Abstract
In this multicenter phase II study, we evaluated the safety and efficacy of imatinib in patients with steroid-resistant chronic graft-versus-host disease (cGVHD) and evaluated the quality of life (QOL) of the enrolled patients using the Short Form 36 (SF-36) health survey questionnaire. Thirty-six patients who were diagnosed with steroid-refractory cGVHD and treated with imatinib between March 2013 and February 2019 received 100 mg/day of imatinib for 2 weeks. Depending on the patient’s condition and investigator’s decision, the imatinib dose was allowed to be increased by 100 mg every 2 weeks up to 400 mg/day. Patients who achieved stable disease (SD), partial remission (PR), and complete remission (CR) at 3-month response evaluations continued imatinib for up to 6 months. The majority of the patients had multi-organ cGVHD, with skin (63.9%), lungs (44.4%), mouth (38.9%), and eyes (38.9%) as the most common sites. The overall response rate was 58.3%, including 3 and 18 patients with CR and PR, respectively, and an overall decline in National Institutes of Health (NIH) severity scores was observed at study completion in the absence of significant adverse effects. The overall response rates were 70.5%, 66.7%, 34.8%, and 25% in patients with gastrointestinal, liver, skin, and lung cGVHD, respectively. Factors representing emotional well-being were significantly improved based on the patient-reported QOL evaluation using SF-36. The effect of imatinib on steroid tapering, which was notable in responders, was also present in 50% of those who achieved SD without worsening cGVHD. Imatinib exhibited therapeutic efficacy in steroid-refractory and steroid-dependent cGVHD with tolerable toxicity.
Clinical Trial Registration: KCT0006785.
Keywords
Introduction
Allogeneic stem cell transplantation (allo-SCT) is the most definitive modality to improve survival outcomes and maintain long-term remission in patients with hematological malignancies1,2. However, graft-versus-host disease (GVHD), in which T and B lymphocytes derived from the graft attack the host, remains a vital hurdle in achieving successful transplantation3–5. In particular, chronic GVHD (cGVHD) occurs in approximately 30%–70% of patients undergoing allo-SCT and is the most important cause of nonrelapse morbidity after allo-SCT6–9.
cGVHD occurs as a result of a break in immune tolerance to self-antigens involving a complex network of molecular events, including thymic dysfunction, deficiency of regulatory T cell, and auto-antibody production by aberrant B cells 10 . Treatment of cGVHD depends on the affected organ and severity. The National Institutes of Health (NIH) Consensus Conference and the European Society for Blood and Marrow Transplantation consensus guidelines recommend topical or systemic steroids as first-line treatment11,12. However, more than 50% of patients require second-line therapy, and standard therapies are not available for steroid-resistant patients13,14. Moreover, morbidity and mortality are higher among patients with steroid-refractory cGVHD 15 .
cGVHD is characterized by fibrosis in multiple organs that manifests as a range of clinical features, including debilitating tissue injury and higher risk of life-threatening events 16 . Platelet-derived growth factor (PDGF) and transforming growth factor β (TGF-β), which are involved in inflammatory and fibrogenic processes, play an important role in the pathogenesis of cGVHD17,18. Anti-TGF-β antibodies were shown to suppress the development of skin fibrosis in a murine model of cGVHD, whereas blockade of the PDGF and TGF-β signaling pathways reduced the development of skin fibrosis18–20. Imatinib, a dual inhibitor of the PDGF and TGF-β signaling pathways, strongly represses intracellular PDGF and TGF-β signaling in vitro 21 . An Italian transplantation group reported that imatinib was effective in 19 patients with refractory cGVHD 22 . The 6-month overall response rate (ORR) was 79%, including seven and eight patients achieving complete remission (CR) and partial remission (PR), respectively, demonstrating imatinib as a promising agent for refractory fibrotic cGVHD. Magro et al. 23 also reported an ORR rate of 50% in 14 patients with sclerodermatous cGVHD treated with imatinib, demonstrating its efficacy. In contrast, Stadler et al. 24 reported that imatinib exhibited limited efficacy in severe, refractory lung cGVHD. Less favorable results with imatinib were also reported in patients with sclerodermatous cGVHD, with an overall improvement rate of only 30% in a French study including 39 patients with cGVHD25,26.
