Abstract
In our daily plastic surgery practice, we have seen many chronic wounds that need new biotechnology to help and improve wound healing. Stem cells play a crucial role in regenerative medicine. Many pre-clinical researches had reported the beneficial paracrine effects of stem cell therapy for chronic wounds. Cell-friendly scaffolds may provide the protection and three-dimensional space required for adherence of stem cells, thus allowing these stem cells to proliferate and differentiate for treatment purpose. A successful scaffold may enhance the effects of stem cell therapy. In this presented series, the authors attempted to identify the most suitable scaffolds from several commercially available wound dressings that could sustain adipose-derived stromal/progenitor cells (ADSCs) survival. Therefore, we isolated ADSCs containing the green fluorescent protein (GFP) from GFP transgenic rats. The GFP (+) ADSCs and their progenies could be easily observed using a fluorescence microscope. Moreover, we analyzed the cytokines secreted in condition medium (CM) to understand the activities of ADSCs in various dressings. Our results showed that the foam dressings, hydrofiber, chitosan, and alginate plus carboxymethylcellulose were identified as the most suitable dressing materials. Higher concentrations of transforming growth factor beta (TGF-β) and vascular endothelial growth factor (VEGF) were observed 48 h after loading them with GFP (+) ADSCs. Therefore, multiple topical cell therapy using ADSCs can be performed by applying suitable dressing scaffolds without repeated needle injections to deliver the stem cells into the wound bed. Based on their fluorescence property, the GFP (+) ADSCs can also possibly be used for testing biocompatibility of medical materials in the future.
Introduction
The current management of chronic wounds includes wound dressing, bedside debridement, infection control, moisture balance, and vascular assessment1,2. Originally, wound dressings were used to protect the wound base. With the advances made in understanding the wound-healing process, several wound dressings have been designed to maintain an optimal amount of moisture and provide an ideal environment for epithelial migration1,2. Numerous types of commercially available wound dressings promote wound healing. Moreover, the selection of dressings is usually based on a physician’s personal experience, availability, wound condition, cost, and patient’s preference 3 .
Zuk et al. 4 first reported the use of adipose-derived stromal/progenitor cells (ADSCs) to improve wound healing with promising results5–9. ADSCs have the capacity of prolonged or unlimited self-renewal and ability to differentiate into multiple lineages 10 . Furthermore, the paracrine effects of the ADSCs regulate the levels of cytokines around the wound site to enhance wound healing11,12. In the clinical setting of cell therapy, simultaneously maintaining the contact between ADSCs and the wound bed and keeping them viable are challenges that require to be solved. However, this involves repeated local injections, which makes using ADSCs in clinical practice difficult 11 . Therefore, ADSC scaffolds have been developed as a reasonable option for chronic wound treatment.
In our earlier studies8,10, we successfully isolated ADSCs from the inguinal fatty tissue of Wistar rats. We reported the use of ADSCs to screen possible scaffold candidates from commercially available materials used during routine surgery 10 . In this study, we explored possible wound dressing materials that can be used as scaffolds to carry ADSCs. We harvested the inguinal fatty tissue from a green fluorescent protein (GFP) transgenic SD rat and cultured ADSCs. These GFP (+) ADSCs can be traced repeatedly under a fluorescence microscope in every generation.
We gathered 12 types of commercially available wound dressing materials in the clinics. The optimal scaffold had to demonstrate a high biocompatibility and be able to enhance the survival of ADSCs. We wanted to identify ADSC-friendly dressings that could serve as ADSC carriers for chronic wound cell therapy in the future.
Materials and Methods
Harvesting Adipose Tissue From GFP Transgenic Rat and Cultivating ADSCs
An SD-Tg (UBC-emGFP) 18Narl transgenic rat was purchased from the Genetic Engineering Murine Model Services, National Laboratory Animal Center (GEMMS-NLAC) in Taipei, Taiwan. The preparation procedures have been described by our colleagues Lin et al.
13
, Lee et al.
14
, and Sheen et al.
