-cell contact of MSCs plays a central role in mediating their beneficial effects
39
, such as inhibition of T cell proliferation, reprogramming macrophages into an anti-inflammatory phenotype, and sustained releasing of therapeutic mediators at the site of injury, mediating immune suppression, nutrient and growth factors, thereby alleviating inflammatory damage, promoting repair, and regeneration of injured cells7,36,37,39. Importantly, the differentiation of MSCs into other tissue types is highly dependent on the microenvironment in which they reside and the specific
The differentiation of MSCs into different lineages is highly dependent on the microenvironment in which they are located7,40. Cell-to-cell communication and signals from the microenvironment, including paracrine/autocrine soluble factors, gap junctions, extracellular vesicles (EVs), all influence the differentiation of MSCs in the damaged tissue microenvironment
40
. This microenvironment is known as the stem cell niche, which is crucial for controlling the differentiation of stem cells into specific lineages
Hh Is an Important Signaling Pathway That Mediates MSCs Differentiation
The differentiation of MSCs involves multiple signaling pathways. During embryonic development, Hh, transforming growth factor-β (TGF-β)/bone morphogenetic protein (BMP), Wnt, Notch, mitogen-activated protein kinase (MAPK), and other pathways are important signaling pathways that maintain the growth rate of all differentiation lineages of stem cells and are responsible for cell fate determination, organogenesis, and other processes
50
. Among them, Hh is an intercellular signaling molecule that participates in early embryonic development and other important cellular processes, including proliferation, apoptosis, migration, and differentiation
53
. It plays a central role in the regulation of the proliferation and differentiation of adult stem cells. The general signaling mechanism for the Hh pathway is conserved from fly to mammal, although more and distinct components have been found in mammalian cells
54
. In the early 1990s, three Hh homologs were discovered in vertebrates: Sonic hedgehog (SHh), Indian hedgehog (IHh), and Desert hedgehog (DHh). Hh signaling plays a critical role in the regulation of pattern formation, growth, stem cell maintenance, and self-renewal in several organs during development55–58. Among them, DHh expression is typically limited to male reproductive tract and will not be further discussed
58
. SHh and IHh are critical during embryological development and postembryonic tissue repair and stem cell regulation59,60. Injury can cause high expression of Hh proteins in damaged tissues, making them important signaling molecules in the damaged microenvironment 43,48,49 . These molecules may be involved in regulating MSCs differentiation when MSCs are in a specific
The primary cilium is a specialized structure located on the cell surface 66 . Primary cilia play a key role in Hh signaling pathway. In mammals, three protein members of Hh (SHh, IHh, and DHh) share the same downstream signaling components 58 . The negative and positive cellular regulation of Hh is controlled by two transmembrane proteins, Patched (Ptch), a negative regulator of Hh signaling, and Smoothened (Smo), a G protein-coupled receptor (GPCR)-like 7-transmembrane receptor that stimulates downstream signaling in response to Hh signal. In the absence of Hh ligand, Ptch induces a closed conformation of Smo, leading to inhibition of its activity and halting all downstream signaling events 67 . When extracellular Hh is present, it binds to and inhibits Ptch, and abrogates its repressive effect on the Smo receptor, a key effector essential for Hh signal transduction 68 . Smo initiates a signaling cascade that results in the translocation of the glioma-associated oncogene (GLI) family of transcription factors into the nucleus.
