Abstract
This article presents a comprehensive review of the factors influencing the efficacy of mesenchymal stem cells (MSCs) transplantation and its association with platelet concentrates (PCs). It focuses on investigating the impact of PCs’ composition, the age and health status of platelet donors, application methods, and environmental factors on the outcomes of relevant treatments. In addition, it delves into the strategies and mechanisms for optimizing MSCs transplantation with PCs, encompassing preconditioning and combined therapies. Furthermore, it provides an in-depth exploration of the signaling pathways and proteomic characteristics associated with preconditioning and emphasizes the efficacy and specific effects of combined therapy. The article also introduces the latest advancements in the application of biomaterials for optimizing regenerative medical strategies, stimulating scholarly discourse on this subject. Through this comprehensive review, the primary goal is to facilitate a more profound comprehension of the factors influencing treatment outcomes, as well as the strategies and mechanisms for optimizing MSCs transplantation and the application of biomaterials in regenerative medicine, offering theoretical guidance and practical references for related research and clinical practice.
Introduction
In the past few decades, various forms of platelet concentrates (PCs) have been progressively developed and applied 1 . Derived from blood, PCs, such as platelet-rich plasma (PRP) and platelet-rich fibrin (PRF) have found widespread use in orthopedics 2 , dentistry 3 , and other medical fields, demonstrating immense potential in regenerative medicine 4 .
Stem cells, a class of undifferentiated cells with the capacity for self-renewal and differentiation, are also frequently employed in regenerative medicine 5 . Among these, mesenchymal stem cells (MSCs) can, under specific induction conditions in vivo or in vitro, differentiate into various tissue cells, such as osteoblasts, chondrocytes, adipocytes, myocytes, and hepatocytes. MSCs offer advantages, such as ease of acquisition, minimal ethical concerns, and low adverse reactions compared with other types of stem cells 6 . For example, bone marrow-derived MSCs (BMSCs), adipose-derived MSCs (AD-MSCs), amniotic-derived MSCs (AF-MSCs), menstrual blood-derived MSCs (Men-MSCs), and umbilical cord-derived MSCs (UC-MSCs) are among the widely studied sources7–9.
However, the therapeutic properties of exogenous MSCs may be compromised following their transplantation into the human body 10 . Encouragingly, current research indicates that the characteristics, proliferation, and migration of stem cells can be regulated and enhanced11,12. Consequently, there is a growing interest in the development of combined stem cell therapies, such as stem cell combined with acupuncture 13 , stem cell combined with immune cells14,15, and stem cell combined traditional Chinese medicine 16 . Capitalizing on the analogous roles of PCs and MSCs in regenerative medicine, there have been proposals to combine PCs and MSCs for the treatment of relevant diseases. Although this combined approach has shown promising results in both animal and clinical experiments, some findings do not consistently support their synergistic effects17,18.
Considering the above, this review aims to provide insights into whether PC preconditioning of MSCs confers benefits and whether the combined therapy involving both approaches surpasses the efficacy of either treatment alone. Furthermore, it seeks to elucidate the underlying mechanisms and integrate the latest applications of biomaterials to enhance the understanding and implementation of regenerative strategies in clinical settings.
Factors Influencing the Efficacy of the Relevant Treatment and Their Correlation With PCs
The Components of PCs Impact the Efficacy of the Relevant Treatment
It is widely acknowledged that PRP is considered the first generation of PCs, with PRF being categorized as the second generation 19 . These distinctions arise from variations in preparation methods, leukocyte content, and fibrin architecture, potentially leading to diverse impacts on cell regeneration, tissue healing 20 , and the potential for heightened local adverse reactions 21 . Scholars have further classified PCs into four families based on leukocyte content and fibrin architecture: pure PRP (P-PRP), leukocyte PRP (L-PRP), pure PRF (P-PRF), and leukocyte PRF (L-PRF) 22 .
The differences in preparation and composition of PRP and PRF in clinical applications may have an impact on treatment outcomes. In most cases, PRP is prepared by drawing blood from the patient, treating it with an anticoagulant, and then centrifuging it within an hour of collection 23 . In contrast, the preparation process of PRF does not require the addition of exogenous anticoagulants because it is obtained by centrifugation of the patient’s own blood 24 . Then, modifications to the centrifuge protocol and centrifuge tube technology led to the development of a liquid injection version of the PRF (i-PRF) 25 . New methods are now being developed to concentrated PRF (C-PRF), and increased platelet and white blood cell production can increase even more 26 . The use of C-PRF improved growth factor delivery and cell activity compared with the traditional i-PRF regimen. This makes PRF potentially more advantageous in certain clinical scenarios, particularly for patients who are allergic to exogenous anticoagulants or prefer not to use them. In general, the presence of anticoagulants poses a potential risk to wound healing, as they can increase the likelihood of bleeding. However, it is important to acknowledge that wound healing is a multifaceted and intricate process. Interestingly, certain current studies have suggested that anticoagulants may not universally hinder wound healing, as evidenced by recent research findings27,28.
In addition to the presence of obvious additives, it is important to consider potential additives. The selection of centrifuge tubes is crucial for the successful preparation of PRF, as it can significantly impact its quality and clinical effectiveness. Research has shown that many centrifuge tubes used for PRF preparation may contain chemical additives, which can potentially impact clot formation, cellular behavior, and clinical outcomes 29 . Therefore, the preferred approach typically involves using additive-free glass centrifuge tubes for PRF preparation to ensure optimal quality and clinical efficacy.
Moreover, some researchers posit that the efficacy of PCs on various MSCs can be attributed to their high content of multiple cell factors and growth factors 30 . For instance, some scholars observed an increase in the release of vascular endothelial growth factor (VEGF) and insulin-like growth factor-1 (IGF-1) in PRF over time. In an intriguing finding, they noted that when the receptors for VEGF and/or IGF-1 were neutralized, the ability of PRF to promote the differentiation of AD-MSCs into endothelial cells was significantly inhibited 31 . However, it is important to note that due to differences in preparation methods, the secretome composition and types of growth factors released by different PCs can vary 32 .
