Abstract
CD8 T cells play a key role in cancer immunotherapy and allograft rejection. However, it is not clear how they kill cells and tissues that do not have the agonist peptide-major histocompatibility complex (MHC) on their surface, as in the settings of MHC class I deficient tumors and indirect rejection of MHC-mismatched transplants. CD8 T cells might respond to agonist antigen cross-presented on hematopoietic cells, leading to a “bystander” rejection. Alternatively, they may recognize agonist antigen cross-presented on recipient endothelial cells and kill the tissue’s vital blood supply. The latter mechanism predicts that all non-vascularized grafts, grafts dependent on in-growth of recipient blood vessels, will be susceptible to CD8 T cell mediated indirect rejection. In contrast, we show here that non-vascularized transplants, bearing the same agonist antigen, are not universally susceptible to this rejection pathway. Non-vascularized skin, but not islet or heart tissue transplants were indirectly rejected by CD8 T cells. Furthermore, CD8 T cells were able to indirectly reject skin grafts when recipient MHC class I expression was restricted to bone marrow derived cells but not when it was restricted to radioresistant cells (e.g. endothelial cells). These findings argue against a major role for endothelial cell cross-presentation in killing of tissue that does not present the agonist peptide-MHC class I. Instead, the data suggests that cross-presentation by recipient hematopoietic cells underlies the CD8 T cell mediated killing of tissue that is unable to directly present the target peptide-MHC class I.
Introduction
Allorecognition and priming of donor specific T cells involves presentation of the alloantigen on either host or donor derived professional antigen presenting cells. Once activated, many individual CD8 effector T cells can kill target cells via direct recognition of alloantigen agonist peptide-major histocompatibility complex (MHC) complexes on the donor cell (the direct pathway). A smaller fraction of CD8 effector T cells, restricted to recognizing the alloantigen peptides presented in the context of host MHC, can also reject donor cells via the indirect pathway 1 . How CD8 T cells indirectly reject grafts is not clear. CD8 T cells have a limited capacity to kill bystander cells in tumors or allografts that do not express, or have lost expression, of the appropriate MHC class I1–3. Nevertheless, the indirect pathway is an important contributor to graft rejection 1 and both CD4 and CD8 T cells can cause indirect rejection4–7. However, there is a differential sensitivity of tissues to indirect rejection 7 where CD8 T cells can indirectly reject a skin graft but not a heart transplant from the same donor 5 . The disparate outcome with heart and skin grafts may be due to differences in the cell types within the graft, or it may be due the origin of the vasculature5,8–11. The heart is typically transplanted as a “vascularized graft” such that the blood vessels in the graft are of donor origin, while skin is typically a “non-vascularized” graft, with vessels of recipient origin. Thus, CD8 T cells might target donor antigen cross-presented on the recipient blood vessel endothelial cells that have grown into the graft tissue 5 , killing host derived blood vessels needed for graft survival. However, direct in vivo evidence for this mechanism of indirect rejection is lacking.
We tested two key predictions of the endothelial cell targeting model: (1) that all non-vascularized grafts would be susceptible to CD8 T cell mediated indirect rejection, and (2) that the effector mechanism of indirect rejection of a skin graft would depend on cross-presentation of donor antigen on recipient cells that are relatively radioresistant (e.g. endothelial cells or their precursors) 12 , and not cross-presentation on recipient hematopoietic cells.
Materials and Methods
Ethics Statement
This study was approved by the Health Sciences Animal care committee at the University of Alberta (AUP00000215). All care and handling of animals were carried out in accordance with the guidelines of the Canadian Council on Animal Care.
Mice
Adult C3H (H-2k), C57BL/6 (B6; H-2b) and B10.BR (H-2k) mice were purchased from Jackson Laboratory (Bar Harbor, ME, USA) and Charles River Canada (Saint-Constant, QC, Canada). Adult CBA mice (H-2k) were obtained from NCI Frederick (Frederick, MD, USA). B6.Rag1-/-, B6-CD45.1, and B6 mice expressing green fluorescent protein, B6.GFP; B6-Tg (UBC-GFP) 30Scha/J, originally from Jackson Laboratory, were bred in-house. B6.Rag1-/--KbDb−/− mice were generated by crossing B6.Rag1-/- with B6.H-2Kbtm1-H-2Dbtm1N12 (originally from the NIAID Exchange Program, NIH: 004215; Class I knockout mice) 13 . B6.Rag1-/--GFP (H-2b) mice were generated by crossing B6.Rag1-/- with B6.GFP mice described above. Female MataHari mice 5 on the B6.Rag1-/- background were bred at University of Alberta.
