Abstract
Stem cell aging is a process in which stem cells progressively lose their ability to self-renew or differentiate, succumb to senescence or apoptosis, and eventually become functionally depleted. Unresolved oxidative stress and concomitant oxidative damages of cellular macromolecules including nucleic acids, proteins, lipids, and carbohydrates have been recognized to contribute to stem cell aging. Excessive production of reactive oxygen species and insufficient cellular antioxidant reserves compromise cell repair and metabolic homeostasis, which serves as a mechanistic switch for a variety of aging-related pathways. Understanding the molecular trigger, regulation, and outcomes of those signaling networks is critical for developing novel therapies for aging-related diseases by targeting stem cell aging. Here we explore the key features of stem cell aging biology, with an emphasis on the roles of oxidative stress in the aging process at the molecular level. As a concept of cytoprotection of stem cells in transplantation, we also discuss how systematic enhancement of endogenous antioxidant capacity before or during graft into tissues can potentially raise the efficacy of clinical therapy. Finally, future directions for elucidating the control of oxidative stress and developing preventive/curative strategies against stem cell aging are discussed.
Introduction
Stem cells are ontogenically diverse cell populations distributed in niches throughout the human body. They are unique in being pluripotent and endowed with a lifelong capacity for self-renewal. As stem cells can develop into a virtually infinite spectrum of cell types in organisms, prospects of their wide application in regenerative medicine, drug screening, and disease modeling have been suggested in the past decades. According to their origins, stem cells can be broadly divided into embryonic stem cells (ESCs) and adult stem cells (ASCs). Although ESCs are totipotent to form a brand new organism, studies on human ESCs are subject to ethical controversies, particularly in countries following the Christian faith. 1 To address this pitfall, ASCs have been extensively studied instead for applications in regenerative medicine, 2 on account of their apparent advantages in wide body distribution (e.g., bone marrow, peripheral blood, fat, cornea, retina, brain, skeletal muscle, dental pulp, liver, kidney, skin, gastrointestinal tract endothelium and pancreas), easy isolation, pluripotency, and immune-regulatory properties. Following transplantation, ASCs are capable of “sensing” locations of wound sites and migrating to those sites for tissue repair, through either direct reoccupying or paracrine actions to stimulate host organ regeneration. 3
Aging is an essential physiological process in most cell types. When growth limits are reached, mammalian cells enter permanent cell cycle arrest, referred to as replicative senescence, mediated by internal factors such as telomere length, telomerase activity, and other aging-related genes. 4,5 Another major aging mechanism, premature senescence, is affected by the cell microenvironment and can be elicited by stressful stimuli to initiate irreversible arrest of proliferation. 6 For example, many intrinsic and extrinsic factors, such as changes in genetic structure and chromatin modifications, and nonoptimal culture conditions (e.g., nutrients, temperature, drugs, radiation, inflammation, mechanical stress, and oxidative stress), can induce replicative and premature senescence. 7 Indeed, replicative senescence and premature senescence share many similarities, including a specific set of alternations in cell function, morphology, gene expression, and positive staining for senescence-associated β-galactosidase activity (SA-βgal). 8 In contrast, genome-wide gene expression analysis show that cell aging due to different factors rarely overlap in the gene expression profile, which implies that the aging process is distinctly regulated by multiple signaling pathways. 9,10
Aging Biology of Stem Cells
Maintaining a balance between self-renewal and differentiation is extremely important for stem cell homeostasis, since excessive self-renewal favors carcinogenesis, while inordinate differentiation induces premature consumption. 11 During replication, various stress stimuli promote stem cell aging through several signaling pathways, resulting in a series of cellular alterations, including cell morphological changes, compromised proliferation capacity, defective nutritional sensing, reduced genomic stability, telomere shortening, epigenetic alterations, impaired protein structures, mitochondrial dysfunction, dysregulated intercellular communication, and loss of cellular pluripotency. 12,13 In a broad sense, major promoters of stem cell aging include accumulation of age-related toxin metabolites, DNA damage, protein oxidation, mitochondrial dysfunction, and stem cell depletion in aged tissues. 14 Indeed, reactive oxygen species (ROS) generated from normal metabolism or extrinsic mediators are among the most important endogenous toxins and regulatory factors in stem cells. They are also the direct inductive factors for DNA, protein, and mitochondrial damage. Thus, from a general perspective, an emerging focus in stem cell aging biology is to understand the mechanisms that underpin the balance between oxidative stress and antioxidative defense in those cells. 15 Concomitant with elevated ROS generation, an increased number of aged stem cells typically leads to a depletion of functional stem cell pools in aged tissues, through a combination of destabilizing events including perturbation of cell cycle activity, decline in self-renewal, improper differentiation, and stress-induced cell death (Fig. 1). 14 Enhancing mitochondrial functions is a logical strategy for restoring aged stem cell function and tissue regeneration, since damaged mitochondria constitute a primary source of ROS, which can act as an inducer of apoptosis. For instance, in the past 100 years, caloric restriction (CR) has been employed to retard aging and extend the life span in diverse organisms including yeast and primates. It has been demonstrated that CR enhances the functions of various types of stem cells through mitochondria-related nutrient sensing and DNA damage pathways. 16 –18 Maintenance of normal mitochondrial functions heavily depends on the oxidized form of cellular nicotinamide adenine dinucleotide (NAD+). Previous studies have shown that supplementation of NAD+ increased the longevity. 19,20 Recently, it was demonstrated that NAD+ acts as a pivotal switch to modulate senescence of muscle stem cells through the mitochondrial unfolded protein response and the synthesis of prohibitin proteins. 20

Aged stem cells are characterized by loss of regenerative function, differentiation, senescence, cell death, and depletion of the functional stem cell pool, which significantly impair their ability to repair damaged tissue.
