Abstract
Hypoglycemia is a frequent and often serious complication of diabetes management. While its acute effects on cognition are well documented, growing evidence suggests that recurrent or severe hypoglycemia exposure may also contribute to long-term cognitive decline and increased risk of dementia in patients with diabetes. In fact, clinical and epidemiological data suggest that repeated hypoglycemic events are associated with lower performance in multiple cognitive domains, especially in pediatric and elderly populations. Moreover, meta-analyses and cohort studies highlight a significant association between severe hypoglycemia and increased risk of dementia while cognitive impairment has been shown to also increase the risk of hypoglycemia. This suggests a potential bidirectional relationship between hypoglycemia and cognitive decline. On the molecular level, both animal and human studies reveal that recurrent hypoglycemia induces oxidative stress, impairs mitochondrial dynamics, and leads to synaptic injury, particularly in vulnerable brain regions such as the hippocampus and cortex. These findings underscore the need for personalized glucose management strategies that minimize hypoglycemia risk while maintaining glycemic targets, particularly in populations vulnerable to cognitive impairment. This review summarizes the current understanding of the impact of hypoglycemia on brain function, including its structural, cellular, metabolic, and cognitive consequences.
Introduction
Cognition is defined by the American Psychological Association as “all forms of knowing and awareness, such as perceiving, conceiving, remembering, reasoning, judging, imagining, and problem solving.” 1 It is influenced by numerous modifiable and non-modifiable conditions such as age, education level, drinking, smoking, social activity, and disease conditions, among others. 2 Any malfunction in the brain or other organ processes particularly those associated with chronic disease can impair cognition. Such conditions include hypertension, heart disease, stroke, obesity, diabetes, and kidney disease. 3 Cognitive impairments can be broadly categorized into neurodegenerative and non-neurodegenerative impairments. Alzheimer’s disease, frontotemporal lobar degeneration, and α-synucleinopathies result in a neurodegenerative type of cognitive impairment. Some nutritional deficiencies, exposure to toxins, metabolic disorders, and cerebrovascular diseases result in a non-neurodegenerative type of cognitive impairment. 4 The present review article focuses on the impact of exposure of patients with diabetes to hypoglycemia on cognitive impairment.
Diabetes
Diabetes is a chronic condition characterized by hyperglycemia due to either insulin deficiency or impaired glucose uptake. 5 According to the Centers for Disease Control and Prevention ~38 million Americans have diabetes (>11% of the U.S. population). 6 Three major types of diabetes exist, with type 1 diabetes (T1D) accounts for 5%–10% of cases, type 2 diabetes (T2D) accounts for 90%–95% of cases, and gestational diabetes occurring in pregnant women. T1D occurs due to insulin deficiency, while T2D results from insulin resistance. Complications of diabetes may include microvascular complications such as neuropathy, nephropathy and retinopathy, as well as macrovascular complications such as cardiovascular disease and stroke. These complications can be episodic or progressive. 5 Sixty-five percent of deaths in people with diabetes are due to cardiovascular disease, 5 with stroke and ischemic heart disease being the primary causes of morbidity. Retinopathy is the most prevalent microvascular complication and typically develops slowly over time. It is both treatable and preventable with early detection. 5 Diabetic nephropathy, characterized by persistent proteinuria, may be present at the time of T2D diagnosis but emerges years after T1D onset. It significantly increases the risk of coronary heart disease and stroke, and death from macrovascular disease. Improved glycemic control can reduce the risk of neuropathy. 5 Peripheral neuropathy and diabetic ketoacidosis are other secondary complications of diabetes.5,7 Diabetes is also associated with geriatric symptoms such as cognitive impairment, falls, and depression. 8 Cognitive decline and dementia are considered late-life complications of diabetes.8,9
Diabetes and hypoglycemia
Diabetes has numerous potential consequences, which are significantly worsened and more dangerous with inefficient treatment. 10 Multiple daily insulin injection therapy with insulin analogs, as well as continuous subcutaneous insulin infusions along with pump therapies, are considered optimal treatment option for effective management of hyperglycemia in T1D. 11 Given that there is progressive beta cell dysfunction in T2D, the specific therapy must be individualized. Basal insulin is added as an initial insulin regimen using either intermediate-acting neutral protamine Hagedorn or long-acting insulin, while short-acting insulin added before meals if needed. Trying to regulate glycemic levels through rigorous insulin therapy, however, may lead to hypoglycemia. Continuous glucose monitoring has shown that most patients with treated diabetes experience hypoglycemia.12 –14 Recurrent exposure to hypoglycemia (RH) leads to severe consequences, such as hindered counterregulatory defense against hypoglycemia, increasing the frequency and severity of future episodes. 15
The American Diabetes Association (ADA) defines hypoglycemia as “all episodes of an abnormally low plasma glucose concentration that leads to patient harm,” 16 while others define hypoglycemia as a temporary state of significantly reduced blood glucose levels. In most individuals, a blood sugar level lower than 70 mg/dL is defined as a hypoglycemic event. 17 Clinically, hypoglycemia is identified by the presence of symptoms caused by a drop in plasma glucose level and the alleviation of symptoms after the blood glucose level is returned to normal. 18 It is the main hindrance in both T1D and T2D in obtaining optimal glycemic control, which the ADA recommends being HbA1c levels <7%.19,20 Hypoglycemia can be divided into symptomatic and asymptomatic episodes. Asymptomatic hypoglycemia is characterized by blood sugar levels below the alert value of 70 mg/dL without symptoms, 21 while symptomatic hypoglycemia involves blood sugar levels below the alert value but with typical hypoglycemia symptoms. Hypoglycemia is classified as mild, moderate, and severe based on the extent of drop in blood glucose levels. Mild (level 1) and moderate (level 2) hypoglycemia refers to blood glucose levels of 54–70 or >54 mg/dL, respectively. Severe (level 3) hypoglycemia is characterized by altered mental and/or physical status requiring assistance.22,23
Hypoglycemia is a common occurrence among patients with diabetes, particularly those who are insulin dependent.12 –14 It often results from insulin therapies reducing glucose levels excessively, leaving patients in a hypoglycemic state. While insulin treatment is necessary for controlling diabetic hyperglycemia, wrong dosage may lead to dangerously low glucose levels. In response to falling glucose levels, the body typically initiates an autonomic response characterized by symptoms of sweating, palpitations, and shakiness. 24 This response also triggers the release of hormones to restore glucose levels. These symptoms serve as early warning signs, prompting patients to take corrective action. However, repeated hypoglycemic events can lead to hypoglycemia unawareness. Hypoglycemia unawareness is defined as the failure to sense a significant fall in glucose below normal levels.
