Abstract
Obesity has been linked to cognitive impairment through systemic low-grade inflammation. High fat and sugar diets (HFSDs) also induce systemic inflammation, either by induced Toll-like receptor 4 response, or by causing dysbiosis. This study aimed to evaluate the effect of symbiotics supplementation on spatial and working memory, butyrate concentration, neurogenesis, and electrophysiological recovery of HFSD-fed rats. In a first experiment, Sprague-Dawley male rats were given HFSD for 10 weeks, after which they were randomized into 2 groups (n = 10 per group): water (control), or Enterococcus faecium + inulin (symbiotic) administration, for 5 weeks. In the fifth week, spatial and working memory was analyzed through the Morris Water Maze (MWM) and Eight-Arm Radial Maze (RAM) tests, respectively, with 1 week apart between tests. At the end of the study, butyrate levels from feces and neurogenesis at hippocampus were determined. In a second experiment with similar characteristics, the hippocampus was extracted to perform electrophysiological studies. Symbiotic-supplemented rats showed a significantly better memory, butyrate concentrations, and neurogenesis. This group also presented an increased firing frequency in hippocampal neurons [and a larger N-methyl-
Introduction
Obesity has increased dramatically in recent decades, leading to a pandemic situation. Obesity increases the risk of several diseases such as type 2 diabetes, cardiovascular disease, hypertension, and even neurodegenerative diseases, such as cognitive impairment 1 .
Both obesity and dietary patterns associated with increased weight have been linked to cognitive impairment, probably due to low-grade inflammation1,2. As adipocytes increase in size, they recruit macrophages, which induce insulin resistance and trigger inflammation by releasing pro-inflammatory cytokines such as tumor necrosis factor-alpha (TNF-α) and interleukin (IL)-6 3 .
As obesity leads to systemic inflammation, it also induces microglial activation and inflammation in the hippocampus, leading to gliosis and inhibiting long-term potentiation (LTP) and impairing neurogenesis 4 . It also promotes the production of beta-amyloid, increasing the risk of both cognitive impairment and Alzheimer’s disease (AD) 5 .
High-fat diets have also been linked to cognitive impairment, as they stimulate lipopolysaccharide (LPS) receptor—Toll-like receptor 4 (TLR4)—on immune cells, initiating an inflammatory cascade 6 . They also change the gut microbiota composition, leading to dysbiosis. Microbiota composition of obese patients is different from non-obese ones and is associated with impaired scores in memory tests 7 . It is now believed that dysbiosis may play a role in cognitive impairment through the “microbiota-gut-brain axis,” as it is also associated with systemic inflammation 8 .
As gut microbiota may be one of the linkers between obesity and cognitive impairment, targeting microbiota with symbiotics may be a possible therapeutic approach. Therefore, in this proof-of-concept study, we aimed to evaluate the supplementation with a symbiotic composed of Enterococcus faecium (E. faecium) and agave inulin in memory and cellular recording of obese high–fat-sugar-diet (HFSD)-fed rats. Previous studies have already demonstrated that this symbiotic is capable of improving cognition of middle-aged rats 9 .
E. faecium is a lactic-acid bacteria (LAB), that is a normal microorganism mainly found in the gastrointestinal tract; it is also found in human milk and is used in the production of various traditional fermented food products10,11. Evidence has shown that the use of E. faecium as a probiotic is safe 10 , and reduces human and animal intestinal inflammation 12 . This probiotic has also cholesterol-lowering abilities and produces bacteriocins against pathogenic bacteria such as Staphylococcus aureus and Vibrio cholerae, and shows immune-stimulation actions11,13. E. faecium has shown to attenuate obesity in mice administered a high-fat diet, as well as reduced endotoxemia levels (by reduction in LPS) and associated inflammation (by inducing an anti-inflammatory effect and an attenuation of macrophage pro-inflammatory responses) 14 .
Agave inulin is a fructo-oligosaccharide prebiotic. Studies performed by Hoffman JD, et al (2019) revealed that higher inulin interventions mitigate neuroinflammation. Inulin leads to an increased microbial metabolism via gut–brain axis components, enhanced glycolytic metabolism in the periphery, and reduced hippocampal inflammatory gene expression in asymptomatic APOE4 (the strongest genetic risk factor for AD) transgenic mice 15 . Inulin is fermented into short-chain fatty acids (SCFA) such as butyrate, which has been related to improvement in intestinal tight-junctions primarily in the colon, an increased production of molecules such as brain-derived neurotrophic factor (BDNF) associated with memory consolidation (by inhibiting histone deacetylase inhibitors)16,17. Inulin has also shown to improve episodic memory and improve recall performance in humans 18 .
Materials and Methods
Animals
Twenty male, 2-month-old Sprague-Dawley rats of 250 g body weight were used. These animals were supplied by the animal breeding center of the Health Sciences Faculty, Anáhuac University. They were housed in acrylic cages under controlled conditions of 22°C ± 2°C, and 12-h light-dark cycle.
Study Design
In order to induce obesity, the rats were fed with HFSD consisting of commercial pellets (Nutricubos 501137, Purina®, México), which were previously immersed in lard at a ratio of 1:0.7 for 6 h at 60°C; the processed pellets had the following nutritional content for 100g: 546.3 ± 0.01 kcal with 21.00% ± 0.12% of fat . The sugar was given as a 30% sucrose solution in the drinking water. This diet was given to the rats for 10 weeks before the supplementation of the symbiotic and was continued for another 5 weeks throughout all the supplementation period. All the procedures were performed in accordance with the National Institutes of Health Guide for the care and use of laboratory animals. In the same way, the Mexican Official Norm on Principles of Laboratory Animal Care (NOM 062-ZOO-1999) was considered. The Animal Bioethics and Welfare Committee of the Health Sciences Faculty at Anahuac University approved all animal procedures (ID: 201850). The experiments of this study were designed and reported according to the ARRIVE guidelines. At the end of the study, the animals were euthanized with intraperitoneal sodium pentobarbital.
To explore the effect of the symbiotic on the cognitive function of obese rats, we performed two experiments. The first investigated the effect in memory, butyrate levels, and neurogenesis. The second one studied the electrophysiological effects of the symbiotic in the brain.
In the first experiment, we evaluated the effect of the symbiotic on spatial memory using the Morris Water Maze (MWM) behavioral test, and on working and reference memory using the Eight-arm Radial Maze (RAM) test. As this was a proof of concept study and the effect of the symbiotic has already been explored on cognitive impairment, we decided to perform a preliminary series of experiments evaluating only two groups. For this purpose, two groups of 10 rats each were randomly formed using the GraphPad QuickCalcs program (http://www.graphpad.com/quickcalcs/)—Group 1: control group, to which the vehicle was administered (water at 2 ml/kg) and Group 2: E. faecium + agave inulin were administered. The sample size for this experiment was calculated using an alpha of 0.05 and a beta of 0.20. At the end of this experiment, a stool sample was randomly obtained from 5 animals (in both groups) to determine butyrate concentrations. Upon termination of the follow-up period in this experiment (6 weeks), 10 rats were used for neurogenesis analysis at hippocampus (n = 5 per group).
Based on the results of the first study, we carried out a second experiment. In this case, we also formed two groups as described previously, but only with five rats each one: Group 1, water (control) and Group 2, E. faecium + agave inulin (symbiotic). Herein, we evaluated the electrophysiological responses of hippocampal neurons located in area CA1, which include evaluation of the induction and expression of LTP at the Schaffer Collaterals and the assessment of pyramidal of pyramidal cells' membrane properties.
In both experiments, rats were fed with HFSD for 10 weeks before the supplementation with the symbiotic and then continued for 5 weeks throughout all the supplementation period.
Symbiotic Administration
After the 10 weeks obesity induction, animals were supplemented through oral gavage. The control group received water (vehicle, 2 ml/kg of body weight), and the experimental group received a symbiotic consisting of E. faecium (4 x 108 colony forming units, CFU/dose) and agave inulin (860 mg/kg) contained in 2 ml of water/kg of body weight. The treatment was administered daily for 5 weeks. In the fifth week, as we continued to supplement the rats, both spatial and working memory tests were performed (with 1 week apart between tests). For the rat electrophysiology groups, we supplemented them for 5 weeks prior to euthanasia 9 .
