Abstract
Recent years have witnessed rapid advancements in 3D bioprinting and the widespread application of mesenchymal stem cells (MSCs) across various medical disciplines. The synergistic integration of 3D bioprinting and MSCs has opened innovative avenues for tissue engineering and regenerative medicine, particularly in bone tissue repair and regeneration. However, the progress of 3D bioprinting in the field of MSCs research still requires further exploration, and there remains a scarcity of related bibliometric analyses in this domain. With the aim of addressing this existing gap, this research systematically searched the Web of Science Core Collection for publications spanning from January 2003 to October 2025. It employed CiteSpace for cluster and evolution analysis, VOSviewer for collaboration network and keyword co-occurrence analysis, and the R package “bibliometrix” for statistical evaluation of bibliometric indicators. This bibliometric analysis focused on tissue engineering research integrating 3D bioprinting with MSCs, encompassing 1,846 original articles. These articles were authored by 10,276 researchers from 2,024 institutions across 69 countries and published in 342 academic journals. From 2014 to 2023, the number of annual publications exhibited a fluctuating yet rapid upward trend. China and the United States emerged as the most influential countries, with China experiencing a particularly substantial increase in research output—though international collaborations among institutions and authors remained limited. Wu C.T. and Bose S. stood out as key contributors to this field, while journals such as Biomaterials and Biofabrication have significantly advanced the discipline. High-frequency keywords including “3D printing” and “tissue engineering” reflected the core research directions, whereas emerging terms such as “MSC-EVs” and “nanocomposites” indicated current frontiers; in addition, “bioink,” “3D scaffold,” “osteogenesis,” and “angiogenesis” represented areas gaining growing research attention. Overall, this bibliometric study provides a thorough overview of the research tendencies and developments related to 3D bioprinting in the MSC field.

Introduction
Mesenchymal stem cells (MSCs) are multipotent stromal cells that can differentiate into various lineages, including osteoblasts, chondrocytes, and adipocytes 1 . These cells are typically isolated from tissues such as bone marrow, adipose tissue, umbilical cord blood, etc. Among these, bone marrow-derived mesenchymal stem cells (BMSCs), due to their early discovery, extensive research, and broad clinical applications, remain one of the primary sources for MSC studies and therapies. However, in terms of cell concentration, sources such as adipose tissue may yield higher cell quantities2,3. Mesenchymal stem cells are characterized by their expression of surface markers like CD105, CD73, and CD90, and by the absence of markers such as CD45, CD79α, and HLA-DR 4 . Their immunosuppressive properties make them valuable for treating acute graft-versus-host disease (aGVHD) and autoimmune disorders, as they modulate immune responses by interacting with immune cells and secreting anti-inflammatory factors. For example, Zhao et al. 5 showed that combining MSCs with basiliximab and calcineurin inhibitors can enhance the therapeutic effect on steroid-refractory aGVHD. This combination reduces the toxicity of second-line drugs and the incidence of chronic graft-versus-host disease (GVHD), does not increase relapse rates, and shows good tolerability. Research into MSC-based therapies spans diverse regenerative applications, including cartilage repair 6 , bone healing 7 , liver regeneration 8 , and myocardial infarction treatment 9 . To enhance therapeutic efficacy, genetic modification of MSCs is also being explored. In the TREAT-ME1 trial, autologous MSCs were transduced with a gamma-retroviral self-inactivating vector carrying a therapeutic promoter-gene construct for advanced gastrointestinal tumors 10 . IL-37-modified MSCs (IL-37-MSCs) substantially increased protection against intestinal ischemia-reperfusion injury via the NLRP3 signaling pathway 11 . Extracellular vesicles derived from MSCs (MSC-EVs), such as exosomes and microvesicles, represent a cell-free approach that retains MSC benefits. These exosomes have been shown to restore IL-1β-impaired sulfated glycosaminoglycan synthesis and suppress IL-1β-induced nitric oxide and MMP13 production. Inhibitors targeting adenosine receptor activation, AKT phosphorylation, ERK phosphorylation, and AMPK phosphorylation partially reverse these effects, which in turn supports the repair of temporomandibular joint osteoarthritis (TMJ-OA) 12 . Although stem cell therapy offers considerable therapeutic promise, its in vivo application faces challenges such as unpredictable cell migration, low post-transplant survival, and poor engraftment efficiency 13 . Biocompatible scaffolds used as stem cell carriers can improve cell distribution and survival at injury sites, promoting tissue repair 14 . Integrating 3D printing with such scaffolds offers a powerful approach in tissue engineering, enabling the fabrication of precise, personalized, and biocompatible constructs 15 .
3D printing, or additive manufacturing (AM), occupies a vital role in tissue engineering 16 . Techniques such as fused deposition modeling (FDM), stereolithography (SLA), selective laser sintering (SLS), digital light processing (DLP), and binder jetting are commonly employed 17 . These methods enable the fabrication of customized medical devices. A prominent medical application lies in producing patient-specific implants and prosthetics, which are widely adopted in orthopedics and sports medicine 18 . Recent advances also permit the simultaneous deposition of multiple materials within a single print, facilitating the construction of complex structures with graded mechanical and aesthetic properties 19 . Such structures can effectively mimic the mechanical structure and stiffness of the native extracellular matrix (ECM) 20 . For instance, Qiao et al. 21 designed a tri-layered stratified scaffold for osteochondral regeneration that incorporated MSCs within a gelatin methacrylamide (GelMA) hydrogel and delivered growth factors in a zone-specific manner to promote cell migration and differentiation. Building on these capabilities, 4D printing has emerged as a progression from 3D printing by introducing a temporal dimension 22 . 4D-printed objects can alter their shape or properties over time in response to environmental stimuli such as light, heat, or humidity 23 . Concurrently, bioprinting utilizes 3D printing to deposit living cells and biological materials, creating tissue constructs for regenerative medicine 24 . The integration of nanotechnology with 3D printing further expands these possibilities. This convergence enhances material properties at the nanoscale, improving strength, conductivity, and other characteristics 25 . Such electrical conductivity is known to support tissue healing and recovery 26 . In summary, 3D printing technology—particularly 3D bioprinting—offers considerable potential for medicine. By employing cells and biomaterials as bioinks, 3D bioprinting enables precise spatial control over cell placement and microenvironmental factors according to pre-designed models 27 . This capability allows directed organization of multicellular architectures and controlled deposition of varying cell densities. Integrating MSCs with 3D bioprinting facilitates the engineering of complex tissue constructs with regulated microenvironments, showing particular promise for repairing and regenerating organs such as the heart 28 and bone 29 , and advancing the future of tissue repair and organ transplantation.
Bibliometrics provides a powerful approach for analyzing scholarly communication and mapping the research landscape, offering both quantitative and qualitative insights into specific fields30,31. This methodology has been widely applied in medical research to investigate diverse topics such as uterine transplantation (UTx) 32 , nanomaterials in gastric cancer 33 , advances in total mesorectal excision (TME) 34 , 3D bioprinting for cardiovascular diseases 35 , and MSC research in liver disease 36 . Through bibliometric analysis, researchers can identify evolving trends, pivotal issues, and dynamic shifts within a discipline. Examining publications, countries, institutions, journals, authors, and their collaborative networks enables a comprehensive overview of a field’s current state. Author keywords and their co-occurrence patterns further help delineate the field’s foundational elements, emerging frontiers, and research highlights. To our knowledge, while bibliometric studies have examined 3D printing in cardiothoracic diseases 37 and cartilage regeneration 38 , no analysis has specifically addressed its application in MSC research. Our study therefore employs bibliometric methods to elucidate the current landscape and future trajectories of 3D bioprinting in the MSC field. We seek to identify leading contributors, institutions, countries, and current research hotspots. This work aims to support future tissue engineering efforts by presenting an innovative framework for advancing precise and personalized tissue construction.
Materials and methods
Data sources and search strategy
The Science Citation Index Expanded (SCIE) within the Web of Science Core Collection (WOSCC) serves as a prominent global database for bibliometric studies 39 . To mitigate potential biases from database updates and inaccuracies, two authors (YCL and BS) independently retrieved data from WOSCC on 10 October 2025. The search strategy employed was TS= (“3D bioprinting” OR “3D printing” OR “3D printed” OR “3D *print*” OR “three dimensional bioprinting” OR “three dimensional printing” OR “three dimensional printed” OR “three dimensional *print*” OR “rapid prototyping” OR “stereolithography” OR “additive manufacturing”) AND TS = ([ stem cells”] OR (“mesenchymal stem cell”)). All data were obtained online and did not require ethical certification.