Those with hypoglycemia unawareness may experience delayed or absent physiological responses, which may prevent them from seeking treatment on time. They instead may experience symptoms of neuroglycopenia. Neuroglycopenia is a severe neurological condition accompanied by reduced consciousness, drowsiness, confusion, loss of consciousness, and even coma.25,26 This cognitive deficit prevents recognition of hypoglycemia, delaying treatment and increasing the risk of acute complications. Studies show that hypoglycemia unawareness affects 25%–40% of T1D patients who have had disease for >15 years. 24 Repeated moderate hypoglycemic episodes increase the risk of severe hypoglycemia due to a decreased counter-regulatory response. 26
Metabolic effects of hypoglycemia exposure
Hypoglycemia significantly impacts brain metabolism, as the brain relies almost exclusively on glucose for energy. 27 In response to hypoglycemia, the brain adapts by upregulating glucose transporters like GLUT1 and GLUT3 to enhance glucose uptake. 27 It also shifts substrate use by promoting uptake and oxidation of amino acids and increasing lactate uptake. 27 However, acute or prolonged hypoglycemia can impair glycolysis, reduce key glycolytic intermediates, and alter the TCA cycle activity. 27 Earlier, we reported that exposure to mild/moderate RH induces changes in the hippocampal metabolomic profile of insulin-treated rats with streptozotocin-induced diabetes (Figure 1). 28
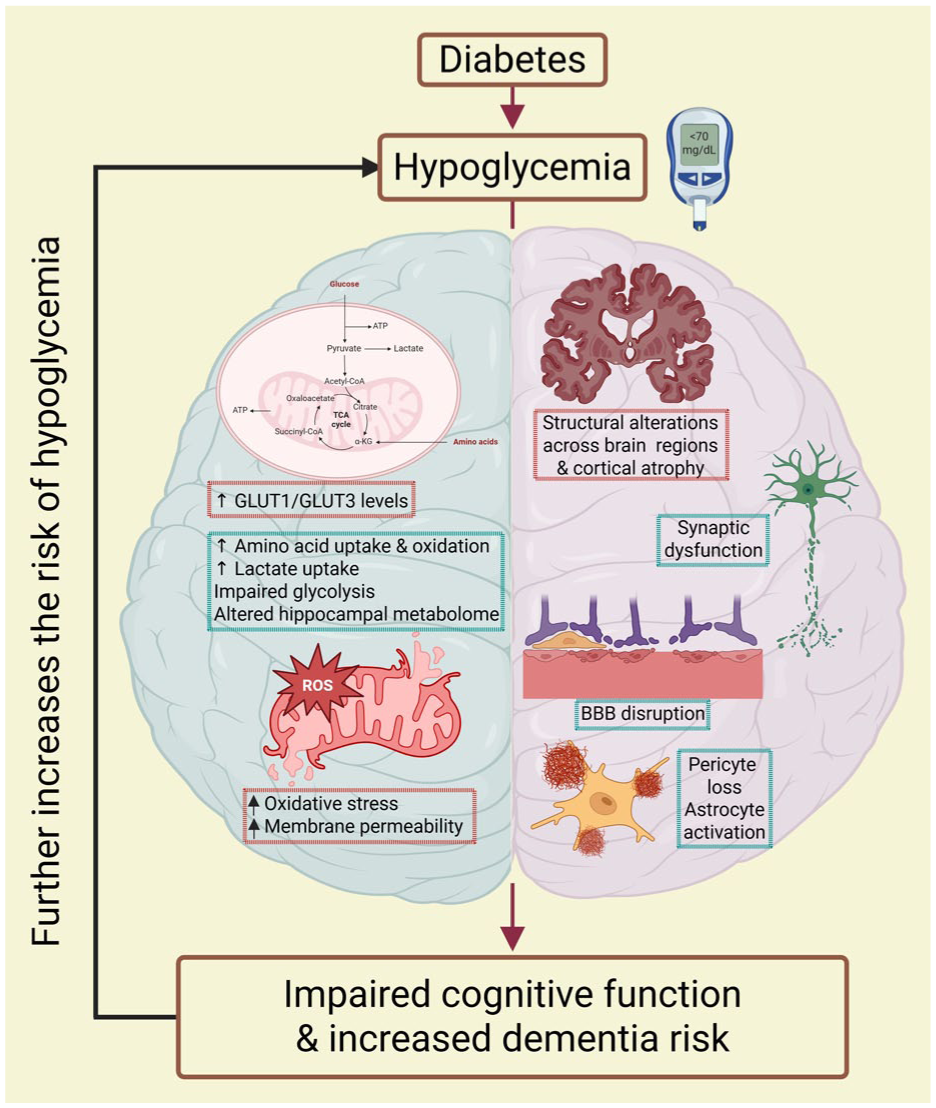
A diagram summarizing potential mechanisms by which recurrent hypoglycemia impairs cognitive function and increases the risk of dementia in diabetes. Recurrent exposure to hypoglycemia may impair cognitive functions through a combination of metabolic alterations and neuropathological changes in brain. Proposed metabolic effects include upregulation of glucose transporters 1 and 3 to enhance glucose uptake, increased utilization and oxidation of amino acids, and enhanced lactate uptake. Additional hypoglycemia-induced metabolic disturbances include impaired glycolysis, reduced levels of key glycolytic intermediates, altered tricarboxylic cycle activity, changes in the hippocampal metabolomic profile, and mitochondrial dysfunction leading to reduced ATP production and increased oxidative stress due to elevated free radical generation. Neuropathological changes associated with recurrent hypoglycemia include reduced cortical volume (frontal, parietal, and temporal lobes), synaptic loss in hippocampal regions, and blood–brain barrier dysfunction, brain edema, pericyte loss, astrocyte overactivation, and histopathological changes within the hippocampus. Changes encircled with green dotted lines indicate potentially recoverable changes, whereas those encircled with red dotted lines indicate potentially non-recoverable changes. Created in BioRender. https://BioRender.com/icish63
