Abstract
Iron dysregulation is a crucial factor in the development of neurological diseases, leading to the accumulation of reactive oxygen species (ROS) and oxidative stress, triggering inflammatory responses, and ultimately causing neurological impairment. Pachymic acid (PA) is an active ingredient extracted from the medicinal fungus Poria cocos, which has been reported with multiple pharmacological effects, including anti-inflammatory, anti-ischemia/reperfusion, and anticancer actions. In this study, we test whether PA have neuroprotection effect aganist ferrous ions induced toxicity in SH-SY5Y cells. It was found that pre-treatment with PA reduced intracellular ROS levels, increased mitochondrial membrane potential, and protected cells from apoptotic death. RNA-seq and qRT-PCR results indicated that PA can regulate the key genes IL1B, CXCL8, CCL7, and LRP1 on the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways, such as NF-κB signaling pathway, IL-17 signaling pathway, to prevent Fe2+-induced apoptotic cell death. Our research indicated that PA has potential therapeutic effects on the neuroprotection by regulating neuroinflammation and oxidative stress damage.
Dear editor,
Iron dyshomeostasis plays a key role in neurological diseases, such as Alzheimer’s disease (AD), Parkinson’s disease (PD), and Huntington’s disease (HD)1–3. In addition, excess free ferrous ions (Fe2+) was involved in the Fenton reaction, which led to the accumulation of reactive oxygen species (ROS) and oxidative stress 1 . The increase of ROS, which induced by excess Fe2+, affected mitochondrial activity and led to mitochondrial dysfunction which has associated with neurodegenerative disease, such as AD, PD, and amyotrophic lateral sclerosis (ALS) 4 , and resulted inflammatory responses 5 . Neuroinflammation was a condition associated with neuron degeneration and involves the production of proinflammatory cytokines, such as interleukin (IL)-1β, IL-6, and tumor necrosis factor (TNF)-α 6 . Besides, the overexpression of these cytokines, which were closely related to inflammatory response, can future activate the downstream apoptotic signaling pathway of neurons and aggravate neurological impairment 7 .
Pachymic acid (PA) is one of the main active ingredients extracted from medicinal fungus Poria cocos 8 . Pachymic acid has been reported with multiple pharmacological effects, including neuroprotection 9 , anti-inflammatory 10 , anti-ischemia/reperfusion effects 11 , and anticancer action 12 . However, whether PA protect Fe2+-induced oxidative stress and preserve the mitochondrial membrane potential which have been the focus of attention in the treatment of nerve injury, remains unknown. In this study, we test whether PA has the potential to inhibit neuroinflammation on ferrous ions induced toxicity in SH-SY5Y cells.
In order to investigate the toxicity of ferrous iron and the preprotection of PA in SH-SY5Y cells, the cells were treated with 50, 100, 150, 200 μmol/L of FeCl2 for 24 h. Cell Counting Kit- 8 (CCK-8) assay were used to assess cell viability. The results showed that the viability of cells incubated with 200 μmol/L FeCl2 for 24 h significantly decreased to 54.91% ± 2.63% compared with the control group (Fig. 1A). So, this concentration of FeCl2 was chosen for the following experiments. In previous studies, PA pretreatment for 1–2 h can inhibit cellular oxidative stress damage 10 . Therefore, to investigate the effect of different concentrations of PA on the cytotoxicity of Fe2+-induced SH-SY5Y cells, the SH-SY5Y cells were treated with different concentrations of PA for 2 h prior to treatment with 200 μmol/L FeCl2 for another 24 h. As shown in Fig. 1B, the viability of cells were significantly higher in PA-treated groups (20 μM PA) than that of the Model group. In addition, cellular apoptosis was examined by annexin V-FITC and PI staining. In the early stage of apoptosis, cells were found to be positive for both Annexin V-FITC and PI. Conversely, in the late stage of apoptosis, cells were found to be positive for Annexin V-FITC but negative for PI. As demonstrated in Fig. 1C, the rate of early apoptosis was significantly lower in the cells treated with PA + FeCl2 compared to the Model group. These results demonstrate that PA blocked Fe2+-induced apoptotic cell death (Fig. 1C).
