Abstract
Many studies have explored cardiac progenitor cell (CPC) therapy for heart disease. However, optimal scaffolds are needed to ensure the engraftment of transplanted cells. We produced a three-dimensional hydrogel scaffold (CPC-PRGmx) in which high-viability CPCs were cultured for up to 8 weeks. CPC-PRGmx contained an RGD peptide-conjugated self-assembling peptide with insulin-like growth factor-1 (IGF-1). Immediately after creating myocardial infarction (MI), we transplanted CPC-PRGmx into the pericardial space on to the surface of the MI area. Four weeks after transplantation, red fluorescent protein-expressing CPCs and in situ hybridization analysis in sex-mismatched transplantations revealed the engraftment of CPCs in the transplanted scaffold (which was cellularized with host cells). The average scar area of the CPC-PRGmx-treated group was significantly smaller than that of the non-treated group (CPC-PRGmx-treated group = 46 ± 5.1%, non-treated MI group = 59 ± 4.5%; p < 0.05). Echocardiography showed that the transplantation of CPC-PRGmx improved cardiac function and attenuated cardiac remodeling after MI. The transplantation of CPCs-PRGmx promoted angiogenesis and inhibited apoptosis, compared to the untreated MI group. CPCs-PRGmx secreted more vascular endothelial growth factor than CPCs cultured on two-dimensional dishes. Genetic fate mapping revealed that CPC-PRGmx-treated mice had more regenerated cardiomyocytes than non-treated mice in the MI area (CPC-PRGmx-treated group = 0.98 ± 0.25%, non-treated MI group = 0.25 ± 0.04%; p < 0.05). Our findings reveal the therapeutic potential of epicardial-transplanted CPC-PRGmx. Its beneficial effects may be mediated by sustainable cell viability, paracrine function, and the enhancement of de novo cardiomyogenesis.
Introduction
Myocardial infarction (MI) and congestive heart failure resulting from post-MI remodeling remain the leading causes of death in the developed world 1 . As adult cardiomyocytes have low proliferation rates 2 and low turnover rates 3 , conventional medical or surgical therapeutic options are limited. Cardiac transplantation is the major curative treatment for end-stage heart failure, but it is only available to a limited number of patients due to organ donor shortages 4 . Cell-based therapy has recently emerged as a viable alternative for heart failure treatment5,6. Clinical reports have shown that transplanted cells improve the function of the ischemic heart. However, other clinical studies have failed to reproduce these favorable findings7–9. A long-term follow-up of randomized clinical trials failed to prove a sustained improvement in heart function10,11.
Recently, a growing body of evidence is suggesting that the beneficial effects of transplanted cells on heart function and remodeling mainly occur via paracrine effects, rather than via the physiologically reverent regeneration of myocardium. Moreover, broad analyses of bioactive paracrine molecules have become more important than ever12,13. However, transplanted cells often suffer from a low engraftment rate, which stems from leakage and washout during injections, and massive cell death. This lessens the efficient release of paracrine factors from the transplanted cells within the host tissue14,15. Biological scaffolds are expected to circumvent the loss of grafted cells, as they provide a three-dimensional (3D) microenvironment for cells and support their survival, proliferation, and function 16 . Many kinds of scaffolds, such as natural protein-based matrices, synthetic polymers, and hydrogels, have been designed to date. However, the clinical application of cardiac cell-based tissue engineering approaches is currently in a premature stage because of complications in matching them to engrafted cells and their compatibility with host tissue 17 .
When injected with self-assembling peptides after MI, cardiac progenitor cells (CPCs) have been reported to be the most effective cells for reducing infarct size and preventing cardiac dysfunction among various types of cells, such as bone marrow mononuclear cells, skeletal myoblasts, and adipose tissue-derived mesenchymal cells 18 . The mechanisms of these beneficial effects have been shown to be partially mediated by paracrine factors such as vascular cell adhesion molecule 1 (VCAM-1) and junctional adhesion molecule A (JAM-A). These factors are secreted in large amounts from CPCs19,20. Polymerized nanoparticles can be used to form hydrogels in situ. CPCs can be entrapped in these hydrogels to prevent cell death within the harsh environment that occurs during the acute phase of MI. This suggests that self-assembling peptides can function as bioscaffolds in a manner that is compatible with CPCs. However, the injectable nanopeptide-CPC system is limited by the number of engrafted cells that it can produce and its ability to withstand mechanical stress in the heart. Therefore, we constructed a 3D thick scaffold (CPC-PRGmx), in which CPCs were cultured in combination with insulin-like growth factor-1 (IGF-1) and RGD-binding peptide-modified self-assembling peptides (PuramatrixTM [PM], 3-D MATRIX, Tokyo, Japan). We examined whether a pericardially delivered cardiac patch could be engrafted and if it could maintain the survival and function of CPCs. Moreover, we examined whether this approach could prevent left ventricular remodeling after MI, while stimulating the endogenous regeneration of cardiomyocytes.
Materials and Methods
Animals
Wild mice (C57BL/6NCr, 11 weeks old) were purchased from Japan SLC (Shizuoka, Japan). Double-transgenic mice (CreLacZ mice) were obtained by crossbreeding transgenic mice, as previously described 21 . Briefly, mice-harboring tamoxifen-inducible Cre recombinase proteins fused to two mutant estrogen receptor ligand-binding domains (MerCreMer), which were expressed under the control of the α-myosin heavy chain promoter (provided by Dr. Jeffery D Molkentin, Cincinnati Children’s Hospital Medical Center, Cincinnati, OH, USA), were crossbred with mice that harbored the cytomegalovirus immediate early enhancer chicken β-actin hybrid promoter (CAG)-chloramphenicol acetyl transferase (CAT)-LacZ. In these mice, β-galactosidase was expressed following the removal of a loxP-flanked stop sequence (provided by Dr. Jyunichi Miyazaki, Osaka University Graduate School of Medicine, Osaka, Japan)22,23. Cre recombination was induced by intraperitoneal (ip) injection of CreLacZ mice daily with tamoxifen (Sigma−Aldrich, St Louis, MO, USA). This was dissolved at 4 mg·mL−1 in corn oil (Sigma−Aldrich), at a dosage of 5 mg·kg−1 body weight, for 14 consecutive days. All strains were maintained on a C57BL/6NCr genetic background after backcrossing. Genotyping was performed by polymerase chain reaction (PCR) using tail deoxyribonucleic acid (DNA) with the following primers: MerCreMer forward, 5′-GTTCGCAAGAACCTGATGGACA-3′; MerCreMer reverse, 5′-CTAGAGCCTGTTTTGCACGTTC-3′; LacZ forward, 5′-CCTTTCGCCAGCTGGCGTAATAGCG-3′; and LacZ reverse, 5′-ACCGTGCATCTGCCAGTTTG-3′.