Data screening
Two authors (YCL and BS) independently assessed and selected all publications, resolving any disagreements through consensus with a third reviewer (HW). The initial screening step limited the selection to English publications in the SCIE database of WOSCC (excluding non-English publications, n = 6; excluding non-SCIE databases, n = 152). The search covered the period from 1900 to 10 October 2025, with retrieved publications dating from January 2003 to October 2025 (no relevant studies were found for 1900–2002). The second step focused on content type: only original articles were retained, leading to the exclusion of 540 documents comprising review articles (496), early access (29), book chapters (6), proceeding papers (7), data papers (1), and retracted publications (1). In addition, the two reviewers performed independent screening of titles and abstracts, with full-text review carried out as needed to thoroughly identify literature related to MSC-based 3D bioprinting. Consequently, 7 articles that did not employ 3D bioprinting technology and 6 articles in which the printed cells or materials did not involve MSCs were excluded. Non-empty core fields were used as a basic criterion for data cleaning. Records were retained only if the Author (AU) field was non-empty, at least one Keyword (DE) was included, and the Publication Year (PY) field contained a value. No records missing these fields were identified, and no duplicates were found. Therefore, we excluded 711 publications that did not meet the requirements, and the final data included 1,846 relevant original articles. The detailed data filtration process is illustrated in Fig. 1.
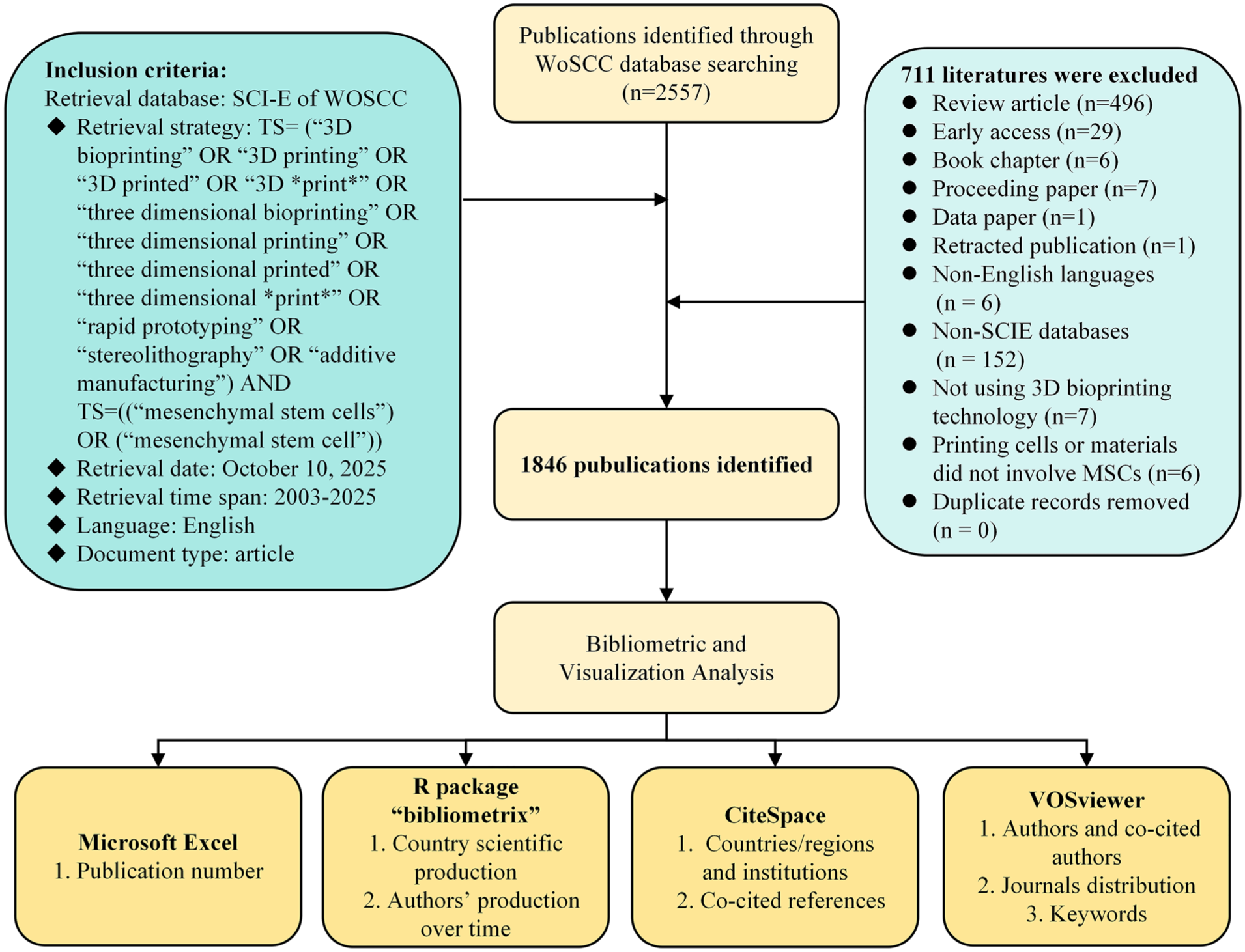
Flowchart of the search strategy and included studies.
Data analysis and visualization
Bibliometric analysis and visualization were performed using CiteSpace (version 6.4.R1), VOSviewer (version 1.6.20), and the R package “bibliometrix (version 4.3.0).” CiteSpace, developed by Professor Chen C.M., enables the detection and visualization of emerging trends and transient patterns in scientific literature 40 . The analysis employed the following CiteSpace parameters: a time span from January 2003 to October 2025 with one-year slices. The term sources and links were configured with the default parameters. We used CiteSpace to conduct an in-depth analysis of collaboration networks across countries and institutions, examine co-citation patterns among references, and generate a dual map overlay of journals. Furthermore, the software’s timeline view and burst detection features were applied to generate keyword time graphs and to identify bursts in keywords and references.
VOSviewer, a software tool developed by the Center for Science and Technology Studies (CWTS) at Leiden University 41 , constructs and visualizes bibliometric networks such as collaboration, co-citation, and co-occurrence networks 42 . Our study employed VOSviewer to analyze authors, co-cited authors, journals, and keyword co-occurrence, with minimum occurrence thresholds set at 10, 70, 5, and 30, respectively. Both author keywords and Keywords Plus were included in the keyword co-occurrence analysis.
The R package “bibliometrix” facilitates comprehensive bibliometric analysis, enabling quantitative scientometric research through the import of bibliographic data, execution of analytical procedures, and construction of networks for co-citation, coupling, collaboration, and co-occurrence analysis43,44. Using its web-based interface “biblioshiny,” 45 we calculated frequency statistics for authors, journals, institutions, and countries, incorporating both publication and citation counts. We also examined collaborative network relationships among authors, institutions, and countries.
CiteSpace represents a foundational bibliometric tool that effectively tracks the temporal evolution of research topics and visually represents associative relationships among clusters. Its burst detection feature precisely identifies emerging research fronts, a capability not intuitively available in VOSviewer or the R package “bibliometrix,” and one that aligns closely with this study’s objective of exploring field development trends46,47. VOSviewer offers superior network visualization, allowing node size and color to be customized according to publication count and cluster affiliation, while its density analysis function quantifies collaboration intensity. Keyword co-occurrence analysis further constitutes a distinctive strength of this tool 48 . The R package “bibliometrix” supports customizable statistical modeling and delivers accurate quantification of diverse bibliometric indicators 49 . Though these tools can interpret data differently, we maintained dataset consistency for cross-tool analysis by having all three read a unified plain-text file. Furthermore, the data readings from different tools were cross-validated to minimize potential significant deviations arising from any single tool’s data interpretation.
Results
Quantitative analysis: annual publication output
Fig. 2 shows the annual number of publications related to the application of 3D bioprinting in the field of MSCs from 2003 to 2025. Between 2003 and 2012 (a period of slow development), annual publications remained below 10, with only 11 articles published by 2013. This was followed by a phase of rapid fluctuating growth: annual output surpassed 200 in 2022 (225 articles) and peaked at 241 in 2023, reflecting an overall fluctuating upward trajectory. These results confirm a fluctuating yet rapid increase in annual publications from 2014 to 2023. Publications on the application of 3D bioprinting in the MSC field declined in 2024, likely reflecting the combined effects of natural research cycle fluctuations, ongoing technological iterations, delays between publication and indexing, and shifting research priorities. As emerging technologies such as 4D printing, cell-free scaffolds, and nanocomposites mature and clinical translation progresses, the number of subsequent publications is expected to rise rapidly again. We therefore anticipate a sustained increase in publication output and growing research interest in 3D bioprinting for MSC applications.

Annual publication output related to the application of 3D bioprinting in MSCs.
