Abstract
Imatinib, a selective BCR-ABL tyrosine kinase inhibitor (TKI), was introduced after allogeneic hematopoietic stem cell transplantation (allo-HSCT) for patients with chronic myeloid leukemia (CML). However, the long-term effects of allo-HSCT in chronic phase CML patients are mostly unknown. We retrospectively analyzed the outcomes of 204 patients with sibling donors who received peripheral stem cells and underwent allo-HSCT of chronic phase I (CP1) in the pre- and post-TKI era at Shariati Hospital in Tehran, Iran, from 1998 to 2017 and followed up till the end of 2021. The median follow-up time for all patients was 8.7 (SD = 0.54) years. Fifteen-year overall survival (OS), disease-free survival (DFS), graft-versus-host disease-free relapse-free survival (GRFS), relapse, and non-relapse mortality (NRM) incidence were 65.70%, 57.83%, 17.56%, 13.17%, and 28.98%, respectively. Using multivariable analyses, the only risk factor increasing the hazard of death was the time between diagnosis to allo-HSCT greater than 1 year compared to this time less than 1 year by 74% [hazard ratio (HR) = 1.74, P = 0.039]. Also, age is a significant risk factor for DFS (HR = 1.03, P = 0.031). Our findings suggested that allo-HSCT is still an important treatment option for CP1 patients, especially those resistant to TKI treatment. TKI consumption can have a desirable effect on NRM after allo-HSCT for CP1 CML.
Keywords
Introduction
Selective BCR-ABL tyrosine kinase inhibitor (TKI) has superseded allogeneic hematopoietic stem cell transplantation (allo-HSCT) for patients with chronic myeloid leukemia (CML) 1 . As a first-line treatment, imatinib reduced the number of allo-HSCTs due to its 98%, 92%, and 83% efficacy in channeling the chronic phase of disease toward complete hematologic responses (CHRs), major cytogenetic responses (MCRs), and complete cytogenetic responses (CCRs), respectively2,3. Based on the guidelines, the indications of allo-HSCT in the TKI era are resistance or intolerance of patients to second-generation TKIs, the existence of T315I mutation after examining more available treatment options, and the classification of transplant and illness risk 4 .
To date, the majority of data on CML transplant-related risk factors were obtained prior to the advent of TKI5,6. Multiple studies analyzing the outcomes of allogeneic SCTs in the era of TKIs have demonstrated that imatinib pretreatment does not enhance transplant-related toxicity incidence7,8. Even some results on transplanted sufferers with chronic phase I (CP1) CML after medication with second-generation TKIs have been reported8,9. However, long-term results of allo-HSCT in CP-CML patients previously treated with or without TKI are lacking7,10,11.
This retrospective analysis examined the consequences of individuals who received allogeneic HSCT for CP with varying TKI treatment histories before transplantation. Therefore, we seek to examine predictive risk factors for transplant results in CP-CML patients with or without prior TKI(s) therapy. The clinical relationship between hematologic responses and the result of long-term engraftment was also studied.
Materials and Methods
Data Collection and Ethical Considerations
This retrospective study was performed using data from 260 patients diagnosed with CML at the Research Institute for Oncology, Hematology, and Cell Therapy (RIOHCT), Shariati Hospital in Tehran, Iran, who received allo-HSCT from 1998 to 2017 and followed up till the end of 2021. TKI drugs have been included in the treatment protocol of this center since 2004. All basic required information was collected through a registered system and supplementary data were gathered via a comprehensive, thorough evaluation of each patient. All patients with allo-HSCT underwent chemotherapy with myeloablative conditioning (MAC) regimens. Also, all patients in the post-TKI era received only imatinib before transplantation. Individuals who were unwilling to participate, as well as pregnant women, were excluded. The ethics committee of Tehran University of Medical Sciences authorized the current investigation (reference IR.TUMS.VCR.REC.1399.477); before enrolling in the study, written permission was sought from each patient.