Preparation of 3D Cellularized Scaffold
CPCs are cell-lined CPCs that were established from adult mouse heart stem cell antigen-1-positive cells, as previously reported19,24. Briefly, a heart of an adult C57Bl/6 mouse (10–12 weeks old) was enzymatically dissociated into a single-cell suspension, and enrichment of stem cell antigen-1 (Sca-1)-positive cells was achieved by sorting using the magnetic cell sorting system (MCAS; Miltenyl Biotec, Sunnyvale, CA). Whole primary cell suspension was incubated with PE-conjugated anti-Sca-1 antibody for 10 min on ice, washed in phosphate-buffered saline (PBS) supplemented with 3% fetal bovine serum (FBS), incubated with anti-PE micro beads for 15 min at 4°C, and washed with PBS supplemented with 3% FBS. The samples were passed through an MACS column set up in a Miltenyl magnet, and the Sca-1 cells were eluted from the column by washing with PBS supplemented with 3% FBS. To increase the purity of the Sca-1 cells, magnetic sorting was performed one more time. Since primary isolated Sca-1-positive cells derived from adult murine hearts consisted of several populations including cardiac stem/progenitor cells, hematopoietic cells, and endothelial cells 24 , we first established the clonal cells from cardiac Sca-1-positive cells. We cultured 104 primary isolated Sca-1-positive cells derived from adult murine hearts on 10-cm dishes. After repeating limited dilution, we established clonal cell lines. The efficiency of the cloning was ~0.1%. The clonal cells can be expanded for more than 500 population doublings. Flow cytometric analysis revealed that almost 100% of cells expressed Sca-1, CD29, and CD44, ~20% expressed CD34, and no cells expressed CD31, CD45, and c-kit through passages. The clonal cells expressed cardiac transcription factors such as Nkx2.5 and GATA4 but not cardiac contractile proteins under culture conditions of 80% confluence. Gene profiles and cell surface marker analysis revealed that CPCs were similar to those of primary isolated Sca-1-positive cells previously reported 24 . Thus CPC possessed almost identical features to the intrinsic cardiac stem/progenitor cells. When CPCs were cultured under confluent conditions for 4 weeks, expression levels of Nkx2.5 and GATA4 were upregulated, and expression of myocyte enhancer factor 2C (MEF2C), atrial natriuretic peptide (ANP), alpha-myosin heavy chain (MHC) and sarcomeric alpha-actinin was detected at the mRNA and protein level 19 . Self-assembling peptides (PM and peptide-modified designer PM (PRG-, SDP-, and PFS-conjugated PM) were kindly provided by 3-D Matrix, Ltd. (Tokyo, Japan). PRGDSGYRGDS (PRG) is a collagen VI-derived functional motif, which consists of a two-unit RGD-binding peptide with two similar RGD-binding sequences, PRGDS and YRGDS 25 . SDPGYIGSR (SDP) is a laminin-derived functional motif 26 . PFSSTKT (PFS) belongs to the family of bone marrow homing peptides 27 . PM and peptide-modified designer PM were dissolved in deionized water and sonicated before use. PM was mixed with PRG-, SDP-, or PFS-conjugated PM in different mix ratios; thereby, making hydrogels with various concentrations of peptide-modified designer PM (PRGmx, SDPmx, and PFSmx, respectively). The cells were suspended in 295 mM sucrose solution (10% w/v) and mixed with deionized water containing 1% PM, PRGmx, SDPmx, or PFSmx at a ratio of 1:1. Therefore, the final concentration of self-assembling peptides was 0.5% (W/V). In some experiments, biotin-conjugated PM (TORAY Industries, Inc., Tokyo, Japan) was substituted with PM to detect transplanted PM. A volume of 100 µL of unpolymerized scaffold solution containing 2 × 105 cells·µL–1 of CPCs was placed onto the cell culture insert (BD Biosciences, Franklin Lakes, NJ, USA). This was then placed in a 24-well culture plate containing Iscove’s modified Dulbecco’s medium (IMDM; Thermo Fisher Scientific, Inc. Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS; Takara Bio USA Inc. Madison, WI, USA), and penicillin/streptomycin (Thermo Fisher Scientific, Inc.). In some experiments, 100 ng·mL-1 of insulin-like growth factor 1 (IGF-1; Sigma-Aldrich, St Louis, MO, USA)—was added to prepare a graftable CPC-scaffold complex. After 10 min, 100 µL of the medium was gently added to the top portion of the gelled PM-cell mixture, and the medium outside of the cell culture insert was changed. In the next 30 min, the medium was changed twice and then once every 3 d. Red fluorescent protein (RFP)-expressing retroviral stocks were generated as previously described 28 . CPCs were infected with the RFP-expressing retroviral vector and selected for growth in the presence of 500 µg·mL-1 neomycin (Sigma-Aldrich) for two weeks. After encapsulation in PM, RFP-positive (RFP+) CPCs were maintained in the medium with 500 µg·mL-1 neomycin until transplantation. The transfection efficiency of the RFP was >90%.
Animal Surgery
CreLacZ mice were subjected to MI 3 days after the 14th tamoxifen injection. The induction of anesthesia was performed primarily with pentobarbiturates, and isoflurane was considered for the maintenance of the anesthesia. The depth of anesthesia was assessed by measuring vital signs, including blood perfusion and oxygenation through evaluation of overall skin color of the ears, tail, and paw pads; respiratory rate by counting the rise and fall of the chest in 1 minute; heart rate by ECG systems. To confirm unconsciousness and a lack of pain perception, we used paw withdrawal to assess mouse anesthetic depth. The paw withdrawal response was confirmed just prior to making a surgical incision and be monitored regularly throughout the anesthetic and surgical procedure. Additionally, post-operative pain assessment was done by documenting weight; noting changes in activity, food, and water intake. MI was produced by ligation of the left anterior descending artery, 1 mm distal to the left atrial appendage, with an 8-0 Prolene suture. Occlusion was confirmed by observing immediate blanching of the left ventricle at the post-ligation site and an accompanying electrocardiogram (ECG) recording that showed corresponding ST-elevation. Mice with major technical problems including failure of intubation, severe bleeding, lung damage, or coronary artery damage; any signs of hypotension before the procedures; ST-elevation less than 0.2 mV on ECG; heart rate <300 bpm; persistent dysrhythmia; outside the weight range of 32 to 48 g; or outside the age range of 12 to 15 weeks were excluded from the final analysis. In addition, we reconfirmed the successful ligation at the timing of sacrifice.
Immunocytochemistry and Immunohistochemistry
The CPC-scaffold complex was fixed for 15 min at room temperature in 4% paraformaldehyde (Wako, Osaka, Japan) in phosphate-buffered saline (PBS) and rinsed with PBS. After permeabilization with PBS containing 0.2% NP40 (Nacalai Tesque, Kyoto, Japan), the hydrogels were stained with rhodamine phalloidin (Thermo Fisher Scientific, Inc.) overnight at 4 °C. Nuclei were stained with 4′, 6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich). To analyze cell viability, the fixed CPC-scaffold complex was treated with a LIVE/DEAD Viability/Cytotoxicity kit (Thermo Fisher Scientific, Inc.). The TUNEL-positive apoptotic CPCs in the scaffold were detected using the In Situ Cell Death Detection Kit, TMR red (Roche Applied Science, Penzberg, Germany).
Paraffin-embedded heart tissues were sectioned at 5 mm thickness, deparaffinized, and stained overnight at 4 °C using anti-Von Willebrand factor (vWF; DAKO, Glostrup, Denmark). To detect TUNEL-positive cells, fresh frozen sections (4 µm) were fixed with cold acetone (Wako) for 5 min at 4 °C and TUNEL staining was performed using an in situ apoptosis detection kit (TAKARA BIO Inc. Kusatsu, Japan). Antibodies were detected using the VECSTATIN ABC kit (Vector Laboratories Inc., Burlingame, CA, USA). Nuclei were stained with hematoxylin (Sakura Finetek Japan, Tokyo, Japan). For fluorescent immunohistochemistry, fresh frozen sections (4 µm) were fixed with 4% paraformaldehyde in PBS or cold acetone. After blocking with PBS containing 2% donkey serum (Jackson ImmunoResearch Laboratory, West Grove, PA, USA), 0.2% NP40, 2% bovine serum albumin (Sigma-Aldrich),− Cy-3-conjugated avidin (Jackson ImmunoResearch Laboratory), mouse monoclonal anti-SA-actinin (Sigma-Aldrich),− rabbit polyclonal connexin40 (Merck Millipore, Darmstadt, Germany), rabbit polyclonal anti-laminin (Sigma-Aldrich),− or rabbit polyclonal anti-RFP (MBL, Nagoya, Japan) antibodies were applied to paraformaldehyde-fixed sections. For acetone-fixed sections, rat monoclonal anti-CD31 (eBioscience, San Diego, CA, USA), mouse monoclonal anti-smooth muscle actin (SMA; Sigma-Aldrich), or rat monoclonal anti-Ly6G (BD Pharmingen, Franklin Lakes, NJ, USA) antibodies were used. The samples were subsequently washed three times in PBS, and Alexa Fluor 488- (Thermo Fisher Scientific, Inc.) or Cy3-conjugated (Jackson ImmunoResearch Laboratory) secondary antibodies were used to visualize the expression of specific proteins. Actin staining was performed with rhodamine or Alexa Fluor 488 phalloidin (Thermo Fisher Scientific, Inc.). Nuclear staining was performed using DAPI (Sigma-Aldrich). To examine β-galactosidase expression, the samples were incubated for 20 h after secondary antibody incubation in a 0.1 mM phosphate buffer (pH 7.3) solution of 1 mg·mL-1 X-gal chromagen (Takara), 5 mM K3Fe(CN)6, 5 mM K4Fe(CN)6, 2 mM MgCl2, 0.01% sodium deoxycholate, 0.01% NP40, and 20 mM Tris-HCl (pH 7.3). All staining procedures using a detection kit were performed according to the manufacturer’s protocol. Images of cells and tissues were obtained using a laser confocal microscope (TCS-SP5, Leica Microsystems, Wetzlar, Germany) or a fluorescence microscope (Axioskop 2 plus, Zeiss, Oberkochen, Germany).