Distribution and collaboration of countries and institutions
In our study, we examined 2,024 institutions from 69 countries or regions, with key publication details summarized in Table 1. Fig. 3a (generated using the R package bibliometrix) shows the country-wise publication distribution, with the top five most productive countries being China (3,245 publications), the United States (1,092), South Korea (449), Germany (329), and Italy (278). Fig. 3b details the number of publications by corresponding authors across countries, distinguishing between single-country publications (SCP) and multicountry publications (MCP). Fig. 3c, d illustrate the international collaboration analysis conducted with CiteSpace, while Table 2 lists the top 10 countries and institutions by publication volume. In the diagrams, each circle represents a nation or institution, with its size proportional to the volume of publications from that entity. China led with 856 publications, followed by the United States (356), Germany (117), South Korea (113), and England (92). China also achieved the highest total citation count (30,084), closely trailed by the United States (23,377), while Germany ranked a distant third (4,889). Shanghai Jiao Tong University (127 publications, 6.87%) and the Chinese Academy of Sciences (111 publications, 6.01%) contributed the most publications, together accounting for 12.88% of all articles, followed by Peking University (58 publications, 3.14%), Sichuan University (50 publications, 2.71%), and Zhejiang University (50 publications, 2.71%). Surprisingly, Chinese institutions comprised all ten of the most productive institutions. Betweenness centrality quantifies the extent to which a node lies on the shortest paths between other nodes in a network, thereby reflecting its bridging role in the overall structure50,51. As shown in Fig. 3c, d, and Table 2, several countries and institutions exhibited high centrality values, including the United States (0.28), Germany (0.24), Italy (0.22), Shanghai Jiao Tong University (0.23), the Chinese Academy of Sciences (0.15), and Peking University (0.10), which are highlighted with purple circles. These results suggest that these countries and institutions have played influential roles in the field. Although the United States ranked second in publication output, it achieved the highest betweenness centrality (0.28), reflecting the substantial influence of its research. The connecting lines between nodes represent international or inter-institutional collaborations, with line thickness indicating the intensity of cooperation. In summary, active collaborative relationships were observed among countries and institutions such as the United States, China, Germany, Shanghai Jiao Tong University, and the Chinese Academy of Sciences.
The main information about data.


(a) Country scientific production related to the application of 3D bioprinting in MSCs. (b) Corresponding authors’ countries. (c) The visual map of national contributions and cooperation. (D) The visual map of institutional contributions and cooperation.
Publications in the 10 most productive countries and institutions related to the application of 3D bioprinting in the field of MSCs.

Visualization analysis of authors and co-cited authors
A total of 10,276 authors and 10,268 co-cited authors were identified from 1,846 publications related to the application of 3D bioprinting in the mesenchymal stem cell (MSC) field, with an average of 5.57 authors per article. Table 3 lists the top 10 productive authors and co-cited authors, highlighting their high publication output and citation impact. VOSviewer was used to visualize the author collaboration network (Fig. 4a) and author co-citation network (Fig. 4b). Among the most productive authors, Wu C.T. from the Chinese Academy of Sciences ranked first in publication output (34 publications, 2,484 citations), followed by Zhang L.G. (27 publications, 3,137 citations) and Wang J.W. (22 publications, 1,587 citations). Co-citation analysis, a standard bibliometric method, identifies authors who are cited together by other publications, revealing potential collaborative relationships, particularly in interdisciplinary fields. Among the top 10 co-cited authors, Bose S. (219 citations) and Murphy S.V. (214 citations) each accumulated over 200 citations, with Hutmacher D.W. closely trailing (181 citations). The thickness of the connecting lines reflects the strength of collaboration between nodes. For the author collaboration network, Zhang L.G. exhibited the strongest collaborative connections (total link strength = 256), followed by Wu C.T. (246) and Shie M.Y. (197). Among co-cited authors, Murphy S.V. (1,477) ranked first in total link strength, followed by Bose S. (972) and Hutmacher D.W. (969), indicating their close integration with other authors. The analysis further revealed a network of communication and collaboration between authors and co-cited authors in this research domain. Fig. 5a (generated via the R package “bibliometrix”) illustrates the relationship between publication volume and time for the top 20 productive authors. In this figure, bubble size corresponds to the annual number of publications, color intensity represents the annual total citations, and the red line traces the publication timeline; the number of publications by most authors shows a gradual increase over time. The graph reveals distinct chronological patterns of academic output among the authors. Fig. 5b presents a tripartite co-occurrence network of authors, countries, and institutions, visually depicting the relationships between authors (AU), their affiliated countries (AU_CO), and institutions (AU_UN). China emerged as the central hub, with dense connections to numerous authors and institutions. Shanghai Jiao Tong University and China Medical University (Taichung, Taiwan, China) exhibited strong linkages with multiple research groups, including Chinese contributors (eg, Wu C.T., Wang J.W., and Zhang Y.) and international researchers (eg, Gellinsky M. and Lee S.J.). This tripartite network clearly demonstrates China’s dominant position in author and institutional clusters within this field, while also reflecting the distribution of international participation. Collectively, these visualizations offer critical insights for analyzing collaborative networks and tracking emerging research trends in the integration of 3D bioprinting and MSCs.
Top 10 authors and co-cited authors related to the application of 3D bioprinting in the field of MSCs.


(a) The visual map of the authors’ collaboration network. (b) The visual map of the co-citation network of outstanding authors.

(a) Output of the top 20 most influential authors in the field of 3D bioprinting combined with mesenchymal stem cells (MSCs), 2010–2025. (b) Visualization of the countries/institutions affiliated with the top 20 most influential authors and their collaborative relationships.
Analysis of journals distribution and information flow
To map the publication landscape and citation dynamics of 3D bioprinting in the MSC field, we analyzed the distribution of literature across journals and disciplines. The most productive and influential journals are identified in Fig. 6a. Table 4 lists the top 10 journals and co-cited journals for 3D bioprinting applications in MSC research. These top 10 journals together published 553 articles, accounting for 29.96% of all publications. Among them, Biofabrication (2024 IF = 8.0, JCR Q1) had the highest publication output (100 articles, 5.42% of the total), followed by Advanced Healthcare Materials (69 articles, 3.74%) and ACS Applied Materials and Interfaces (63 articles, 3.41%). Journal co-citation analysis helps elucidate the academic network and the influence of core publications in the field. In the top 10 co-cited journals, two were cited more than 4,000 times: Biomaterials (Citations = 8,217, 2024 IF = 12.9) and Acta Biomaterialia (Citations = 4,457, 2024 IF = 9.6). Biofabrication (Citations = 2,175, 2024 IF = 8.0) followed. The high citation frequency of these journals reflects their considerable influence in the field.
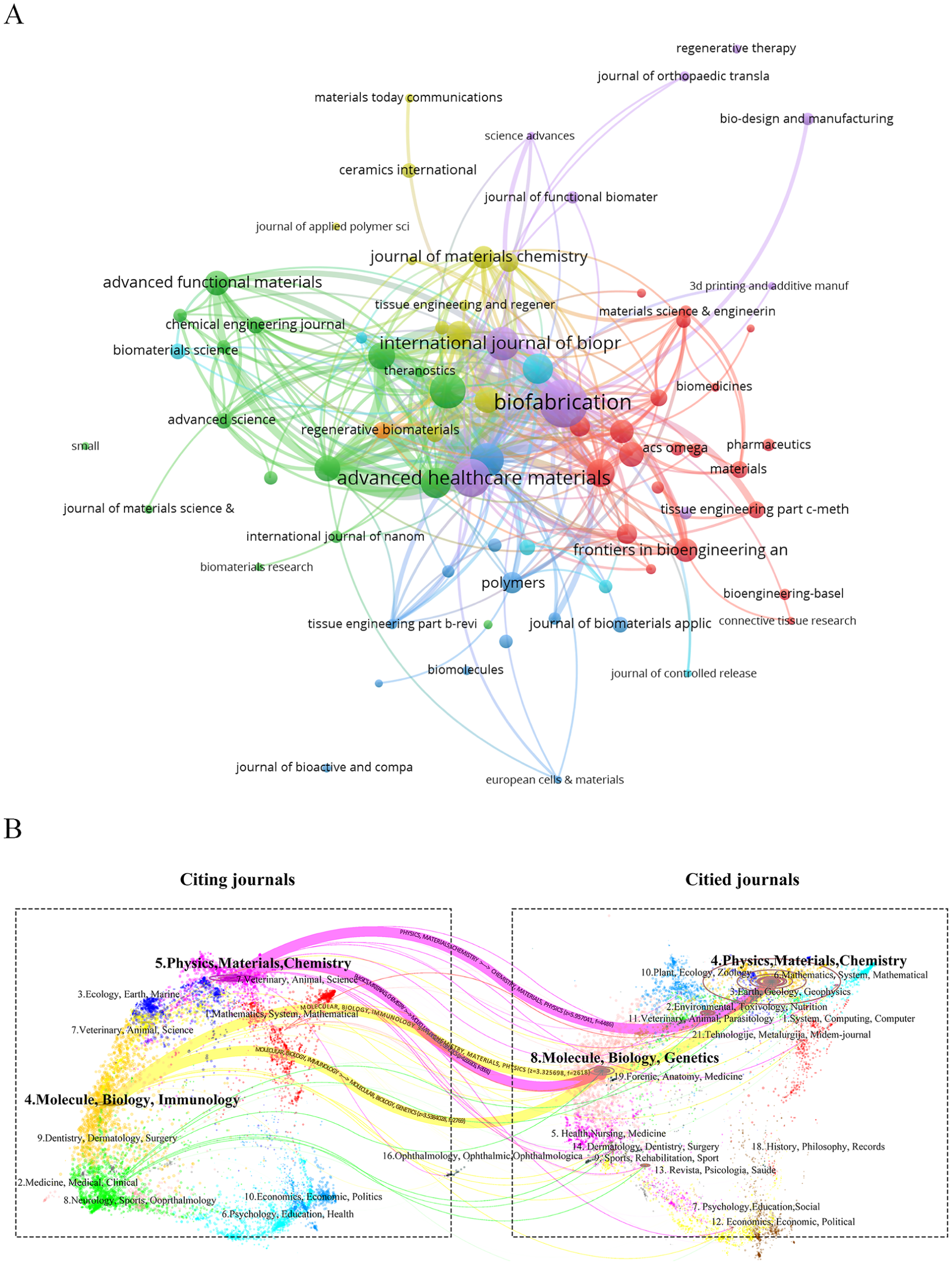
(a) The visual map of journals where papers are published related to the application of 3D bioprinting in MSCs. (b) The dual-map overlay of all journals.
