Abstract
Multiple system atrophy (MSA) is a sporadic, progressive neurodegenerative disease characterized by autonomic nervous dysfunction with parkinsonism or cerebellar ataxia. Mesenchymal stem cell therapy or transplantation of human umbilical cord blood mononuclear cells (hUCB-MCs) may inhibit progression in MSA, but long-term studies are lacking. In addition, injection of stem cells via lateral atlanto-occipital space puncture (LASP, or Gong’s puncture) may efficiently target areas of brain injury and avoid the disadvantages of other methods. This prospective study investigated the long-term clinical efficacy of transplantation of hUCB-MCs via LASP for the treatment of MSA. Seven patients with MSA who received hUCB-MC transplantation via LASP were followed for 3 to 5 years. Neurological function was evaluated before (baseline), at 3, 6, and 12 months, and annually after the first transplantation using the Unified MSA Rating Scale (UMSARS); a lower score indicated improvement. Adverse events were recorded. The best therapeutic effect was observed 3 to 6 months after the first hUCB-MC transplantation. The total UMSARS score at the timepoint of best effect (25.71 ± 11.87) was significantly lower than the score before treatment (42.57 ± 7.96; P = 0.001), but also significantly lower than at the end of follow-up (35.14 ± 18.21; P = 0.038). The UMSARS II score (findings on neurological examination) at the timepoint of best effect was significantly lower than before treatment (P = 0.001). There were no serious adverse events. In conclusion, transplantation of hUCB-MCs via LASP is a safe and effective treatment for MSA.
Keywords
Introduction
Multiple system atrophy (MSA) is a sporadic, progressive neurodegenerative disease characterized by autonomic nervous dysfunction 1 . There are two clinical phenotypes of MSA: MSA with predominant parkinsonism (MSA-P) and MSA with dominant cerebellar ataxia (MSA-C) 2 . The worldwide incidence of MSA has increased dramatically in recent years. Rapid disease progression often leads to death within 8 to 9 years after the onset of symptoms3,4. The current treatments for MSA are mostly symptomatic management for autonomic nervous disorders and parkinsonism 5 . In the past 10 years, the effectiveness of recombinant growth hormone, riluzole, minocycline, lithium, rifampicin, and rasagiline in the treatment of MSA has been evaluated; however, no positive results were observed3,6.
In 2012, a randomized controlled trial showed that treatment with autologous bone marrow mesenchymal stem cells (MSCs) delayed the progression of neurological deficit in patients with MSA-C, suggesting the therapeutic potential of MSCs in MSA treatment 7 . In 2019, Singer et al. 8 reported that intrathecal transplantation of adipose-derived MSCs was a safe, well-tolerated treatment method that significantly suppressed the progression of MSA in a dose-dependent manner. The patients with MSA in both studies were followed for only 1 year, and the long-term efficacy of stem cell therapy for MSA remains unclear.
Human umbilical cord blood mononuclear cells (hUCB-MCs) contain a variety of stem cell populations (eg, hematopoietic stem cells, endothelial stem cells, lymphoblasts, small embryonic stem cells, lymphocytes, and bone marrow cells), as well as neurotrophic factors 9 . The therapeutic effects of hUCB-MCs have been observed in animal models of stroke and spinal cord injury10,11, but the clinical efficacy of hUCB-MCs in MSA has not been investigated.
The main approaches of cell therapy for central nervous system diseases include vein infusion, lateral ventricle puncture, lumbar puncture, and stereotactic transplantation 12 . Although intravenous infusion of cells is a simple procedure, the majority of cells that are infused through the vein are trapped and cleared in the lung so that fewer reach the injured site 13 . Furthermore, the number of cells that can enter the brain is very limited because of the blood–brain barrier 13 . Cells injected via lateral ventricle puncture can migrate to the injured brain area and integrate well with host tissues 14 . However, lateral ventricle puncture has certain shortcomings, such as large injury areas and high infection rate. The major disadvantage of lumbar puncture is cell depletion at each nerve root 15 .