Mitochondria play a crucial role in cellular energy production through oxidative phosphorylation, which involves the transfer of electrons from molecular oxygen to generate adenosinetriphosphate (ATP). If mitochondria are damaged or functioning improperly, the normal flow of electrons will be disrupted and result in the buildup of ROS or oxidative stress 13 . To further determine whether PA prevented apoptotic cell death in SH-SY5Y cells by alleviating Fe2+-induced mitochondrial dysfunctions, the ROS level and mitochondrial membrane potential were measured using DCFH-DA and JC-1 staining. DCF fluorescence increases in proportion to the level of ROS in the cell, making it a useful tool for studying the effects of ROS on cellular function and health 14 . The results of immunofluorescence showed that PA significantly attenuated Fe2+-induced intracellular ROS levels (Supplemental Figure S1, Figure S2; Fig. 1E). As shown in Fig. 1D, compared with the Model group, pretreated with 20 μM PA for 2 h prior to treated with 200 μM Fe2+ for 24 h significantly increased the mitochondrial membrane potential (Fig. 1E) 10 . In conclusion, PA attenuated oxidative stress, preserved mitochondrial membrane potential in Fe2+-induced oxidative damage in SH-SY5Y cells model, which is a commonly used in vitro model for assessing basic neurophysiological processes potentially involved in neurodegeneration or brain injury.
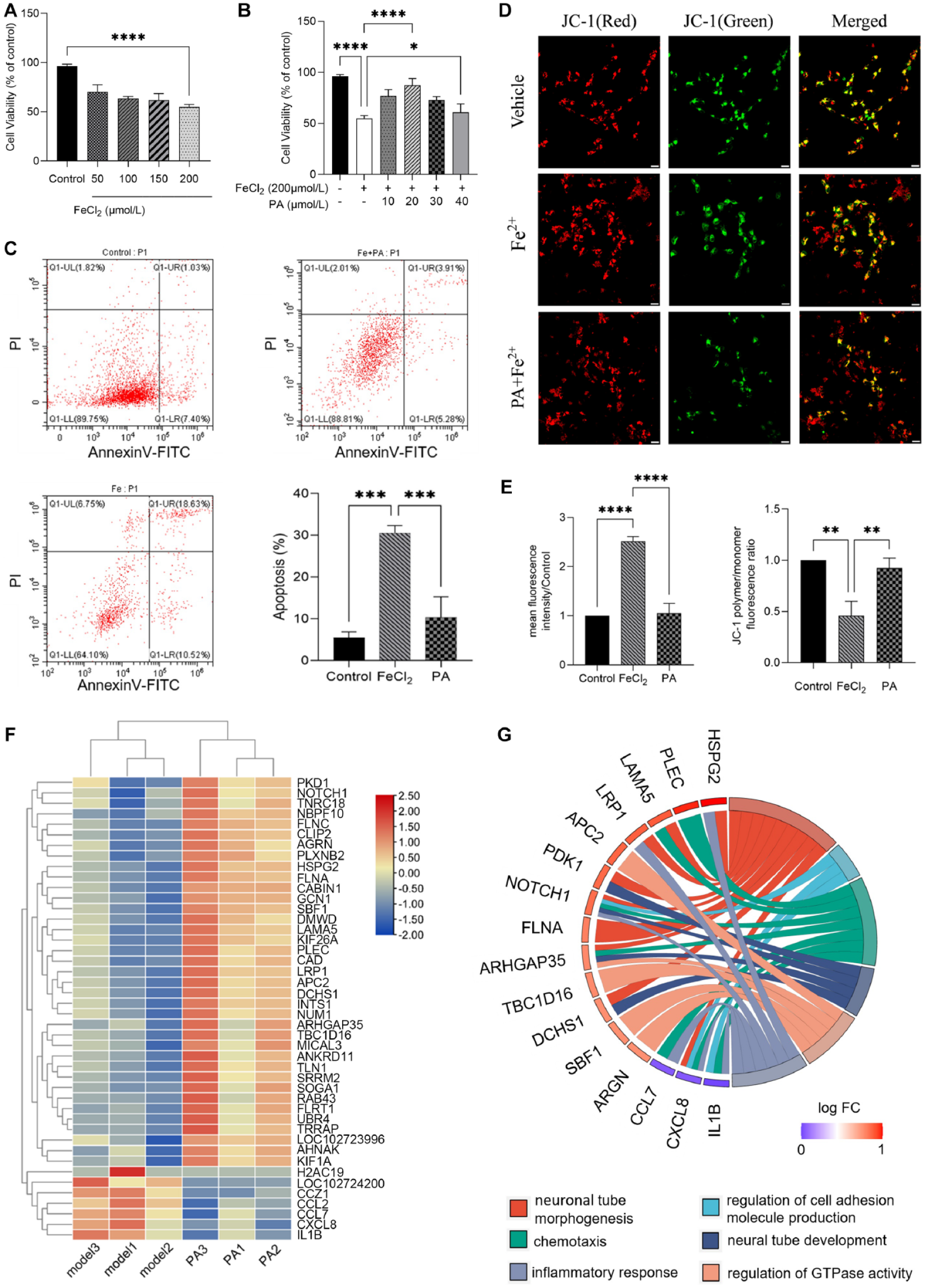
Effect of PA on Fe2+-induced apoptosis of SHSY-5Y cells. (A) CCK-8 assay of SH-SY5Y cells (Human Neuroblastoma Cell Line) with the treatment of Fe2+ for 24 h. (B) CCK-8 assay of SH-SY5Y cells with pretreatment of PA (0, 10, 20, 30, 40 μM) for 2 h and then incubation with 200 μM Fe2+ for 24 h. Mean ± SD (n = 4). *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. (C) Apoptotic