The top 10 journals and co-cited journals related to the application of 3D bioprinting in the field of MSCs.

To analyze the distribution of citing and cited journals, a dual-map overlay was generated to reveal patterns within the scientific corpus against the backdrop of global scientific literature (Fig. 6b). This visualization presents a dual-map overlay of 3D bioprinting applications in MSC research from 2003 to 2025 (projected), identifying two primary citation paths. These paths indicate that papers published in journals specializing in Physics/Materials/Chemistry or Molecular/Biology/Genetics are frequently cited by journals in either Physics/Materials/Chemistry or Molecular/Biology/Immunology. This dual-map overlay enables a macro-level understanding of the distribution and evolutionary trajectories of scientific literature in the field, thereby uncovering key interdisciplinary connections.
Co-cited literature analysis and clustering network
Small and Marshakova first proposed co-citation analysis to evaluate relationships among scholarly articles 52 . This method was subsequently applied and refined during the development of CiteSpace, highlighting the connections between citing and cited articles 53 . Analyzing typical clusters therefore reveals the structure and dynamics of academic fields, particularly for 3D bioprinting in MSC research. We examined 1,846 citing articles and their references to identify homogeneous clusters of highly cited literature in this domain.
Table 5 lists the top 10 most co-cited references related to 3D bioprinting applications in MSC research. The most frequently co-cited reference was a 2014 Nature Biotechnology article by Murphy S.V. et al., titled “3D bioprinting of tissues and organs” (180 citations), followed by contributions from Karageorgiou V. et al. (2005; 165 citations), Kang H. et al. (2016; 109 citations), and others. All 10 of the top co-cited references were classified in the JCR Q1 category, reflecting their high academic standing. Among these, the 2006 Cell paper “Matrix elasticity directs stem cell lineage specification” had the highest impact factor (2024 IF = 42.5). CiteSpace visualized the co-citation network of 69,079 references (Fig. 7a). Each circle represents a reference, with size proportional to its citation frequency. Links between nodes indicate that the corresponding references were co-cited by one of the 1,846 citing articles analyzed, and line thickness corresponds directly to the co-citation frequency. The co-cited references were clustered into 10 major groups using indexing terms: 3D bioprinting (cluster #0), tissue engineering (cluster #1), polylactic acid (cluster #2), 3D printing (cluster #3), cartilage regeneration (cluster #4), cartilage tissue engineering (cluster #5), calcium silicate (cluster #6), bone tissue engineering (cluster #7), osteochondral defect repair (cluster #8), and gradient structure (cluster #9). These cluster labels represent the principal research themes or concepts within each group (Fig. 7b).
Top 10 co-cited references related to the application of 3D bioprinting in the field of MSCs.


(a) The visual map of co-cited references related to the application of 3D bioprinting in MSCs. (b) The clustering network map of relevant co-cited references. (c) The top 25 references with the strongest citation bursts related to the application of 3D bioprinting in MSCs.
A citation burst refers to a sharp, substantial rise in the number of citations a publication receives over a specific period 54 . This pattern of heightened citation frequency highlights the influential nature of these references and their pivotal role in advancing scholarly discourse within the field, typically indicating that the publications have exerted considerable impact on the academic community or sparked new research trends. Using CiteSpace, we identified 25 bursty references with a minimum burst duration of two years (Fig. 7c). This figure presents the top 25 references with the strongest citation bursts, along with their respective burst durations.
Keyword and hotspots analysis
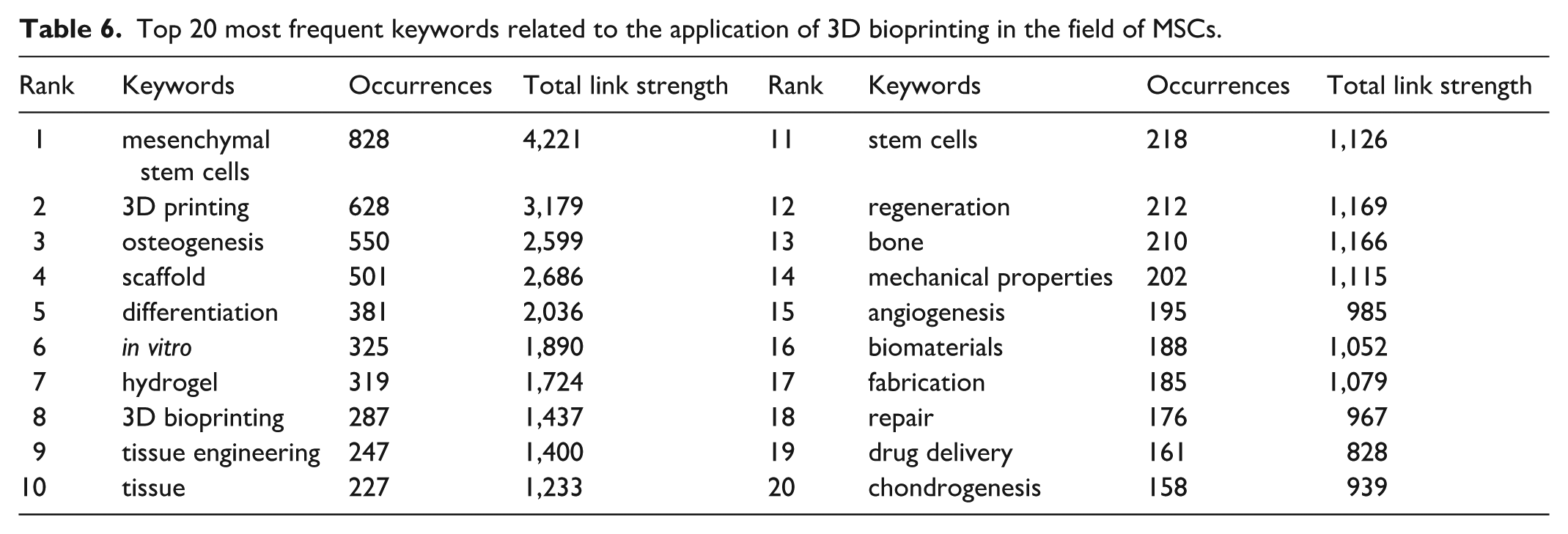
Keyword co-occurrence analysis constitutes a core analytical module in VOSviewer. We employed a combined “network graph and density map” visualization to delineate current research hotspots and emerging trends more clearly (Fig. 8a, b). Prior to generating these visualizations, a thesaurus file (Supplementary File) was applied to all co-occurrence analyses. This file unified different keyword variants: for example, “scaffolds,” “Scaffold,” and “3d scaffold” were standardized to “scaffold,” and “hydrogels” and “Hydrogel” to “hydrogel.” This process reduced terminological dispersion and enhanced the coherence of resulting clusters. Fig. 8a, b present a keyword co-occurrence analysis of all keywords, identifying 6,034 keywords in total, with 246 appearing more than 10 times and 115 occurring more than 20 times. Table 6 lists the 20 most frequent keywords associated with the application of 3D bioprinting in the MSC field. “Mesenchymal stem cells” ranked first (828 occurrences; total link strength = 4,221), confirming its central role in the field, followed by “3D printing” (total link strength = 3,179), “osteogenesis” (total link strength = 2,599), “scaffold” (total link strength = 2,686), and “differentiation” (total link strength = 2,036). Node colors represent the average publication year of keywords, ranging from blue (2020) to yellow (2021.5), which illustrates the temporal evolution of research hotspots. For instance, more recent studies tend to emphasize “3D bioprinting,” “angiogenesis,” and “osteogenesis,” whereas earlier research frequently focused on “scaffold” and “tissue.” Fig. 9 presents a keyword timeline visualization generated using CiteSpace, displaying keyword frequency along a timeline from 2005 to 2025. Labels on the right (eg, #0 3D printing and #1 tissue engineering) denote distinct clusters corresponding to major research areas, as indicated by their larger node size and earlier coloration (red). The figure further demonstrates the progression of technologies such as nanomechanical characterization and MSC-EVs, which have seen increasing application in tissue engineering.

