Abstract
The advent of tyrosine kinase inhibitors (TKIs) has profoundly changed the therapeutic landscape of chronic myeloid leukemia (CML) in the chronic phase (CML-CP). Imatinib is the first-generation, and the first and as yet the most widely used TKI, and the recommended dose is 400 mg/day for treating CML-CP. Most patients tolerate this treatment well, and prolonged hematological toxicities have rarely been reported. In this manuscript, we report a newly diagnosed CML-CP patient who developed prolonged myelosuppression (lasting for more than three months) following only one week of imatinib at 400 mg/day as the solitary treatment. Imatinib was discontinued, and pancytopenia persisted, with a continuous decrease in hemoglobulin levels. After restoration of autologous hematopoiesis, reintroduction of imatinib at 100 mg/day resulted in recurrent myelosuppression, and subsequent treatment with imatinib at 50 mg/day achieved good hematological homeostasis. We hypothesized that extraordinary sensitivity resulted in severe and prolonged myelosuppression.
Keywords
Introduction
The advent of tyrosine kinase inhibitors (TKIs) has profoundly changed the therapeutic landscape of chronic myeloid leukemia (CML) in the chronic phase (CML-CP), with the majority of patients treated solely with TKIs achieving sustained molecular remission and normal expected longevity. Imatinib is the first-generation, and the first and as yet the most widely used TKI in CML treatment, and the recommended dose is 400 mg/day for treating CML-CP. Most patients tolerate this treatment well, and hematological toxicities ameliorate rapidly following discontinuation or tapering of imatinib.1,2 Prolonged myelosuppression has rarely been reported and develops usually several months after imatinib administration.3–9 A newly diagnosed CML-CP patient developed prolonged severe myelosuppression (lasting for more than 3 months) following only one week of imatinib at 400 mg/day as the solitary treatment. Imatinib was discontinued, and pancytopenia persisted. Autologous hematopoiesis eventually recovered with supportive treatment by recombinant human granulocyte colony-stimulating factor (rHG-CSF) and recombinant human erythropoietin (rH-EPO). More interestingly, after restoration of autologous hematopoiesis, reintroduction of imatinib at 100 mg/day resulted in recurrent myelosuppression, and subsequent treatment with imatinib at 50 mg/day achieved good hematological homeostasis. Unfortunately, molecular relapse occurred 17 months later.
Case presentation
An 82-year-old Chinese male had abnormalities in complete blood count (CBC) found during a preoperative examination for abdominal aortic aneurysm. At that time, CBC showed the following results: white blood cells (WBCs), 32.46 × 109/L; absolute neutrophil count (ANC), 26.44 × 109/L; red blood cells (RBCs), 4.18 × 1012/L; hemoglobulin (Hb), 148 g/L; and platelets (Plts), 286 × 109/L. He was diagnosed with CML-CP based on the morphological appearances of marrow and blood smears and identification of the Philadelphia chromosome translocation and BCR/ABL1 fusion gene. He denied having diseases in hematological, immunological or other systems in the past. He was prescribed imatinib at 400 mg/day as the solitary drug for the treatment. One week later, CBC monitoring showed the following results: WBCs, 2.48 × 109/L; ANC, 0.98 × 109/L; RBCs, 3.56 × 1012/L; Hb, 135 g/L; and Plts, 21 × 109/L. Imatinib was discontinued, but pancytopenia persisted, with a continuous decrease in Hb levels being the most prominent. He was intermittently transfused with packed RBCs. On Day 94, CBC monitoring showed the following results: WBCs, 2.51 × 109/L; ANC, 1.16 × 109/L; RBCs, 1.50 × 1012/L; Hb, 68 g/L; Plts, 19 × 109/L; and absolute reticulocytes (Rets), 29.50 × 109/L. Marrow aspirate was performed, and morphological examination showed normal cellularity, with erythroid progenitors accounting for 7.5% of the total nucleated cells. Various stages of neutrophilic granulocytes without obvious morphological abnormalities predominated on the smears. Examination of blood smears confirmed the reduction in nucleated cells without evident morphological abnormalities. The morphological features suggested a diagnosis of pure red cell aplasia (Figure 1). Minimal residual disease (MRD) evaluation detected the BCR/ABL1 (p210) fusion gene (b3a2) in 0.242% of hematopoietic progenitors. Morphological examination of marrow and blood smears. (a) Examination of marrow smear under a low-power lens (10 × 10) showed normal cellularity. (b) Examination of marrow smears under a high-power lens (10 × 100) showed a decreased percentage of nucleated erythrocytes (accounting for 7.5% of the total nucleated cells) without differential arrest. Various stages of neutrophilic granulocytes predominated on the smear without obvious morphological abnormalities. (c) Examination of blood smears under a low-power lens (10 × 10) confirmed the reduced number of nucleated cells. (d) Examination of blood smears under a high-power lens (10 × 100) showed no evident morphological abnormalities in erythrocytes, granulocytes or lymphocytes. The morphological features of marrow and blood smears suggested a diagnosis of pure red cell aplasia.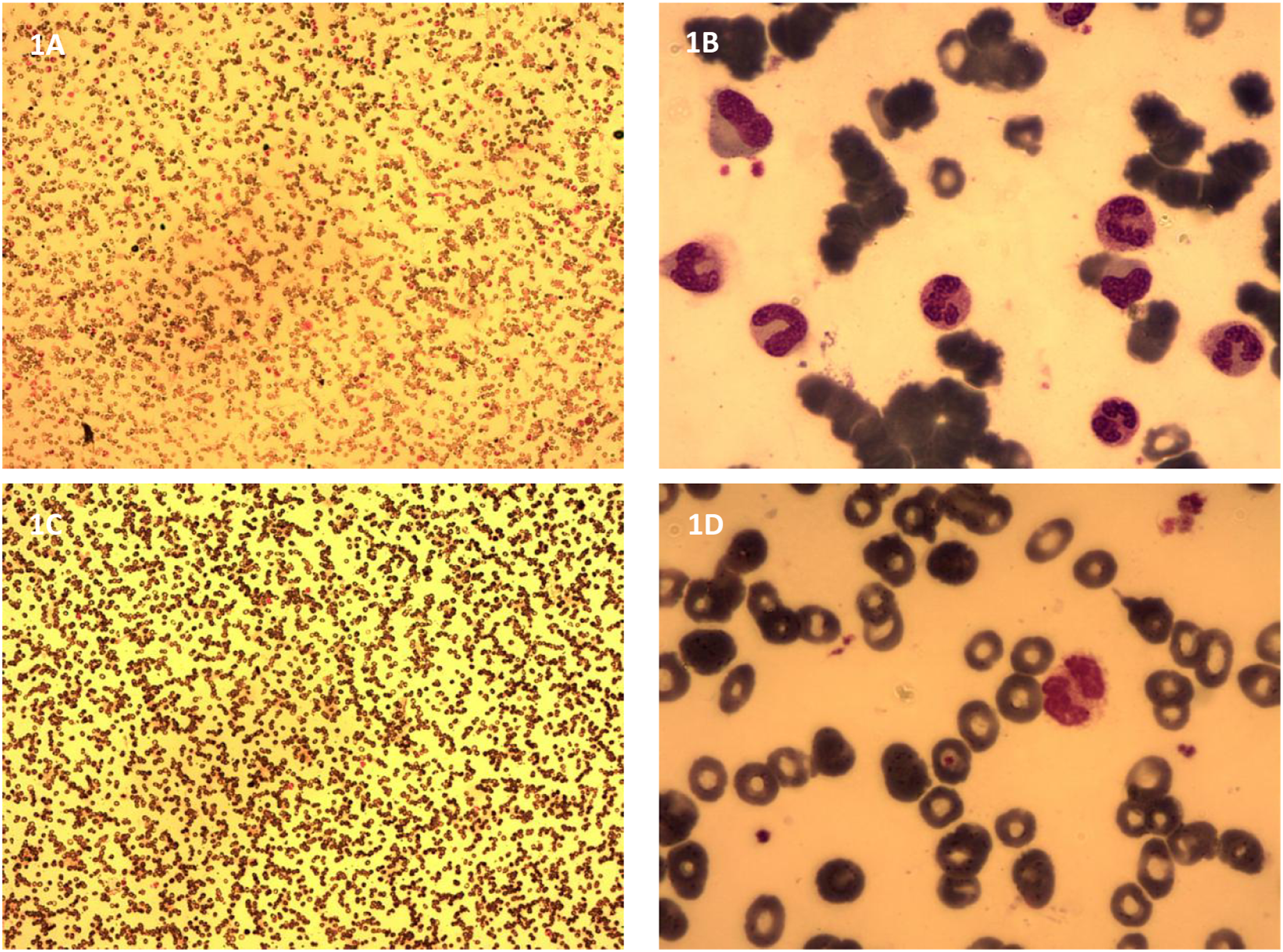
Considering the prolonged hematopoietic suppression, supportive treatment with rHG-CSF at 200 μg/day and rH-EPO at 3000 U every other day was initiated. Autologous hematopoiesis recovered within one month of treatment. After restoration of autologous hematopoiesis, imatinib treatment was reintroduced at 100 mg/day, and the severe myelosuppression rapidly recurred. The patient then received imatinib at 50 mg/day. This treatment yielded stable hematological homeostasis. WBCs, Hb, and Plts in CBC monitoring are delineated in Figure 2. MRD evaluation on Day 592 detected the BCR/ABL1 fusion gene in 94.8% of hematopoietic progenitors. WBCs, Hb and Plts in blood cell monitoring. Granulocytopenia and thrombocytopenia occurred 1 week following imatinib treatment at 400 mg/day, and then Hb levels rapidly decreased and became transfusion dependent. MRD evaluation on Day 94 detected the BCR/ABL1 fusion gene in 0.242% of marrow cells. This severe myelosuppression lasted for more than 3 months and was eventually resolved by supportive treatment with rHG-CSF and rH-EPO. Reintroduction of imatinib at 100 mg/day again resulted in severe myelosuppression. After restoration of autologous hematopoiesis, imatinib treatment at 50 mg/day yielded hematological homeostasis. MRD evaluation on Day 592 detected the BCR/ABL1 fusion gene in 94.8% of marrow cells. WBCs (×109/L), Hb (×10 g/L) and Plts (×10 × 109/L). WBCs: white blood cells; MRD: Minimal residual disease.
