Abstract
Chronic graft-versus-host disease (cGVHD) is a potentially life-threatening complication after allogeneic hematopoietic stem cell transplantation. Standard steroid first-line treatment could not satisfy therapeutic needs due to limited efficacy. As a highly selective Janus kinase (JAK) 1 inhibitor, SHR0302 exhibits a reduced inhibition effect on JAK2 and might have less effect on hematopoiesis. This phase I clinical trial investigated the tolerability and safety of SHR0302 in combination with prednisone, and its early efficacy evidence as a potential first-line treatment to moderate/severe cGVHD. The standard 3 + 3 dose escalation was implemented to find the optimal dose of SHR0302. And prednisone was concurrently administrated with a dose of 1 mg/kg/d and then gradually tapered after 2 weeks. Eighteen patients were enrolled into the study. Grade ≥ 3 treatment-related adverse events were observed in 38.9% of patients. Only one patient developed DLT (grade ≥ 3 hypercholesterolemia) in the highest dose-level group who had pre-existing hypercholesterolemia. The maximum tolerated dose was not reached. No patient discontinued treatment due to AEs. Sixteen out of 18 patients were evaluable for responses, the ORR at week 4 and week 24 were 94.4 and 87.5%, respectively. Overall, the treatment of SHR0302 combined with prednisone was safe and well-tolerated, preliminary clinical results presented a high response for previously untreated cGVHD and a significant reduction in prednisone use in this study. A phase II trial will be conducted to further investigate its therapeutic effects clinically.
Introduction
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is a potentially curative treatment option for a variety of hematological diseases. However, chronic graft-versus-host disease (cGVHD) remains the main cause of late morbidity and mortality after allo-HSCT 1 . About 30% to 70% of allo-HSCT recipients suffer from cGVHD 2 and have experienced decreased quality of life and even death3,4.
For decades, steroids with or without calcineurin inhibitors have undoubtedly been the first-line treatment for cGVHD. However, approximately 30% to 60% of patients have to receive subsequent lines of therapy due to the limited efficacy of first-line treatment5,6. Moreover, the long-term usage of steroids inevitably brings about various disorders, such as diabetes, osteoporosis, severe infections, and so on. Several randomized trials have evaluated the efficacy of adding other agents, such as azathioprine 7 , thalidomide 8 , mycophenolate mofetil 9 , or hydroxychloroquine 10 for previously untreated cGVHD, the results fail to exhibit any clinically significant benefit. Still, an unmet need remains for the first-line treatment of cGVHD.
In recent years, the knowledge of the pathophysiology of cGVHD has gradually improved. Chronic GVHD develops via a complex cellular and molecular network involving thymic damage and aberrant antigen presentation, leading to aberrant T and B cell activation and differentiation, alloantibody formation, and tissue fibrosis 11 . Kinases about pathogenesis of GVHD, such as Rho-associated coiled-coil-containing kinase 2 (ROCK2) 12 , Janus kinase 1/2 (JAK1/2) 13 , and Bruton’s tyrosine kinase (BTK) 14 have been well investigated. Several small molecule inhibitors targeting these kinases have exhibited improved efficacy in animal models and clinical trials15,16. Three agents, ruxolitinib, ibrutinib, and belumosudil have been approved by the U.S. Food and Drug Administration (FDA) for the treatment of cGVHD after failure of first-line and/or second-line therapies 17 . Notably, ruxolitinib has been widely used in both acute graft-versus-host disease (aGVHD) and cGVHD based on the results of the REACH1, REACH2, and REACH3 trials18–20.
Preclinical studies showed that JAK1/2 signaling played a crucial role in the inflammation and tissue impairment in both aGVHD and cGVHD 15 . Blocking the JAK/STAT pathway can potentially inhibit the pathological and physiological process of GVHD21,22. Oral SHR0302 exhibited good tolerance and efficacy in Chinese adult patients with moderate to severe atopic dermatitis 23 . Treatment with SHR0302 at 4 and 8 mg doses in patients with moderate or severe alopecia areata for 24 weeks was found efficacious and generally well tolerated 24 . Preliminary results have reported good and fast efficacy, as well as an acceptable safety profile of the investigational SHR0302 in psoriatic arthritis 25 . In addition, our previous study demonstrated that SHR0302 could effectively prevent and reverse aGVHD and preserve graft-versus-tumor effect with less impact on hematopoietic recovery in mouse model 22 . Therefore, considering the safety and initial efficacy of SHR0302 in the treatment of chronic inflammation-related diseases, we choose to use SHR0302 to treat chronic GVHD to improve the survival of patients and their quality of life.
On the contrary, due to an increased risk of cytopenia, especially thrombocytopenia, the most common adverse event associated with ruxolitinib significantly hinders its clinical application for the treatment of cGVHD 20 . Given the crucial role of JAK2 signaling in normal hematopoiesis26,27, we hypothesized whether a highly selective JAK1 inhibitor could potentially block pathways contributing to cGVHD development while preserving JAK2 function to reduce the incidence of cytopenia. Our preclinical study has indicated that SHR0302 can inhibit JAK1-mediated interleukin (IL)-6, interferon (IFN)-γ, and other cytokine pathways related to the pathological occurrence of aGVHD 22 . Its high selectivity on JAK1 but reduced inhibitive effect on JAK2, are potentially helpful to minimize the risks of neutropenia and anemia 22 .