(a) The visual map of keyword co-occurrence related to the application of 3D bioprinting in MSCs. (b) The heatmap of keywords.
Top 20 most frequent keywords related to the application of 3D bioprinting in the field of MSCs.
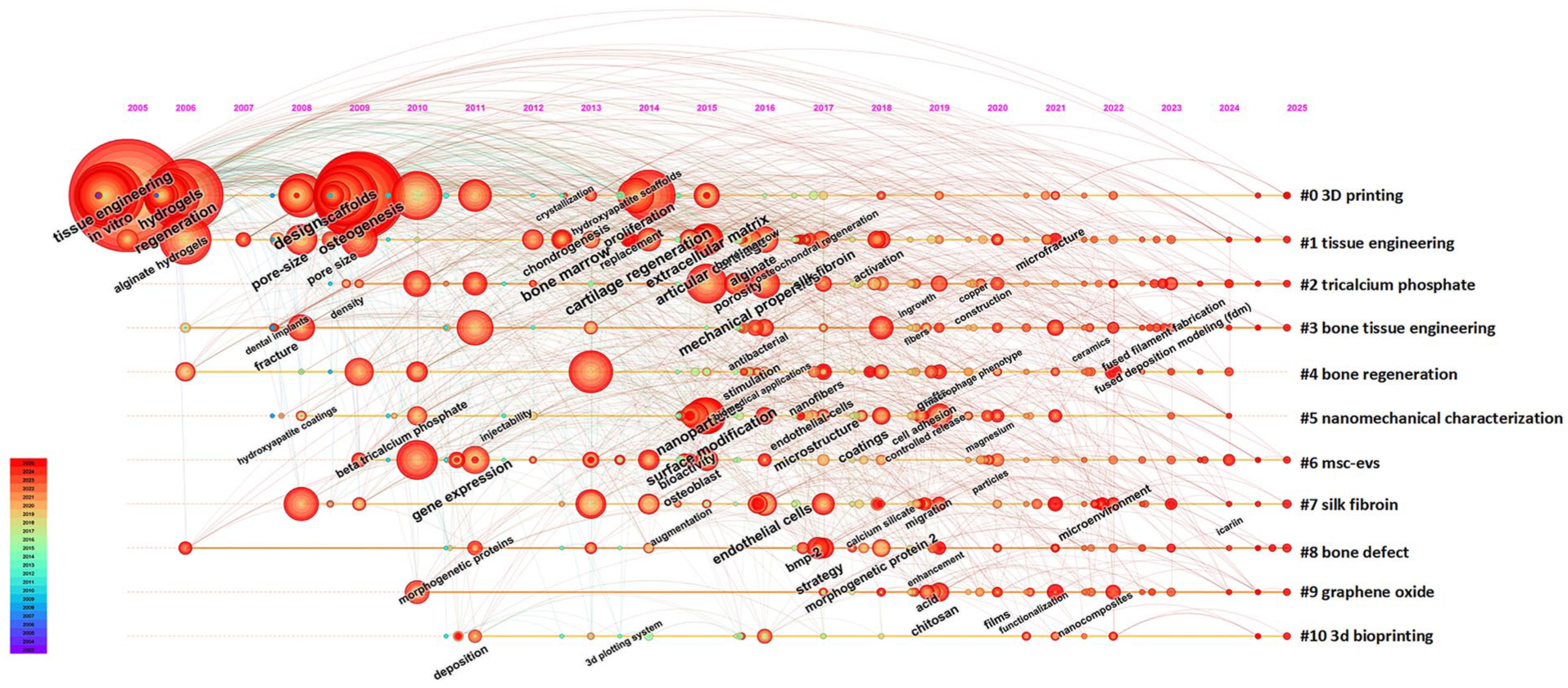
The keyword timeline map related to the application of 3D bioprinting in MSCs.
Discussion
To delineate the knowledge structure and dynamic evolution of 3D bioprinting in the context of mesenchymal stem cells (MSCs), we conducted a comprehensive bibliometric and visual analysis of their integration from 2003 to 2025. As of 10 October 2025, 10,276 authors from 2,024 institutions across 69 countries had published 1,846 papers on 3D bioprinting in the MSC field, spanning 342 distinct journals. These data indicate substantial research output and extensive collaborative research in this field. To characterize publication trends, we analyzed contributions across multiple dimensions, including countries, institutions, authors, journals, references, and keywords. Co-citation analysis and keyword co-occurrence mapping further identify the foundational knowledge, research hotspots, and thematic evolution in this field. These analyses delineate the main development pathways of key research topics. On this basis, we have pinpointed the current research frontiers of MSC-based 3D bioprinting. This work addresses critical gaps in the literature on MSC-3D bioprinting integration and provides novel perspectives on the progress of this interdisciplinary field, laying a data-driven foundation for future research.
Prior to 2003, research on 3D bioprinting in MSCs was scarce, indicating the field had not yet been established. The seminal paper in this area was published in 2003 by Schantz J.T. and colleagues, entitled “Repair of calvarial defects with customized tissue-engineered bone grafts II. Evaluation of cellular efficiency and efficacy in vivo” 55 . Their work utilized rapid prototyping to fabricate customized polycaprolactone (PCL) scaffolds with precisely controlled microstructures, which were seeded with bone marrow-derived mesenchymal progenitor cells (MPCs) to treat calvarial defects. These scaffold-cell constructs, featuring defined external geometry and microstructure, successfully delivered osteogenic cells and enhanced bone formation in critical-sized defects in vivo, thereby creating new possibilities for the functional and aesthetic reconstruction of complex craniofacial defects. Following this pioneering 2003 study, the field began to gain increased attention. Between 2003 and 2013, only a limited number of studies emerged, reflecting an early developmental phase. A pivotal milestone was reached in 2014 with the article “3D bioprinting of tissues and organs” by Murphy S.V. et al. This article examines the fabrication of complex, functional living tissues through 3D bioprinting, a process that employs biocompatible materials, cells, and supporting components. It addresses the pressing demand for transplantable tissues and organs within regenerative medicine. The discussion highlights the inherent complexities of the technology, including the challenges of selecting appropriate materials, cell types, and growth factors. In addition, the text outlines its applications in generating tissues for transplantation, such as skin, bone, and cardiac structures. This technology also provides meaningful guidance for the development of advanced tissue models used in research, drug discovery, and toxicology studies. Following the article “Vascularized 3D printed scaffolds for promoting bone regeneration” published by Yan et al. 56 in 2018—which was the most cited document locally with 39 citations—the authors prepared a 3D-printed degradable scaffold for the controlled release of deferoxamine (a compound that promotes angiogenesis and osteogenesis), inspired by the clinical need for bone tissue regeneration and the process of bone development. This scaffold exhibited good biocompatibility (supporting MSC growth and differentiation) and mechanical properties comparable to natural cancellous bone, representing a significant advance for orthopedic tissue engineering with potential clinical translation for segmental bone defect treatment. A critical technical component in this field is bioink—a hydrogel-based solution that typically encapsulates one or more biomaterials and the desired cell types—used for fabricating tissue constructs 57 . These advancements are expected to drive rapid expansion in MSC-based 3D bioprinting applications, with an increasing number of clinical trials likely employing 3D bioprinting to fabricate MSC-laden biological scaffolds for regenerating and repairing cartilage, bone, muscle, blood vessels, hair follicles, and other tissues58–60.
