Abstract
Outcomes after repair of chronic rotator cuff injuries remain suboptimal. Type-1 collagen-rich tendon hydrogel was previously reported to improve healing in a rat chronic rotator cuff injury model. Stem cell seeding of the tendon hydrogel improved bone quality in the same model. This study aimed to examine whether there was a synergistic and dose-dependent effect of platelet-rich plasma (PRP) on tendon–bone interface healing by combining PRP with stem cell–seeded tendon hydrogel. Human cadaveric tendons were processed into a hydrogel. PRP was prepared at two different platelet concentrations: an initial concentration (initial PRP group) and a higher concentration (concentrated PRP group). Tendon hydrogel was mixed with adipose-derived stem cells and one of the platelet concentrations. Methylcellulose, as opposed to saline, was used as a negative control due to comparable viscosity. The supraspinatus tendon was detached bilaterally in 33 Sprague-Dawley rats (66 shoulders). Eight weeks later, each detached tendon was repaired, and a hydrogel mixture or control was injected at the repair site. Eight weeks after repair, shoulder samples were harvested and assigned for biomechanical testing (n = 42 shoulders) or a combination of bone morphological and histological assessment (n = 24 shoulders). Biomechanical testing showed significantly higher failure load and stiffness in the concentrated PRP group than in control. Yield load in the initial and concentrated PRP groups were significantly higher than that in the control. There were no statistically significant differences between the initial and concentrated PRP groups. The addition of the highly concentrated PRP to stem cells–seeded tendon hydrogel improved healing biomechanically after chronic rotator cuff injury in rats compared to control. However, synergistic and dose-dependent effects were not seen.
Introduction
Rotator cuff tears are a common problem causing shoulder pain and disability. Chronic rotator cuff tears are initially treated conservatively 1 ; however, surgical treatment is often pursued due to failure of conservative measures to alleviate pain and dysfunction. The number of rotator cuff surgical repairs has increased over the last decade 2 , yet re-tear rates remain high despite surgical and technological advancements3,4. Various reasons including patient background, surgical techniques, poor healing potential, and structural features at the tendon–bone interface (TBI) can be attributed to these failures4,5.
In our previous study, we developed thermoresponsive type-1 collagen-rich tendon hydrogel (tHG) derived from decellularized human tendon. tHG alone improved TBI healing biomechanically after chronic rotator cuff injury repair in rats 6 . Augmenting tHG with adipose-derived stem cells (ADSCs) further improved bone structure at the TBI after chronic rat rotator cuff injury compared to tHG alone. tHG+ ADSCs also improved tendon healing in an acute rat Achilles tendon repair model7,8.
In addition to ADSCs, platelet concentrate, well known as platelet-rich plasma (PRP), is another potent stimulator of healing 9 . Because platelet concentrate promotes cell proliferation and angiogenesis, it has been used clinically to improve rotator cuff tear healing3,10. However, the use of PRP is controversial 11 as some reports demonstrate a positive effect of PRP on rotator cuff healing in humans3,10 and in animal models9,12 while other studies fail to demonstrate improved healing with PRP13–15. Furthermore, there are no data examining the dose-dependent effect of platelet concentrate for TBI healing of chronic rotator cuff injuries.
We hypothesized PRP would have a positive, synergistic, and dose-dependent effect on TBI healing after chronic rotator cuff injury and repair in a rat model when combined with ADSC-seeded tHG.
Materials and Methods
Animal Studies and Research Ethics
Thirty-four specific pathogen-free Sprague-Dawley (SD) rats (12–14 weeks, 300–360 g, male, Inotiv, West Lafayette, IN) were used in compliance with the regulations of the Institutional Animal Care and Use Committee. Two SD rats were housed per cage in a 12/12 hours light/dark cycle and given food and water ad libitum. Twenty-one rats were randomly assigned to biomechanical testing while 12 were assigned to histology and micro-computed tomography (μCT) assessment. A flowchart of animal surgeries and analysis is shown in Fig. 1. One rat was used for histology of an intact shoulder. This study was carried out in compliance with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines.

Flowchart of animal use. This flowchart shows the number of the animals, shoulders, and analysis of each group. SD: Sprague-Dawley; MC: methylcellulose; tHG: type-1 collagen-rich tendon hydrogel; ADSC: adipose-derived stem cell; PRP: platelet-rich plasma; TBI: tendon–bone interface.
