Abstract
Introduction
Background
Inflammatory bowel disease (IBD) is a multifaceted inflammatory condition with a complex pathogenesis that remains incompletely understood. 1 Recent studies have shed light on the potential involvement of autophagy in the development of IBD.2–4 Autophagy, a process vital for cellular metabolism and survival, has been implicated in the onset and progression of various diseases,5,6 including IBD. As research delves deeper, monoclonal antibodies targeting relevant pathways have been utilized in the treatment of this disease.7–9 However, a significant concern arises with the decreased treatment sensitivity and the emergence of drug resistance following monoclonal antibody therapy, which profoundly impacts therapeutic efficacy.10,11
Although the mechanism of drug resistance produced by monoclonal antibodies in the treatment of IBD has not been fully revealed, relevant studies have uncovered many molecular mechanisms leading to treatment drug resistance.12,13 One significant factor is the production of anti-antibodies. 14 Anti-antibodies can neutralize Fab fragments attached to proteins and promote the formation of immune complexes, 15 both of which lead to increased drug clearance through the reticuloendothelial system, leading to altered drug pharmacokinetics and reduced therapeutic efficacy. However, in the case of anti-antibody-independent resistance to monoclonal antibody therapy for IBD, the mechanism may involve mechanical failures, such as insufficient lymphocyte trafficking, or insufficient drug concentration due to pharmacokinetic variations. In this context, autophagy has emerged as a critical regulator of drug resistance. 16
Despite the optimization of biological therapies, a significant proportion of patients have a secondary loss of response or are primary nonresponders to therapy. Approximately only 40% of patients with response to biological therapies maintain clinical remission at 1 year. The development of resistance to biological agents poses an inevitable challenge in the treatment of IBD. The diverse mechanisms of action exhibited by different biologics offer patients the opportunity to regain clinical remission by transitioning to alternative medications. The discovery of new therapeutic targets holds promise for the development of emerging biological agents. However, the frequent switching of targeted drugs may not be feasible for many patients due to the high initial treatment costs associated with such interventions.17,18 In cases where drug resistance occurs, alternative strategies such as increasing the dosage, shortening the dosing interval, or considering the addition or adjustment of immunosuppressants can be considered to restore clinical response and prevent the discontinuation of effective treatment.19,20
Traditional Chinese medicine (TCM) is widely utilized in the treatment of IBD.21–23 Most Chinese herbal medicines are herbs, and more and more pharmacological effects of herbs are being revealed, including anti-inflammatory, anti-tumor and antioxidant effects.24,25 Among the various TCM formulations, Peilan and Fuling are highly regarded and commonly used medicines for invigorating the spleen and removing dampness. Peilan, which contains p-Cymene as its primary component, 26 has been scientifically demonstrated to possess a range of beneficial effects. Through its modulation of the PI3K (phosphoinositide 3-kinase)/AKT (Ak strain transforming)/mTOR (mammalian target of rapamycin) signaling pathway, 27 p-Cymene exhibits antioxidative, anti-inflammatory, antiviral, antitumor, immune-regulatory, vasodilatory, and neuroprotective properties.28,29 Similarly, Fuling, primarily composed of pachymic acid, 30 exerts its effects by activating the AMPK (AMP-activated protein kinase) and mTOR cell autophagy signaling pathways.31,32 Additionally, it can inhibit FFA-induced (free fatty acid-induced) expression of autophagosomes and autophagy-related proteins in HepG2 (hepatoblastoma) cells. 33
Objective
The objective of this study is to investigate the potential of p-Cymene and pachymic acid in modulating the autophagy signaling pathway. The study aims to assess whether these compounds can alleviate drug resistance observed in adalimumab-resistant inflammatory cell lines and alleviate inflammatory response.