Infarct Size Measurement and Myocyte Quantification
Hearts were fixed in 10% PBS-buffered formalin (Wako) and embedded in paraffin. Then, several transverse sections (5 µm) were collected every 0.4 mm from the apex, mid, and base. The fibrous infarct area was detected visually by Masson trichrome staining (Muto Pure Chemicals Co., Ltd, Tokyo, Japan). The lengths of the scar and non-infarcted muscle were measured for both the endocardial and epicardial surfaces of each histological section. The final infarct size was expressed as a percentage of the average endocardial and epicardial scar lengths and surface circumferences multiplied by 100 29 . Myocyte numbers were quantified according to the method reported by Jiang et al. 30 , with minor modifications. Heart sections were double-stained with laminin and SA-actinin; they were imaged using a fluorescence microscope (Zeiss). The images, in which the plasma membrane boundaries were demarcated by laminin, were processed using Axiovision software (Zeiss). Individual cardiomyocytes were highlighted as particles and resolved as distinct particles that were close together. The number of particles was automatically measured as the number of myocytes. Particles were included based on the size of cardiomyocytes (ranging from 50 to 3,000 pixels·cell-1); non-myocytes were excluded by size, and by the absence of SA-actinin staining.
Echocardiography
Echocardiography in spontaneous breathing mice was performed using a Vevo770 (Visual Sonics, Toronto, Canada) with a 25-MHz imaging transducer. Two-dimensional (2D) images and M-mode tracing were recorded from the parasternal long-axis view at the midpapillary level to determine LVIDD and LVISD. %FS was calculated as %FS = [(LVIDD—LVISD) / LVIDD]×100. We further confirmed the anterior wall motion abnormality by both B-mode long and short axis views. We evaluated the cardiac function 14 days and 28 days after MI.
MTT Assay
CPCs encapsulated in PM, PRG-, SDP-, or PFS-conjugated PM were cultured in IMDM supplemented with 0.2% FBS for 72 h, in the presence or absence of 500 ng·mL-1 of IGF-1. After aspirating the medium, the cells were washed with PBS once, and PBS was added (400 μL·well 1 ). After 2 h of incubation with 5 mg·mL-1 MTT (Sigma-Aldrich) solution, 10% sodium dodecyl sulfate (SDS) solution was added and incubated overnight. The OD570 of the supernatant was measured.
Enzyme-Linked Immunosorbent Assay (ELISA)
Cardiac mesenchymal cells (CMCs) were isolated according to a previously reported method 31 . Briefly, 11-week old C57BL/6NCr mice hearts were cut into four pieces and digested in trypsin- Hanks' Balanced Salt Solution (HBSS; Thermo Fisher Scientific, Inc.) at 4 °C, followed by digestion with collagenase type II (Worthington Lakewood, NJ, USA) in HBSS at 37 °C. The CMCs were separated from myocytes and endothelial cells by differential plating on plastic dishes. They were then cultured in Dulbecco’s modified eagle medium (DMEM) containing penicillin/streptomycin, 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES; 25 mM), glutamine (2 mM), and 10% FBS until confluency was reached. To obtain conditioned medium (CM) of CPCs and CMCs, cells were cultured in PRGmx or on gelatin-coated dishes in IMDM supplemented with 10% FBS and penicillin/streptomycin for 4 days. The prepared cells were washed with PBS three times, and the medium was changed to serum-depleted IMDM. After 2 days, the supernatant was collected as CM. The concentrations of vascular endothelial growth factor (VEGF), JAM-A, and soluble vascular adhesion molecule 1 (VCAM-1) were measured using a Quantikine mouse ELISA kit (R&D Systems, Minneapolis, MN, USA). The volume of supernatant and number of residual cells in PRGmx, or on the dishes, were measured to calculate the amount of cytokines secreted from 105 cells.
Fluorescence in situ Hybridization (FISH) Analysis
The CPC-scaffolds were transplanted into the pericardial sac of the infarcted hearts of wild-type female mice. Four weeks after transplantation, the mice were sacrificed, and whole hearts were snap-frozen in nitrogen. Frozen sections were fixed with 4% paraformaldehyde in PBS overnight and subjected to FISH analysis (Chromosome Science Labo Inc., Sapporo, Japan). The sections were digested with 0.004% pepsin/0.1 M HCl solution for 3 min. They were then dehydrated by exposure to a graded series of ethanol solutions. After applying mouse X and Y FISH probes, the sections were denatured by incubation at 70°C for 10 min and hybridized at 37°C overnight. Hybridized sections were washed with 50% formamide at 37°C. X FISH probes were detected using Cy3-conjugated anti-digoxin antibodies, and Y FISH probes were Alexa488 conjugated anti-fluorescein isothiocyanate (FITC) antibodies. Nuclei were stained with DAPI.
Statistical Analysis
The normality and variance of data were analyzed and adjusted with a p value of 0.05, as rejection criteria. Results are presented herein as means ± standard error (SE). Statistical significance was calculated using unpaired Student’s t-tests for comparisons between two groups, or by one-way analysis of variance (ANOVA) Turkey–Kramer post hoc tests for multiple comparisons. In cases where the data were not normally distributed and/or the variance was not homogeneous, the significance was calculated using Mann–Whitney U-tests for comparisons between two groups, or by Kruskal–Wallis tests (followed by Steel–Dwass tests) for multiple comparisons. Statistical analysis was performed using Microsoft Excel with the add-in software Statcel3 (OMS, Japan). Statistical significance was set at p < 0.05.
Results
PRGmx, in Combination With IGF-1, Prevents CPC Cell Death in a 3D-Scaffold
To determine the optimal mixture ratio of peptide-modified PM and pure PM, we cultured CPCs in PRGmx, PFSmx, or SDPmx, consisting of different mixture ratios of peptide-modified PM and pure PM, for 3 days. When PRG was increased from 0% to 30% in PRGmx, there was an increase in cell viability (as determined by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide [MTT] assay). Cell viability reached a plateau between 30% and 70% PRGmx, before dropping at 100% PRGmx (Fig. 1A). For PFSmx, cell viability was proportional to the PFS mixture ratio (Fig. 1B); adding SDP had no detectable effect on cell viability (Fig. 1C). These results suggest that ~50% and 100% mixture ratios are optimal for PRGmx and PFSmx, respectively. However, PFSmx hydrogel containing more than 70% PFS was fragile and incompatible for 3D culturing.