Material Preparation
SD rat ADSCs were commercially purchased (Cyagen Bioscience Inc., Santa Clara, CA). Cells were used at passage 5 or 6 and seeded at 4 M/ml in tHG 8 .
tHG was prepared from chemically decellularized human cadaveric flexor tendons following a previously described protocol 16 . Briefly, flexor tendons were collected from human cadavers of mixed sex and age (>18 years) to ensure consistent quality. Muscle tissue was carefully removed, and the tendons were chemically decellularized with sodium dodecyl sulfate and ethylenediamine tetra-acetic acid. The decellularized tendons were lyophilized with the FreeZone 1 Freeze Dry System (Labconco, Kansas City, MO) and milled using Wiley Mini-Mill (Thomas Scientific, Swedesboro, NJ) until the tendon became a fine powder, which was stored at −80°C until use. Tendon powder was digested with pepsin (Sigma-Aldrich, St. Louis, MO) in hydrochloric acid solution. After confirmation of the quality of the gel by microscopy, the tendon solution was neutralized to pH 7.4. Gelation was confirmed following incubation at 37°C.
PRP was commercially purchased (Innovative Research, Novi, MI), obtained from SD rat’s blood. We used two different platelet concentrations of PRP to investigate the dose-dependent effect of PRP platelet. PRP was used at its initial manufacturer’s platelet concentration for the initial PRP group. To create a concentrated PRP group, commercially purchased PRP was centrifuged at 400 × g for 10 minutes 17 into a pellet. Ninety percent of the supernatant plasma was carefully removed. Then the pellet was resuspended with the remaining 10% of the plasma, creating a high platelet concentration. Density of platelets in both solutions was counted manually.
Seventy microliters of tHG was mixed with 0.4 million (M) ADSCs and then with 10 µl of 10× minimal essential media (Applied Biosystems, Waltham, MA), 10 µl of fetal bovine serum (Sigma-Aldrich), and 10 µl of either of the platelet concentrates immediately before use. The tHG mixture was fabricated at a final concentration of 2% of tendon powder and 4 M/ml of ADSC.
Methylcellulose (MC) was used as control instead of saline, which we have previously used. In our study model, saline likely leaked out through the gap of the closed muscle right after the injection. Therefore, we desired a control more similar to tHG. By using a substance with viscosity comparable to that of tHG, we would be able to determine whether the advantage of tHG can be attributed to merely the occupation of any substance that can create a scaffold or to the biochemical effects of tHG specifically 18 . MC was purchased commercially (Sigma-Aldrich) and prepared with distilled water to a 2% solution.
Surgical Procedure
Supraspinatus detachment surgery
Rats were anesthetized with isoflurane. The surgical sites were disinfected, and enrofloxacin (5 mg/kg; Bayer DVM, Leverkusen, Germany) and buprenorphine hydrochloride (0.05 mg/kg; Reckitt Benckiser Healthcare Ltd., Hull, UK) were injected. Bilateral supraspinatus (SSp) tendons were exposed as previously reported8,19. Briefly, the deltoid was split and detached from the acromion. The SSp tendon was dissected from the long head of biceps and infraspinatus tendons. The insertion of the SSp tendon was detached from the humerus (Fig. 2A, B). After detachment, the deltoid muscle and skin were closed. Buprenorphine hydrochloride was administered every 12 hours for 3 days, and enrofloxacin was administered daily for 5 days.