The spatial distribution of countries/regions and institutions, illustrated in the map and Fig. 3a–d, identifies China, the United States, and Germany as the most productive nations. China leads in publication volume, yet it exhibits a relatively low betweenness centrality of 0.07 (Table 2). The United States ranks second in publications but achieves the highest betweenness centrality (0.28). This pattern indicates that China has produced a substantial body of independent research, which explains its lower centrality. In contrast, the United States plays a pivotal bridging role in the global collaboration network, as evidenced by its high betweenness centrality. China and the United States dominate overall publication volume. Other nations exhibit lower output but maintain active international research partnerships. China produces the highest number of SCPs, substantially exceeding all other countries and reflecting its considerable independent research capacity. The United States also generates a high volume of SCPs, though slightly fewer than China. Furthermore, the United States shows a relatively large count of multiple-country publications (MCPs), underscoring its robust involvement in international collaborations. Overall, multicountry collaborative documents remain less common overall. This pattern suggests that despite growing international cooperation, SCPs continue to dominate academic literature. Notably, Singapore was one of the earliest nations to adopt 3D bioprinting for MSC research, with the first article in this field published in 2003 55 . All top 10 institutions are Chinese: Shanghai Jiao Tong University leads with the most publications (127) and highest betweenness centrality (0.23), followed by the Chinese Academy of Sciences (111 publications, 0.15 centrality).
We ranked authors based on their total number of publications in the MSC-related 3D bioprinting field, incorporating citation counts, H-index, and total link strength to identify the most productive and influential researchers (Table 3). Wu C.T. from the Chinese Academy of Sciences led with 34 publications, followed by Zhang L.G. from George Washington University (27 publications) and Wang J.W. from Shanghai Jiao Tong University (22 publications). Wu C.T. has accumulated 2,484 citations and an H-index of 90, while Zhang L.G. has 3,137 citations and an H-index of 66—both substantially higher than other top-ranked scholars. Furthermore, the total link strength of these two authors exceeds 200, indicating their work is not only prolific but also of high quality and impact. They hold considerable prestige in the academic community, with their findings widely recognized and cited. As shown in Fig. 4a, the author co-authorship network is divided into seven main clusters. Leading researchers such as Wu C.T., Zhang L.G., and Gelinsky M. have not formed interconnected networks, indicating limited academic interaction among them. Promoting collaboration between Chinese, American, and other international research groups could help overcome academic barriers and accelerate progress in MSC-based 3D bioprinting, thus advancing tissue engineering. Fig. 4b displays the author co-citation network generated via VOSviewer, which clusters into three groups. The top 10 co-cited authors each have over 100 co-citations, with Bose S. ranking first (219 co-citations), followed by Murphy S.V. (214) and Hutmacher D.W. (181). Notably, Murphy S.V. (1,477) and Bose S. (972) exhibit remarkably high total link strength values. Fig. 5a provides a temporal perspective on academic output and influence, tracking publications from 2010 to 2025. Overall, the number of publications by these authors has increased significantly since 2016, with the 2020–2024 period representing the peak of research output—reflecting a substantial surge in field activity. For instance, Fisher J.P. maintained a steady publication rate during this period and was the earliest among the top 20 authors to enter this research field (in 2010). Zhang L.G. produced notable output in 2016 (depicted as large bubbles in Fig. 5a) and subsequently maintained a stable publication record. In contrast, Kelly D.J. demonstrated a marked increase in research activity after 2020. This graph clearly delineates the temporal contributions and distribution of core researchers, aiding in identifying active authors and tracking the evolution of research focus over time. Fig. 5b illustrates collaborative ties between authors, institutions, and countries, clarifying the structure of academic partnerships. Wang J.W. from Shanghai Jiao Tong University is linked to multiple institutions, whereas Fisher J.P. from the University of Maryland appears more limited in institutional collaborations. We also observed substantial collaborative activity among Shanghai Jiao Tong University, the Chinese Academy of Sciences, George Washington University, and other organizations—all making significant contributions to the application and advancement of 3D bioprinting in MSC research.
Journal and co-citation analysis are crucial tools for evaluating academic research and identifying key journals in a research field 61 . Table 4 reveals that Biofabrication (100 publications, 2024 IF = 8.0), an English journal, has the highest publication output and ranks first in total citations (6,340 citations). As a citing journal, Biofabrication focuses on cutting-edge research on fabricating biological systems and therapeutic products using cells, proteins, and biomaterials as fundamental components. In contrast, the cited journal Biomaterials spans a broad spectrum of physical, biological, and chemical sciences that underpin biomaterials design and clinical application, encompassing polymer synthesis and characterization, drug and gene carrier design, host response biology, immunology, toxicology, and nanoscale self-assembly. Both journals are closely linked to the fields of biomaterials and cell and tissue engineering, a finding that aligns with the dual-map overlay presented in Fig. 6b. The dual-map overlay of journals illustrates the disciplinary distribution of academic publications and reveals points of convergence between research fields 62 . Fig. 6b illustrates the intersection between the left (citing journals) and right (cited journals) sections, depicting the interactions and citation relationships across different disciplinary fields in the application of 3D bioprinting in the MSC field. The first citation pathway is from Physics/Materials/Chemistry and Molecular/Biology/Genetics journals to Physics/Materials/Chemistry journals, and the second citation pathway is from Physics/Materials/Chemistry and Molecular/Biology/Genetics journals to Molecule/Biology/Immunology journals, showing a convergent pattern in development of disciplines. This implies that interdisciplinary integration can foster the emergence of novel scientific ideas and methods in the fields of Physics/Materials/Chemistry and Molecule/Biology/Immunology, such as the advancements of 3D bioprinting 63 and 4D bioprinting 64 in terms of physical materials and chemical properties. In addition, it can promote the further extraction of MSC-EVs as biomaterials, leveraging their smaller molecular size, lower immunogenicity, and stronger tissue repair properties, thus accelerating the rapid development of tissue engineering 65 . Among the top 10 journals and co-cited journals, 90 and 80% are JCR Q1 journals, respectively. This reflects their relatively high academic status and influence in the field of 3D bioprinting and mesenchymal stem cell research. Therefore, researchers should prioritize these journals to efficiently locate relevant articles and stay updated on cutting-edge advancements in 3D bioprinting for MSC applications.
Fig. 7 presents a literature clustering analysis chart illustrating 3D bioprinting applications in the MSC field, revealing research hotspots, core literature, and temporal evolution. Core literature from the early period (2013–2016)—such as works by Bose S. (2013), Murphy S.V. (2014), and Kang H.W. (2016)—is represented by large red nodes, indicating these are highly cited classic studies that laid the foundation of the field. Recent literature (2018–2021), including contributions from Derakhshamer S. (2018), Zhang L.G. (2019), and Schwab A. (2020), has also maintained high influence, reflecting research continuity and innovation (Fig. 7a). The articles by Murphy S.V. (2014), Hutmacher D.W. (2016), and Zhang L.G. (2019) are the three most co-cited, representing pivotal literature, research hotspots, and trends. All top 10 co-cited articles belong to JCR Q1 journals, underscoring their high academic standing (Table 5). Tissue engineering (TE) is an interdisciplinary field integrating biology, materials science, engineering, and medicine 66 , aiming to regenerate or repair damaged tissues and organs via in vitro cultivation or construction 67 . The concept was first formally introduced and published in the journal Science by American scientists Vacanti J.P. and Langer R. in 1993 68 . As shown in Fig. 7b, the earliest cluster—tissue engineering (cluster #1)—represents the field’s origin. The core principles of tissue engineering—cells, biomaterials, and biochemical factors—have remained consistent since the early experiments by Bell and colleagues in 1979 69 . In 1997, Cao Y. and colleagues successfully formed auricular cartilage with skin coverage in nude mice using tissue engineering 70 . This marked the potential of tissue engineering technology to form tissues and organs with complex three-dimensional spatial structures, highlighting the immense potential of tissue engineering. Therefore, 3D bioprinting (cluster #0) occupies an important position in tissue engineering. As the largest of the 10 clusters, it has undergone substantial development. Since 1997, researchers have engineered auricular-shaped cartilage tissue using various cells and scaffolds, first in vitro, then in immunodeficient nude mice, and later in immunocompetent animals71,72. Later clinical reports described a two-step scaffold-free approach, in which microtia chondrocytes were injected subcutaneously into the abdomen; the resulting cartilage was then manually carved into an ear-shaped framework and re-implanted in its final position 73 . A subsequent study reported the first clinical use of this auricular reconstruction technique to achieve in vitro regeneration of patient-specific ear-shaped cartilage 74 . Cartilage regeneration (cluster #4) is the main core issue and research foundation of early studies in this field. Calcium silicate (cluster #6) is fabricated into porous scaffolds or hydrogels to provide a 3D growth microenvironment for chondrocytes. The silicon ions (Si) and calcium ions (Ca) released during its degradation in the body can activate cell signaling pathways (eg, AMPK/ERK1/2 and Wnt/β-catenin), thereby promoting the proliferation and differentiation of chondrocytes. Clusters #1, #0, #4, and #6 collectively form the core early research foundation in this field, providing significant reference basis for the subsequent research frontiers such as 3D printing (cluster #2), to-bone interface regeneration (cluster #3), cartilage tissue engineering (cluster #5), bone tissue engineering (cluster #7), osteochondral defect repair (cluster #8), and gradient structure (cluster #9). Moreover, there are interdependent relationships between different clusters, and they influence and cite each other. Although significant progress has been made in osteochondral repair through tissue engineering, there are still major challenges, including identifying suitable cell sources, adverse reactions of the host to the graft, difficulties in manufacturing pre-designed three-dimensional structures, and insufficient mechanical properties to maintain structural integrity75,76. One approach selects MSCs or induced pluripotent stem cells (iPSCs), which can differentiate into any cell type, as seed cells to guide their differentiation into specific lineages for repairing or replacing damaged tissues 77 . For example, Zhu et al. 78 found that short-term low-temperature pretreatment can stimulate MSCs to activate the PI3K/AKT pathway, which has the advantages of promoting angiogenesis and maintaining cell viability. A second strategy employs 3D printing to fabricate personalized biological scaffolds with defined mechanical properties, addressing individual patient needs while supporting tissue growth and vascularization79,80. These advancements and frontiers in tissue engineering are also the core content of this study.