Spatial Memory
Spatial memory was assessed using the MWM, which consists of a circular pool with a hidden platform submerged 2 cm below water surface in southwest (SW) quadrant, that has to be found by the animal. Three visual cues were positioned equidistant above the water level. In the acquisition (training) phase, four trials were assessed by animal for 5 consecutive days; on each day, the animal was introduced into the pool from four different starting points that change in order through the training period. Each animal had 1 minute to get to the platform, with a 20-s interval between trials. On the sixth day, a final trial was carried out to assess memory retention. During this phase, the platform was removed and the animals had 1-min to swim around the pool 9 . All trials were video tracked by means of a computerized system (Smart v3.0.02 Panlab Harvard Apparatus®). Escape latency and time spend on each quadrant were analyzed.
Working and Reference Memory
Working and reference memory were assessed using the RAM test. This test consisted of an eight arm maze in which food is placed for the rodent to eat. The food is placed in 4 of the arms, hidden from the rodents view; the other 4 arms have food that cannot be accessed by the rodent, to avoid finding the reachable food by smell. The objective of the test is to evaluate both working and reference memory at the same time, as the rats must find the arms with food using extra-maze cues, avoiding the arms previously used were they already ate the food (working memory) as well as avoiding the arms with no reachable food on them (reference memory). Re-entering into those arms indicate memory deficit 19 . The results are reported as percentage of correct answers, dividing the number of correct entries in the arms with food, by all the entries in the other arms or repeated arms.
Fecal Butyrate Concentrations
For the first experiment in which we quantified butyrate levels butyric acid concentrations were determined from feces. For this purpose, feces were collected and then frozen at -80°C until measurement. Fecal samples were then lyophilized and rehydrated in distilled water. After pH was adjusted (between 2 and 3 with 5M HCl), 1 ml chloroform was added, and samples were centrifuged for 25 min at 4,000 × g. Afterward, the concentration of butyric acid was determined by gas chromatography using a GC2010 apparatus (Shimadzu, Japan). In this procedure, a DB-1701 capillary column with 30 m length, 0.25 mm inner diameter, and 0.25 mm film thickness (Agilent, Santa Clara, CA, USA) was used. The conditions for performing this procedure were as follows—oven: 250°C, split ratio: 20, colum: 95°C (Isothermal), FID: 300°C, make up gas helium: 4 ml/min, H2 flow: 40 ml/min, airflow: 400 ml/min, retention time butyric acid: 3.5–3.6 min. For quantification of SCFA, an external standard (SupelcoTM WSFA-1 Mix, Supelco Sigma Aldrich Co., Bellefonte PA, USA) was used. Results were reported as nanograms/dry weight.
Immunofluorescence
In the first experiment, we also assessed neurogenesis by using a double stain with 5-bromo-2’-deoxyuridine (BrdU) and doublecortin (Dcx). BrdU incorporates into active post mitotic cells and Dcx is a marker for neural precursor cells (NPC). Therefore, BrdU+/Dcx+ cells were considered new formed neurons. In this way, animals were evaluated at the end of follow-up. Three days before euthanasia, rats were injected with BrdU (Roche, 50 mg/kg; IP) intraperitoneally every 12 h for five doses. Brains were fixed by paraformaldehyde perfusion. Coronal cuts of subventricular (SVZ) and subgranular (SGZ) zones were obtained by cryosectioning (three slices 40 µm thick with 200 µm spacing, a total of 9 sections per animal were counted) and placed on slides using the free float method. Afterwards the slides were washed twice for a period of 10 min with phosphate-buffered saline + Triton (PBT) and incubated with ImmunoRetriever for 30 min at 65ºC. Subsequently, the samples were subjected to two washes of 5 min with phosphate-buffered saline (PBS) and then again incubated with 1N HCl at 37ºC. Afterwards, the slides were incubated for 10 min with sodium borate 0.1 M and then washed three times with PBT. Unspecific binding sites were blocked with standard blocking solution for 30 min. The primary antibodies against BrdU (Roche, mouse IgG, 1:250) and Dcx (EMD-Millipore, 1:250) were incubated for 16 h overnight. After incubation, slides were washed three times for 10 min with PBT, and the secondary antibodies (BrdU: donkey IgG; Dcx: rabbit IgG all at 1:500 from Invitrogen) were incubated for 2 h. PBT washes eliminated the excess of antibodies. Slides were counterstained with DAPI. The total number of cells in SVZ and SGZ were quantified by a blinded evaluator using cell counting software (Image-Pro Plus, Media Cybernetics, USA). The total number of BrdU+/Dcx+ cells was obtained by averaging the total number of cells from all nine slides. Photomicrographs were taken in a confocal microscope (Zeiss LSM 800).
Electrophysiological Studies
For the second experiment, we evaluated electrophysiological responses of hippocampal neurons located in area CA1 through various procedures, as follows.
Brain slice preparation
The procedures were performed following the Mexican Official Norm for the use and care of laboratory animals (NOM-062-ZOO-1999) and were approved by the Ethics Committee for Animal Research (CICUAL) of The Center for Research and Advanced Studies of the National Polytechnic Institute (CINVESTAV; Protocol number 0090-14). Animals were deeply anesthetized with pentobarbital (50 mg/kg−1, intraperitoneal) and decapitated. Brains were exposed and rapidly placed for 2–3 min in an ice-cold sucrose solution containing (in mM): 210 sucrose, 2.8 KCl, 2 MgSO4, 1.25 Na2HPO4, 26 NaHCO3, 6 MgCl2, 1 CaCl2, and 10 D-glucose with pH = 7.20-7.30 and saturated with carbogen mixture (95% O2 /5% CO2). Subsequently, hemispheres were separated through the midsagittal line, and the resulting blocks of tissue were glued to the plate of a VT1000S vibratome (Leica, Nussloch, Germany) to obtain sagittal hippocampal slices (385 mm thick). The resulting slices were transferred and maintained 30 min at 33°C ± 2°C in an incubation solution composed of (in mM): 125 NaCl, 2.5 KCl, 1.2 Na2HPO4, 25 NaHCO3, 2 MgCl2, 1 CaCl2, and 10 D-(þ)-glucose; pH = 7.20–7.30 and continuously bubbled with the carbogen mixture. After incubation, the slices were stabilized at room temperature for at least 1 h. Individual slices were transferred to a submersion recording chamber (total volume 400 µl) 10 min before the beginning of the experiments. The slices were maintained at a constant flow rate (3.5–4 ml/min-1) and perfused with a standard artificial cerebrospinal fluid (ASCF) solution containing (in mM): 125 NaCl, 3 KCl, 12.5 Na2HPO4, 250 NaHCO3, 2.5 CaCl2, 1.5 MgCl2, and 10 glucose; maintained at 33°C ± 2°C with the help of an inline solution heater coupled to a single-channel temperature controller (TC-324C, Warner Instruments).
Patch-clamp whole-cell recordings
CA1 pyramidal cell bodies were localized 50–150 µm from the slice surface and identified with infrared video microscopy and differential interference contrast optics coupled to a Nikon FN1 microscope. The patch pipettes were pulled from borosilicate glass with a micropipette puller (P57, Sutter Instruments, Novato, CA, USA) with a tip resistance of 4–7 MΩ and were filled with an intracellular solution containing (in mM): 135 K+-gluconate, 10 KCl, 5 NaCl, 1 EGTA, 10 HEPES, 2 Mg2+-ATP, 0.4 Na+-GTP, 10 phosphocreatine, and pH = 7.20–7.28. Current- and voltage-clamp recordings were obtained with an Axopatch 200B amplifier (Molecular Devices, San José, CA, USA), digitized and sampled at 10 kHz, and filtered at 5 kHz with a Digidata 1440A (Axon Instruments, Palo Alto, CA, USA). The off-line analyses were performed with pCLAMP10.6 software (Molecular Devices).
Measurements of intrinsic membrane properties and firing frequency
The resting membrane potential (RMP) was measured after the initial break-in from gigaseal to whole-cell configuration. Once the RMP was determined and stabilized, the cells were switched to current-clamp mode, and a series of negative and positive current steps (1 s, 30 pA increments, starting from -300 pA) were injected to determine the input resistance (RN), membrane time constant (tau) and the rheobase current required to evoke at least one action potential (AP). RN was determined as the slope of a lineal function (f(x)=mx+b) fitted to the current-voltage relationship around RMP; the membrane time constant was calculated by fitting a mono-exponential equation
Action potential kinetic analysis
Action potential (AP) threshold was calculated using the third derivative method, as previously described 20 . The AP kinetic properties were determined with phase plots, which were constructed plotting the first derivative of the membrane potential dV/dt (mV.ms-1) against the membrane potential, as previously reported 21 .