Materials and methods
Experimental design
The adalimumab-resistant inflammatory cell lines in the logarithmic growth phase were taken, adalimumab was added to the control group, and different concentrations of p-Cymene and pachymic acid were added to the experimental group based on the control group, the concentrations were 0.1 μg/ml and 0.2 μg/ml, 1 μg/ml, 5 μg/ml, 10 μg/ml. 34
Cell culture
A RAW 264.7 murine macrophage cell line was purchased (Jiangsu ALF Biotechnology Co.) and cultured in DMEM (Dulbecco’s Modified Eagle Medium) supplemented with 10% FBS (Fasting blood sugar), 100 u/ml penicillin and 100 mg/ml streptomycin. The cells were maintained in a humidified incubator at 37 °C with 5% CO2 for optimal growth conditions. The RAW 264.7 murine macrophage cell line was treated with LPS (Lipopolysaccharide,final concentration of 1 μg/mL) for 24 hours. We assessed the expressions of inflammatory cytokines using ELISA (enzyme-linked immunoassay).
Cell growth curve and cell viability
Inflammatory cells cultured with adalimumab (AbbVie China Co.) of unequal degree, adopted step-by-step induction method, were initially introduced at a dose of different concentrations (gradients: 2 μg/μL, 10 μg/μL, 50 μg/μL, 250 μg/μL) for 24 hours after induction, the normal culture base was maintained, and the cell growth rate was measured using CCK-8 (cholecystokinin octapeptide) assay.
The viability of the cells was determined using the CCK-8 assay. The cell survival rate was expressed as a percentage relative to normal cells. Based on the obtained viability data, growth curves were calculated to visualize the proliferation and growth patterns of the cells over time.
ELISA
The expression of cytokines, including Tumor Necrosis Factor-α (TNF-α), Interleukin-1β (IL-1β), Interleukin-6 (IL-6), and Interleukin-10 (IL-10), was assessed using mouse ELISA kits (Jiangsu ALF Biotechnology Co.). For the experiment, cells were seeded in 12-well plates and pre-treated with different concentrations of p-Cymene and pachymic acid (Shanghai TAUTO Biotech Co.). The treatments were performed in the absence or presence of LPS for a duration of 24 hours. Following the incubation period, the supernatant from each well was collected into individual tubes. The content of each cytokine was then determined according to the instructions provided with the respective ELISA kit.
Quantitative reverse transcriptase-polymerase chain reaction (qRT-PCR)
The RNA samples were quantified using an ultraviolet spectrophotometer to determine their concentration and purity. Following quantification, equal amounts of RNA were subjected to reverse transcription using a High-Capacity cDNA Reverse Transcription Kit, converting RNA into complementary DNA (cDNA). PCR amplification was carried out using TaqMan gene expression master mix, utilizing 2 μL of the synthesized cDNA in a final reaction volume of 20 μL. The amplification cycles were performed using a real-time PCR instrument. To analyze the quantitative fluorescence results, a standard curve was constructed using known reference samples. The obtained fluorescence data were then analyzed using Bio-Rad iq5 software, enabling quantification and analysis of gene expression levels.
Western blot analysis
Cells were harvested and suspended in RIPA lysis buffer containing 1 mmol/L PMFS fluoride. Antibodies for p‐AKT, p‐PI3K and p‐mTOR, and β‐actin were purchased from Cell Signaling Technology. Total protein was tested for cytokine expression. Densitometry was conducted on blots using ImageJ. Signal quantitation was calculated using Quantity One software and signals were normalized to the β‐actin signal.
The calculation of the IC50 value
To establish the adalimumab-resistant cell line, the concentration of adalimumab was gradually increased until the drug resistance index (RI) exceeded 5. The RI is calculated as the IC50 (half-maximal inhibitory concentration) of the drug-resistant cell line divided by the IC50 of the parental cell line. It is generally accepted that when the RI surpasses 5, the drug-resistant cell line meets the required threshold. The calculation of the IC50 value was analyzed using Prism 9.
Statistical analysis
If the data fitted in the distribution of normality and the variance was homogeneous, one-way analysis of variance (ANOVA) was used for comparison between groups, followed by Tukey’s multiple comparisons as post hoc test. p < .05 was considered statistically significant. For data that did not fit in the distribution of normality, the Kruskal-Wallis tests (followed by the Dunn’s test) were used. Values of p < .05 were considered statistically significant. The data was analyzed using Prism 9, with default settings and parameters. Additional details about the experimental design, such as the sample size and control groups, can be found in the supplementary materials.