10
previously. Briefly, this GFP (+) SD rat was anesthetized using an intraperitoneal injection with Zoletil 50 (50 mg/kg, Virbac Taiwan Co, Ltd, Taipei, Taiwan). Approximately 15 g subcutaneous tissue was excised from the inguinal area bilaterally. The tissue was rinsed using
Adipogenesis Differentiation of ADSCs
Following the induction procedure reported by Lin et al. 13 , these primary cultured cells were treated by adipogenesis supplementations in DMEM4,13. Oil red stain was used to monitor their successful differentiation into adipocytes.
Fluorescence-Activated Cell Sorting Analysis of ADSCs
According to the Concise Review of ADSCs written by Schaffler and Buchler 15 , the surface markers CD90.1, CD44H, CD11b/c, CD45, and CD79α were examined. The ADSCs within the third passage were harvested and fixed in neutralized 2% paraformaldehyde solution for 30 min. These fixed cells were washed twice with PBS and incubated with antibodies against CD90.1, CD44H, CD11b/c, CD45, and CD79α for 30 min. The surface antigens of these cells were identified using flow cytometric analysis.
Seeding the ADSCs Into 12 Types of Wound Dressing Scaffolds and Synthesis of the Condition Medium
The following 12 types of wound dressings were collected in plastic surgery clinics to test their biocompatibility: Acticoat® Dressing (polyethylene mesh), AnsCare® ChitoClot Gauze (chitosan), Aquacel® foam (hydrofiber + foam), Aquacel® (hydrofiber), Aquacel® Ag (hydrofiber), Atrauman-Ag® (polyamide mesh), Biatian® Alginate Dressing [alginate + carboxymethylcellulose (CMC)], Collawound® matrix wound dressing (collagen Sponge), Mepilex Ag® (foam), PELNAC® (collagen sponge), SeaSorb Ag® (alginate + CMC), and Versiva® XC (foam). A 5-mm punch was used to cut the dressing materials and placed in a 96-well nonadhesive tissue culture plate. Keratinocyte-SFM (serum-free medium) was used to moisten these wound dressings; then, 3 × 105 GFP (+) ADSCs were loaded on each dressing material (n = 5) (Figs. 4 and 5). This culture plate was incubated at 37°C for 48 h and then the condition medium (CM) was collected for enzyme-linked immunosorbent assay (ELISA) examination to determine the concentration of three different cytokines.
ELISA for Condition Medium
Three types of cytokines namely platelet-derived growth factor (PDGF), transforming growth factor beta (TGF-β), and vascular endothelial growth factor (VEGF) were selected, as they were involved in wound healing. DuoSet ELISA development kits (R&D Systems, Minneapolis, MN, USA) were purchased. The assay was performed according to the manufacturer’s instructions. PDGF, TGF-β, and VEGF were monitored for 48 h after loading ADSCs. The TGF-β kit required a serum-free cell culture condition.
Statistical Analysis
All data values are presented as the mean values with standard deviations. Five independent experiments were performed for each wound dressing. Data were analyzed using Tukey’s test. Statistical significance was assumed at a P value of <0.05.
Results
Characterization and Verification of GFP (+) ADSCs
The cell morphology of the primary cultured cells from the SD-Tg(UBC-emGFP) 18Narl rat was assessed under a fluorescence microscope (Fig. 1). The results of adipogenesis differentiation with/without oil red stain are illustrated in Fig. 2. The flow cytometry analysis of GFP-ADSCs surface antigen, CD90.1, and CD44H were positive, whereas that of CD11b/c, CD45, and CD79α were negative. The isotype control was also negative (Fig. 3). These data proved that the primary cultured GFP (+) cells had the characteristics of ADSCs. The cumulative population doubling level (CPDL) was performed. At 24, 48, 72, 96, and 120 h, the level of proliferation are 140%, 320%, 660%, 1,450%, and 2,330%, respectively (supporting data).
