Abstract
Sanfilippo syndrome type III B (MPS III B) is an inherited disorder characterized by a deficiency of α-N-acetylglucosaminidase (Naglu) enzyme leading to accumulation of heparan sulfate in lysosomes and severe neurological deficits. We have previously shown that a single administration of human umbilical cord mononuclear cells (hUCB MNCs) into Naglu knockout mice decreased behavioral abnormalities and tissue pathology. In this study, we tested whether repeated doses of hUCB MNCs would be more beneficial than a single dose of cells. Naglu mice at 3 months of age were randomly assigned to either a Media-only group or one of three hUCB MNC treatment groups-single low dose (3 × 106 cells), single high dose (1.8 × 107 cells), or multiple doses (3 × 106 cells monthly for 6 months) delivered intravenously; cyclosporine was injected intraperitoneally to immune suppress the mice for the duration of the study. An additional control group of wild-type mice was also used. We measured anxiety in an open field test and cognition in an active avoidance test prior to treatment and then at monthly intervals for 6 months. hUCB MNCs restored normal anxiety-like behavior in these mice (p< 0.001). The repeated cell administrations also restored hippocampal cytoarchitecture, protected the dendritic tree, decreased GM3 ganglioside accumulation, and decreased microglial activation, particularly in the hippocampus and cortex. These data suggest that the neuroprotective effect of hUCB MNCs can be enhanced by repeated cell administrations.
Introduction
Sanfilippo syndrome type III B [mucopolysaccharidosis (MPS) III B] is an inherited lysosomal storage disorder caused by mutations in the gene encoding α-N-acetylglucosaminidase (Naglu) on chromosome 17q21 that leads to a deficiency of the Naglu enzyme in the degradative pathway of heparan sulfate, a glycosaminoglycan (GAG) (28). Initial development proceeds normally, but as heparan sulfate accumulates within lysosomes, damage occurs to multiple organ systems, including the brain. Neurologic symptoms of the disorder usually manifest after 2 years of age and include delayed development, progressive mental retardation, hyperactivity, aggressive behavior, and sleep disorders (35). Other organ systems are also affected, with these children often having hepatosplenomegaly, respiratory, cardiac, gastrointestinal, and vision problems (1). Death usually occurs between 11 and 20 years of age. There is no specific treatment for the disease, but with the advent of a knockout mouse model of MPS III B (25) in which the Naglu gene has been knocked out, it is hoped that more progress in the development of new therapeutic strategies will be possible.
Cell therapy has received recent attention as a potential treatment for lysosomal storage diseases. In 2005, Escolar and associates reported that infants with Krabbe disease who received cord blood transplants prior to the appearance of symptoms predominantly had normal cognitive development, whereas children treated after symptom development had little neurologic improvement (8). In a more recent study, bone marrow-derived adult stem cells were transplanted into the lateral ventricles in the MPS I (Hurler) mouse model, reducing accumulation of GAG in the brain and improving sensorimotor function (27). Our approach has been to examine the ability of human umbilical cord blood (hUCB) cells to reduce the underlying disease pathology and improve neurologic symptoms in the MPS III B Naglu knockout mouse model. These cells have been used successfully in many models of neurodegenerative disease and brain injury (33) and may be preferable to other cell sources such as bone marrow due to hUCB cells' low pathogenicity and immune immaturity. The mononuclear cell (MNC) fraction from hUCB is relatively rich in multipotent progenitors with extensive proliferative capacity (26,36). We have previously shown that a single hUCB MNC administration into the cerebral ventricle of presymptomatic Naglu-/- mice (15) or intravenous (IV) cell delivery at different disease stages (early symptomatic or late stage) (13) had a beneficial effect, probably due to enzyme delivery into the enzyme-deficient mutant mice. The benefits of enzyme replacement therapy with hUCB MNCs include decreases in disorder-related behavior and decreases of heparan sulfate accumulations in lysosomes of various internal organs. However, because of the gene mutation in MPS III B, there is a chronic deficiency in the Naglu enzyme, and it is not clear whether a single administration of hUCB cells is sufficient to replenish enzyme levels and arrest disease progression for the lifetime of the patient. Therefore, in this study, we examined whether intravenously administering repeated doses of hUCB MNCs would have a greater therapeutic effect than a single cell dose. Our outcome measures included behavioral outcomes, brain pathology, and accumulation of secondary degradation products, inflammatory state of microglia, and distribution of the administered cells within the brain. It was our hypothesis that repeated hUCB MNC doses would prevent the progressive neurodegeneration in the Naglu-/- mouse model of Sanfilippo type III B.
Materials and Methods
Animals
This study was carried out in accordance with NIH guidelines for the care and use of laboratory animals under the purview of the University of South Florida's Institutional Animal Care and Use Committee. The mice were housed three to five per cage in a temperature-controlled room on a 12-h on and 12-h off light schedule (lights on at 6:00 A.M.). Food and water were available ad libitum. The animals were housed in an isolation hood to reduce the likelihood of opportunistic infections that may occur with cyclosporine administration. All animals were obtained from our established Naglu mouse colony. Seventy-seven mice [63 homozygote (31 males, 32 females) and 14 wild-type (six males, eight females)] 3 months of age when the study began were assigned to one of the following groups: 1) Nontreated wild-type controls (No-Tx+/+, six males, eight females); 2) Media-treated homozygous Naglu mice (Media-/-, seven males, eight females); 3) homozygous Naglu mice treated with a single injection of hUCB MNCs (Single, Low, eight males, nine females); 4) homozygous Naglu mice treated with a single injection of hUCB MNCs (Single, High, eight males, eight females); 5) homozygous Naglu mice treated with six monthly injections of hUCB MNCs (Multiple, eight males, seven females).
Phenotyping of Mice
Phenotyping of the mice was determined from tail biopsies prior to entry into the study. Naglu enzyme activity was assayed as described by Li et al. (24), using a fluorogenic substrate. Briefly, tissue homogenates (25 μl) were incubated for 1 h at 37°C with an equal volume of 0.2 mM 4-methylumbelliferyl α-N-acetylglucosaminide (EMD Millipore, Billerica, MA, USA) in 0.1 M Na acetate buffer (Fisher Scientific, Pittsburgh, PA, USA), pH 4.3, containing 0.5 mg/ml bovine serum albumin (BSA; Sigma Aldrich, St. Louis, MO, USA); the fluorescence of released 4-metheylumbelliferone was measured after addition of 1.0 ml of glycine buffer (EMD Millipore), pH 10.5. One unit of activity corresponds to the hydrolysis of 1 nmol substrate per hour. Enzyme activity was determined fluorometrically using a microplate reader (Bio-Tek Instruments, Winooski, VT, USA).