Outcomes and Definitions
Disease status was characterized in accordance with the World Health Organization (WHO) criteria 12 . Patients transplanted in CP >1 were either diagnosed in accelerated phase (AP) or blast crisis (BC) or had progressed to more advanced disease but returned to CP at the time of allo-HSCT. Indications for allo-HSCT were defined as follows: (1) accelerated and blastic phase at diagnosis; (2) progression to AP/BC (accelerated phase/blast crisis); (3) failure of treatment owing to TKI resistance; and (4) T315I mutation 13 . TKI resistance was defined based on the European LeukemiaNet (ELN) 2009 criteria 14 . Resistance before pre-TKI era is one of the indications for allo-HSCT. The hematologic response was determined based on the ELN criteria 14 : white blood cell <10 × 109/L, basophils <5% with the absence of myelocytes, promyelocytes, myeloblasts in the differential, platelet count <450 × 109/L, and not palpable spleen. Imatinib response was monitored at diagnosis and every 15 days until CHR was achieved and confirmed, followed by at least every 3 months (or more if required) 14 . BCR-ABL evaluation was also performed before transplantation at the reference laboratory. A standardized quantitative real-time reverse transcription-polymerase chain reaction (RT-PCR) method was utilized to identify the BCR-ABL. Acute and chronic graft-versus-host disease (GvHD) were categorized based on clinical agreement criteria by the National Institutes of Health15,16. Overall survival (OS) was considered from the date of transplantation, and patients alive at the time of the final follow-up were calculated. Disease-free survival (DFS) was specified as survival without relapse; patients who were alive after transplantation and did not have relapses until final follow-up were evaluated 17 . Graft-versus-host disease-free relapse-free survival (GRFS) was clarified as the absence of grade III-IV aGvHD, severe cGvHD, disease relapse, and mortality following allo-HSCT 18 . Relapse incidence was defined as progression to AP/BC, hematological relapse, cytogenetic relapse, and molecular relapse 13 . Due to the conditions and limitations of our study to evaluate relapse, hematological tests were performed in all patients, and molecular examination was conducted in some patients. Non-relapse mortality (NRM) incidence was determined as the time of HSCT to the date of death in the absence of disease relapse 19 .
Statistical Analyses
Categorical variables were described by frequency and percentage, and continuous variables by mean, standard deviation, median, and interquartile range (IQR). Chi-square and Fisher’s exact tests were used to determine the homogeneity of categorical between the pre- and post-TKI groups, and Student’s t-tests or Wilcoxon rank-sum tests were used to determining homogeneity for continuous variables. Using the log-rank test for comparison, Kaplan–Meier curves were constructed to estimate OS, DFS, and GRFS 20 . The reverse Kaplan–Meier method was used to determine the median follow-up time 21 .
A multivariate Cox proportional hazards model (PH) was fitted using variables selected based on P-values ≤0.2 in univariate models 22 . The TKI era variable is the focus of our research. It was therefore included in every multivariable model regardless of its significance in univariable models. In each of the three multivariable models, the global proportionality of hazards assumption was evaluated using Schoenfeld residuals. The Gray test was used to compare each cumulative incidence calculated by Gray’s method 23 . Death without relapse was considered a competing event for both relapse and relapse for NRM. A Fine-Gray PH regression model with competing events is used to assess variables’ effects on relapse and NRM incidences 24 . Variable selection in multivariable incidence regression was based on the same scenario as in multivariable Cox regression. A two-sided P-value of 0.05 or lower was considered statistically significant. Analyses were done with STATA version 11.2 and Packages “survival” and “cmprsk” in R software version 3.3.1 25 .
Results
Patient Characteristics
Two hundred sixty patients underwent allo-HSCT from 1998 to 2017 at RIOHCT, Shariati hospital, affiliated by Tehran University of Medical Sciences (TUMS) as the main center for oncology and stem cell transplantation in Iran. They were followed up till the end of 2021. Patients were all under 55 years of age, and the average age was 30.16 (SD = 9.62) years. Among these patients, 166 individuals underwent allo-HSCT before 2004 (in the pre-TKI era) and 94 patients after 2004 (in the post-TKI era). Other characteristics of patients are also listed in Supplemental Table S1.