In this study, we reported the results of SHR0302 combined with prednisone for patients with previously untreated moderate or severe cGVHD and assessed the safety and initial efficacy. This phase I trial was registered at www.clinicaltrials.gov as #NCT04146207.
Material and Methods
Patients
Patients who underwent allo-HSCT for hematological diseases were enrolled in the study. The inclusion criteria were as follows: (1) 18≤ age ≤70 years old; (2) being diagnosed with moderate or severe cGVHD according to the National Institutes of Health (NIH) consensus criteria 28 ; (3) no previous systemic treatment including extracorporeal photochemotherapy (ECP); (4) Karnofsky Performance Status >60% or Eastern Cooperative Oncology Group performance status (ECOG PS) 0 to 2; (5) patients who had received other prophylactic immunosuppressants or treatment for aGVHD were eligible; (6) other JAK inhibitors such as ruxolitinib for aGVHD must be discontinued for at least 4 weeks before the first dose of SHR0302; (7) no recurrent primary disease; (8) the willingness and ability to participate. The exclusion criteria were as follows: (1) pregnant or lactating women; (2) absolute neutrophil count (ANC) <1.0 × 109/L or platelet count <50 × 109/L; (3) uncontrolled active infections or active hepatitis B, active hepatitis C requiring antiviral therapy; (4) Human immunodeficiency virus positive; and (5) overlap syndrome.
Study Design
The study was an open, phase I clinical trial with a sample size of 15 to 30. The primary objective was to evaluate the safety of SHR0302 in combination with prednisone in the first-line treatment of moderate or severe cGVHD, and the secondary objective was to evaluate its efficacy. The sample sizes for the dose-escalation phase were based on a standard “3 + 3” design 29 . Standard 3 + 3 dose escalation was performed to determine the optimal single dose of SHR0302, which corresponded to five dose levels of 1, 2, 4, 6, and 8 mg orally once a day. For each patient, prednisone (or equivalent dose of methylprednisolone) was administered at a fixed starting dose of 1 mg/kg/d. The dose of prednisone was tapered off after at least 2 weeks of treatment as clinically indicated (Supplemental Table 1) 30 . SHR0302 will be administered for at least 6 months unless cGVHD progression, initiation of another systemic cGVHD treatment, unacceptable toxicity, relapse of underlying disease, or discontinuation for any other reason. The primary endpoint was safety and tolerability. The secondary endpoints included overall response rate (ORR; complete [CR] or partial response [PR]) at day 28 and week 24, proportion of patients with prednisone discontinuation, event-free survival (EFS; survival without cGVHD progression, relapse of underlying disease, or start of subsequent cGVHD therapy), and overall survival (OS).
The study protocol was approved by the Ethics Committees of Shanghai General Hospital (Approval ID: 2019-110). All enrolled patients provided written informed consent. The study was carried out in accordance with the principles of the Declaration of Helsinki and the International Conference on Harmonization Good Clinical Practice guidelines.
Assessments and Definitions
The safety and tolerability were assessed by the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) v5.0. Dose-limiting toxicity (DLT) was defined as all hematological toxicity of grade 4 or above and nonhematological toxicity of grade 3 or above that occurred within 28 days after the first application of SHR0302. The maximum tolerated dose (MTD) was defined as the dose level at which more than 1/3 of patients experienced DLTs during the observation period. The efficacy was assessed according to the 2014 National Institutes of Health (NIH) Consensus28,31, including CR (resolution of all manifestations in each organ or site), PR (improvement in at least one organ or site without progression in any other organ or site), mixed response (MR, improvement in at least one organ accompanied by progression in another organ), progression disease (PD, worsening of at least one organ and no improvement in any other organ), and unchanged (outcomes that do not meet the criteria for CR, PR, PD or MR). Lack of response was defined as no CR or PR. Study visits occurred at baseline and every 2 weeks through week 12, every 4 weeks from week 13 through 1 year after treatment.
Statistical Analysis
The DLT set included all patients who received at least 4 weeks of treatment or discontinued study treatment due to intolerable AEs within 4 weeks following treatment initiation. Safety and efficacy were analyzed in all patients who received at least one dose of the study drug. Demographic and clinical characteristics were presented as counts and percentages for the categorical variables; medians and ranges were provided for the continuous variables. ORR was calculated with a 95% Clopper–Pearson confidence interval (CI). Survival analysis was performed according to the Kaplan–Meier method.
Results
Patients Baseline Characteristics
From April 2020 to July 2022, 18 patients were enrolled in the trial. Up to May 2023, the median follow-up period was 20.7 months (95% CI: 17.1–24.2). Currently, there are still four patients undergoing SHR0302 treatment in the study. The baseline characteristics of these patients were summarized in Table 1 and Supplemental Table 2. The median age of the patients was 47 years (range, 31–64). The median time from allo-HSCT to cGVHD diagnosis was 6.6 months (range, 4.0–18.0). Eleven (61.1%) patients had severe cGVHD, and the other 7 had moderate cGVHD. At baseline, the most common sites of involvement in the 18 evaluable patients, including responders and nonresponders, were skin (66.7%) and mouth (50.0%); and lung (5.6%) and genital tract (5.6%) involvement also appeared [Table 1 and Supplemental Table 2). Eight patients were enrolled along with immunosuppressive agents for GVHD prophylaxis, of whom 7 received cyclosporine A (CsA) and one received tacrolimus (TAC). Six out of the eight patients left off their immunosuppressive agents during the study.