Exogenous anticoagulants are not required in the preparation of PRF, which circumvents the potential effect of exogenous anticoagulants on the release of MSCs and growth factors33,34. In addition, the relatively more stable three-dimensional fibrin matrix structure of PRF contributes to the protection and gradual release of the enclosed growth factors 35 . This stable structure facilitates the sustained release of growth factors, providing continuous stimulation for cellular regeneration and tissue repair during the wound healing process, thus promoting longer-term activity of growth factors. In summary, PRF may offer potential advantages over PRP in terms of growth factor release, especially in the context of cell activity, and regeneration in vivo. For example, PRF, PRP, and concentrated growth factor were injected into the shoulder region of adult rabbits with rotator cuff lesions, and the corresponding indexes of cell proliferation and angiogenesis were the best in the PRF group 36 . But, some scholars believe that although PRF is better than PRP in the area of wound healing, the healing speed is not as good as PRP 37 .
Therefore, it is suggested that the discussion should be based on more diseases and indicators, and PRF and PRP cannot be simply evaluated. Whether the effects of specific cells or growth factors alone can fully replicate the therapeutic potential of PCs is similarly a subject that requires further research. This highlights the need for continued research to clarify the specific mechanisms underlying the effects of PCs and their individual components on regenerative processes, thereby enhancing our understanding of their potential in clinical applications 38 .
The Age and Health of PCs Donors Affect the Efficacy of the Relevant Treatment
The efficacy of PCs is impacted not only by the previously mentioned preparation methods, but also by the age and underlying health conditions of the donors39,40. Scholars conducted a study using a rat model of myocardial infarction, in which they observed that the application of PRP derived from young donors in combination with BMSCs led to a reduction in the infarct area. This favorable outcome was attributed to the heightened secretion of growth factors, notably, including IGF-1, epidermal growth factor (EGF), and platelet-derived growth factor BB (PDGF-BB), in PRP obtained from younger individuals 41 . Conversely, some scholars discovered that culturing BMSCs in autologous plasma from patients with type 2 diabetes mellitus had a detrimental effect on their vitality and stem cell characteristics, indicating that the differences in donors can indeed influence the efficacy of combined therapy 42 . These findings highlight the critical influence of donor age and health status on the therapeutic potential of PCs, emphasizing the necessity of considering donor-specific factors in the optimization of combined therapeutic approaches. Further research is warranted to elucidate the precise mechanisms underlying these donor-related effects, thereby advancing our understanding of the complexities involved in the therapeutic application of PCs in conjunction with other regenerative therapies.
The Application Methods and Environmental Factors of PCs Affect the Efficacy of the Relevant Treatment
Apart from the age and health condition of the donor, the method of application and environmental factors of PCs may also have a significant impact on their therapeutic effects. Studies have shown that the method of application, injection site, and environmental factors may significantly affect the regenerative therapeutic effects of PCs.
First, the method of application is crucial to the therapeutic effects of PCs. Different application methods, such as direct injection, local application, or internal implantation, may have different effects on the biological utilization and regenerative efficacy of PCs. For example, some studies suggest that directly injecting PCs into damaged tissue sites may be more effective in promoting regeneration than local application 43 . Delivery methods are also limited by technology. Until 2014, PRF was still clinically available only in solid clotting form 44 . Therefore, choosing the appropriate application method is crucial for maximizing the regenerative potential of PCs.
Second, the choice of injection site may also have a significant impact on the therapeutic effects of PCs. Different injury sites or tissue types may require different injection techniques and site selections to maximize the release and action of growth factors and cytokines in PCs. For example, subcutaneous injection may be chosen to promote wound healing 45 , while intra-articular injection may be preferred for relieving knee osteoarthritis 46 (OA). Therefore, personalized treatment plans and technique selections are crucial for achieving optimal therapeutic effects.
Finally, environmental factors, such as hypoxia status, may also affect the efficacy of PCs. When PRP and AD-MSCs are used simultaneously, they have a significant stimulating effect on the proliferation of endothelial cells, with their promoting effect involving angiogenesis45,47. However, under hypoxic conditions, studies have found that PRP-treated AD-MSCs inhibited the phosphorylation of endothelial nitric oxide synthase in endothelial cells, thereby reducing nitric oxide production and inhibiting angiogenesis 48 . Therefore, environmental factors should be considered for their impact on the efficacy of PCs, to develop more effective treatment strategies. Guidelines for the storage temperature, duration, and container materials should be established for PCs to ensure the preservation of growth factors and other bioactive components. This is crucial because the storage time and method of PCs can impact their environment.
Strategy and Mechanism of PCs to Optimize the Efficacy of MSCs Transplantation
The Role and Mechanism of PCs Preconditioning in MSCs Transplantation
Signaling pathways related to PCs preconditioning of MSCs
The therapeutic effects and underlying mechanisms of PCs preconditioning on MSCs have garnered substantial attention within the scientific community. Preconditioning MSCs with PCs have been demonstrated to significantly enhance their biological functions, elevate repair and regenerative capabilities, and ultimately amplify their therapeutic potential. Multiple studies have confirmed the positive impact of PCs preconditioning on the therapeutic efficacy of MSCs, showcasing enhanced effectiveness in facilitating tissue repair and functional recovery across a spectrum of disease models. For example, in a non-healing wound model, preconditioned MSCs have exhibited the capacity to actively promote epithelialization and neovascularization, thereby substantially contributing to the process of wound healing 49 . Furthermore, in the context of cartilage repair, preconditioned MSCs have exhibited the capability to stimulate chondrocyte proliferation and enhance the synthesis of the cartilage matrix, suggesting promising advantages in the treatment of cartilage defects 50 .
