Abstract
Between the completion of the mesenchymal stem cell (MSCs) preparation and the transplantation into the patient, there is a time interval during which the quality control and transport of MSC products occur, which usually involves suspending the cells in normal saline in an oxygen–glucose deprivation (OGD) microenvironments. Thus, how to effectively maintain MSC viability during the abovementioned time interval is bound to play a significant role in the therapeutic effect of MSC-based therapies. Recently, menstrual blood–derived endometrial stem cells (MenSCs) have attracted extensive attention in regenerative medicine due to their superior biological characteristics, including noninvasive protocols for their collection, abundant source material, stable donation, and autotransplantation. Therefore, this study aimed to mainly determine the effect of storage temperature on the maintenance of MenSC viabilities in an OGD microenvironment, and to preliminarily explore its potential mechanism. Simultaneously, the effects of solvents commonly used in the clinic on MenSC viability were also examined to support the clinical application of MenSCs. Consequently, our results demonstrated that in the OGD microenvironment, a nonfreezing low temperature (4°C) was suitable and cost-effective for MenSC storage, and the maintenance of MenSC viability stored at 4°C was partly contributed by the sustained releases of autophagy-produced energy. Furthermore, the addition of human serum albumin effectively inhibited the cell sedimentations in the MenSC suspension. These results provide support and practical experience for the extensive application of MenSCs in the clinic.
Introduction
As of April 2021, more than 1,200 clinical trials of mesenchymal stem cell (MSC)-based therapies have been registered based on ClinicalTrials.gov data, and the safety of MSC transplantation has been extensively confirmed1–3. Currently, it is well known that MSC treatment–derived paracrine effects, immunomodulation and differentiation, have exhibited promising therapeutic effects on various diseases in basic and clinical studies, and provide opportunities for diseases without effective treatments4–6. Furthermore, as a novel type of MSCs, menstrual blood–derived endometrial stem cells (MenSCs) have attracted extensive attention in regenerative medicine due to their superior biological characteristics, including noninvasive protocols for their collection, abundant source material, stable donation, and autotransplantation7–9. Empirically, approximately 0.5 × 105 primary MenSCs can be isolated from 5 ml of menstrual blood sample, and the harvested primary MenSCs can be amplified to approximately 1 × 108 cells in vitro after normal subculture to passage 3, which satisfies the requirement for not only autologous transplantation but also for family members 8 . Until now, MenSC transplantation has exhibited promising therapeutic effects on Duchenne muscular dystrophy, heart failure, and intrauterine adhesion in the clinic, and no safety problems have been reported9–11. Moreover, in addition to the superior capacity to promote injured tissue regeneration, MenSC-derived immunomodulation also plays an important role in diseases with inflammatory dysfunction 12 . Especially, MenSC-based therapies have emerged as a promising therapeutic option for the treatment of COVID-1913.
However, unlike long-term cryopreservation of MSCs, which has been a concern for the majority of studies14,15, with MSCs intended for clinical application, there is a time interval between the completion of fresh MSC preparation and transplantation into the patient for actual clinical application. The most basic quality control of MSC products needs to be performed during this period, including but not limited to endotoxin, mycoplasma, and cell surface marker detection16,17. Simultaneously, MSC production is often completed at commercial cell preparation companies, but MSC applications are usually carried out in hospitals. Therefore, the packaged MSC suspension should be immediately transported to the hospital once the MSCs are encapsulated, and this transport process still requires extra time 18 . Moreover, configured MSC products usually suspend cells in normal saline, in a typical oxygen–glucose deprivation (OGD) microenvironment. Currently, the in vitro OGD model is often used to mimic the interruption of the supply of oxygen and metabolic fuel to the cells and tissues that occurs during an ischemic event and has been extensively developed to elucidate the role of key cellular and molecular mechanisms underlying ischemia-induced injury in the heart and brain, mainly including myocardial infarct, ischemic stroke, and cardiac arrest19,20. As both oxygen and glucose are critical to cell survival, the duration of OGD is positively correlated with reduced cell viability in a variety of in vitro models. Accordingly, it has been demonstrated that under hypoxic conditions, glucose deprivation accounts for the widespread loss of neurons seen in OGD and reperfusion 19 . However, Kanazawa et al. indicated that both microglia and peripheral blood mononuclear cells (PBMCs) pretreated by optimal OGD might be novel therapeutic strategies against ischemic stroke21,22.