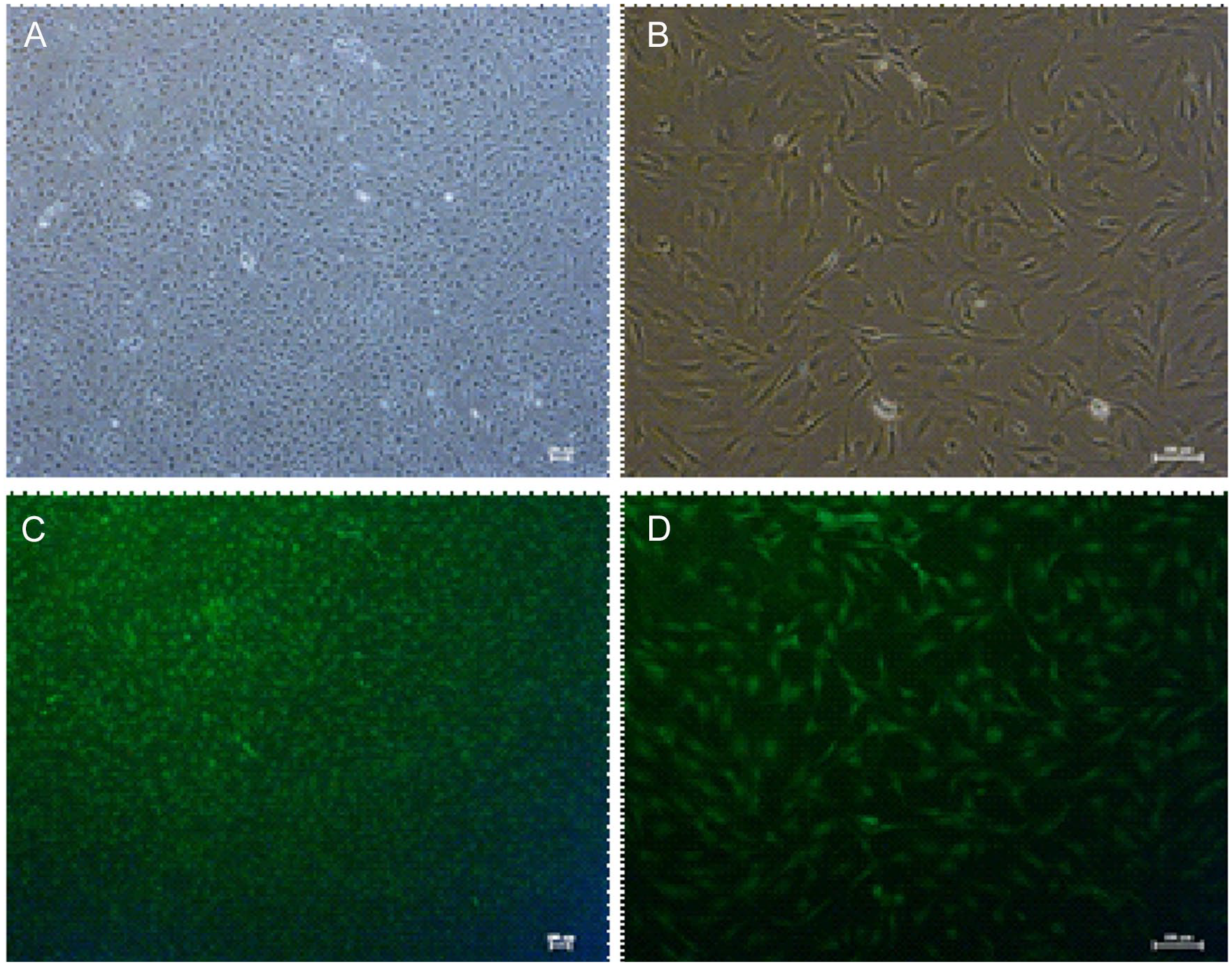
(A) Morphology of green fluorescent protein (+) adipose-derived stromal/progenitor cells (ADSCs) from subcutaneous fat pads cultured in vitro, (B) bright field, (C) fluorescent images of ADSCs, magnification: 40×, and (D) fluorescent images, magnification: 100×.

Multilineage differentiation—adipogenesis, green fluorescent protein (GFP) (+) adipose-derived stromal/progenitor cells (ADSCs) Under Oil Red Stain, (A) GFP (+) ADSCs control, magnification: 100×, (B) GFP (+) ADSCs control, magnification: 200×, (C) GFP (+) ADSCs post induction, magnification: 100×, and (D) GFP (+) ADSCs post induction, magnification: 200×.