Patient Baseline Characteristics.

Safety
At the time of data cutoff, the median duration for SHR0302 was 6.3 months (range, 1.1–20.3), and for SHR0302-prednisone was 4.5 months (range, 1.1–20.3). Overall, all patients experienced treatment-related adverse events (TRAEs), and grade 3 or 4 TRAEs were observed in 7 (38.9%) patients (Table 2). The most common TRAEs included hypercholesterolemia (61.1%, n = 11), hypertriglyceridemia (50.0%, n = 9), platelet count decreased (38.9%, n = 7), and anemia (16.7%, n = 3). After a median of 3 weeks of treatment (range, 0–9 weeks), 6 patients experienced infections such as urinary tract infection, skin infection, pneumonia, viral infection, Epstein–Barr Virus reactivation, and hepatitis B reactivation, of which 3 were from the 2-mg group and one each from the 1, 4, and 8-mg groups, respectively. Four cases of infections were of grade 1–2, while the other two were of grade 3. No patient developed cytomegalovirus (CMV) reactivation. One patient (No. 15) experienced grade 1 hepatitis B virus reactivation during a follow-up of 6 to 12 months and had been taking Tenofovir alafenamide Fumarate tablets for antiviral treatment. And one patient (No. 3) developed grade 2 hyperglycemia and recovered with oral antidiabetic drugs. There were no serious adverse effects (SAEs) related to SHR0302, and no patient experienced SHR0302 dose reduction/discontinuation or death due to AEs. Only one patient developed DLT (grade ≥3 hypercholesterolemia) in the highest dose-level group who had pre-existing hypercholesterolemia. The MTD was not reached.
Treatment-Related Adverse Events.
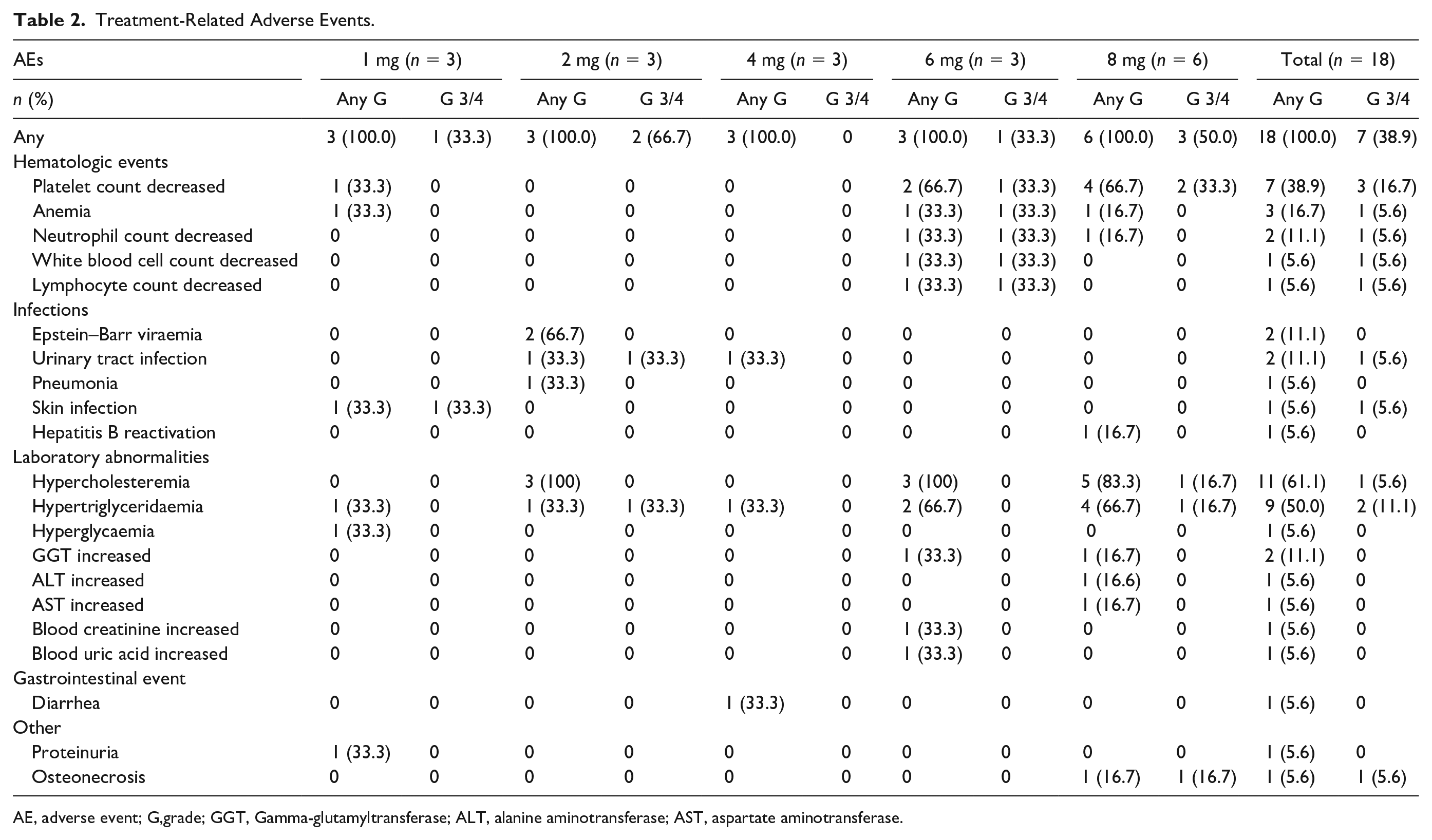
AE, adverse event; G,grade; GGT, Gamma-glutamyltransferase; ALT, alanine aminotransferase; AST, aspartate aminotransferase.