Due to a small number of subjects in certain subclassifications, in order to maintain the homogeneity of the data, we considered only patients with CP1 for the phase of disease before allo-HSCT, patients with sibling donors, and those that received peripheral stem cells for our final analysis, decreasing our study population to 204 patients as in Table 1.
Baseline Characteristics of CML Transplanted Patients Based on TKI Era.

AP: accelerated phase; BC: blast crisis; CML: chronic myeloid leukemia; HSCT: hematopoietic stem cell transplant; TKI: tyrosine kinase inhibitor.
Bold P values are significant at 0.05.
Indications for and Outcomes of Allo-HSCT
Indications of allo-HSCT for patients consist of resistance to standard drugs, progression to AP/BC, accelerated phase of the disease, blastic phase of the disease, and having T315I mutation. According to our data, resistance to standard drugs and T315I mutation are the most and least frequent indications of allo-HSCT. Although the indications showed different distributions in the pre- and post-TKI era (P = 0.003) owing to very low individuals in several categories, the distribution in the two groups is almost similar (Table 1).
In this study, 159 individuals experienced acute GvHD, and 111 patients faced chronic GvHD. In contrast with cGvHD, aGvHD patients have a different distribution in the pre-and post-TKI era (P = 0.04) (Table 2).
Post-transplant Outcomes of CML Patients Based on TKI Era.

aGvHD: acute graft-versus-host disease; cGvHD: chronic graft-versus-host disease; CML: chronic myeloid leukemia; TKI: tyrosine kinase inhibitor.
Bold P values are significant at 0.05.
The organs most damaged by aGvHD were the liver and skin, with grade II (44.08%) and grade I (30.92%) as the highest grade of aGvHD in these organs. Moreover, the most common type of cGvHD was the moderate type in the pre- and post-TKI era (53.33% and 47.22%, respectively). As the last measured outcome, the total number of relapses of CML did not occur differently in the pre- and post-TKI era (P = 0.427) (Table 2).
Analyses of Overall, Disease-free, and Graft-free Relapse-free Survival
We followed patients for up to 15 years with a median of 8.7 [95% confidence interval (CI): 7.6–9.62] years to determine the OS, DFS, and GRFS of patients. The 15-year probability for OS, DFS, and GRFS were 63.12%, 55.54%, and 17.43% in pre-TKI and 75.22%, 66.34%, and 17.68% in the post-TKI era; P = 0.234, P = 0.451, P = 0.462, respectively (Fig. 1A, B, C). Out of six variables, OS had no significant differences except for the time between diagnosis and allo-HSCT (P = 0.045). No variable significantly affected DFS and GRFS (Supplemental Table S2). After a multivariable Cox PH regression analysis of the outcomes, the only risk factor increasing the hazard of death in OS is the time between diagnosis and allo-HSCT greater than 1 year compared to this time less than or equal to 1 year by 74% (HR = 1.74, P = 0.039). As for the DFS, the only significant risk factor in the multivariable model is age, so with every year increase in age, the chance of death or relapse will be about 3% more (HR = 1.03, P = 0.031). No other risk factors were associated with OS and DFS. Finally, for GRFS, no risk factor was found to be significantly linked (Table 3). We did not also check the effect of aGvHD on GRFS as it does not make sense considering the definition of GRFS.

Survival outcomes of CML transplanted patients based on TKI era. (A) Kaplan–Meier estimates of overall survival (OS). (B) Kaplan–Meier estimates of disease-free survival (DFS). (C) Kaplan–Meier estimates of graft-versus-host disease-free, relapse-free survival (GRFS). CML: chronic myeloid leukemia; TKI: tyrosine kinase inhibitor.
Univariable and Multivariable Cox Regression Analyses for Overall Survival (OS), Disease-free Survival (DFS), and GvHD-free, Relapse-free Survival (GRFS).