Preparation of hUCB MNCs for Transplantation and Transplantation Procedure
Cryopreserved hUCB MNCs (U-CORD-CELL™, Saneron CCEL Therapeutics, Inc., Tampa, FL, USA) were used. The cells were quickly thawed in a 37°C water bath and transferred into Isolyte S, pH 7.4 (BBraun/McGaw Pharmaceuticals, Irvine, CA, USA) and centrifuged (180 × g/7 min). The cells were resuspended in Isolyte S, pH 7.4 and viability and cell numbers assessed using the 0.4% trypan blue (Invitrogen, Carlsbad, CA, USA) dye exclusion method prior to and following transplantation. The cell concentration was adjusted to 60,000 or 360,000 cells/μl.
Either a single dose of hUCB MNCs (3 × 106 or 18 × 106) or Media (Isolyte S, pH 7.4) was delivered intravenously in 50 μl. In multiple hUCB MNC administrations, 3 × 106 cells were delivered monthly for 6 months. Cell dosage for a single injection was 3 × 106 (control for each multiple injection) or 18 × 106 cells (control for the entire series of multiple injections). A 31-gauge needle (Hamilton, Reno, NV, USA), attached to a 10-μl syringe (Hamilton), was placed into the lumen of the tail vein of mice anesthetized with isoflurane (2–5% in O2, 2 L/min; Abbott, Abbott Park, IL, USA). Once the cells or media had been delivered, the needle was removed and pressure applied to the vein for 30–60 s to prevent blood loss. All animals, except No-Tx+/+ mice, were immunosuppressed with cyclosporine A (10 mg/kg IP daily; Novartis, New York, NY, USA) during the posttransplantation period.
Behavioral Tests
The animals were examined for general condition on a weekly basis throughout the study, and body weight was measured. Upon euthanization, bladder urine volume was measured as an indicator of disease state.
Anxiety behavior in an open field test and cognitive function (active avoidance) were performed at 1 week pretransplant and at monthly intervals for 6 months posttransplant. In mice with multiple cell injections, behavioral tests were performed 1 week after each injection.
Open Field
We measured anxiety of the mice in an open field using the Versamax® system (Accuscan Instruments, Inc., Columbus, OH, USA). The mice were placed in a Plexiglas box (35 × 20 × 30 cm) surrounded by an array of infrared beams. When the mice moved, the beams were broken, and the information was transmitted to a computer. The test period was 5 min in length during the light phase of the day. The specific parameters of motor behavior that were measured were the distance (cm) the animal traveled around the outside margin or corner of the cage (MRGDIST) and the distance traveled in the center of the cage (CTRDIST).
Active Avoidance
The active avoidance test was performed using the Shuttleflex Active Avoidance System (Accuscan Instruments, Inc.). Data were generated with the use of the Shuttle Flex Software. In the acquisition test (learning), the mouse was placed in a dark chamber and given 10 s (adaptation period) to explore the chamber before testing began. The conditioned stimulus (CS, 200 lux light, pulsed at 2,500 HZ) was administered for 5 s prior to delivery of the unconditioned stimulus, a mild electric shock (UCS, 0.5 mA). The shock was continued for a maximum of 10 s or until the mouse crossed to the other chamber. Trials were separated by 1-min intervals, and each test consisted of six trials daily during a 3-day period. The response time (latency) from conditioned stimulus onset until crossing to the other chamber and response time from unconditioned stimulus onset until last crossing was automatically measured in the acquisition phase. On the fourth day, the retention test (memory) was performed. The animal received only the conditioned stimulus for 5 s. The latency from conditioned stimulus onset until crossing to the other chamber was measured as was total test length during retention testing. The mice were tested prior to the initial hUCB or vehicle transplant and then at monthly intervals for 6 months.
Histology and Immunohistochemistry
After the last behavioral test, mice were euthanized under pentobarbital anesthesia (Henry Schein, Melville, NY, USA) and perfused transcardially with 4% paraformaldehyde (PFA; Sigma Aldrich) in 0.1 M phosphate buffer (PB; Sigma Aldrich), pH 7.2. The brains were then removed, postfixed, and cryoprotected in 20% sucrose (Sigma Aldrich) in PB overnight. Coronal or sagittal sections were cut at 30 μm in a cryostat (Richard Allen Scientific, Kalamazoo, MI, USA).
In order to examine hippocampal histology, cresyl violet (Sigma Aldrich) was used to stain Nissl according to standard techniques. Briefly, slides were rehydrated in distilled water and then placed in a solution of 0.1% cresyl violet in 1% acetic acid (Fisher Scientific) for 10 min. Slides were rinsed in distilled water for 2 min and then dehydrated through a series of alcohols, rinsed in xylene, and coverslipped with Permount (both Fisher Scientific).
Dendritic morphology of hippocampal neurons was examined from Golgi staining on selected tissue blocks from mice in the No-Tx+/+, Media-/-, and Multiple hUCB injection groups. The tissue blocks were fixed in 10% neutral-buffered formalin (Fisher Scientific) and stained using the Rapid Golgi staining method. Briefly, blocks of formalin-fixed tissue are immersed in a solution containing potassium dichromate (Sigma Aldrich) and osmium tetroxide (Sigma Aldrich) for 5–7 days. After rinsing the tissue, the tissue blocks are subsequently immersed in a solution of silver nitrate (Sigma Aldrich) for 28–42 h, resulting in the formation of a silver chromate precipitate in ~5% of neurons. The stained tissue blocks were then embedded in nitrocellulose (ICC Chemical, New York, NY, USA), sections cut on a sliding microtome (American Optical, Buffalo, NY, USA) at 120 μm and mounted before clearing in α-terpineol (Sigma Aldrich), rinsing with xylene and methylbenzoate (Sigma Aldrich), and coverslipping with Permount.
Distribution of hUCB MNCs in the brain was also determined. Three males and three females were selected from each of the transplant groups. An immunofluorescence technique with an antibody directed toward human nuclear antigen (HuNu; 1:50; EMD Millipore) was used to identify the transplanted cells within the brain. The primary HuNu antibody was preincubated with a monovalent goat anti-mouse Fab′ fragment antibody conjugated to fluorescein isothiocyanate (FITC; 1:200; Jackson Immunoresearch, West Grove, PA, USA) for 2 h. The brain tissue was blocked with 1% normal human serum (Sigma-Aldrich) in phosphate-buffered saline (PBS; ThermoScientific, Waltham, MA, USA) with 0.5% Triton X-100 (Sigma Aldrich) for 30 min prior to applying this antibody complex. The tissue sections were then incubated 24 h at 4°C. The next day, tissue sections were rinsed three times in PBS. The brain tissues were rinsed three times in PBS and then coverslipped with Vectashield with DAPI (Vector Laboratories, Burlingame, CA, USA). The slides were examined under epifluorescence using an Olympus BX60 research microscope (Olympus, Center Valley, PA, USA). Immunopositive cells were counted in the entire brain section (n = 3–5/mouse).