The key role of Smo in Hh signaling is to control the activation of the GLI transcription factors69,70. GLI1 is exclusively a transcriptional activator, whereas GLI2 and GLI3 have dual activity 71 . GLI2 acts primarily as a transcriptional activator of the Hh pathway, GLI3 primarily acts as a suppressor of Hh signaling72–75. Sufu, a negative regulator of GLI 1–3, was shown to abduct GLI 1–3 in the cytoplasm to prevent it from undergoing nuclear translocation. In cells, Sufu is phosphorylated by protein kinase A (PKA) to form a stable phosphorylated form of Sufu, which affects its interaction with GLI. Sufu and GLI form a complex in the cytoplasm after entering the primary cilia resulting in different phosphorylation states of the two proteins and determining transcriptional activator form (GLIA) or repressor form (GLIR) production, depending on the presence or absence of Hh ligand stimulation 71 . In the absence of Hh ligand, Ptch induces a closed conformation of Smo (its activity is inhibited). The inactive Smo is in the cytoplasm. At the same time, cyclic adenosine monophosphate (cAMP) increases and activates PKA, which phosphorylates both proteins of the Sufu/GIL complex. GLI is further phosphorylated by CK1 and Glycogen Synthase Kinase-3β (GSK3β) and forms GLIR by proteolytic cleavage. In the presence of Hh ligand, Ptch derepresses Smo and Smo activates to initiate Hh downstream signaling pathways in receptor cells. Ptch is transported to lysosomes in the cytoplasm for degradation. Activation of Smo translocates onto the ciliary membrane and carboxy-terminal phosphorylation. Meanwhile, activated Smo inhibits PKA activity and is not sufficient to phosphorylate the Sufu/GLI complex. The unphosphorylated complex is transported to the cilia tip. The unphosphorylated Sufu is degraded. The GLI is released and is phosphorylated by CK1 to form GLIA. GLIA translocates to the nucleus, where it binds to the promoter of the Hh-related target genes to initiate transcription. These target genes participate in important biological processes, including cell growth and survival (Bcl2, FGF, IGF2), proliferation (Cyclin D1, Wnt, Myc, Ptch), differentiation (BMP, Wnt), migration, and GLI1 expression (Fig. 3). Since GLI1 is both a key transcriptional factor and a downstream target protein of Hh signaling pathway, thus upregulation of GLI1 expression is a reliable marker for Hh signaling activity76–79. Some studies suggest that the Hh signaling pathway directs MSCs toward specific differentiation pathways43,46,49,62,80,81.
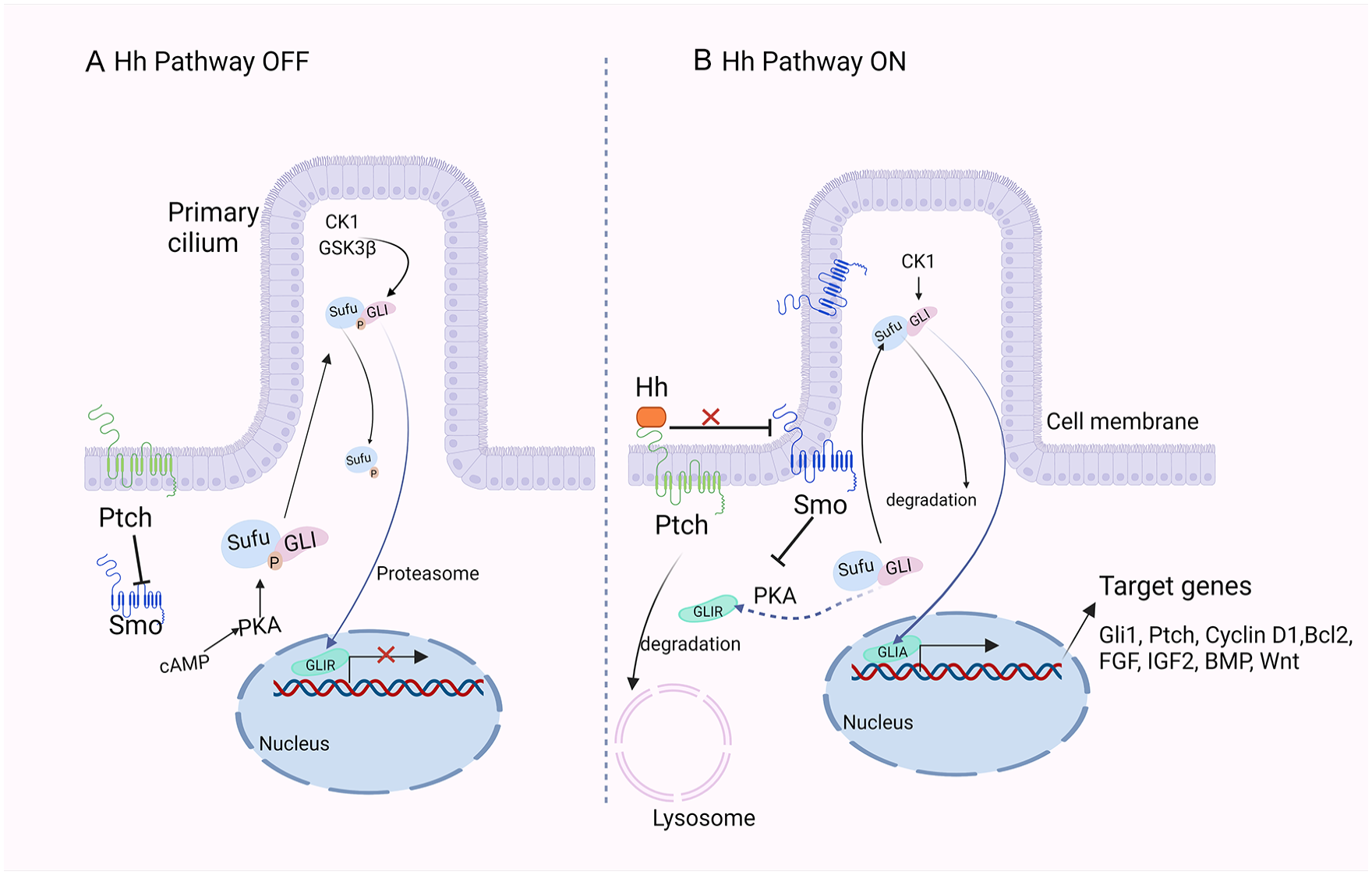
Simplified diagram of the hedgehog (Hh) signaling pathway in mammals. (A) In the absence of Hh ligand, Ptch inhibits the activity of Smo causing Hh pathway inhibition. In this pathway-inactive state, PKA, CK1, and GSK3β phosphorylate the Sufu/GLI complex, which dissociates, and the transcription factor GLI triggers a partial proteasomal degradation by phosphorylation to produce the corresponding repressor form (GLIR). Subsequently, GLIR enters the nucleus and inhibits the transcription of target genes. (B) The Hh ligand binds to Ptch and is translocated