Flow cytometry analysis of green fluorescent protein (GFP)-adipose-derived stromal/progenitor cell (ADSC) surface antigens. CD90.1 and CD44H were positive, whereas CD11b/c, CD45, and CD79α were negative, and the isotype control was also negative.
Images of GFP (+) ADSCs Incubated in the 12 Dressing Materials at 48 h
The fluorescence microscope was used to assess these dressing materials. The GFP (+) ADSCs could be seen in some of the images. In some dressing, the ADSCs gathered together to become “spheres” (Figs. 4 and 5). However, these images did not represent the entire amount of ADSCs on the dressing materials. They provided information only on the qualitative analysis.

(A) Green fluorescent protein (GFP) (+) adipose-derived stromal/progenitor cells (ADSCs) incubated in Aquacel foam 48 h, bright field, magnification 100×, (B) fluorescent images of the same view of (A), ADSC spheres noted; (C) GFP (+) ADSCs incubated in Aquacel hydrofiber 48 h, bright field, magnification 100×, and (D) fluorescent images of the same view of (C), ADSC spheres noted.
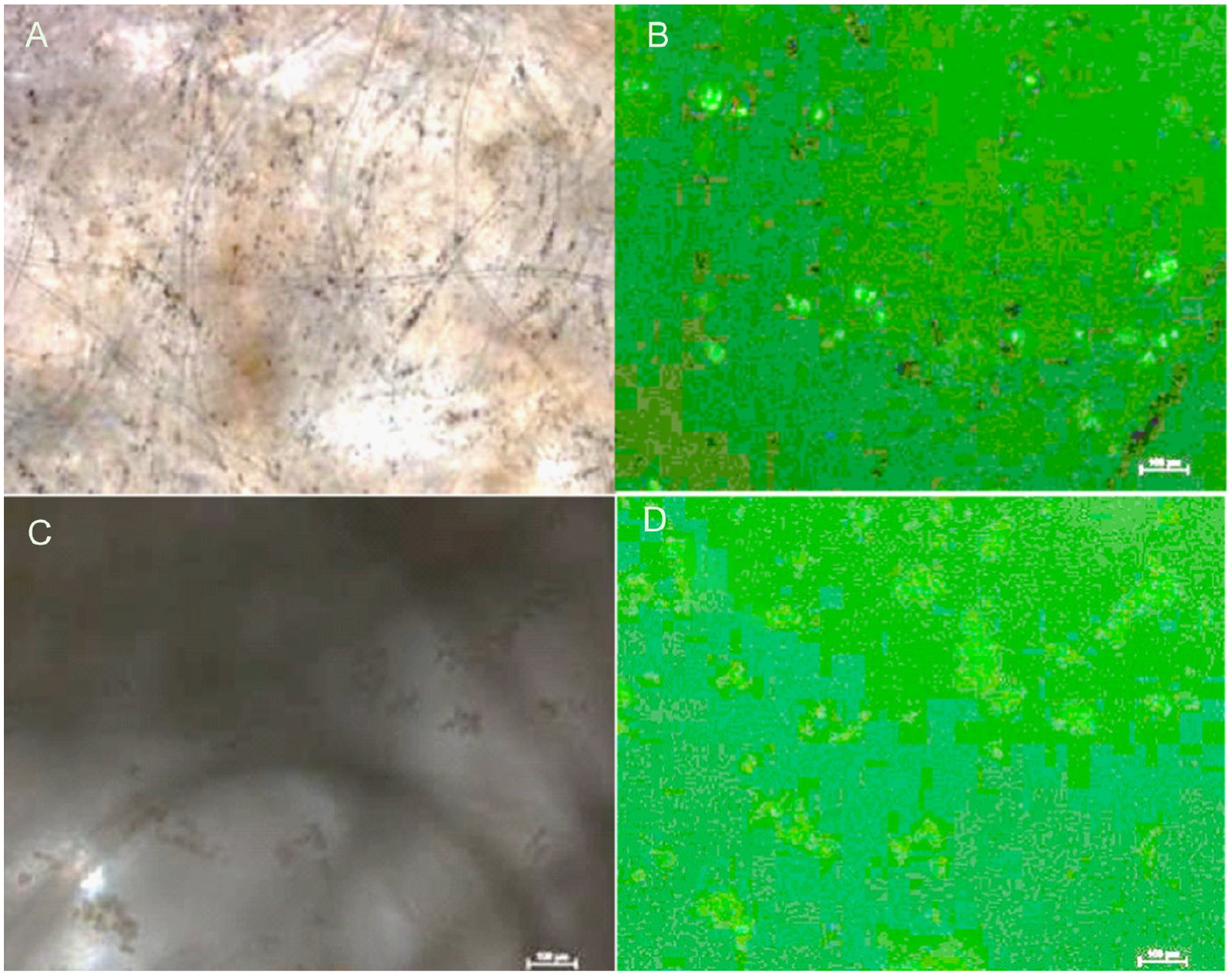
(A) Green fluorescent protein (GFP) (+) adipose-derived stromal/progenitor cells (ADSCs) incubated in Aqucel-Ag hydrofiber 48 h, bright field, magnification 100×, (B) fluorescent images of the same view of (A), ADSC spheres noted; (C) GFP (+) ADSCs incubated in Seasorb Ag 48 h, bright field, magnification 100×, and (D) fluorescent images of the same view of (C), ADSC spheres noted.
Cytokine Concentration in the CM
ADSCs secrete various cytokines. To understand the quantities of ADSCs and their responses when implanted on various scaffolds, the culture media that were used for these GFP (+) ADSCs in daily care were collected. These media were checked for PDGF-αβ, TGF-β1, and VEGF. However, no difference was noted in the concentrations of PDGF-αβ among these CMs (Table 1). Significantly high concentrations of TGF-β1 and VEGF were noted in the foam dressing, chitosan dressing, hydrofiber, and alginate plus CMC groups (Tables 2 and 3).