To identify microglia, the ABC method of immunohistochemistry was used. Sections were washed in PBS (pH 7.4) for 10 min and then incubated for 20 min in 40% methanol (Sigma Aldrich), 30% hydrogen peroxide (Sigma Aldrich) in PBS. The sections were again washed three times for 10 min in PBS and then blocked in 10% normal goat serum (NGS; Vector Laboratories), 0.03% Triton X-100 (Sigma Aldrich) in PBS. Tissue was then incubated overnight at 4°C in primary antibody solution [rabbit antiionized calcium-binding adapter molecule (IBA-1) 1:2,000 for microglia (Wako Chemicals, Richmond, VA, USA) in 3% NGS, Triton X-100, and PBS]. On day 2, the tissue was washed in PBS and incubated in secondary antibody solution [goat anti-rabbit 1:500 (Vector Laboratories, Burlingame, CA, USA), 3% NGS, 0.03% Triton X-100, PBS] for 1 h at room temperature, followed by three times 10 min of PBS. The sections were incubated in ABC (Vector Laboratories) solution for 1 h, and then the label was visualized using a diaminobenzidine kit (Vector Laboratories) for about 5 min, followed by three times 10-min rinses of PBS. Next, the tissue was dehydrated through a series of alcohols, cleared in xylene, and coverslipped with Permount. The sections were examined on an Olympus BX60 microscope. Photomicrographs of the fourth, fifth, and sixth cerebellar lobules, M1 motor cortex, dentate gyrus (DG) of hippocampus, and striatum were taken at 20× with an Olympus Magnafire camera. The density of Iba-1 immunolabeling per area (μm2) of all microglia and activated microglia, based on morphology, was determined using Image Pro Plus (Media Cybernetics, Rockville, MD, USA) and expressed as mean Iba-1+ cells/μm2.
A similar procedure was used to label monosialodihexosylganglioside (GM3 ganglioside) in the brain with the following modifications. The sections were blocked in 1% BSA with 1.5% NGS in 0.05% Triton X-100 and PBS for 1 h before being placed in primary antibody (monoclonal anti-GM3, 1:200, Cosmo Bio Co., Ltd., Carlsbad, CA, USA; in 1% BSA, 1.5% NGS, 0.05% Triton X-100, and PBS) overnight at 4°C. The sections were then washed before being placed in biotinylated goat anti-mouse antibody (1:200) for 1 h at room temperature. The remainder of the procedure was the same as for microglial labeling (above). The density of GM3 immunolabeling per area (μm2) was determined using Image Pro Plus and expressed as mean GM3+ cells/μm2.
Statistical Analysis
For analysis of the open field measure of anxiety, the data are expressed as means ± SEM for each month. A repeated measures analysis of variance (ANOVA) was performed with Bonferroni post hoc tests being administered where appropriate. Significance was set at p < 0.05. For the active avoidance test, the data across trials (6/day) across days (3 days for acquisition and 1 day for retention) were averaged for each month of testing and expressed as mean ± SEM. A repeated measures analysis of variance (ANOVA) was performed as described for anxiety. For microglial and GM3 immunohistochemical analysis, the data are expressed as mean positively labeled cells/μm2 ± SEM. Data were analyzed with a one-way ANOVA followed by a Tukey post hoc test. Bladder urine volume data are expressed as mean ± SEM and were analyzed with a one-way ANOVA followed by a Tukey post hoc test.
Results
General Condition of the Mice
As an indicator of well-being, we measured body weight throughout the study. Mice in all groups continued to gain weight for the duration of the study, but there were no significant differences in body weight between the groups. We observed five mice with alopecia distributed between all the groups, including the No-Tx+/+ controls; this is consistent with our previous observation that this is a condition found even in the background strain on which the Naglu mice were bred (16). Other complications included one mouse with a skin lesion, one mouse with an abscess, and two mice from the Media-/- group with a distended abdomen as a result of severe urine retention.
Open Field Anxiety Behavior After Administration of hUCB MNCs
The parameters that examined anxiety in the open field test measure the distance traveled around the perimeter of the cage (anxious, MRGDIST) or in the center of the cage (nonanxious, CTRDIST). Significant differences between groups were observed in the initial overall ANOVAs of both anxiety measures, with significant differences between groups (p < 0.0001), over time (p < 0.001) and a group by time interaction (p < 0.01) for MRGDIST (Fig. 1A) and significant differences between groups (p < 0.0001), over time (p < 0.001) and a group by time interaction (p < 0.0001) for CTRDIST (Fig. 1B). Post hoc testing indicated that there were significant differences between groups on both of the variables from 3 to 6 months after the initial transplant. The specific comparisons, mean ± SEM, and probability of the significant group differences are presented in Table 1. By the 5-month time point, the Media-/- mice were more active than all other animals in both the distance traveled in the margins and center of the cage (Fig. 1). This was most noticeable for CTRDIST where activity was 308.3 ± 118.8% of baseline at 5 months and 200.4 ± 71.1% at 6 months (Fig. 1B). Mice treated with hUCB cells exhibited the least amount of activity.

Human umbilical cord blood (hUCB) cells altered anxious behavior. Anxiety was measured based on whether the animal spent time next to the walls of the cage in the open field test (A) margin distance (MRGDIST) or in the open center of the cage (B) center distance (CTRDIST). +Significant differences between groups (p values varied between 0.05 and 0.0001).
Significant Differences in Anxiety Between Groups

MRGDIST, margin distance; CTRDIST, center distance.
Cognitive Function After hUCB MNC Administration
Learning and memory were tested in an active avoidance paradigm. During the acquisition phase of the test, the mice underwent six trials daily for 3 days followed by a retention test on the fourth day. In total, cognitive function was measured seven times, once prior to the initial hUCB MNC or vehicle transplants and monthly thereafter for 6 months. Data are reported for total duration of the test. There were significant differences between the groups (p < 0.0001) and over time (p < 0.001) as well as a group by time interaction (p< 0.001) for the total duration of the test during the learning acquisition phase (Fig. 2A). There were no significant differences prior to transplantation. Total test time for the No-Tx+/+ mice at the 2-month time point was 8.57 ±0.50 s. This was significantly less than the total test time of the Single, Low dose group 11.91 ±0.89 s (p<0.05) and the Single, High dose group (12.15 ±0.54 s, p<0.05) at this time. The No-Tx+/+ group also had faster test times than the Single, High dose group at 5 (9.04 ±0.58 s vs. 12.71 ± 0.50 s, p < 0.01) and 6 months (8.04 ± 0.60 s vs. 12.65 ± 0.50 s, p < 0.0001). The Media-treated group of Naglu-/- mice performed the active avoidance test faster than the mice treated with a Single, High dose of cells at 3 (8.67 ± 0.68 s vs. 12.24 ± 0.72 s, p< 0.01), 4 (8.53 ± 0.73 s vs. 12.33 ± 0.65 s, p < 0.01), 5 (8.64 ± 0.54 s vs. 12.71 ± 0.50 s, p< 0.001), and 6 months (7.88 ±0.18 s vs. 12.65 ±0.50 s, p< 0.0001). There were also differences between the hUCB MNC-treated groups on total test time. The Single, Low group took significantly longer to complete the test than the multiple injection group at 1 (11.16 ± 0.68 s vs. 7.44 ± 0.84 s, p < 0.001) and 2 months (11.91 ±0.89 s vs. 8.66 ±0.80 s, p< 0.05). The Single, High dose group also took longer to complete the test than the Multiple injection group at 2 (12.15 ±0.54 s vs. 8.66 ±0.80, p<0.05), 3 (12.24 ±0.72 s vs. 8.79 ± 0.85 s, p<0.05), 4 (12.33 ±0.65 s vs. 8.74 ±0.79 s, p<0.01), 5 (12.71 ±0.50 s vs. 9.43 ±0.60 s, p<0.05), and 6 months (12.65 ± 0.50 s vs. 8.71 ± 0.67, p < 0.01).