At doses of 1 to 4 mg, hematologic toxicity was mild, and only one patient experienced grade 1 platelet count decreased and anemia. Although hematologic toxicities were increased in dose levels of 6 and 8 with six patients exhibiting thrombocytopenia, only one patient in dose level 6 and two in dose level 8 experienced grade 3 thrombocytopenia. Notably, patients with grade 3 thrombocytopenia recovered rapidly after thrombopoietic growth factor treatment without platelet transfusion or SHR0302 dose reduction. No patient developed grade 4 hematologic toxicities and experienced any dose reduction of SHR0302 at any dose level.
In addition, throughout the clinical study, we observed varying degrees of hypercholesteremia and hypertriglyceridaemia, among which the proportion of such AEs occurring in the 6- and 8-mg dose groups was higher than that in the 1- to 4-mg dose group. However, only one patient experienced grade 3 or 4 TRAEs, and symptomatic lipid-lowering treatment was given to alleviate and control the symptoms. Simultaneously, grade 1–2 abnormal liver enzyme indicators (including aspartate aminotransferase [AST], alanine aminotransferase [ALT], etc.) were also found in the 6- and 8-mg dose group. These aforementioned findings should be more or less related to abnormal lipid metabolism, which was consistent with previous researches about the influence of JAK inhibitors on lipid metabolism in some clinical trials32–34.
Efficacy
During the brief 4-week treatment period, one patient (No. 1) did not respond (Fig. 1A) and belonged to the SHR0302 1-mg group with 73.9 kg body weight (BW). Meanwhile, another patient (No.3) in the 1-mg group with 90 kg BW experienced slight progression of mouth symptoms after achieving an early transient response. As the patient in 1-mg group who achieved CR had a relatively lower BW of 60 kg, indicating that the initial SHR0302 dose of 1-mg was too low for adult patients. The proportion of moderate or severe cGVHD patients significantly decreased to 16.7% (3/18) after 4-week treatment (Fig. 1B), and of the 18 evaluable patients, 17 patients (94.4%, 95% CI: 0.727–0.999) had an objective response, including 5 (27.8%) with CR and 12 (66.7%) with PR.
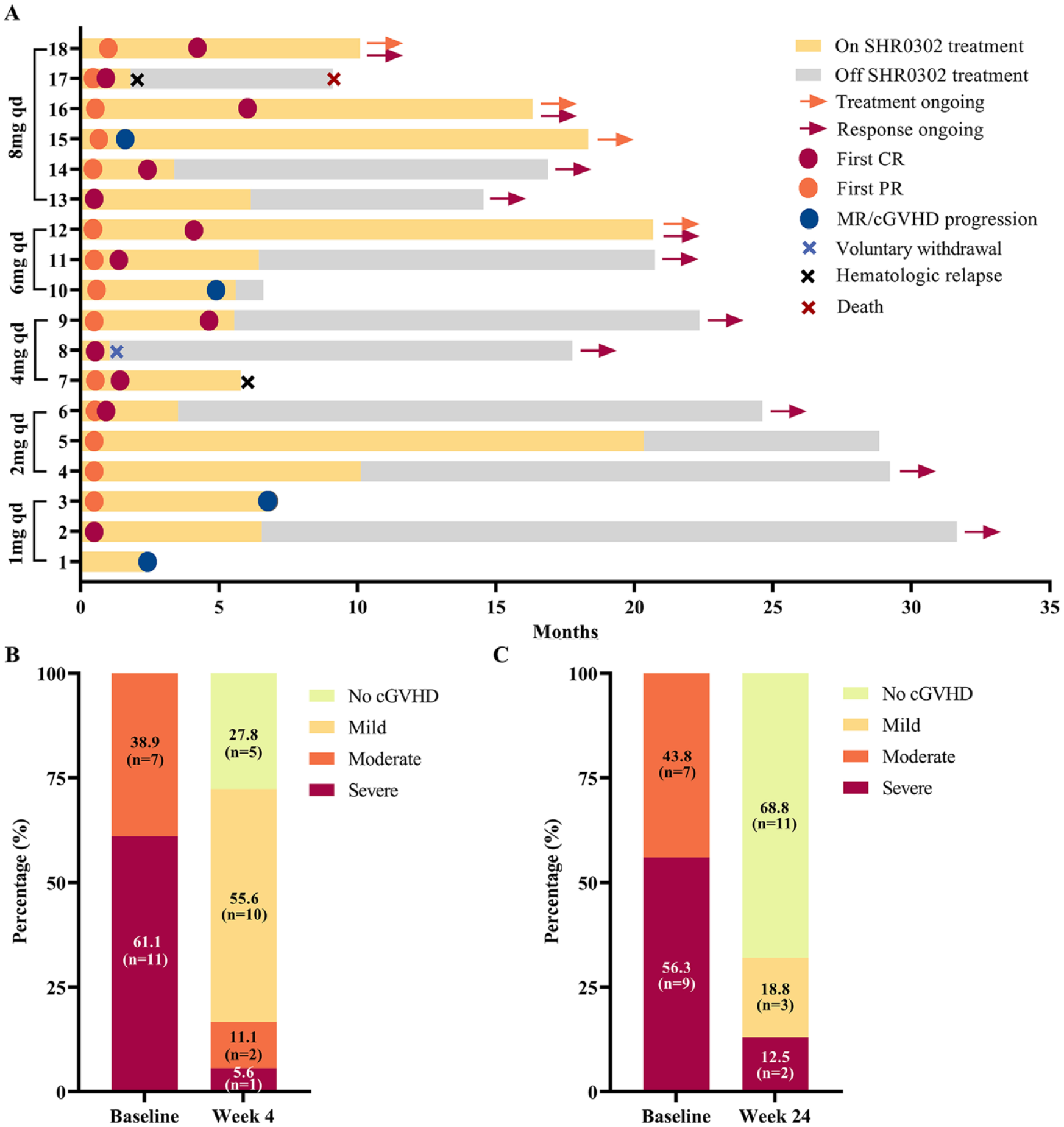