Consequently, how to effectively maintain the cell viability of suspended MSCs under adverse conditions is bound to have a significant impact on the therapeutic effect of MSC transplantation for various diseases. Previous studies have indicated that a nonfreezing low temperature (2°C–8°C) is appropriate and conducive to the short-term preservation of cell viability23–25. Low temperature can inhibit temperature-dependent cell metabolism events, mainly involving a slowdown of biochemical reactions, reduced membrane fluidity, inhibited DNA fragmentation and enhanced protein stability which lead to reduced oxygen and ATP consumption, and finally contribute to cell viability maintenance26,27. In addition to the storage temperature, the suspension medium also has critical significance17,24,25. Therefore, this study aimed to determine the effect of temperature on the maintenance of MenSC viability in an OGD microenvironment and to preliminarily explore its potential mechanism. Simultaneously, the effects of solvents commonly used in the clinic on MenSC viability were also examined to provide support for the clinical application of MenSCs.
Materials and Methods
Cells
This study was approved by the Ethics Committee of Xinxiang Medical University. MenSCs were kindly provided by Zhongyuan Stem Cell Research Institute (Xinxiang, Henan, China). All experimental procedures for MenSCs were carried out in accordance with the approved guidelines. Written informed consent was obtained from all donors prior to sample collection, and all donors provided consent for the use of their MenSCs for scientific research. Frozen MenSCs were recovered in 75 cm2 cell culture flask (7 × 105 cells per flask) in Dulbecco’s modified Eagle’s medium (Zhong Qiao Xin Zhou Biotechnology Co, Ltd, Beijing, China) supplemented with 10% fetal bovine serum (Gibco, Australia), and a 1% mixture of penicillin and streptomycin (100×, Genevie, Beijing, China). The fresh medium was replaced every 3 days. When the cells reached 80% to 90% confluence, the cells were conventionally detached and subcultured into new flasks. P3-P6 MenSCs were used in this study.
MenSC Storage and Treatment
MenSCs were conventionally digested into single cells with trypsin and treated as follows according to the experimental design: (1) MenSC suspension was centrifuged and resuspended in saline at a density of 1 × 106 cells/ml, the cell suspensions were aspirated into the 1 ml syringes, all the air was removed, and the syringe head was sealed with silica gel plugs and parafilm. Thereafter, the syringes containing cells were placed at different temperatures (4°C, 25°C, and 37°C) and stored for different times (6 h, 12 h, and 24 h). At the end of storage, the MenSCs in the syringes were gently mixed and transferred to 1.5 ml tubes for the following viability examinations, including cell density, viability and diameter assays, cell counting kit-8 (CCK8) assays and apoptosis detection by flow cytometry and western blotting (WB). (2) MenSCs suspended in saline at a density of 1 × 106 cells/ml were sealed in syringes as performed above, and then stored at 4°C and 37°C for 2 h, 4 h, and 6 h. At the end of storage, the MenSCs were used for sbusequent assays, including CCK8 assays and autophagy activation assays by WB. (3) MenSCs suspended in saline, saline with spautin-1 (final concentration: 10 μM, Beyotime, Shanghai, China), and saline with SMER28 (final concentration: 100 μM, Beyotime, Shanghai, China) at a density of 1 × 106 cells/ml were sealed in syringes as performed above and were then stored at 4°C for 6 h and 12 h. At the end of storage, the MenSCs were used for subsequent assays, including apoptosis detection by flow cytometry and autophagy activation assays by WB. (4) MenSCs suspended in saline, saline + 5% human serum albumin (HSA, Hualan Bio, Xinxiang, China), multiple electrolytes (Tiancheng Pharmaceutical Co, Ltd, Hebei, China), and multiple electrolytes + 5% HSA at a density of 1 × 106 cells/ml were, respectively, sealed in syringes as performed above and then stored at 4°C for 6 h and 12 h. At the end of storage, the MenSCs were used for subsequent assays, including CCK8 assay, apoptosis detection by flow cytometry, and apoptosis detection by WB.
Cell Density, Viability, and Diameter Assays
After storage at different temperatures for different times, the MenSC suspensions were transferred to centrifuge tubes. Then, the cell density, viability (trypan blue staining), and diameter were detected by a cell counter (Luna-II, Logos Biosystems, Gyeonggi-do, Korea).
CCK8 Assay
The MenSC suspension was diluted to 1.5 × 105 cells/ml with fresh medium according to the initial density (1 × 106 cells/ml) and then added to the 96-well plates (200 μl of cell suspension per well). After culturing for 24 h, 48 h, and 72 h (based on the experimental design), 10 μl of CCK8 solution was added and incubated at 37°C for 2 to 3 h, and the absorbance was measured at 450 nm by SpectraMax® i3 microplate reader (Molecular Devices, San jose, CA, USA).