Synaptic currents measurements
Excitatory postsynaptic currents (EPSCs) were evoked with a bipolar stimulation electrode placed in the stratum radiatum of CA1, ≈300 µM away from the recording site. Electrical stimulation was delivered with a frequency of 0.1 Hz. The recording of the EPSC was performed in voltage-clamp mode in the presence of picrotoxin (PTX, 50 µM). After 5 min of a stable baseline recording, the cell was held at −70 mV to evoke AMPAR-mediated EPSC. After that, the holding potential was slowly switched to +40 mV to evoke N-methyl-
Extracellular recordings
Individual slices were transferred to an extracellular submersion recording chamber (total volume 300 µL) and perfused with artificial ACSF (constant flow rate 2.5 ml/min-1). The composition of the ACSF was identical to the one used in the patch-clamp whole-cell experiments. The temperature in the recording chamber was maintained at 32°C ± 2°C with a single-channel temperature controller (TC-324C, Warner Instruments). After 10 min of slice stabilization, a nichrome bipolar stimulation electrode (38 μM bare diameter) was placed in the stratum radiatum (see Fig. 6A), and field excitatory postsynaptic potentials (fEPSPs) were acquired with a recording pipette. The recording pipette had a tip resistance of 1–2 MΩ when filled with 3 M NaCl solution. The stimulation frequency for test stimuli was 0.066 Hz. The stimuli consisted of paired monopolar pulses (100 µs duration) with 60 ms interstimulus interval. According to input-output (I-O) curves carried out previously, a current intensity ≈100–200 µA evoked 30%–50% of maximal fEPSP response.
Test stimuli were delivered with a high-voltage isolator unit (A365D; World Precision Instruments, Sarasota, FL, USA) and controlled with a Master-8 pulse generator (AMPI, Israel). A Dagan BVC-700A amplifier (Minneapolis, MN, USA) coupled to an extracellular headstage (Dagan 8020) was used to amplify and high-pass filter (0.3 Hz) the analog responses. Additionally, a Humbug noise eliminator (Quest Scientific Instruments, North Vancouver, BC, Canada) was used to suppress any electrical noise. Recorded responses were displayed on a PC-based oscilloscope and digitized for storage and off-line analysis with LabVIEW 7.1 software (National Instruments, Austin, TX, USA).
Synaptic stimulation
Bidirectional plasticity experiments were carried out in the same acute slice with intact GABAergic inhibition. After the slice stabilization in the recording chamber, a 25-min baseline of fEPSPs was acquired with a stimulation frequency of 0.066 Hz. Next, 900 pulses were delivered to the Schaffer collaterals at 1 Hz to induce long-term depression (LTD) and fEPSP responses were recorded for additional 60 min. After this, the current intensity was adjusted to match similar baseline fEPSP values, and a second baseline of fEPSPs was acquired for 15 min. Then, two trains of HFS (100 pulses at 100 Hz, repeated twice with a 10 s interval) was delivered to the Schaffer collaterals, and the fEPSP responses were recorded for additional 60 min.
Determination of the magnitude of synaptic potentiation
The fEPSP responses were normalized to baseline values. The normalized responses were expressed as mean ± S.E.M. The magnitude of LTD or LTP was calculated by comparing fEPSP values from the last 5 min of recording (minute 56–60 for LTD or minute 131–135 for LTP) baseline values. The magnitude of post-tetanic potentiation (PTP) was calculated by comparing fEPSP values from the first minute post-HFS vs. baseline value. Wilcoxon (dependent groups) or Mann-Whitney (independent groups) tests were used to compare the means between groups. A P value < 0.05 was considered statistically significant.
Drugs used for electrophysiological studies
All chemicals and drugs were obtained from Sigma-Aldrich Chemicals Co. (St. Louis, MO, USA) and freshly prepared directly in the required solution.
Statistical Methods
Statistical analysis was performed using Prism 5 software (Prism 5.01, GraphPad Software Inc., San Diego, CA, USA). Data are expressed as mean ± standard error of the mean (S.E.M.). All data sets were analyzed for normality using Shapiro Wilk normality test. MWM evaluations were analyzed using two-way analysis of variance (ANOVA) and Mann-Whitney U post-hoc test for escape time, and Student’s t test for consolidation phase. Working memory was evaluated using two-way ANOVA. Butyrate concentration was analyzed using Mann Whitney U test mean while unpaired Student’s t test was used to compare neurogenesis results. A P value < 0.05 was considered statistically significant. To calculate the size of the effect on spatial memory evaluation, we used Rosenthal’s r, an effect size test for data that are not normally distributed. This test can be used alongside Mann Whitney U-test results 22 .
For the whole-cell patch clamp recordings, group measures are numerically expressed as mean ± SEM. The box plots show the median, mean, first and third quartiles plus 10%–90%, and the 5th and 95th percentiles. All statistical comparisons were performed between control values (obtained from obese animals in the control group) and those obtained from animals treated with symbiotics (experimental group). The Gaussian distribution of data was validated with the Kolmogorov–Smirnov test (P = 0.05). Comparability among experimental groups was evaluated using a two-tailed unpaired Student’s t-test or Mann-Whitney U test for independent groups. Two-way analysis of variance (ANOVA) followed by a Holm–Šidák post-hoc test was used for multiple comparisons among dependent groups, as appropriate. For all experiments, a P value <0.05 was considered statistically significant. All fits were performed using Clampfit 10.7 (Molecular Devices).
Results
Supplementation With E. Faecium and Agave Inulin Has No Effect in Bodyweight in Rats Fed With a High Fat-Sugar Diet
All rats were weighed at the onset and at the end of the experiment; no difference was observed between groups. Weight before intervention was 396 ± 4.54 g in the control group; 394 ± 9.27 g in the symbiotic group. The final weight (after intervention) in the control group was 633 ± 11.74 g, while the one in the symbiotic group was 631 ± 10.92 g (P > 0.05; Student’s t-test; Fig. 1).
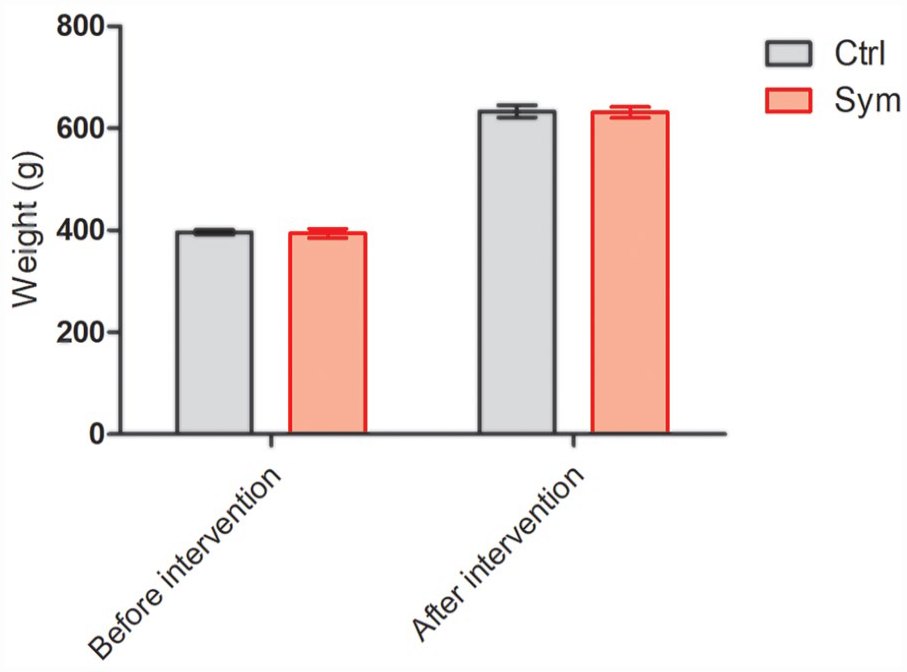
Weight before intervention and after the 5 weeks of symbiotic or placebo supplementation. Results are presented as mean ± SEM; n = 10; Student’s T test (ns). SEM: standard error of the mean.