Beyond glucose metabolism, hypoglycemia-associated substrate limitation reduces ATP production, affects mitochondrial function and increases oxidative stress due to elevated free radical formation.27,29 An earlier study reported lower mitochondrial membrane potential, increased levels of oxidative damage, imbalance of mitochondrial dynamics, and increased mitophagy in the hippocampus of RH-exposed streptozotocin (STZ)-diabetic mice when compared to STZ-diabetic mice. 30 Similarly, another study observed that the exposure of cultured astrocytes to recurrent low glucose resulted in mitochondrial dysfunction and increased accumulation of mitochondrial reactive oxygen species. 31 This dysfunction, particularly in vulnerable brain regions like the hippocampus, may contribute to cognitive impairment. However, inhibition of PARP-1 and pyruvate treatment was found to reduce the extent of acute severe hypoglycemia exposure induced cognitive impairment and neuronal cell death in the CA1 region of the hippocampus (Figure 1).32,33 Therefore, literature collectively indicates that hypoglycemia impairs mitochondrial function and elevates oxidative stress, contributing to neuronal damage.
The actual physiological effects of hypoglycemia on the brain remain incompletely understood. Chabriat et al. used PET scans to analyze brain metabolism after recurrent insulin-induced severe hypoglycemic episodes in 15 insulin-dependent patients with T1D. 25 They found no significant differences in brain metabolism between those with frequent comas and those without, suggesting that brief insulin-induced comas may not cause lasting brain damage. In people with diabetes mellitus, hypoglycemia results in decreased brain glucose levels regardless of hypoglycemia awareness. However, the rate of glucose metabolism was higher in the moderate hypoglycemia-aware group when compared to the moderate hypoglycemia-unaware group. 34 Another study demonstrated that the global brain glucose uptake was 20% lower during both moderate hypoglycemia and euglycemia in the moderate hypoglycemia-unaware patients when compared to the hypoglycemia-aware patients. 35 This observation is contrary to the expected increased glucose uptake due to elevated levels of glucose transporters in the brain following exposure to moderate hypoglycemia.36,37 Collectively, these findings underscore that while the brain adapts to short-term glucose deprivation, repeated or severe hypoglycemia can disrupt cellular metabolism, leading to potential long-term neurological effects such as cognitive impairment. The potential metabolic mechanisms causing RH-induced cognitive impairment are shown in Figure 1.
Overall, the literature suggests that RH may drive cognitive impairment through multiple mechanisms acting in synergy, including metabolic, oxidative, and inflammatory changes in the brain (Tables 1 and 2). 38 Based on the available evidence, we propose a model in which hypoglycemia-induced substrate limitation leads to increased mitochondrial reactive oxygen species production and impaired mitochondrial function, including reduced ATP production efficiency and further amplification of oxidative stress. 29 These early events may subsequently activate additional metabolic and inflammatory pathways during the sub-acute to chronic phases post-hypoglycemia exposure. The cumulative effects of these processes may preferentially affect vulnerable brain regions, such as hippocampus, leading to sustained structural and functional deficits. 39 However, the mechanisms underlying the disproportionate susceptibility of the hippocampus to hypoglycemia-induced damage remain unknown.