The efficacy of imatinib in cGVHD is debatable. Therefore, we conducted a multicenter phase II study to evaluate the efficacy and safety of imatinib in patients with steroid-resistant cGVHD based on the previous data. In addition, we assessed patient-reported quality of life (QOL) using a Short Form health survey questionnaire administered during the study period.
Methods
Definition of Steroid-Refractory cGVHD
cGVHD was diagnosed and graded based on the NIH consensus criteria 8 . Treatment of cGVHD was conducted according to the experience and competence of the treating clinician. Patients with moderate or severe cGVHD received prednisone with or without calcineurin inhibitors. Steroid-refractory cGVHD was defined as (1) worsening cGVHD even with the use of 1 mg/kg/day prednisone for 2 weeks, (2) cGVHD that was stable or not improving with the use of ≥0.5 mg/kg/day prednisone for 4–8 weeks, and (3) failure to reduce prednisone dose below 0.5 mg/kg/day 27 .
Patient Enrollment
Thirty-six patients from the 13 institutes who fulfilled the following criteria were enrolled in this study, which was conducted between 2014 and 2019: (1) age ≥20 years, (2) Eastern Cooperative Oncology Group performance status score ≤2, (3) cGVHD that meets the NIH diagnostic criteria after allo-SCT, (4) moderate or severe cGVHD based on the NIH global scoring system, (5) steroid-refractory cGVHD, and (6) resistance to calcineurin inhibitors such as cyclosporine and tacrolimus 8 . Patients with liver GVHD alone, those with the progression of underlying disease, and those who underwent donor lymphocyte infusion were excluded.
Assessment of Treatment Response
Response to treatment was evaluated based on the NIH consensus criteria for cGVHD by scoring each involved organ site using a 4-point scale 28 . CR was defined as the resolution of all reversible manifestations of cGVHD. PR of cGVHD was defined as a clinical score reduction of at least 1 point in one or more of the affected organs. ORR was defined as the sum of CR and PR. Disease progression or treatment failure was defined as a clinical score increase of at least 1 point in one or more organs or the development of new symptoms or signs of cGVHD. Patients not meeting the CR, PR, or disease progression are considered stable disease (SD).
Imatinib Treatment
Enrolled patients received 100 mg/day imatinib for 2 weeks. The imatinib dose was allowed to increase by 100 mg every 2 weeks up to 400 mg/day depending on the patient’s condition and the investigator’s decision. Patients who achieved CR, PR, or SD in the 3-month response evaluation continued treatment with imatinib for up to 6 months. Treatment response was evaluated every 2 weeks for 6 months according to the NIH global scoring system. Survival outcomes of the enrolled patients were evaluated for 3 years.
Assessment of Patient-Reported QOL
Patient-reported health-related QOL was evaluated using Short Form 36 (SF-36) version 2 (1996, 2004, 2012 Medical Outcomes Trust and QualityMetric Incorporated), which includes 36 items that measure the following eight scales of QOL: physical functioning, role physical, bodily pain, general health, vitality, social functioning, role emotional, and mental health. Two summary scales of the SF-36 questionnaire show a physical dimension represented by the physical component score (PCS) and a mental dimension represented by the mental component score (MCS) (https://www.qualitymetric.com/health-surveys-old/the-sf-36v2-health-survey/).