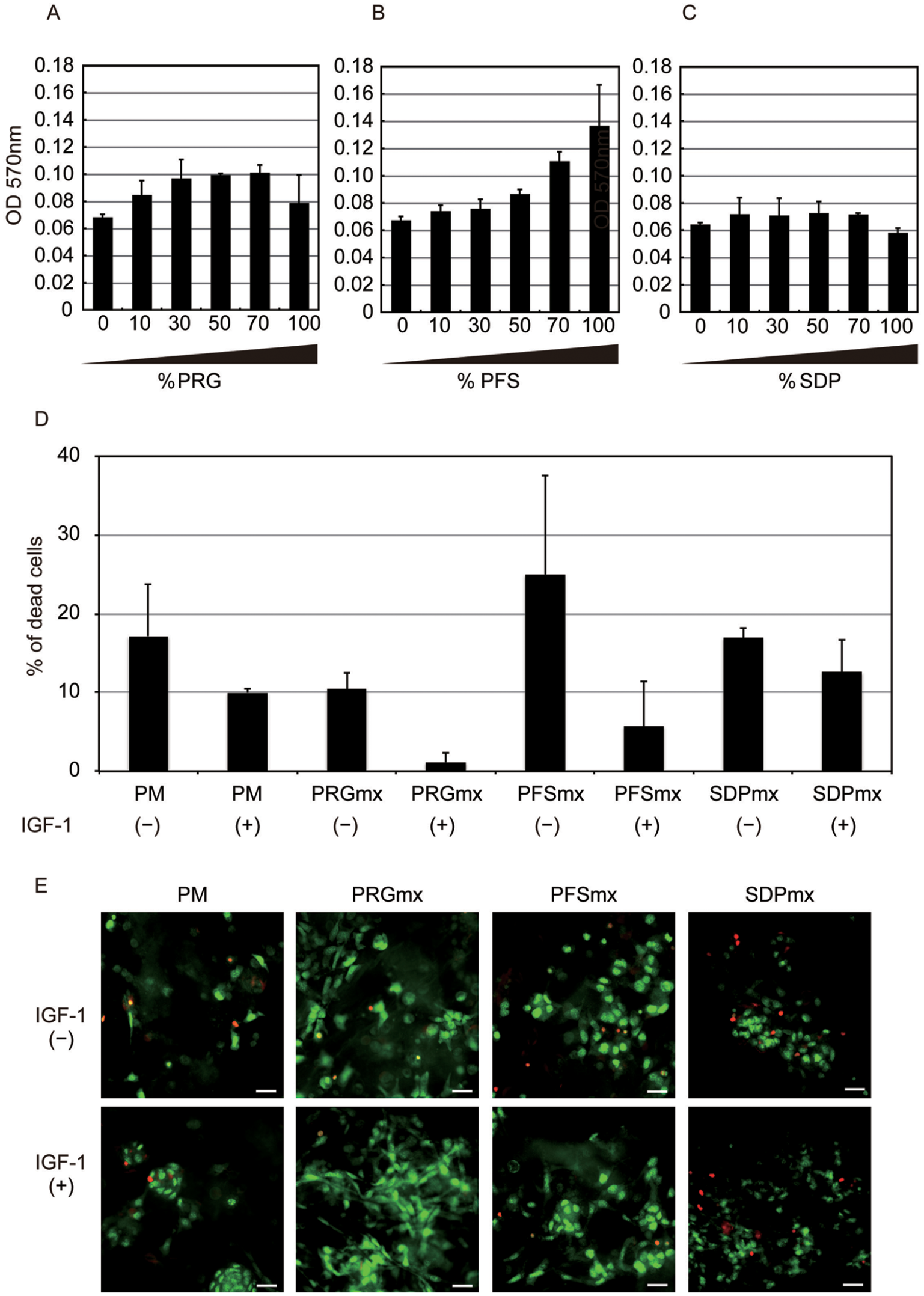
Effects of peptide-modified peptides and IGF-1 on CPC viability: (A)-(C) Cell viability in different scaffolds with different mix ratios of (A) PRG, (B) PFS, and (C) and SDP after 3 days of culture. Numerical values on horizontal axis indicate mixture ratio; vertical axis shows optical density at 570 nm. Averaged values of three samples are shown. (D) Effect of IGF-1 on viability of CPCs cultured in PM, PRGmx, PFSmx, or SDPmx (mixture ratio = 50%). Results are expressed as percentage of dead cells to total cells. Data are representative of three independent experiments. Similar results were observed in three independent experiments. (E) live/dead staining fluorescent images. Viable (green) and non-viable (red) cells are shown; scale bars = 20 μm. CPC: cardiac progenitor cell; IGF-1: insulin-like growth factor-1.
When CPCs were cultured in PM, 50% PRGmx, 50% PFSmx, or 50% SDPmx for 3 days, IGF-1 treatment reduced the frequencies of dead cells across all scaffolds, particularly for PRGmx (Fig. 1D). Live/dead staining revealed few red dead CPCs and large numbers of green live CPCs in PRGmx with IGF-1 (Fig. 1E). The beneficial effects of PRGmx and IGF-1 on CPCs cultured in a 3D environment were further evaluated by terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining and MTT assays. TUNEL-positive apoptotic CPCs were more common in PM than in the other groups (45.5 ± 6.9% in PM, 17.5 ± 5.6% in PRGmx, 17.6 ± 6.8% in IGF-1-treated PM, and 6.2 ± 1.1% in IGF-1-treated PRGmx, *p < 0.05, **p < 0.01; Fig. 2A). We identified a substantial number of TUNEL-positive (red) CPCs in PM (Fig. 2B, upper left), with significantly fewer TUNEL-positive CPCs in PM with IGF-1 and PRGmx with or without IGF (Fig. 2B, lower left, upper right, and lower right, respectively). Notably, a substantial number of actin-positive (green) elongated CPCs were present in IGF-treated PRGmx. MTT assays revealed that IGF-1 treatment enhanced cell proliferation in CPCs cultured in PM (Fig. 2C). Although PRGmx could not enhance cell proliferation, combining PRGmx with IGF-1 had a remarkable synergic proliferative effect on CPCs (1.4 ± 0.12% in PRGmx, 2.9 ± 0.28% in IGF-1-treated PM, 4.8 ± 0.99% in IGF-treated PRGmx; cell proliferation expressed as fold change relative to PM, *p < 0.05; Fig. 2C). Actin staining revealed that most CPCs cultured in PRGmx, but not PM, showed elongated morphology, irrespective of IGF treatment (Fig. 2D). The average cell surface area of CPCs cultured in PRGmx was significantly larger than that of CPCs cultured in PM (Fig. 2E). IGF-1 failed to facilitate efficient cell spreading in each peptide scaffold (188 ± 24.1 μmm2 in PM, 336 ± 39.1 μmm2 in PRGmx, 184 ± 31.7 μmm2 in IGF-1-treated PM, and 334 ± 47.2 μmm2 in IGF-1-treated PRGmx, *p < 0.05; Fig. 2D). These findings suggest that PRGmx promotes cytoskeletal formation and the spreading of CPCs. Moreover, IGF-1 synergistically protects against massive cell death and apoptosis of CPCs grown in 3D culture. Therefore, we used PRGmx (with 50% PRG and 50% pure PM) with IGF-1 to construct a transplantable CPCs-PRGmx for subsequent experiments.

Synergistic effects of PRGmx and IGF-1 on CPC viability and spreading. (A) Percentages of TUNEL-positive apoptotic CPCs cultured in PM, PRGmx, PM with IGF-1, or PRGmx with IGF-1 (mixture ratio of PM to PRG-conjugated PM = 50%). (B) Representative TUNEL staining images of CPCs under each culture condition (TUNEL staining shown in red). CPCs were co-stained with Alexa-fluor 488 phalloidin for actin (green) and DAPI for nuclei (blue); scale bars = 100 μm. (C) MTT viability assay of CPCs cultured in PM, PRGmx, PM with IGF-1, or PRGmx with IGF-1. (D) Morphology of CPCs cultured in PM, PRGmx, PM with IGF-1, or PRGmx with IGF-1. CPCs were stained with rhodamine phalloidin (red) for actin and DAPI (blue) for nuclei; scale bars = 30 µm. (E) Average cell surface area in each culture condition is expressed via pixel area of each fluorescent image; *p < 0.05. CPC: cardiac progenitor cell; DAPI: 4′,6-diamidino-2-phenylindole; IGF-1: insulin-like growth factor-1; PM: Puramatrix.
Cellularized Pericardial Scaffold Retains Transplanted CPCs, Accompanying Scaffold Degradation
When cultured for 8 weeks, CPCs-PRGmx resembled a cuboid semi-lucent hydrogel (Fig. 3A). Rhodamine phalloidin and 4′,6-diamidino-2-phenylindole (DAPI) staining revealed that CPCs were uniformly distributed over the scaffold (Fig. 3B). After ligation of the left anterior descending artery, an incision was made in the parietal pericardium, creating a perforation (Fig. 3C, left and middle, white arrowheads). A trimmed CPC-PRGmx was then transplanted into the pericardial sac through said perforation; this was visible as white semi-opaque material on the anterolateral wall of the left ventricle (Fig. 3C, middle, white arrow). A perforation of the parietal pericardium was closed with a 10-0 Prolene suture (Fig. 3C, right, black arrow).