Duration of response, overall cGVHD score at week 4 and week 24. (A) Swimming plot of SHR0302 treatment exposure, duration, and response; #08 voluntarily withdrew from the study and did not receive subsequent treatment or experience disease progression at the data cut-off date; #10 and #15 had a decrease in % FEV1 after improvement in other involved organs and were defined as MR; (B) The proportions of patients with different grade cGVHD at baseline and 4 weeks after SHR0302 (n = 18); (C) The proportions of patients with different grade cGVHD at baseline and 24 weeks after SHR0302 (n = 16). Two patients were excluded from the 24-week efficacy analysis: patient No.17 had a primary disease relapse at 6 weeks after initiation of SHR0302, and patient No.1 had joint progression at week 12 and received methotrexate as a subsequent treatment.
Then, at the 24-week efficacy evaluation, two patients were excluded: patient No.17 had a primary disease relapse at 6 weeks after initiation of SHR0302, and patient No.1 had joint progression at week 12 and received methotrexate as a subsequent treatment (Fig. 1A). And only two (No. 10 and 15) of 16 evaluable patients were assessed with severe cGVHD at 24 weeks after the first dose administration of SHR0302 (Fig. 1C). These two patients had a decrease in % FEV1 after improvement in other involved organs and were defined as MR at 21 and 6 weeks after treatment, respectively (Fig. 1A). Patient No.10 dropped out of this study at week 24 and received ruxolitinib 1 month later, while patient No.15 remained on SHR0302 treatment without progression. Besides, almost all involved organs were controlled or alleviated. Thus, 14 (87.5%, 95% CI: 0.617–0.984) out of the 16 evaluable patients achieved objective response, including 11 (68.8%) with CR and 3 (18.8%) with PR. Moreover, the median follow-up duration was 20.7 months (95% CI: 17.1–24.2), during which only one patient experienced mortality attributed to relapse of the primary hematological disorder. And, the median survival has not yet been reached.
Meanwhile, as shown in Fig. 2 and Supplemental Table 3, these majority of involved organs responded quickly to the combination therapy and their site-specific scores decreased to zero at 4 weeks after treatment, including skin (n = 4), mouth (n = 3), liver (n = 3), gastrointestinal (GI) tract (n = 2), and genital tract (n = 1). However, the resolution of symptoms pertaining to eyes and lungs was slow. Notably, the results of organ-specific response at week 24 were encouraging. It seemed that the CR of the combination treatment was durable in the mouth (n = 8), skin (n = 6), GI tract (n = 4), liver (n = 3), joints (n = 3), eyes (n = 1), and genital tract (n = 1).