Statistical Analysis
Categorical variables were reported as numbers with proportions, and continuous variables were reported as medians with ranges. Overall survival (OS) was calculated from the initiation of imatinib treatment until death due to any cause or the date of last follow-up. Event-free survival (EFS) was calculated from the initiation of imatinib treatment until death, treatment failure, progression of the underlying hematologic malignancy, or the date of last follow-up. The Kaplan–Meier method was used to analyze OS and EFS, and survival curves were compared using the log-rank test. Patient-reported QOL by SF-36 was assessed before the initiation of the study and after the end of the study. The changes of each quantified subscale were analyzed using Wilcoxon rank-sum test. All data were analyzed using the R statistical software program version 3.6.2 (R Foundation for Statistical Computing, Vienna, Austria; available at http://www.r-project.org).
Results
Patient Characteristics
The baseline patient characteristics are summarized in Table 1. The current study included 36 patients with a median age of 47.5 years (range, 23–63 years), and 17 patients (47.2%) were male. All patients received peripheral blood stem cells, including 25 (69.4%), 8 (22.2%), and 3 (8.3%) patients receiving from matched sibling, matched unrelated, and haploidentical donors, respectively. In addition, 88.9% of the patients were administered myeloablative conditioning regimen. Thirty-one patients (86.1%) experienced acute GVHD (aGVHD) and received steroids as first-line aGVHD treatment. aGVHD was not completely resolved in 11 patients (35.5%), and 6 patients (19.4%) were dependent or refractory to steroids. The majority of the patients had multi-organ cGVHD, and skin (63.9%), lungs (44.4%), mouth (38.9%), and eyes (38.9%) were the most commonly involved sites. Moderate and severe cGVHD developed in 20 (55.6%) and 16 (44.4%) patients, respectively, according to the NIH global severity score. Twenty-five patients (69.4%) had been treated with ≥0.5 mg/kg/day steroids at the time of study enrollment. Most patients were also treated with calcineurin inhibitors (88.9%) or mycophenolate mofetil (19.4%) due to uncontrolled cGVHD.
Patient Characteristics.

allo-SCT: allogeneic stem cell transplantation; CR: complete remission; RAEB: refractory anemia with excess blasts; SAA: severe aplastic anemia; CML: chronic myeloid leukemia; BuFlu: busulfan plus fludarabine; BuCy: busulfan plus cyclophosphamide; GVHD: graft-versus-host disease; GI: gastrointestinal; NIH: National Institutes of Health.
Response
After 3 months of imatinib treatment, 1 (2.8%), 18 (50%), 15 (41.7%), and 2 (5.6%) patients achieved CR, PR, SD, and PD, respectively. Two patients discontinued imatinib treatment due to the progression of GHVD, one patient withdrew from the study due to imatinib intolerance, and one patient experienced progression of the underlying disease. The remaining 32 patients continued the study. Following three more months of imatinib treatment, one patient with CR maintained their status and two patients with PR improved their status to CR. In addition, 18 (56.3%), 10 (31.3%), and 1 (3.1%) patient had PR, SD, and PD, respectively. Overall, 3, 18, and 10 patients achieved CR, PR, and SD, respectively, whereas 3 patients experienced cGVHD progression and 2 discontinued the study because of withdrawal of consent and progression of underlying disease. The ORR was 58.3% (21/36). At study completion, there was an overall decline in NIH severity scores except for eye involvement. In addition, complete resolution of cGVHD was reported in all involved organs except for the eyes and joints (Table 2). The efficacy of imatinib was better in liver and gastrointestinal cGVHD than in skin and lung cGVHD. CR was achieved in 66.7% (2/3), 42.9% (3/7), 17.4% (4/23), and 12.5% (2/16) of the patients with liver, gastrointestinal, skin, and lung cGVHD, respectively (Fig. 1). The ORR of patients with gastrointestinal and liver cGVHD was 70.5% and 66.7%, respectively, whereas the ORR of those with skin and lung cGVHD was 34.8% and 25%, respectively (Table 2).
Changes in the NIH Severity Score After Imatinib Treatment.

NIH: National Institutes of Health; CR: complete response; PR: partial response; ORR: overall response rate; GI: gastrointestinal.