The effect of hUCB cells on learning and memory of α-N-acetylglucosaminidase knockout (Naglu-/-) mice in an active avoidance paradigm. The mice were placed in a shuttle box with an electrified grid. A warning light came on for 5 s followed by 10 s of shock for a total possible test time of 15 s. The shock turned off when the mouse escaped to the nonelectrified side of the shuttle box or the 15 s elapsed. The mouse could avoid the shock entirely by running to the nonelectrified compartment when the warning light came on. During the acquisition or learning phase of this test, there were six trials per day for 3 consecutive days. Performance was averaged across test days. During the retention phase of the test, the mice were placed in the same test box, but they did not receive a shock. (A) Total duration of the test during the learning phase. (B) Total duration of the test during the memory retention phase. +Significant differences between groups (p values varied between 0.05 and 0.0001).
When we examined total test time during the retention phase of the active avoidance test, there was a significant effect of group membership (p < 0.0001), time (p < 0.0001), and a group by time interaction (p < 0.05) (Fig. 2B). The No-Tx+/+ group (7.53 ± 0.85 s) completed the test faster than the Single, Low group (13.09 ± 0.73 s, p < 0.0001) at month 6, the Single, High group at 4 (9.05 ± 0.80 s vs. 13.47 ± 0.61 s, p < 0.01) and 6 months (7.53 ± 0.85 s vs. 11.83 ± 0.81 s, p < 0.01), and the Multiple injection group at 4 (13.36 ± 0.85, p < 0.01) and 6 months (12.29 ± 0.73, p < 0.001). The Media group performed significantly faster than the Single, Low group at month 6 (7.48 ± 0.64 s vs. 13.09 ± 0.73 s, p < 0.0001). The Media group also performed the test significantly faster than the Single, High group at 4 (9.34 ± 0.79 s vs. 13.47 ± 0.51, p < 0.01) and 6 months (7.48 ± 0.64 s vs. 11.83 ± 0.81 s, p < 0.01). Similar results were observed between the Media group and the Multiple dose group also at 4 (13.36 ± 0.85 s, p < 0.01) and 6 months (12.29 ± 0.73, p < 0.001). There were no significant differences between the hUCB MNC-treated groups.
hUCB MNCs Were Found Throughout the Brain
hUCB MNCs were identified in the brain (n = 6/group, three sections per mouse) using fluorescence immunohistochemistry with the human nuclear antibody, so they could be distinguished from mouse cells. Using this procedure, cells were found distributed throughout the brains of animals that had undergone all three cell-dosing regimens (Fig. 3). In all three groups, the cells were concentrated within the hippocampus, cerebellum, and olfactory bulb. Further, more cells were observed in those animals that received the greatest number of cells, based on counts from three sections/mouse. We observed approximately 23 hUCB MNCs per mouse in the Single, Low group, while mice that received a Single, High dose had approximately 32 cells. The most cells were observed in the Multiple dose group with a mean of 43 cells/mouse.
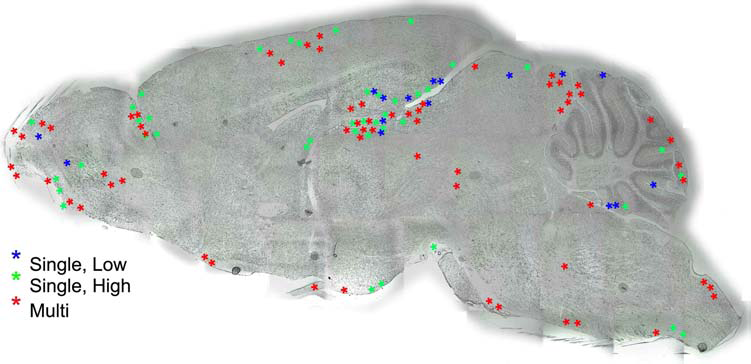
Distribution of hUCB cells in the brain of Naglu-/- mice. Sagittal sections through the brain were immunolabeled with an antibody to human nuclei and the location of labeled cells within the brain recorded.
hUCB MNCs Restored the Neuronal Cytoarchitecture of the Hippocampus
Serial sections of the brain in nontreated and celltreated Naglu mice were stained with 0.1% cresyl violet to examine the neuronal cytoarchitecture of the hippocampus. In Figure 4, representative photomicrographs from the CA3 region of the hippocampus are shown. Normal neurons were seen in control wild-type mice (No-Tx+/+; Fig. 4A). In CA3 of the Media-/- mice, the neurons were rounded and swollen, and many were vacuolated (Fig. 4B). In addition, the neuronal layer was thinner than in the No-Tx+/+ mice. Treating the Naglu-/- mice with hUCB MNCs tended to restore CA3 hippocampal architecture. The Single, Low dose group had more surviving neurons than the Media-/- mice, and fewer neurons appeared rounded and swollen (Fig. 4C). Mutant mice receiving the Single, High dose of cells, however, demonstrated disorganized neuronal hippocampal architecture: many swollen and vacuolated neurons were observed, and some had degenerated similarly to Media-/- mice (Fig. 4D). In contrast, Naglu mice receiving repeated administrations of hUCB MNCs (the Multiple group) showed many surviving neurons in CA3 (Fig. 4E). Similar results were observed in CA1, CA2, and the dentate gyrus (data not shown).