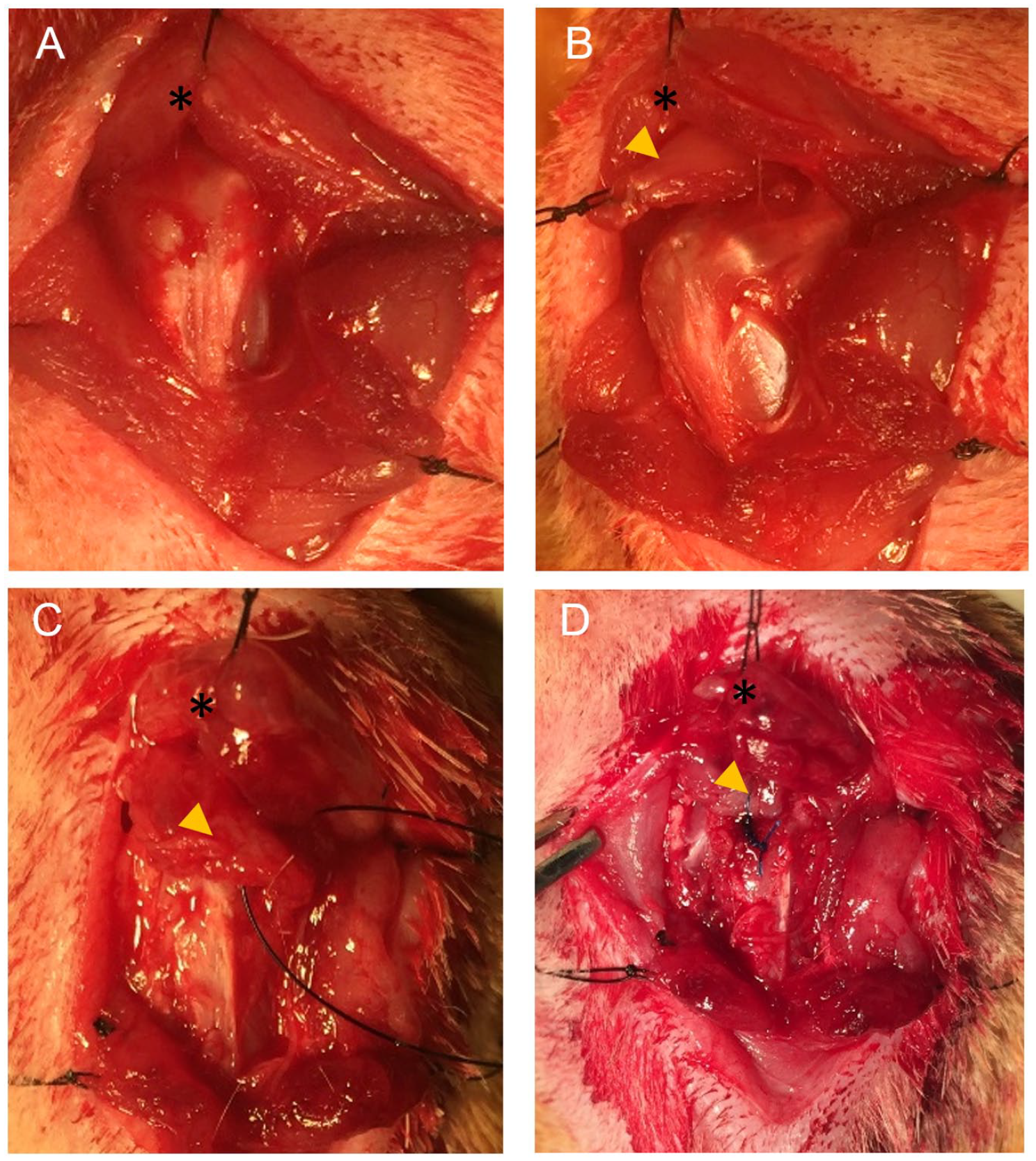
Surgical procedures. The supraspinatus tendon was dissected (A) and completely detached from the greater tuberosity of the humerus (B). Eight weeks after the first surgery, the tagged tendon was dissected (C), scar was excised, and the supraspinatus tendon was sutured to the greater tuberosity of the humerus via a bone tunnel (D). The asterisk denotes acromion; arrowhead denotes supraspinatus tendon.
SSp repair surgery
Eight weeks later, bilateral SSp tendons were repaired under anesthesia6,8,19. Briefly, the distal stump of the SSp tendon was identified through the same approach (Fig. 2C). Scar tissue was removed from the SSp distal stump and the greater tuberosity. A bone tunnel was created in the humeral head with a 23G needle. The SSp tendon was sutured with a double-armed 5-0 Prolene suture (Ethicon, Bridgewater, NJ) using a modified Mason-Allen technique through the bone tunnel onto the greater tuberosity (Fig. 2D) 19 . After SSp repair, the deltoid muscle was approximated. Before completing deltoid closure, 100 μl of one of the following gels was injected on to the repair site: 1) 2% MC (MC group), 2) tHG + ADSC + initial-concentration PRP (initial PRP group), or 3) tHG + ADSC + highly concentrated PRP (concentrated PRP group) (Table 1). On the opposite shoulder, a second gel was randomly selected and injected. Buprenorphine hydrochloride and enrofloxacin were administered.