to the lysosome for degradation via endocytosis. At this point, the restrictive effect of Ptch on Smo is lifted, allowing Smo to enter the cilium where it is activated. Inhibition of PKA activity by activated Smo is not sufficient to phosphorylate the Sufu/GLI complex, blocking the phosphorylation and proteolytic processing of the GLI proteins and enabling them to become transcription activators (GLIA). GLIA then enters the nucleus to initiate transcription of downstream target genes (figure was created with BioRender.com).
The Role of Hh Pathway in Multi-Lineage Differentiation
Studies have confirmed that the Hh signaling plays a critical role in controlling the proliferation and differentiation of stem cells or progenitor cells61,62. Therefore, the Hh signaling pathway is crucial for the proper differentiation of MSCs and the maintenance of stem cell homeostasis. It has been shown that this pathway plays a key role in the differentiation of MSCs into various cell types, regulating the expression of various genes involved in MSCs differentiation, and determining cell differentiation into different cell lineages, such as mesodermal cells (osteoblasts, chondrocytes, adipocytes, and myocytes)43,44, endodermal cells (hepatocytes64,82–85, pancreatic beta-cells [β-cells]45,86–88, and lung epithelial cells7,89–91), and ectodermal cells (neuroglial cells46,92–99, endothelial cells100–102).
The Role of Hh in Mesodermal Differentiation of MSCs
The Hh Signaling Pathway Regulates Osteogenic Differentiation of MSCs
Osteoblasts develop from the MSCs through a process called osteogenesis. Osteogenesis is an important developmental event that results in bone formation. Bone-forming cells or osteoblasts develop from MSCs through a highly controlled process regulated by several signaling pathways, such as Hh, BMPs, Notch, neural epidermal growth factor-like 1 protein (NELL-1), and Wnt/β-catenin signaling 103 . These signaling pathways individually as well as in coordination with other signaling molecules, regulate the osteogenic lineage commitment of MSCs by activating several osteo-lineage specific transcription factors 103 . Among these factors, Hhs play an important role in bone formation and homeostasis, as their vital roles in modulating the osteogenesis of MSCs, and key functions both individually as well as in coordination with other signaling cascades in terms of Wnt, BMP, and parathyroid hormone-related protein (PTHrP) during skeletal development, bone repair, and bone regeneration103–107. During embryonic development, SHh plays a critical role in skeletal development, participating in the shaping of the trunk, limbs, and facial bones 108,109, while IHh has an important role in skeletal development and regulates endochondral bone formation, and intramembranous ossification 110 . In fact, disruption of the Hh signals can lead to bone-related diseases in terms of osteoarthritis, osteoporosis, bone defects, and severe skeletal deformities 111 . Inhibition of Hh signaling using the Hh inhibitor cyclopamine blocks Hh signal and leads to a lack of peripheral cartilage ossification in the head 112 .