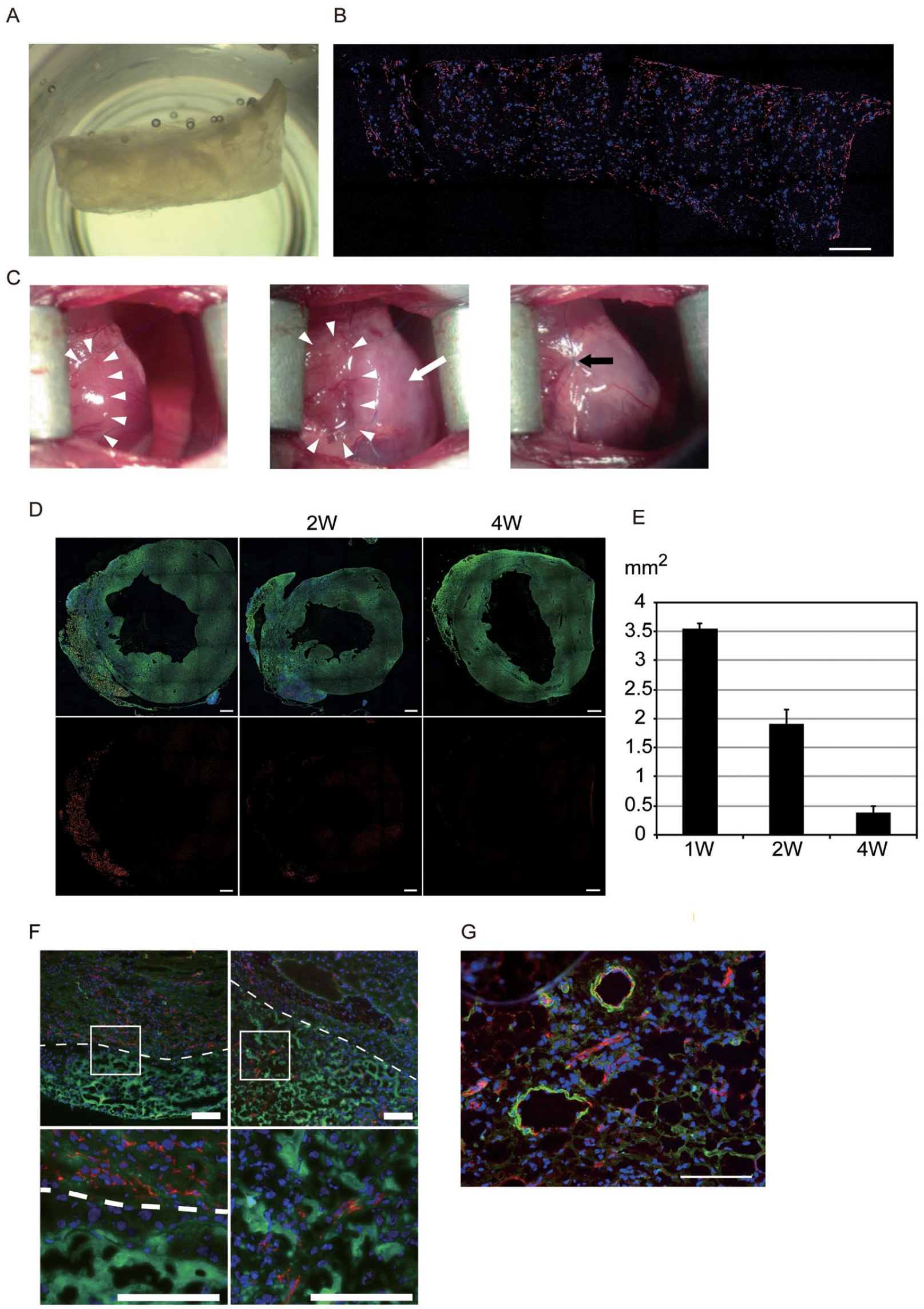
Effect of coordinate degradation of PM after transplantation on new tissue organization in CPC-PRGmx. (A) Front view of CPC-PRGmx after culturing for 8 weeks. (B) Immunofluorescent image of CPC-PRGmx shown in (A) with staining for actin (red) and nuclei (blue); scale bar = 500 μm. (C) Delivery of CPC-PRGmx into pericardial space. Small incision was made in parietal pericardium to create a perforation (left panel, white arrowheads). Edge of parietal pericardium was lifted and CPC-PRGmx was delivered into pericardial space through the perforation (middle panel, white arrowheads). CPC-PRGmx resembles semi-lucent material (white arrow). Perforation of parietal pericardium was sutured (black arrow) and then CPC-PRGmx was fixed, covering anterolateral wall of left ventricle. (D) Macro-images of hearts transplanted with CPC-PRGmx containing biotin-conjugated PM. Frozen heart sections were stained for SA-actinin (green) and nuclei (blue; upper panels). PM detected using Cy3-conjugated avidin (red; upper and lower panels); scale bars = 500 µm. (E) Quantitative analysis results of biotin signal-positive area. Three heart sections were examined per mouse. Values are averages from two mice for each group. (F) Immunofluorescent images of borderline between transplanted CPC-PRGmx and host myocardium after 3 days (left panels) and 7 days (right panels) post-transplantation. Sections were stained for CD31 (red) and nuclei (blue). PM resembles amorphous substance with green auto-fluorescence; scale bars = 100 µm. (G) Immunofluorescent image of epicardial patch area 4 weeks post-transplantation. Sections were stained for CD31 (red), SMA (green), and nuclei (blue); scale bars = 100 µm. CPC: cardiac progenitor cell; PM: Puramatrix; SMA: smooth muscle actin.
To quantify scaffold degradation in vivo, CPC-PRGmx, in which PM was replaced with biotin-labeled PM, was transplanted. The heart sections were stained with SA-actinin antibody, followed by Alexa Fluor 488-labeled secondary antibody and Cy3-conjugated avidin. One week after transplantation, the fluorescent red signal derived from biotin-labeled PM was co-localized with an epicardial patch covering the infarct area, characterized by wall thinning (Fig. 3D, left). The red signal decreased over the implantation time, although the epicardial patch (reminiscent of the former PM scaffold) remained (Fig. 3D, middle [2 weeks] and right [4 weeks]). Fig. 3E shows quantitative analysis of the biotin signal-positive area (3.5 ± 0.1 μmm2 after 1 week, 1.9 ± 0.3 μmm2 after 2 weeks, and 0.4 ± 0.1 μmm2 after 4 weeks). Three days after transplantation, the epicardial patch was occupied by PM, showing a non-specifically stained amorphous structure (Fig. 3F, left). Seven days after transplantation, a substantial number of DAPI-positive cellular nuclei and CD31-positive capillaries were detected between the sparse PM (Fig. 3F, right). The epicardial patch was further cellularized and capillarized, and some SMA-positive arterioles were observed 4 weeks after transplantation (Fig. 3G). These findings suggest that coordinated PM degradation, combined with tissue ingrowth within the scaffold, led to the engraftment of the stable epicardial patch.
Next, we examined whether PRGmx supported the engraftment of CPCs in vivo by using fluorescent probes or sex-mismatched transplantation. After the transplantation of red fluorescent protein (RFP) + CPC-PRGmx, many RFP + CPCs were observed in the scaffold area, which was detected as a nonspecific Alexa Fluor 488-stained area (Fig. 4A, left three panels in upper and middle row) or as an epicardial patch after hematoxylin and eosin (HE) staining (Fig. 4A, left three panels in bottom row). However, only a faint RFP signal was detected 3 days after the direct injection of PRGmx with RFP + CPCs (Fig. 4A, right panels). The numbers of RFP-positive CPCs per graft area decreased during implantation, and RFP signals were not observed in animals sacrificed 4 weeks after transplantation (Fig. 4B). When male CPC-PRGmx was transplanted to the infarcted heart of female mice, many cells in the graft area possessed a pair of X and Y chromosomes 4 weeks after transplantation (Fig. 4C, arrowheads). Cells in the myocardium, distant from the graft area, only possessed X chromosomes (Fig. 4C, double arrows). We observed a few cells with three X chromosomes and one Y chromosome in the nucleus, suggesting that cell fusion occurred between host- and donor-derived cells (Fig. 4C, arrows and inlet). These findings suggest that PRGmx supports the survival of CPCs in scaffolds cellularized with host-derived cells.