The circulation of cerebrospinal fluid in the spinal canal is passive, and it is difficult for cells to enter the ventricular system 16 . There is evidence that microglia and macrophages are active in areas of brain injury, and the local microenvironment of the brain is highly complex 17 . Cells that are implanted in the injured area may be cleared. This impedes cell survival and expansion 18 .
Lateral atlanto-occipital space puncture (LASP, also known as Gong’s puncture since 2018) 14 injects stem cells into the occipital cistern, where they migrate to the target area via endogenous migration. This prevents microglia and macrophages from entering the site of injury and therefore improves cell survival 18 . This prospective study investigated the long-term clinical efficacy of hUCB-MC transplantation via LASP for the treatment of MSA.
Materials and Methods
This study was approved by the medical ethics committee of the local hospital (Approval No: 2013165) with informed consent signed by all patients. The procedure for blood collection and isolation of hUCB-MCs was approved by the local ethics committee.
Patients
The study population comprised seven patients with MSA who were treated with hUCB-MC transplantation via LASP in the Department of Neurology of our hospital from January 2014 to March 2016. For inclusion, MSA was diagnosed according to the criteria for probable MSA proposed by Gilman et al. in 2008 19 . Furthermore, patients had no serious cerebrovascular or other major digestive, respiratory, or blood diseases; malignant tumors; or unconscious disorders or mental system diseases.
Preparation of hUCB-MCs
Fresh cord blood (150 ml) was donated by a healthy woman who underwent cesarean section in our hospital. She had no infections, including hepatitis B, hepatitis C, human immunodeficiency virus, or syphilis. The blood was collected under sterile conditions. The hUCB-MCs were separated from blood samples using a cord blood treatment kit (cat. no. SCR-200I; Ningxia Zhonglianda Biological, China). A 5-ml suspension containing 2–3 × 108/ml hUCB-MCs was obtained.
Transplantation of hUCB-MCs
All operations were performed in compliance with aseptic principles. The hUCB-MCs were transplanted into patients with MSA via LASP. Each patient was placed supine without a pillow. The highest point of the mastoid was located and the puncture site was 1 cm inferior, and 1 cm posterior, to that point. After local anesthesia, the skin was pierced by a needle.
The direction of LASP was parallel to the imaginary line of the external auditory canal and perpendicular to the longitudinal axis of the cervical vertebra. The needle was inserted along the lower edge of the occipital bone. A sense of breakthrough was experienced two times during the puncture. The puncture was considered successful when cerebrospinal fluid flowed freely after the needle was pulled out. Three milliliters of cerebrospinal fluid were collected. Five milliliters of pre-warmed hUCB-MCs were injected slowly into the cisterna magna. The needle was then removed and the puncture point was pressed with a sterile dressing cover.
The patient was placed supine with a pillow under the head. The pillow was removed 6 to 8 hours after the procedure. Each patient was treated with hUCB-MC transplantation via LASP 2 to 4 times, at intervals of 1 month.
Follow-up
Patients were followed for 3 to 5 years. Neurological function was evaluated before the first hUCB-MC transplantation (baseline), at 3, 6, and 12 months, and annually thereafter. Neurological function was scored using the Unified Multiple System Atrophic Rating Scale (UMSARS) 20 . The UMSARS consists of UMSARS I, which quantifies patients’ symptoms and neurological function, and UMSARS II, which quantifies the findings on neurological examination. The timepoint of best effect was considered the follow-up at which patients had the lowest UMSARS total score and the lowest UMSARS II score. Adverse events were also recorded.
Statistical Analysis
Data were analyzed using SPSS 20.0 and are shown as mean ± standard deviation. Repeated-measures analysis of variance was applied to compare the UMSARS scores at different timepoints. The Bonferroni test was used to compare the UMSARS scores at each timepoint in pairs. P < 0.05 was considered statistically significant.