Supplementation With E. Faecium and Agave Inulin Enhances Spatial Memory in Rats Fed With a High Fat-Sugar Diet
Obesity has shown to impair cognitive functions, especially memory. Thus, we conducted a task to evaluate spatial memory in obese rats fed with a HFSD. During the acquisition phase, the results confirmed that spatial memory is quite poor in obese rats. After all acquisitions, the control group did not decrease the time to get to the hidden platform. On the contrary, we observed an increased time to get to the hidden platform, showing no spatial memory consolidation. On the other hand, the symbiotic-supplemented group presented a decrease in the time to get to the hidden platform, showing a significant difference on day 4 of acquisition (Escape latency: control = 37.38 ± 9.04 s, mean ± SD; symbiotic = 11.46 ± 3.75 s; P = 0.0117, Mann Whitney U test; Fig. 2A). To know the effect size (the clinical efficacy of the treatment), the Rosenthal’s r was calculated. In this case, a large size effect was obtained in the spatial memory of the symbiotic group (r = 0.51).

Results from the MWM test. (A) Escape latency time (seconds) from starting point in the South East (SE) quadrant and platform in Southwest (SW) quadrant shows a decrease in time in the symbiotic group, whereas the control group increased the escape latency time. Mann Whitney U test; *P = 0.0117. (B) Path length to platform (m) measured during the last day (5th) of the acquisition test. Mean ± SEM, n = 10, Student’s T test; **P = 0.02. (C-D) Illustrative images of rats trajectories in MWM during last days’ trials of study groups: control (C), symbiotic (D). SEM: standard error of the mean.
We also analyzed the path length to platform. Interestingly, the symbiotic-supplemented group showed a significant lower distance to the platform (14.37 ± 3.95 vs 44.26 ± 13.47 m; symbiotic and control group, respectively; P = 0.02, Student’s t-test; Fig. 2B). Representative pads of the last day of acquisition phase for each group are illustrated in Fig. 2C, D.
Supplementation With E. Faecium and Agave Inulin Does Not Affect Working Memory in Rats Fed With a High Fat-Sugar Diet
To complement the cognitive study, 1 week after the MWM test we carried out another test that analyzes working memory, as it is also reported to be affected in obesity 7 . No difference was observed between the groups regarding the percentage of correct responses (control: 42.85% ± 4.14%, mean ± SD; symbiotic: 39.52 ± 4.77 s; Student’s t-test; P > 0.05; Fig. 3A) or the time to complete the task (P > 0.05, two-factor ANOVA for repeated measures; final values of time on day 5: control: 2.29 ± 0.39 min; symbiotic: 2.57 ± 0.45 min; Fig. 3B).
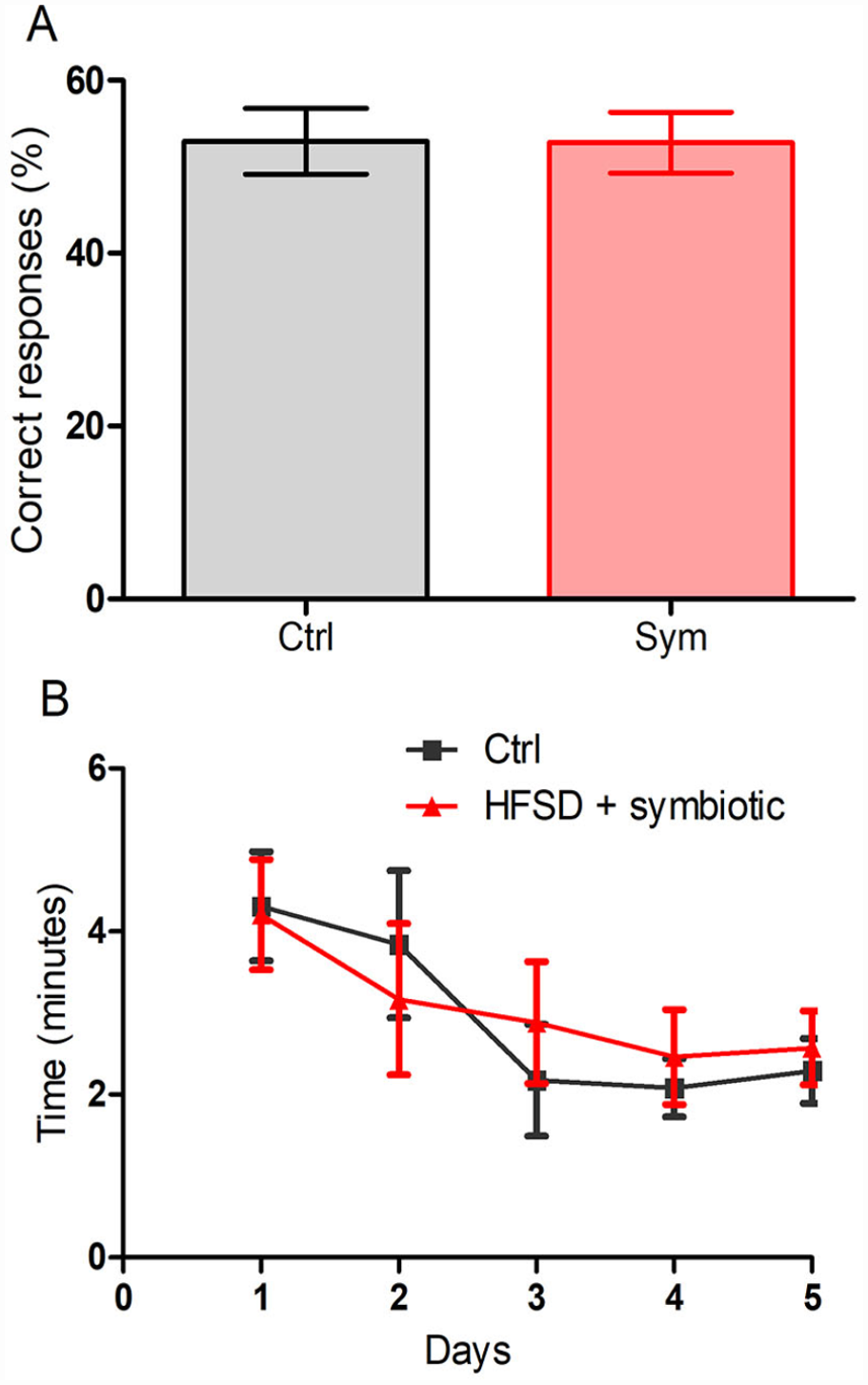
Results from Eight-arm Radial Maze. (A) Percentage of correct responses (%) which represent working and reference memory, after 5 days of acquisition. Results are presented as Mean ± SEM; n = 10; Student’s T test (ns). (B) Time (min) taken to complete the task. Two-way ANOVA for repeated measures followed by post hoc Bonferroni; P > 0.05. SEM: standard error of the mean.
Symbiotic Supplementation Induced a Significant Increase in Butyrate
Previous investigations have already reported a significant increase in butyrate after symbiotic supplementation 9 . As butyrate is an anti-inflammatory postbiotic that could influence inflammation, we measured its concentration in fecal samples of the studied animals. The results showed a significant increase in the concentration of butyrate after symbiotic supplementation (68.56 ± 5.9, ng/dry weight; mean± SD) as compared to that observed in the control group (48.27 ± 2.3, P = 0.05, Mann Whitney U test; Fig. 4).
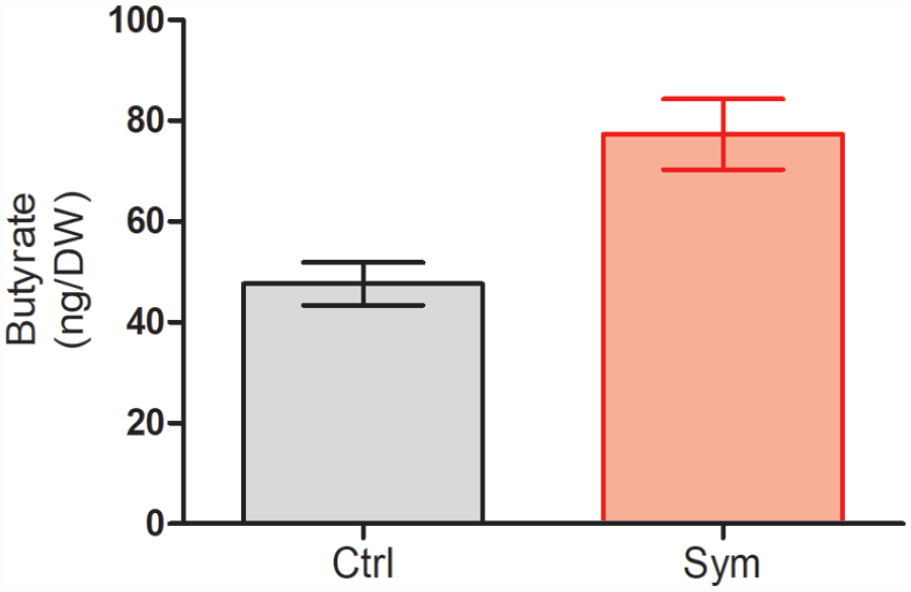
Butyrate concentrations in fecal samples. Results are presented as Mean ± SD; n = 5; Mann Whitney U test; P = 0.05.