Skin Transplantation
Full thickness trunk skin was transplanted onto the dorsum of recipient mice. Briefly, 1 cm2 of donor skin was secured with sutures to the recipient graft bed and bandaged for 7 days. Skin grafts were considered rejected at the time when >90% surface area was necrotic. Histological analysis of skin grafts was performed at the times indicated.
Islet Isolation and Transplantation
In all, 500 islets were transplanted beneath the renal capsule as previously described 14 . Briefly, recipients were made diabetic by a single i.p. injection of streptozotocin (Sigma-Aldrich Canada, Oakville, ON, Canada). Diabetes was confirmed as blood glucose of >20.0 mmol/L and grafts were considered rejected when blood glucose levels exceeded 15 mmol/L over 2 days. When indicated, MataHari mice were primed by an i.p. injection of 5 million irradiated B6.Rag1-/- male splenocytes 5 days prior to islet transplantation.
Heterotopic Neonatal Heart Transplantation
Donor neonatal hearts were transplanted either under the skin of the ear pinna or under the renal capsule, as described previously 15 .
Generation of Bone Marrow Chimeras With Monoclonal CD8 T Cells
Recipient mice were lethally irradiated with 12 Gy in split doses prior to receiving intravenously (i.v.) 20–40 million bone marrow cells and received TMX/SMP antibiotic (Strathcona Pharmacy, Edmonton, AB, Canada). Chimeras received 0.8 to 1.0 million FACS sorted splenic T cells (CD8β+TCR Vβ8+; 85-95% purity) from MataHari female mice that had been immunized 5 days earlier; immunization was by an i.p. injection of 5 million irradiated (20 Gy) B6-CD45.1 male splenocytes. To increase survival of transferred T cells 16 , the chimeric recipients were given NK cell depleting antibody (PK136; 0.3 mg per injection, produced in house), at day -5, -2, and +1, relative to T cell transfer. Chimeras were given skin grafts 1 day after T cell transfer.
Flow Cytometry
Chimerism was assessed by flow cytometry for GFP, H-2Kb and H-2Db. Antibodies against TCR Vβ8, CD8β, CD44, H-2Kb, and H-2Db were from BD Pharmingen (San Diego, CA, USA) and eBioscience (San Diego, CA, USA). Data were acquired using an LSR II (Becton Dickson, Sunnyvale, CA, USA) and analyzed with FlowJoTM (Treestar software, OR, USA).
Statistical Analysis
Flow cytometry data were compared by the Mann-Whitney U Test. Graft survival was compared using the log-rank (Mantel-Cox) test. All statistical analyses were done using Prism (GraphPad Software, La Jolla, CA, USA). A P value of ≤ 0.05 was considered statistically significant.
Results
Indirect CD8 T Cells Reject Non-Vascularized Grafts of Skin, but not Islets or Heart Tissue
MataHari females have a monoclonal population of CD8 T cells specific for the WMHHNMDLI peptide of the male antigen (HY) in the context of H-2Db. When on the B6.Rag1-/- (H-2b) background, as in our study, they lack or have greatly reduced numbers of CD4 T cells 5 . HY from male grafts of an MHC-mismatched donor (e.g. C3H; H-2k) can only be recognized through the indirect pathway (Fig. 1A). To begin to test whether the vascularized versus non-vascularized nature of donor grafts is the key determinant of indirect rejection, we assessed indirect rejection of islet grafts (Fig. 1A). Islet grafts, like skin grafts, are non-vascularized (revascularization is primarily by recipient neoangiogenesis).17,18

Rejection of skin but not islet grafts by CD8 T cells via the indirect pathway. MataHari female mice received a skin or islet graft from the donor mice indicated. (A) List of donors and the potential rejection pathways in a MataHari female recipient. (B) Top: Skin graft survival for C3H male (n = 8) and B6.Rag1-/- male (n = 9) donors. Bottom: Islet graft survival from C3H male (n = 12), C3H male* (n = 5), C3H female (n = 2), CBA male (n = 2), B10.BR male (n = 4) donors, B6.Rag1-/--KbDb-/- male (n = 4) donors, all surviving to > 100 days, and B6.Rag1-/- male (n = 10) donors; P < 0.0001. For C3H male* donors, recipient MataHari female mice were primed to the HY antigen 5 days prior to islet transplantation.