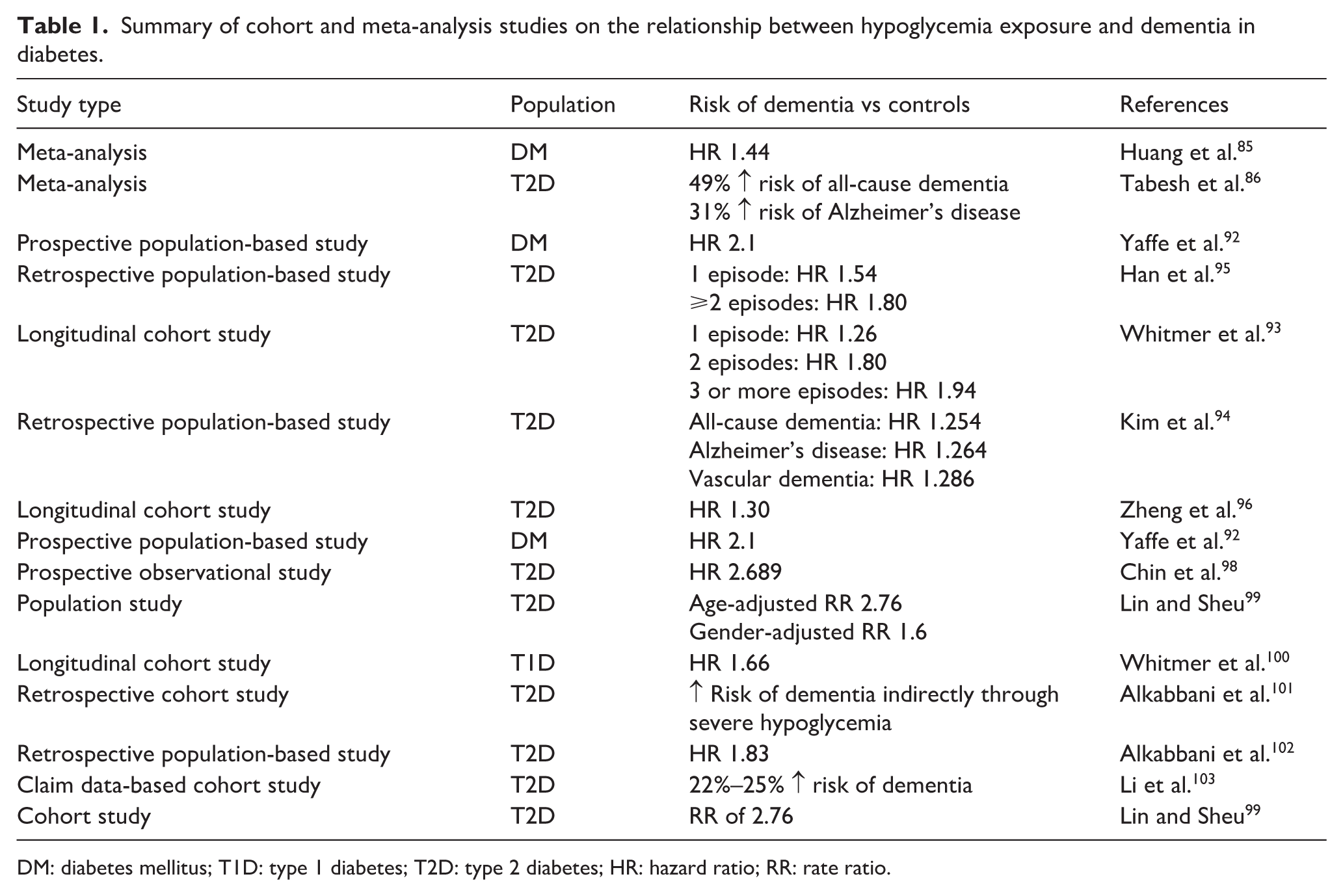
Summary of cohort and meta-analysis studies on the relationship between hypoglycemia exposure and dementia in diabetes.
DM: diabetes mellitus; T1D: type 1 diabetes; T2D: type 2 diabetes; HR: hazard ratio; RR: rate ratio.