Apoptosis Detection by Flow Cytometry
A 0.5 ml cell suspension (1 × 106 cells/ml) was centrifuged at 1,200 rpm for 5 min, and the supernatant was discarded. MenSC apoptosis was determined by an Annexin V/PI (prodium iodide) staining kit according to the instruction of the manufacture (Beyotime, Shanghai, China). Briefly, 195 μl of binding solution was used to gently suspend the cell sedimentation, and 5 μl of Annexin V-FITC and 10 μl of PI solution were gently added and mixed with the cell suspension. After incubating at 4°C for 20 min, 20 μl of the suspension was dripped into the cell counting plate, observed and imaged under a fluorescence microscope. The rest of the sample was diluted with phosphate-buffered saline to a volume of 500 μl and filtered with a 100 μm cell filter. Then, the samples were detected by flow cytometry (FACSCalibur, BD, Franklin Lakes, NJ, USA).
Western Blotting Analysis
The protein samples of MenSCs stored at different temperatures for different times were conventionally collected. Briefly, the cell samples were lysed with radio-immunoprecipitation assay (RIPA) lysis buffer containing 150 mM NaCl, 1% NP-40, 0.5% deoxycholate, and 1% sodium dodecyl sulfate (SDS) on ice for 30 min. Then, the supernatant was collected and the concentration of total protein was determined by the bicinchoninic acid (BCA) assay. Subsequently, the protein samples (10 μg per lane) were electrophoresed in 10% to 12% SDS–polyacrylamide gel electrophoresis, transferred to a nitrocellulose membrane under constant current for 1 h, and blocked with 5% nonfat milk in tris buffered saline (TBS) containing 0.1% Tween 20. Thereafter, the primary antibodies (Bcl2, Bcl-xL, Bad, Caspase3, Caspase9, AKT, Beclin, p62, p65, Lc3A/B, and GAPDH; Table S1) were incubated overnight at 4°C, and the horseradish peroxidase (HRP)-conjugated secondary antibody was incubated for 2 h at room temperature. Finally, the expression of targeted proteins was developed using the ECL WB substrate kit and detected using chemiluminescence instrument (Amersham Imager 600, GE Healthcare Life Sciences, Boston, MA, USA), and the gray value of the targeted proteins were quantified by ImageJ software. GAPDH was used as the internal control, and the expression level of the target proteins is indicated by the ratio to GAPDH.
Cell Sedimentation Assay
Two milliliters of MenSC suspension (1×106 cells/ml), suspended in saline or saline containing 5% HSA, was blended evenly and transferred into transparent tubes and incubated for 120 min at room temperature. During the standing time (5 min, 30 min, 60 min and 120 min), photos of cell sedimentation were collected, and the turbidity of the cell suspension was measured at 600 nm using a microplate reader, which indirectly indicated the sedimentation speed of cells.
Statistical Analysis
All quantitative results are presented as the mean ± SEM. The results were analyzed using the Student’s t test for comparisons between two groups and one-way analysis of variance followed by Dunnett’s test for comparisons among ≥3 groups. For all analyses, P < 0.05 was considered statistically significant.
Results
Low Temperature Storage Effectively Maintains MenSC Viability
In the OGD microenvironment, the cell density and cell viability gradually decreased with the extension of the storage time at different storage temperatures, but MenSCs stored at 4°C exhibited higher cell density and cell viability during the same storage time (Fig. 1A, B). Simultaneously, when MenSCs were stored at 4°C, the cell diameter only showed a slight decrease with the extension of storage time; however, when MenSCs were stored at 25°C and 37°C, the cell diameter first exhibited obvious shrinkage after storage for 6 h, and then the cell diameter showed a robust increase, especially for cells stored at 37°C, which suggested cell death caused by cell swelling (Fig. 1C). In accordance with the above results, the subsequent apoptosis staining results confirmed that the percentages of apoptotic cells positively correlated with the extension of storage time and the increase in storage temperature, and the MenSCs stored at 4°C exhibited superior viability (Fig. 1D–F).

Low temperature maintains the viability of MenSCs in the OGD microenvironment. (A–C) MenSCs were stored at 4°C, 25°C, and 37°C for 6 h, 12 h, and 24 h, and then the cell density, viability (trypan blue staining), and diameter were measured by a cell counter. (D–F) MenSCs were stored at 4°C, 25°C, and 37°C for 6 h, 12 h, and 24 h, and then the apoptosis (Annexin V/PI staining) of suspended MenSCs was detected by flow cytometry and imaged under a fluorescence microscope. MenSCs: menstrual blood–derived endometrial stem cells; OGD: oxygen–glucose deprivation; PI: prodium Iiodide.