The signaling pathways associated with PCs preconditioning of MSCs play a pivotal role in mediating the amplified therapeutic effects observed in preconditioned MSCs. These pathways act as intricate molecular networks that regulate a myriad of cellular processes and responses, ultimately contributing to the enhanced biological functions and therapeutic potential of MSCs after preconditioning with PRP. The ERK/MAPK (Extracellular Signal-Regulated Kinase/Mitogen-Activated Protein Kinase) signaling pathway stands out as one of the key pathways implicated in enhancing MSCs function through PCs preconditioning. Activation of this pathway has been linked to the promotion of cell proliferation, survival, and differentiation. Upon PRP preconditioning, the activation of ERK/MAPK signaling in MSCs may engender heightened regenerative and reparative capabilities, thus improving their therapeutic efficacy across diverse disease models15,51–54. Similarly, the PI3K/Akt signaling pathway plays a crucial role in mediating the effects of PCs preconditioning on MSCs, known for its involvement in regulating cell survival, growth, and metabolism15,51,55,56. Following PRP preconditioning, the activation of PI3K/Akt signaling in MSCs can contribute to increased cellular viability, improved regenerative potential, and heightened resistance to cellular stress, all of which are essential for the therapeutic function of MSCs in tissue repair and regeneration.
Furthermore, it is imperative to recognize that the ERK/MAPK and PI3K/Akt pathways do not act in isolation; rather, they frequently interact and crosstalk with each other and with other signaling cascades to orchestrate a comprehensive cellular response to PCs preconditioning. The integration of these signaling pathways, along with potential modulation of other interconnected pathways, forms a complex regulatory network that underpins the multifaceted enhancements observed in preconditioned MSCs. In summary, the signaling pathways related to PCs preconditioning of MSCs, particularly the ERK/MAPK and PI3K/Akt pathways, represent critical molecular mechanisms through which PRP preconditioning augments the therapeutic potential of MSCs15,51. Understanding the intricate signaling crosstalk and the specific molecular event within these pathways is essential for unraveling the precise mechanisms underlying the enhanced biological functions and therapeutic effects of preconditioned MSCs (Table 1).
PCs Regulates MSCs Through Several Signaling Pathways.
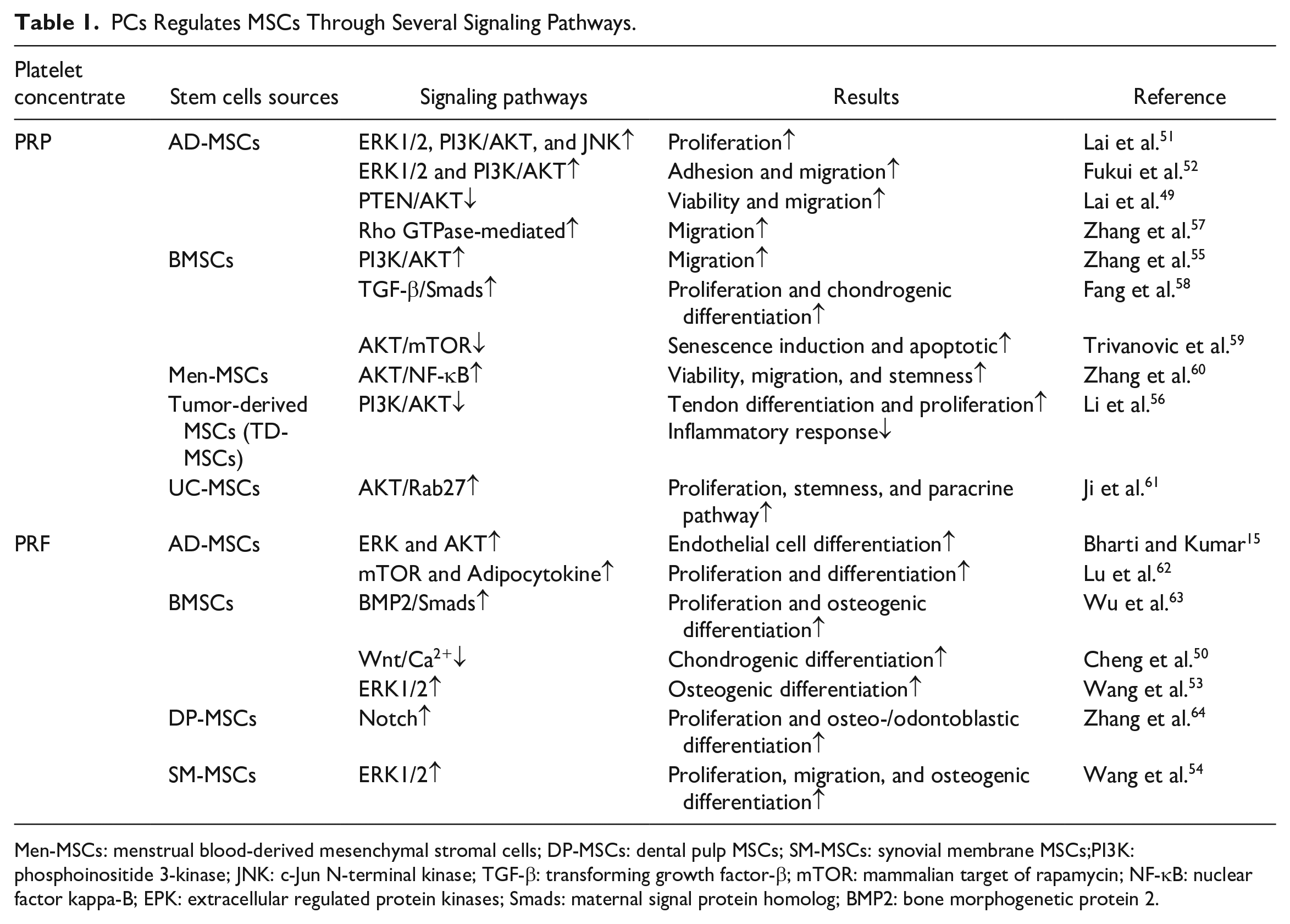
Men-MSCs: menstrual blood-derived mesenchymal stromal cells; DP-MSCs: dental pulp MSCs; SM-MSCs: synovial membrane MSCs;PI3K: phosphoinositide 3-kinase; JNK: c-Jun N-terminal kinase; TGF-β: transforming growth factor-β; mTOR: mammalian target of rapamycin; NF-κB: nuclear factor kappa-B; EPK: extracellular regulated protein kinases; Smads: maternal signal protein homolog; BMP2: bone morphogenetic protein 2.