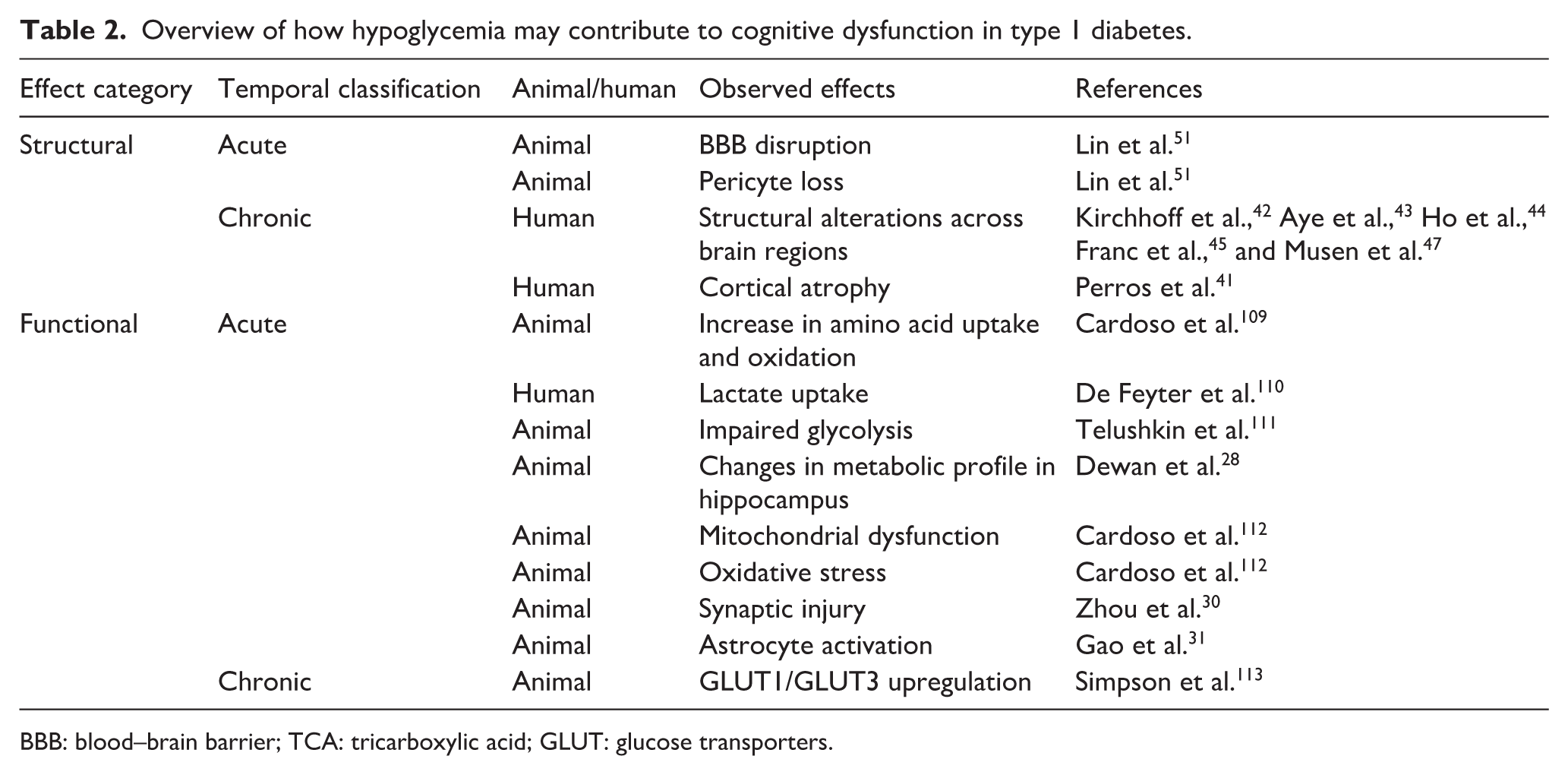
Overview of how hypoglycemia may contribute to cognitive dysfunction in type 1 diabetes.
BBB: blood–brain barrier; TCA: tricarboxylic acid; GLUT: glucose transporters.
Cellular and structural vulnerability of the brain to hypoglycemia
Due to its high energy requirements and limited glucose storage capacity, the brain is particularly vulnerable to hypoglycemic stress. This section explores how acute and RH affect brain cells, disrupt synaptic function, impair mitochondrial dynamics, and lead to structural changes in various brain regions (Table 2).
Limited information exists on how hypoglycemia affects brain structure. A retrospective MRI study comparing T1D subjects with normal and impaired awareness of hypoglycemia, and non-diabetic controls observed that those with impaired awareness had a smaller volume of the whole cortex (frontal lobe, parietal lobe, and temporal lobe) compared to patients with normal awareness of hypoglycemia, while no significant differences were observed between patients with normal awareness and control. The results of this study suggest that the observed changes may be due to long-term exposure to RH. 40 Similarly, Perros et al. observed an association between the history of severe hypoglycemia exposure and cortical atrophy in patients with T1D, 41 while a case report also linked a reduced volume of gray matter in the hippocampus with memory impairment in a patient with T1D exposed to recurrent severe hypoglycemia. 42 Early-onset T1D is associated with increased structural abnormalities in the brain.43,44 Patients with diabetes display cognitive deficits potentially related to both a decrease in the white matter volume and reduction in gray matter density.45 –48 Two studies specifically found reduced white matter in patients diagnosed with T1DM, correlating this loss with cognitive decline (Figure 1).46,48
At the cellular level, hypoglycemia affects different brain regions unevenly. A review article proposes that not all areas of the brain are equally affected by hypoglycemia. 49 The hippocampal area, temporal area, cerebral cortex, substantia nigra, and basal ganglia area of the brain appear more sensitive to hypoglycemic injury. In studies with normal and STZ-diabetic rats, moderate RH (1–2 mM for 1 h over 5 days) caused sparse neuronal death in the cortex without detectable neuronal death in the hippocampus. 50 The exposure of RH to STZ-diabetic mice resulted in disorganized pyramidal cells with smaller cell bodies and shrunken or absent nuclei. The morphological condition of the pyramidal cell layer appeared worse in the RH-exposed STZ-diabetic group when compared to the STZ-diabetic group. 30 Severe hypoglycemia (<2 mM for 90 min) in STZ-diabetic mice results in blood–brain barrier (BBB) damage, reduced expression of markers of the BBB, brain edema, pericyte loss, and histopathological changes in the hippocampus. 51 Exposure of STZ-diabetic mice to RH results in excessive activation of astrocytes in the cortex, and lower levels of neurotrophins (BDNF/GDNF) and downregulation of related signaling (Figure 1). 31 The same study observed that the exposure of cultured astrocytes to recurrent low glucose resulted in increased accumulation of mitochondrial reactive oxygen species and mitochondrial dysfunction.