Since the invention of stereolithography by Charles W. Hull in 1986 81 , the technology has been subsequently applied to biomaterials, particularly in tissue engineering. 3D bioprinting (cluster #0) enables the precise layer-by-layer assembly of biological materials, biochemical factors, and living cells for fabricating 3D constructs 82 . The main methods of 3D bioprinting include inkjet-based printing, digital light processing (DLP), extrusion-based printing, and freeform reversible embedding (FRE) 83 . These techniques are typically selected based on printing mechanism, resolution, processing time, and material compatibility to meet diverse application scenarios and research requirements. The medical applications of 3D bioprinting in the MSC field rely on the integration of multiple disciplines—including materials science, bioengineering, cell biology, biophysics, and medicine—for continuous advancement. By integrating 3D printing (cluster #2) with photo-crosslinking curing technology, the properties of bioinks (eg, mechanical strength, degradation rate, and bioactive factor loading capacity) can be precisely tailored. This approach enables the fabrication of complex 3D biomimetic structures while maintaining MSC viability. It further supports the regulation of MSC proliferation and differentiation, facilitating the formation of heterogeneous or personalized tissue constructs such as bone-cartilage interfaces and vascularized tissues (cluster #3). Ultimately, these capabilities promote the clinical translation of tissue engineering and regenerative medicine.
In cartilage and bone tissue engineering (clusters #5 and #7), gradient structures (cluster #9) represent a key design concept that leverages technologies such as 3D printing to create continuous or stepwise spatial variations in scaffold composition, mechanical properties, pore architecture, and degradation rate. This approach aims to replicate the heterogeneity of natural tissues—such as that of the bone-cartilage interface (cluster #3)—and to guide cell behavior. Gradient structures address the challenges of interfacial regeneration, enabling precise repair of complex biological tissues. For example, DLP printing can be used to progressively direct BMSCs to accelerate osteochondral defect repair (cluster #8).
Researchers are developing methods to print personalized tissues and organs, enabling customized treatment strategies for patients. For example, liver tissue models (eg, functional 3D human liver buds) containing MSCs have been fabricated for research and drug screening 84 . The freeform reversible embedding in suspension hydrogel (FRESH) technique has been utilized for 3D bioprinting of collagen to reconstruct cardiac tissue 85 . This approach produces cardiac patches that promote the migration of functional myocardial cells to repair cardiac defects; these constructs can also form in vitro heart muscle models for studying fundamental cardiac physiology or serve as high-throughput drug screening platforms86,87. In one study, Zhang et al. found that artesunate targeted myeloid differentiation factor 2 (MD2) and inhibited the MD2/Toll-like receptor 4 (TLR4) signaling pathway, thereby alleviating fibrotic gene expression in cardiac fibroblasts within 3D-engineered heart tissues. Engineering a functional heart—that is, constructing a complete organ with performance equivalent to native tissue—represents the ultimate goal of cardiac engineering. In the field of nerve regeneration, umbilical cord mesenchymal stem cells (UC-MSCs) can be made into 3D bio-catheters to treat ulnar nerve defects, and BMSCs can be used to treat traumatic brain injuries, promoting the regeneration of blood vessels and nerves 88 . 3D tissue printing typically leverages bioinks like elastin and collagen to simulate the extracellular matrix (ECM). These bioinks can also incorporate various bioactive tissue engineering materials, including polyethylene glycol hydrogels, alginate 21 , gelatin methacryloyl (GelMA), and chitosan 89 . By mimicking ECM’s hardness, elasticity, pore size, and porosity, 3D bioprinting enables MSCs to adhere and migrate appropriately, thereby facilitating tissue repair 90 . In tissue engineering, signaling pathways such as Wnt/β-catenin 91 , TGF-β1 92 , and Notch1 93 regulate MSCs, enhancing their differentiation into cardiac-specific lineages including cardiomyocytes, endothelial cells, and smooth muscle cells. Mesenchymal stem cells also exhibit immunomodulatory properties by secreting anti-inflammatory cytokines like transforming growth factor-beta (TGF-β) and interleukin-10 (IL-10), which reduce inflammation and modulate immune responses 94 . In addition, they contribute to ECM remodeling and may support tissue integration and functional recovery via direct and indirect intercellular communication (eg, gap junctions and chemical signaling) 95 . The ECM plays a critical role in 3D bioprinting: its mechanical properties (eg, stiffness and elasticity) influence cellular mechanotransduction, thereby affecting cell proliferation, differentiation, and gene expression 96 . For example, altering culture substrate stiffness can modulate the secretion and uptake behaviors of MSC-EVs 97 . In bone tissue regeneration engineering (cluster #7), 3D-bioprinted scaffolds can be designed to induce MSC osteogenic differentiation via key transcription factors and signaling pathways, including Wnt, TGF-β, PI3K/AKT, MAPK, Hippo, and JAK-STAT 98 . Beyond vascularization, anti-inflammatory/immunomodulatory effects, and mechanical stimulation, specific genes and proteins can be directly printed onto scaffolds: for instance, fibroblast growth factor-2 (FGF-2) incorporated into biological scaffolds interacts with FGF receptors (FGFR) to activate downstream signaling that promotes angiogenesis, wound healing, and cell proliferation/differentiation 99 . These molecular cues further regulate MSC behavior and tissue regeneration.
Nanomaterials have been used to improve stem cell engraftment in cellular therapy 100 , enhance material mechanical stability in tissue repair 101 , increase electrical conductivity in nerve regeneration 102 , strengthen tissue adhesion 103 , and boost antibacterial capacity in wound dressings 104 . Incorporating nanocomposites enhances bioink printability and mechanical properties, including viscosity, shear-thinning behavior, structural integrity, biocompatibility, and cell–material interactions 105 . In addition, 3D-printed polylactic acid (PLA)-based nanocomposite scaffolds can be modified for bone tissue engineering by coating with ordered mesoporous strontium bioactive glass (SrBG) or a SrBG/nano-hydroxyapatite (nHA) combination. These coatings enhance scaffold hydrophilicity, elastic modulus, and hardness, thereby promoting cell growth and differentiation. Fig. 7c shows that Murphy S.V. (2014) exhibited the highest citation burst strength (29.24) between 2015 and 2019—indicating substantial academic impact during this period. This article is also the most frequently co-cited reference. These co-cited references primarily originate from fields such as biomaterials, tissue engineering, and biofabrication, reflecting sustained research activity and innovation in these domains. At least one publication per year showed strong citation bursts from 2012 to 2020. Among the 2020 publications, the articles by Koons G.L. (2020), Wang C. (2020), and Schwab A. (2020) have citation bursts continuing through 2024, indicating these studies may remain influential in the current and upcoming years.