cGVHD severity scores before and after imatinib treatment. GI: gastrointestinal; cGVHD: chronic graft-versus-host disease.
Steroid Reduction Effect
Systemic steroid treatment could be discontinued in 8 of the 32 patients who completed the study. In addition, steroid treatment was tapered off without worsening cGVHD in 14 patients. Importantly, 5 of the 10 patients who achieved SD (50%) experienced the steroid-reducing benefit of imatinib treatment, despite the lack of improvement in cGVHD (Fig. 2).

Steroid-tapering effect of imatinib therapy.
Imatinib Dose and Response
Target imatinib dose (400 mg/day) was achieved in seven patients (19.4%). Twenty (55.6%) patients increased imatinib to 300 mg/day, and six (16.7%) patients received 200 mg/day of imatinib. The other three (8.3%) patients continued 100 mg/day of imatinib. Median dose of 300 mg/day of imatinib was administered during the study. However, there was no statistically significant difference in the imatinib dose between the imatinib responders and nonresponders (Supplementary Table 1).
Safety
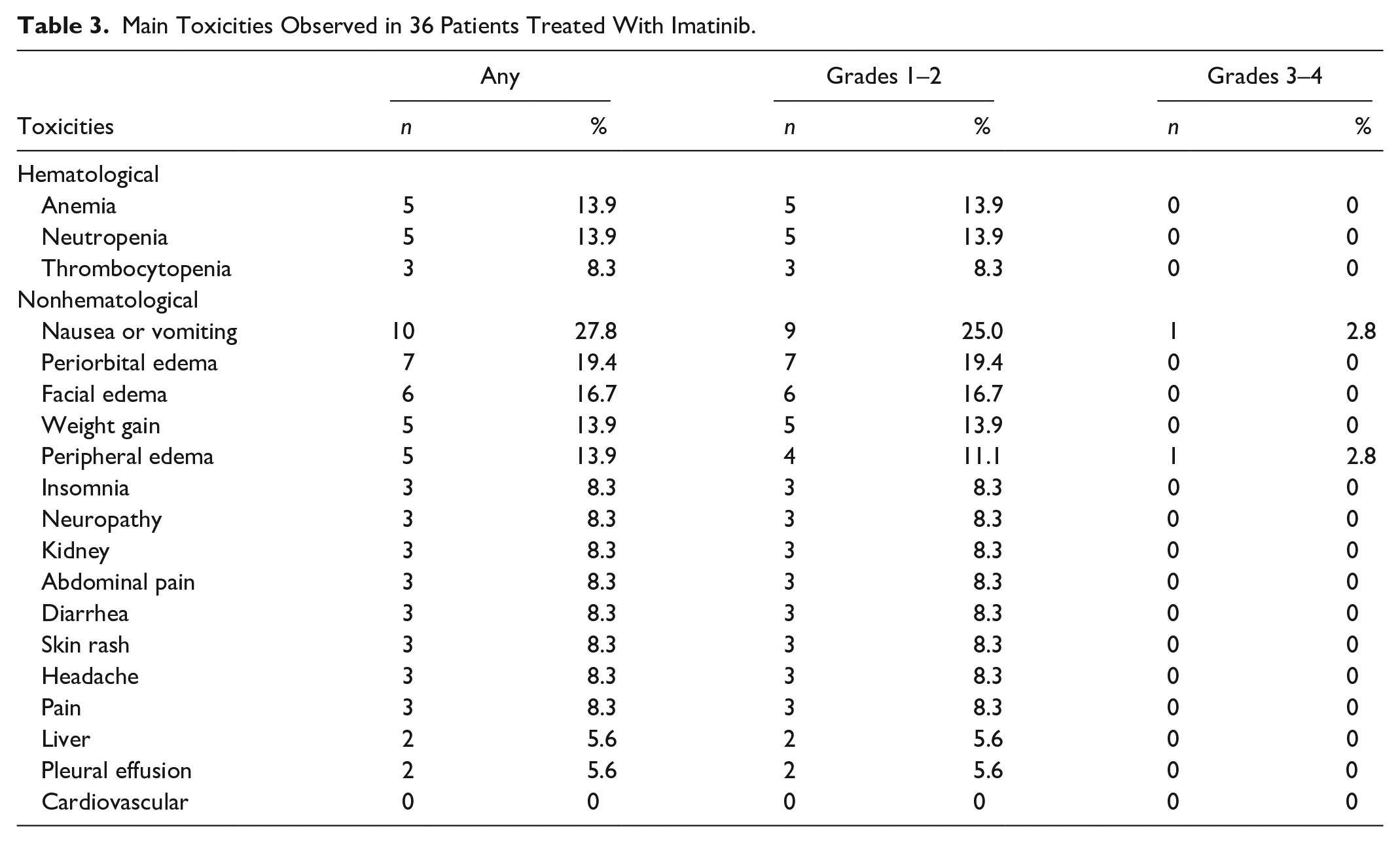
One patient withdrew from the study due to severe nausea, whereas imatinib-related fatal events did not occur during the study period. The details on hematological and non-hematological toxicities are summarized in Table 3. Hematological toxicities included grade 1/2 mild anemia, neutropenia, and thrombocytopenia. Nausea/vomiting, periorbital edema, facial edema, weight gain, and peripheral edema were common nonhematological adverse events. No severe cardiovascular, liver, and kidney dysfunction were reported. Severe toxicities leading to withdrawal from the study were rare. Nausea/vomiting and edema were the main causes for failure to reach the target imatinib dose.