At the subcellular level, the exposure of RH to STZ-diabetic mice results in injury to hippocampal synapses. 30 Electron microscopy ultrastructural studies revealed irregular synapses with fewer synaptic vesicles, smaller postsynaptic regions and densities, widened synaptic cleft, and swollen synaptic membrane in RH-exposed STZ-diabetic mice when compared to normal control and STZ-diabetic mice. These subtle changes in synaptic organization likely impair synaptic transmission, membrane excitability, and synaptic plasticity. The study also found an absence of many cristae within mitochondria and a lower numeric density and specific surface area of mitochondria, suggesting significantly swollen mitochondria and a lower number of mitochondria in RH-exposed STZ-diabetic mice when compared to normal control mice. Additionally reported lower mitochondrial membrane potential, increased levels of oxidative damage, imbalance of mitochondrial dynamics, and increased mitophagy in the hippocampus was also found in RH-exposed STZ-diabetic mice when compared to STZ-diabetic mice. 30 Another study observed oxidative injury in the hippocampal dendritic area in both normal and STZ-diabetic rats exposed to moderate RH (1–2 mM for 1 h, for 5 consecutive days). 50 The exposure of differentiated N2a neuroblastoma cells to glucose/pyruvate deprivation resulted in increased tau phosphorylation (Ser262 and Ser396). 52 Starvation of mice also results in tau hyperphosphorylation. 53
The literature demonstrates that hypoglycemia exerts profound effects on the brain at multiple levels, from structural alterations in vulnerable regions to subcellular impairments in synaptic and mitochondrial function as summarized in Figure 1.
Effect of hypoglycemia exposure on cognitive function
Research shows a strong association between cognitive impairment and diabetes with many diabetic individuals exhibiting decreased cognitive functioning and physiological changes within the brain (Table 1). 54 This is observed in both T1D and T2D patients. Severe hypoglycemia is increasingly recognized as a contributor to cognitive decline in diabetes. 55 Effects of hypoglycemia can be temporary or long-term. Immediately following a hypoglycemic event, diabetic patients often experience signs of cognitive deficits including irrational behavior, confusion, poor vision, and balance, etc. While these effects are temporary, there are also possible long-term irreversible cognitive effects of hypoglycemia that have been investigated.
Studies offer mixed evidence on the long-term cognitive implications of hypoglycemia. Refuting this theory, the DCCT/EDIC study followed 1441 T1D patients for 6–18 years to assess whether hypoglycemic episodes from intensive insulin therapy caused lasting cognitive decline. Results showed no significant link between hypoglycemic events and cognitive decline. The study concluded that despite an increased risk of severe hypoglycemia, intensive insulin treatment did not have harmful long-term effects on cognitive function. Despite a higher number of severe hypoglycemic events during the DCCT phase in the intensive group (653 vs 205 in the conventional group), the follow-up EDIC phase recorded similar event numbers in both groups (243 vs 254), possibly masking long-term effects of higher number of hypoglycemic comas or seizures observed in intensive treatment group when compared to the conventional treatment group during the DCCT phase of the study. 56 Another small study of adult T1D patients with retinopathy found no differences in cognitive ability between those with and without a history of recurrent severe hypoglycemia. However, the sample size was too small to draw firm conclusions. 57 Similarly, adult-onset T1D patients with and without hypoglycemic comas showed no differences in Mini-Mental State Exam scores or P300 latencies, suggesting no lasting impairment. 58 A study evaluating the long-term effect of recurrent severe hypoglycemia in adult T1D patients found that prior RH exposure does not have any effect on cognitive function. However, an interaction between RH exposure and neuropathy was found to exaggerate the extent of some neurobehavioral dysfunction. 59
In contrast, other studies highlight factors such as age, frequency, and severity of hypoglycemic events as key determinants of long-term cognitive effects. The Edinburgh Type 2 Diabetes Study reported a link between increased cognitive decline, both in the history of hypoglycemia and incident hypoglycemia during the follow-up period. 55 In contrast, data from the Outcome Reduction with Initial Glargine Intervention (ORIGIN) trial did not observe a significant link between severe hypoglycemia exposure and incident cognitive impairment. Also, the study reported a non-significant inverse relationship between non-severe hypoglycemia and incident cognitive impairment. 60
In a review article, Firer explains age as a determining factor in how severe the cognitive decline will be following hypoglycemia exposure. Children with hypoglycemic occurrences at a young age are likely to show significant cognitive decline later in life. Children with hypoglycemic occurrences at a young age also showed decreased IQ and cognitive performance later in life. 61 Another study by Hannonen et al. found similar results. 62 The study included 11 children with a history of T1D and recurrent severe hypoglycemia, 10 children with T1D without a hypoglycemic history, and 10 healthy children as controls. They found that the children with a history of severe hypoglycemia had more neurophysiological deficits and learning impairments and were more likely to need special-education measures. 62 A 7-year prospective study in pediatric T1D patients found that exposure to hypoglycemic seizures was associated with deficits in perceptual, motor, memory, and attention tasks. 63 Similar findings were reported in another pediatric study comparing intensive and conventional diabetes therapies. Children receiving intensive therapy experienced more severe hypoglycemia and showed selective impairments in spatial declarative memory and pattern recognition, potentially linked to temporal lobe dysfunction. 64 A meta-analysis of pediatric T1D studies further confirmed that recurrent severe hypoglycemia was associated with lower performance in domains such as intelligence, memory, learning, verbal fluency, and language. 65 In neonates with hypoxic-ischemic encephalopathy treated with therapeutic hypothermia, RH, particularly glucose levels below 2.6 mmol/L, was associated with poorer outcomes in cognitive, language, and socioemotional development, and a significantly higher risk of death or long-term disability. 66