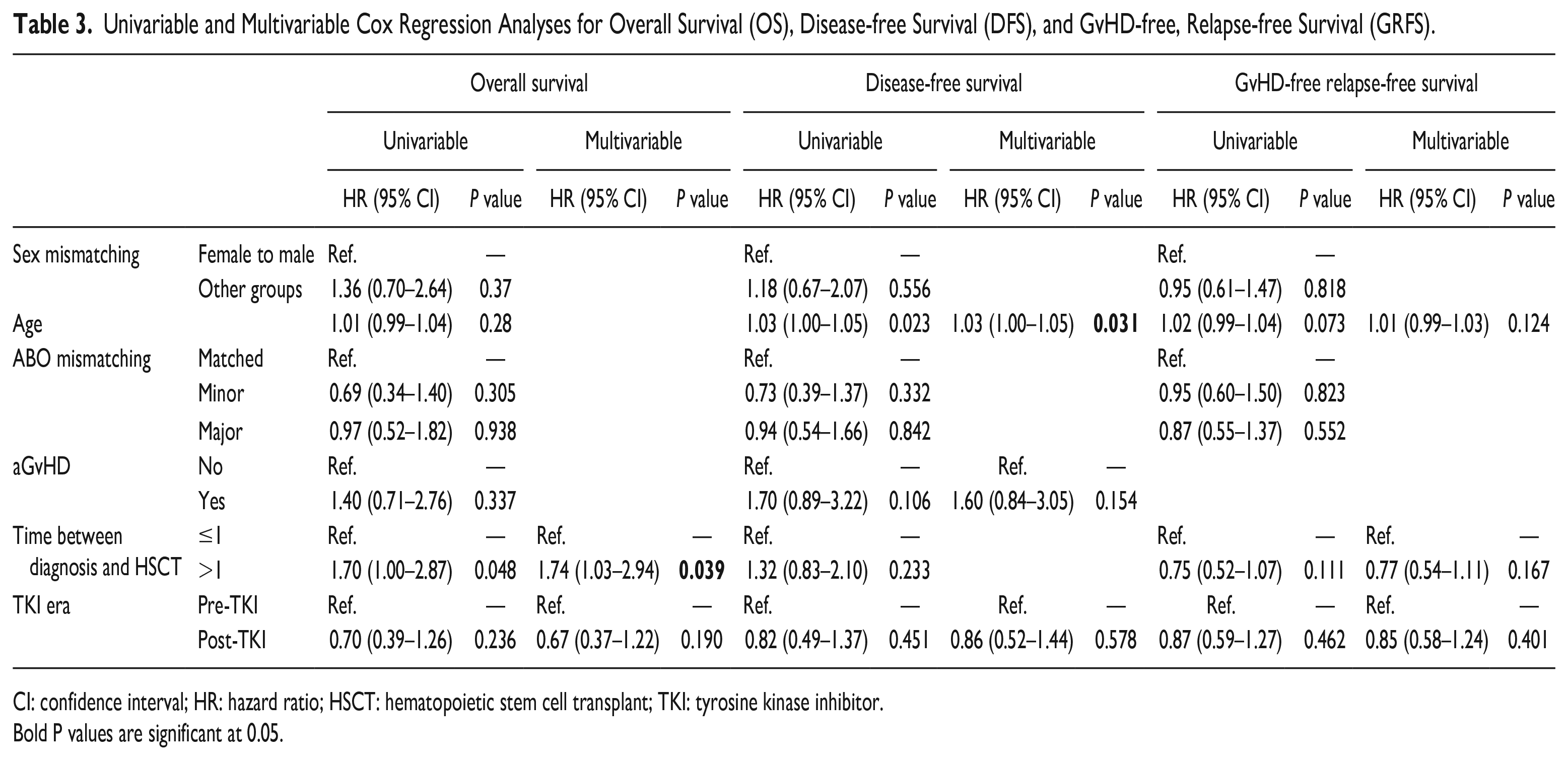
CI: confidence interval; HR: hazard ratio; HSCT: hematopoietic stem cell transplant; TKI: tyrosine kinase inhibitor.
Bold P values are significant at 0.05.