hUCB cells protect neurons in the hippocampus. Representative photomicrographs of the CA3 region of hippocampus in (A) wild-type No-Tx+/+ mice. (B) Media-/-; (C) Single, Low; (D) Single, High; (E) Multiple. (A) Normal neurons with welldefined cell processes in CA3 hippocampus were visible in the No-Tx+/+. (B) In Media-/- mice, swollen and rounded neurons (arrows) are noted. Narrowing of the neuron layer in CA3 was visible. (C) Most neurons in CA3 of Naglu-/- mice treated with the low cell dose appeared normal, but cell processes were not well defined. (D) Cytoarchitecture of CA3 in Single, High dose-treated mice showed disorganized neuron layers with numerous swollen cells. A number of glial cells dispersed in the stratum radiatum adjacent to the CA3 were also observed (asterisks). (E) Well-defined neurons were observed in the CA3 layer with multiple cell processes in the Multiple hUCB cell treatment group. Scale bar: 50 μm.
hUCB MNCs Protected the Dendritic Tree of Hippocampal Neurons
Structural integrity of the hippocampal neurons in Naglu mice receiving multiple cell transplants compared to nontreated (Media-/-) mutant and wild-type (No-Tx+/+) mice was evaluated using Golgi staining (Fig. 5). The Golgi stain, a sensitive method to determine neuronal plasticity, allows microscopic observation of the soma and entire dendritic domain of the neuron. In the No-Tx+/+ mice, we observed a normal neuronal dendritic tree structure with widespread branches both in CA1 (Fig. 5A, B) and the DG (Fig. 5G, H). Multiple spines are visible on dendritic branches (Fig. 5B and H). In nontreated Naglu mice (Media-/-), neurons were characterized by swelling and varicosities. The dendritic tree was distorted, and dendritic spines were lost along the branches (Fig. 5C, D, I, J). In contrast, morphology of hippocampal neurons in mutant mice with multiple cell administrations appeared normal (Fig. 5E, F, K, L). The dendrites were extensively branched, and spine density was very high. There were no varicosities on the dendritic branches.

hUCB cells altered dendritic morphology in the hippocampus. Representative photomicrographs of Golgi staining in CA1 region (A–F) and DG (G–L) of hippocampus. Wild-type No-Tx+/+ (A, B, G, H), Media-/- (C, D, I, J), and Multiple (E, F, K, L). Magnification in A, C, E, G, I, K = 40×; magnification in B, D, F, H, J, L = 1,008×.
hUCB MNCs Decreased GM3 in the Brain
GM3 ganglioside was used as a marker for accumulation of secondary storage products in the brain. Very slight GM3 immunolabeling was apparent in the brain of No-Tx+/+ wild-type mice, although there was some visible immunolabeling in the cortex (Fig. 6A–D). In contrast, the Media-treated Naglu-/- mice had higher GM3 immunoexpression in hippocampus (Fig. 6E), cortex (Fig. 6F), cerebellum (Fig. 6G), and striatum (Fig. 6H). In the Single, Low cell-treated group, there was modest GM3 immunostaining in the hippocampus (Fig. 6I), but GM3 immunoreactivity was visible in the cortex (Fig. 6J), cerebellum (Fig. 6K), and striatum (Fig. 6L).

GM3 gangliosides in the brains of Naglu-/- mice. Immunohistochemistry for GM3 ganglioside was used to label secondary degeneration products. In the No-Tx+/+ wild-type mice, there was very little GM3 in the (A) hippocampus, (B) cortex, (C) cerebellum, or (D) striatum. In the Media-treated Naglu-/- mice, there was extensive GM3 labeling in all regions examined (E–H). In mice treated with a Single, Low dose (I–L), Single, High dose (M–P), and Multiple doses (Q–T) of hUCB cells, there appeared to be less GM3 staining. Scale bar: 50 μm.
Similar results were observed in brain tissue from the Single, High dose group (Fig. 6M–P). Multiple doses of hUCB MNCs reduced GM3 labeling in all regions examined (Fig. 6Q–T).
When the density of GM3 immunoreactivity was quantified in the brain, there were significant differences between the experimental groups in hippocampus (p < 0.001) (Fig. 7A), cerebellum (p < 0.05) (Fig. 7C), and striatum (p < 0.001) (Fig. 7D) but not cortex (Fig. 7B). In the No-Tx+/+ group, the density of GM3 immunolabeling was 0.017 ± 0.003 (Fig. 7A). This was significantly less than the amount observed in the Media-/- group (0.12 ± 0.01; p < 0.001), the Single, Low group (0.094 ± 0.013; p < 0.001), and the Single, High group (0.10 ± 0.02; p < 0.001), but not the Multiple injection group (0.05 ± 0.01). However, the Multiple dose group had significantly less GM3 than the Media-/- (p < 0.01) and the Single, High dose group (p < 0.05). In cerebellum (Fig. 7C), the only significant difference between groups was between the No-Tx+/+ group (0.0046 ± 0.0008) and the Media-/- group (0.31 ± 0.17; p < 0.05), even though there was a tendency for hUCB MNCs to decrease GM3 immunolabeling. Similar to the observations in the hippocampus, GM3 labeling in the striatum (Fig. 7D) was significantly lower in the No-Tx+/+ group (0.011 ± 0.003) compared to Media-/- (0.08 ± 0.01; p < 0.001), and Single, High (0.066 ± 0.011, p < 0.001) groups. GM3 expression in the Single, Low (0.035 ± 0.004) and Multiple (0.014 ± 0.003) injection groups were similar to expression in the No-Tx+/+ group. Similarly, the density of GM3+ cells/μm2 in these two groups were also significantly lower than in the Media-/- group (p < 0.01 and p < 0.001, respectively) and the Single, High group was significantly different from the Multiple group (p < 0.01).
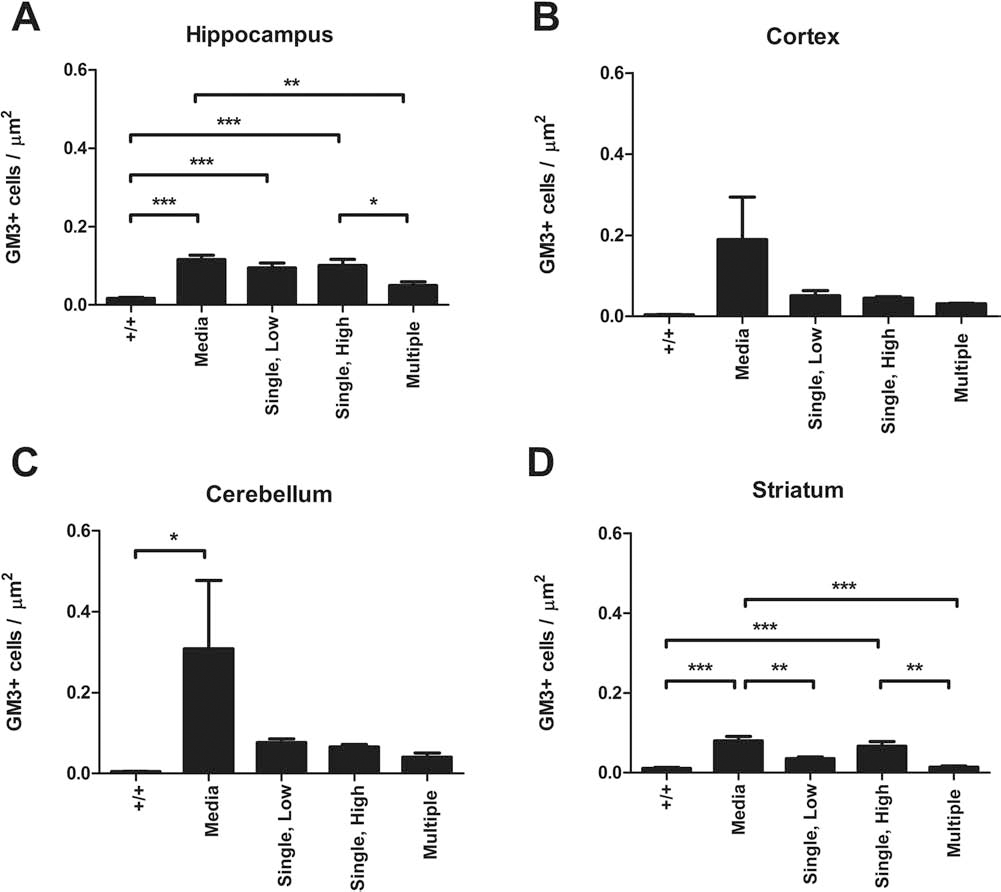
hUCB cells reduce GM3 ganglioside buildup in the brains of Naglu-/- mice. The number of monosialodihexosylganglioside-positive (GM3+) cells was quantified in (A) hippocampus, (B) cortex, (C) cerebellum, and (D) striatum. *p < 0.05; **p < 0.01; ***p < 0.001.