Oxidative Stress–Evoked Pathways in Stem Cell Aging
In simple terms, oxidative stress can be conceptualized as a suboptimal cellular status arising from deficits in the antioxidant reserve relative to ROS production. High levels of endogenous/exogenous active oxygen and excessive ROS are detrimental factors for the induction of cell aging and a large array of diseases, including cancer. 21 ROS include free radicals such as superoxide (O2 •–) and hydroxyl radicals (•OH) and nonpolar molecules such as hydrogen peroxide (H2O2), which are thought to be some of the principal ROS contributing to an oxidative stress environment. In general, high levels of ROS mediate oxidation of biological macromolecules such as DNA, lipids, and proteins, which consequently causes cellular damage through upsetting intracellular redox homeostasis, exhausting repair machineries, and inducing cell death via intrinsic and extrinsic apoptotic signaling. 22 Emerging evidence suggests that oxidative stress plays a key role in stem cell aging induction and the progression of various diseases. As mentioned, ROS- mediated damage to biological macromolecules can take the form of protein denaturation, lipid peroxidation, DNA modifications, and mitochondrial dysfunction, all of which ultimately promote cellular senescence. 23,24 Other studies also emphasize that accumulation of damaged DNA and protein in stem cells may cause the senescence and loss of organ function. 25,26 Given that mitochondria are a key hub of energy production and regulators of bioenergetics in stem cells, many studies provided corroborative evidence for a direct link between mitochondrial dysfunction and stem cell aging. 27 –29 Thus, understanding the complex signaling networks impacted by oxidative stress as well as interactions between those pathways and diseases is of paramount importance to establish the molecular basis for stem cell antistress capacities (Fig. 2). Here, we summarize several key pathways contributing to oxidative stress-related stem cell aging.
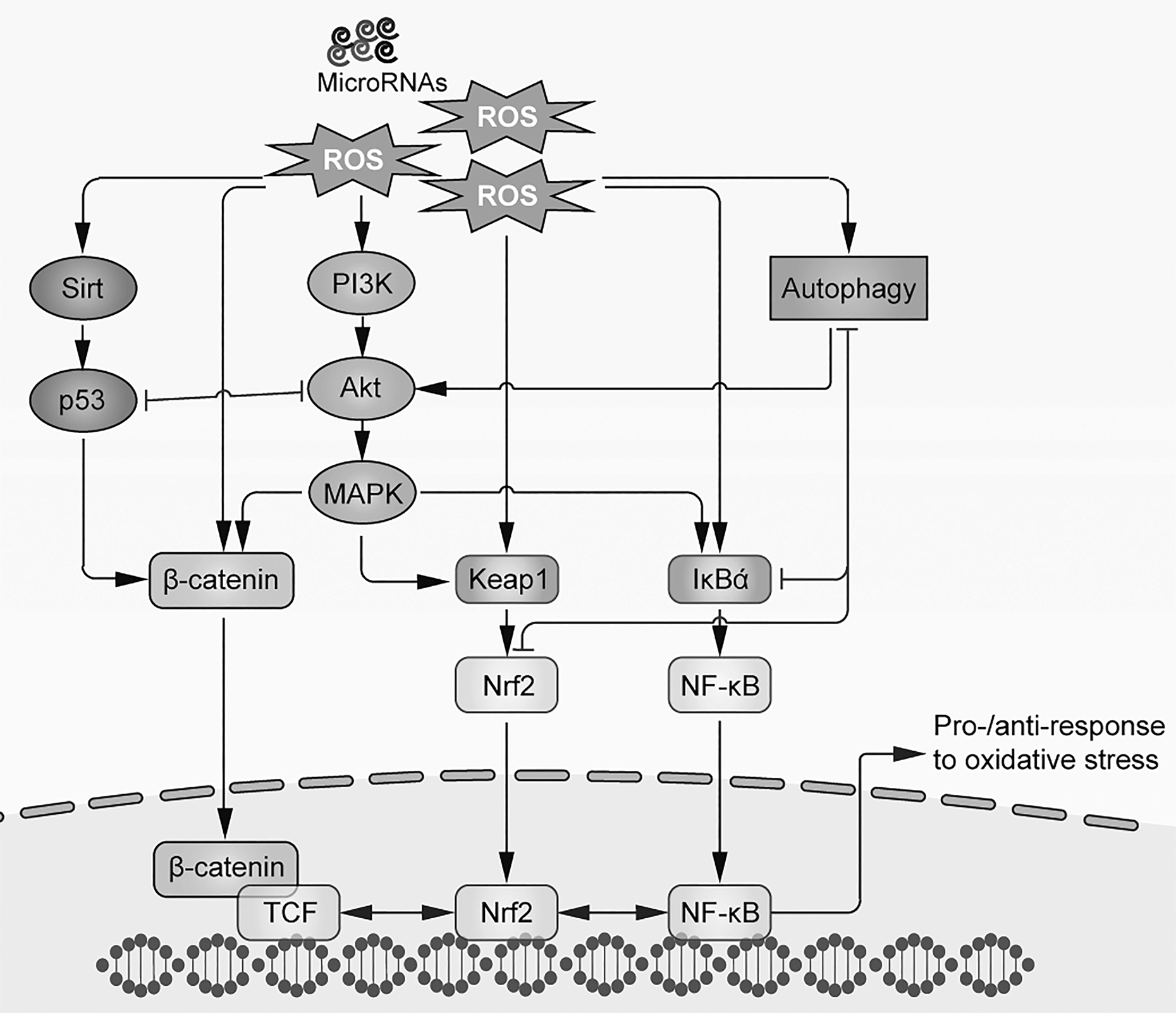
Illustration of oxidative stress–evoked signaling pathways in aged stem cells.