Incidence of Relapse and Non-relapse Mortality
When assessing the effectiveness of treatment and transplantation, evaluating the incidence of relapse and NRM is essential. Following 15 years of observation, the cumulative incidence of relapse in the pre- and post-TKI era were 12.63% and 13.88%, respectively (P = 0.401, Fig. 2A). Furthermore, the 15-year cumulative incidence of NRM was 31.74% and 19.92% in pre- and post-TKI, respectively (P = 0.144, Fig. 2B). Acute GvHD, however, was significantly associated with a higher chance of relapse incidence (15-year incidence of 16.35% vs 2.22%, P = 0.029) (Supplemental Table S3).

Incidence outcomes of CML transplanted patients based on TKI era. (A) Cumulative incidence (CuI) of non-relapse mortality (NRM). (B) CuI of relapse incidence (RI). CML: chronic myeloid leukemia; TKI: tyrosine kinase inhibitor.
Univariable and multivariable analyses of relapse and NRM incidence have been revealed in Table 4. Multivariable analysis of relapse incidence showed that age (HR = 1.08, P = 0.001) and occurrence of aGvHD (HR = 6.63, P = 0.008) will increase the hazard of relapse. This increase for the occurrence of aGvHD is such that it increases the hazard of relapse up to more than 6.5 times. Moreover, transplantation later than 1 year after diagnosis compared to earlier time augments the hazard of the NRM around 52% (HR = 1.52, P = 0.041) while receiving treatment in the post- than pre-TKI era had a significant preventive effect up to 40% (HR = 0.6, P = 0.025) on NRM (Table 4).
Univariable and Multivariable Fine-Gray PH Regression Analyses for Relapse Incidence (RI) and Non-relapse Mortality Incidence (NRM).
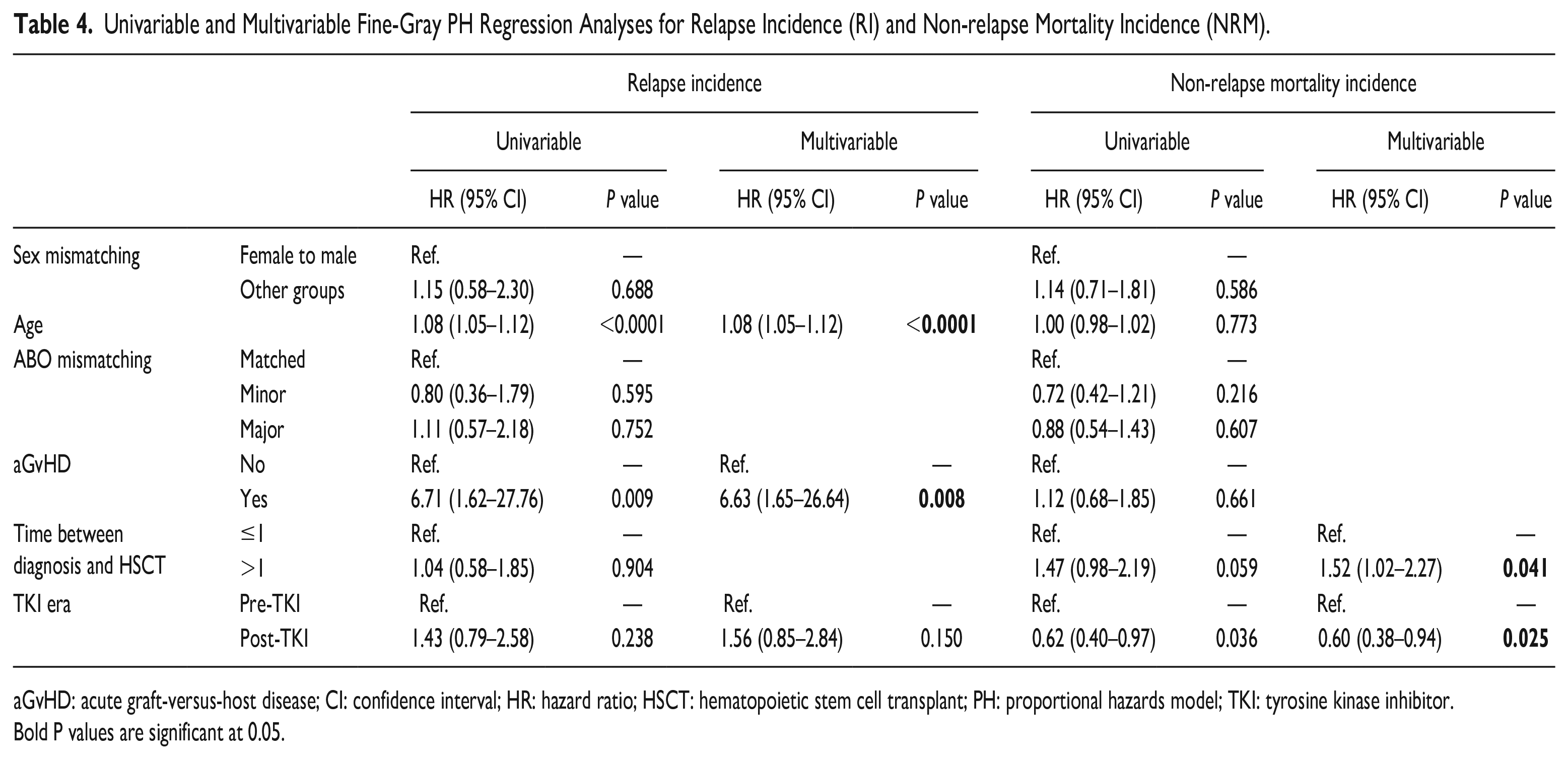
aGvHD: acute graft-versus-host disease; CI: confidence interval; HR: hazard ratio; HSCT: hematopoietic stem cell transplant; PH: proportional hazards model; TKI: tyrosine kinase inhibitor.
Bold P values are significant at 0.05.
Acute and Chronic GvHD Incidence