Proteomic profiles related to PCs preconditioning of MSCs
The term “proteomic profiles” refers to the complete set of proteins expressed by an organism 65 . On the other hand, “Exosomes” represent a distinct category of small extracellular vesicles released by cells, containing a diverse array of bioactive molecules, such as microRNAs, proteins, and growth factors 66 . In summary, proteomic analysis usually involves the study of proteins within Exosomes 67 . The term “secretome” encompasses the complete set of proteins and other molecules that are actively secreted by cells 68 . The secretome encompasses various types of secreted proteins, constituting nearly 30% of the proteomic profile, including antibodies, cytokines, chemokines, growth factors, among others. Studies have revealed that certain preconditioning methods can influence the therapeutic efficacy of MSCs by modulating their proteomic profiles69,70. Similarly, PRP preconditioning of MSCs can impact therapeutic efficacy through similar mechanisms (Table 2), particularly via the secretome.
Part of the Proteome of MSCs is Regulated by PCs.
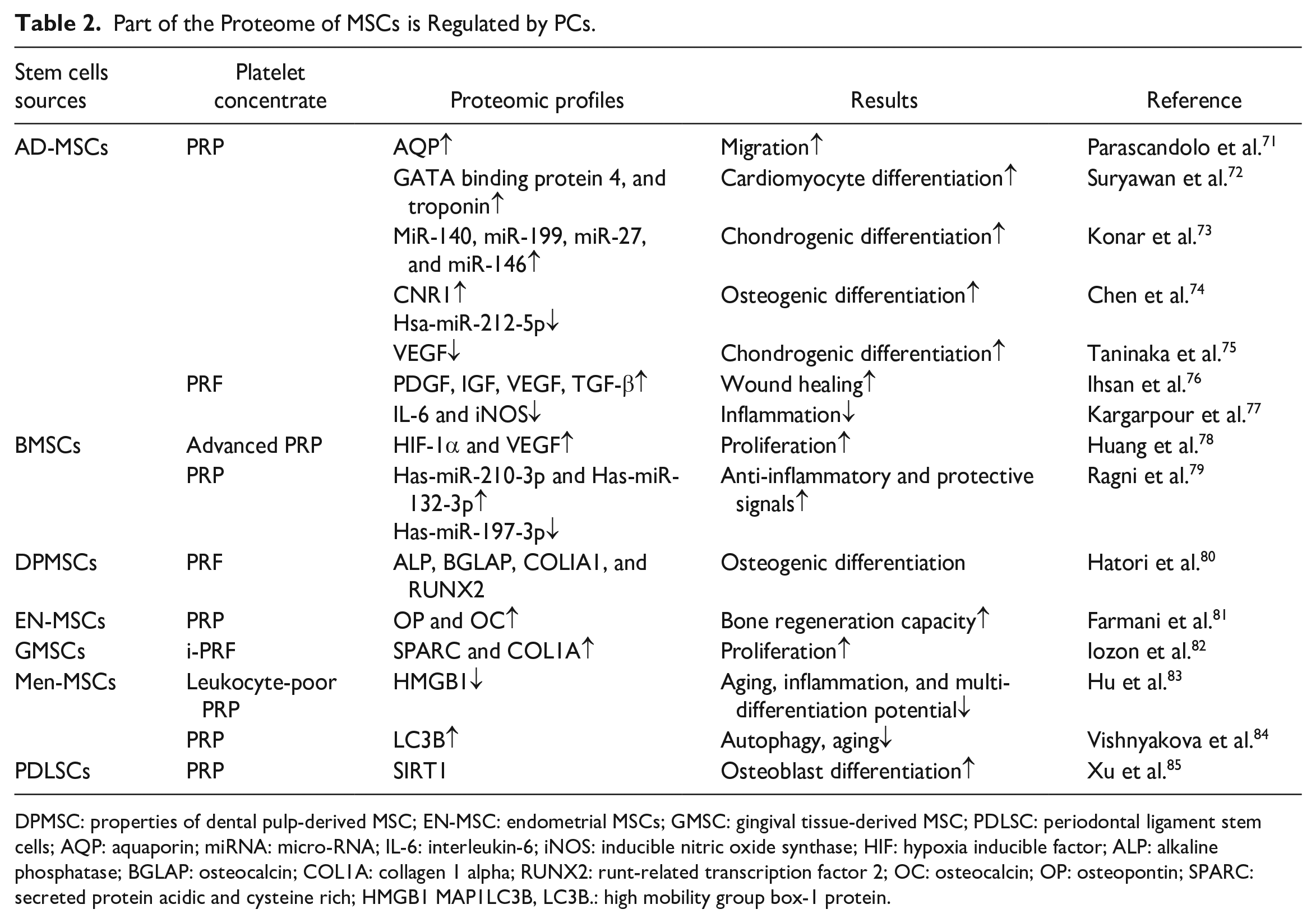
DPMSC: properties of dental pulp-derived MSC; EN-MSC: endometrial MSCs; GMSC: gingival tissue-derived MSC; PDLSC: periodontal ligament stem cells; AQP: aquaporin; miRNA: micro-RNA; IL-6: interleukin-6; iNOS: inducible nitric oxide synthase; HIF: hypoxia inducible factor; ALP: alkaline phosphatase; BGLAP: osteocalcin; COL1A: collagen 1 alpha; RUNX2: runt-related transcription factor 2; OC: osteocalcin; OP: osteopontin; SPARC: secreted protein acidic and cysteine rich; HMGB1 MAP1LC3B, LC3B.: high mobility group box-1 protein.