Nuclear Factor-E2-Related Factor 2–Kelch-like ECH-Associated Protein 1 Pathway
Nuclear factor (NF)-E2 Nuclear factor erythroid 2-related factor 2 (Nrf2) is an important transcription factor that controls the cellular antioxidation machinery. In conjunction with Kelch-like ECH-associated protein 1 Kelch-like ECH-associated protein 1 (Keap1) and antioxidant response element (ARE), which make up the Nrf2/Keapl/ARE pathway, the Nrf2 regulates the expression of a series of antioxidant enzymes in stem cells. In the absence of stress conditions, Keap1 is tightly bound to Nrf2 to suppress its activity, allowing a resting status. As oxidative stress sets in, redox-sensitive cysteine residues in the intervening region of Keap1 become oxidized, followed by dissociation of Nrf2 and Keap1 and Nrf2 nuclear translocation. Nrf2 further combines with transcription factor avian musculoaponeurotic fibrosarcoma (Maf), activating transcription factor 4, and Fos-related antigen proteins to form a heterodimer. ARE thus becomes activated to initiate transcription of a host of antioxidant genes so as to mobilize cellular detoxification. 30
A study conducted by Mohammadzadeh et al. proved that transient overexpression of Nrf2 protected mesenchymal stem cells (MSCs) against apoptosis triggered by hypoxia and oxidative stress via upregulation of superoxide dismutase (SOD) and heme oxygenase 1. 31 Elsewhere, it was demonstrated that Nrf2 was crucial for several aspects of hematopoietic stem cell (HSC) homeostasis. Specifically, deficiency of Nrf2 induced cell-intrinsic hyperproliferation and impaired the migration and retention of HSCs in their bone marrow niche, partly through direct interaction between Nrf2 and C-X-C chemokine receptor type 4 (CXCR4). 32 Subsequent studies of Keap1-knockout mice further revealed a critical role of Nrf2 in cell fate determination and cellular ROS regulation of HSCs and human airway basal stem cells. 33,34 A very recent study identified a coordinated regulating system of a cilium-autophagy-Nrf2 control axis and cell cycle progression that directs human ESCs toward neuroectoderm, which was the first decision during ESC differentiation. 35 Antioxidant treatment (e.g., edaravone and N-acetylcysteine [NAC]) has been shown to relieve oxidative stress–suppressed Nrf2 activity in MSCs (e.g., from umbilical cord and muscle), leading to enhanced antistress capacities against exogenous toxin challenge and improved transplantation efficacy in animal models of acute liver failure. 36,37
Wnt/β-Catenin Pathway
When the Wnt ligand binds to cell membrane co-receptors’ low-density lipoprotein receptor-related proteins 5/6 (LRP5/6) or Frizzled receptors, it either activates disheveled protein in the cytoplasmic to directly suppress glycogen synthase kinase 3beta (GSK-3 β) activity or induces the accumulation of β-catenin in the cytoplasm. When a certain cytoplasmic threshold level is reached, β-catenin translocates itself into the nucleus to act as a transcriptional coactivator of transcription factors that belong to the family of T-cell factor/lymphoid-enhancer factor. Numerous studies supported an indispensable role of the Wnt/β-catenin pathway in cell proliferation, differentiation, apoptosis, cell position decision, and carcinogenesis. 38
It has been suggested that the Wnt/β-catenin pathway is implicated in the process of stem cell aging brought on by microenvironmental changes. 39 For example, several studies found that sustained Wnt/β-catenin exposure was an important factor to promote ASC aging in both in vitro and in vivo models. 40 –42 When incubated with old rat serum, rat MSCs exhibited typical senescence phenotypes including reduced proliferation and increased SA-βgal and ROS production. This was partly a result of the activation of the DNA damage response and the p53/p21 pathway evoked by the Wnt/β-catenin pathway. 43 A recent seminal work by Florian et al. confirmed that during HSC aging, elevated levels of Wnt5a caused an unexpected shift from canonical to noncanonical Wnt signaling. Subsequent studies with transgenic animals found that Wnt5a treatment of young HSCs exhibited aging-associated phenotypes, while haplo-insufficiency of Wnt5a attenuated HSC aging. 44 It was reported that chronic ethanol administration suppressed osteoblastogenesis and enhanced adipogenesis of MSCs, leading to evident reduction in bone mineral density. Treatment with antioxidants (e.g., NAC) significantly blocked bone loss and rebalanced stem cell differentiation directly through the recovery of Wnt/β-catenin signaling inhibited by ethanol. 45
p53 Pathway
p53 (tumor protein p53, or tp53) was recognized as one of the most important genes with cellular tumorigenesis. In response to myriad stressors, including DNA damage and oxidative stress, p53 is activated as a transcription regulator, leading to a quick accumulation of p53 in stressed cells. 46 In addition, p53 exerts its influence over a variety of key protein pathways through posttranslational modifications including protein phosphorylation, acetylation, methylation, and ubiquitination. Thus, p53 serves as a “genome guardian” to monitor the integrity of the genome and maintain cell homeostasis. 47