Consistent with previous studies showing CD8 T cells can indirectly reject MHC class I deficient skin grafts 19 or grafts with the wrong MHC allele for the CD8 T cell,5,20 MataHari (H-2b) female mice indirectly rejected male C3H (H-2k) skin grafts (Fig. 1B). However, MataHari female mice did not reject male C3H islet transplants via the indirect pathway (Fig. 1B) despite their ability to rapidly directly reject MHC-matched (H-2b) donor male islets 21 . MataHari female mice were also unable to indirectly reject islets from male CBA (H-2k), B10.BR (H-2k), and B6.Rag1-/--KbDb−/− (H-2b) donors (Fig. 1B). A lack of T cell priming did not appear to be responsible for the lack of rejection, as even MataHari female mice previously primed with male antigen 21 did not indirectly reject islets (Fig. 1B).
To test whether differences in graft location might affect the outcome, we gave MataHari female recipients non-vascularized, rather than vascularized heart grafts, either under the renal capsule 15 or under the skin of the ear pinna15,22. While it can be difficult to visually detect the beating of a heart graft under the ear skin, we could discern visual signs of muscle contraction in most male C3H (H-2k) and male CBA (H-2k) heart grafts (Supplementary Table 1). In contrast, none of the male MHC-matched (H-2b) heart grafts could be seen contracting, even when observed early post-transplant (day 35). All male CBA, male C3H, and female C3H heart grafts under the ear pinna had well organized cardiac myocytes with no lymphocytic infiltration. In contrast, male MHC-matched (B6.Rag1-/-) grafts showed extensive fibrosis with no cardiac muscle fibers present (Supplementary Fig. 1A). For grafts under the kidney capsule at 100 days, contractions and intact cardiac myocytes (Supplementary Fig. 1B) were seen for all male CBA grafts while male B6.Rag1-/- neonatal hearts had been completely rejected. These results demonstrate that a non-vascularized heart transplant, like its vascularized counterpart 5 , resists indirect rejection by CD8 T cells.
MHC Class I is Required on Hematopoietic Cells but not on Recipient Radioresistant Cells for Indirect Rejection by CD8 T Cells
We directly examined the requirement for MHC class I expression by radiosensitive hematopoietic cells (such as antigen presenting cells (APCs)) or radioresistant cells (vascular endothelium). We generated reciprocal radiation chimeras of the H-2b haplotype between B6.Rag1-/--GFP and B6.Rag1-/--KbDb−/− mice (Supplementary Fig. 2), transferred to them primed and sorted MataHari female T cells 6 weeks post bone marrow transplantation, and then challenged these mice with two male skin grafts, from C3H and B6.Rag1-/- donors (Fig. 2A). If HY cross-presented by host endothelial cells were the target of indirect rejection, chimeras with MHC I on radioresistant cells (MHC I−/− to MHC I+/+ chimeras) would be expected to reject the C3H male skin graft due to the ingrowth of recipient radioresistant endothelial cells cross-presenting HY. However, chimeras with MHC I on radioresistant cells and lacking MHC class I on radiosensitive cells accepted male C3H skin grafts for greater than 100 days despite rapid rejection of B6 male skin grafts in the same recipients (Fig. 2B and Supplementary Fig. 3). These data suggest that MHC class I is required on radiosensitive cells for CD8 T cell-mediated indirect rejection and that any cross-presentation by recipient endothelial cells does not provide a sufficient target for indirect rejection. In contrast, MHC I+/+ to MHC I-/- chimeras (lacking Class I on radioresistant cells) could reject both male C3H and B6 skin grafts (Fig. 2B and Supplementary Fig. 3) despite the lack of cross-presentation on recipient radioresistant endothelial cells. The CD8 T cells expressed similarly high levels of CD44 (Supplementary Fig. 2B) and rejected B6 male skin grafts with equal kinetics in the two groups of chimeras (Fig. 2B), despite rejection of male C3H grafts only occurring in the MHC I+/+ to MHC I-/- chimeras. Although a fraction (30%) of C3H grafts survived long-term in MHC I+/+ to MHC I-/- chimeras, these grafts had evidence of rejection (Supplementary Fig. 3B). Taken together, there is a requirement of MHC class I to be present on radiosensitive cells but not radioresistant cells for indirect rejection by CD8 T cells to occur.