Main Toxicities Observed in 36 Patients Treated With Imatinib.
Patient-Reported QOL
The SF-36 questionnaire was completed by 29 patients (80.6%). The scoring used in the SF-36 questionnaire is described in Supplementary Table 2. Overall, all QOL domains improved after the imatinib treatment, as shown in Fig. 3. Particularly, factors representing emotional well-being (role emotional, mental health, and MCS) were significantly improved. However, there was no statistical difference in the current patient-reported QOL questionnaires between the imatinib responders and nonresponders (data not shown).
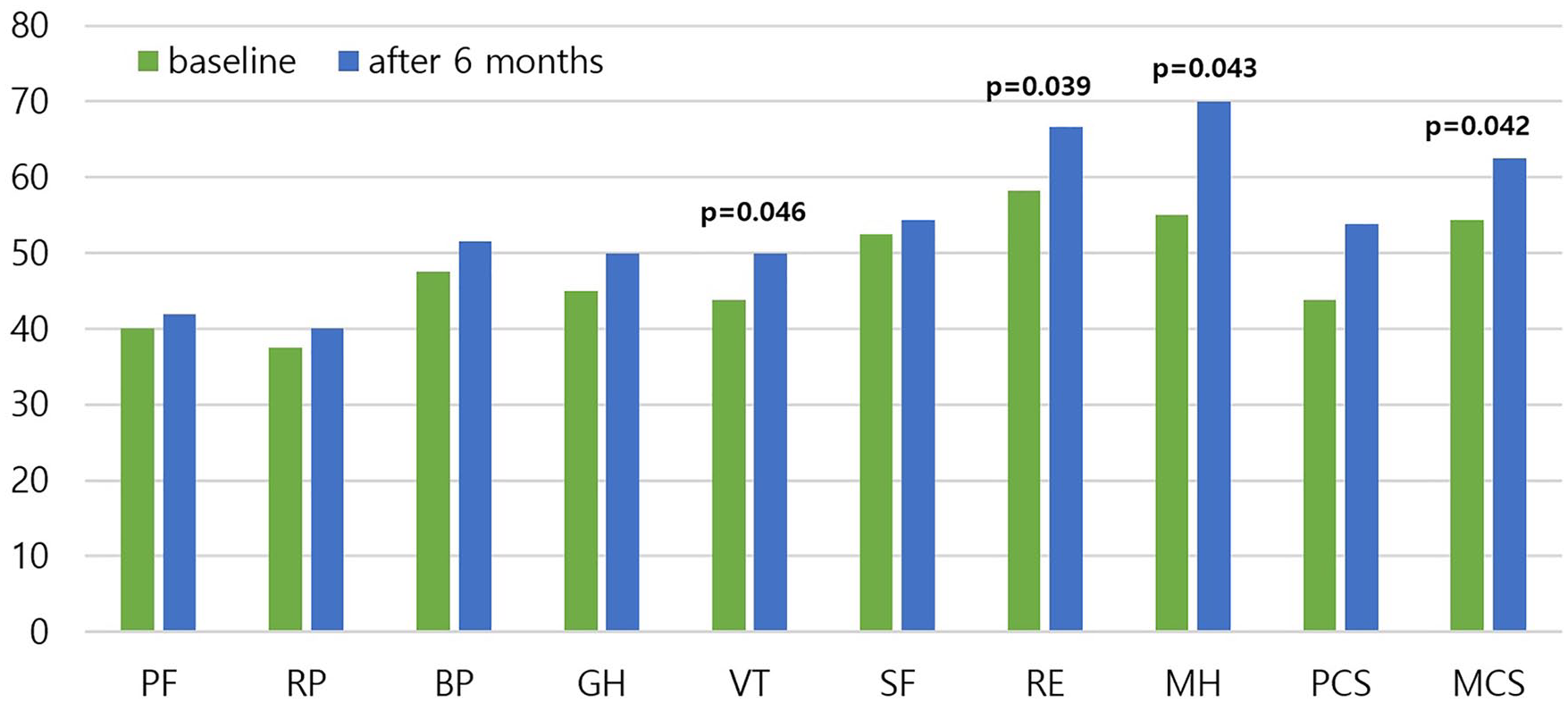
Quality of life domain scores according to the patient-reported SF-36 questionnaire after imatinib treatment. SF-36: Short Form 36; PCS: physical component score; MCS: mental component score; PF: physical functioning; RP: role-functioning physical; BP: body pain; GH: general health; VT: vitality; SF: social functioning; RE: role emotional; MH: mental health.
Long-term Outcomes
The median follow-up duration was 37 months (range, 4–54 months). Overall, 28 of the 36 patients were alive at the last follow-up. Five patients who did not respond to imatinib treatment, including two patients with SD and three patients with PD, died due to the progression of lung (n = 3) and skin (n = 2) cGVHD. Two patients with PR died due to pneumonia, whereas one patient died from relapsed acute leukemia. The 3-year EFS and OS at 3 years were 65.6% and 76.7%, respectively (Supplementary