The Level of PDGF-αβ Secreted From GFP (+) ADSC in Different Medical Materials.

Groups not sharing a same letter (a, b, c, d, e) are significantly different and group sharing (*) are significantly different by Tukey’s test (P < 0.05) (n = 5). ADSC: adipose-derived stromal/progenitor cell; CMC: carboxymethylcellulose; GFP: green fluorescent protein; PDGF: platelet-derived growth factor.
The Level of TGF-β1 Secreted From GFP (+) ADSC in Different Medical Materials.

Groups not sharing a same letter (a, b, c, d) are significantly different and group sharing (*) are significantly different by Tukey’s test (P < 0.05) (n = 5). ADSC: adipose-derived stromal/progenitor cell; CMC: carboxymethylcellulose; GFP: green fluorescent protein; TGF-β: transforming growth factor beta.
The Level of VEGF Secreted From GFP (+) ADSCs in Different Medical Materials.

Groups not sharing a same letter (a, b, c, d) are significantly different and group sharing (*) are significantly different by Tukey’s test (P < 0.05) (n = 5). ADSC: adipose-derived stromal/progenitor cell; CMC: carboxymethylcellulose; GFP: green fluorescent protein; VEGF: vascular endothelial growth factor.
Discussion
The wound-healing process is highly regulated by secretion of various growth factors, cytokines, and chemokines 16 . Chronic wounds require an extended healing period because their endogenous healing mechanisms are disrupted. With technological advancements, administration of exogenous growth factors has shown promise in stimulating chronic wound healing 17 . However, the cost of recombinant human growth factors is high, and proper conservation to maintain their bioactivities is an uncertain issue. Cell therapy has recently gained attention because it provides new hope for patients who have not experienced successful wound healing by conventional treatments 6 . Different cells (fibroblasts, keratinocytes, platelets, ADSCs) have been used to convert the wound bed into an environment where maximum wound healing can be achieved 18 .