hUCB MNCs Decreased Urine Retention
One of the significant systemic pathologies in this mouse model is urine retention (17). Upon euthanization, bladder urine content was measured in all mice as a general indicator of hUCB cell efficacy to treat the systemic disease (17). As can be seen in Figure 8, bladder urine retention was significantly elevated in untreated Media-/- group (p < 0.01) and Single, Low hUCB group (p < 0.01) compared to No-Tx+/+ mice. As the dose of cells increased, there was a tendency for urine retention to decrease (Single, High hUCB group), until with Multiple hUCB injections, urine retention was significantly less than in the Media-/- and Single, Low groups, and not significantly different from the No-Tx+/+ group.
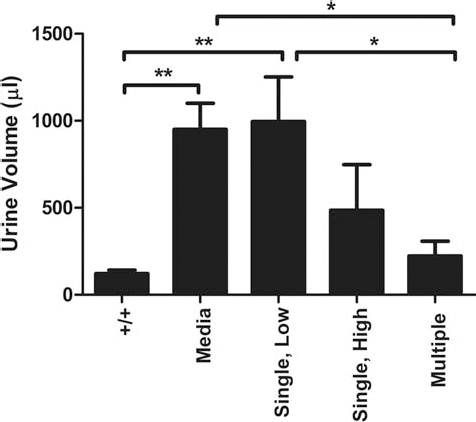
Bladder urine retention. The volume of urine was greater in the Media-only group compared to wild-type mice. While the Single, Low dose of hUCB cells did not alter urine volume, multiple doses significantly lowered urine retention. *p < 0.05; ** p < 0.01.
hUCB MNCs Altered the Phenotype and Number of Microglia
Microglia in the hippocampus, cortex, cerebellum, and striatum of wild-type (No-Tx+/+) mice exhibited a resting morphology with small cell bodies and thin processes (Fig. 9A–D). In contrast, microglia in hippocampus, cortex, and cerebellum of the Media-treated Naglu-/- mice had thicker, bushier processes, indicative of an activated state (Fig. 9E–G). In addition, amoeboid microglia, which are phagocytic, were also visible. Microglial cells in the striatum of these mice were similar to those of the wild-type mice (Fig. 9H). Treating Naglu-/- mice with hUCB MNCs altered microglial morphology. In animals that received a Single, Low dose of cells, the microglia had large cell bodies and long thin processes in all examined regions (Fig. 9I–L). A similar morphology was observed in the Single, High dose group (Fig. 9M–P). In the Multiple dose group, microglia in the striatum were similar to those of the other cell-treated groups, but in hippocampus, cortex, and cerebellum, the Iba-1-labeled cells were small with few processes (Fig. 9Q–T).

Iba1-positive microglia in the brains of Naglu-/- mice. In the No-Tx+/+ wild-type mice, microglia exhibited a small cell body and long thin processes typical of a resting state in (A) hippocampus, (B) cortex, (C) cerebellum, or (D) striatum. In the Media-treated Naglu-/- mice (E–H), the labeled microglia exhibited multiple phenotypes, with both ramified cells with bushy processes typical of activated cells and an ameboid morphology indicating the cells were phagocytic. Only in the striatum of the Media-/- was a resting morphology observed (H). In the Single, Low group (I–L), microglia morphology was altered further, with many microglia having large cell bodies, but thin processes emanating from the cell soma. A similar phenotype was observed in the Single, High dose group (M–P). In the Multiple dose group (Q–T), few ionized calcium-binding adapter molecule (Iba1) microglia were observed. Scale bar: 50 μm.
We also quantified the density of total labeled microglia and activated microglia in hippocampus, cortex, and cerebellum (Fig. 10). Only in the hippocampus was there a significant difference between the groups in the density of Iba-1+ cells per micrometer (p < 0.02) (Fig. 10A). Upon further examination, there were significantly more Iba-1+ cells in the Media-/- group (0.0018 ± 0.0011 cells/μm2) compared to the No-Tx+/+ group (0.00069 ± 0.00023 cells/μm2; p < 0.05). Treating the mice with hUCB MNCs decreased the density of microglia, but the differences were only significant in the Single, High (0.0006 ± 0.00004 cells/μm2, p < 0.05) and Multiple transplant groups (0.00054 ± 0.000007 cells/μm2; p < 0.01). When we only quantified the density of activated microglia, there were also significant differences between the groups (p < 0.02) (Fig. 10B). While there was a tendency for mice treated with a Single, Low dose of hUCB MNCs to have a greater density of activated microglia (0.00051 ± 0.00005 cells/μm2) than the Media-/- (0.00031 ± 0.00013 cells/μm2) and No-Tx+/+ (0.00036 ± 00010 cells/μm2), these differences were not significant. However, there was a significantly lower density of Iba-1 immunolabeling in the Multiple transplant group (0.00015 ± 0.00004 cells/μm2) compared to the Single, Low group (p < 0.05). In neither the cortex (Fig. 10C) nor the cerebellum (Fig. 10E) did the density of Iba-1 labeling of all microglia change significantly with treatment condition. However, there were significant differences in the amount of activated microglia in the cortex of cell-treated mice (p < 0.01). There were more labeled microglia in both the Single, Low (0.00033 ± 0.00006 cells/μm2; p < 0.01) and Single, High (0.00028 ± 0.00005 cells/μm2; p < 0.05) groups compared to the Multiple dose group (0.000006 ± 0.00002 cells/μm2) (Fig. 10D). There were no significant differences in the number of activated microglia in the cerebellum (Fig. 10F).