Low Temperature Storage Enhances the Antiapoptotic Capacity of MenSCs
CCK8 assays indicated that the proliferative capacity of MenSCs was negatively correlated with the extension of storage time and the increase in storage temperature, and the cells stored at 25°C for 24 h or 37°C for 12 h completely lost their viability (Fig. 2A). Furthermore, we concluded that the expression of antiapoptotic indicators (Bcl2 and Bcl-xL), proapoptotic indicators (Bad, Caspase9, and Caspase3), and proliferative indicators (Akt) remained stable in MenSCs stored at 4°C. However, with the extension of storage time the expression of antiapoptotic indicators (Bcl2 and Bcl-xL), proapoptotic indicators (Bad, Caspase9, and Caspase3), and proliferative indicators (Akt) exhibited significant decreases in the cells stored at 25°C or 37°C (Fig. 2B–H). In addition, based on the gray value of GAPDH, our results indicated that the total protein of cell samples significantly decreased in the cells stored at 25°C or 37°C with the extension of storage time, which was consistent with the decrease in cell density (Fig. 1A).

Low temperature enhances the antiapoptotic capacity of MenSCs in the OGD microenvironment. (A) MenSCs were stored at 4°C, 25°C, and 37°C for 6 h, 12 h, and 24 h, and then the MenSCs were transferred in 96-well plates and cultured in complete medium for 24 h, 48 h, and 72 h. Cell proliferation activity was detected by CCK8 assay. (B-H) MenSCs were stored at 4°C, 25°C, 37°C for 6 h, 12 h, and 24 h, and then the apoptotic markers were examined by conventional WB. The gray value of the targeted proteins was quantified with the ImageJ software. MenSCs: menstrual blood–derived endometrial stem cells; OGD: oxygen–glucose deprivation; WB: western blotting; CCK8: cell counting kit-8; OD: optical density; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; NS: no statistical significance.
Autophagy Activation Is Involved in Maintaining MenSC Viability at Low Temperature
To explore the potential mechanism of low temperature on MenSC viability maintenance in the OGD microenvironment, MenSCs were stored at 4°C and 37°C for 2 h, 4 h, and 6 h for subsequent examination. The CCK8 assay indicated that although the viability of MenSCs stored at 4°C or 37°C gradually decreased with the extension of storage time, the rapid viability decline of the cells stored at 37°C was obvious with the extension of storage time (Fig. 3A). Further examination found that the expressions of Beclin and LC3A/B-II and the ratio of LC3A/B-II/LC3A/B-I were gradually upregulated with the extension of storage time at 4°C; p65 expression remained stable, while p62 exhibited the opposite expression pattern. However, when the cells were stored at 37°C, Beclin expression and the ratio of LC3A/B-II/LC3A/B-I reached their peak when stored for 4 h, while p65 expression was significantly downregulated with the extension of storage time (Fig. 3B–G).

Autophagy activation participates in the maintenance of MenSC viability in the OGD microenvironment. (A) MenSCs were stored at 4°C and 37°C for 2 h, 4 h, and 6 h, and then the suspended MenSCs were transferred to 96-well plates, and cultured for 24 h, 48 h, and 72 h in complete medium. Cell proliferation activity was detected by CCK8 assay. (B–H) MenSCs were stored at 4°C and 37°C for 2 h, 4 h, and 6 h, and then autophagy-related markers were examined by conventional WB. The gray value of the targeted proteins was quantified with the ImageJ software. MenSCs: menstrual blood–derived endometrial stem cells; OGD: oxygen–glucose deprivation; WB: western blotting.
Autophagy Inhibition Impairs the Maintenance of Low Temperature–Derived Viability for MenSCs
To further confirm the role of autophagy in maintaining MenSC viability in OGD microenvironments, the autophagy inhibitor spautin-1 and autophagy enhancer SMER28 (Beyotime, China) were used to perform interventional experiments. The flow cytometry results showed that compared with the control group, the number of apoptotic cells treated with spautin-1 was significantly increased, but no significant change was observed in the cells treated with SMER28 in the OGD microenvironment (Fig. 4A, B). In accordance with the above results, the subsequent WB results also demonstrated that compared with the control group, the expressions of Beclin and LC3A/B-II and the ratio of LC3A/B-II/LC3A/B-I were significantly downregulated, and p62 expression was upregulated after spautin-1 treatment. Akt expression levels decreased significantly after treatment with spautin-1 for 6 h but returned to normal levels after treatment for 12 h. Moreover, the expressions of Beclin was significantly upregulated and p62 was downregulated in MenSCs treated with SMER28 compared with the control group, but no significant changes were observed in the expression of LC3A/B-II or Akt or the ratio of LC3A/B-II/LC3A/B-I (Fig. 4C–H).