Studies have shown that undifferentiated human MSCs have a basal level of Hh signaling, which may be maintained by autocrine secretion of SHh and IHh expressed in these cells
49
. An early study has shown that recombinant N-terminal Sonic Hedgehog (rSHh-N) promotes proliferation and osteogenic differentiation of rat BM-MSCs, as evidenced by increased alkaline phosphatase (ALP) activity, upregulation of the osteogenesis-related genes collagen type I, core-binding factor alpha1 (Cbfa1), and osteocalcin (OCN), and increased matrix mineralization in differentiating BM-MSCs113,114. Implantation of rSHh-N overexpressing MSCs significantly accelerates bone formation
MSCs, give rise to osteo-chondro progenitors, which develop into osteo-precursors upon Runx2 (Runt-related transcription factor 2) activation or chondrocytes if Sox9 (SRY box transcription factor 9) is activated 120 . SHh enhances osteogenic differentiation of MSCs in Runx2-dependent and manner and is accompanied by expression of bone progenitor cell markers and matrix mineralization of differentiated MSCs114,121–124. Runx2, the master regulator of osteogenesis, has an important role in early osteogenic lineage commitment125,126. And, it is a specific phenotype and key transcription factor that directs MSCs toward osteoblastic differentiation and inhibits them from differentiating into adipocytes and chondrocytes 127 . The secreted molecule NELL-1 is another critical factor that binds with apoptosis-related protein 3 (APR3) 128 and colocalizes with Cntnap4 on the cell surface to promote osteogenic differentiation of human AD-MSCs62,129. NELL-1 is a transcriptional target of the transcription factor Runx2, where Runx2 increases NELL-1 transcription in a dose-dependent manner103,118. Mechanistically, NELL-1 is preferentially expressed in osteoblasts, and its expression level is like that of Runx2, and is highest during skeletogenesis130,131. Combined activation of Hh, NELL-1 signaling by SHh-N (SHh, N-terminus protein) and NELL-1 addition had an additive pro-osteogenic effect on human AD-MSCs 62 . NELL-1 promotes osteogenesis accompanied by activation of Hh signaling 62 . Therefore, Runx2 and NELL-1 are key players for Hh signaling to promote MSCs differentiation into osteoblasts.
In addition to SHh protein, some small molecule compounds (purmorphamine [PMA] and oxysterols) and drugs (simvastatin) can also promote the osteogenic differentiation of MSCs by enhancing Hh signaling. PMA and oxysterols are originally identified as effective osteogenic factors in MSCs and are identified as activators of the Hh pathway132–136,94. They upregulated gene expression of the mediators of Hh pathway, SMO, PTCH1, GLI1, and GLI2, and increased the expression of several genes (eg, RUNX2 and BMPs) related to osteogenesis through the activation of Hh pathway 89,92,94. Nevertheless, they activate the Hh signal through different mechanisms134,136,137. PMA activates the Hh pathway by targeting the seven transmembrane pathway component Smoothened (SMO), a critical component of the Hh signaling pathway89,92,94. Oxysterols stimulate Hh pathway activity by indirectly activating SMO90,91. Simvastatin administration resulted in an enhanced osteogenic differentiation capacity, indicated by upregulated expression of COL1, ALP, and OCN, and increased ALP activity. More importantly, bone marrow mesenchymal stem/stromal cells (BMSCs) treated with simvastatin expressed higher levels of IHh and GLI1, and more nuclear translocation of GLI1 was observed 138 .
Although several pathways individually influence the bone formation, osteogenesis is achieved by the coordinated action of multiple signaling pathways 103 . For instance, Hh can coordinate other signaling pathways to promote osteogenic differentiation of MSCs. Bone morphogenetic protein 9 (BMP9) promotes osteogenic differentiation of mouse MSCs, and the Hh signaling pathway plays an important regulatory role in BMP9-induced MSCs osteogenic differentiation 139 . Specifically, BMP9 induces the expression of early osteogenic markers (such as ALP) and late osteogenic markers (such as matrix mineralization) as well as osteopontin (OPN) and OCN in MSCs, but this effect is significantly inhibited by the Hh signaling inhibitor cyclopamine. Hh-induced osteoblastogenesis requires BMP signaling, and together they elicit a synergistic expression of alkaline phosphatase activity 140 .