hUCB cells reduce activated microglia in the brains of Naglu-/- mice. The density of Iba-1-immunolabeled microglia was quantified in (A, B) hippocampus, (C, D) cortex, and (E, F) cerebellum. In addition to quantifying all microglia (A, C, E), the number of activated microglia were determined based on morphology (B, D, F). *p < 0.05.
Discussion
The premise behind administration of hUCB cells for the treatment of MPS III B is that the cells will provide the missing enzyme, thereby slowing or stopping the progression of the disease. We have previously shown that hUCB MNCs produce and have an ability to release the Naglu enzyme in vitro (15). When the cells are administered into pregnant dams, they transmigrate into the embryos, widely distributing in the tissues of the developing embryos where they restored enzyme activity prenatally (12). The hUCB MNCs also restore heparan sulfate levels in multiple organ systems after IV administration into Naglu mutant mice at early symptomatic or late disease stage (13), which is the likely reason that liver and kidney pathology are reduced. Similarly, the cells restore hippocampal cytoarchitecture when administered intracerebroventricularly (15) and intravenously (13). In the present study, we examined whether administering repeated doses of hUCB MNCs would have a greater therapeutic effect than a single dose. Our results suggest that repeated administration of hUCB MNCs has a greater effect on the underlying pathology than a single administration of cells.
Anxiety Behavior of Naglu-/- Mice Is Altered by hUCB MNCs
Previous studies have demonstrated that the Naglu-/- mice appear less anxious than wild-type mice. In the open field test, time spent in the open center of the cage is considered to be indicative of less anxiety; these mice have been observed to enter the center more often and travel further at higher speed than wild-type controls (23). Our results are consistent with this earlier observation; the Media-treated mice in our study spent more time in the center of the open field test chamber and covered more distance in this region than mice from any of the other groups. Langford-Smith et al. (23) hypothesized that this decreased “anxiety” was actually a function of the observed hyperactivity and not reflective of anxiety per se. In support of this hypothesis, the Media-treated mice also traveled further along the margins of the cage, suggesting that this apparent lack of anxiety is a function of hyperactivity. However, distance traveled in the margins at 5 months was only 140% of baseline compared to distance traveled in the center (308% of baseline); hyperactivity cannot fully explain the preponderance of distance traveled in the center of the cage. Further, performance of the Naglu-/- mice is consistent across different tests of anxiety. In the elevated plus maze, these mice were shown to be less “anxious” than normal mice, spending more time in the open arms of the test apparatus than normal mice (5). Further, there were consistent differences observed between the Media-treated Naglu-/- mice and mice treated with hUCB MNCs. Regardless of dose of cells, the treated mice decreased the distance traveled in the center of the cage. Similar observations have been made in the permanent middle cerebral artery occlusion model of stroke in rats where hUCB MNCs delivered intravenously decreased hyperactivity and anxiety-like behaviors (39).
These results raise an interesting point. Repeated injection of cells was not more effective than a single low-dose injection of cells at reducing anxiety in these mice. Further, the single injection maintained these behavioral changes for 6 months posttransplant. However, it is not clear how the cells induced these changes. Some cells did survive in situ for the duration of the study and could have been capable of directly interacting with host cells to produce enzyme (15). Alternatively, the cells produce many trophic factors and cytokines that could support neural function (4). Finally, the cells may have induced epigenetic changes in the host mouse to induce long-term changes. For example, administration of hUCB MNCs in an animal model of stroke increases glial-derived neurotrophic factor in host cells (3). Moreover, a recent study demonstrated that multiple small doses of hUCB MNCs (2.5 × 106 per injection) intravenously administered even into symptomatic mice modeling ALS significantly delayed motor functional deterioration and increased mouse life span by protecting motor neurons from inflammatory effectors (14). Similarly, injecting 106 hUCB MNCs into the transgenic presenilin–amyloid precursor (PSAPP) mouse model of Alzheimer's disease monthly for 6 months, beginning at 6 months of age, decreased cognitive impairment, amyloid load, and microgliosis (6). In addition, injections of hUCB MNCs every second day beginning 7 days after myocardial infarction and ending on the 13th day showed that cardiac function improved more with repeated injections and significantly increased the level of interleukin (IL)-10 in the heart (40).
In addition to examining anxiety behavior in the Naglu-/- mice, investigators have also examined cognitive function, but the results of these tests are less consistent than observations of anxiety. In the Morris water maze test, Naglu-/- mice consistently took longer to find the hidden platform than wild-type controls (11). In a fear conditioning paradigm of learning and memory testing, these mice, young and old, performed normally in the contextual test, but older mice failed to learn the association between a tone and a mild shock to the paws (19). The authors hypothesized that the mice may have had a deficit in hearing as opposed to a learning deficit since the mice behaved normally on the contextual test (19). We have previously shown that learning and memory had a tendency to improve after a single administration of hUCB MNCs compared to Media-treated animals, but this improvement was variable between trials in the active avoidance paradigm and was inconsistent over time (13). In our current study, we averaged all learning or retention trials to reduce spurious variations during testing. However, this analysis of the test results demonstrated that Naglu-/- mice were no longer impaired compared to the wild-type mice. Further, administration of hUCB MNCs, especially at the highest single dose actually lengthened exposure to shock and total length of the test, suggesting that the cells interfered with cognitive performance. The exception to this is with repeated cell administrations, where total length of the test was indistinguishable from that of the wild-type and media-treated groups. Our data suggest that this mouse model poorly reflects cognitive/behavioral effects of enzyme depletion even while remaining a very good biochemical model for examining the underlying pathology of the enzyme deficiency. Part of the issue may be the nature of the cognitive tests. All tests that have been reported in the literature rely on either visual or hearing acuity, which may be impaired in this mouse model (18). Further, it was recently reported that these mice also have peripheral neuropathies that significantly reduced their pain sensitivity, which could directly interfere with their ability to perform the active avoidance test that we performed (10). Another issue that may interfere with the behavioral outcomes is the pathology of other organ systems in this mouse model. We have observed abnormalities in this model such as hydrocephalus, growth retardation, and urine retention (Fig. 8) that are not consistent with the disease profile in children and that can interfere with the ability of the mouse to perform behavioral tests (16,17).