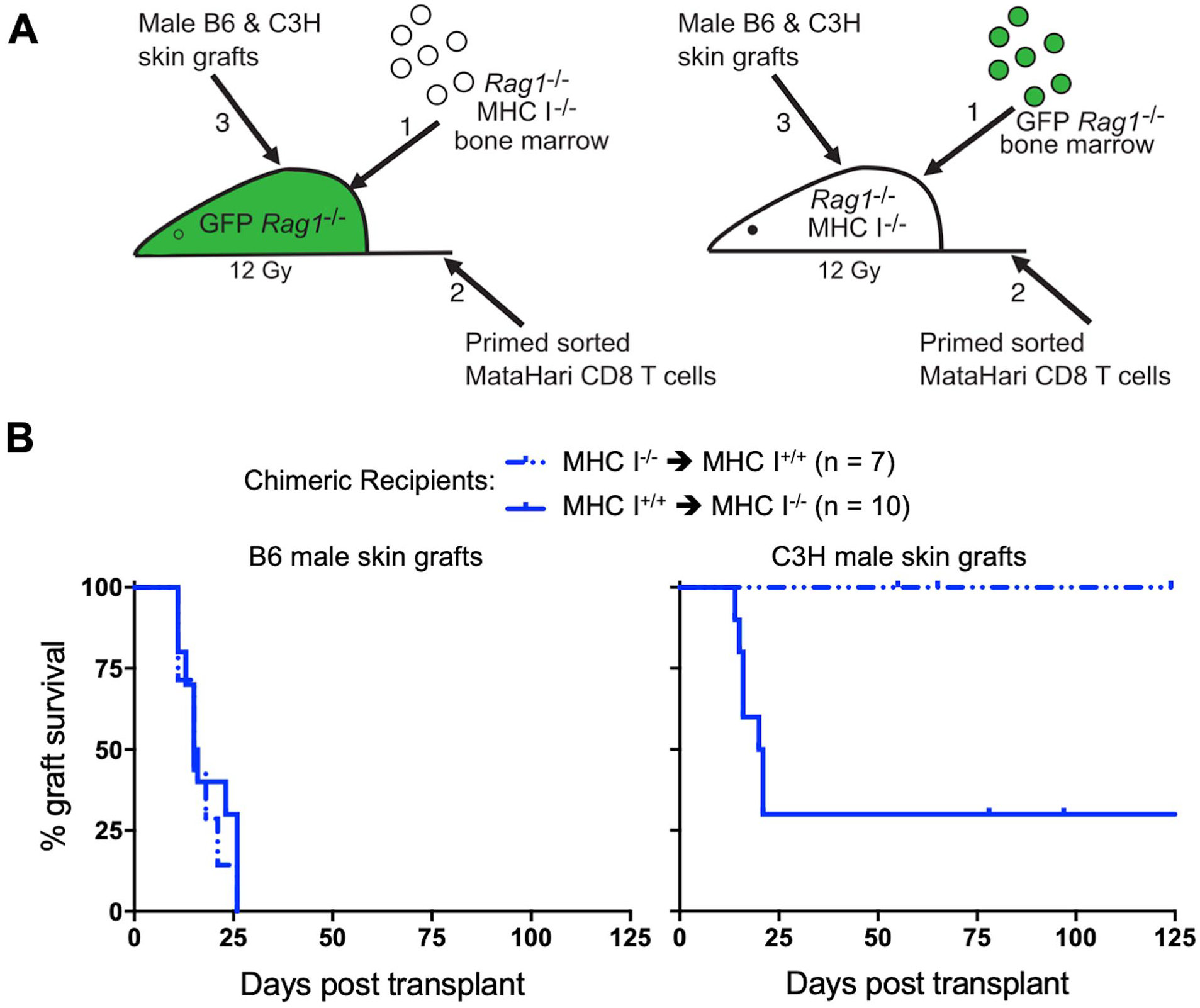
Indirect CD8 T cell mediated indirect rejection of skin depends on MHC class I expression on hematopoietic cells and not radioresistant cells. (A) Diagram of the experimental approach. Sorted CD8 T cells from MataHari female mice primed to HY 5 days earlier were transferred to chimeras 6 weeks post bone marrow transplantation. Two skin grafts, one from male C3H and one from male B6 donors, were given to each chimera one day post T cell transfer. (B) Survival curves of male skin grafts on bone marrow chimeras, with B6 skin on the left and C3H skin on the right (solid line for MHC I+/+ to MHC I-/-, n = 10; dashed line for MHC I-/- to MHC I+/+, n = 7; P = 0.0065 for C3H male skin grafts).