Results
Clinical Characteristics
Among the seven cases, five were diagnosed as MSA-C and two were categorized as MSA-P (Table 1). There were five men and two women, aged between 53 and 65 years, with a disease course of 18 to 48 months. Five patients were treated with hUCB-MC transplantation, three times. The other 2 patients were treated two and four times, respectively. The follow-up time was 3 to 5 years and the best effect was observed 3 to 6 months after the first transplantation.
Demographics, Clinical Characteristics, and Follow-up Data of Seven Patients With MSA.

FFU: final follow-up; MSA: multiple system atrophy; MSA-C: final follow-up; MSA-P: final follow-up; Tx: number of treatments.
MSA disease course.
Clinical Efficacy of hUCB-MC Transplantation
For six patients (Pts #1, 2, 3, 4, 6, and 7), both the UMSARS total score and the UMSARS II score were significantly lower at the timepoint of best effect (3–6 months after the first transplant) compared with baseline (before treatment; Table 2). At the end of follow-up, the scores of these patients were also lower than the scores before treatment. The UMSARS score of one patient (Pt #5) did not change significantly after treatment, and the condition of this patient, as indicated by the UMSARS total score and UMSARS II score, had aggravated by the end of follow-up.
UMSARS Scores of Seven MSA Patients at Baseline, Best Score, and Final Follow-up and After hUCB-MC Transplantation.
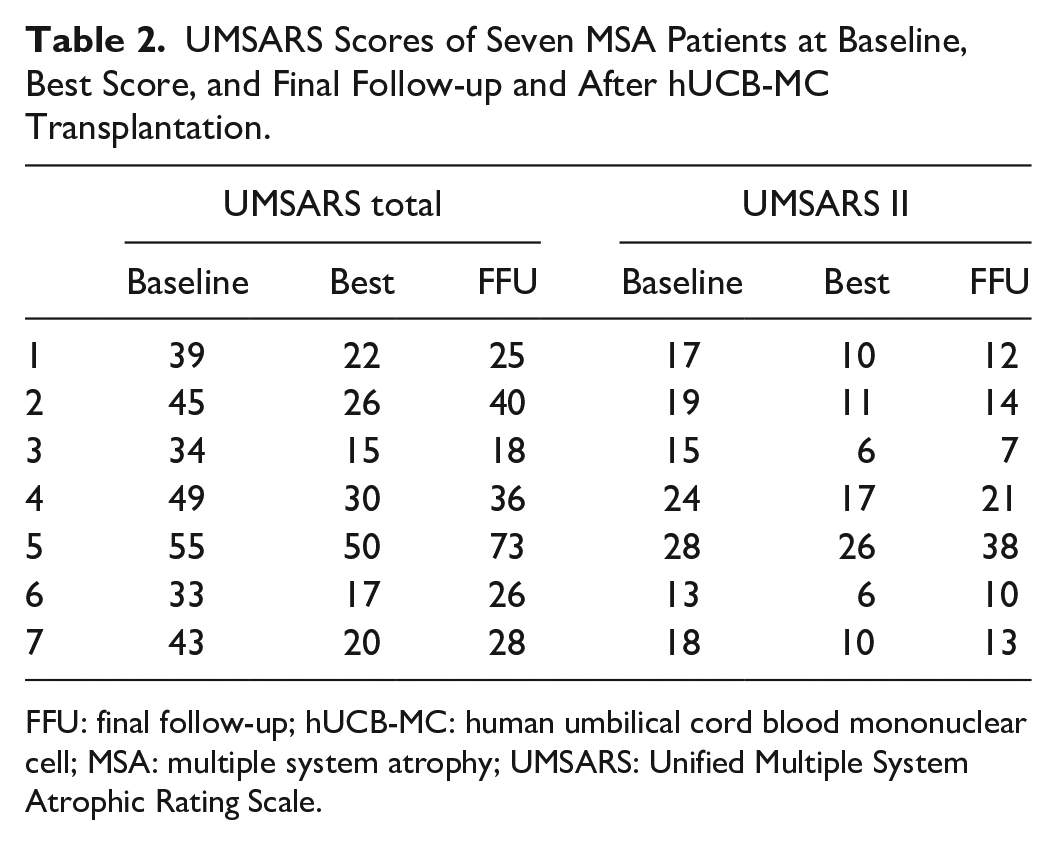
FFU: final follow-up; hUCB-MC: human umbilical cord blood mononuclear cell; MSA: multiple system atrophy; UMSARS: Unified Multiple System Atrophic Rating Scale.