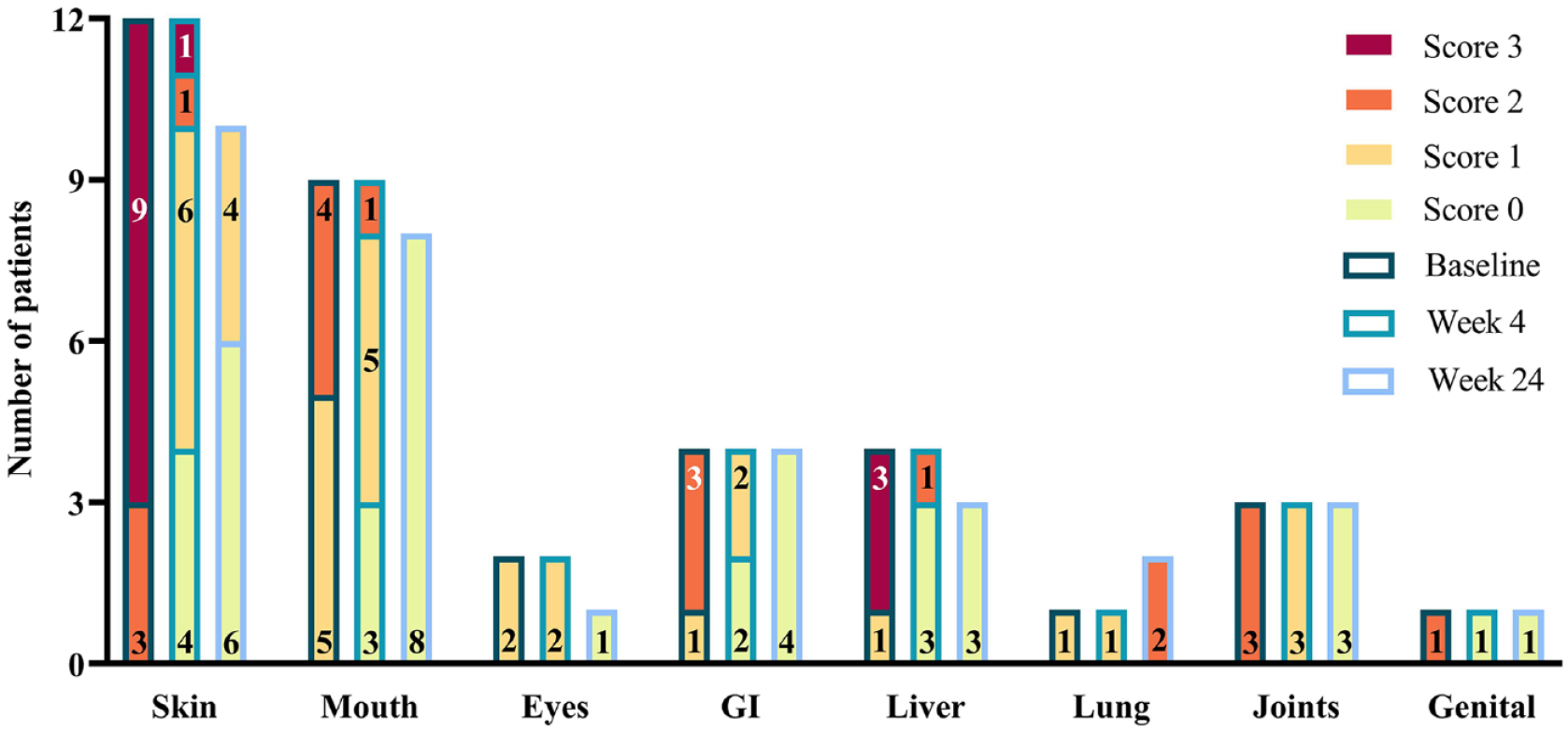
NIH 0–3 score changes of each involved organs over time. Patient #1 and #17 were excluded from the 24-week efficacy analysis. Consequently, the numbers of skin, mouth, eyes, and liver organs evaluated at week 24 were lower compared to that at the baseline.
Dosage of Prednisone
Prednisone dose reduction from baseline to week 24 was shown in Fig. 3. Prednisone was administrated at an initial dose of 1 mg/kg/day and then tapered according to the study protocol (Supplemental Table 1). The median prednisone dose was 0.60 (range: 0.41–0.87) mg/kg/day at week 4, 0.2 (range: 0–0.6) mg/kg/day at week 12, and 0 (range: 0–0.2) mg/kg/day at week 24. Furthermore, prednisone was discontinued in 8 patients at week 24 and all immunosuppressant drugs, including SHR0302, were discontinued in 4 patients with CR before the 24-week assessment. Therefore, there was a reduction in prednisone usage after the combined treatment with SHR0302.
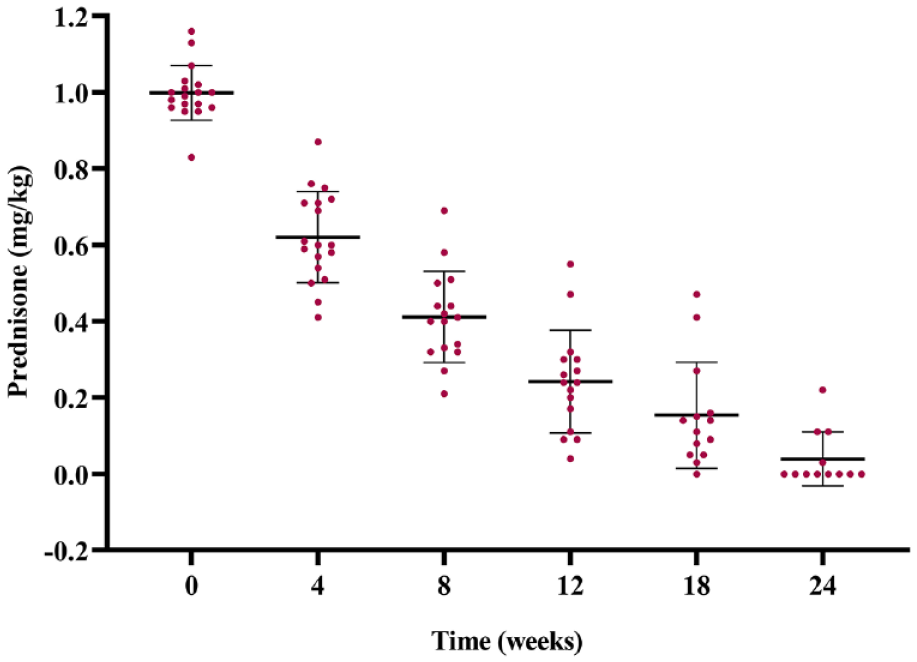
Prednisone dose reduction from baseline to week 24.