Engraftment of PRGmx scaffold embedded with CPCs in a mouse MI model. (A) Representative immunofluorescent images of sections of RFP + CPC-PRGmx-transplanted heart. Upper row shows magnified images of engrafted CPC-PRGmx obtained 3 days, 1 week, and 2 weeks post-transplantation (left three panels); right panel shows immunofluorescent image of needle-injured area 3 days post-CPC injection. Corresponding immunofluorescent and HE staining macro images are shown in middle and bottom rows, respectively. White squares indicate area where magnified image was taken. For immunohistochemistry, sections were stained for RFP (red) and nuclei (blue). PRGmx scaffolds were nonspecifically stained with Alexa-fluor 488-conjugated anti-mouse antibodies; scale bars = 500 µm. (B) Quantitative analysis of RFP + cells in section at indicated time. Three heart sections were examined per mouse. Values are averages from two mice for each group. (C) Results of FISH analysis; upper two panels show immunofluorescent images obtained from graft areas indicated by white square in macro image. Transplanted CPCs containing a pair of X (red) and Y chromosomes (green) in nuclei (arrowheads in upper two panels), and fused cells containing three X chromosomes (red) and a Y chromosome (green) in nuclei (arrows) were detected. Inset shows magnified image of fused cell (arrow). Lower right panel shows immunofluorescent image obtained from remote area indicated by white square in macro image. Host-derived cells containing X chromosomes (red) in nucleus (double arrows) were observed. Scale bars = 500 µm for macro image, 10 µm for magnified images. CPC: cardiac progenitor cell; FISH: fluorescence in situ hybridization; MI: myocardial infarction; RFP: red fluorescent protein.
Transplantation of CPC-PRGmx Attenuates Left Ventricular Remodeling by Enhancing Angiogenesis and Reducing Apoptotic Cell Death
Four weeks after transplantation, the CPC-PRGmx-treated group exhibited significantly lower fibrosis levels than the non-treated MI group (Fig. 5A). The average scar areas in the non-treated MI and CPC-PRGmx-treated groups were 59 ± 4.5% (n = 18) and 46 ± 5.1% (n = 17), respectively (Fig. 5B, p

Transplantation of CPC-PRGmx attenuates left ventricular remodeling by enhancing angiogenesis and reducing apoptotic cell death and neutrophil accumulation. (A) Representative Masson’s trichrome-stained myocardial sections from non-treated MI heart (MI, left) and CPC-PRGmx-treated heart (CPC-PRGmx, right); scale bars = 2.5 mm. (B) Quantification of the infarct size 4 weeks post-transplantation. Data show means ± standard error (SE; *p < 0.05; non-treated MI group [MI] versus CPC-PRGmx-treated group [CPC-PRGmx]). (C) Representative images from sections of non-treated (MI, left) and CPC-PRGmx-treated (CPC-PRGmx, right) hearts stained for capillaries with anti-vWF antibodies; scale bars = 100 µm. (D) Quantification of vWF-positive capillaries 4 weeks post-transplantation. Data are means ± standard deviation (SD; *p < 0.05; non-treated MI group [MI] versus CPC-PRGmx-treated group [CPC-PRGmx]). (E) Representative images from sections of non-treated (MI, left) and CPC-PRGmx-treated (CPC-PRGmx, right) hearts stained using TUNEL method (24-h post-MI); scale bars = 100 µm. (F) Quantification of TUNEL-positive cells 24-h post-transplantation; data are means ± SD (*p < 0.05; non-treated MI group [MI] versus CPC-PRGmx-treated group [CPC-PRGmx]). (G) Representative images from sections of non-treated (MI, left) and CPC-PRGmx-treated (CPC-PRGmx, right) hearts stained with anti-Ly6G (24-h post-MI). PRGmx scaffolds were nonspecifically stained with Alexa-fluor 488-conjugated anti-mouse antibodies; scale bars = 100 µm. (H) Quantification of Ly6G-positive neutrophils 24 h post-transplantation. CPC: cardiac progenitor cell; MI: myocardial infarction; vWF: Von Willebrand factor.
Echocardiographic Measurement of Hearts 4 Weeks After Cell Transplantation.

CPC: cardiac progenitor cells; %FS: left ventricular fractional shortening; HR: heart rate; HW/BW: heart weight to body weight ratio; LVIDD: left ventricular internal diastole diameter; LVISD: left ventricular internal systole diameter; LVPWTd: left ventricular posterior wall thickness at end-diastole; LVSWTd: left ventricular septum wall thickness at end-diastole; MI: myocardial infarction; n: number of hearts.
P < 0.05 in comparison with MI.
Echocardiographic Measurement of Hearts 2 Weeks After Cell Transplantation.

CPC: cardiac progenitor cells; %FS: left ventricular fractional shortening; HR: heart rate; LVIDD: left ventricular internal diastole diameter; LVISD: left ventricular internal systole diameter; MI: myocardial infarction; n: number of hearts.
P < 0.05 in comparison with MI.
The CPCs are characterized by their potent ability to secrete JAM-A and sVCAM-1, which both mediate the beneficial effects of CPC transplantation after MI through anti-apoptotic, angiogenic, and anti-inflammatory effects19,20. Here, CPCs secreted large amounts of JAM-A and VCAM-1, but not vascular endothelial growth factor (VEGF), compared with dormant CMCs cultured on gelatin-coated dishes (Fig. 6). CPC-PRGmx secreted more VEGF, a comparable amount of JAM-A, and less VCAM-1 than 2D CPCs. However, CMCs cultured in PRGmx (CMC-PRGmx) hardly secreted these three factors.
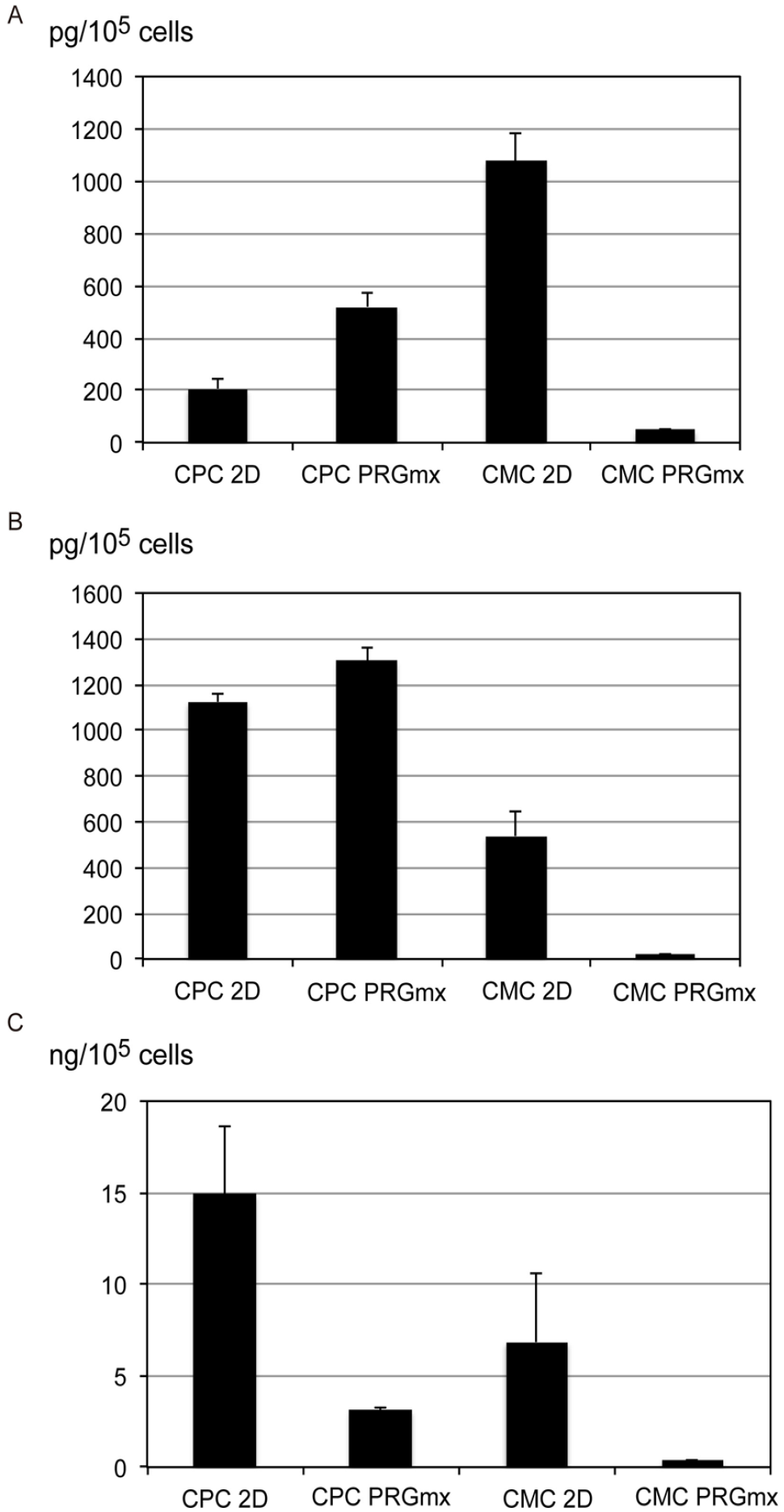
Cytokine secretion profiles of CPCs and CMCs. Concentrations of (A) VEGF, (B) JAM-A, and (C) VCAM-1 for CPCs and CMCs. Amounts of secreted cytokines are represented as quantities per 105 cells. CMC: cardiac mesenchymal cells; CPC: cardiac progenitor cell; JAM-A: junctional adhesion molecule-A; VCAM-1: vascular adhesion molecule 1; VEGF: vascular endothelial growth factor.