The Amount and Final Concentration of Each Component in Experimental Groups.

This table shows the detail of the gel components in each group. The platelet concentration became theoretically one-tenth after mixing with tHG. ADSC: adipose-derived stem cell; MC: methylcellulose; M: million; PRP: platelet-rich plasma; tHG: type-1 collagen-rich tendon hydrogel.
Eight weeks after repair surgery, (16 weeks after the first surgery) shoulder samples were harvested, labeled by study number to blind the evaluator to study group, and randomly assigned for biomechanics (n = 42 shoulders) or a combination of histology and μCT (n = 24 shoulders).
Biomechanical Analysis
Fourteen shoulders per group were evaluated using the same biomechanical testing protocol in our previous report 8 . Samples were kept in PBS at −20°C until use. Specimens were preconditioned from 0% to 5% strain at a rate of 1% strain/s for five cycles then pulled until failure at a rate of 1% strain/s (Supplemental Fig. 1)20,21. Five biomechanical parameters were measured: load to failure, stiffness, yield load, energy, and strain at failure. Yield load was defined as the load at which there was a deviation of 10% from the linear region of the load versus displacement curve. Biomechanical testing and analysis were performed by a blinded investigator (H.S.).
microCT Evaluation
Eight shoulders per group were used to evaluate the bony parameters and were fixed in 10% formalin for 48 h at room temperature after harvest. Bony quality of the greater tuberosity of the humerus was evaluated with μCT (Explore CT-120; GE Healthcare, Little Chalfont, UK) at a resolution of 50 μm, 100 kV, and 50 mA. Acquired images were processed with MicroView2.2 (GE Healthcare) software. The following bony parameters were evaluated: bone mineral density (BMD), bone volume fraction, trabecular thickness, trabecular number, and trabecular separation 22 .
Histologic Assessment
After μCT assessment, samples were decalcified in Immunocal (Decal Congers, NY) at room temperature for 48 hours and embedded in paraffin, and 5-μm-thickness coronal sections were prepared at the mid-portion of the SSp TBI. Hematoxylin and eosin, Safranin-O, and Picrosirius Red staining were performed. An intact control shoulder sample was processed via the same protocol. The area of metachromasia at the surgery site was measured using ImageJ (National Institute of Health, Bethesda, MD) with Safranin-O images6,8,23. Collagen fiber organization was assessed with Picrosirius Red staining according to the birefringence with the captured image under a polarized microscope (Leica DMi8; Leica, Wetzlar, Germany)6,8,23. Ten rectangular areas (2500 μm2 each) were randomly selected at the surgical site per image and the gray scale of the selected area was calculated with ImageJ. The higher average grayscale values were considered more organized and matured collagen. The area of type-1 collagen accounting for the TBI area was calculated based on the images of Picrosirius Red staining under a polarized microscope. The color of collagen changes according to the fiber thickness, and collagen type-1 typically appears red to yellow 24 . Therefore, the percentage of type-1 collagen area accounting for TBI area was calculated with ImageJ24–26. Briefly, the digital image was segmented using hue, saturation, and brightness. Then the area of interest was marked. The image was converted to a binary image and measured.
Statistical Analysis
A priori power analysis was performed to estimate the sample size. Using a primary outcome of load to failure and an increased strength of 20% for clinical significance6,8,27,28, a sample size of 14 shoulders in each group achieved a power of 0.80 with α = 0.05. All the data are expressed as mean ± standard deviation. For statistical analysis, a one-way analysis of variance was performed, and the Tukey-Kramer honestly significant difference test was performed as a post hoc analysis (JMP, SAS Institute, Cary, NC). A value of P < 0.05 was considered statistically significant.
Results
Concentration of Platelets
The initial manufacturer’s platelet concentration was 1.50 × 106/µl (initial PRP group), and it increased to 7.24 ± 1.46 × 106/μl after concentration (concentrated PRP group) (Table 1). Both platelet concentrates were verified as leukocyte-poor by microscopic analysis.