Studies in adult T1D patients found that those with impaired hypoglycemia awareness and a history of coma had slower psychomotor speed, suggesting lasting cognitive consequences. 67 The acute severe hypoglycemia exposure in both normal and diabetic subjects causes cognitive impairment in terms of verbal fluency, reaction time, and verbal and visual memory.68 –70 Lincoln et al. also reported a significant correlation between frequent major hypoglycemic episodes and declines in intelligence in T1D patients. 71 Acute severe hypoglycemia has been shown to transiently impair cognitive function, with recovery taking up to 1.5 days post-episode.72,73 A meta-analysis of elderly diabetes patients (T1D and T2D) found worse general intelligence scores in those exposed to severe hypoglycemia, with T2D patients also performing worse on memory and psychomotor tests. 74 Another meta-analysis found that patients with recurrent severe hypoglycemia showed impaired memory in both T1D and T2D populations, with T2D patients also experiencing slower processing speed. However, no significant differences were noted in executive function or general intelligence between groups. 75
Interestingly, some studies suggest that prior exposure to moderate RH might protect the brain from later severe hypoglycemia. For example, Puente et al. found that moderate RH preconditioning reduced neuronal injury and cognitive decline after severe hypoglycemia. 76 However, conflicting evidence from another group showed that similar preconditioning worsened outcomes, increasing neuronal death and memory deficits. 77 Additional studies in non-diabetic rats reported that RH exposure enhanced hippocampal-dependent learning,78 –80 though the same group later found that RH improved spatial learning but impaired set-shifting ability, potentially due to reduced medial prefrontal cortex glucose metabolism. 81
Several studies by multiple groups of investigators demonstrated deleterious effects of both severe and non-severe hypoglycemia exposure on cognitive function. Moderate RH in STZ-diabetic rats led to oxidative damage in the hippocampus and cortex as well as cognitive deficits. 50 Our lab similarly found that moderate RH impaired spatial learning and memory in STZ-diabetic rats. 82 Other studies demonstrated that severe hypoglycemia exposure to STZ-diabetic mice and non-diabetic rats also causes changes in hippocampal histopathology and cognitive impairment.51,83 RH-induced cognitive decline is linked to astrocyte overactivation, reduced levels of neurotrophins, increased mitochondrial reactive oxygen species, and mitochondrial dysfunction.30,31
The effect of hypoglycemia on cognition is complex and can depend on numerous factors. The cognitive deficits seen in one patient may vary from those seen in another patient. This can be due to factors such as frequency of hypoglycemic events, severity of hypoglycemic events, age, type of diabetes, etc. Therefore, using clinical studies, it can be difficult to make broad generalizations about the effects of hypoglycemia on cognitive decline. However, well-controlled animal model studies demonstrate that despite all of these external factors, hypoglycemia is linked to cognitive deficits, both short-term and long-term. A study in T2D subjects reported decreased serum levels of neuroproteins like epiregulin (EREG), abhydrolase domain contain 14B, and inositol monophosphate 1, along with increased levels of cysteine-rich intestinal protein 2 (CRIP2) in subjects with mild cognitive impairment (MCI). Moreover, the combined assessment of EREG and CRIP2 was found to moderately differentiate patients with MCI. 84 However, future studies are required to establish the predictive value of these biomarkers in identifying the relationship between hypoglycemia exposure and cognitive impairment in diabetes.
Exposure to hypoglycemia and risk of dementia
Diabetes is a known risk factor for dementia, but the role of hypoglycemia in this association remains unclear. Huang et al. conducted a systematic review and meta-analysis of over 1.4 million diabetic patients from 10 studies, finding a 44% increased risk of dementia in those with severe hypoglycemic episodes compared to those without. 85 Notably, this analysis focused solely on dementia, excluding individuals with mild cognitive impairment. 85 Another meta-analysis of 40 studies representing over seven million patients with diabetes also found that hypoglycemia exposure was linked to increased risk of all-cause dementia (49%) and Alzheimer’s disease (31%). 86 Consistent with these findings, an earlier review article also summarized multiple studies linking hypoglycemia and increased risk of cognitive decline and dementia in patients with T2D. 49 The ADVANCE and ACCORD trials further supported this association, demonstrating that impaired cognitive function was both a consequence and a predictor of severe hypoglycemia, suggesting a bidirectional relationship where hypoglycemia contributes to cognitive decline, which in turn impairs diabetes self-management and increases the risk of further hypoglycemic episodes.87 –92
Additional evidence indicates that severe hypoglycemia may accelerate cognitive deterioration and contribute to the development of dementia, particularly in older adults. 9 A retrospective study found that exposure to one, two, or three or more severe hypoglycemic episodes was associated with progressively higher risks of dementia by 26%, 80%, and 94% compared to patients with no history of hypoglycemia, respectively. 93 This dose-dependent relationships were observed in South Korean T2D patients. 94 A cohort study in T2D patients reported that exposure to a single episode of severe hypoglycemia causes a 54% higher risk of developing dementia (hazard ratio (HR) = 1.54). This risk further increases to 80% with exposure to two or more episodes of hypoglycemia (HR = 1.80). 95 This dose-dependence was observed for Alzheimer’s disease but not vascular dementia. 95 It is plausible that higher number of hypoglycemia exposures are needed to increase the risk of Alzheimer’s disease further. While mechanisms of vascular dementia can be activated by lower number of hypoglycemia exposures and the risk plateaus without further increase by exposures to higher number of hypoglycemia exposures. Future studies are required to clarify the differential effects of hypoglycemia on these dementia subtypes.