Transplantation of CPC-PRGmx Enhances de novo Cardiomyogenesis
The beneficial effects of cell therapy may be mediated not only by cardiac protection, but also by the replenishment of the myocardium with newly formed cardiomyocytes 32 . Here, RFP-positive CPCs were retained in the PRGmx scaffold after transplantation. However, RFP- and SA-actinin-double-positive striated cardiomyocytes were not detected (data not shown). We subsequently examined whether CPC-PRGmx transplantation enhanced endogenous regeneration by quantifying the number of newly formed cardiomyocytes using CreLacZ mice. Nearly 100% of working cardiomyocytes have been shown to be irreversibly labeled with β-galactosidase in CreLacZ mice, so the emergence of β-galactosidase-negative cardiomyocytes can act as a marker of de novo cardiomyogenesis 21 . The limited number of pre-existing specialized cardiomyocytes, including the atrioventricular node, bundle of His, and Purkinje fibers, evaded the expression of β-galactosidase after tamoxifen treatment in CreLacZ mice. Thus, we excluded such cells based on the expression of connexin40 (a marker of conduction system cells). Following tamoxifen administration, CreLacZ mice subjected to MI were randomly divided into non-treated MI (n = 5) and CPC-PRGmx-treated (n = 4) groups. X-gal-negative cardiomyocytes were present in the remote (upper two rows in Fig. 7A, B) and MI areas. This included the border and infarct areas (lower two rows in Fig. 7A, B) 28 days after infarction. These X-gal-negative cardiomyocytes exhibited SA-actinin-positive fine sarcomeres and were delineated by laminin. Adjacent sections were negative for connexin40, unlike the X-gal-negative and connexin40-positive (X-gal-Cx40+) cardiac conduction cells (Fig. 7, arrowheads). Compared with the non-treated MI group (Fig. 7A), X-gal-negative and connexin40-negative cardiomyocytes (X-gal-Cx40-CM) occurred frequently in the CPC-PRGmx-treated group (Fig. 7B) in both remote and MI areas.

Effects of transplantation of CPC-PRGmx on de novo cardiomyogenesis. Representative images of X-gal-negative cardiomyocytes in (A) non-treated and (B) CPC-PRGmx-treated MI hearts of CreLacZ mice. Immunofluorescent images of two adjacent sections obtained from remote (upper two rows in A and B) or MI (lower two rows in A and B) areas are shown. One section was stained with antibodies specific for SA-actinin and laminin, the other was stained with antibodies specific for SA-actinin and CX40, followed by DAPI and X-gal staining. Arrows indicate X-gal-Cx40- newly formed cardiomyocytes. Arrowheads indicate X-gal-Cx40+ cardiac conduction cells; scale bars = 20 µm. CPC: cardiac progenitor cell; DAPI: 4′,6-diamidino-2-phenylindole; MI: myocardial infarction.
After 28 days, in the MI area, the CPC-PRGmx-treated group had 0.98 ± 0.25% of X-gal-Cx40-CM (n = 4), whereas the non-treated MI group had 0.25 ± 0.04% (n = 5; *p < 0.05; Fig. 8). The two groups also differed significantly in the remote area: the CPC-PRGmx-treated group had 0.28 ± 0.05% of X-gal-Cx40-CM (n = 4), whereas the non-treated MI group had 0.11 ± 0.04% (n = 5; *p < 0.05; Fig. 8). The percentage of X-gal-Cx40-CM in the MI area was significantly higher than that in the remote area, regardless of the treatment (#p < 0.05; Fig. 8). Thus, the transplantation of CPC-PRGmx enhanced cardiac injury-mediated endogenous regeneration in remote and MI areas.

Frequency of X-gal-Cx40- cardiomyocytes versus total cardiomyocytes in remote and MI areas, in non-treated (open bar) and CPC-PRGmx-treated (closed bar) mice (after MI). Asterisks above a line spanning two columns indicate a significant difference between two groups (*p < 0.05). Number signs indicate significant differences between remote and MI areas. Two pairs of adjacent heart sections were examined per mouse. Values presented are averages of four (non-treated group) and five (CPCs-PRGmx-treated group) mice. CPC: cardiac progenitor cell; MI: myocardial infarction.
Discussion
Here, we successfully cultured CPCs in a self-assembling peptide hydrogel. In doing so, we obtained high cell viability and uniform cell distribution. PRG motifs on the self-assembling peptide and IGF-1 in the medium synergistically enhanced CPC survival and cell spreading; they created a 3D graft (CPC-PRGmx) for cardiac repair. Transplanting CPC-PRGmx into the pericardial space reduced the infarct area and improved cardiac function and remodeling after MI. These beneficial effects may have been associated with the enhancement of angiogenesis, inhibition of apoptosis, and reduction of emigrated neutrophils in the infarcted area. In turn, these benefits may have been promoted by paracrine factors derived from the CPCs (which were adapted to the 3D environment). In addition, genetic fate mapping revealed that de novo cardiomyogenesis was induced via the intra-pericardial transplantation of CPC-PRGmx.
The transplantation of engineered tissue patches containing either CPCs or cardiomyocytes represents an emerging strategy for cardiac repair. A variety of biomaterials such as prefabricated porous solid matrices, decellularized heart tissue, cell sheets, and hydrogels, have been studied to create 3D heart tissue 33 . The 3D scaffolds of porous solid matrices have been fabricated from natural biological materials or synthetic polymers. The epicardial transplantation of collagen and basement membrane in extract-based engineered heart tissue containing neonatal rat cardiomyocytes has been shown to prevent cardiac remodeling and improve cardiac function in infarcted hearts 34 . Using similar biological scaffolds, Nathaniel et al. engrafted human embryonic stem (ES) cells and human-induced, pluripotent, stem cell-derived cardiomyocytes as a cardiac patch onto the hearts of athymic rats 35 . Liau et al. 36 , meanwhile, fabricated fibrin-based engineered heart tissue, in which mouse ES cell–derived cardiomyocytes developed significant contractile forces ex vivo. Synthetic polymers such as polyglycolic acid, poly ε-caprolactone-co-l-lactide, and polyglycerol-sebacate were used as 3D scaffolds in this case, but poor adhesiveness meant that they had to be coated with natural extracellular matrix proteins to ensure cardiomyocyte attachment. The rigid structures and pores of this matrix isolate cardiomyocytes, thereby preventing direct intracellular contact. The immune response to the presence of natural biological matrices may therefore represent an additional limitation 37 . The decellularized heart technique utilizes the almost intact connective tissue architecture of the myocardium and blood vessels, after eliminating cells from the tissue 38 . Recently, natural scaffolds made from decellularized bladders and myocardial tissue have been used to create 3D cardiac patches by combining them with cardiomyocyte cell sheets39,40. Turner et al. successfully cultured ES-derived endothelial cells with ES-derived cardiac cell sheets on a decellularized porcine bladder matrix. Godier-Furnémont et al., meanwhile, seeded fibrin hydrogels containing human mesenchymal progenitor cells on decellularized sheets of human myocardium. They then transplanted them into a nude rat model of MI. This engrafted patch enhanced vascular growth in the infarcted region and attenuated cardiac remodeling after MI.