It has been suggested that oxidative stress elicits a specific p53 transcriptional response, which is mediated by p44/p53 and p66 to control cellular senescence and aging. Studies have also found that oxidative stress could enhance the protein acetylation levels of p53 and promote cell aging, implicating the NAD+-dependent deacetylase enzyme sirtuin 1 (SIRT1). 48,49 However, oxidative stress environments do not always induce cell aging, as evidence exists that when cells are maintained at a low level of oxidative stress, p53 primarily induces the expression of antioxidant genes to prevent cell death. By contrast, high levels of p53 could accelerate the generation of ROS and induce cell death under severe cellular stress. 50 In stem cells, in response to telomere attrition and oncogenic stimuli, activation of p53 depletes HSCs. 51 Besides mTOR and Wnt, p53-induced phosphatase 1 (Wip1) was also shown to regulate HSC aging through direct p53 actions, since Wip1-knockout mice exhibited multifaceted phenotypes of HSC aging while deletion of p53 rescued the condition. 52 However, the precise role of p53 in stem cell aging is complex as others have found that increased p53 function in aged HSCs (isolated from 12-month old p53+/m transgenic mice with more p53 activity than wild-type mice) was associated with slower rate of cell proliferation but a relatively younger status at a molecular level. 53 Furthermore, transgenic mice with p53 overexpression did not display signs of accelerated aging. 54 A possible explanation is that p53 may help maintain tissue homeostasis by suppressing pathologic hyperproliferation and aberrant stem cell differentiation. 12
Inhibition of p53 activity has been suggested as a strategy for preventing stem cell quiescence since deficiency of connexin 43 in bone marrow-MSCs exhibited hyperactivated p53 and treatment with antioxidant NAC restored stem cell stemness via p53 suppression. 55 Moreover, NAC enhanced hESC stemness and maintained cellular homeostasis by regulating hypoxia-inducible factor-2α-suppressed p53 activity. 56
Phosphatidylinositol 3-Kinase /Akt/Mechanistic Target of Rapamycin Signaling Pathway
Phosphatidylinositol 3-kinase (PI3K)-Akt pathway is thought to be the most important prosurvival pathways in cells. Upon activation by various factors such as epidermal growth factor, sonic hedgehog, insulin growth factor 1 (IGF-1), and insulin, PI3K quickly mobilizes Akt that localizes to the cell membrane. The PI3K/Akt pathway directly regulates cellular quiescence, proliferation, cancer, and longevity. 57 Mechanistic target of rapamycin (mTOR) is a direct target of Akt for the regulation of cell growth, autophagy, and metabolism. Under diverse conditions including oxidative stress, they form the PI3K/Akt/mTOR pathway to coordinately direct cell fate. 58
Evidence has shown that the reduction in the activation of PI3K/Akt/mTOR signaling pathway extends life span in healthy organisms, that is, from yeast to mammals. Moreover, aberrant signal transduction in this pathway is one of the major pathogenic factors of aging. 59 In vitro study suggested that this pathway inhibited aging and promoted self-renewal of human skin-derived precursors. 60 In a myocardial ischemia/reperfusion injury model, MSC-derived exosomes were found to enhance myocardial viability and ameliorate oxidative stress through the PI3K/Akt pathway. 61 It was found that high-density lipoprotein protected MSCs from oxidative stress–induced cell death through regulation of the PI3K/Akt pathway. 62 Furthermore, a recent study reported that blocking of the PI3K/Akt/mTOR pathway prevented aging phenotypes and enhanced proliferative capacity of MSCs. Reduction in intracellular oxidative stress, prevention of DNA damage, and induction of pluripotency gene expression (e.g., Nanog and octamer-binding transcription factor 4) were thought to be the main mechanisms underlying the observations. 63
Nuclear Factor-Kappa B Pathway
Nuclear factor-kappa B (NF-κB) is a master transcriptional regulator of immune response and cell death. It is well-known that oxidative stress triggers inflammatory cascades that are primarily mediated by NF-κB. Study found that ROS activated inhibitors of NF-κB (IKBS) ubiquitination, NF-κB translocation, the stimulation of interleukin 8 (IL-8) expression, and/or increase of p53 protein stability, leading to cell aging intervention. 64 This finding was further confirmed in induced pluripotent stem cells (iPSCs); NF-κB was repressed during cell reprogramming toward their pluripotent state while hyperactivation of aging-associated NF-κB inhibits iPSC generation via eliciting the reprogramming repressor DOT1-like histone H3K79 methyltransferase (DOT1L). 65 Furthermore, p65 isoform of NF-κB was activated and accumulated in aged HSCs, most likely increasing the expression of P-selectin and reflecting a time-dependent increase in inflammation. 53 IGF-1, mTOR, SIRT1, and p53 are reported to be the upstream signaling regulator of the NF-κB pathway during aging. 66 Attenuation of NF-κB activity (primarily p65) by heat shock protein 90 (HSP90) inhibitor, 67 NAC, 37 myoblast determination protein (MyoD), 68 and NF-κB small molecule inhibitor 69 was reported to reduce cellular oxidative stress, alleviate cell death, and enhance stemness in various stem cell types.