Biomechanical Results
Thirty-eight samples out of 42 (90.5%) failed at the TBI. One sample in the MC group and one in the concentrated PRP group failed at mid-substance while two samples in the concentrated PRP group failed at the clamp interface. These samples were included in the analysis because the true load to failure at the TBI of these samples should be higher than the results obtained during testing. There was one outlier in both the MC and concentrated PRP groups, defined as outside of the range of mean ± 2SD. Outliers were excluded from biomechanical analysis, resulting in 13 shoulders in the MC group, 14 in the initial PRP group, and 13 in the concentrated PRP group for the biomechanical analysis.
Overall, biomechanical testing showed significantly higher load to failure (27.86 ± 7.02 N) and stiffness (16.75 ± 3.74 N/mm) in the concentrated PRP group than in the MC group (21.22 ± 4.74 N, P = 0.041 and 12.04 ± 3.73 N/mm, P = 0.012, respectively, Fig. 3). Load to failure in the initial PRP group (27.26 ± 7.81 N) versus the MC group was P = 0.062. Yield load in the initial PRP group (25.55 ± 8.59 N) and the concentrated PRP group (27.69 ± 6.95 N) were significantly higher than that in the MC group (18.58 ± 5.24 N, P = 0.039 and P = 0.006, respectively). The energy and strain at failure in the initial PRP group, the concentrated PRP group, and MC group did not reach statistical significance. There was no statistically significant difference in any biomechanical parameter between the initial and concentrated platelet concentration groups.
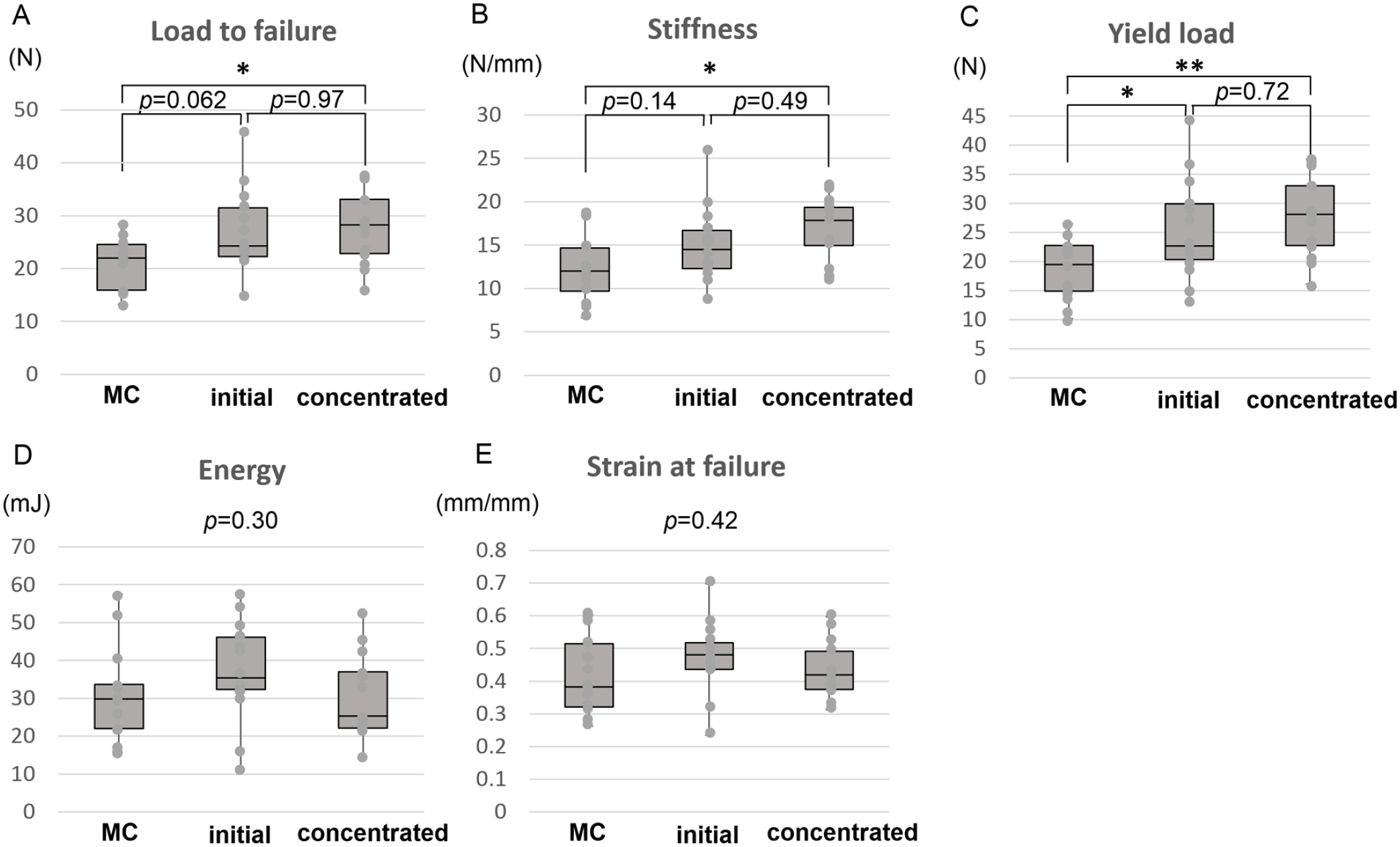
Biomechanical evaluation. Load to failure and stiffness in the concentrated PRP group were significantly greater than those in MC control (A and B). Yield load in the initial and concentrated PRP groups were significantly larger than that in MC control (C). Energy and strain at failure were not significantly different between groups (D and E). MC: methylcellulose; PRP: platelet-rich plasma. *P < 0.05; **P < 0.01.