Symbiotic Supplementation Increased Neurogenesis in the SVZ and SGZ of Hippocampus
Cognitive impairment could be induced by the loss of neurons in the hippocampus and thus, the induction of neurogenesis could result in memory improvement. Since E. faecium is capable of increasing BDNF expression and this molecule can potentially improve neurogenesis 9 , we decided to asses this parameter in studied animals. As is shown in Fig. 5, symbiotic-supplemented animals exhibited significantly more neurogenesis in the SVZ (Fig. 5A, Symbiotic vs. Control: 380 ± 30 vs. 110 ± 10; mean ± SD, P < 0.0001; Student’s t-test) and SGZ (Fig. 5B, 2500 ± 135 vs. 1064 ± 95; P = 0.03; Student t-test) as compared to control group.

Effect of the symbiotic supplementation on neurogenesis. (A) Neurogenesis in the subventricular zone (SVZ); P < 0.0001. (B) Neurogenesis in the subgranular zone (SGZ); P = 0.03. Results are presented as Mean ± SD; n = 5; Student’s t test.
Symbiotic Supplementation Modifies Biophysical Properties and Output of CA1 Pyramidal Cells
Because symbiotic supplementation improves cognitive performance in the spatial memory MWM test of obese rats, we next evaluated the effects of symbiotic supplementation on the electrophysiological properties of hippocampal neurons. Patch-clamp whole-cell recordings were performed on CA1 pyramidal cells (CA1 PCs) from dorsal hippocampal brain slices (see methods for details) (n = 15 PCs / 5 animals of each experimental group). Fig. 6A shows representative voltage responses to somatic current injection. From the responses, we determined that RMP is more depolarized in the symbiotic-supplemented cells compared to control ones (RMP in control = -74.16 ± 0.73 mV; in symbiotic = -70.64 ± 0.70 mV; Student’s t-test; t(28) = 3.467; P = 0.002; Fig. 6B). Likewise, we found an increased somatic membrane resistance (RN) in the symbiotic group (RN in control = 114.62 ± 4.99 MΩ; in symbiotic = 135.86 ± 5.69 MΩ; Student’s t test; t(28) = 2.789; P = 0.010; n = 15 cells; Fig. 6C1). On the other hand, membrane time constant and the “sag” conductance mediated by HCN channels did not exhibit statistical difference (membrane time constant in control = 17.21 ± 0.68 ms; in symbiotic = 18.16 ± 0.72 ms; Student’s t test; t(28) = 0.960; P = 0.346; Fig. 6C2. Sag conductance in control = 6.45 ± 0.61 mV; in symbiotic = 7.37 ± 0.48 mV; Student’s t-test; t(28) = 1.201; P = 0.240; Fig. 6C3).
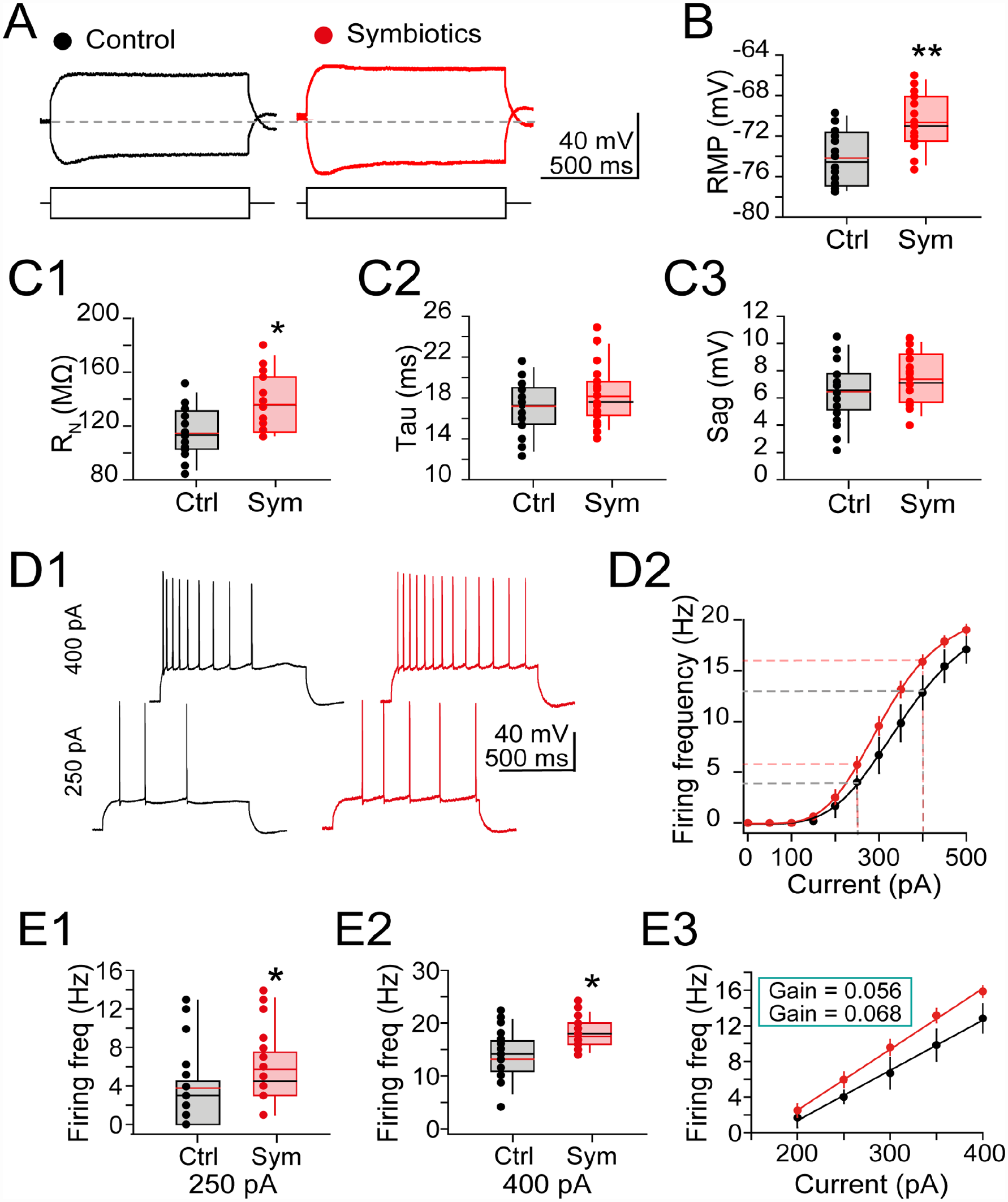
Symbiotics supplementation modifies electrophysiologic properties of CA1 pyramidal cells. (A) Representative voltage responses to current injections. Horizontal dashed line indicates resting membrane potential of control cell. (B) Box plot graphs summarizing the RMP; symbiotic-supplemented cells (symbols: values for each independent experiment; middle line in the box, average response ± SEM) were more depolarized compared to control cells (black symbols). (C) Box plots contrasting different kinetic values of intrinsic properties of control and symbiotic-supplemented cells: (C1) Somatic RN was increased in the symbiotic supplemented cells. (C2) Membrane time constant and (C3) “Sag” conductance was unchanged in symbiotic-supplemented cells. (D1) Representative firing responses to depolarizing current steps (1-sec duration, 250 pA or 400 pA). Notice the increased AP frequency in supplemented cells. (D2) Scatter plot; the sigmoid fitting summarizes the changes in firing frequency at the indicated current injections. The leftward shift in the symbiotic-supplemented group indicates increased firing output of symbiotic-supplemented cells compared to control. The vertical dashed lines indicate current values (250 pA and 400 pA) used to compare the firing frequency of both experimental groups (see below) (E1-2) Box plots showing the changes in firing frequency with 250 and 400 pA. (E3) Scatter plot; the increased slope of the linear fitting shows the increase in firing gain of the symbiotic-supplemented cells. *P < 0.05; **P < 0.01; n = 15 cells/5 animals for each condition. AP: action potential; RMP: resting membrane potential; RN: resistance; SEM: standard error of the mean.