The average UMSARS total score and UMSARS II score of all patients with MSA at different timepoints were significantly different (UMSARS total score: mean F = 13.301, P = 0.009; UMSARS II score: mean F = 9.536; P = 0.021; Table 3).
Average UMSARS Total Score and UMSARS II Score of Seven Patients With MSA at Baseline and After hUCB-MC Transplantation.

FFU: final follow-up; UMSARS: Unified Multiple System Atrophic Rating Scale.
P < 0.05 compared with the scores before treatment.
P < 0.05 compared with the scores at the timepoint of best effect.
The paired comparison analysis showed that the average UMSARS total score at the timepoint of best effect (25.714 ± 11.870) was significantly lower compared with the score at baseline (42.571 ± 7.955; mean difference = −16.857; P = 0.001). The average UMSARS total score at the end of follow-up (35.142 ± 18.206) was also lower than that before treatment, but not statistically significant. The average UMSARS total score at the end of follow-up (35.142 ± 18.206) was significantly higher than that at the timepoint of best effect (25.714 ± 11.870; mean difference = −9.429; P = 0.038).
The UMSARS II score at the timepoint of best effect was significantly lower than before treatment (mean difference = −6.857, P = 0.001) and had remained low at the end of follow-up (16.428 ± 10.438 cf. 19.142 ± 5.209), but the difference was not statistically significant (mean difference = −2.714, P = 0.797). The UMSARS II score at the end of follow-up was slightly higher than that at the timepoint of best effect (16.428 ± 10.438 cf. 12.285 ± 7.087), but the difference was not statistically significant (mean difference = 4.143, P = 0.070; Table 3).
Adverse Events
One patient (Pt #2) had a fever on the second day after treatment, with a maximum body temperature of 38.0°C. After antipyretic treatment, the body temperature returned to normal. Another patient (Pt #6) experienced pain and discomfort at the puncture site. The symptoms disappeared on the third day after transplantation without special treatment. There were no other adverse events or complications during follow-up.
Discussion
The current study found that the neurological function of patients with MSA improved after hUCB-MC transplantation, with the best effect observed at 3 to 6 months after the first treatment, and no patient experienced any severe adverse event related to transplantation. These findings suggest that hUCB-MC transplantation via LASP is a safe and effective treatment method for MSA.
In this study, hUCB-MC transplantation via LASP appeared to improve MSA symptoms and delayed disease progression at the 3- to 5-year follow-up, as reflected by changes in the UMSARS and UMSARS II scores. Gong et al. 14 in 2018 optimized the puncture position and route of LASP based on cervical 1–2 lateral puncture and cerebellomedullary cistern puncture (Gong’s puncture), which effectively prevented injuries in the vertebral artery and medulla oblongata. Since then, more than 1,000 operations with Gong’s puncture have been performed, with high effectiveness and safety. Stem cells that are transplanted into the cisterna magna by lateral ventricle puncture can reach the target area via endogenous migration, which prevents these cells from directly entering the inflammatory environment of the injury site, thereby improving cell survival 21 . Gong’s puncture also prevents cell loss in the lung via intravenous infusion or at the nerve root via lumbar puncture. The cisterna magna connects with the fourth ventricle. Therefore, theoretically, LASP is as effective as lateral ventricle puncture, but with less injury or complications.