Acute and chronic GvHD are two important possible consequences of allo-HSCT that can affect patients’ short- and long-time survival. We estimated the cumulative incidence of acute and chronic GvHD to determine whether there are any significant differences between pre- and post-TKI era treatments.
During a 100-day follow-up, the incidence of aGvHD was significantly greater in pre- than post-TKI era treatment (82.01% and 69.23%, respectively, P = 0.016, Fig. 3A). Meanwhile, there was no significant difference between the 15-year cumulative incidence of cGvHD in the pre- and post-TKI era (48.88% and 62.78%, respectively, P = 0.888, Fig. 3B).

GvHD incidence outcomes of CML transplanted patients based on TKI era. (A) Cumulative incidence (CuI) of acute graft-versus-host disease (aGvHD). (B) CuI of chronic GvHD (cGvHD). CML: chronic myeloid leukemia; GvHD: graft-versus-host disease; TKI: tyrosine kinase inhibitor.
Causes of Death
The most prevalent reasons for mortality among the 58 patients who passed away throughout this research were acute GvHD, infection, and recurrence (46.55%, 13.79%, and 10.34%, respectively).
Discussion
This is a retrospective study based on the RIOHCT registry, the main center for hematology, oncology, and stem cell transplantation in Iran. We analyzed the outcomes of 204 patients in CP1 who received allo-HSCT from 1998 to 2017 in the pre- and post-TKI era. The 15-year OS, DFS, GRFS, relapse incidence, and NRM incidence in the pre-TKI era compared with post-TKI era are about (63.12% vs 75.22%), (55.54% vs 66.34%), (17.43% vs 17.68%), (12.63% vs 13.88%), and (31.74% vs 19.92%), respectively. Our analyses confirmed that the post-TKI era was associated with lower NRM. The time between diagnosis and allo-HSCT more than 1 year was an unfavorable prognostic factor for OS and NRM. The age of the patient was significantly associated with increased DFS and relapse incidence. aGvHD was also significantly associated with an elevated risk of relapse incidence (15-year incidence of 16.35 vs 2.22%).