Micro-RNAs (miRNAs) are important regulators of gene expression involved in osteogenesis, and regulate the Hh signaling pathway in MSCs. The miR-324-5p regulates the Hh signaling pathway through different mechanisms in human BM-MSCs and mouse C3H10T1/2 cells (MSCs), thereby inhibiting osteogenic differentiation, miR-324-5p targets SMO and GLI1 in the human Hh signaling pathway, while targeting Glypican 1 (GPC1) in mice. MiR-196a can activate the Hh signaling pathway by targeting GNAS to reverse osteogenic differentiation in osteoporotic mice MSCs 141 , whereas miR-467g has been found to be an inhibitor of osteoblast differentiation, negatively regulating osteogenesis through the IHh/Runx2 signaling pathway 142 .
Additionally, in high glucose and oxidative stress conditions, partially inhibiting MSCs osteogenic differentiation through regulating the Hh signaling pathway is possible. High glucose inhibits SHh protein expression in rat BM-MSCs143,144, while the activation of the SHh signaling pathway can upregulate the expression of bone morphogenetic protein 4 (BMP4), bone sialoprotein (BSP) and OPN in MSCs through transfection with lentivirus SHh vectors
145
. Thus, SHh overexpression significantly increases the osteogenic ability in rat BMSCs
Oxidative stress inhibits the Hh signaling pathway and Hh-mediated osteogenic differentiation of MSC
144
. The addition of exogenous H2O2 (125–500 mM) can decrease ALP activity in human BM-MSCs
147
. Therefore, H2O2 can prevent SHh-mediated osteogenic differentiation of mice and other MSCs lines
The Hh Signaling Pathway Regulates Chondrogenic Differentiation of MSCs
The Hh signaling pathway plays an important role in the chondrogenic differentiation of MSCs149–154 and in cartilage development
A study demonstrates that Hh, TGF-β1, and BMP-2 proteins are equivalent inducers of MSCs chondrogenesis 154 . Nevertheless, compared with BMP, TGF, and other growth factors, SHh plays a stronger role in promoting human MSCs proliferation and chondrocyte differentiation. It enhances the expression of chondrocyte-specific markers collagen type II (Col II), chondroitin sulfate, Sox9, and CEP-68 149 . Reprogramming human MSCs to increase the activity of the SHh signaling pathway can promote MSCs chondrogenesis 159 . Atractylenolide is enriched in atractylodis macrocephalae, which is commonly used to treat arthritis. One study has shown that adding atractylenolide induces GLI promoter and promotes chondrogenic differentiation of rat BM-MSCs. Compared with untreated BM-MSCs, the expression of chondrogenic markers Sox9, Col II, and sulfated proteoglycans is increased in the atractylenolide-treated group, suggesting that atractylenolide enhances MSCs chondrogenic differentiation by activating the SHh signaling pathway 150 . Furthermore, when the Hh signaling pathway inhibitor cyclopamine was used to inhibit SHh signaling 151 , the effect of atractylenolides on promoting chondrogenic differentiation of MSCs was weakened, confirming that the promotion of chondrogenic differentiation by atractylenolide depends on the activation of the SHh signaling pathway. In addition, the SHh signaling pathway is an important regulatory factor in maintaining the chondrocyte phenotype. Under hypoxic conditions, SHh signal can stimulate the production of the transcription factor Nkx3.2, induce chondrogenesis, and inhibit the expression of another transcription factor Runx2 that induces osteogenesis, thereby maintaining the chondrocyte phenotype and promoting the secretion of cartilage matrix 160 .
In addition to SHh, IHh is also considered to be involved in chondrocyte differentiation. IHh is mainly expressed in prehypertrophic chondrocytes and osteoblasts at puberty stages 161 . Genetic studies have shown that IHh signaling regulates the proliferation and differentiation of osteoblasts and chondrocytes during bone development and repair162–166. IHh mainly regulates chondrocyte proliferation and differentiation through PTHrP163,167. One study has found that SHh and IHh have the same effect on BMSCs in promoting chondrogenic differentiation. There is no significant difference in the expression levels of Sox9, ACAN, and Col II proteins in the IHh and SHh transfection groups 152 . These results may be because both IHh and SHh promote chondrogenesis by stimulating the Hh pathway168,169. Moreover, several other signaling systems have been found to interact with IHh or SHh during chondrogenic differentiation169–171. Previous studies have suggested that IHh is also important for maintaining endogenous TGF-β and BMP signaling, and these three signaling pathways collectively drive the optimal chondrogenesis in human BM-MSCs 153 .