On one hand, the secretome of PCs and MSCs is often utilized independently for disease treatment. For instance, several scholars conducted a comparative analysis of the secreted proteome of BMSCs and L-PRF, concluding that BMSCs conditioned medium (BMSCs-CM) exhibited enrichment in processes/functions related to cell function, tissue morphogenesis, and extracellular matrix tissue in bone healing, while L-PRF-CM was most enriched in immune regulation, coagulation, and platelet function 86 . Hence, the secreted proteome of PCs and the secreted proteome of MSCs demonstrate both overlap and unique advantages. Moreover, PCs have the capability to directly modulate the secretome of MSCs. Preconditioning amplifies the secretion of proteins by MSCs, upregulating favorable factors and potentially downregulating the expression of unfavorable secreted proteins. PCs preconditioning may also impact the immunomodulatory effects of MSCs. MSCs possess the ability to regulate immune responses and suppress inflammatory reactions, and PCs preconditioning may further potentiate this effect. Preconditioned MSCs may release more immunosuppressive factors, such as TGF-β 76 , thereby modulating inflammatory responses, enhancing tissue microenvironments, and promoting tissue repair and regeneration during the treatment process 87 .
In addition, PCs preconditioning of MSCs can also impact efficacy by regulating non-secreted proteins. For example, preconditioning has been found to promote MSCs migration through the modulation of membrane proteins 71 . These findings underscore the comprehensive and multifaceted effects of PCs preconditioning on MSCs, shedding light on its potential to optimize the therapeutic outcomes of MSC-based transplantation.
Efficacy and Mechanism of Simultaneous Transplantation of PCs and MSCs
The concurrent transplantation of PCs and MSCs represents a synergistic approach to regenerative therapy, capitalizing on the complementary properties of these cell types (Table 3). This strategy aims to enhance the therapeutic efficacy of MSCs transplantation by harnessing their combined effects on survival, engraftment, immunomodulation, angiogenesis, and tissue regeneration. The co-transplantation of PCs and MSCs is expected to augment paracrine signaling, as both cell types secrete a multitude of bioactive factors that collectively enhance the regenerative process. Currently, there are limited preclinical trials investigating the use of PRF and MSCs in disease treatment. Further research is necessary to thoroughly compare the potential benefits and drawbacks of combination therapy involving PRP. Moreover, given the presence of its three-dimensional fibrin matrix, PRF can be considered a superior vehicle for delivering stem cells (MSCs) into the body. This aspect holds considerable promise in the ongoing surge of biomaterials research 105 .