After adjusting for multiple covariates, another cohort study in T2D patients also observed an association between the exposure to hypoglycemia and risk of dementia (HR = 1.30). 96 A retrospective propensity-score matched cohort study using the IQVIA Medical Research Data database showed that diabetic patients with a dementia diagnosis have a significantly higher risk for hypoglycemia exposure. 97 Another prospective study evaluating the potential association between exposure to hypoglycemia and dementia in older adults with diabetes observed two-fold higher risk of dementia in those who experienced hypoglycemia versus those who did not. 92 The study also reported that older adult diabetes patients with dementia had a significantly greater risk of experiencing a subsequent hypoglycemic event (HR = 3.1). 92 A study in older adults with diabetes reported that lower cognitive ability was associated with a two-fold increased risk of severe hypoglycemia over a 4-year follow-up period. 55 However, the mechanism by which impaired cognitive ability increases the risk of hypoglycemia is unknown and represent an important area for future investigation. A prospective observational study using the Korea National Diabetes Program also linked hypoglycemic episodes to increased dementia risk (HR = 2.689). 98 A Taiwanese study also reported increased crude rate ratios (RR) for dementia (age-adjusted RR = 2.76 and gender-adjusted RR = 1.6) in diabetic subjects with hypoglycemia compared to those without hypoglycemia. 99 A longitudinal cohort study that followed 2821 T1D patients also reported 66% higher risk of dementia in those who experienced severe hypoglycemic events when compared to those without (HR = 1.66). 100
A mediation analysis study that examined whether the relationship between insulin use and dementia risk is direct (independent of hypoglycemia) or indirect (mediated through hypoglycemia), observed that the insulin use is associated with dementia through the mediating effect of severe hypoglycemia. 101 Similarly, a population-based cohort study using exposure density sampling also reported significantly increased risk of dementia in T2D patients who experienced severe hypoglycemia (HR = 1.83). 102 Compared to late manifestation of severe hypoglycemia early manifestation of severe hypoglycemia contributes more to the risk of dementia in T2D patients. 103 Supporting these findings, an animal study using streptozotocin-induced diabetic APP/PS1 mice demonstrated that exposure to moderate hypoglycemia led to faster cognitive deterioration in this model of Alzheimer’s disease. 104 Overall, a growing body of clinical and experimental evidence strongly links hypoglycemia to increased risk of cognitive decline and dementia in diabetes. Table 1 summarizes key cohort and meta-analysis studies on this relationship.
Neuroinflammation and associated neurodegenerative processes are key mechanisms contributing to compromised BBB permeability in Alzheimer’s disease and other dementia subtypes.105,106 Consistent with this, pharmacological modulation that improved BBB integrity also alleviated cognitive dysfunction in a mouse model of sepsis-associated encephalopathy. 107 In addition, inhibition of β amyloid aggregation and associated inflammatory response exerts a beneficial effect on cognitive deficits in APP/PS1 mice. 108 Therefore, these findings suggest that neuroinflammation, BBB dysfunction, and protein aggregation are linked to cognitive impairment. However, whether these mechanisms contribute to RH-associated cognitive impairment in diabetes remains to be determined.
Conclusion
The current body of evidence points to hypoglycemia as a critical factor causing cognitive decline in people with diabetes. While acute episodes can cause transient cognitive deficits, recurrent and severe hypoglycemia may lead to structural brain changes, synaptic damage, mitochondrial dysfunction, and long-term cognitive decline. These effects appear to be particularly pronounced in children, older adults, and patients with impaired awareness of hypoglycemia. Importantly, the relationship between hypoglycemia and cognitive dysfunction is likely bidirectional. Cognitive impairment can reduce a patient’s ability to effectively treat diabetic hyperglycemia, thereby increasing the risk of hypoglycemic episodes (Figure 1). Emerging data link hypoglycemia exposure to a significantly increased risk of developing dementia, especially in individuals with T2D. Despite some inconsistencies in literature, the overall trend supports a detrimental effect of hypoglycemia on brain health. Moving forward, greater emphasis must be placed on preventing hypoglycemia through individualized treatment regimens, the use of continuous glucose monitoring, and patient education. Future studies should aim to identify specific mechanisms underlying hypoglycemia-induced brain dysfunction.
Footnotes
Acknowledgements
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Institutes of Health grant NS122808 and the Evelyn F McKnight Brain Institute, University of Miami. SM received funds from American Heart Association 24POST1198296. The funding organizations had no role in the design of the study, data collection, analysis, interpretation of results, writing of the manuscript, or the decision to publish the article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