Keyword co-occurrence analysis reveals research hotspots, thematic connections, and developmental trends within academic disciplines (Fig. 8). Fig. 8a includes core methods and materials in tissue engineering, such as “biofabrication,” “3D printing,” “fabrication,” “hydrogel,” “collagen,” “chitosan,” and “alginate.” It also features terms from interdisciplinary fields, including “angiogenesis,” “osseointegration,” and “surface modification,” indicating that tissue engineering extends beyond biomaterials and cells to integrate biomechanics and biochemistry. These interdisciplinary keywords underscore the field’s breadth and its capacity to address complex biological challenges. The evolution of research hotspots has introduced emerging terms such as “bioink,” “exosomes,” and “nanoparticles,” which are expected to define future investigative priorities. The combination of “silk fibroin” and “electrospun nanofibers” technology exemplifies the “material-structural-function” collaborative design paradigm in tissue engineering. By replicating the nanotopology and biochemical signals of the native extracellular matrix (ECM), this combination offers an effective strategy for multitissue regeneration. These materials can also act as bioink components in 3D bioprinting to simulate the natural ECM 106 . Such bioinks may encapsulate MSCs, growth factors, and other bioactive molecules to support cell viability and function 107 . Modifying the chemical composition and physical structure of electrospun fibers enables regulation of extracellular matrix signaling, thereby directing MSC differentiation into specific lineages 108 . Incorporating vascular endothelial cells and angiogenic factors within electrospun nanofibers or printed vascular conduits further promotes angiogenesis 109 . Integrating electrospinning with 3D bioprinting facilitates the development of novel bioreactors that provide dynamic culture environments for MSCs. These systems simulate in vivo conditions—including mechanical stress and fluid dynamics—to optimize cellular behavior and tissue maturation 110 . In addition, such bioreactors allow precise control over parameters such as pH, temperature, oxygen tension, and perfusion rates 111 . Patel et al. 112 reported that perfusion bioreactor culture markedly enhances MSC-EV production compared with conventional methods. Kronstadt et al. 113 demonstrated that MSC-EVs generated in a perfusion bioreactor system significantly accelerate wound healing in diabetic mouse models, underscoring the therapeutic potential of bioreactor-expanded MSCs. Extracellular vesicles derived from MSCs modulate inflammation and promote cartilage regeneration; for example, they enhance chondrocyte function via the miR-205-5p/PTEN/AKT pathway, a key mechanism in cartilage repair 114 . Extracellular vesicles derived from MSCs also contribute to liver repair by crossing the microvasculature and dispersing throughout injured tissue to aid regeneration 115 . The convergence of 3D bioprinting, MSCs, and associated technologies—such as electrospun nanofibers, bioreactors, and EVs—illustrates how interdisciplinary integration fosters innovation in tissue engineering and regenerative medicine. This synergistic approach holds promise for new strategies in tissue and organ repair, though future research will require rigorous clinical validation.
Fig. 9 presents a keyword timeline visualization, illustrating the distribution of research hotspots, thematic interconnections, and spatiotemporal evolution in tissue engineering. This map shows that cluster #0 (3D printing) and cluster #1 (tissue engineering) represent two major current research hotspots. These clusters span a longer timeline and exhibit numerous connections to other keywords, reflecting their sustained activity and broad interdisciplinary relevance. The keyword clustering results align with the high-frequency keyword analysis, with “3D printing,” “tissue engineering,” “3D bioprinting,” and “bone regeneration” appearing most frequently (Table 6). The prominence of cluster #2 (bone tissue engineering), cluster #4 (bone regeneration), and cluster #8 (bone defect) underscores a sustained research emphasis on bone repair and regeneration. Additional clusters, including cluster #2 (calcium phosphate), cluster #6 (MSC-EVs), cluster #7 (silk fibroin), and cluster #9 (graphene oxide), further reveal significant research interest in biomaterials and engineered scaffolds. The analysis identifies ten major clusters of core research themes, encompassing technical approaches (eg, 3D printing and bioprinting), application areas (eg, bone tissue engineering), and emerging fields (eg, nanomechanical characterization). Research foci have progressively shifted from foundational materials and cell differentiation to technological advancements, material refinement, and emerging frontiers (eg, extracellular vesicle therapy and nanocomposite materials). Furthermore, a high degree of cross-integration among these themes is evident, forming a research landscape defined by multitechnique convergence and multiscale regulation.
Limitations
This study employs quantitative analysis to elucidate application trends, research patterns, and collaborative networks related to 3D bioprinting in the mesenchymal stem cell (MSC) field. Data were exclusively retrieved from English-language publications in the Web of Science Core Collection (WoSCC)—a database renowned for its authority and comprehensiveness in the medical field. However, the high citation rate of review articles may introduce bias into the study’s findings 116 ; we therefore excluded review articles to enhance the accuracy of our conclusions. In addition, citation counts are influenced by publication timing, potentially leading to the underrecognition of impactful recently published papers 117 . To ensure a rigorous analysis, we employed multiple tools for cross-validation (including CiteSpace, VOSviewer, and the R package “bibliometrix”) to guarantee data accuracy. Considering differences in data processing among software platforms, we primarily used CiteSpace, supplemented by VOSviewer and “bibliometrix.” All three tools analyzed the same data file to minimize result discrepancies. We acknowledge that we utilized the pre-configured templates of the R package; while no customization was performed, this tool sufficiently meets standard bibliometric requirements. Furthermore, our analyses were performed using multiple bibliometric software platforms, and—most importantly—our procedures followed a standardized systematic review and analysis protocol.
Conclusion
The integration of 3D bioprinting with the immunomodulatory and tissue repair capabilities of MSCs holds considerable potential for tissue engineering applications, such as bone and cardiac regeneration. Bibliometric analysis offers an objective, quantitative approach to assess developmental trends and frontiers in this interdisciplinary area. Using CiteSpace, VOSviewer, and the R package “bibliometrix,” we analyzed the research landscape, evolutionary pathways, primary directions, and future outlooks over the past 23 years. From 2014 to 2023, annual publications in this field showed a trend of fluctuating rapid growth. China has markedly increased its output in this specific domain, with both China and the United States emerging as the most influential countries. Enhanced collaboration between China, the United States, and other global research institutions and authors is essential. Leading journals such as Biomaterials and Biofabrication have played a pivotal role in advancing the field. High-frequency keywords cluster around 3D printing, tissue engineering, and bone tissue engineering, indicating current research priorities. Research areas such as “MSC-EVs,” “graphene oxide,” “nanocomposites,” and “nanomechanical characterization” have also garnered increasing attention. This trend reflects the emergence of cutting-edge research directions, such as MSC-derived exosome therapy and nanocomposite materials. Keyword co-occurrence analysis further indicated that “bioink,” “exosomes,” “nanoparticles,” “osteogenesis,” and “bioactive glass” may represent key foci for future investigations. This pioneering bibliometric study comprehensively synthesizes research trends and progress related to the application of 3D bioprinting in the field of MSCs. The resulting analysis provides a novel perspective on the application of 3D bioprinting in MSC research, assisting researchers in selecting suitable journals, identifying potential collaborators, and staying updated on emerging topics and cutting-edge developments to advance the field.
Supplemental Material
sj-txt-1-cll-10.1177_09636897251410650 – Supplemental material for 3D bioprinting and mesenchymal stem cells: A bibliometric analysis of emerging trends and advancements
Supplemental material, sj-txt-1-cll-10.1177_09636897251410650 for 3D bioprinting and mesenchymal stem cells: A bibliometric analysis of emerging trends and advancements by Yi-cheng Li, Bo Shao, Kui Ye, Xu Liu, Hong-da Wang, Cheng-lu Sun, Yi-yi Xiao and Hao Wang in Cell Transplantation
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
Not applicable.
Author Contributions
YCL: Conceptualization, Data curation, Formal Analysis, Methodology, Software, Visualization, Writing – original draft, Writing – review and editing. BS: Conceptualization, Data curation, Formal Analysis, Methodology, Software, Validation, Visualization, Writing – original draft. KY: Conceptualization, Formal Analysis, Investigation, Validation, Writing – original draft. XL: Conceptualization, Formal Analysis, Investigation, Validation, Writing – original draft. HDW: Conceptualization, Formal Analysis, Investigation, Validation, Writing – original draft. CLS: Conceptualization, Data curation, Validation, Writing – original draft. YYX: Conceptualization, Data curation, Validation, Writing – original draft. HW: Funding acquisition, Data curation, Project administration, Resources, Supervision, Writing – review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants to Hao Wang from the National Natural Science Foundation of China (No. 82270794), Natural Science Foundation of Tianjin (No. 25JCYBJC00500), Horizontal Research Project of Tianjin Medical University General Hospital (No. 2023048), and Tianjin Key Medical Discipline Construction Project (Nos TJYXZDXK-3-001B and TJYXZDXK-3-013C).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are available within the article and its supplementary materials; further inquiries should be directed to the corresponding author.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