ADSCs were evaluated for regenerative application because these cells are easy to isolate, relatively abundant, and multipotent 19 . The stem cells’ therapeutic potential is largely due to their capability to secrete pro-regenerative cytokines, causing them to be an attractive choice for the treatment of chronic wounds 16 . ADSCs can promote wound healing and trigger neovascularization through their ability to differentiate endothelial cells and release VEGF 20 . The ADSCs could differentiate within the epithelium or secrete several types of cytokines (paracrine effect) that stimulated angiogenesis to improve wound healing7,8,19,21. Tremolada et al. 22 reported a novel Lipogems® technology which helped to enhance the healing of lower leg chronic ulceration, especially in diabetic patients. The exosome content has been demonstrated to be significantly greater in fat tissue processed with Lipogems® than with the enzymatic method. The Lipogems® system yields a microfragmented autologous adipose tissue that acts as a large-scale tool to supply damaged tissues with a regenerative environment in a one-step intervention 22 . This method can activate the natural healing process without using enzymes, additives, or separation centrifugation and relying instead on mild mechanical forces 23 . ADSCs are extremely promising as an alternative tool for the regenerative strategy for wound therapy 16 .
Kallmeyer et al. 24 reported local injections of ADSCs for rat wound model cell therapy. Their ADSCs were transduced to express both GFP and firefly luciferase to enable tracking by immune-histological analysis and bioluminescence imaging. They found that these local injected ADSCs could become distributed within the wound bed in 24 h. And these ADSCs were strongly detectable up to 7 days. In our earlier animal studies 8 , the authors had reported ADSC injections for the animal chronic wound model cell therapy. However, merely applying the ADSCs over the wound surface would be less invasive. Topical coverage with ADSCs might make it easier for the institutional review board to approve our human clinical trial in the future. Therefore, identification of suitable scaffolds that could carry as many ADSCs as possible should be explored in future research.
Clinical physicians are not specialized in chemical synthesis and are unable to create new scaffold materials. Commercially available dressings might reduce the concerns of biotoxicity from new chemical synthetic materials. Hence, we tested several commercially available wound dressings to evaluate their potential as ADSC carrier scaffolds and examine their biocompatibility.
In this present series, we harvested the adipose tissue from the GFP (+) transgenic SD rat to isolate GFP (+) ADSCs. Moreover, the surface antigens of our primary cultured cell lines were CD90.1(+) and CD44H(+), whereas CD11b/c, CD45, and CD79α were all negative as observed using flow cytometry analysis 15 . We have proven that our primary culture cell lines possess the characteristics of stem cells10,13. GFP (+) ADSCs were used to serve as a biocompatibility testing tool.
In this study, we identified the most cell-friendly commercial wound dressings and sought potential scaffold candidates that could serve as ADSC carriers during cell therapy for difficult wounds. Because each generation of GFP (+) ADSCs could be observed under a fluorescence microscope, we could trace the positions of GFP (+) ADSCs repeatedly. However, these wound dressings were not transparent. We also detected fluorescence in some of the dressing materials under the fluorescence microscope. Fluorescence microscope observations could be considered only as a “qualitative analysis.” We were unable to identify which dressing material was superior using only fluorescence microscope findings. The ADSCs that survived within the scaffolds would be able to secrete various cytokines. The amount of cytokine secretion is proportional to the survived number of ADSCs. Therefore, the cytokine levels in the CM were studied.
The ADSCs can emanate a variety of cytokines, and several studies have reported on the ADSC-CM12,25–27. The ADSCs promote proliferation of dermal fibroblasts by using not only cell-to-cell contact but also paracrine cytokines 28 . In this series, we collected and analyzed the CMs from 3 × 105 GFP (+) ADSCs in different commercially available dressing materials. We measured the concentrations of PDGF, TGF-β1, and VEGF by using the ELISA technique because these three cytokines were crucial growth factors that might improve wound healing. From the viewpoint of cell therapy, a better scaffold should possess a higher concentration of these cytokines in their CMs. In clinical practice, the chronic wounds shall be inspected and changed dressing daily or every other day to prevent wound infection. The old dressing will be removed and discarded. A new dressing will be applied to the wound. So, in the clinical cell therapy “ADSCs carrying dressing” may cover the wound only for 24 to 48 h. Then, another new ADSCs carrying dressing will replace the old one. Therefore, in this study, we evaluate the cytokine levels from the 48 h of ADSC dressings’ CM.