Combining PRP With MSCs: Clinical and Animal Experiments for Disease Treatment.
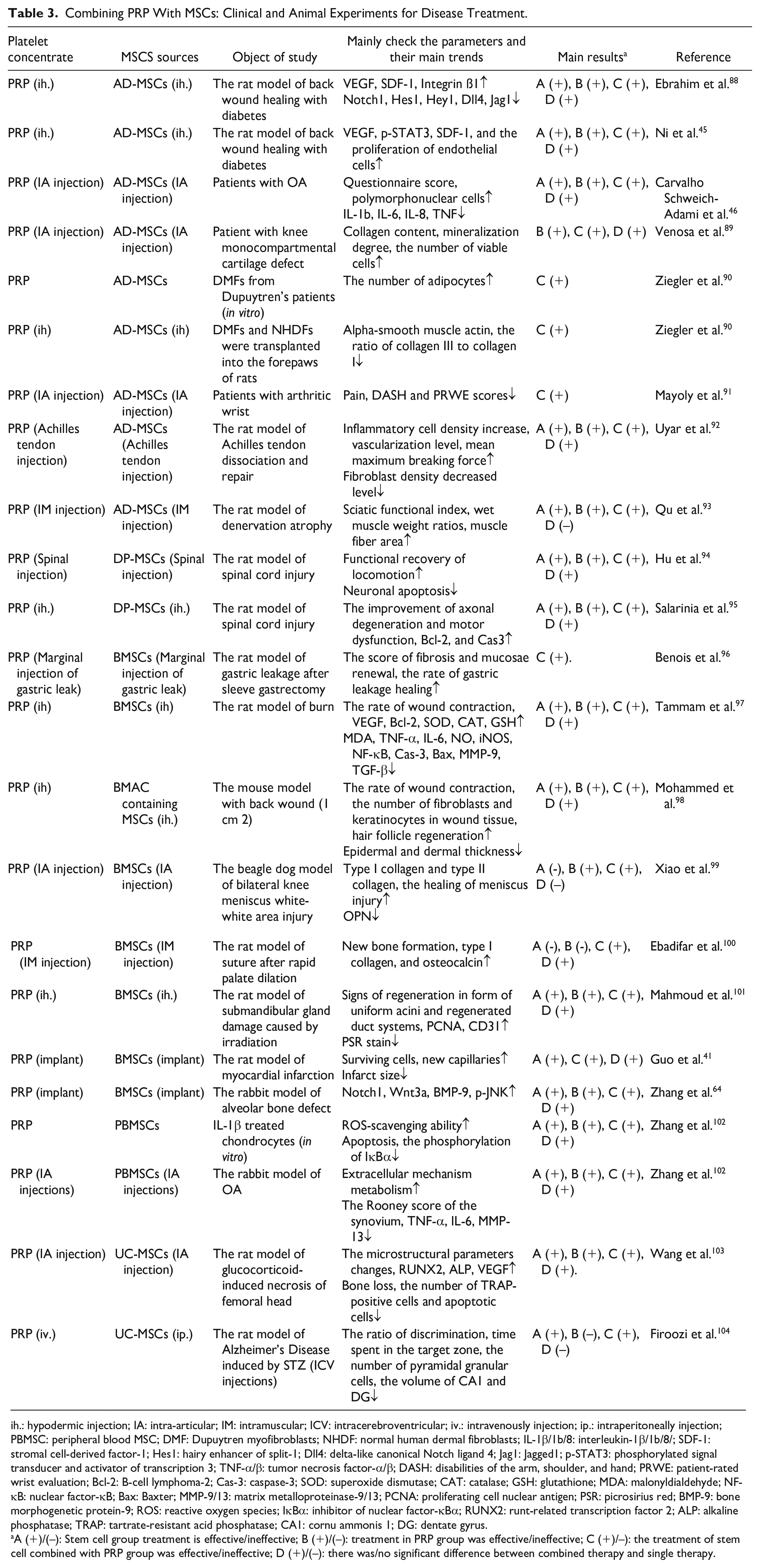
ih.: hypodermic injection; IA: intra-articular; IM: intramuscular; ICV: intracerebroventricular; iv.: intravenously injection; ip.: intraperitoneally injection; PBMSC: peripheral blood MSC; DMF: Dupuytren myofibroblasts; NHDF: normal human dermal fibroblasts; IL-1β/1b/8: interleukin-1β/1b/8/; SDF-1: stromal cell-derived factor-1; Hes1: hairy enhancer of split-1; Dll4: delta-like canonical Notch ligand 4; Jag1: Jagged1; p-STAT3: phosphorylated signal transducer and activator of transcription 3; TNF-α/β: tumor necrosis factor-α/β; DASH: disabilities of the arm, shoulder, and hand; PRWE: patient-rated wrist evaluation; Bcl-2: B-cell lymphoma-2; Cas-3: caspase-3; SOD: superoxide dismutase; CAT: catalase; GSH: glutathione; MDA: malonyldialdehyde; NF-κB: nuclear factor-κB; Bax: Baxter; MMP-9/13: matrix metalloproteinase-9/13; PCNA: proliferating cell nuclear antigen; PSR: picrosirius red; BMP-9: bone morphogenetic protein-9; ROS: reactive oxygen species; IκBα: inhibitor of nuclear factor-κBα; RUNX2: runt-related transcription factor 2; ALP: alkaline phosphatase; TRAP: tartrate-resistant acid phosphatase; CA1: cornu ammonis 1; DG: dentate gyrus.
A (+)/(–): Stem cell group treatment is effective/ineffective; B (+)/(–): treatment in PRP group was effective/ineffective; C (+)/–): the treatment of stem cell combined with PRP group was effective/ineffective; D (+)/(–): there was/no significant difference between combined therapy and single therapy.
MSCs are renowned for their multi-lineage differentiation potential, and the presence of PCs may offer guidance for MSCs differentiation toward specific lineages relevant to the target tissue. This orchestrated differentiation process holds promise for optimizing tissue regeneration and functional recovery. Furthermore, PCs play a crucial role in vascular stabilization and maturation by releasing angiogenic factors, while MSCs can differentiate into perivascular cells and facilitate vascular remodeling. Thus, the concurrent presence of PCs and MSCs is anticipated to synergistically promote vascular stability and functionality in the target tissue, thereby enhancing regenerative outcomes.
The synergistic action of concurrently transplanting PCs and MSCs is anticipated to produce a more potent and sustained therapeutic impact compared with the transplantation of either cell type in isolation. Numerous in vivo and in vitro studies involving human and animal subjects have consistently shown the superior effectiveness of combined PCs and MSCs in promoting regenerative processes and enhancing therapeutic outcomes. Fig. 1 shows some of the diseases for which efficacy may be better when PCs and MSCs are transplanted together.