Mitogen-Activated Protein Kinase Signaling Pathway
Mitogen-activated protein kinase (MAPK) is a family of serine/threonine protein kinases that are widely distributed in mammals and mainly includes extracellular signal-regulated kinase 1/2 (ERK1/2), c-JUN N-terminal kinase (JNK), p38, and ERK5 members. MAPK has been identified as a major regulator in cell growth, differentiation, stress environment, cell death, and inflammatory response. This pathway can be activated by various extracellular stimuli such as physical cues, inflammatory cytokines, growth factors, and bacterial components. 70
The roles of MAPK in cell aging have been investigated in a number of studies. For example, when compared with young mice, aged mouse livers exhibited decreased ERK1/2 level but increased JNK1/2 and p38 MAPK levels. 71 However, data from rat aorta and human skeletal muscle were distinct, indicating that MAPK is affected by aging in a tissue-specific manner. 72,73 It was also found that signaling pathways evoked by oxidative stress were stimulus source dependent, since endogenous H2O2 produced from hypoxic stress induced ERK activation, while exogenous H2O2 primarily induced p38 MAPK activation. 74 In HSCs, p38 MAPK is activated by ROS to augment the expression of p16 and p19 alternate reading frame (ARF), thus limiting the self-renewal capacity of HSCs as well as the life span of mice. 75 Interestingly, in aged skeletal muscle, a stem cell autonomous loss of self-renewal potential was investigated with alterations in p38 MAPK and fibroblast growth factor receptor 1 signaling. Pharmacological manipulation of p38 MAPK significantly alleviated age-associated stem cell self-renewal defects. 76 Furthermore, evidence exists for crosstalk between the MAPK and Nrf2 pathways, since oxidative stress activates the MAPK-Nrf2-ARE axis to regulate phase II enzyme (e.g., SOD and glutathione S-transferase) expression and activity, which consequently modulates cell growth and aging. 77 By treating stem cells with antioxidant compounds (e.g., NAC, edaravone, or zeaxanthin dipalmitate), the oxidative stress–evoked activation of MAPK pathway components (e.g., p38 MAPK and ERK) was significantly suppressed, which facilitates ROS detoxification and attenuation of cell injury. 36,37,78 However, it seems that other key proteins, particularly those downstream of MAPKs, also play substantial roles in the antioxidative defense of stem cells against injury and aging. 36
Sirtuin Pathway
Sirtuins are a class of proteins with NAD-dependent deacetylase activity. They are functionally significant in regulating stem cell proliferation, death, differentiation, and aging, particularly in the contexts of oxidative stress. 79 SIRT1 and adenosine monophosphate-activated protein kinase (AMPK) were reported to coordinately restore aging-impaired cell growth, differentiation, and mitochondrial functions of MSCs. 79,80 A recent study found that miR-34a mediates this process by acting as pro-apoptotic and pro-senescence factors in MSCs via direct targeting of SIRT1. Inhibition of miR-34a resulted in improved viability and antistress functions of MSCs in an in vitro model of hypoxia and serum starvation. 81 In addition, the osteogenic and adipogenic differentiation of tendon or dental pulp stem cells were also controlled by SIRT1, which involved signal transduction of both Wnt/β-catenin and PI3K/Akt pathways. 82,83 Unlike SIRT1, studies on the roles of SIRT2 in stem cell biology are scarce. Si et al. found that SIRT2 was upregulated during embryoid body differentiation of mouse ESCs. Knockdown of SIRT2 shifted ectoderm to mesoderm/endoderm differentiation through the GSK-3β activation. 84 As a regulator of mitochondrial homeostasis and oxidative stress, SIRT3 is dispensable for young HSC functional maintenance but becomes essential at an old age. Controlling mitochondrial plasticity and SIRT3 expression is a promising strategy to reverse age-related degenerations. 85 Other studies have also suggested that decreased SIRT3 and NAD+ levels in aged MSCs and somatic cells increased cell vulnerability to oxidative stress and senescence. 86,87 The roles of SIRT6 in stem cell oxidative stress and aging have been intensively investigated over the past decade. It is one of the few known genes that can modulate both longevity and progeria, since overexpression of SIRT6 led to enhanced longevity and SIRT6-deficiency-induced progeroid phenotypes. 88,89 A recent study illustrated a protective role of SIRT6 against oxidative stress–induced MSC injury, in which SIRT6 coactivates Nrf2 and RNA polymerase II. 90 Data from an HSC homeostatic study highlighted the importance of epigenetic regulation of Wnt signaling for ASC homeostasis and self-renewal capacity. 91 Furthermore, the NF-KB pathway was exhibited to be quite crucial in SIRT6-mediated stem cell differentiation and senescence. 92,93
An appropriate treatment using the antioxidant NAC has been found to postpone the aging process of mouse oocytes partly through the upregulation of both SIRT1 and SIRT2. 94 In a mouse model of laminopathy-based progeria, a potent antioxidant agent from grape seed, resveratrol, reportedly rescued SIRT1-dependent ASC decline and extended the life span of mice. Lamin A was identified as a direct activator of SIRT1 to induce deacetylase inactivation. 95 In hESC-derived mesenchymal progenitors, resveratrol promoted cell osteogenesis but inhibited adipogenesis via the SIRT1/ forkhead box O3a (FoxO3a) axis. 96 Although the direct involvement of oxidative stress was not investigated in details, it was speculated that major ROS-related pathways were preferentially modulated to enhance the therapeutic potential of antioxidants.