The Hh Signaling Pathway Regulates Adipogentic Differentiation of MSCs
Adipogenesis is a complex process that involves the coordinated regulation of multiple signaling pathways. The Hh signaling pathway has been widely reported as a negatively regulator of adipose tissue development113,172. With most of its downstream targets having been shown to play important roles in adipose tissue development. Using genome-wide RNAi screening, 500 candidate obesity genes are identified, with many of them enriched in the Hh signaling pathway and having anti-adipogenic effects
173
. Studies have shown that SHh is a key regulator of adipogenesis
173
. Specifically, activation of the Hh signaling pathway
Many studies have shown that there is a potential relationship between osteogenic and adipogenic differentiation of MSCs in rodents122,176–178. Osteogenic and adipogenic differentiation are opposing biological processes178–182. There is a balance between osteogenesis and adipogenesis, and promoting osteogenic differentiation while inhibiting adipogenic differentiation of MSCs is a key step for bone regeneration in tissue engineering
183
. To support this relationship, numerous
The Role of Hh in Ectodermal Differentiation of MSCs
Neuronal Differentiation of MSCs
In addition to possessing mesodermal tri-lineage differentiation potential43,44, MSCs also have the ability to differentiate across embryonic layers into ectodermal cell lineages, such as nerve cells
46
and lung epithelial cells7,89–91. The differentiation of MSCs into neural cells is a complex process regulated by multiple factors, including transcription factors, growth factors, and signaling molecules. One of the most important signaling molecules in the process is SHh. Loss of neurons is a common feature of many neurodegenerative diseases (such as Alzheimer’s disease [AD], Parkinson’s disease, spinal cord injury, stroke, and Huntington’s disease [HD]), which are associated with function loss and disability188,189. Because drugs cannot effectively improve damaged neurons in the brain190,191, stem cell therapy is considered to be an effective therapeutic strategy for restoring lost cells in human nervous system diseases92,192. Many sources of MSCs for neural differentiation can serve as alternative cell sources for therapy193–195. MSCs from different sources, such as adipose, dental, and bone marrow can differentiate into various types of neural cells93–98,196–199, including oligodendrocytes
99
, motor neurons195,200–204, dopaminergic-like cells, and cholinergic-like cells94,205. These cells can express neural and cholinergic-specific markers at the protein level, including Nestin, SMI-32, Islet-1, and AChE, in the presence of Retinoic Acid (RA) and SHh94–98,205. Several studies have also shown that MSCs isolated from adult teeth and shed deciduous teeth can differentiated into neurons and express neuronal markers in specific inducing culture media206,207. MSCs from human periapical cysts (hPCy-MSCs) have higher neural differentiation potential and exhibit neural progenitor-like characteristics, spontaneously expressing neuron-specific proteins and genes following neural induction. Under appropriate neural stimulation conditions, hPCy-MSCs acquire neural morphology and significantly overexpress several neural markers
208
.
SHh is a secreted protein involved in the development and maintenance of the nervous system. It is known to play a critical role in MSCs differentiation into neurons 210 . The therapeutic effects of MSCs can be mediated by the SHh pathway, which increases synaptic plasticity and synaptogenesis after MSCs treatment. SHh has been shown to promote the biological process of MSCs differentiation into neurons by upregulating the expression of transcription factors, such as Neurogenin-2 (Ngn2), NeuroD1, and Olig2203,211, and by downregulating the expression of pluripotency- maintaining transcription factors Sox2 and Oct4212,213. MSCs genetically modified with the SHh gene also readily differentiation into neurons and astrocytes 212 . It has been demonstrated that MSCs mediate an increase in tissue plasminogen activator through the SHh pathway, thereby promoting brain plasticity and neurological function 210 . However, the SHh inhibitor cyclopamine can block the therapeutic effects of MSCs by inhibiting the SHh pathway 210 .
Activation of the SHh pathway not only upregulates the expression and secretion of basic fibroblast growth factor and vascular endothelial growth factor in MSCs but also mediates their differentiation into neurons by upregulating downstream gene products of the SHh pathway, such as BDNF, NT-3, and GDNF48,68,214. Chemical factors that control signaling pathways involved in neuronal development can enhance the neuronal differentiation of MSCs 215 . For example, the SHh signaling pathway agonist PMA can improve neuronal differentiation by directly enhancing SMO receptor activation136,216,217. Another study has shown that PMA promotes the differentiation of MSCs into motor neuron-like cells when cultured on polycaprolactone (PCL) scaffolds 46 . Therefore, activation of the SHh signaling pathway is a necessary condition for MSCs to differentiation into neural cells.