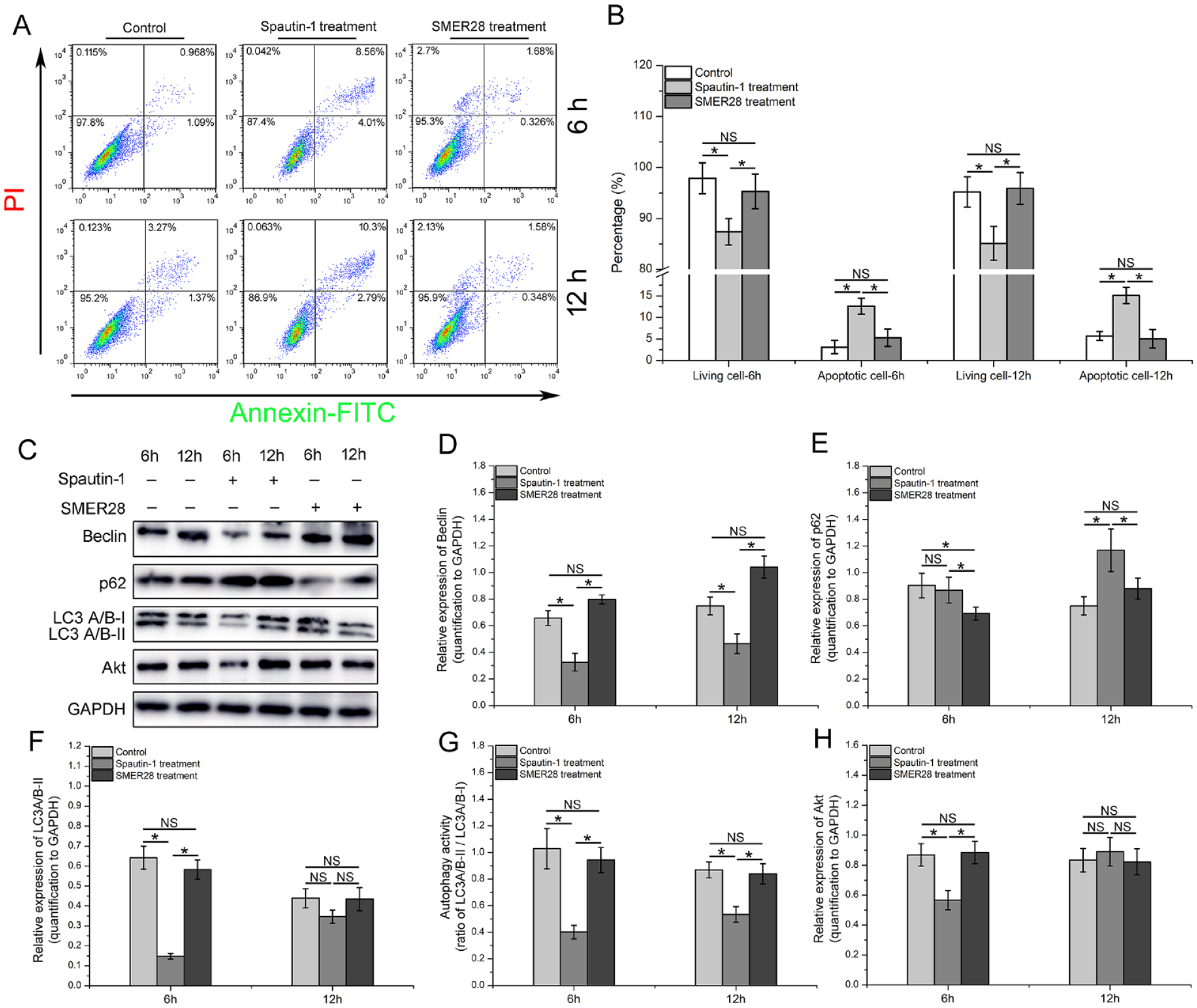
Autophagy inhibition significantly impaired MenSC viability in the OGD microenvironment. (A, B) MenSCs treated with spautin-1 and SMER28 were stored at 4°C for 6 h and 12 h, and the apoptosis of suspended MenSCs was detected by flow cytometry. (C–H) MenSCs treated with spautin-1 and SMER28 were stored at 4°C for 6 h and 12 h, and the expression of autophagy-related markers was examined by conventional WB. The gray value of the targeted proteins was quantified with the ImageJ software. MenSCs: menstrual blood–derived endometrial stem cells; OGD: oxygen–glucose deprivation; WB: western blotting.
The Effect of Suspension Medium on MenSC Viability During Storage
We observed that the addition of 5% HSA to saline could effectively inhibit MenSC sedimentation within 2 h (Fig. 5A, B). Subsequently, both the metabolic activity assay (Fig. 5C) and apoptosis assay (Fig. 5D, E) indicated that there were no differences between the viability of MenSCs suspended in multiple electrolytes and saline. However, the addition of 5% HSA to multiple electrolytes significantly promoted MenSC apoptosis in this solution. Furthermore, consistent with the above results, the WB results also demonstrated that Bcl2 expression was significantly downregulated in MenSCs suspended in multiple electrolytes containing 5% HSA, while Bax and Akt were significantly upregulated.