cells were examined by annexin V-FITC/PI double-staining in the Model group and PA Treated group for 24 h. (D) Mitochondrial damage assay via JC-1 (5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimidazolylcarbocyanine iodide) staining. Scale bar: 20 μm. (E) Quantitative analysis of fluorescence intensity as an indication of ROS levels and MMP levels. Mean ± SD (n = 4). Scale bar: 50 μm. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. (F) DEGs in Model group and PA Treated group. (G) The bar plots show the changes in the expression of genes between Model group and PA Treated group. PA: Pachymic acid; CCK-8: Cell Counting Kit-8; PT: propidium iodide; ROS: reactive oxygen species; MMP: matrix metalloproteinase; DEGs = differentially expressed genes.
To identify the specific molecular network and mechanism of PA protection of SH-SY5Y cells apoptosis, RNA-sequencing was performed for SH-SY5Y cells in Model group, PA-Treated group, and the Control group, followed by bioinformatics analyses. The RNA-sequencing results showed that compared to the Control group, 107 up-regulated and 312 downregulated differentially expressed genes (DEGs) were identified in the Model group (Figure S3). The raw data have been uploaded to the NCBI database (https://www.ncbi.nlm.nih.gov/sra/PRJNA972428). The differential expression of these genes may regulate the apoptosis of SH-SY5Y cells. However, compared with the Model group, there were 37 up-regulated and 7 downregulated DEGs in the PA-Treated group, (|log2foldchange|(log2FC) > 0.3, Padj < 0.05), (Fig. 1F). Hierarchical clustering analysis was performed for the 44 common DEGs in PA-Treated group and Model group. The results showed that the expression level of the same gene in the PA-Treated group was significantly different from that in the Model group (Fig. 1F).