Because RN influences the excitability of hippocampal neurons, changes in this passive electrical property are likely to modify the output of CA1 PCs (see Romo-Araiza et al. 9 ). Thus, we compared several active membrane properties from the control condition and CA1 PCs supplemented with the symbiotic. First, we found that the rheobase current, which is the minimal current required to elicit an action potential (AP), did not reach statistical difference in the symbiotic group compared to control cells [rheobase in control = 250.33 + 15.72 pA; in symbiotic = 213.33 + 10.31 pA; Student’s t -test; t(28) = 1.976; P = 0.058; data not shown]. However, the firing frequency (FF) did increase its prevalence in the symbiotic group, [F(1, 10) = 14.806; P = 0.003; Two-way ANOVA; Fig. 6C]. Representative AP trains from control and symbiotic CA1 PCs, and the resulting FF plot is shown in Fig. 6D1, D2. We also conducted independent FF analyses in response to current injections (250 and 400 pA) from the individual cells (see dashed lines in Fig. 6D2). The resulting analyses are summarized in the box plots of Fig. 6E1 (FF response to 250 pA) and Fig. 6E2 (FF to 450 pA). For both analyzed loci, the FF was increased in the symbiotic group compared to control cells [FF elicited with 250 pA in control = 3.78 + 0.69 Hz; in symbiotic = 6.07 + 1.02 Hz; Student’s t-test; t(28) = 1.895; P = 0.049. FF elicited with 400 pA in control = 14.86 ± 0.80 Hz; in symbiotic = 17.99 ± 0.71 Hz; Student’s t-test; t(28) = 2.762; P = 0.011]. Lastly, the rising slope observed in the linear portion of the FF graph confirmed an increase in the gain of firing of the symbiotic-supplemented animals (gain fire in control = 0.056 ± 0.007 Hz/pA; in symbiotic = 0.068 ± 0.003 Hz/pA; Mann-Whitney U; P = 0.043. Fig. 6E3). Together, these data demonstrate that symbiotic supplementation enhances the electrophysiologic responses from CA1 PCs and eases their ability to fire action potentials.
Symbiotic Supplementation Modifies the Action Potential Waveform of CA1 PCs
Higher magnification of the AP spike showed that symbiotic supplementation causes broadening of the AP half-width, as depicted in the superimposed spikes of control and symbiotic supplemented cells (Fig. 7A). Further confirmation of the increased half-width and slower spike repolarization was obtained by plotting the first derivative action potential spike 20 (Fig. 7B). Remarkably, the spike threshold did not exhibit differences in symbiotic supplemented cells compared to control CA1 PCs (arrowheads in Fig. 7A, B indicates AP threshold) [spike threshold in control = -36.35 ± 1.59 mV; spike threshold in symbiotic = -36.28 ± 0.75 mV; Student’s t-test; t(22) = 0.043; P = 0.966; Fig. 7C]. Regarding the kinetic parameters of the spike, we did not find change in the AP amplitude in both groups [spike amplitude in control = 126.70 ± 2.63 mV; spike amplitude in symbiotic = 123.99 ± 0.99 mV; Student’s t-test; t(22) = 1.017; P = 0.319; Fig. 7D]. As previously stated, the AP half-width (H-W) was significantly increased the symbiotic-supplemented cells compared to control cells [H-W in control = 1.26 ± 0.03 ms; H-W in symbiotic = 1.35 ± 0.03 ms; Student’s t-test; t(22) = 2.255; P = 0.033; Fig. 7E]. As expected, the maximal depolarization slope (MDS), a parameter that mirrors voltage-sensitive Na+ conductance, did not exhibit changes [spike MDS in control = 266.01 ± 9.26 mV/ms; spike MDS in symbiotic = 272.91 ± 7.11 mV/ms; Student’s t-test; t(22) = 0.600; P = 0.554; Fig. 7F]. Contrary to these observations, the maximal repolarizing slope (MRS), a kinetic parameter that mirrors activity of K+ conductance responsible of the fast repolarization of the AP exhibited a significant slope reduction [spike MRS in control = -108.21 ± 6.21 mV/ms; in symbiotic = -92.35 ± 3.63 mV/ms; Student’s t-test; t(22) = 2.282; P = 0.046; Fig. 7G]. These results indicate that symbiotic supplementation controls the AP broadening through the modulation of K+ conductances and has no effect on the voltage-sensitive Na+ conductances mediating the fast depolarization of the AP spike.

Symbiotics supplementation modifies the action potential kinetics of CA1 pyramidal cells. (A) Superimposed spikes of control and symbiotic-supplemented CA1 PCs. (B) Representative phase plots obtained from the AP shown in panel A; arrowhead in both panels indicate AP threshold. (C) Box plots showing the action potential threshold for each experiment. No difference was found in the AP threshold of the supplemented group compared to control cells. D-G) Box plots contrasting kinetic parameters acquired from the phase plots in control and symbiotics supplemented cells: (D) the spike amplitude was similar in both groups. (E) symbiotics supplementation increases the duration of spike half-width. (F) symbiotics supplementation did not alter the maximal depolarization slope of the spike. (G) symbiotics supplementation decrease the maximal repolarization slope of the spike. *P < 0.05; n = 15 cells/5 animals for each condition. AP: action potential; PC: pyramidal cell.
Symbiotic Supplementation Modifies the NMDA/AMPA-Receptor Ratio at the Schaffer—CA1 Pyramidal Cell Synapse
Metabolic disorders and obesity alter the synaptic machinery required for induction of LTP23–25, the physiologic substrate of memory and cognition 26 . Since symbiotic supplementation increased cognitive performance, we hypothesize that symbiotic may alter the density of NMDA/AMPA-receptors-mediated current ratio, a mechanism that could explain the improved cognitive performance of the obese animals. Therefore, we measured the NMDA/AMPA ratio of control and symbiotic supplemented CA1 PCs. To isolate the NMDA/AMPA transmission, picrotoxin (50 µM) was bath perfused for at least 10 min, and a stimulation electrode was placed in the stratum radiatum, 250–300 µM behind the recorded CA1 PC (see schematic representation in Fig. 8A). The recorded neurons were held in voltage-clamp mode, and excitatory postsynaptic currents (EPSC) were elicited at -70 mV and + 40 mV to evoke an AMPA-, and NMDA-mediated current, respectively. The NMDA/AMPA current ratio was larger in the symbiotic supplemented animals, as illustrated in the representative currents in Fig. 8B, and the individual experiments grouped in the boxplot of Fig. 8C [NMDA/AMPA ratio in control = 1.07 ± 0.07; in symbiotics = 1.37 ± 0.11; Student’s t-test; t(18) = 2.156; P = 0.045; Fig. 8C]. On the other hand, the kinetic properties of the NMDA-, and AMPA-receptor-mediated currents did not differ in both groups [AMPA rise time in control = 4.68 ± 0.21 ms; in symbiotics = 4.95 ± 0.15 ms; t(18) = 1.071; P = 0.301. AMPA decay time in control = 31.05 ± 1.47 ms; in symbiotics = 28.31 ± 1.68 ms; t(18) = 1.222; P = 0.239. NMDA rise time in control = 14.08 ± 0.49 ms; in symbiotics = 14.35 ± 0.31 ms; t(18) = 0.468; P = 0.646. NMDA decay time in control = 63.63 ± 3.85 ms; in symbiotics = 64.83 ± 3.13 ms; t(18) = 0.241; P = 0.813; Student’s t-tests; data not shown] suggesting increased expression of NMDA receptors rather than modified receptor kinetics in response to symbiotic supplementation.