Figs. 1, 2). The 6-month EFS was significantly better among imatinib responders (ie, those who achieved PR or CR) than imatinib nonresponders (P = 0.007). However, there was no statistical difference in the OS between the responders and nonresponders in the current study (Fig. 4).

Survival outcomes. The imatinib responders (partial remission/complete remission) at 6 months exhibited superior event-free survival compared with imatinib nonresponders (A), whereas there was no statistical difference in overall survival between the imatinib responders and nonresponders (B). EFS: event-free survival; OS: overall survival.
Discussion
Severe cGVHD requires long-term immunosuppressive treatment. Although steroids have been used as first-line treatment of cGVHD affecting numerous organs, more than half of the patients with cGVHD remain refractory to or dependent on steroids. As alternatives, numerous candidate approaches, including small-molecule inhibitors, antibodies, and cellular therapies, have been tested in clinical trials 3 . In the current prospective trial including 36 patients with steroid-refractory severe cGVHD, 32 patients completed 6 months of treatment with imatinib. The median imatinib dose was 300 mg/day, and severe adverse events were not observed. After 6 months of imatinib treatment, 21 of the 36 patients exhibited partial or complete improvement in cGVHD. The EFS was significantly better in imatinib responders than in imatinib nonresponders. Moreover, the effect of imatinib on steroid tapering, which was notable in imatinib responders, was also present in 50% of those who achieved SD without worsening cGVHD.