Results
The effect of inflammatory cytokines in LPS-induced RAW264.7 cell line
The results showed that LPS stimulation significantly elevated TNF-α, IL-1β, IL-6 and IL-10 expressions (Figure 1). As shown in Figure 1, levels of the inflammatory cytokines, IL-6(p = .022, t = 3.654, df = 4), IL-10(p < .001, t = 25.41, df = 4) and TNF-α (p = .029, t = 3.328, df = 4) were significantly increased in LPS group as compared with the control group. This means that the inflammatory cell line modeling of RAW 264.7 murine macrophage cell line induced by LPS was successful. Cytokines levels in the LPS induced RAW 264.7 murine macrophage cell line. IL-1, IL-6, IL-10, and TNF-α levels were measured by ELISA from the Control and LPS groups. Significance was determined by ANOVA with Tukey’s analysis.
Figure 1 Cytokines levels in LPS induced RAW 264.7 murine macrophage cell line. IL-1, IL-6, IL-10, and TNF-α levels were measured by ELISA from the control and LPS groups. Significance was determined by ANOVA with Tukey’s analysis.
Inflammatory cell line develops resistance upon adalimumab treatment
In the absence of adalimumab treatment, the cell proliferation rate remained comparable to the control group (Figure 2(a)). After calculating the cell proliferation curve, the logIC50 of the adalimumab-resistance inflammatory cell line was 14.38, and the logIC50 of the parental cell line was 7.64. The results (Figure 2(b)) showed that the IC50 value of the adalimumab-resistance inflammatory cell line was on the order of hundreds of trillions, while that of the parental cell line was on the order of millions. We considered this result to be related to the small toxicity of adalimumab. (a). From the first to the seventh day, the OD value of the adalimumab-resistance inflammatory cell line and parental cell line was measured by the CCK-8 method. (b). After calculating the cell proliferation curve, the logIC50 of the adalimumab-resistance inflammatory cell line was 14.38, and the logIC50 of the parental cell line was 7.64.
The effects of cell proliferation in adalimumab-resistance inflammatory cell line