Symbiotics supplementation modifies the NMDA/AMPA ratio and improves long-term potentiation of CA1 pyramidal cells. (A) Schematic representation of synaptic stimulation of Schaffer collaterals to evoke EPSC in CA1 pyramidal cells (patch-clamp recordings) and for extracellular recordings (field EPSPs), in which the recording pipette was also positioned in the Stratum Radiatum. (B) Representative NMDA-, and AMPA-mediated currents, evoked at +40 and -70 mV, respectively. (C) Box plots contrasting the NMDA/AMPA ratio from control (black symbols) and symbiotic-supplemented cells (red symbols). (D) Upper panel: Representative EPSCs from control (black traces) and symbiotics supplemented cells (red traces). Both groups exhibited reliable PTP and LTP. Symbiotics supplemented cells exhibited robust LTP compared to control cells. Lower panel: Time course graph of LTP experiments. (E) Bar graph summarizing individual values of potentiation (symbols) and average response ± SEM (bar graphs) for both experimental groups. F) Cumulative probability chart for EPSC amplitude after HFS for both experimental conditions. The statistical graph was constructed using the steady-state EPSC amplitude after induction of LTP; therefore, the first five minutes of EPSC responses (after PTP) were discarded. AMPA: α-amino-3-hydroxyl-5-methyl-4-isoxazole-propionate; EPSC: excitatory postsynaptic currents; fEPSP: field excitatory postsynaptic potential; HFS: high-frequency stimulation; LTP: long-term potentiation; NMDA: N-methyl-D-aspartate; PTP: post-tetanic potentiation; SEM: standard error of the mean. *P < 0.05; n = 10 cells/5 animals for each condition.
Subsequently, we explored the induction of LTP in control and symbiotic-supplemented cells. Because of the increased NMDA/AMPA ratio found in the latter group, we hypothesize that the magnitude of LTP will be increased in the synapses of the supplemented CA1 PCs. For both experimental conditions, a baseline of CA1 EPSCs was acquired at 0.16 Hz for 10 min, followed by trains of HFS (100 pulses at 100 Hz repeated every 10 s) delivered to the Schaffer collaterals. In response to HFS, the EPSCs exhibited a robust PTP followed by a sustained increase in the excitatory postsynaptic currents’ (EPSC) amplitude that continued up to 30 min. Although CA1 LTP was reliable expressed in both groups, the synapses of the symbiotics supplemented cells exhibited an increased response compared to control CA1 PCs [PTP in control = 369.06% ± 34.09% of baseline; PTP in symbiotics = 423.75% ± 20.22% of baseline; Student’s t-test; t(12) = 1.755; P = 0.105. EPSCs amplitude at 30 min after HFS in control = 147.83% ± 13.24% of baseline. EPSC amplitude in symbiotics = 197.50% ± 20.22% of baseline; Student’s t-test; t(12) = 2.393; P = 0.034]. The time-course graph in Fig. 8D shows the averaged LTP from both groups acquired (black and red symbols for control and symbiotic supplemented cells, respectively). The individual magnitude of LTP obtained from each cell tested and the average response ± SEM is shown in Fig. 8E, and the cumulative probability of LTP for control and symbiotic supplemented cells is shown in Fig. 8F. Together, these data show that symbiotic supplementation modifies the expression of glutamate receptors in CA1 PCs and enhances hippocampal LTP.
Symbiotic Supplementation Increases the Bidirectional Plasticity at CA1 PCs Synapses
A property of the glutamatergic synapses in CA1 PCs is the long-term bidirectional modifications mediated by NMDA receptors and governed by the level of presynaptic activity. Indeed, a CA1 synapse that is depressed by LFS (i.e., 900 pulses at 1 Hz) can be subsequently potentiated in response to HFS (100 pulses at 100 Hz, repeated twice, 10-s interval). Because our previous findings demonstrated increased NMDA receptor-mediated responses (Fig. 8B, C), we explored bidirectional plasticity 27 in response to symbiotic supplementation.
Since bidirectional plasticity requires prolonged periods of data acquisition (up to 3 h), we switched to the non-invasive technique of extracellular recordings to overcome the intracellular washout associated with the whole-cell patch-clamp technique. A bipolar stimulation electrode was placed in the stratum radiatum of CA1, and field excitatory postsynaptic potentials (fEPSPs) were also acquired in the stratum radiatum (see schematic representation in Fig. 8A for electrode placement). After 10–15 min of stabilizing the synaptic response, a 25-min baseline of fEPSPs was acquired at 0.06 Hz. Then, LFS (900 pulses at 1 Hz) was delivered to the Schaffer collaterals. In control slices, LFS failed to induce a stable LTD (fEPSP slope at 60 min post-LFS = 83.4% ± 11.01% of baseline response, P = 0.16 Wilcoxon test, n = 6 slices/5 animals. Black symbols in the time-course graph, Fig. 9A; see also Representative fEPSPs in the upper panel of Fig. 9A, acquired at the indicated times of the time-course graph). In contrast, in symbiotic slices, LFS induced a stable, long-lasting depression of the fEPSP slope (fEPSP slope at 60-min post-LFS = 71.7% ± 6.55% of baseline response, P = 0.03, Wilcoxon test, n = 7 slices/5 animals. Red symbols in the time-course graph, Fig. 9A). After 60 min of continuous fEPSPs acquisition (post-LFS), the stimulation current was adjusted to match the original baseline response. Then, a second baseline of fEPSPs was acquired for 15 min, and HFS (100 pulses at 100 Hz, repeated twice, 10-s interval) was delivered to the Schaffer collaterals. In response to HFS, PTP was observed in both groups (PTP in control slices = 241.6% ± 31.7% of baseline response; PTP in symbiotics slices = 259% ± 26.8% of baseline response). The PTP was followed by a long-lasting enhancement of the fEPSPs slope in both experimental conditions (fEPSP slope at 60 min post-HFS in control slices = 133.1% ± 13.6% of basal response, P = 0.031, Wilcoxon test, n = 6 slices/5 animals. fEPSP slope at 60 min post-HFS in symbiotic group = 180.4% ± 14% of baseline response, P = 0.03, Wilcoxon test, n = 7 slices/5 animals). The bar graphs in Fig. 9B summarize the averaged response ± SEM and the individual magnitude of LTD, PTP, and LTP (symbols) from each slice and experimental condition included in this analysis. An additional comparison of the magnitude of LTP revealed that the symbiotic-supplemented slices exhibited ≈47% increased potentiation compared to control slices (P = 0.035, Mann-Whitney test; Bars in Fig. 9B). Fig. 9C summarizes the cumulative probability distribution of the bidirectional plasticity (LTD and LTP) for control and symbiotic supplemented slices. These findings show that symbiotic supplementation increases the plasticity capabilities at the Schaffer collateral synapses of obese animals.

Symbiotics supplementation increases the bidirectional plasticity at CA1 PCs synapses. (A) Representative fEPSPs (averaged from 12 consecutive sweeps, except PTP whose trace was averaged from 4 consecutive sweeps) recorded at the indicated time in the time-course graph (lower panel). The time-course graph summarizes the average of the fEPSPs slope (control n = 6 slices/5 animals, black symbols; symbiotic supplemented slices n = 6 slices/5 animals, red symbols). Arrowheads below LFS and above HFS indicate delivery of high-frequency stimulation (900 pulses at 1 Hz for LFS and 100 pulses, repeated twice, 10-s interval for HFS, respectively). After 60 min of recording post-LFS, the intensity of the test stimulation was readjusted (increased) to match the original baseline fEPSP amplitude. (B) Bar graph summarizing the changes in fEPSP slope magnitude in response to LFS, PTP, and HFS from control (white bars, n = 6 slices/5 animals) and symbiotic supplemented slices (red bars n = 6 slices/5 animals). The horizontal dashed line represents the baseline response of the normalized fEPSP slopes. (C) Cumulative probability chart shows the magnitude of LTD (left panel) and LTP (right panel) for control and symbiotic-supplemented slices (black and red lines, respectively). The vertical dashed line represents the EPSP baseline. Arrowheads indicate a change in the slope below (for LTD) and above (for LTP) of 25% of the baseline value. Changes in the fEPSP slope below 25% were discarded. *P < 0.05 vs. baseline response (Wilcoxon test) or control group (Mann-Whitney test). fEPSP: field excitatory postsynaptic potential; HFS: high-frequency stimulation; LFS: low-frequency stimulation; LTD: long-term depression; LTP: long-term potentiation; PC: pyramidal cell; PTP: post-tetanic potentiation.
Discussion
Obesity is a worldwide problem associated with different comorbidities, including cognitive impairment 28 . Obesity has shown an important association with cognitive decline, particularly affecting executive function and both episodic and working memory2,4. Preclinical models of diet-induced obesity show that cognition is impaired, primarily affecting the hippocampus and thus spatial learning and memory 1 .