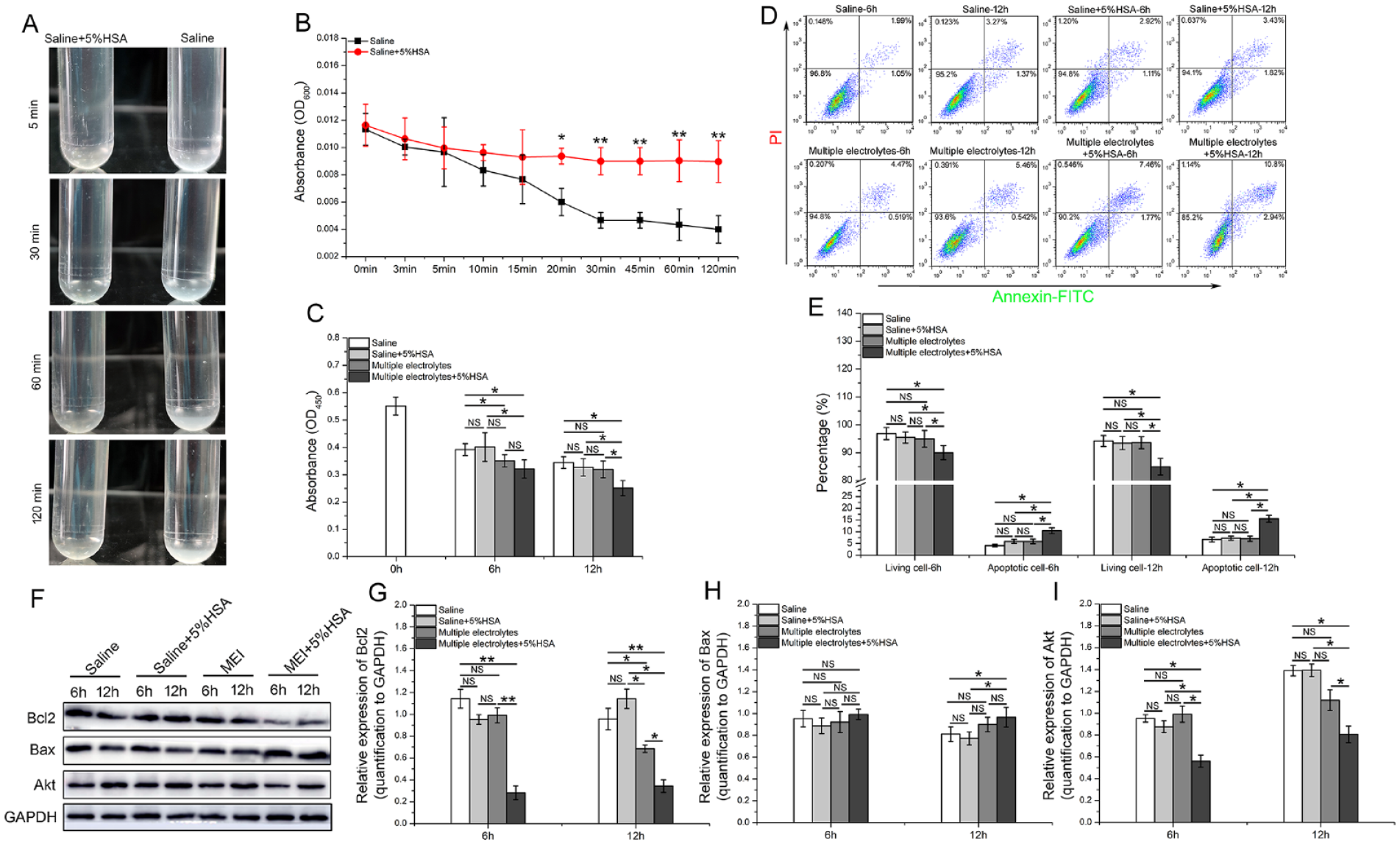
The effect of solvents on the viability of suspended MenSCs. (A, B) MenSCs suspended in saline with or without 5% HSA were incubated for 120 min at room temperature. Then, photos of cell sedimentation were imaged, and the sedimentation speed was qualitied by microplate reader. (C) MenSCs suspended in saline, saline + 5% HSA, multiple electrolytes and multiple electrolytes + 5% HSA were stored at 4°C for 6 h and 12 h. Then, the MenSCs were transferred to 96-well plates and cultured for 48 h in complete medium, and cell proliferation activity was detected by CCK8 assay. (D, E) MenSCs were suspended in the abovementioned solvents at 4°C for 6 h and 12 h and then the apoptosis of suspend MenSCs were detected by flow cytometry. (F–I) MenSCs were suspended in the abovementioned solvents at 4°C for 6 h and 12 h and then conventional WB was used to identify the expression of antiapoptotic proteins. The gray value of the targeted proteins was quantified with the ImageJ software. MenSCs: menstrual blood–derived endometrial stem cells; OGD: oxygen–glucose deprivation; HSA: human serum albumin; WB: western blotting.