In addition to leukemia, imatinib also targets and inhibits the PDGFR and TGF-β pathways, which play pivotal roles in fibrosis that occurs in cGVHD. Several studies have already investigated the efficacy of this first-generation tyrosine kinase inhibitor (TKI) for the treatment of refractory cGVHD, mainly focusing on skin and lung involvement17,29. However, Olivieri et al. 5 were the first to report that imatinib was effective against all types of GVHD, including that with visceral involvement, based on the analysis of long-term outcomes of 39 patients with steroid-refractory cGVHD receiving imatinib. In a recent study including the hematopoietic transplantation data of 66 patients with steroid-refractory cGVHD from Spain, skin, gastrointestinal tract, and liver were more responsive to imatinib treatment compared with the lungs and eyes 30 . Similarly, the current study evaluating imatinib treatment for severe cGVHD involving the liver and gastrointestinal tract shows promising results. Among the patients with liver and gastrointestinal cGVHD, the CR rates were 66.7% and 42.9% and the ORRs were 66.7% and 71.4%, respectively; these rates were significantly higher than those observed among the patients with skin and lung involvement. Increased fibrosis and loss of bile ducts are characteristic histopathologic features of long-standing liver GVHD 31 . In addition, significant fibrosis and the distortion of mucosal architecture are the main histologic changes in gastrointestinal cGVHD 32 . Numerous studies have already demonstrated the involvement of the PDGF and TGF-β signaling pathways in liver and intestinal fibrosis; therefore, further investigation is warranted to elucidate the specific role of TKIs on fibrosis observed in liver and gastrointestinal cGVHD associated with severe symptoms33–35.
Although high-dose systemic steroids remain a major therapeutic approach for moderate and severe cGVHD, long-term use of steroids is associated with a wide range of adverse events including infections15,36,37. The addition of calcineurin inhibitors or other immunosuppressants may allow the reduction in steroid dose; however, whether this approach is effective for long-term outcomes remains unclear38,39. Previous studies have already proposed that imatinib therapy might allow the discontinuation or significant dose reduction of steroids5,22. Our analyses also revealed that imatinib had a beneficial effect on steroid tapering and dose reduction even in patients with SD. Moreover, the role of emotional and mental health scales and the MCS was significantly improved compared with the functional scales based on the patient-reported QOL questionnaire. The improvement in cGVHD symptoms and the reduction in steroid dose might have contributed to the observed improvement in the emotional well-being of the study patients. Given that the QOL is a central concern of transplant survivors, these results are meaningful and clearly demonstrate the benefits of imatinib treatment.
Patient-reported QOL is as important as clinician-reported disease severity in assessing therapeutic response to cGVHD; therefore, numerous studies aimed to measure the QOL of patients with refractory cGVHD. Some studies suggested that patient-reported symptoms and QOL were associated with the severity of clinician-reported cGVHD27,40–42. Unlike the current study results, Rosenthal et al. 16 demonstrated that reducing symptom burden by imatinib treatment was strongly correlated with improved PCS but not with the emotional well-being based on the SF-36 questionnaire. However, further investigation with larger cohorts and sufficient statistical power is necessary to identify consistent patterns of patient-reported QOL who receive salvage therapy for unresolved cGVHD.