The underlying mechanism of obesity in inducing cognitive impairment is not clearly known, but some mechanisms have been proposed. It is well known that obesity and an unbalanced diet lead to dysbiosis, which in turn is associated with leaky gut, as well as an increase in blood brain barrier (BBB) permeability and neuroinflammation 29 . The latter is a pathological state that is strongly associated with changes in hippocampal transmission and synaptic plasticity. Neuroinflammation inhibits also the expression of neurotrophic factors and the action of other essential mechanisms for cognitive function30,31. Likewise, the inflammatory reaction in the brain induces oxidative stress and an increase in reactive oxygen species (ROS), another important factor contributing to hippocampal degeneration 28 .
Another possible mechanism of obesity-induced cognitive impairment is insulin resistance (IR). Both obesity and HFSD are associated with IR, as free fatty-acids (FFAs) affect insulin receptor function. Development of IR affects the use of insulin by cells, including those in the hippocampus. This could be a critical inducer of cognitive alterations since the modification in hippocampal insulin signaling could be associated with cognitive impairment 32 . Insulin is very important for the brain, as it participates in neuronal survival, neuroplasticity and cognition. Thus, IR can potentially affect these functions. Insulin also modulates LTP and long-term depression (LTD) by reducing the amount of AMPA receptors 33 . Moreover, IR has also shown to induce mitochondrial dysfunction, increased ROS production, and neuronal death in the brain33,34.
On the other hand, as mentioned before, microbiota imbalance is perhaps one of the most important factors participating in the induction of obesity co-morbidities. Microbiota composition changes according to diet and body weight increases. Therefore, interventions that improve microbial composition—as supplementation with the symbiotic did in the present investigation—also improve the metabolic dysfunction associated with obesity, as well as some cognitive functions through the microbiome-gut-brain axis1,7. The microbiome–gut–brain axis establishes bidirectional interactions between gut microbiome and the brain either by neuronal, immune, or neuroendocrine pathways. Microbial metabolites such as SCFA are important for gut microbiota-brain signaling as they can both establish a bottom-up signaling system mainly through an immune pathway, as well as cross the BBB and act in brain cells directly 35 .
In addition, the enhancement of microbiota leads to an improvement in the function and integrity of the BBB and brain environment partly by increasing occluding expression and modulating the concentration of some neurotransmitters36,37. Thus, the supplementation with a symbiotic, without changing other parameters of lifestyle, could improve different aspects of memory and electrical function of hippocampal neurons.
The symbiotic combination (E. faecium and agave inulin) used in the present investigation has already demonstrated to improve cognition in middle-aged rats 9 . These results prompted us to evaluate if it could also improve cognition in an obesity model, considering that cognitive impairment is also an important co-morbidity in obesity. In this way, in the present investigation it was shown that spatial memory in obese animals did indeed improve with symbiotic supplementation.
Previous investigations have shown that the probiotic used in this investigation (E. faecium) is capable of increasing the amount of butyrate, a postbiotic produced by E. faecium with anti-inflammatory properties that could diminish neuroinflammation38,39. In fact, this probiotic is one of the strains commonly used for inflammatory bowel disease; butyrate activates G protein–coupled receptors (GPCR), inhibiting neutrophile expression of proinflammatory mediators, and also by promoting Th1 cell production of IL-10, which has an anti-inflammatory effect 40 . In the present investigation, the symbiotic-supplemented group showed a significant increase in butyrate. On the other hand, the symbiotic (E. faecium + inulin) is known to increase the expression of the BDNF by inhibiting histone deacetylase (HDAC)-4, which inhibits BDNF expression, a critical molecule for memory consolidation9,40,41; germ-free mice show reduced expression of BDNF and NMDA receptors in cortex and hippocampus 42 . Therefore, by either reducing inflammation or increasing BDNF concentrations, the symbiotic could improve cognitive function.
Additionally, recent investigations have reported the usefulness of probiotic supplementation, specifically with E. faecium, in the amelioration of IR. This could also be an additional effect contributing to cognitive improvement43–45.
As memory was affected in non-supplemented rats, and this function is directly related to the hippocampus, we decided to evaluate the electrophysiology of this specific area of the brain. We found that obesity induced by a HFSD affects action potentials, and supplementation with the symbiotic eases the ability to fire action potentials and modulates K+ conductances 46 .
As it is well known, K+ efflux regulates membrane repolarization and dictates the ceasing of the AP, leading to the loss of positive charges that return the cell to its RMP or even to hyperpolarization 47 . These channels are critical to set neuronal excitability, synaptic integration capabilities and plasticity through induction and expression of LTP at the glutamatergic synapses. In this study, the supplementation of the symbiotic caused a broadening of the AP, a phenomenon dependent of K+ conductances.
For instance inactivation of potassium voltage-gated channel subfamily A member 1 (Kv1) expressed in the hippocampal CA1 neurons, produces a broadening of the AP, enhancing spike-evoked Ca2+ entry and neurotransmitter release; this leads to a facilitation in synaptic transmission 48 . Potassium channels inactivation limits their conductance during periods of high neuronal activity, as this decay current limits the afterhyperpolarization, thus quickly restoring the high membrane resistance and facilitating high-frequency firing47,49. In the present study, we found increased firing frequency that might be associated with regulation of several K+ conductances. In this sense, other potassium currents such as that mediated by the M channels, also modulate neuronal fire frequency, as the inhibition of this current leads to the firing of many rhythmical AP after a single excitatory imput. Blockers of these channels, such as acetylcholine, have been associated with increased learning 50 . In this regard, gut microbiota is linked to acetylcholine release, since some microorganisms can activate the vagal nerve fibers and promote the release of this neurotransmitter 51 . In addition, SCFA also stimulate the secretion of acetylcholine 52 .
The symbiotic supplementation increases the NMDA/AMPA ratio, enhancing thus, the magnitude of LTP when compared to the control group. In a previous study, we demonstrated that this symbiotic enhances both LTP and NMDA/AMPA ratio in middle-aged rats 9 . Other probiotics supplementation has also shown to modify LTP. In a study carried out by Jena et al. 53 , mice fed with a western diet showed reduction in LTP-based neuroplasticity, and B. infantis supplementation returned the potentials to normal levels, with no difference between the control group fed with a healthy diet.
As symbiotic supplementation increased NMDA receptor-mediated responses, we explored bidirectional plasticity and found a significant increase in the plasticity capabilities at the Schaffer collateral synapses and neurogenesis of obese animals. This finding suggests that a symbiotic supplementation has the ability to enhance or perhaps restore the metaplastic capabilities of the glutamatergic synapses, a phenomenon that requires additional experimentation. These results could also explain the cognitive improvement in supplemented rats. Since symbiotic administration increases BDNF production, this could partly explain the increase in synaptic plasticity, and neurogenesis since this molecule is capable of generating this type of processes9,54.
Finally, we found no significant differences between the studied groups in working and reference memory. A study carried out by Yoshizaki et al. 55 showed that a high-fat diet enhances working memory and reduces anxiety-like behavior in rats undergoing the elevated plus maze test, indicating that this type of diet might have beneficial effects in some cases; another study carried out by Deshpande et al. 56 showed that a high-fat diet administered for 8 weeks to 7-month-old male Sprague Dawley rats did not affect their performance on the water radial arm maze. That could be the reason why we found no differences between the studied groups.
Conclusions
Obesity causes cognitive impairment through different pathways, such as systemic inflammation, oxidative stress, and dysbiosis. The administration of a symbiotic (E. faecium and agave inulin) to obese rats improves cognitive impairment, demonstrated by an improvement in spatial memory, increased neurogenesis, and LTP generation; one of the mechanisms of action might be the increase in butyrate showed in fecal samples, as butyrate helps the induction of BDNF and thus of LTP.
Footnotes
Acknowledgements
We thank Diego Incontri-Abraham for language supervision.
Author Contributions
Conceptualization, A.I and A.R.A.; methodology, R.I.P.A, E.G., L.A.M, Y.C., A.M.F.P, A.C.G., R.R.B., A.P.A.C., C.R.Q., R.J.C.; formal analysis, A.R.A., A.I., E.J.G.; writing—original draft preparation, A.R.A., E.J.G., A.I.; writing—review and editing, A.I., E.J.G.; supervision, A.I., E.J.G., A.R.A; project administration, A.R.A, A.I., E.J.G.; funding acquisition, A.I., A.R.A., E.J.G. All authors have read and agreed to the published version of the manuscript.
Ethical Approval
The animal study protocol was approved by Animal Bioethics and Welfare Committee of the Health Sciences Faculty at Anahuac University approved all animal procedures (ID: 201850).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research received financial support from Universidad Anáhuac México.