Discussion
Stem cell–based regenerative medicine is a promising strategy for the treatment of various diseases that have no effective treatments2,28. Currently, MSC transplantation–induced paracrine effects, immunomodulation, and transdifferentiation exhibit superior functional recovery of injured tissues or organs, but bring challenges to mechanistic studies and their clinical application in diseases4–6,28. MenSCs were the focus of this research project and have attracted notable attention as a promising alternative MSC-based therapy for various diseases due to their unique advantages, such as regular and noninvasive sample collection, abundant availability, superior proliferative capacity, and autologous transplantation potential7,8,29.
With the development of MSC products for clinical application, it has become more critical to clarify the different responses of living MSCs to their stored microenvironment, which is not only of fundamental scientific interest, but is indispensable for ensuring the quality of these MSC products, and plays critical roles in the success of their clinical application17,30. However, challenges faced during the storage and shipping of therapeutic MSC products from commercial cell preparation companies to hospitals have drawn significant attention in emerging regenerative medicine. To date, methods for the long-term cryopreservation of MSCs in liquid or gaseous nitrogen under the protection of various kinds of cryoprotective agents have been extensively established and tested14,15. The challenges related to short-term storage and transportation of MSC products before in vivo transplantation remain poorly studied, because of the existence of a time interval between the completion of MSC production and the actual clinical application following the quality control and transport of MSC products. Simultaneously, most MSC-based clinical applications require the supply of living cells for immediate and easy administration by the clinical end-user, preferably without the presence of a cryoprotectant, which has potential toxicity and/or irreversible impairment to the cell and body 31 , and packaged MSC products usually suspend MSCs in normal saline with an OGD microenvironment.
Nonfreezing low temperature is extensively used as a preservative method in both organs and cells that are capable of recovering from exposure to low temperatures 32 . Thus, it seems reasonable to apply low temperature to the short-term storage of MSCs for therapeutic procedures. As early as 1993, 4°C storage was used to maintain the viability of epidermal cells in grafted skin 33 , and 4°C was recognized as the optimal temperature for storage and delivery of bone marrow–derived MSCs, as it minimally affected their viability 24 . Recently, Wu et al. 25 also recommended that adipose‑derived stem cells (ADSCs) for clinical application should be maintained at 4°C during short‑term storage to maintain their viability. As expected, our results were consistent with previously published reports and demonstrated that in the OGD microenvironments, the viability of suspended MenSCs was negatively correlated with storage time and temperature, and 4°C was suitable for the maintenance of MenSC viability. The mechanism of cell viability maintenance under low temperature is likely to be contributed by slowing metabolic activity and cell cycle progression (energy-dependent processes), such as protein synthesis and transport systems, and the upregulation of cold inducible RNA-binding protein (CIRBP) also plays a protective role in cell viability maintenance under low temperature32,34. Simultaneously, during the storage process, MenSCs can protect themselves by shrinking their cell volume (decreasing their cell diameter), which is an adaptive way that cells resist low temperature–induced adverse microenvironments 35 . However, low temperature–induced stress may also affect the integrity of the membrane lipid bilayer and cytoskeletal structure and promote the production of free radicals, which collectively result in further cell injury, apoptosis, or necrosis36,37. This is why although 4°C storage was capable of effectively maintaining the density, viability, and antiapoptotic capacity of suspended MenSCs, long-term storage still impaired MenSC viability. Presumably, a shorter storage time theoretically resulted in better preservation of cell viability and function.
Although 4°C was suitable for the maintenance of MenSC viability in the OGD microenvironments, 4°C storage could not completely stop MenSC metabolic activity, which still required energy to sustain metabolic activity that is indispensable for cell survival. Therefore, autophagy is first considered when we attempt to explore where energy comes from, especially in the OGD microenvironment. Autophagy activation in cells is an adaptive mechanism under various kinds of stress, protects the cell against adversity, and promotes cell survival by delaying apoptosis and necrosis38,39. Generally, autophagy activation can upregulate the expression of the antiapoptotic factor Bcl2, and enhance the antiapoptotic capacity of cells; however, when the autophagy-produced energy is insufficient to protect cells against apoptosis, p53 transcriptional activity is promoted, the expression of proapoptotic Bid is upregulated, and the downstream caspase-3 apoptotic pathway is activated 40 . Simultaneously, Akt activity is positively correlated with cell viability, and previous reports have demonstrated that the enhancement of Akt activity could significantly ameliorate OGD-induced impairment of cell viability41,42.