microCT Results
BMD at the greater tuberosity in the initial PRP group and concentrated PRP group were not significantly different from that in the MC group (P = 0.195). There were no significant differences between the three groups in the other bone parameters (Table 2).
Bone Parameters Evaluated by Micro-Computed Tomography.

No parameter in bone morphological study was significantly changed among the three groups. MC: methylcellulose; M: million; PRP: platelet-rich plasma; ANOVA: analysis of variance; BMD: bone mineral density; BVF: bone volume fraction; Tb Th: trabecular thickness; Tb Sp: trabecular separation; Tb N: trabecular number.
Histology Results
Cellularity was high at the repair site in all three groups and covered by scar-like tissue (Fig. 4). However, the repair site and the structure of enthesis appeared better organized in the initial and concentrated PRP groups than in the MC groups (Supplemental Fig. 2). Although there was no significant difference in the area of fibrocartilage calculated from Safranin-O staining (P = 0.20, Fig. 4B), the area was slightly greater in the initial (0.58 ± 0.49 mm2) and concentrated (0.42 ± 0.20 mm2) PRP groups than that in the MC groups (0.27 ± 0.18 mm2). Similarly, using Picrosirius Red staining, collagen birefringence was better (P = 0.42, Fig. 4C) and the area of type-1 collagen was wider (P = 0.74, Fig. 4D) in PRP-treated groups than in the MC control group.

Histologic analysis. Sample images from H&E, Safranin-O, and Picrosirius Red staining of the MC group [(b) and (c), respectively], initial PRP group [(e) and (f), respectively], and concentrated PRP group [(h) and (i), respectively] (A). The area of fibrocartilage was calculated based on Safranin-O staining (B), and birefringence was calculated based on Picrosirius Red staining (C). The area of collagen 1 was also measured (D). Picrosirius Red staining area is a magnification of the box in the H&E stains. The collagen appears better organized in the initial and concentrated PRP groups; however, the amount of fibrocartilage and collagen produced was not significantly different between three groups. A: Bar, 100 µm; B, C, and D: values shown as average ± SD. H&E: hematoxylin and eosin; MC: methylcellulose; PRP: platelet-rich plasma.
Discussion
This study revealed that the combination of concentrated PRP with tHG and ADSC biomechanically improved healing in a chronic rotator cuff injury and repair model compared to a 2% MC control. When assessing yield load, both groups with platelet augmentation demonstrated higher plasticity. Despite no significant difference in histologic parameters, the histology and higher organization of the collagen fibers likely influenced the biomechanical results. However, a dose-dependent effect of platelet augmentation was not clearly demonstrated.
To determine whether the addition of PRP enhances healing at the TBI in a chronic rat rotator cuff injury model, the current data were compared with our prior studies. As shown in Fig. 5, load to failure, stiffness, and yield load of initial and concentrated platelet augmentation groups were nearly comparable to those of repair with ADSC-seeded tHG, ADSC alone, and tHG alone6,8, demonstrating no significant advantage of PRP-augmented, ADSC-seeded tHG over tHG alone. Similarly, in histological assessment and bone quality, the results from the current study were almost identical to the result of previous studies. Thus, a synergistic effect of PRP addition to ADSC-seeded tHG on biomechanical parameters, histology, and bone quality was not demonstrated.