In our study, there was a reduction in allo-HSCT in the post-TKI era that was aligned with the European Society for Blood and Marrow Transplantation (EBMT) study; an annual HSCT reduction of 1,400 to 800 for CML in the chronic phase from 1999 to 2003, and one the Center for International Blood and Marrow Transplant Research (CIBMTR) study that showed a reduction of 617 to 223 in the TKI era2,26,27. In addition, Kantarjian et al. 28 indicated that imatinib consumption in CP patients is associated with higher survival. Meanwhile, Ciftciler and Haznedaroglu, in a review article in 2021, sought to assess the best TKI choice for CML treatment. They argued that TKIs extend the patient’s life. However, it must be noted that selecting the proper generation of TKI is linked to important factors such as individual features of patients, compliance with drugs, lifestyle choices, comorbidities, the specific toxicity profile of the TKI medicine, and physician experience 29 . Conversely, Deininger et al. 7 showed that imatinib before allogeneic SCT had no effect on OS, DFS, or NRM versus historical controls. Interestingly, the global viewpoint noted that the use of second-generation TKIs as first-line treatment did not influence incremental survival during a 10-year follow-up period 30 . Under our study, the sex mismatching, age of the recipient, ABO mismatching, aGvHD, and time between diagnosis and allo-HSCT are independently associated with the long-term OS, DFS, and GRFS and must be considered.
The 3-year OS in our study was about 76%, which is low compared with previous studies that showed a 3-year survival rate of 85% to 92% for CP131 –33. This inconsistency could be attributed to other known risk factors for mortality (comorbidity score, cytomegalovirus serum status, cytokine gene polymorphism)32,34,35. Our study showed that OS depended on the time between diagnosis and allo-HSCT. The German Registry of Stem Cell Transplantation (DRST) was partially aligned with our results. The DRST indicated that in multivariable analysis, OS was improved for the younger age, HSCT in CP1, HLA-matched HSCT, and HSCT in the first year from diagnosis 26 . Furthermore, according to the study by Ciftciler and Haznedaroglu 29 , allo-HSCT in resistant and progressed AP/BC patients who were returned to the chronic phase increased the chance of improvement.
The NRM for patients transplanted in CP1 was between 5% (95% CI: 4%–6%) in a decade and 10% (95% CI: 9%–12%) 15 years after transplantation in other studies5,13,17,32. In our study, NRM was 16% in the first year, reached 29% in 15 years of follow-up, and was influenced by the time between diagnosis and the allo-HSCT and the post-TKI era as well. The higher NRM might be due to advancing ages at HSCT, the use of a female donor for male recipients, and acute and chronic GvHD7,17. Moreover, Bornhäuser et al. 36 showed that intervals >12 months between diagnosis and transplantation had the most significant negative impact on survival and NRM. On the contrary, Oehler et al. 37 compared 145 patients treated with imatinib followed by allo-HSCT with 231 CML patients who received a first full allo-HSCT and investigated no significant association between consumption of imatinib and NRM. Similarly, TriNetX analyzed the impact of the first-line TKI on 10-year survival after the start of treatment. The results showed no significant difference in mortality in any of the three cohorts comparing survival probability between imatinib and dasatinib or nilotinib, nor comparing dasatinib with nilotinib 30 .
DFS rates in our study at 3 years were 70.02 (95% CI: 63.13–75.86), similar to those reported by others38 –40. However, the DFS at 10 and 15 years were lower than those in the CBTMR study. Lower DFS might be due to older age at transplantation, a female donor for a male recipient, and the experience of GvHD as independent predictors 17 . In addition, Warlick et al. 41 showed that older age had an increased risk of relapse and lower DFS, and most had advanced disease at HSCT. Accordingly, our study supports that the DFS is significantly associated with the age of allo-HSCT recipients.
GRFS, as a health-related quality of life after transplantation for young CML patients, is a composite endpoint for transplantation in current clinical trials. In our result, the incidence of GRFS in 5-year follow-up was 29.83 (95% CI: 22.93–37.02), in line with the study by Zheng et al. 42 In multivariable analysis, we showed no independent factor for GRFS after transplantation. Several studies supporting our results have been published43 –45. We need more studies on GRFS in CML patients to investigate details.