Discussion
Our previous studies showed that allogeneic islets, skin and B cells are susceptible to indirect rejection by CD4 T cells; however, allogeneic T cells were not rejected by this mechanism. 7 Indirect rejection by CD8 T cells has not been extensively examined. We investigated the susceptibility of different tissues to indirect CD8 T cell mediated rejection and the nature of the cells targeted by this pathway. We found that the HY specific monoclonal population of CD8 T cells could indirectly reject male skin grafts; however, male islet and heart grafts were resistant to indirect rejection even in recipients previously primed to HY. Potential reasons for the differential susceptibility include size and location of the graft, origin of the vasculature, the level and type of antigen presentation, or the presence of tissue-specific antigens23–25. Our data suggest that antigen location, tissue specific antigens, and the presence of host vasculature in the graft could not account for the differential susceptibility of non-vascularized transplants to undergo indirect rejection. Instead, properties intrinsic to the tissue may determine the outcome. Skin possesses potent antigen presentation by Langerhans cells or dermal dendritic cells (DCs) and high concentrations of extracellular glycoproteins that may facilitate re-stimulation of T cells.26,27 Antigens from the high numbers of skin-derived APCs, or their exosomes, may be taken in by recipient cells and cross-presented28–30. Skin grafts might also recruit higher numbers of recipient APCs into skin grafts, promoting indirect rejection.
Primed MataHari female T cells were shown to respond in vitro to cultured aortic endothelial cells co-incubated with HY peptide and in vivo experiments showed that skin could only elicit an indirect CD8 response if grafted onto a recipient that expressed the H-2b MHC 5 . It was concluded that MHC class I was required on recipient derived vascular endothelium to cross-present male antigen providing for indirect rejection 5 . However, this conclusion was not directly tested in vivo. The lack of indirect rejection that we observed with both islet and neonatal heart grafts suggests the presence of other mechanisms of indirect CD8 rejection. Furthermore, we found that MHC class I was required on the recipient’s radiosensitive cells for the indirect rejection of skin grafts by primed CD8 T cells. When hematopoietic cells lacked MHC class I, these T cells were unable to reject a C3H male skin graft despite the presence of MHC I on radioresistant cells (e.g. neovascular endothelial cells) and despite being fully competent to reject an MHC-matched male skin graft on the same recipient.
Cross-presentation on MHC class I, in physiologic settings, has been a function largely attributed to specific DC subsets 31 . This fits with the concept that it would be evolutionarily disadvantageous for many other types of cells, such as endothelial cells, to have a strong capacity for cross-presentation. For example, if endothelial cells were to readily cross-present antigens, it is not clear what would prevent lethal organ ischemia when healthy non-infected vascular endothelial cells become targets of our anti-virus cytotoxic T cells during a systemic viral infection.
In summary, we conclude that CD8 T cells kill cells/tissues lacking the appropriate MHC through a bystander mechanism following recognition of cognate peptide-MHC on APCs of bone marrow origin, rather than through antigen presentation by vascular endothelial cells within the graft.
Supplemental Material
sj-docx-1-cll-10.1177_09636897221136149 – Supplemental material for CD8 T Cells Target Antigen Cross-Presented by Bone Marrow Derived Cells to Induce Bystander Rejection of Grafts Lacking the Cognate Peptide-MHC
Supplemental material, sj-docx-1-cll-10.1177_09636897221136149 for CD8 T Cells Target Antigen Cross-Presented by Bone Marrow Derived Cells to Induce Bystander Rejection of Grafts Lacking the Cognate Peptide-MHC by David P. Al-Adra, Govindarajan Thangavelu, Jiaxin Lin, William F.N. Chan, Kristofor K. Ellestad, Louis Boon and Colin C. Anderson in Cell Transplantation
Footnotes
Acknowledgements
We thank Deb Dixon for islet cell transplantation, Mike Bui for pilot studies assessing skin and islet grafts, Catherine Ewen for help with cell sorting, Perveen Anwar for technical assistance, HSLAS staff for animal care, and Peter Bretscher for critical review of our manuscript.
Ethics Approval
This study was approved by the Health Sciences Animal care committee at the University of Alberta (AUP00000215), Edmonton Canada. All care and handling of animals were carried out in accordance with the guidelines of the Canadian Council on Animal Care.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the guidelines of the Canadian Council on Animal Care and was approved by the Health Sciences Animal care committee at the University of Alberta.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research has been supported by operating funds to CCA from the Canadian Institutes of Health Research (PS148588, FRN79521) and awards from the Alberta Heritage Foundation for Medical Research (to DPA and CCA).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