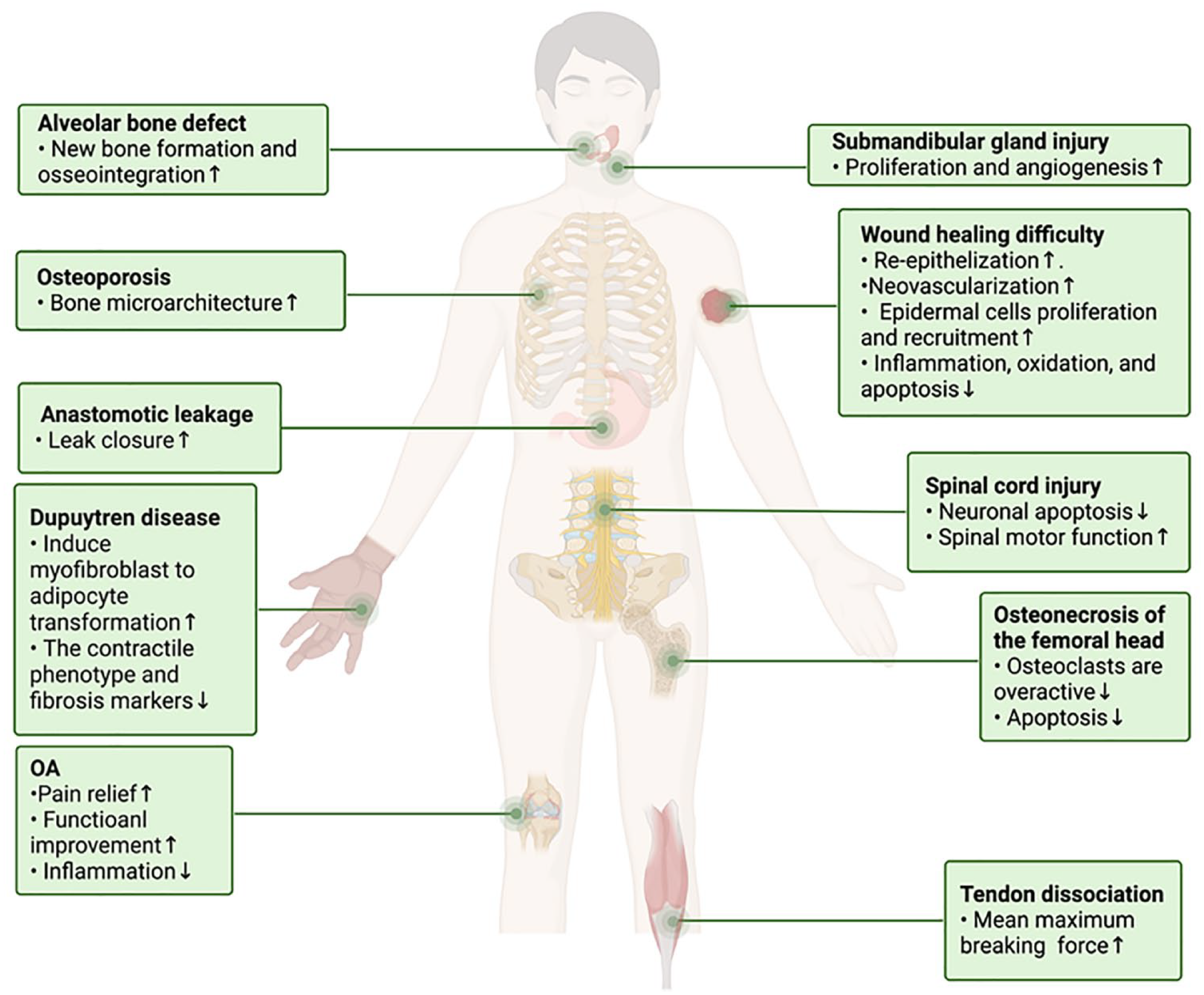
The combination therapy exhibits a synergistic effect in certain diseases.
However, it is important to note that some studies have reported mixed results, with the combined treatment not consistently yielding significantly greater outcomes than individual treatments in certain scenarios. For example, muscle atrophy was observed in rats after superior sciatic nerve dissection following single or combined treatment 93 . In addition, in a 12-month follow-up study of knee OA from Brazil, researchers found that intra-articular injection of BMSCs, even without PRP, was effective in improving function and reducing symptoms caused by knee OA 106 . Similarly, in Beagle dogs with meniscal injury, PRP alone was found to promote the clinical healing rate of meniscal white-white area injury regardless of BMSCs injection 99 . These observations suggest that the effectiveness of the combined treatment may also be influenced by the source of transplanted MSCs. Notably, a systematic review comparing bone marrow MSCs with other therapeutic injections for the treatment of knee arthritis alone also failed to demonstrate a clinical advantage of bone marrow MSCs 107 .
In conclusion, while the concurrent transplantation of PCs and MSCs holds significant promise in regenerative therapy, further research is needed to fully understand the nuanced interactions and potential variations in efficacy across different experimental and clinical contexts.
The Latest Applications of Biomaterials: Optimizing Regenerative Medicine Strategies
In addition to traditional pretreatment or combination therapy to optimize MSCs transplantation, there are emerging technologies, including the use of innovative delivery systems, such as hydrogels, nanofibers, titanium alloys, and other advanced materials to combine PCs and MSCs.
Hydrogels, which are composed of polymer networks capable of absorbing and retaining large amounts of water, have been recognized for their potential to serve as biological scaffolds 108 . For instance, some scholars have demonstrated the use of PRP hydrogel scaffolds in conjunction with AD-MSCs to construct injectable tissue-engineered nucleus pulposus in vitro 109 . This innovative approach has shown promise in minimally invasive repair of degenerative intervertebral disks. Furthermore, PRP, when utilized as a hydrogel, could mimic the 3D microenvironment of cells 59 .
Injectable hydrogels can also serve as carriers for drug delivery in disease treatment and as vehicles for regenerative or tissue engineering purposes 110 . These hydrogels have the capacity to protect growth factors from degradation, overcome the biomechanical properties of PRP and MSCs-derived exosomes, and enable sustained release of growth factors and exosomes. Natural biomaterials, such as silk fibroin (SF), have been explored for designing hydrogels loaded with MSCs, showcasing their potential for applications in cartilage reconstruction and wound healing111–113.
It is crucial to acknowledge that the synergistic effects of PRP with stem cells may not consistently manifest across all applications. For example, in the context of promoting bone regeneration, PRP used as a cell carrier for MSCs has demonstrated favorable outcomes, resulting in higher cell proliferation rates, accelerated bone growth, and enhanced interface strength114,115. However, there have been instances, such as with decellularized human placental (DHP), where the combination of BMSCs and PRP did not significantly enhance joint cartilage regeneration in certain scaffold models 116 . These observations underscore the complexity of interactions between PRP and stem cells in different tissue microenvironments, highlighting the need for comprehensive understanding and context-specific evaluation when considering the application of PRP in conjunction with stem cell therapies.
Moreover, they emphasize the importance of continued research to elucidate the underlying mechanisms and optimize the utilization of PRP in combination with stem cells for diverse regenerative applications. Derivatives of PCs have also been explored for their potential to be incorporated into hydrogels. Autologous platelet-rich gel (APG), derived from PRP, has been investigated as a carrier system for cell delivery and as a release system for endogenous growth factors. In addition, second-generation PCs, known as PRF, has shown promise as an efficient matrix material for encapsulating stem cells and as a biodegradable scaffold material.