The rate of imatinib response varies from 17% to 79% in patients with refractory cGVHD, whereas a statistically significant association between daily imatinib dose and therapeutic effect has not yet been established5,22–26,30. A study with low-dose daily imatinib (50–100 mg) showed some effect for refractory cGVHD with fibrotic features 22 . Furthermore, Olivieri et al. 5 showed that a mean dose of 270 mg/day of imatinib was administered and most patients received 200 mg/day of imatinib in a study for steroid-refractory cGVHD, although the dosage of imatinib was planned to increase to 400 mg/day. We also aimed to determine the appropriate imatinib dose in the current study. The target imatinib dose of 400 mg/day was achieved in only seven patients. Increasing the imatinib dose was difficult in many patients because of nausea, vomiting, and edema. However, the mean imatinib dose did not differ between the imatinib responders and nonresponders, in agreement with previous studies2,5,22,30,43. Therefore, careful selection of patients who might obtain clinical benefit from imatinib therapy should be considered. Studies investigating pretreatment anti-PDGF receptor alpha subunit (anti-PDGFRA) antibody level as a predictive marker to identify such patients reported that anti-PDGFR activity was significantly associated with response to imatinib therapy in patients with cGVHD5,22. Moreover, patients with high pretreatment levels of anti-PDGFRA antibodies showed improved risk/benefit ratio to TKI treatment in a recent study 44 . In addition, second-generation TKIs such as dasatinib might be considered as an alternative approach to target the PDGF and TGF-β pathways for cGVHD management 45 .
Although our data showed the promising result of the imatinib in patients with steroid-refractory cGVHD, ibrutinib is currently the only US Food and Drug Administration (FDA)-approved agent for steroid-refractory cGVHD with 67% ORR. In addition, belumosudil was also approved by the FDA for the treatment of cGVHD after failure of two or more lines of systemic therapy in 20217,46–48. Belumosudil therapy reported 76% of ORR with 5% of CR rates in all organs including pulmonary cGVHD 48 . Ruxolitinib which was FDA-approved for aGVHD reported 43% to 85% of ORR in the retrospective studies with steroid-reducing effect49,50. Other agents including rituximab have shown benefits, but it is difficult to select an agent for steroid-refractory cGVHD because of the insufficient evidence3,51.
In conclusion, imatinib exhibited therapeutic efficacy with tolerable toxicity in patients with steroid-refractory and steroid-dependent cGVHD, who also exhibited improved emotional well-being. Besides skin and lung cGVHD, those with gastrointestinal and liver cGVHD might potentially benefit from daily low-dose imatinib, and the steroid dose might be reduced in patients treated with imatinib.
Supplemental Material
sj-docx-1-cll-10.1177_09636897221113789 – Supplemental material for Results of Multicenter Phase II Study With Imatinib Mesylate in Allogeneic Recipients With Steroid-Refractory Chronic GVHD
Supplemental material, sj-docx-1-cll-10.1177_09636897221113789 for Results of Multicenter Phase II Study With Imatinib Mesylate in Allogeneic Recipients With Steroid-Refractory Chronic GVHD by Dong Won Baek, Hee Jeong Cho, Ju-Hyung Kim, Jae Sook Ahn, Hyeoung-Joon Kim, Sung Nam Lim, Jun Won Cheong, Sung-Yong Kim, Ho Sup Lee, Jong Ho Won, Ho-Young Yhim, Sang Kyun Sohn and Joon Ho Moon in Cell Transplantation
Footnotes
Authorship
D.W.B. collected and interpreted the data and drafted the manuscript. H.J.C., J.H.K., J.S.A., H.J.K., S.N.L., J.W.C., S.Y.K., H.S.L., J.H.W., and H.Y.Y. contributed to patient enrollment, data collection, and revision of the manuscript. S.K.S. and J.H.M. contributed to the study design and data interpretation and revised the manuscript critically for important intellectual content. All authors provided final approval of the version to be submitted.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author.
Ethical Approval
This study was approved by the Institutional Review Board of Kyungpook National University Hospital (KNUH 2013-08-021) and each other participating center.
Statement of Human and Animal Rights
All procedures in this study that involved human participants were performed in accordance with the Institutional Review Board of Kyungpook National University Hospital (KNUH 2013-08-021) and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Statement of Informed Consent
Written informed consent was obtained from all patients for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