Discussion
The cGVHD remains a significant complication following allo-HSCT and is the primary cause of late post-allo-HSCT treatment-related mortality and morbidity 35 . Despite numerous clinical trials investigating various steroid-sparing agents, only limited trials have demonstrated clinical efficacy, resulting in unfavorable outcomes for patients with steroid-refractory cGVHD (SR-cGVHD) 36 . The pathophysiology of cGVHD is intricate, involving both T-cell and B-cell-mediated immune responses, and certain cytokines including IL-6, IL-13, and IFN-γ, have been implicated in the pathogenesis of cGVHD 11 . JAK, a nonreceptor tyrosine kinase, plays a crucial role as a signal transducer for these cytokines 37 . Given the secretion and release of cytokines are critical for the initiation and maintenance of cGVHD, the blockade of their secretions has been evaluated as a means of preventing and potentially treating GVHD in preclinical models 38 .
In one clinical trial, SHR0302 tablet has been safely used in patients of atopic dermatitis for dose-ranging phase II study, with ongoing efficacy and improvement in quality-of-life metrics 23 . In a preclinical study, SHR0302 showed promise in preventing aGVHD 22 . In this study, we evaluated the safety and efficacy of oral SHR0302 in patients with cGVHD. Over 60% TRAEs were grade 1–2 and got ameliorated without additional clinical interventions, and the incidence of grade ≥3 TRAEs was only 38.9%. SHR0302 was overall well-tolerated, though all enrolled patients experienced AEs associated with the SHR0302 combination regimen. Hematopoietic toxicities like anemia and thrombocytopenia, were quite common for patients of allo-HSCT receiving treatment with JAK inhibitors like ruxolitinib 18 . In this study, three patients in high dose level of 6 and 8-mg also experienced grade 3 thrombocytopenia, but no patient developed grade 4 hematopoietic toxicity. All hematologic toxicity recovered rapidly after thrombopoietic growth factor treatment, without platelet transfusion or SHR0302 dose reduction. Considering that SHR0302 preclinical data brings out rapid hematopoietic recovery in aGVHD mouse model, these clinical results further indicate that SHR0302 has mild hematologic toxicity, which may be ascribed to its highly selective activity against JAK1 rather than to JAK2 22 .
In addition, in this study, abnormal lipid metabolism was one of the most common AEs. Although 11/18 patients developed hypercholesterolemia and 8 developed hypertriglyceridemia, the only one patient who developed DLT (grade ≥3 hypercholesterolemia) was in the highest dose group with a high baseline cholesterol level. The DLT was well managed after antilipidemic therapy and recovered rapidly within 2 weeks. Such observations coincide with other research findings investigating the influence of JAK inhibitors on lipid metabolism in clinical trials of ruxocitinib 32 , baricitinib, 33 and TQ05105 34 . In principle, a host of cytokines and hormones can regulate lipid metabolism by activating the JAK-STAT signaling pathway 39 . And activated STATs mediated by JAK1 can regulate lipid metabolism directly by influencing the expression of enzymes 40 .
Besides, infection occurred in 6 patients (6/18, 33.3%), of which 2 had grade 3 infection episodes and 4 had grade 1 events during the follow-up period. The majority of infections developed at taking a high dose of prednisone concurrently. In addition, only one patient experienced grade 1 hepatitis B virus reactivation during a follow-up of 6 to 12 months. Therefore, the high dose of prednisone might be the primary factor contributing to such infection events. As in other studies, the conventional first-line prednisone-calmodulin inhibitor (CNI) therapy reported in the BMT CTN 0801 study also observed grade 3 infections 5 , in which 39.1% of patients experienced severe to life-threatening/fatal infection episodes. More recently, the combination of itacitinib (JAK1 inhibitor) and steroids for new-set cGVHD resulted in a relatively high incidence of ≥grade 3 infection 41 .
The data from this study showed that SHR0302 tablet in combination with prednisone had a promising efficacy in first-line treatment of cGVHD. The response rates in our study were encouraging; SHR0302 seemed to be more effective in alleviating cGVHD symptoms in targeted organs, especially the skin, mouth, eyes, GI tract, joints, and liver. Previous studies suggested that JAK1/2 signaling played a crucial role in the inflammation and tissue impairment in both aGVHD and cGVHD 42 . JAK/STAT were described as a family of rapid membrane to nucleus signaling molecules that act downstream of over 50 cytokines 43 . SHR0302 is a selective JAK1 inhibitor that effectively blocks the effects of cytokine storms. The main clinical symptoms of the skin and other mucosal areas are cytokines induced inflammatory reactions, and SHR0302 may lead to a faster response to symptom improvement by inhibiting the JAK1 pathway. Evidence also suggests that organs derived from the same embryologic origin, like ectodermal skin, eyes, and oral mucosa share some common cGVHD pathways44,45. Therefore, mucosal effector sites appear to respond particularly well to this agent, while further clinical validation is needed. In addition, lung symptoms did not improve at week 4 post treatment, which are mainly caused by long-term inflammatory reactions leading to fibrosis, resulting in irreversible clinical symptoms and poor drug response. These data indicate, like other JAK inhibitors, cGVHD of lung is insensitive to SHR030236,46,47. Due to the limited sample size in this study, lung cGVHD symptoms will be closed monitored in phase II trial in the future.