Bioengineering strategies have been employed to mitigate the potential adverse effects of PRP on MSCs. For instance, researchers have proposed methods to address toxic metabolites present in PRP, aiming to enhance the adhesion and proliferation of MSCs and endothelial cells. These innovative approaches hold potential for expanding the utilization of autologous PCs and offering improved outcomes for patients. Furthermore, in addition to the intrinsic properties of the novel biomaterials, alterations in the microenvironment, such as PH, may also influence the ultimate efficacy of these bioengineered systems 117 .
In summary, the combination of PCs and MSCs using advanced delivery systems presents exciting opportunities for diverse applications, ranging from tissue engineering to regenerative medicine. While there are promising outcomes, further research is warranted to elucidate the optimal conditions and potential limitations of these innovative approaches.
Discussion
The concept of this combination therapy assumes that the two work synergistically. PCs, when subjected to preconditioning or utilized in conjunction with MSCs, have exhibited promising outcomes in laboratory settings and clinical applications, particularly within the realm of regenerative medicine.
When considering the type of PCs, it is essential to define specific centrifugation parameters and timing to ensure optimal platelet enrichment. For PRF, optimized centrifugation parameters can produce a more stable three-dimensional fibrin matrix. Similarly, for PRP, determining the optimal centrifugation parameters and blood separation process is critical to ensure the desired platelet enrichment. Detailed descriptions of the extraction method for C-PRF are necessary, including the equipment used, centrifugation parameters, centrifugation time, and temperature control. Moreover, quality control criteria for C-PRF should be provided to ensure consistent platelet enrichment and growth factor release performance for each preparation. In addition to the preparation of PCs, standardized treatment protocols and optimal clinical application strategies need to be considered to optimize clinical guidelines. This may include specific indications, dosages, and frequencies for different clinical scenarios, as well as methods for evaluating and monitoring treatment outcomes. Establishing these guidelines will help ensure consistent and repeatable clinical use of PCs, maximizing their potential in tissue repair and regenerative therapies.
When considering the source of MSCs, it is crucial to consider the impact of extraction methods on therapeutic efficacy. The extraction process can potentially lead to cell damage, reduced survival rates, or varying levels of impairment, all of which may impact the quality and function of MSCs. Therefore, it is essential to assess the influence of different extraction methods on the characteristics and therapeutic potential of MSCs to select the most suitable source and extraction method for optimizing therapeutic outcomes. Among all stem cells, AD-MSCs are indeed noteworthy due to their abundance, non-invasive isolation, and lack of ethical restrictions 118 . AD-MSCs are considered as promising candidates for cell therapy due to their extensive immunomodulatory properties and paracrine activity 119 . After transplantation, AD-MSCs have demonstrated excellent potential for in vitro expansion and differentiation into various cell types 120 . Furthermore, the selection of the most appropriate MSCs source needs to be evaluated and chosen on a case-by-case basis, depending on specific clinical applications and research objectives121–123.
In general, each MSCs source has its own unique advantages and limitations, so the selection of the appropriate MSCs source needs to be combined with the specific clinical needs and research purposes.
However, in many studies, numerous variables are often not comprehensively considered, which may affect the interpretation and application of experimental results. Thus, several key considerations need special attention when optimizing clinical guidelines and treatment protocols. Clear definitions of diverse sources of PCs and MSCs, as well as methods of delivery into the body, are essential. Furthermore, researchers need to gain a deeper understanding of how PCs and MSCs behave in different settings to effectively leverage their properties for optimizing treatment outcomes124,125. In experimental design, consideration should be given to whether multiple injections can achieve better results than a single injection. If multiple injections are more effective, further consideration can be given to whether to administer them simultaneously or separately. In addition, the existence of dose dependence should be investigated by establishing different dose groups to determine the most appropriate dose range126,127.
Through the pretreatment of MSCs, PCs can directly influence the properties of MSCs by modulating signaling pathways or indirectly impact the functionality of MSCs by altering their proteome. With the continuous exploration of exosomes, the exosomes of MSCs may be able to replace the direct delivery of MSCs, thereby reducing some of the known side effects128,129. Therefore, we should pay attention to the growth factors and cytokines of PCs interacting with the bioactive components of MSCs exosomes.
Several ongoing randomized controlled trial (RCT) studies have focused on the individual therapeutic potential of PCs and MSCs for various diseases. For instance, a notable RCT demonstrated that BMSCs or leukocyte-poor PRP (Lp-PRP) treatment combined with rehabilitation for chronic patellar tendinopathy effectively reduced pain and improved activity levels in active participants, with those receiving BMSCs showing greater improvement in tendon structure 130 . Therefore, there is merit in incorporating pretreatment and combined applications into experimental designs, potentially integrating novel biological materials, to ascertain the optimal scheme for optimizing MSCs transplantation.
While the utilization of biological materials for the delivery of PCs and stem cells into the human body can enhance the efficacy of combination therapy, it is imperative to consider the potential toxicological effects of new materials 131 . Furthermore, the specific interactions of PCs and stem cells with novel materials may not universally yield favorable outcomes, necessitating careful consideration in the development of new stem cell-based therapies.
In summary, the combination of PRP with MSCs represents a promising approach for disease treatment, which can potentially be further augmented by the integration of novel materials to facilitate the transplantation of MSCs into the human body.
Footnotes
Acknowledgements
Not applicable.
Author Contributions
Conceptualization, X.L. and X.Y.; figure preparation, X.L.; table preparation, X.L., H.X. and Y.P.; writing—original draft preparation, X.L. and H.X.; writing—review and editing, X.X., H.X., Z.W. and X.T. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Ethical Approval
Not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by National Natural Science Foundation of China (grant number: 82360879).