Comparison of biomechanical parameters in current study and prior studies. Load to failure (A) and stiffness (B) and yield load (C) of the initial and concentrated PRP groups (tHG + ADSCs + PRP) from the current study were compared with the data from treatment of the TBI after tendon repair with tHG alone, ADSC alone, and ADSC-seeded tHG from our prior studies. There were no significant differences in the biomechanical parameters among these five groups demonstrating no synergistic effect of PRP. ADSC: adipose-derived stem cell; PRP: platelet-rich plasma; tHG: type-1 collagen-rich tendon hydrogel; TBI: tendon-bone interface.
MC was selected as a control due to comparable viscosity to tHG. MC is chemically stable at room temperature and has been used as vehicle to apply a variety of drugs, and some of its analogs have been used as control substances. MC has been reported to be inert with no effect on tendon healing or adhesions 29 . Therefore, MC is a more ideal control for examining the specific effect of our hydrogel on TBI healing rather than saline. In biomechanical testing, MC group values were comparable to historical saline values in our prior work. Therefore, the viscosity of the injected substance itself did not contribute to the positive effect on TBI healing seen biomechanically.
In clinical cases, the use of PRP remains controversial. One of the reasons for this controversy is that there is confusion about the term “PRP.” PRP has been classified into 4 subgroups,30,31 and many articles do not specify which subgroup was used. In those that do, the subgroup of platelet concentrate and its effect on rotator cuff repair is inconsistent between studies3,10,12–14. Despite using pure (leukocyte-poor) PRP in the current study, which is considered as the most effective platelet concentrate among the 4 subgroups5,31, a synergistic effect of PRP was not demonstrated.
We previously reported the synergistic effect of platelet concentrate and ADSC mixed with tHG on healing of rat acute Achilles’ repair model and Achilles’-tendon-calcaneus tendon-bone grafting model7,32. Similarly, Han et al. reported the synergic effect of combined therapy of mesenchymal stem cell and PRP on rotator cuff repair 33 . One main difference between this study and prior studies was the use of a chronic model in this study and the use of acute injury and repair models in other studies. Platelet concentrates contain growth factors and promote angiogenesis, cell proliferation, and cell migration 34 . ADSCs also excrete growth factors. As previously reported, PRP promotes growth factor secretion, migration, and proliferation of stem cells32,34. In addition, Han et al. reported the gene for collagen 1 expression also increased in vitro 33 . Therefore, the combination of platelets and ADSCs potentially promotes the regeneration of collagen 33 and affects the first 2 phases of healing: inflammation and proliferation 7 . In contrast to Han et al. who only use PRP and stem cells, we used type-1 collagen-rich hydrogel with PRP and ADSCs. Therefore, the effect caused by PRP and ADSC might be masked by tHG, which already has rich type-1 collagen and provides an excellent supportive environment for host cells. In a chronic model, some areas of injury are renewed by the surgical procedure; however, some areas, like the tendon, are scarred and retracted and may not be as affected by platelet augmentation. In addition, the positive effect of platelet augmentation seen at short-term evaluation disappeared in long-term evaluations in several clinical case series15,35,36. Therefore, beside the possibility that there was no synergic effect of platelet concentrates, there is also a possibility that a synergy between platelets and ADSC effects might have been seen at earlier time points. Furthermore, we previously reported the synergic effect of PRP with ADSC-seeded tHG on the repair between tendon and tendon or bone and bone, which are tissues with similar moduli7,32. On the other hand, the TBI, or enthesis, is composed of a transition between tendon, fibrocartilage, calcified fibrocartilage, and bone. Therefore, the increase of type-1 collagen may be more effective on repair of tissues with similar moduli rather than the TBI.
We used two concentrations of platelet augmentation to determine whether there was a dose-dependent effect of PRP augmentation. Weibrich et al. warned that exceedingly highly concentrated platelets seemed to have a negative effect caused by the inhibitory and cytotoxic effect of growth factors 37 . After mixing the platelets with the tHG in this study, the concentration of platelets in the concentrated group was within the optimal range advocated by Weibrich et al. While we did not demonstrate a dose-dependent effect of platelet concentration, a higher platelet concentration tended to demonstrate improved TBI healing when compared to MC for several biomechanical properties. A dose-dependent effect may be revealed by comparing PRP across 3 or more higher concentrations.