We took an adalimumab-resistance inflammatory cell line in the logarithmic growth phase, and added 250 μg/μL adalimumab to the control group. In the experimental group, various concentrations of p-Cymene and pachymic acid were added in addition to the control group. The cell growth curve was plotted, and the cell cycle was measured using the CCK-8 assay. The results showed that (Figure 3), from the proliferation curve, it can be seen that both p-Cymene and pachymic acid have the effect of inhibiting the proliferation of the adalimumab-resistance cell line. On the seventh day, the cell proliferation rates of all groups were significantly different from those of the control group (p < .001, t = 279.2, df = 4). (a). proliferation of adalimumab-resistance cell line treated with adalimumab. (b). proliferation of adalimumab-resistance cell line treated with adalimumab and different concentrations of p-Cymene. (c). proliferation of adalimumab-resistance cell line treated with adalimumab and different concentrations of pachymic acid.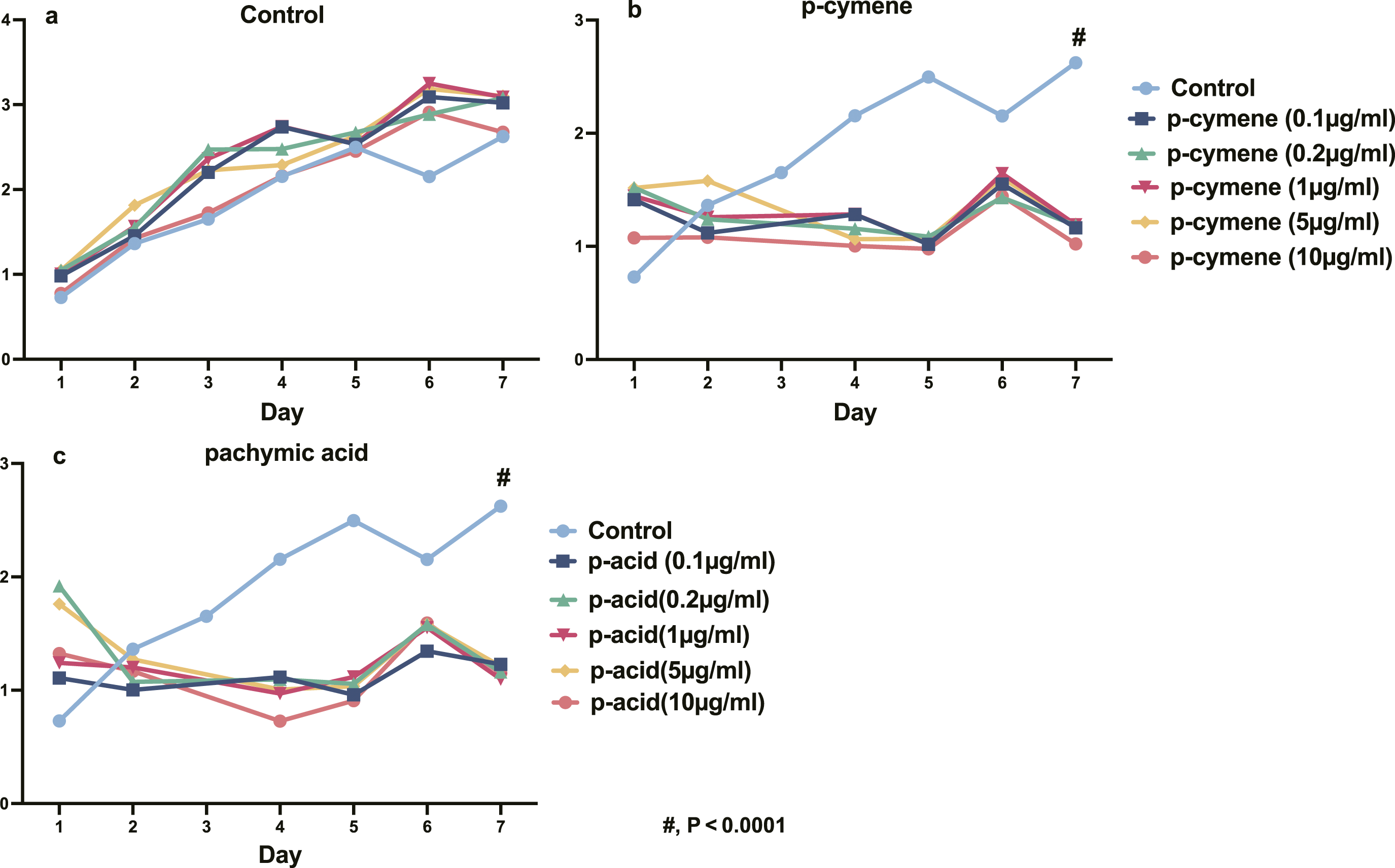
The expression of inflammatory response in adalimumab-resistance inflammatory cell line
We assessed expressions of inflammatory cytokines in adalimumab-resistance inflammatory cell lines using ELISA assays. The results showed that p-Cymene and pachymic acid can alleviate the expression of adalimumab on pro-inflammatory cytokines in an adalimumab-resistance cell line, and can enhance the expression of anti-inflammatory cytokines at the same time (Figure 4). p-Cymene and pachymic acid modulate cytokines levels in adalimumab-resistance cell line treated with adalimumab. IL-1, IL-6, IL-10 and TNF-α, levels were measured by ELISA in adalimumab-resistance cell lines from the Control, Control+ p-Cymene and Control + pachymic acid groups. Significance was determined by Dunn’s multiple comparisons test based on kruskal-wallis analysis. *p < .05, **p < .01, ***p < .001, ****p < .0001.
PI3K/AKT/mTOR signaling pathway plays a significant role in the occurrence of resistance
List of the primers used in quantitative PCR.


(a). representative experiment of PI3K, AKT and mTOR proteins examined by Western blot. (b) the expression of mTOR, Akt and PI3K mRNA using qRT-PCR. Significance was determined by Dunn’s multiple comparisons test based on kruskal-wallis analysis. *p < .05, **p < .01, ***p < .001, ****p < .0001.