Autophagic Pathway
Since accumulation of damaged proteins and organelles is closely associated with aging and senescence, emerging evidence suggests that deficiency of autophagy–lysosomal activity underpins cell aging. 97,98 In HSCs, deletion of autophagy-related gene 7 (Atg7) induced mitochondrial dysfunction, DNA damage, and oxidative stress. Warr et al. further proved that during HSC aging, FoxO3A-induced autophagy was indispensable for metabolic stress protection. 99 Notably, mTOR signaling in intestinal stem cells was shown to regulate the self-renewal and intestinal niche function maintenance. 100 To preserve cell integrity, young quiescent muscle stem cells often use autophagy to guarantee cellular homeostasis and as a protein quality control strategy. However, autophagic regulation is significantly impaired in aged stem cells, which negatively affects the ability of the cells to provide nutrients for the transition from quiescence to activation or maintain stemness by preventing senescence. 101,102 Although not well-defined, the mechanistic link between autophagy and other key cellular events (e.g., inflammation and apoptosis) should also be quite important for stem cell aging. 103,104
Numerous lines of evidence have converged on the idea that autophagy could be a therapeutic target of antioxidant agents. For example, human umbilical cord MSCs with irradiation injury were protected by starvation- or rapamycin-induced autophagy through decreasing ROS production. Treatment with NAC partially mimicked this process. 105 Moreover, H2O2-induced hMSC mitoptosis, necroptosis, and apoptosis were attenuated by a synergistic protection of NAC and ascorbic acid 2-phosphate. The antiapoptotic B-cell lymphoma 2 (Bcl-2), as well as histone-2AX and dynamin-related protein 1 pathways were defined as the main protective mechanisms. 106 Interestingly, autophagic pathway was shown to be induced during high-glucose-induced bone marrow-derived MSC senescence. Application of the autophagic inhibitor 3-Methyladenine (3-MA) or NAC could prevent cell growth arrest and restore stemness via ROS elimination. 107
MicroRNA-related Pathways
MicroRNAs (miRNAs) are endogenous nontranscriptional small RNA of 20–24 nucleotides in length. They function as crucial posttranscriptional regulators in diverse processes including gene expression, protein synthesis, and even cell differentiation and apoptosis. In particular, an essential role of miRNAs in ESC differentiation has been recognized. For example, loss of miRNA-processing enzyme Dicer caused ESC differentiation defects and the death of mouse embryos during early development. The pathological association between oxidative stress and microRNA-related pathways has been established by several recent studies. In adipose tissue-derived stem cells, ROS from hypoxia, antimycin, rotenone, or platelet-derived growth factor (PDGF) upregulated miR-210 expression and increased stem cell proliferation/migration via protein tyrosine phosphatase, nonreceptor type 2. 108 Our study found that regulation of miR-210 expression by zeaxanthin dipalmitate was important for the maintenance of cellular detoxification capacity against oxidative stress and the enhancement of transplantation efficacy in an acute liver failure model. 109 It was subsequently found that overexpression of miR-210 could enhance MSC antioxidant ability and survival after ROS challenge through the activation of c-mesenchymal–epithelial transition. 21 In addition, it was found that direct binding of miR-141-3p to zinc metallopeptidase STE24 transcripts was responsible for prelamin A accumulation in the nuclear envelope of human MSCs, which led to cellular senescence. 110 Maturation of lethal 7 was demonstrated to bind high-mobility group AT-hook 2 to regulate the inhibitor of cyclin-dependent kinase type 4/Arf expression, which was crucial for the decrease in the ability of neural stem cell self-renewal. 111
The Antioxidant Response Pathways in Stem Cells
Cellular redox status significantly influences stem cell homeostasis. The existence of oxidative stress frequently demands an adaptive response from the endogenous antioxidant stress machinery, which in turn significantly modulates the level of oxidative stress. On close inspection, stem cells may respond differentially to different levels of oxidative stress. Under a mild stress condition, cells mainly regulate apoptosis-related gene expression, antioxidant enzyme activity, and defensive transduction pathways to fulfill antioxidative needs. In contrast, sustained and intense oxidative stress dampens stem cell proliferation and promotes premature aging, apoptosis, and even tumor formation. 112 Therefore, understanding the defensive antioxidative responses of stem cells has both theoretical and practical significance for improving stem cell homeostasis and clinical transplantation.