There were limitations to this study. First, commercially purchased allogenic PRP was used in this study, which may cause an immune reaction, and the positive effect of platelet concentrate might wane. However, allogeneic PRP is in line with other studies due to the limited source of the whole blood in a small animal.9,12,38 Second, in Fig. 5, we compared data from this study to those of our prior studies to determine the potential synergistic effect of PRP with tHG and tHG with ADSCs. While all study groups could have been repeated for this study, ethically adhering to the NIH principles of replace, reduce, and refine, this would be inappropriate. We used the same animal facilities, testing conditions, and research team to control for parameters between studies. In addition, we used only 1 rat for an intact shoulder sample. Because this was used for gross histology and not biomechanical comparison, we felt this was appropriate.
In conclusion, the addition of highly concentrated PRP to ADSC-seeded tHG improved TBI healing after chronic rotator cuff injury and repair in rats compared to control. However, a synergistic effect of platelet augmentation was not demonstrated compared to prior studies examining ADSCs and tHG alone. In addition, a dose-dependent effect of platelet concentrate was not clearly detected. Further investigation is needed to reveal if platelet augmentation influences early TBI healing after chronic rotator cuff injury and repair.
Supplemental Material
sj-tif-1-sco-10.1177_09636897231190174 – Supplemental material for Assessment of a Synergistic Effect of Platelet-Rich Plasma and Stem Cell-Seeded Hydrogel for Healing of Rat Chronic Rotator Cuff Injuries
Supplemental material, sj-tif-1-sco-10.1177_09636897231190174 for Assessment of a Synergistic Effect of Platelet-Rich Plasma and Stem Cell-Seeded Hydrogel for Healing of Rat Chronic Rotator Cuff Injuries by Hiroki Oda, Yukitoshi Kaizawa, Austin Franklin, Uriel Sanchez Rangel, Hunter Storaci, Jung Gi Min, Zhen Wang, Geoffrey D. Abrams, James Chang and Paige M. Fox in Cell Transplantation
Supplemental Material
sj-tif-2-sco-10.1177_09636897231190174 – Supplemental material for Assessment of a Synergistic Effect of Platelet-Rich Plasma and Stem Cell-Seeded Hydrogel for Healing of Rat Chronic Rotator Cuff Injuries
Supplemental material, sj-tif-2-sco-10.1177_09636897231190174 for Assessment of a Synergistic Effect of Platelet-Rich Plasma and Stem Cell-Seeded Hydrogel for Healing of Rat Chronic Rotator Cuff Injuries by Hiroki Oda, Yukitoshi Kaizawa, Austin Franklin, Uriel Sanchez Rangel, Hunter Storaci, Jung Gi Min, Zhen Wang, Geoffrey D. Abrams, James Chang and Paige M. Fox in Cell Transplantation
Footnotes
Authors’ Contributions
A substantial contribution to the concept or design of the work, or acquisition, analysis or interpretation of data: all authors. Drafting the article or revising it critically for intellectual content: H.O., Y.K., U.S.R., H.S., G.D.A., and P.M.F. Approval of the version to be published: H.O., Y.K., A.F., U.S.R., J.G.M., H.S., Z.W., G.D.A., J.C., and P.M.F. All the co-authors have read and approved the final submitting manuscript.
Availability of Data and Material
All the data collected or analyzed in this study and the materials used in this study are included in the main text, tables, and figures.
Ethical Approval
The protocol of this work was approved by VA Palo Alto Health Care System (VAPAHCS) Institutional Animal Care and Use Committee (IACUC) (Approval #FOX1616).
Statement of Human and Animal Rights
This article does not contain any studies with human subjects. All animal studies were carried out under IACUC approval.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GDA had relevant financial activities outside the submitted work (Sparta Biomedical: stock options, Orthofix Inc.: research support, AxGen Inc.: stock options, Cytonics Inc.: stock options, and Arthrex: educational support).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: One or more of the authors (J.C., P.M.F.) has received funding. This work was supported by the United States Department of Veterans Affairs Rehabilitation Research and Development Service (J.C.) and American Foundation for Surgery of the Hand (P.M.F.) (grant #1211). J.C. has received funding from the United States Department of Veterans Affairs Rehabilitation Research and Development Service and had intellectual property related to the work (Tendon Hydrogel).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