GO functional enrichment analysis showed that the DEGs between PA-Treated group and Model group were significantly enriched in 30 terms (Supplemental Figure S4), including 14 terms in biological processes (BPs), 10 terms in cellular components (CCs), and 6 terms in molecular functions (MFs), (padj < 0.05). As shown with Circos plots (https://www.bioinformatics.com.cn), there were 6 significantly enriched GO terms (neural tube morphogenesis, regulation of cell adhesion molecule production, chemotaxis, neural tube development, regulation of GTPase activity, inflammatory response) related to apoptotic cell death and neurological impairment (Fig. 1G), invloving 13 up-regulated genes (HSPG2, PLEC, LAMA5, LRP1, APC2, PDK1, NOTCH1, FLNA, ARHGAP35, TBC1D16, DCHS1, SBF1, and ARGN,) and 3 downregulated genes (CCL7, CXCL8, IL1B). KEGG pathway analysis identified 20 significantly enriched pathways (Padj < 0.05) (Figure S5[a]), which involved Malaria, Salmonella infection, IL-17 signaling pathway, NF-κB signaling, mitogen-activated protein kinase (MAPK) signaling pathway 15 . In addition, the interactions of significantly enriched KEGG pathways with DEGs showed that there were 3 key downregulated genes (IL1B, CXCL8, CCL7) and 9 key up-relgulated genes (LRP1, NOTCH1, UBR4, AGRN, ARHFAP35, HSPG2, FLNA, FLNC, LAMA5) regulated the 20 significant KEGG pathway (Figure S5[b]). The more important genes were IL1B, CXCL8, CCL7, LRP1, LAMA5, which regulated pathways related to apaoptosis, inflammation, nerve injury and oxidative stress, such as Malaria, NF-κB signaling pathway, IL-17 signaling pathway, ECM-receptor interaction.
In addition, five DEGs (IL1B, CXCL8, CCL7, LRP1, LAMA5) were selected for validation using qRT-PCR, based on the results of the RNA-seq analysis. Consistent with the results from the RNA-seq analysis, the expression of IL1B, CXCL8, and CCL7 was found to be significantly decreased in the PA Treated group compared to the Model group (*P < 0.05; **P < 0.01). Although LRP1 were significantly decreased in RNA-seq analysis, the qRT-PCR analysis showed only a 10% decrease (Figure S6 supplemental). This discrepancy could be due to the different sensitivities of these two methods. In general, the qRT-PCR results validated the expression pattern of selected DEGs from the RNA-Seq data. This result suggests that PA can inhibit Fe-induced ROS and apoptosis by downregulating IL1B, CXCL8, and CCL7 genes which regulated NF-κB signaling pathway and IL-17 signaling pathway.
In summary, this study demonstrated that PA can significantly block Fe2+-induced apoptotic cell death, reduce Fe2+-induced intracellular ROS levels and increase mitochondrial membrane potential, by downregulating key genes IL1B, CXCL8, and CCL7 in the NF-κB and IL-17 signaling pathway. The research indicates that PA has potential therapeutic effects on the neuroprotection by regulating neuroinflammation and oxidative stress damage.
Supplemental Material
sj-docx-1-cll-10.1177_09636897231218382 – Supplemental material for RNA-Seq Analysis Reveals Potential Neuroprotective Mechanisms of Pachymic Acid Toward Iron-Induced Oxidative Stress and Cell Death
Supplemental material, sj-docx-1-cll-10.1177_09636897231218382 for RNA-Seq Analysis Reveals Potential Neuroprotective Mechanisms of Pachymic Acid Toward Iron-Induced Oxidative Stress and Cell Death by Shuyang Hu, Baili Yang, Binbin Li, Qianqian Fan, Tinglong Wu, Shanshan Li, Dong Wang, Tao Yang and Zhenghua Song in Cell Transplantation
Footnotes
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Hainan Natural Science Foundation (821MS154), Science and Technology Special Fund of Hainan Province (grant no. ZDYF2022SHFZ289), Hainan Province Clinical Medical Center (grant no. 0202067) and National Natural Science Foundation of China (grant no. 32260244).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