The Role of Hh in Endodermal Differentiation of MSCs
In addition to their potential to differentiation into mesodermal lineages, MSCs also have the potential to differentiate into endodermal cell lineages (such as lung epithelial cells, pancreatic cells, and hepatocytes). The Hh pathway plays important roles in the endodermal differentiation of MSCs.
Lung Epithelial Differentiation of MSCs
Lung disease is a growing global health concern and a major cause of death and disability worldwide. In 2017, 3.2 million people died from chronic obstructive pulmonary disease (COPD) 218 (accounting for 5.7% of global deaths), and it is projected to become the third leading cause of death globally by 2030. ALI and its severe form, acute respiratory distress syndrome (ARDS), are also a concern for the global medical community 219 , with mortality rates as high as 30%–40%. The pathogenesis of ALI/ARDS is mainly due to an imbalance of the body’s immune response, leading to severe acute hypoxic respiratory failure caused by diffuse alveolar epithelial and endothelial cell damage from excessive inflammation, which is characterized by decreased lung compliance and increased permeability leading to pulmonary edema, frequently progressing to multiple organ failure and pulmonary fibrosis220,221. In addition, the critical condition caused by COVID in 2019 222 highlights the urgent need for innovative treatment strategies for ALI/ARDS.
More data support the potential and application prospects of MSCs in the treatment of pulmonary diseases 223 . Currently, phase I and II clinical trials have accumulated evidence confirming the safety and tolerability of these cells. Based on the anti-microbial, anti-inflammatory, regenerative, angiogenic, anti-fibrotic, anti-oxidative stress, and anti-apoptotic effects demonstrated by MSC therapy, these effects can block the physiopathological mechanisms of ALI/ARDS222–224. In recent years, although more and more studies have shown that therapeutic potential of MSC-derived EVs appears to be comparable with that of MSC therapy, and can effectively simulate the therapeutic effects of MSCs in animal models of lung injury 36 . The fact remains that using EVs alone cannot fully realize the potential of MSCs to regenerate and repair injured alveolar epithelial cells by cell differentiation potential and direct mitochondrial transfer, to save dying cells39,89,90. Importantly, some studies have shown that MSC therapy is more effective than its EVs in alleviating lung injury, regardless of the etiology, because promoting MSCs differentiation into Alveolar Epithelial Type II (AT II) cells may be more favorable for accelerating re-epithelialization36,37, ultimately repairing damaged alveolar structure and facilitating lung function improvement7,89,90.
So far, studies have demonstrated that MSCs can differentiate into alveolar epithelial cells
Pancreatic Cell Differentiation of MSCs
MSCs are a cellular therapy approach for treating type I and II diabetes45,86,227. In particular, the transplantation of artificial insulin-producing cells (IPCs) is a practical approach with permanent results in the treatment of type I diabetes mellitus228–230. Numerous studies have shown that MSCs can successfully differentiate into functional pancreatic islet cells45,86,87,227. In terms of differentiating into IPCs, MSCs can be obtained from multiple sources, with bone marrow and adipose tissue MSCs being the most extensively studied cells45,88.
In humans, the Hh pathway plays an important role in the differentiation of endoderm to pancreatic β-cells in the embryonic gut231,232. Studies have demonstrated that although during early stages of pancreas organogenesis, Hh signaling has been shown to inhibit tissue morphogenesis and cell differentiation, it is important for the reactivation of the SHh pathway during the late maturation of pancreatic β-cells 231 , especially in maintaining the insulin secretion function of IPCs differentiated from MSCs231–237. Therefore, manipulation of SHh signaling pathway can be used as reliable approach to improve the generation of functional IPCs from MSCs238,239. Studies have found that inhibiting Hh activity in the early stages of induced differentiation is required for MSCs to successfully differentiate into IPCs233,234,239. Dayer et al 239 also confirmed that early inactivation and late reactivation of the SHh signaling pathway during differentiation of AD-MSCs can beneficially affect the expression of functional and developmental pancreas-related genes (Pdx1, MAFA, Ngn3, Isl1, Nkx2.2, and Nkx6.1) and insulin secretion in differentiated cells.