The relapse incidence rate was 5.44% in the first year to 13.17% in 15 years after allo-HSCT, and age and acute GvHD were linked to a higher risk of relapse incidence in our study. Liu et al. demonstrated that the incidence rate of relapse 5 years following the transplant was 10.7% for the total group (95% CI: 3.6%–17.8%), and based on univariable analyses, grade 0–I acute GvHD and the absence of chronic GvHD were associated with a higher relapse incidence rate. Yet, the multivariable analysis revealed no statistically significant risk factors for the relapse incidence rate 38 . Notably, Goldman et al. showed a higher relapse incidence rate was seen in patients 20 years or older [23% (95% CI: 14%–35%)] compared with younger patients [8% (95% CI: 3%–21%)], but this finding had only borderline significance (P < 0.03). Also, patients with moderate to severe aGvHD had a risk for treatment failure (death or relapse), 3.2 times higher than patients with no or mild aGvHD (P < 0.0001) 46 . These observations were in line with our results.
Strength and Limitation
We presented the results of a 15-year long-term follow-up of CML patients who underwent allo-HSCT. Also, we had patients of a wide range of ages and highlighted important factors occurring within years after allo-HSCT, which may help predict long-term outcomes. Nonetheless, we could not assess all patients for cytogenetic and molecular response due to high costs and lack of facilities in the study’s early years. In addition, we excluded approximately 25% of the population due to the heterogeneity of the data.
In conclusion, this investigation revealed the predictive criteria for allo-HSCT results in CP1 CML. We concluded that allo-HSCT remains an essential therapeutic alternative for CP1 patients, particularly those who are unsusceptible to TKI therapy. These individuals have more long-term survival and less NRM. TKI consumption had a favorable influence on NRM after allo-HSCT for CP1 CML. Patients who received CP1 transplants had equal survival results, independent of prior TKI treatment. However, long-term results should be investigated by randomized studies of many homogeneous patients.
Supplemental Material
sj-docx-1-cll-10.1177_09636897231163212 – Supplemental material for Comparable Outcomes of Pre- Versus Post-Tyrosine Kinase Inhibitor Era Treatment in Chronic Myeloid Leukemia: A Retrospective Cohort Study With Long-term Follow-up
Supplemental material, sj-docx-1-cll-10.1177_09636897231163212 for Comparable Outcomes of Pre- Versus Post-Tyrosine Kinase Inhibitor Era Treatment in Chronic Myeloid Leukemia: A Retrospective Cohort Study With Long-term Follow-up by Sahar Tavakoli, Fattaneh Khalaj, Amir Kasaeian, Seyed Ali Mousavi, Amir-Hossein Mousavian, Fatemeh Arabi, Soroush Rad, Shahrbano Rostami, Maryam Barkhordar, Mohammad Biglari, Heydar Ali Mardani-Fard, Hediyeh Alemi, Naghmeh Khavandgar, Hossein Kamranzadeh Fumani, Ghasem Janbabai, Seied Asadollah Mousavi, Ardeshir Ghavamzadeh and Mohammad Vaezi in Cell Transplantation
Footnotes
Acknowledgements
We thank our colleagues for clinical management and our data registry team for providing data to the study.
Author Contributions
ST and AK conceptualized and organized the study; ST, FK, AK, A-HM, and FA wrote the text; AK analyzed the data and wrote the results; all authors participated in collecting data and approval process of the manuscript. All authors agreed to the submitted version of the manuscript.
Availability of Data and Materials
The raw data associated with the conclusions of this article will be made accessible by the corresponding author, without undue reservation.
Ethical Approval
The ethical committee of the Vice-Chancellor in Research Affairs, Tehran University of Medical Sciences (TUMS), approved the study (reference IR.TUMS.VCR.REC.1399.477). The patients provided their written informed consent to participate in this study.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to express thanks to the Research Institute for Oncology, Hematology and Cell Therapy (RIOHCT), Tehran University of Medical Sciences (TUMS), Tehran, Iran, for their financial support through small grant (TUMS, Grant No. 98-3-107-45749).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