The cellular antioxidation machinery encompasses several key pathways. Firstly, through the regulation of endogenous antioxidant enzymes (e.g., SOD, catalase, glutathione peroxidase) and other nonenzymatic molecules (e.g., ergothioneine, vitamin C, microelement), cells efficiently remove excessive oxidants and protect important organelles from ROS-induced damage. Secondly, with the facilitation of antioxidant molecules, cells are able to inhibit cellular senescence and apoptosis by regulating the expression of apoptosis-related genes such as Bcl-2, Bax, p53, and inhibitors of apoptosis proteins. Essentially, the antioxidant effect in stem cells does not take place in a single approach but through the multiple signaling pathways. 113
Controlling Oxidative Stress as a Therapeutic Strategy in Regenerative Medicine
Although stem cell transplantation has been shown to improve clinical outcomes in a number of diseases, poor cell survival following transplantation has limited its efficacy. This is mainly attributable to the severe inflammation and oxidative stress that negatively impact the microenvironment at the sites of injury. Some animal studies and clinical trials have also suggested that oxidative stress is negatively associated with stem cell transplantation efficacy. 114,115 Accordingly, it has been posited that through systematic intervention that enhances resistance to oxidative stress, the therapeutic efficacy of stem cells could significantly improve. 23,37,116 Indeed, resistance to stress are regarded as a cardinal characteristic among stem cells. 117 A growing body of evidence has revealed that oxidative stress tolerance was mainly due to intrinsically high expression and activity of antioxidant enzymes. 118 Meanwhile, it has also been reported that treatment with antioxidants such as NAC and edaravone with stem cells allows significantly improved survival and tissue repair capacity. 36,37 In summary, stem cell aging not only diminishes their structural integrity and ability to repair tissue but is also detrimental to stem cell transplantation efficacy. Clearly, a better understanding of the complex regulatory networks controlling stem cell health and aging is critical for delaying the senescence and improving the clinical outcomes of stem cell transplantation. 119 Recent studies using antioxidants to improve stem cell therapy efficacy are summarized in Table 1.
Summary of Studies Using Antioxidants to Improve Efficacy of Stem Cell Therapy.
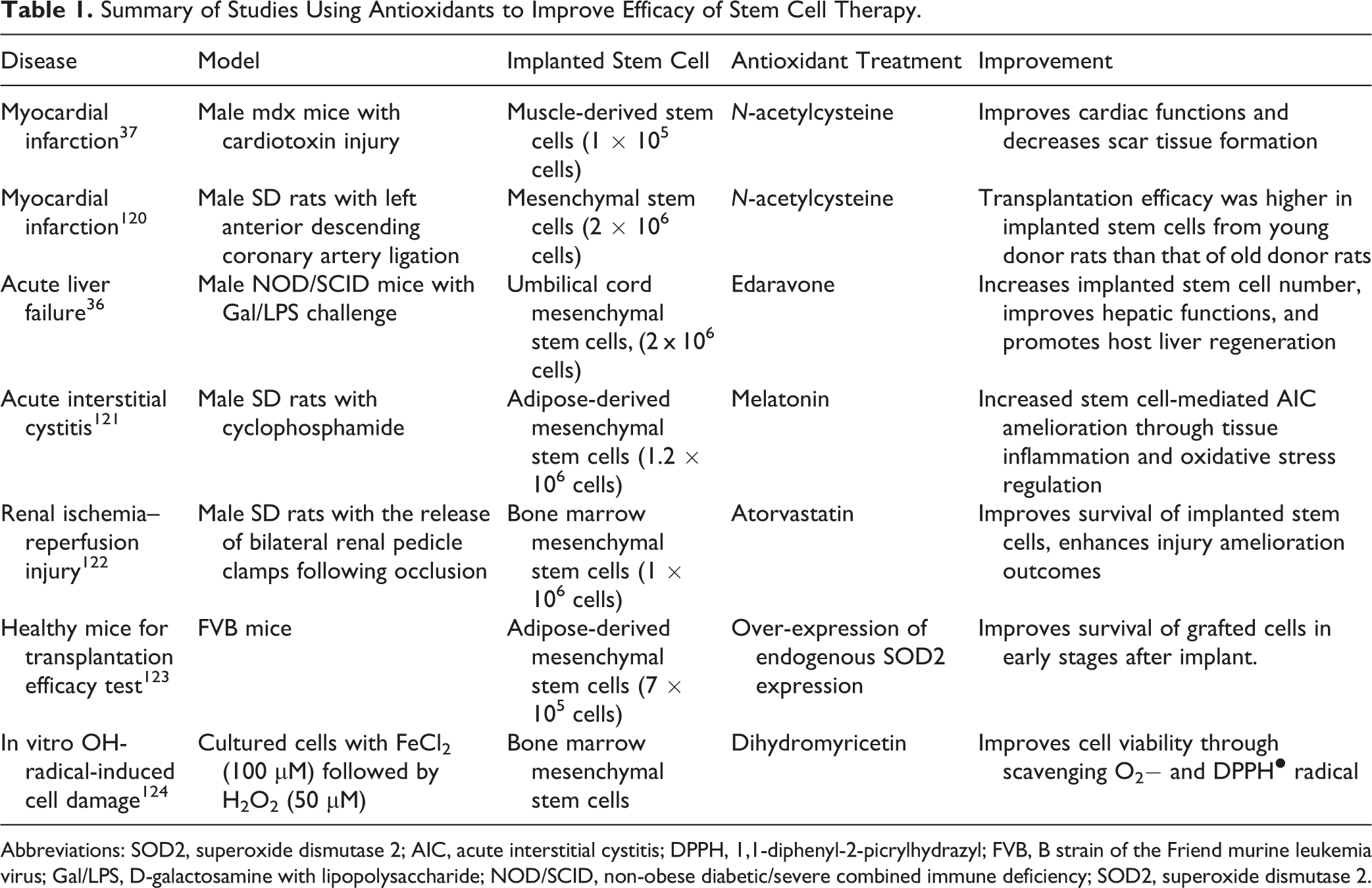
Abbreviations: SOD2, superoxide dismutase 2; AIC, acute interstitial cystitis; DPPH, 1,1-diphenyl-2-picrylhydrazyl; FVB, B strain of the Friend murine leukemia virus; Gal/LPS, D-galactosamine with lipopolysaccharide; NOD/SCID, non-obese diabetic/severe combined immune deficiency; SOD2, superoxide dismutase 2.