Pancreatic and duodenal homeobox 1(Pdx1) is a key gene in pancreatic development and insulin regulation, and is required for β-cell maturation and maintenance of β-cell function235,236, and plays a key role in the differentiation of MSCs into IPCs and maintenance of IPCs 240 . Thomas et al233,237 demonstrated that the promoter of Pdx1 contains an Hh response element, and thus SHh affects Pdx1 expression thereby inducing insulin expression and secretion in mature pancreatic β-cells. Hashemi Tabar et al 238 proposed a novel scheme to optimize the differentiation of AD-MSCs to IPCs using Pdx1 overexpression and SHh manipulation (early repression late reactivation). These findings suggest that inhibition of the SHh signaling pathway during early MSCs differentiation and reactivation of this pathway in later stages are important biological processes for MSCs differentiation into IPCs. Because increasing evidence indicates that the differentiation state of IPCs is not permanent, and they lose their identity and functionality in response to a variety of signals, including changes in the transcriptional profile normally present in mature β-cells241,242. Manipulation of the pancreatic signaling pathway (such as SHh pathway) is a practical approach to increase the efficiency of differentiation of MSCs to IPCs 243 . Although maintaining a certain level of SHh activity in β-cells and IPCs is important and necessary for optimal β-cell function 244 , excessive SHh activity can impair mature β-cells function 239 .
Hepatocyte Differentiation of MSCs
Liver disease is the 11th leading cause of death globally 245 . Regardless of the underlying etiology of the disease, patients will develop similar liver dysfunction in the later stages of the disease. Currently, only liver transplantation has a curative effect on late-stage disease, but the availability of donor liver is limited. The use of liver cell-like cells manufactured from MSCs for the treatment of liver disease is a hopeful strategy for late-stage liver disease patients, making MSCs of significant research value and having broad application prospects in liver diseases. Due to the ability of MSCs to promote liver regeneration and alleviate liver damage by differentiating into liver cells, inhibiting the release of inflammatory factors, and enhancing the proliferation of intrahepatic cells, MSC-based therapy has become a focus for promoting liver regeneration and repairing liver damage64,246–248. Based on the demonstrated efficacy and safety of MSC therapy in preclinical studies in patients with no other treatment options, MSC-based therapy has become a promising approach for the treatment of liver disease64,82,245–248.
Although numerous studies have demonstrated the MSCs transplantation can promote liver regeneration through differentiation into liver cells64,82–85,245,246, reports on the role and mechanism of Hh pathway in MSCs differentiation into hepatocytes are rare. In the damaged liver, dying liver cells have been shown to release Hh ligands249,250. These Hh ligands trigger the proliferation of Hh-responsive cells, such as liver progenitor cells (liver MSCs) and hepatic stellate cells (HSCs), and these cells also produce Hh ligands, amplify the activity of the Hh signal in these cells through autocrine and paracrine mehanisms249–252. One study report that MSCs can promote liver regeneration by a mechanism that has a significant increase in miRNA-125b levels expressed by MSCs, leading to a reduction in Smo and inhibition of the Hh signaling, thereby reducing fibrosis, and ultimately promoting liver regeneration 81 . Although some studies have indicated that MSCs play an important role in liver regeneration by differentiating into liver-like cells64,82–85,245, the exact mechanism of the Hh pathway in MSCs differentiation into hepatocytes requires further exploration.
Future Perspectives
Cell therapy aims to replace damaged resident cells by restoring a cellular and molecular environment suitable for tissue repair and regeneration
253
. Although the mechanism of action of MSCs is mainly related to their secretome (including chemokines, cytokines, growth factors, and nucleic acids), direct differentiation of cells into functional target cells can better and more quickly promote the regeneration and repair of damaged tissues. Nevertheless, the low efficiency of differentiation limits use of MSCs in clinical disease treatment. Optimizing the directed differentiation potential of MSCs may be one of the important strategies to significantly improve MSC therapy efficacy. More importantly, the observation of MSCs demonstrate their capacity to transdifferentiate into distinct forms of mesodermal, ectodermal, and endodermal cells when exposed to a particular concoction of growth factors and substrates
7
. These target cells induced by
Footnotes
Authors’ Contributions
M.W. wrote the manuscript. H.H. and J.J. designed the framework of the manuscript. J.M. and G.Q. wrote about the role of Hh in the differentiation of endodermal cells of MSCs. C.G. and Q.C. collated references. S.Z., Y.J., and J.L. revised the manuscript. All authors read and approved the final manuscript.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This paper was supported by the National Natural Science Foundation of China (nos. 82272282, 82020108021, and 81571912).