Our results revealed no significant differences in PDGF levels (Table 1). However, obvious variations were noted in the concentrations of TGF-β1 and VEGF in different dressing materials (Tables 2 and 3). In general, dressing materials with nanosilver particles were less cell-friendly in comparison with dressings without these particles. Moreover, the dressings with high concentrations of TGF-β1 (>30 pg/ml) were Versiva® XC (foam), Aquacel® foam (hydrofiber + foam), and AnsCare® (chitosan). Also, Versiva® XC (foam), Aquacel® foam (hydrofiber +foam), Aquacel® (hydrofiber), AnsCare® (chitosan), and SeaSorb Ag® (alginate + CMC) demonstrated high concentrations of VEGF (>100 pg/ml).
Limitation
Our study has several limitations. First, the GFP (+) cells can only be seen in the transparent scaffold. The non-transparency and some unknown fluorescent material in the dressings make our observation difficult. Second, the ADSCs may survive on the dressing longer than 48 h. However, this study design did not investigate the pattern or growth of ADSCs on the dressing longer than 48 h. Further research may be needed to study the longevity of ADSCs on these dressing. Third, in clinical situation, some ADSCs may translocate to the wound bed. This study did not provide information about this issue. Further analyses and investigations are suggested.
Conclusion
In summary, we successfully isolated GFP (+) ADSCs from the GFP (+) transgenic SD rat. The adipogenesis differentiation and sorting of surface antigen cells proved that these primary cultured cell lines had the characteristics of stem cells. The GFP (+) ADSCs were then used to test 12 types of commercially available wound dressings. We identified three types of dressings with TGF-β1 concentrations of >30 pg/ml in the CM and five types of dressings with VEGF concentrations of >100 pg/ml in the CM. VEGF and TGF-β1 are growth factors vital for wound healing. This study demonstrated that we can select appropriate scaffold dressings for autologous ADSCs seeding to perform cell therapy in human patients with chronic wounds. Moreover, based on their fluorescence property, the GFP (+) ADSCs can possibly be used for testing biocompatibility of medical materials in the future.
Research Data
sj-jpg-2-cll-10.1177_09636897221113798 – Supplemental material for Foraging for the Optimal Dressing Scaffold to Carry Adipose-Derived Stromal/Progenitor Cells for Cell Therapy
Supplemental material, sj-jpg-2-cll-10.1177_09636897221113798 for Foraging for the Optimal Dressing Scaffold to Carry Adipose-Derived Stromal/Progenitor Cells for Cell Therapy by Chia-Chieh Wu, Ying-Che Chen, Yi-Chia Wu, Shu-Hung Huang, Yur-Ren Kuo and Su-Shin Lee in Cell Transplantation
Supplemental Material
sj-pptx-1-cll-10.1177_09636897221113798 – Supplemental material for Foraging for the Optimal Dressing Scaffold to Carry Adipose-Derived Stromal/Progenitor Cells for Cell Therapy
Supplemental material, sj-pptx-1-cll-10.1177_09636897221113798 for Foraging for the Optimal Dressing Scaffold to Carry Adipose-Derived Stromal/Progenitor Cells for Cell Therapy by Chia-Chieh Wu, Ying-Che Chen, Yi-Chia Wu, Shu-Hung Huang, Yur-Ren Kuo and Su-Shin Lee in Cell Transplantation
Footnotes
Acknowledgements
The authors would like to thank the Genetic Engineering Murine Model Services (GEMMS), National Core Facility Program for Biotechnology, Ministry of Science and Technology for providing the SD-Tg (UBC-emGFP) 18Narl transgenic rat used in this study.
Ethical Approval
This study had been approved by the Institutional Animal Care and Use Committee (IACUC) of Kaohsiung Medical University, Kaohsiung, Taiwan.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care and Use Committee guidelines of Kaohsiung Medical University, Kaohsiung, Taiwan.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from Changhua Christian Hospital-Kaohsiung Medical University Research Funding (10-CCH-KMU-011 and KMUH101-1R22) and Kaohsiung Municipal Siaogang Hospital (KMSH P-110-12 and KMSH H-109-06).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