Distribution of hUCB MNCs in the Brain
The majority of hUCB MNCs observed in the brains from all transplanted groups tended to cluster in the cerebellum, hippocampus, and olfactory bulb, although cells from the repeated and high-dose groups were also found in the medulla and cortex. The preponderance of cells in the hippocampus and cerebellum is interesting since these regions are known to have a greater degree of degeneration than other brain regions in MPS III B (5,25). These data are encouraging, but the number of surviving cells found was small, even when considering the number of cells in the entire brain. To make these calculations, we multiplied the average number of cells per section by the number of 30-μm-thick sagittal sections through the brain [~125 per hemisphere based on the published size of C57BL/J5 mouse brain (30)]. When expressed this way, approximately 0.02% of the transplanted cells in the repeated administration group and 0.01% cells in the single cell dose groups survived in the brain. This is consistent with observations in other models of brain injury in which cells are delivered intravenously (9,21); the majority of cells are found in spleen, lung, liver, and kidney. The difference between those models and this one is that in the Naglu-/- mouse there is widespread organ damage. We previously showed that hUCB MNCs were found in liver, kidney, and spleen in addition to the brain 6 months after a single IV administration, and these cells continued to produce heparan sulfate (13) to ameliorate damage in these organs as well as the brain. This widespread distribution is one of the major benefits of IV hUCB MNC delivery. Given these earlier observations, a biodistribution analysis was not performed in this study; future studies will examine this issue more thoroughly.
hUCB MNCs Reduce the Pathology in the Brain
Not only is there a loss of neurons with age in the Naglu-/- mouse model, but those neurons that survive have dystrophic axons as well as pre- and postsynaptic pathology (38). Repeated injections of hUCB MNCs produced the greatest neuroprotection. Hippocampal structural architecture remained intact in the repeated cell dose-treated mice compared to the Media-treated Naglu-/- mice or mice treated with single cell doses. Not only were neuronal cell bodies protected, but hUCB MNC administration resulted in restoration of the dendritic tree, with many branching dendrites and extensive spine formation. Alterations in the dimensions of the dendritic domain (branches and spines) reflect changes in the functional capacity of the neuron (31,32,40). Dendritic spines are the postsynaptic neurostructural component and the primary anatomical loci for transfer of information between neurons (41). The increase in spine density would suggest that synaptic connectivity is increased after repeated injections of hUCB MNCs. This is consistent with a recent report showing that hUCB cells increased spine density in the aged brain (34).
The underlying pathology in MPS III B is the buildup of heparan sulfate in tissue because of the lack of the Naglu enzyme. That GM3 ganglioside levels were reduced with repeated cell dosing to the level observed in wild-type mice, suggests that heparan sulfate was being broken down and cleared from these mice and that the administered cells were providing the missing enzyme. Further support is provided by the observed decrease in urine retention in the bladder. This interpretation is consistent with our previous demonstration that hUCB MNCs produce and release the Naglu enzyme (15). We had previously shown that prenatal transplantation of a single dose of 3 × 106 cells became incorporated into organs throughout the body and restored enzyme levels to that of the pups' heterozygote parents (12). While prenatal delivery may be the best way to treat and minimize the severity of the disease, prenatal diagnosis is rare, making it essential to develop a treatment that can be delivered to the child at early age to minimize organ damage throughout the body and stop the progression of the disease. While the single hUCB MNC doses did alter GM3 levels in the brain, the greatest effect was with repeated cell dosing. The repeated cell treatment would continually provide new cells that could lead to a gradual increase in surviving cells over time or a replacement of hUCB MNCs that did not survive long term. The latter may be a problem since in other models of hUCB MNC transplantation for brain injury or disease, few cells survive long term (3). Even so, the hUCB MNCs were found distributed throughout the brain, with more cells observed after IV administration of repeated doses than with a single injection of either a low or high dose.
hUCB MNCs Reduce Microgliosis and Activated Microglia
There was a striking reduction in microgliosis and microglial activation after hUCB MNC treatment in this study. In the Naglu-/- mice, there is a significant upregulation of inflammatory responses that involve both microglia and astrocytes (7,29). When inflammatory mediator and neurotrophin expression were evaluated in this mouse model, there was a loss of brain-derived neurotrophic factor (BDNF) expression in the cerebrum but an increase in the cerebellum (37). In contrast, chemokine (C-C motif) ligand 3 [CCL3; also known as macrophage inflammatory protein 1α (MIP1α)] increased in both cerebrum and cerebellum, along with caspase 11, an activator of caspase 1 and 3, which is therefore capable of modulating apoptotic pathways. In conjunction with this increase in inflammatory mediators, there were significant increases in the superoxide ion (37). In addition to the local brain inflammatory response, there is activation of the immune system that results in infiltration of T-cells, B-cells, and macrophages (7) into the central nervous system (CNS) and induction of autoimmune responses. Administering hUCB MNCs may decrease neuropathology through modulation of these inflammatory and immune processes. Indeed, we have shown that IV hUCB MNCs and hUCB-derived T-cells decrease the number of activated microglia in the hippocampus in the aging brain, thereby increasing neurogenesis (2,34). In our current study, all three cell-dosing regimens reduced the density of Iba-1 microglial immunostaining in the hippocampus. Interestingly, the pattern of the density of activated microglia in this region and in the cortex was different, with most microglia being observed in the single, lowdose group and the least in the multiple dose group. This might suggest that the single low-dose stimulated proliferation of microglia. It has been hypothesized in a stroke model of brain injury that proliferation of microglia is essential to their neuroprotective functions (20); blocking microglial proliferation early after stroke leads to greater brain damage and exacerbation of proinflammatory conditions in the injured brain (22). We have shown that the hUCB MNCs produce numerous neurotrophic and growth factors, including granulocyte and macrophage colonystimulating factor (GM-CSF), that may be instrumental in initiating this proliferation and the production of insulinlike growth factor-1 (IGF-1) by microglia (4). The decrease in activated microglia in the single, high, and multiple injection groups may reflect a resolution of the underlying pathology as more heparan sulfate is cleaved, leading to a decrease in the proinflammatory state. Alternatively, the direct effect of hUCB MNCs on inflammation may reduce the proinflammatory toxicity of the environment allowing compromised, but surviving, neurons to maintain function.
In summary, we demonstrated that hUCB MNCs are particularly effective at modulating anxiety in the Naglu-/- mice, although the dosing regimen has only a small influence on the behavioral effects. The cells are found throughout the brain with more accumulation in olfactory bulb, hippocampus, and cerebellum than in other brain regions. In the hippocampus, neuronal damage is further limited by repeated cell injections versus a single dose of cells. Similarly, in hippocampus, cortex, cerebellum, and striatum, repeated cell administrations reduce GM3 accumulation more than single cell doses, and this reduction is accompanied by greater reductions in microglial activation. These data suggest that repeated administrations of hUCB MNCs produce a greater amelioration of the underlying disease pathology. Further studies will be necessary to determine if this treatment regimen can slow the progression of the disease, thereby increasing survival and minimizing disease symptoms and whether the improved outcome is a function of enzyme administration, decreased inflammation, or both.
Footnotes
Acknowledgments
This research was supported in part by the Children's Medical Research Foundation, Inc., and the International Organization of Glutaric Acidemia. The cord blood cells were provided by Saneron CCEL Therapeutics, Inc. A.E.W. and S.G.D. are consultants to Saneron CCEL Therapeutics, Inc. In addition, S.G.D., A.E.W., P.R.S., C.D.S., and N.K.N. are inventors on multiple cord blood patents. C.D.S. is Senior Vice President of Research and Development, and N.K.N. is President, Corporate Development and Operations at Saneron CCEL Therapeutics, Inc. P.R.S. is cofounder of Saneron CCEL Therapeutics, Inc.