Discussion
In this manuscript, we describe a CML-CP patient who developed severe and prolonged myelosuppression following only one week of imatinib treatment at the recommended dose. Because imatinib was the only drug administered and because no evident infections were present in this period, it is reasonably believed that the severe myelosuppression was caused by imatinib treatment. After restoration of autologous hematopoiesis, reinstitution of imatinib treatment at 100 mg/day recapitulated, despite a relatively short duration, severe myelosuppression. The recapitulated myelosuppression substantiated the causative role of imatinib treatment in the development of aplastic cytopenia. A very low dose of imatinib (50 mg/day) was able to maintain an excellent hematological profile. We hypothesized that extraordinary sensitivity resulted in the severe and prolonged myelosuppression.
The mechanisms underlying imatinib-induced myelosuppression are not fully understood. Imatinib inhibits marrow cell proliferation in a dose-dependent manner. The inhibitory degree of imatinib treatment is much greater than the degree of stem cell factor withdrawal or c-kit functional inhibition, suggesting that other signaling pathways are also involved. 10 A reduction in imatinib dosage was able to reduce myelosuppressive activity, 11 indicating dose-dependent hematological toxicity. Severe myelosuppression has also been reported with other TKIs.7,12 All evidence suggests that myelosuppression in TKI treatment may be an intrinsic pharmaceutical effect rather than an immune-mediated pathogenesis.13,14
Nevertheless, the degrees of hematological toxicity are different in individual patients. Complete molecular remission that was maintained by very-low-dose imatinib has been reported in a patient with chronic eosinophilic leukemia who developed severe myelosuppression after short-term imatinib treatment at 100 mg/day. 9 Asian patients are sometimes intolerant to the recommended dose, and TKIs can achieve an effective plasma level at a lower does. 6 However, it is unlikely that ethnic differences can cause such extreme differences in drug tolerance. Although cumulative drug toxicity could explain the prolonged myelosuppression in previously reported cases who took imatinib for several months before the emergence of severe myelosuppression, our patient’s persistent myelosuppression lasting for more than 3 months following only one week of imatinib treatment cannot be explained by the cumulative effect.
In addition to advanced age, idiosyncrasy due to defects in drug degradation seems to be the most reasonable interpretation for the unusual hypersensitivity in our case. The metabolism and clearance of imatinib are dependent on cytochrome P450 3A4 and CYP3A metabolic activity,13,14 defects in which can affect imatinib degradation and thus enhance hematological toxicities. However, tests for CYP450 3A4 and CYP3A metabolic activity were not performed due to our laboratory limitations, leading to the limitation of further discussion.
Although very-low-dose imatinib was capable of effectively maintaining good hematological homeostasis, it is unfortunate that molecular relapse later occurred. The occurrence of molecular relapse raises the conjecture that, under close monitoring, CML patients may benefit from an appropriate myelosuppressive status by individually tailoring the imatinib dosage in accordance with their drug sensitivities,15,16 as indicated by previous reports in which deep and durable molecular remission was achieved in almost all patients with severe myelosuppression.3–9
Conclusion
Conventional dose of imatinib can cause severe myelosuppression in some patients, and intensive CBC monitoring is required in the initiation of imatinib treatment. If severe myelosuppression occurs, imatinib dosage should be tailored in the subsequent treatment. The benefit of an appropriate myelosuppressive status warrants extensive investigations.
Footnotes
Acknowledgements
We would like to thank Fan-Jun Meng (Department of Hematology, The Affiliated Hospital of Qingdao University) for guiding the treatment.
Author contributions
Xi-Chen Zhao developed the idea; Jia Xu and Bo Ju analyzed the data and drafted the manuscript; Jia Xu, Bo Ju, Xiao-Dong Yang and Nuan-Nuan Xiu participated in the treatment; Xi-Chen Zhao supervised the treatment and revised the manuscript; all authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Specialized Scientific Research Fund Projects of the Medical Group of Qingdao University (No: YLJT20201002).
Ethical approval
Ethical approval to report this case was obtained from the Institutional Review Board of Huangdao Central Hospital Affiliated with The Medical Group of Qingdao University (2022-10-08).
Informed consent
Written informed consent was obtained from the patient.
Data availability
All data generated or analyzed are included in this case report.