Discussions
IBD is associated with an abnormal immune system response, which can result in intestinal mucosal epithelial cell death and tissue damage. 1 The conventional treatment approaches for IBD typically involve the use of corticosteroids and immunomodulators, as well as antagonists targeting TNF, inhibitors of IL-12 and IL-23, among others. 35
Autophagy is a highly conserved cellular process in which cytoplasmic materials are degraded and recycled to maintain energy homeostasis. When autophagy exhibits a cytoprotective function, the use of autophagy inhibitors can enhance the sensitivity of biological agents, suggesting that autophagy may play a protective role in drug-resistant cells. 36 Conversely, when autophagy expresses cytotoxic functions, it can induce the death of drug-resistant cells. Understanding the dual role of autophagy and its involvement in drug resistance is crucial for developing effective therapeutic strategies. Manipulating autophagy, either by inhibiting or inducing it, may offer potential approaches to overcome drug resistance and improve the sensitivity of biological agents in the treatment of IBD and other conditions. Further research is needed to elucidate the precise mechanisms and identify optimal strategies for targeting autophagy in drug-resistant cells. The mTOR (mammalian target of rapamycin) pathway is a key negative regulator of autophagy. 37 Inhibition of the mTOR pathway can induce autophagy and promote cell death in various cell types, including NIH3T3 cells, drug-resistant LoVo/ADR colon cancer cells, 13 and cisplatin-resistant cervical cancer cells. By targeting the autophagy pathway, mTOR inhibitors have shown promising effects in overcoming drug resistance.38,39 In situations where drug-resistant cells lack the ability to undergo apoptosis or exhibit resistance to apoptosis, inducing autophagy can be an alternative strategy to prevent the development of drug resistance. By promoting autophagy, drug-resistant cells may undergo autophagic cell death, leading to improved treatment outcomes. The mTOR complex also mediates other signaling pathways, including the MAPK and PI3K/AKT pathways. 40 Activation of these pathways can contribute to drug resistance in various tumor therapies. For example, reversing cisplatin resistance in lung cancer can be achieved by targeting the mTOR pathway, while PI3K inhibitors have shown efficacy in breast cancer, and TORC1 (target of rapamycin complex 1) inhibitors have demonstrated potential in melanoma treatment. 37
Research has indeed demonstrated that inhibition of autophagy can be a promising strategy to prevent drug resistance and enhance the effectiveness of anti-tumor therapy.41,42 Autophagy, a highly conserved cellular process across eukaryotes, is responsible for the degradation of cytoplasmic components within lysosomes. It plays a protective role in the early stages of tumor development and regulates oncogenes and molecular processes in normal cells. Evidence suggests that autophagy can suppress neoplastic transformation. Both preclinical studies and clinical trials have provided insights into the potential benefits of autophagy inhibition in improving the efficacy of anti-tumor drugs and monoclonal antibodies targeting tumor-associated antigens.43,44 By inhibiting autophagy, tumor cells may become more susceptible to the effects of anti-cancer treatments, leading to enhanced therapeutic outcomes. This approach has shown promise in overcoming drug resistance and improving the effectiveness of monoclonal antibody therapies.
Traditional Chinese medicine (TCM) provides a different perspective on the etiology of IBD.45,46 According to TCM principles, the development of IBD is attributed to imbalances in the body, particularly related to the spleen and kidney functions. TCM theory suggests that chronic damage to the temper (a concept related to the body’s vital energy) can lead to disturbances in blood circulation and the stagnation of Qi (vital energy) and blood. These imbalances can result in the accumulation of dampness and the formation of "rotten flesh” within the body. Furthermore, TCM emphasizes the role of the kidneys in IBD. According to TCM theory, the continuous spread of these imbalances can eventually affect the kidneys, leading to further impairment of both spleen and kidney functions. The spleen is considered responsible for the digestion and absorption of nutrients, while the kidneys are associated with the storage of essence and the regulation of water metabolism. In TCM practice, the treatment of IBD involves strategies to invigorate the spleen, remove dampness, and restore the balance of Qi and blood circulation.