Conclusion and Perspectives
The consequences of stem cell aging can be both subtle and overt, ranging from impairment of cellular homeostasis, vulnerability to cellular damage, and loss of regenerative function, to increased cell death. This process is modulated by many different intrinsic and extrinsic pathways, whose signaling is more complex due to complex cross talk. The importance of oxidative stress in inducing stem cell aging has been amply illustrated by recent studies, which emphasized the possibility of maintaining cellular homeostasis and enhancing transplantation efficacy through regulation of the endogenous antioxidation machinery. In addition, pathways for DNA damage repair, protein translation/stability, maintenance of mitochondrial function, and stem cell pool replenishment in aged tissues are receiving increasing attention in recent years as targets to prevent stem cell aging.
14
However, several major problems need to be addressed in the context of oxidative stress control and stem cell aging retardation in the near future. Specifically: Interdependence of aging pathways means that it would be difficult to ascertain the pathway that is responsible for certain aging phenotypes or clinical conditions. In most cases, some signals have a greater influence over an aging event. For example, Nrf2 is a general regulatory transcriptional factor for regulation of antioxidant genes and NF-κB is the master regulator of cellular inflammation and apoptosis in aged or damaged stem cells.
125,126
Defining the detailed signaling network is critical for the study of stem cell aging biology. Although many studies supported the effectiveness of antioxidant treatments (e.g., NAC) in improving stem cell antistress ability, antiaging processes, and transplantation efficacy, it remains unclear how those treatments affect age-dependent deficits of stem cells. In addition, further identification and characterization of the direct binding partners or molecular targets of specific antioxidants is required. It is clear that uncontrolled ROS is the major inducer of stem cell aging. However, the pleiotropic roles of ROS in different stem cell populations during distinct phases of aging need to be established. Moreover, whether low levels of endogenous ROS are beneficial to stem cell homeostasis is also a cogent question to answer. Transplantation of functional stem cells has been well explored recently as a method to replenish aged or damaged cells in the context of degenerative diseases, such as neurological disorders.
127,128
However, for the sake of clinical therapy, simple replacement of aged stem cells with young ones is unlikely to be sufficient since a variety of nonautonomous signals contribute to stem cell aging (e.g., extracellular stress signals). Therefore, potentiating the paracrine effects of implanted stem cells and safeguarding the residual aged stem cell functions seem to be practical therapeutic strategies.
76
Genetic manipulation of stem cells (e.g., overexpression of antioxidant gene by lentiviruses) has been shown to enhance the antistress capacity of stem cells against exogenous stressors. This strategy should be handled very carefully since uncontrolled alteration of genomic DNA may induce carcinogenesis. Furthermore, prevention of telomere attrition by upregulating the telomeric pathway is capable of rescuing lost functions of aged stem cells, but this pathway can also be exploited by cancer (stem) cells to overcome replicative senescence.
129
Transient overexpression of key genes involved in antioxidant and/or telomere restoration could be a feasible method to enhance stem cell survival posttransplantation without the risk of carcinogenesis.
It should be noted that although the oxidative stress theory of aging has been explored for past decades, knockout or overexpression of key antioxidant enzymes (except a few examples) did not significantly influence the life span. 130,131 In addition, there has been a lack of clinical trials to validate any correlation between the levels of antioxidants and the human life span. The complexity of the issues may be understood in terms of the sources, nature, subcellular localization and dynamics of cellular oxidative species formed, and which antioxidants could potentially neutralize an oxidant with specificity. Future investigation in this area would demand the development and application of even more sophisticated technologies and methods for ROS detection and molecular imaging so as to unravel the chemical and biological aspects of mechanisms underlying stem cell aging. Overall, initiatives to understand how antioxidant levels of stem cells can be controlled before or during transplantation promise to provide a viable approach for developing translational applications in regenerative medicine.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Major Science and Technology Projects of Guangdong Province (No. 2015B020225005), Basic Research Fund of Shenzhen City (JCYJ20150402111430633), and National Natural Science Foundation of China (31300813, 81370971, 81570552).