Consistent with the above reports, our results confirmed autophagy activation in MenSCs suspended in saline (OGD microenvironment) stored at 4°C and 37°C. Autophagy was gradually activated in MenSCs stored at 4°C, but autophagy in MenSCs stored at 37°C immediately reached a peak after storage for 4 h. We postulated that this is because cells stored at 37°C require substantial energy to maintain normal metabolic activity; therefore, autophagy-produced energy is quickly promoted and released, and excessive autophagy activation induces cell apoptosis. However, the cells stored at 4°C required only minimal energy to maintain indispensable metabolic activity, and 4°C storage played a role in the sustained release of autophagy-produced energy, which contributed to the maintenance of cell viability 18 . Furthermore, subsequent results also confirmed that autophagy inhibition significantly induced the apoptosis of MenSCs stored at 4°C, but autophagy enhancement had no effect on MenSC viability, which is partly explained by the low temperature–induced enzyme activity inhibition in the entire autophagy signaling pathway. Interestingly, a substantial number of MenSCs (>80%) survived after autophagy inhibition for 12 h when stored at 4°C, which strongly suggested that in addition to autophagy activation, other signaling pathways must cooperatively maintain the viability of MenSCs stored at 4°C. Moreover, Kanazawa et al. not only found that intravascular administration of microglia pretreated by optimal OGD promoted functional recovery after focal cerebral ischemia by upregulating the expression of remodeling factors 21 but also further demonstrated that the administration of OGD-pretreated PBMCs promoted functional recovery after cerebral ischemia by prompting angiogenesis and axonal outgrowth 22 . Therefore, it is reasonable to postulate that short-term storage of MenSCs in the OGD microenvironments might improve the homing capacity, survival, and paracrine effects of cells in vitro and in vivo and thereby improving the therapeutic effects of MenSCs in various diseases.
In addition, stem cells are usually suspended in solvent and intravenously injected into patients in clinical applications. In line with the rules of solvent use for intravenous infusion (adding as few foreign substances as possible), normal saline is the preferred option17,24,25. Our results demonstrated that the viability of MenSCs stored at 4°C for 12 h was not significantly different between those stored in saline and those stored in multiple electrolytes. Human serum albumin (HSA) is conventionally added to MSC suspensions to inhibit cell aggregation and sedimentation43,44, although it can cause potential health risks during MSC transplantation45,46. As indicated in our study, HSA addition was not only able to effectively inhibit MenSC sedimentation within 2 h, which is more than enough time to completely finish MenSC transplantation, but also had no negative effect on MenSC suspended in saline. However, HSA addition was able to promote the apoptosis of MenSC suspended in multiple electrolytes, but the potential mechanism (not the focus of this study) is still unknown45,46.
In summary, in the OGD microenvironment, nonfreezing low temperature (4°C) was suitable and cost-effective for MenSC storage, and the maintenance of MenSC viability stored at 4°C was partly contributed by the sustained release of autophagy-produced energy. Furthermore, HSA addition effectively inhibited cell sedimentation in the MenSC suspension. These results provide support and practical experience for the extensive application of MenSCs in the clinic.
Supplemental Material
sj-docx-1-cll-10.1177_09636897221086971 – Supplemental material for Nonfreezing Low Temperature Maintains the Viability of Menstrual Blood–Derived Endometrial Stem Cells Under Oxygen–Glucose Deprivation Through the Sustained Release of Autophagy-Produced Energy
Supplemental material, sj-docx-1-cll-10.1177_09636897221086971 for Nonfreezing Low Temperature Maintains the Viability of Menstrual Blood–Derived Endometrial Stem Cells Under Oxygen–Glucose Deprivation Through the Sustained Release of Autophagy-Produced Energy by Tongtong Chen, Shenghui Zhang, Hongzhang Jin, Xiaofei Fu, Lingrui Shang, Yilin Lu, Yuliang Sun, Badrul Hisham Yahaya, Yanli Liu and Juntang Lin in Cell Transplantation
Footnotes
Author Contributions
JL, YL, and BYH conceived and designed the experiments. TC, SZ, and XF analyzed the data and prepared the manuscript. TC, HJ, LS, and YL performed the experiments. YS, HC, and SZ analyzed the data, wrote the manuscript, and prepared the figures. All authors reviewed the manuscript.
Ethical Approval
This study was approved by the Ethics Committee of Xinxiang Medical University.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the ethics committee of Xinxiang Medical University of Henan Province approved protocols (XYLL-2020038).
Statement of Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Data Availability
The data used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Henan Province Foundation (NO. 202300410307 and NO.212102310611); Xinxiang City Foundation (NO. GG2020009); and the Xinxiang Medical University Foundation (NO. YJSCX202054Y)
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