Mononuclear cells can be readily separated from human umbilical cord blood and used for allogeneic transplantation with low immunogenicity 22 . There is evidence that hUCB-MCs are neuroprotective, and this highlights their potential for the treatment of neurodegenerative diseases 23 . In animal models of stroke and spinal cord injury, hUCB-MCs can migrate to the damaged brain area and replace damaged neurons, thereby improving the local brain microenvironment, reducing inflammation, and promoting regeneration of blood vessels and nerves10,11. Karlupia et al. 24 found that hUCB-MC therapy is more effective than cord MSC therapy in improving the neurological function of rodents with ischemic stroke.
In the present study, hUCB-MC transplantation via LASP showed good therapeutic effects for patients with MSA. A significant improvement in the neurological function was observed at 3 to 6 months after the first transplantation, as evidenced by significantly lower total UMSARS and UMSARS II scores compared with before treatment. Both scores tended to increase from the timepoint of best effect to the end of follow-up. The average annual increase in UMSARS score of patients with MSA is 17.2 25 , which is much higher than the experience of our patients (−1.6). This suggests the long-term efficacy of hUCB-MC transplantation via LASP.
Previous studies showed that stem cells can live in the brain and spinal cord for 3 months in animal models, including primate models26,27. This may explain why the timepoint of best effect after hUCB-MC transplantation in the present study was 3 to 6 months. The stable, long-term effects of transplantation of hUCB-MCs via LASP may be the result of activating endogenous neural stem cells, improving the brain microenvironment, regulating the immune system, and promoting secretion of various neurotrophic factors 28 .
In the present study, the neurological function of one MSA patient did not significantly improve after treatment and had even aggravated by the end of follow-up. This may be due to a long disease course (4 years) and severe symptoms (high UMSARS score) before treatment. Hence, early treatment for MSA is highly recommended.
In this study, we did not include a placebo surgery group because previous evidence confirmed that a placebo effect on neurological function could last up to 1 year after treatment 29 . To eliminate any influence of LASP on efficacy, we followed the patients for 3 to 5 years, and both the total UMSARS and UMSARS II scores were applied to evaluate neurological function. Nevertheless, this study was limited in that only seven patients with MSA were recruited and they were from the same hospital, which may impede the generalization of the current findings. In addition, only one assessment tool was used to evaluate neurological function. Future investigations with other evaluation methods are needed. Finally, it is worth noting that the effects of cell therapy on central nervous diseases have been investigated mostly in uncontrolled and nonrandomized clinical studies, and could not be shown definitively in placebo-controlled, randomized trials 30 . Therefore, multicenter, double-blind, placebo-controlled, randomized clinical trials need to be performed to confirm the findings of the present study.
In conclusion, this study showed that the neurological function of patients with MSA improved after hUCB-MC transplantation, and no severe adverse events related to transplantation were observed. These findings suggest that hUCB-MC transplantation via LASP is a safe and effective treatment method for MSA.
Footnotes
Acknowledgements
We thank the Mayora Group for funding support for Dr Dianrong Gong, Chief Physician of Neurology, Liao Cheng People’s Hospital, and her team to conduct research on the treatment of multiple system atrophy (MSA) using umbilical cord blood stem cell (UCBSC) transplantation through lateral puncture of the occipital space. We also thank all the authors for providing us with additional data upon our request.
Author Contributions
D.G. provided the concept and study design. M.Z. finished data collection. X.Y. and H.Y. finished data analysis. W.W. was a major contributor in writing the manuscript. All authors read and approved the final manuscript.
Ethical Approval
This study was approved by the medical ethics committee of the Liaocheng People’s Hospital (Approval No: 2013165).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the ethics committee of the Liaocheng People’s Hospital (Approval No: 2013165) approved protocols. The procedure for blood collection and isolation of hUCB-MCs was approved by the local ethics committee. This article does not contain any studies with animal subjects.
Statement of Informed Consent
All the participants were informed of the right to withdraw from the study at any time, and all participants provided written informed consent. Issues of privacy and confidentiality were strictly maintained.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by the Mayora Group.