Moreover, the use of SHR0302 in cGVHD in this study has resulted in decreased prednisone use. Within only 4 weeks after starting combination treatment, the median dose of prednisone decreased to 0.60 mg/kg/day, corresponding to a 40% reduction from its initial dose of 1.0 mg/kg/day. In contrast, the dose tapering took up to 12 months to decrease from the mean baseline dose (0.9 mg/kg) to the reduced mean dose (0.7 mg/kg) in another study using corticosteroid alone therapy 5 . The reduced total dose of prednisone and shorter dose tapering time might decrease the morbidity associated with corticosteroid usage in the long run 36 . Therefore, considering the safety and effectiveness of SHR0302 that we have observed in Chinese patients with moderate to severe cGVHD, immunomodulation via blocking some cytokine pathways with SHR0302 represents a novel and promising approach for the treatment of cGVHD. Yet, one DLT was reported in the 8-mg dose group (n = 6), 6 and 8 mg were selected for dose expansion study to determine the recommended phase 2 dose (RP2D). The researcher has initiated the dose expansion study, the findings will be disclosed later.
Although the clinical trial results seemed encouraging, this study had several limitations. First, this study was conducted at a single center with a small sample size and a relatively short follow-up. Simultaneously, as cGVHD is a heterogeneous disease, the entire course of moderate to severe cGVHD may not have been included in this trial study, so the 18 patients cannot fully represent the full spectrum of moderate to severe cGVHD. Second, the contributing effects of glucocorticoid agents cannot be disregarded. And the diagnosis of cGVHD was according to clinical manifestations without pathological evidence. Moreover, due to the COVID-19 pandemic, three patients lacked lymphocyte test data after 28 days of treatment, and the other 15 patients also lacked data on immune cell types at some time points. Thus, based on limited data, meaningful clinical significance could not be analyzed. Therefore, we are going to conduct a randomized controlled clinical trial with a larger sample size to further clarify the therapeutic effects and safety of SHR0302 on cGVHD.
Conclusion
In summary, the treatment of SHR0302 combined with prednisone was safe and well-tolerated. Although this study was conducted at a single center with a small sample size and may not include the entire course of moderate to severe cGVHD, preliminary clinical results also presented a high response for previously untreated cGVHD and a significant reduction in prednisone use in this study. A phase II trial will be conducted to investigate further clinically.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241254678 – Supplemental material for A Novel JAK1 Inhibitor SHR0302 Combined With Prednisone for First-Line Treatment of Chronic Graft-Versus-Host Disease: A Phase I Clinical Trial
Supplemental material, sj-docx-1-cll-10.1177_09636897241254678 for A Novel JAK1 Inhibitor SHR0302 Combined With Prednisone for First-Line Treatment of Chronic Graft-Versus-Host Disease: A Phase I Clinical Trial by Qiaomei He, Xi Sun, Jiahua Niu, Jun Yang, Ying Wang, Chongmei Huang, Kun Zhou, Yin Tong, Yu Cai, Baoxia Dong, Liping Wan, Xianmin Song and Huiying Qiu in Cell Transplantation
Footnotes
Acknowledgements
The authors thank all the faculty and staff in the Department of Hematology, Shanghai General Hospital, Shanghai Jiaotong University School of Medicine. They also appreciate Jiangsu Hengrui Pharmaceuticals Co., Ltd. for providing the agent of SHR0302. They are grateful to all the patients, their families, and all members of the collaborative group in this trial.
Author Contributions
H.Q. and X.S. designed and supervised the clinical study; X.S. and Q.H. collected clinical data and wrote the manuscript; Y.W. participated in the study design; H.Q. and J.N. conducted clinical data collection; J.Y., C.H., K.Z., Y.T., Y.C., B.D., and L.W. enrolled and took care of the patients; H.Q. contributed to response monitoring of the patients. All authors contributed to the article and approved the submitted version.
Data Availability
The data generated in this study are not publicly available due to information that could compromise patient privacy but are available upon reasonable request from the corresponding author.
Statement of Prior Presentation
Presented in poster form at the 48th annual meeting of the EBMT, Prague Hybird, 19-23 March 2022 (P214); the 64th annual meeting of the American Society of Hematology, New Orleans, 11 December 2022 (Publication Number: 3394); and the 65th annual meeting of the American Society of Hematology, San Diego, 10 December 2023 (Publication Number: 3554).
Ethical Approval
The Ethics Committees of Shanghai General Hospital (Approval ID: 2019-110) approve the study.
Statement of Human Rights
All procedures in this study were conducted in accordance with the Ethics Committees of Shanghai General Hospital approved protocols.
Statement of Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Three-year development project from Shanghai Shen Kang Hospital Development Center (SHDC2020CR1012B for Xianmin Song); Clinical Research Innovation Plan of Shanghai General Hospital (grant no. CTCCR-2019D02 for Jiahua Niu); the Clinical Research Innovation Plan of Shanghai General Hospital (grant no. CTCCR-2019B03 for Chongmei Huang).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