Traditional Chinese herbal medicines, such as Peilan and Fuling, are commonly used to address these imbalances and alleviate the symptoms of IBD. These herbs are believed to have properties that invigorate the spleen and remove dampness, aiming to restore the normal functions of the affected organs and promote overall health. In the treatment of IBD, the main method is to jianpi, qushi and qingre (invigorate the spleen, dissipate dampness and clear away heat), and also to jianpi and gushen (invigorate the spleen and strengthen the kidney), liangxue and sanyu (cool blood and remove stasis), lianyang and shengji (astringe ulcer and promote muscle).22,47 It’s important to note that TCM treatments for IBD are highly individualized based on the patient’s specific symptoms, constitution, and pattern of disharmony. Qualified TCM practitioners will assess each patient and develop a personalized treatment plan incorporating various herbs, acupuncture, dietary adjustments, and lifestyle recommendations. Experiments have shown that traditional Chinese medicine has the effect of regulating the autophagy signaling pathway. Fuling is commonly used in TCM to invigorate the spleen and remove dampness, which are essential therapeutic principles in the treatment of IBD. 33 Pachymic acid, as a triterpenoid compound found in Fuling, has been investigated for its effects on autophagy. 48 Research has demonstrated that pachymic acid can activate the AMPK (adenosine monophosphate-activated protein kinase) and mTOR (mammalian target of rapamycin) signaling pathways, which are involved in the regulation of autophagy. By activating AMPK, pachymic acid promotes autophagy induction, while inhibiting mTOR signaling can enhance autophagy activity. These effects have been observed in various cell types, including HepG2 cells, 49 where pachymic acid has been shown to inhibit the expression of autophagosomes and autophagy-related proteins induced by free fatty acids (FFA). Peilan is another commonly used traditional Chinese medicine for jianpi and huashi. Its main active ingredient of Peilan is a natural aromatic compound called p-Cymene. It has been proved that p-Cymene has anti-oxidative, anti-inflammatory, anti-viral, anti-tumor, immunomodulatory, vasodilation and neuroprotective effects.50,51 Studies have shown that p-Cymene inhibits the proliferation of tumor cells such as HL-60, MCF-7, and PC-3 by regulating the PI3K/AKT/mTOR signaling pathway. p-Cymene significantly inhibited the expression of PI3K, AKT and mTOR at the concentration of 50ug/ml 52 p-Cymene has good anti-inflammatory properties. Taking p-Cymene improved the symptoms of TNBS-induced enteritis mice, decreased the expression of TNF-α, IL-1β, and decreased the transcription of COX-2, IFN-γ, iNOS, and NF-κB mRNA. 53
In general, TCM has shown a synergistic property in the treatment of IBD, providing a beneficial adjunct to the treatment of IBD. In addition, the recent research literature also shows that improving the abundance and diversity of intestinal flora also has the potential of synergy for the treatment in IBD , 52 and this mechanism can also become the next research direction. The synergistic effect of traditional Chinese medicine provides more options for the treatment of patients with IBD.
Conclusions
In the current landscape, the quest for effective therapies to combat resistance to biologics in treating IBD remains an elusive pursuit. However, our recent cell experiments have unveiled a beacon of hope. Through meticulous investigation, we have illuminated the potential of p-Cymene and pachymic acid to transcend the barriers of adalimumab cell drug resistance. This profound revelation is rooted in the PI3K/AKT/mTOR signaling pathway.
The limitations of this study include the fact that the results are based solely on cellular experiments and are not supported by animal experimental data. Furthermore, the effects observed on the PI3K/AKT/mTOR signaling pathway may differ in an in vivo context. This study heralds a transformative solution in the field of TCM, which is expected to improve the efficacy of monoclonal antibodies in the treatment of IBD and to improve the efficacy of monoclonal antibodies in the anti-tumor field.
Footnotes
Author contributions
Author 1 (First Author, Jia-Bo Gu): Conceptualization, Methodology, Software, Investigation, Formal Analysis, Writing - Original Draft; Author 2(co-author, Jun Wang): Data Curation, Writing - Original Draft; Author 3: Visualization; Author 4: Data Curation; Author 5: Software; Author 6 and Author 7: Visualization; Author 8 (Corresponding Author, Hei-Ying Jin): Resources, Supervision, Writing - Review & Editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Authors receive research support fees from Special foundation for key Research & Development plans of Jiangsu Province (BE2023786). No other author has reported a potential conflict of interest relevant to this article.