Here, we used synthetic hydrogels made from PM, consisting of alternating hydrophilic and hydrophobic amino acid residues that could adopt β-sheet structures and form a stable 3D hydrogel comprising >99.5% water (depending on the pH, salt, and time) 41 . It has recently been reported that PM can be decorated with a variety of functional motifs derived from the extracellular matrix, or from secreted molecules. These peptide-modified designer PMs can tune signaling to cell surface receptors; they can generate 3D environments for cell culturing and tissue engineering applications, for a variety of cell types 42 . We found that PRG-conjugated PM improved the viability of CPCs and enhanced actin filament formation, compared to other forms of PM. A PRG motif contains an RGD cell attachment sequence for integrin receptors, which promotes the formation of focal adhesive regions, as found in 2D cultures. This plays important roles in cell proliferation and differentiation 43 . Similar advantages of PRG-conjugated PM have also been reported in pre-osteoblast proliferation and differentiation, neural stem cell adhesion and differentiation, and angiogenic activity of endothelial cells44–46.
Since Davis et al. 47 first reported that injected PM confers 3D microenvironments for endogenous angiogenesis, the injection of platelet-derived growth factor-, IGF-1-, or proteinase-resistant stromal cell–derived factor-1-conjugated PM has been reported to enhance angiogenesis, decrease infarct size, improve cardiac function, and increase survival for co-transplanted cells48–52. However, in situ cardiac tissue engineering with multiple needle-base injections has several drawbacks, including the transient disruption of the extracellular matrix, loss of anchorage-dependent signals, and an inhomogeneous distribution of cells; this may be attributable to a potential proarrhythmic risk. Thus, creating an epicardially implantable 3D biodegradable hydrogel that preserves cell survival and proliferation is advantageous. Synthetic hydrogels are generally mechanically weak and unsuitable for tissue engineering applications that require gel structures with high mechanical integrity. In our study, supplementation with IGF-1 reduced apoptotic cell death during hydrogel formation and enhanced the proliferation of CPCs in the hydrogel, leading to a homogenous distribution of CPCs that maintained their actin cytoskeleton architecture. Other growth factors, such as epidermal growth factor, hepatocyte growth factor, and VCAM-1, did not enhance cell viability in the scaffolds (data not shown). The initial culture phase in PM is critical because PM in sterile water exists at pH ~3.0; thus, cells must initially encounter acidic conditions. IGF-1 has been reported to increase CPC growth and survival, in vitro and in vivo 53 . This suggests that CPC-scaffolds comprising CPCs, PRG-conjugated PM, and IGF-1 provided strength to the hydrogel, allowing it to bear the mechanical stress of the beating heart. This allows the hydrogel to function as an epicardial patch hosting coordinated PM degradation and tissue ingrowth.
Reliable methods for quantifying cell engraftment are crucial for accurately assessing efficacy 54 . Here, we examined the engraftment of CPCs using two different methods: retroviral transduction of the reporter gene and sex-mismatched transplantation. Donor-derived CPCs were detected 4 weeks after sex-mismatched transplantation by fluorescence in situ hybridization (FISH). However, the RFP+ cells disappeared simultaneously. Recently, Zhou et al. 55 reported that RFP exerts cytotoxic effects by inhibiting B-cell lymphoma-extra large in HeLa cells. The discordant results between these two methods may stem from the faster elimination of RFP+ CPCs against non-labeled CPCs. FISH has the advantage that it does not require cell labeling to detect transplanted cells. FISH images of CPC-scaffolds revealed many Y chromosome-positive donor-derived CPCs, suggesting that homogenous engraftment was highly efficient.
Given that transdifferentiation of the engrafted CPCs into cardiomyocytes occurred less frequently, the observed benefits may have arisen from cardiac patch-associated paracrine support in myocardial protection. Our in vitro experiment revealed that CPCs retained the ability to secrete VEGF and JAM-A in the 3D PRG-mx environment. Less VCAM-1 was secreted from CPCs in PRG-mx than from those in the 2D culture environment; however, the amount of VCAM-1 secreted from PRG-mx CPCs was more than that of CMCs in the same 3D PRG-mx milieu. The CPC-scaffold was well vascularized and integrated into the host myocardium. Therefore, the CPC-scaffold may exert its beneficial effects through sustained release of its own paracrine factors, such as, VEGF, JAM-A, and VCAM-1, and each factor may play a critical role to ameliorate cardiac remodeling and dysfunction through anti-apoptosis, angiogenesis, or anti-inflammatory effects18–20.
Here, we could not find cardiomyocytes derived from transplanted CPCs. The beneficial effect of cell therapy for heart injury is considered not be related to the differentiation of transplanted cells into cardiomyocytes 56 . However, establishing 3D self-assembling peptide scaffolds containing more cardiomyogenic cells, such as induced pluripotent stem cell–derived or reprogramed cardioblasts, combined with our pericardial grafting method, could realize ideal myocardial remuscularization.
In addition, our genetic fate mapping showed that more endogenously regenerated new cardiomyocytes were observed in the CPC-PRGmx-transplanted hearts than in the non-transplanted MI hearts. Interestingly, the de novo regeneration of cardiomyocytes was enhanced in both ischemic and remote areas. This may have occurred due to CPC-PRGmx in the pericardial sac. Moreover, the unknown cardiomyogenic factors secreted from the cardiac patch may affect the whole area of the heart. For endogenous cardiac regeneration, cardiac stem or progenitor cells are thought to contribute very small, if any, during normal turnover, or at the timing of cardiac injury57,58, but endogenous cardiomyocyte renewal may be enhanced by cell therapies 56 . In our study, the percentage of non-cardiomyocyte-derived cardiomyocytes was 0.25% in non-treated group and 0.98% in CPC-PRGmx-treated group. Although, the absolute effect on endogenous cardiomyocyte is still small, identifying the noble cardiomyogenic factor from epicardially transplanted CPC-scaffolds could reveal a new therapeutic strategy aimed at endogenous myocardial regeneration. Our findings shed light on the therapeutic potential of epicardial patch-like scaffolds comprising CPCs and self-assembling peptides; they may trigger the establishment of a new cardiac tissue engineering therapy.
Footnotes
Acknowledgements
The authors thank M. Tokunaga and A. Furuyama for their excellent technical assistance. The authors thank 3-D Matrix, Ltd. for providing Puramatrix.
Authors’ Contribution
M Kanda, T Nagai, N Kondo, H Akazawa, I Komuro, and Y Kobayashi participated in research design.
M Kanda and T Nagai participated in the writing of the paper.
M Kanda, T Nagai, N Kondo, and K Matsuura participated in the performance of the research.
Availability of Data and Material
The data that support the findings of this study are available from the corresponding author [ email:
Ethical Approval
This study was approved by our institutional review board. The study protocol was approved by the Institutional Animal Care and Use Committee of Chiba University (project number 27-214).
Statement of Human and Animal Rights
All animal procedures were performed in accordance with the recommendations of the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid for Scientific Research, Developmental Scientific Research, and Scientific Research on Priority Areas from the Ministry of Education, Science, Sports, and Culture JSPS KAKENHI (grant nos. 25293180, 18K15839, and 17K17636). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
